Abstract
Despite their limited cargo capacity (<5 kb), adeno-associated viral (AAV) vectors remain the gold standard for in vivo delivery of therapeutic genes. Dual AAV vectors have emerged as a valuable tool for delivering large therapeutic genes and CRISPR tools to overcome this limitation. Here we provide a detailed protocol for the design, production and evaluation of dual AAV vectors. We offer guidelines for selecting a suitable dual AAV strategy, designing and cloning the genes to be delivered, and conducting in vitro evaluations of expression efficiency. In addition, we detail the production of dual AAVs and their assessment in human cellular models, such as induced pluripotent stem cell-derived retinal organoids. Finally, we outline the administration of dual AAVs via different routes in mice and the assessment of transgene-derived RNA and protein expression in various tissues. Overall, the instructions in this Protocol will aid in the efficient in vivo delivery of large DNA fragments using dual AAVs. This Protocol is adaptable to a wide range of model organisms as well as to human organoid cultures and, depending on the application, can be completed in 15–44 weeks.
Key points
-
Adeno-associated viral (AAV) vectors are widely used to deliver therapeutic genes in vivo. Dual AAVs, whereby the gene of interest is delivered by two viral vectors and reconstituted upon delivery, are used to overcome the limited cargo capacity of the AAV genome.
-
This Protocol presents three strategies for dual AAV design and provides guidance for the production and evaluation of dual AAV vectors in wild-type and disease mouse models and in human organoids.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
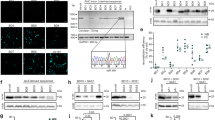
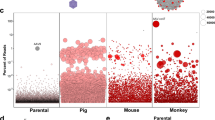
Figures 5 and 6 show example data that were obtained using this Protocol. Source data are provided with this Protocol. Additional data related to this Protocol can be found in the original study20 or may be requested from the authors. Source data are provided with this paper.
References
Wang, D., Tai, P. W. L. & Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat. Rev. Drug Discov. 18, 358–378 (2019).
Li, C. & Samulski, R. J. Engineering adeno-associated virus vectors for gene therapy. Nat. Rev. Genet. 21, 255–272 (2020).
Ibraheim, R. et al. Self-inactivating, all-in-one AAV vectors for precision Cas9 genome editing via homology-directed repair in vivo. Nat. Commun. 12, 6267 (2021).
Jang, M. J. et al. Spatial transcriptomics for profiling the tropism of viral vectors in tissues. Nat. Biotechnol. 41, 1272–1286 (2023).
Rittiner, J. E., Moncalvo, M., Chiba-Falek, O. & Kantor, B. Gene-editing technologies paired with viral vectors for translational research into neurodegenerative diseases. Front. Mol. Neurosci. 13, 148 (2020).
Srivastava, A. In vivo tissue-tropism of adeno-associated viral vectors. Curr. Opin. Virol. 21, 75–80 (2016).
Heo, Y. A. Etranacogene dezaparvovec: first approval. Drugs 83, 347–352 (2023).
Gao, J., Hussain, R. M. & Weng, C. Y. Voretigene neparvovec in retinal diseases: a review of the current clinical evidence. Clin. Ophthalmol. 14, 3855–3869 (2020).
Hoy, S. M. Delandistrogene moxeparvovec: first approval. Drugs 83, 1323–1329 (2023).
Wu, Z., Yang, H. & Colosi, P. Effect of genome size on AAV vector packaging. Mol. Ther. 18, 80–86 (2010).
Naso, M. F., Tomkowicz, B., Perry, W. L. & Strohl, W. R. Adeno-associated virus (AAV) as a vector for gene therapy. BioDrugs 31, 317–334 (2017).
Lai, Y. et al. Efficient in vivo gene expression by trans-splicing adeno-associated viral vectors. Nat. Biotechnol. 23, 1435–1439 (2005).
Yan, Z., Zhang, Y., Duan, D. & Engelhardt, J. F. Trans-splicing vectors expand the utility of adeno-associated virus for gene therapy. Proc. Natl Acad. Sci. USA 97, 6716–6721 (2000).
