Abstract
Coronary artery disease (CAD) imposes a significant economic burden in developing countries like India. Timely diagnosis and treatment should be prioritized to mitigate the disease. Current diagnostic tools being invasive and less specific raise the need to develop less invasive and more reliable molecular biomarkers. MicroRNAs (miRNAs) are an emerging class of molecules that can serve as a potential source of non-invasive biomarkers for CAD. The objective of this study was to determine the potential of circulatory miRNAs as diagnostic biomarkers in CAD. In this study, we have reported two microRNAs, miR-128-3p and miR-195-5p in the serum of CAD patients in Indian Population. A total of 124 subjects were recruited which included 89 angiographically proven CAD patients and 35 control subjects. Our results show a significant decrease in the levels of miR-128-3p in CAD patients while there were no significant changes in the levels of miR-195-5p. Further bioinformatics analysis revealed the potential role of miR-128-3p in cholesterol homeostasis. Altered homeostasis due to cholesterol accumulation in macrophages is the driving force behind formation of foam cells which in turn accelerates the progression of CAD. Here, we have shown that miR-128-3p increases cholesterol levels in macrophages by decreasing cholesterol efflux in-vitro.
Similar content being viewed by others
Introduction
Coronary artery disease is characterized by obstruction of coronary arteries due to gradual buildup of atherosclerotic plaque in them1. Plaque can lead to stenosis that limits the blood flow causing ischemia. It can also provoke thrombus formation leading to interruption in blood flow and myocardial infarction. CAD is a leading cause of mortality in low and middle-income countries like India (death rate of 145 per 100,000 individuals) and poses a big economic burden2. Most of the CAD related deaths occur due to lack of timely diagnosis and medical intervention2,3,4.
Currently several diagnostic approaches are being used for the detection of CAD such as electrocardiogram (ECG), echocardiogram, angiography and other molecular biomarkers such as cardiac troponin (cTnT), Creatine kinase-mB (CK-MB), C-reactive protein (CRP)5. However, most of the diagnostic methods are invasive or have limitations based on their specificity, sensitivity or their half-life5. Therefore, there is an urgent need for some reliable, non-invasive biomarkers for optimal detection and management of CAD.
MicroRNAs (miRNA) are a class of small noncoding RNAs (18 to 25 nucleotides) that mediate gene silencing by binding to 3' Untranslated region (UTR) of target genes6. They are involved in diverse biological processes including developmental timing, differentiation, proliferation, cell death and metabolism6. Circulatory micro RNAs are remarkably stable in the serum and are being explored as useful biomarkers for various human disease7,8. This stability for extended periods of time is achieved due to two mechanisms, i.e., formation of a ribonucleoprotein complex with Argonaute proteins and incorporation of miRNAs into exosomes where they are protected against degradation by RNAses in the blood9. Circulating miRNAs are also specific to tissue and disease type which further increase their potential as a diagnostic marker10,11. Recently few studies have also described the role of circulating miRNAs as early diagnostics biomarkers in CAD12,13.
Our previous work showed that microRNAs miR-128-3p & miR-195-5p regulate lipid metabolism. MiR-195 has been shown to regulate cellular cholesterol and triglyceride levels by targeting ACACA, FASN, HMGCR and CYP27B1 the key genes involved in fatty acid and cholesterol synthesis14. MiR-128-3p regulates cholesterol homeostasis by directly targeting Cholesterol efflux via binding to 3′ UTR (untranslated region) of ATP-binding cassette transporters (ABCA1 and ABCG1)15,16. Since lipid metabolism and transport play a major role in development and progression of coronary artery disease, we hypothesize that miR-128-3p & miR-195-5p might be ideal candidates for biomarkers for CAD.
The clinical diagnostic potential of these circulatory microRNAs has not yet been evaluated. Therefore, the present study was designed to explore the diagnostic biomarker potential of miR-128-3p and miR-195-5p for the first time in Indian cohort. In this study, we used serum of CAD patients as well as healthy control subjects and checked for the expression of miR-195-5p and miR-128-3p using quantitative Real Time-PCR (qRT-PCR). Further, using bioinformatics analysis and current literature we show here the important role of these micro-RNAs in CAD.