Lopes, V. S. et al. Retinal gene therapy with a large MYO7A cDNA using adeno-associated virus. Gene Ther. 20, 824–833 (2013).
Duan, D., Yue, Y. & Engelhardt, J. F. Expanding AAV packaging capacity with trans-splicing or overlapping vectors: a quantitative comparison. Mol. Ther. 4, 383–391 (2001).
Trapani, I. et al. Improved dual AAV vectors with reduced expression of truncated proteins are safe and effective in the retina of a mouse model of Stargardt disease. Hum. Mol. Genet. 24, 6811–6825 (2015).
Ghosh, A., Yue, Y., Lai, Y. & Duan, D. A hybrid vector system expands adeno-associated viral vector packaging capacity in a transgene-independent manner. Mol. Ther. 16, 124–130 (2008).
Carvalho, L. S. et al. Evaluating efficiencies of dual AAV approaches for retinal targeting. Front. Neurosci. 11, 503 (2017).
Dyka, F. M., Boye, S. L., Chiodo, V. A., Hauswirth, W. W. & Boye, S. E. Dual adeno-associated virus vectors result in efficient in vitro and in vivo expression of an oversized gene, MYO7A. Hum. Gene Ther. Methods 25, 166–177 (2014).
Riedmayr, L. M. et al. mRNA trans-splicing dual AAV vectors for (epi)genome editing and gene therapy. Nat. Commun. 14, 6578 (2023).
Liu, X. et al. Partial correction of endogenous DeltaF508 CFTR in human cystic fibrosis airway epithelia by spliceosome-mediated RNA trans-splicing. Nat. Biotechnol. 20, 47–52 (2002).
Lindley, S. R. et al. Ribozyme-activated mRNA trans-ligation enables large gene delivery to treat muscular dystrophies. Science 386, 762–767 (2024).
Tornabene, P. et al. Intein-mediated protein trans-splicing expands adeno-associated virus transfer capacity in the retina. Sci. Transl. Med. 11, eaav4523 (2019).
Blair, H. A. Onasemnogene abeparvovec: a review in spinal muscular atrophy. CNS Drugs 36, 995–1005 (2022).
Srivastava, A. Rationale and strategies for the development of safe and effective optimized AAV vectors for human gene therapy. Mol. Ther. Nucleic Acids 32, 949–959 (2023).
Martin, F. J. et al. Ensembl 2023. Nucleic Acids Res. 51, D933–D941 (2023).
Trapani, I. & Auricchio, A. Seeing the light after 25 years of retinal gene therapy. Trends Mol. Med. 24, 669–681 (2018).
Willett, K. & Bennett, J. Immunology of AAV-mediated gene transfer in the eye. Front. Immunol. 4, 261 (2013).
Riedmayr, L. M. et al. dCas9-VPR-mediated transcriptional activation of functionally equivalent genes for gene therapy. Nat. Protoc. 17, 781–818 (2022).
Chavez, A. et al. Highly efficient Cas9-mediated transcriptional programming. Nat. Methods 12, 326–328 (2015).
Yeo, N. C. et al. An enhanced CRISPR repressor for targeted mammalian gene regulation. Nat. Methods 15, 611–616 (2018).
Nunez, J. K. et al. Genome-wide programmable transcriptional memory by CRISPR-based epigenome editing. Cell 184, 2503–2519 e17 (2021).
Zhang, E., Neugebauer, M. E., Krasnow, N. A. & Liu, D. R. Phage-assisted evolution of highly active cytosine base editors with enhanced selectivity and minimal sequence context preference. Nat. Commun. 15, 1697 (2024).
Chen, L. et al. Engineering a precise adenine base editor with minimal bystander editing. Nat. Chem. Biol. 19, 101–110 (2023).
Anzalone, A. V. et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576, 149–157 (2019).
Bohm, S. et al. A gene therapy for inherited blindness using dCas9-VPR-mediated transcriptional activation. Sci. Adv. 6, eaba5614 (2020).
Levy, J. M. et al. Cytosine and adenine base editing of the brain, liver, retina, heart and skeletal muscle of mice via adeno-associated viruses. Nat. Biomed. Eng. 4, 97–110 (2020).
Davis, J. R. et al. Efficient prime editing in mouse brain, liver and heart with dual AAVs. Nat. Biotechnol. 42, 253–264 (2024).
Chen, F. et al. Multiplexed base editing through Cas12a variant-mediated cytosine and adenine base editors. Commun. Biol. 5, 1163 (2022).
Schmitt-Ulms, C. et al. Programmable RNA writing with trans-splicing. Preprint at bioRxiv (2024).
An, M. et al. Engineered virus-like particles for transient delivery of prime editor ribonucleoprotein complexes in vivo. Nat. Biotechnol. 42, 1526–1537 (2024).
Banskota, S. et al. Engineered virus-like particles for efficient in vivo delivery of therapeutic proteins. Cell 185, 250–265.e16 (2022).
Kulkarni, J. A. et al. Design of lipid nanoparticles for in vitro and in vivo delivery of plasmid DNA. Nanomedicine 13, 1377–1387 (2017).
Pozzi, D. & Caracciolo, G. Looking Back, moving forward: lipid nanoparticles as a promising frontier in gene delivery. ACS Pharmacol. Transl. Sci. 6, 1561–1573 (2023).
Mendell, J. R. et al. Testing preexisting antibodies prior to AAV gene transfer therapy: rationale, lessons and future considerations. Mol. Ther. Methods Clin. Dev. 25, 74–83 (2022).
Duan, D. Lethal immunotoxicity in high-dose systemic AAV therapy. Mol. Ther. 31, 3123–3126 (2023).
van der Loo, J. C. & Wright, J. F. Progress and challenges in viral vector manufacturing. Hum. Mol. Genet. 25, R42–R52 (2016).
Han, X. et al. Ligand-tethered lipid nanoparticles for targeted RNA delivery to treat liver fibrosis. Nat. Commun. 14, 75 (2023).
Musunuru, K. et al. In vivo CRISPR base editing of PCSK9 durably lowers cholesterol in primates. Nature 593, 429–434 (2021).
Kularatne, R. N., Crist, R. M. & Stern, S. T. The future of tissue-targeted lipid nanoparticle-mediated nucleic acid delivery. Pharmaceuticals 15, 897 (2022).
Kulkarni, J. A. et al. The current landscape of nucleic acid therapeutics. Nat. Nanotechnol. 16, 630–643 (2021).
Charlesworth, C. T. et al. Identification of preexisting adaptive immunity to Cas9 proteins in humans. Nat. Med. 25, 249–254 (2019).
van Haasteren, J., Li, J., Scheideler, O. J., Murthy, N. & Schaffer, D. V. The delivery challenge: fulfilling the promise of therapeutic genome editing. Nat. Biotechnol. 38, 845–855 (2020).
Wagner, D. L. et al. High prevalence of Streptococcus pyogenes Cas9-reactive T cells within the adult human population. Nat. Med. 25, 242–248 (2019).
Lee, C. S. et al. Adenovirus-mediated gene delivery: potential applications for gene and cell-based therapies in the new era of personalized medicine. Genes Dis. 4, 43–63 (2017).
Counsell, J. R. et al. Lentiviral vectors can be used for full-length dystrophin gene therapy. Sci. Rep. 7, 44775 (2017).
Jaffe, H. A. et al. Adenovirus-mediated in vivo gene transfer and expression in normal rat liver. Nat. Genet. 1, 372–378 (1992).
Rosenfeld, M. A. et al. Adenovirus-mediated transfer of a recombinant alpha 1-antitrypsin gene to the lung epithelium in vivo. Science 252, 431–434 (1991).