Material and methods
Study population
A total of 124 cases, 89 CAD patients (69 male and 20 female) and 35 control individuals (12 male and 23 female) aged 30–76 years were recruited for the study from Department of Cardiology, AIIMS, New Delhi between March 2022 to February 2024. Eighty-nine patients were clinically suspected and angiographically proven cases of CAD with stenosis > 50% and followed the inclusive criteria. Patients with dilated cardiomyopathy, chronic kidney disease, other valves and genetic malformations of the heart were excluded from the study. Control participants of similar age group were angiographically shown to have < 10% stenosis. Clinicopathological history of all the subjects and their medication records were procured from the Department of Cardiology, AIIMS.
Ethics statement
The protocols adopted were according to the ethical principles of the declaration of Helsinki. The study protocol complies with all the relevant national regulations (Indian Council of Medical Research, ICMR, India), Institutional Ethical Committee of CSIR-IGIB, Delhi and AIIMS, New Delhi (IEC-153/06.03.2020, RP-33/2020). A written and signed informed consent form was obtained from all the participants.
Sample collection and serum isolation
5 ml of peripheral blood was collected from the subjects in primary blood collection tube with clot activator (S-Monovette® Serum-Gel 5 ml tubes, Sarstedt, Germany, cat. no. 02.1388). Serum was separated using two rounds of centrifugation (1900×g at 4 °C for 10 min and then 16,000×g at 4 °C for 10 min). The serum was collected and stored at − 80 °C until further processing.
Biochemical profiling
Biochemical parameters including serum HbA1c (%), Total Cholesterol (TC), (mg/dl), Triglyceride (TG), (mg/dl), High density lipoproteins (HDL-C) (mg/dl), Low density lipoproteins (LDL-C) (mg/dl), Very low-density lipoproteins (VLDL-C) (mg/dl) were analyzed in the SMART Lab at AIIMS, New Delhi on automated chemistry analyzers.
RNA isolation, cDNA synthesis and quantitative real time PCR
RNA was isolated from serum using miRNeasy serum/plasma advanced kit (Qiagen, USA, Cat No. 217204) as per manufacturers protocol. cel-miR-39 (Norgen Biotek, Canada, Cat No. 59000) was used as a spike-in control. cDNA preparation was done using High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, USA, Cat No. 4368814) as per manufacturer’s protocol. Stemloop RT primers were used for cel-miR-39, miR-128-3p and miR-195-5p. Primer sequences are mentioned in Table 1.
For qRTPCR, reaction mixture was prepared using Kapa SYBR fast (Sigma-Aldrich, USA, Cat no. KM4101) dye as per manufacturer’s protocol and PCR was done on Roche lightcycler96 (Roche Diagnostics, Switzerland).
Bioinformatic analysis
The list of gene targets for microRNA hsa-miR-128-3p were downloaded from databases such as targetscan 8.0, miRDB 6.0 and miRTarBase 9.0. A list of common gene targets were obtained from a Venn-diagram using Venny 2.1 (https://bioinfogp.cnb.csic.es/tools/venny). The common targets were further uploaded to ShinyGO database (http://bioinformatics.sdstate.edu/go/) for pathway analysis. Top KEGG pathways and top GO (Gene Ontology) Biological Processes were selected for further biological experiments17,18,19.
Cell culture
THP-1 macrophage cells were cultured using RPMI 1640 medium (Ref no. R4130, Thermo Fisher Scientific, MA, USA) supplemented with 10% Fetal Bovine Serum (Ref no. 10082-147, Thermo Fisher Scientific, MA, USA) and 100 IU/mL penicillin–streptomycin (Gibco, Thermo Fisher Scientific, MA, USA). The cells were incubated at 37 °C and 5% CO2 level. For differentiation into macrophages, the cells were treated with 100 nm PMA (phorbol 12-myristate 13-acetate) for 48 h. Transient transfection was carried out in differentiated THP-1 cells for further experiments. Cells were transfected with miR-128 plasmid20 or antimiR-128-3p (AM-128) (AM17000, assay ID AM11746, Thermo Fisher Scientific, MA, USA) using Ingenio Electroporation Kit (MIR 50112, mirus bio, USA) as per manufacturer’s instructions. Cells transfected with pSilencer 4.1 vector and anti-miR negative control (NC) was used as control for miR-128-3p plasmid and antimiR-128-3p respectively15.