Zak, D. E. et al. Merck Ad5/HIV induces broad innate immune activation that predicts CD8+ T-cell responses but is attenuated by preexisting Ad5 immunity. Proc. Natl Acad. Sci. USA 109, E3503–E3512 (2012).
Khare, R., Hillestad, M. L., Xu, Z., Byrnes, A. P. & Barry, M. A. Circulating antibodies and macrophages as modulators of adenovirus pharmacology. J. Virol. 87, 3678–3686 (2013).
Raper, S. E. et al. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer. Mol. Genet. Metab. 80, 148–158 (2003).
Shirley, J. L., de Jong, Y. P., Terhorst, C. & Herzog, R. W. Immune responses to viral gene therapy vectors. Mol. Ther. 28, 709–722 (2020).
Qian, W. et al. Prolonged integration site selection of a lentiviral vector in the genome of human keratinocytes. Med. Sci. Monit. 23, 1116–1122 (2017).
Wu, C. & Dunbar, C. E. Stem cell gene therapy: the risks of insertional mutagenesis and approaches to minimize genotoxicity. Front. Med. 5, 356–371 (2011).
Cesana, D. et al. Whole transcriptome characterization of aberrant splicing events induced by lentiviral vector integrations. J. Clin. Invest. 122, 1667–1676 (2012).
Cesana, D. et al. Uncovering and dissecting the genotoxicity of self-inactivating lentiviral vectors in vivo. Mol. Ther. 22, 774–785 (2014).
Liu, M. et al. Genomic discovery of potent chromatin insulators for human gene therapy. Nat. Biotechnol. 33, 198–203 (2015).
Hargrove, P. W. et al. Globin lentiviral vector insertions can perturb the expression of endogenous genes in beta-thalassemic hematopoietic cells. Mol. Ther. 16, 525–533 (2008).
Duverge, A. & Negroni, M. Pseudotyping lentiviral vectors: when the clothes make the virus. Viruses 12, 1311 (2020).
Hirsch, M. L., Agbandje-McKenna, M. & Samulski, R. J. Little vector, big gene transduction: fragmented genome reassembly of adeno-associated virus. Mol. Ther. 18, 6–8 (2010).
Trapani, I. Adeno-associated viral vectors as a tool for large gene delivery to the retina. Genes 10, 287 (2019).
Becirovic, E. et al. AAV vectors for FRET-based analysis of protein–protein interactions in photoreceptor outer segments. Front. Neurosci. 10, 356 (2016).
Barbon, E. et al. Development of a dual hybrid AAV vector for endothelial-targeted expression of von Willebrand factor. Gene Ther. 30, 245–254 (2023).
Chen, Z. R. et al. Co-transduction of dual-adeno-associated virus vectors in the neonatal and adult mouse utricles. Front. Mol. Neurosci. 15, 1020803 (2022).
Kodippili, K. et al. Dual AAV gene therapy for duchenne muscular dystrophy with a 7-kb mini-dystrophin gene in the canine model. Hum. Gene Ther. 29, 299–311 (2018).
Omichi, R., Yoshimura, H., Shibata, S. B., Vandenberghe, L. H. & Smith, R. J. H. Hair cell transduction efficiency of single- and dual-AAV serotypes in adult murine cochleae. Mol. Ther. Methods Clin. Dev. 17, 1167–1177 (2020).
Ghosh, A., Yue, Y. & Duan, D. Efficient transgene reconstitution with hybrid dual AAV vectors carrying the minimized bridging sequences. Hum. Gene Ther. 22, 77–83 (2011).
Trapani, I. et al. Effective delivery of large genes to the retina by dual AAV vectors. EMBO Mol. Med. 6, 194–211 (2014).
Ivanchenko, M. V. et al. PCDH15 dual-AAV gene therapy for deafness and blindness in usher syndrome type 1F models. J. Clin. Invest. (2024).
Ferla, R. et al. Efficacy, pharmacokinetics, and safety in the mouse and primate retina of dual AAV vectors for Usher syndrome type 1B. Mol. Ther. Methods Clin. Dev. 28, 396–411 (2023).
Occelli, L. M. et al. Development of a translatable gene augmentation therapy for CNGB1-retinitis pigmentosa. Mol. Ther. 31, 2028–2041 (2023).
Kügler, S., Kilic, E. & Bähr, M. Human synapsin 1 gene promoter confers highly neuron-specific long-term transgene expression from an adenoviral vector in the adult rat brain depending on the transduced area. Gene Ther. 10, 337–347 (2003).
Kuhn, B., Ozden, I., Lampi, Y., Hasan, M. T. & Wang, S. S. An amplified promoter system for targeted expression of calcium indicator proteins in the cerebellar cortex. Front. Neural. Circuits 6, 49 (2012).
Prasad, K. M., Xu, Y., Yang, Z., Acton, S. T. & French, B. A. Robust cardiomyocyte-specific gene expression following systemic injection of AAV: in vivo gene delivery follows a Poisson distribution. Gene Ther. 18, 43–52 (2011).
Schröder, L. C., Frank, D. & Müller, O. J. Transcriptional targeting approaches in cardiac gene transfer using AAV vectors. Pathogens 12, 1301 (2023).
Zincarelli, C., Soltys, S., Rengo, G. & Rabinowitz, J. E. Analysis of AAV serotypes 1–9 mediated gene expression and tropism in mice after systemic injection. Mol. Ther. 16, 1073–1080 (2008).
Xu, J. Preparation, culture, and immortalization of mouse embryonic fibroblasts. Curr. Protoc. Mol. Biol. 28, 28.1 (2005).
Cowan, C. S. et al. Cell types of the human retina and its organoids at single-cell resolution. Cell 182, 1623–1640.e34 (2020).
Muralidharan, V. & Muir, T. W. Protein ligation: an enabling technology for the biophysical analysis of proteins. Nat. Methods 3, 429–438 (2006).
Shah, N. H. & Muir, T. W. Inteins: nature’s gift to protein chemists. Chem. Sci. 5, 446–461 (2014).
Stevens, A. J. et al. A promiscuous split intein with expanded protein engineering applications. Proc. Natl Acad. Sci. USA 114, 8538–8543 (2017).
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 3, 1101–1108 (2008).
Goldman, A., Harper, S. & Speicher, D. W. Detection of proteins on blot membranes. Curr. Protoc. Protein Sci. 86, 10.8.1–10.8.11 (2016).
Ni, D., Xu, P. & Gallagher, S. Immunoblotting and immunodetection. Curr. Protoc. Immunol. 114, 8.10.1–8.10.36 (2016).
D’Costa, S. et al. Practical utilization of recombinant AAV vector reference standards: focus on vector genomes titration by free ITR qPCR. Mol. Ther. Methods Clin. Dev. 5, 16019 (2016).
Kim, S. et al. Generation, transcriptome profiling, and functional validation of cone-rich human retinal organoids. Proc. Natl Acad. Sci. USA 116, 10824–10833 (2019).
Völkner, M., Pavlou, M., Büning, H., Michalakis, S. & Karl, M. O. Optimized adeno-associated virus vectors for efficient transduction of human retinal organoids. Hum. Gene Ther. 32, 694–706 (2021).
Weinmann, J. et al. Identification of broadly applicable adeno-associated virus vectors by systematic comparison of commonly used capsid variants in vitro. Hum. Gene Ther. 33, 1197–1212 (2022).
Tessmer, K., Borsch, O., Ader, M. & Gasparini, S. J. in Brain Organoid Research (ed. Gopalakrishnan, J.) 81–98 (Springer US, 2023).
Collins, A. L. et al. Mild overexpression of MeCP2 causes a progressive neurological disorder in mice. Hum. Mol. Genet. 13, 2679–2689 (2004).
Xu, Z. et al. Trans-splicing adeno-associated viral vector-mediated gene therapy is limited by the accumulation of spliced mRNA but not by dual vector coinfection efficiency. Hum. Gene Ther. 15, 896–905 (2004).
Lek, A. et al. Death after high-dose rAAV9 gene therapy in a patient with duchenne’s muscular dystrophy. N. Engl. J. Med. 389, 1203–1210 (2023).
Hinderer, C. et al. Severe toxicity in nonhuman primates and piglets following high-dose intravenous administration of an adeno-associated virus vector expressing human SMN. Hum. Gene Ther. 29, 285–298 (2018).
Chew, W. L. et al. A multifunctional AAV–CRISPR–Cas9 and its host response. Nat. Methods 13, 868–874 (2016).
Lim, C. K. W. et al. Treatment of a mouse model of ALS by in vivo base editing. Mol. Ther. 28, 1177–1189 (2020).
Riedmayr, L. M., Böhm, S., Biel, M. & Becirovic, E. Enigmatic rhodopsin mutation creates an exceptionally strong splice acceptor site. Hum. Mol. Genet. 29, 295–304 (2020).
Stevens, A. J. et al. Design of a split intein with exceptional protein splicing activity. J. Am. Chem. Soc. 138, 2162–2165 (2016).
Tornabene, P. et al. Inclusion of a degron reduces levelsof undesired inteins after AAV-mediated proteintrans-splicing in the retina. Mol. Ther. Methods Clin. Dev. 23, 448–459 (2021).
Klauer, A. A. & van Hoof, A. Degradation of mRNAs that lack a stop codon: a decade of nonstop progress. Wiley Interdiscip. Rev. RNA 3, 649–660 (2012).
Becher, B., Waisman, A. & Lu, L. F. Conditional gene-targeting in mice: problems and solutions. Immunity 48, 835–836 (2018).
Lancaster, M. A. & Knoblich, J. A. Generation of cerebral organoids from human pluripotent stem cells. Nat. Protoc. 9, 2329–2340 (2014).
McCracken, K. W., Howell, J. C., Wells, J. M. & Spence, J. R. Generating human intestinal tissue from pluripotent stem cells in vitro. Nat. Protoc. 6, 1920–1928 (2011).
Miller, A. J. et al. Generation of lung organoids from human pluripotent stem cells in vitro. Nat. Protoc. 14, 518–540 (2019).
Takasato, M., Er, P. X., Chiu, H. S. & Little, M. H. Generation of kidney organoids from human pluripotent stem cells. Nat. Protoc. 11, 1681–1692 (2016).
Acknowledgements
This work was supported by the Swiss National Science Foundation (grant nos. 310030_212190 and 320030E_221942 to E.B.) and the German Research Foundation (DFG) (project nos. 513025799/FOR5621 to E.B. and M.B.). This work has also been supported by the University Research Priority Program of the University of Zurich (URPP) ITINERARE—Innovative Therapies in Rare Diseases (to E.B.).
Author information
Authors and Affiliations
Contributions
D.M.M. L.M.R. and Z.G. designed and performed the experiments. D.M.M, L.M.R., Z.G., V.J.W., V.M., B.S., E.U., D.Y.O., C.G., T.H., M.B. and E.B. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
E.B. and M.B. are authors on a patent application covering the splice site module and its applications (PCT/EP2019/086454, filed by ViGeneron GmbH, status: published). The other authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key reference
Riedmayr, L. M. et al. Nat. Commun. 14, 6578 (2023): https://doi.org/10.1038/s41467-023-42386-0
Supplementary information
Supplementary Information (download PDF )
Supplementary Fig. 1.
Source data
Source Data Figs. 5 and 6 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download PDF )
Unprocessed western blots and gels.
Source Data Fig. 6 (download PDF )
Unprocessed western blots.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mittas, D.M., Riedmayr, L.M., Gavrilov, Z. et al. Dual AAV vectors for efficient delivery of large transgenes. Nat Protoc 21, 1466–1522 (2026). https://doi.org/10.1038/s41596-025-01243-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41596-025-01243-8