Cholesterol efflux assay
The cholesterol efflux of differentiated THP-1 macrophages was examined using cholesterol efflux assay kit (ab196985, Abcam, USA) following the manufacture's instruction. Briefly, transfected THP-1 cells post 24 h were labelled with cholesterol for 1 h. After labelling cells were kept in equilibration buffer for 24 h and were then loaded with cholesterol acceptors for 4 h. Finally, cells were lysed and both the supernatant and lysate were transferred to a 96 well plate for measuring fluorescence (Ex/Em = 485/523 nm).
Statistical analysis
Descriptive data on the participants recruited was summarized using measures of central tendency and dispersion. The normality of data was analysed using the Shapiro–Wilk test which was significant (non-parametric) in the case of CAD patients. Therefore, the differences between patient and control groups were tested using the Mann–Whitney U test. The expression levels of the miRNAs were calculated using the delta CT method. All the graphs were constructed using GraphPad prism version 8.1.1 (GraphPad software, San Diego, CA, USA). The Receiver Operating Characteristic (ROC) curve analysis was done and the respective area under curve (AUC) was calculated in order to assess the diagnostic potential of the circulatory miRNAs between CAD patients and healthy control group. Multiple logistic regression was done to adjust the effect of potential confounders of the study on miRNA expression. Pearson correlation analysis was done for correlation of miRNA expression and other cardiovascular risk factors associated with the subjects involved in the study. All statistical tests were done using GraphPad prism version 8.1.1 (GraphPad software, San Diego, CA, USA).
Results
Clinical characteristics along with CAD risk factors among the study participants.
A total of 124 cases (89 CAD patients and 35 control individuals) were recruited for the study. Age of participants showed an average of 55.90 ± 9.92 (CAD group) and 50.32 ± 8.25 (Control group). Gender, body mass index (BMI), Diabetes mellitus status (DM), hypertension, blood lipid profiles were recorded (Table 2). The male gender was more represented in CAD groups with 69 (77.53%) as compared to 12 (34.28%) in the control group, respectively. Total cholesterol, LDL, HDL, VLDL, Triglyceride, other medications and therapies were also recorded as shown in Table 2. 95% of CAD patients were on Aspirin, 92.5% on Statin and 90% were on beta blockers whereas 63.4% were taking ACE inhibitors and 14.6% were on Angiotensin receptor blockers. On the other hand, 53.8% of Control subjects were on Statin, 46% on beta blockers, 76.9% were taking Aspirin, 30.8% were taking Angiotensin receptor blockers and only 15.4% were taking ACE inhibitors.
Circulating miR-128-3p and miR-195-5p levels in CAD vs. control subjects
We first did an extensive review of literature for finding if there are any validated targets of miR-128-3p and miR-195-5p that might play a role in various aspects of coronary artery disease14,15,16,21,22,23. As shown in Fig. 1, both miR-128 and miR-195 target genes affecting major cell types involved in CAD i.e., macrophages, vascular smooth muscle cells and endothelial cells. Both miRNAs also target genes involved in lipid metabolism and synthesis as well as vascular smooth muscle proliferation and migration. Hence, we further evaluated the expression level of both miR-128-3p and miR-195-5p in patients with established CAD and control subjects using quantitative Real Time-Polymerase Chain Reaction (qRT‐PCR).
Functionally validated target genes of miR-128-3p and miR-195-5p in coronary artery disease. The figure is a representation of the role of both miR-195 and miR-128 in various aspects of coronary artery disease. The validated target genes of each miRNA with respect to atherosclerosis and other cardiovascular disorders are shown.
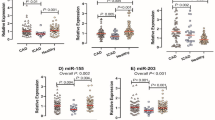
As shown in Fig. 2a, we found the average delta-Ct values of miR-128-3p to be 17.50 ± 3.28 in CAD patients in contrast to 15.47 ± 3.89 in control subjects. In case of miR-195-5p the average delta-Ct values ranged from 16.85 ± 3.92 in case of CAD patients and 15.79 ± 3.74 in case of control subjects as depicted in Fig. 2b. Relative expression pattern [log2 fold change (Mean ± SEM)] of both the microRNAs have been depicted in Fig. 2c and d. Comparing the expression levels of both the miRNAs using Mann–Whitney U test, we can see that miR-128-3p is 2.03 ± 0.35 fold (p-value = 0.0053) decreased in CAD patients as compared to control subjects, whereas miR-195-5p was shown to be 1.24 ± 0.37 fold (p value = 0.1221) decreased in CAD patients vs control. Therefore, our findings were significant for miR-128-3p, but not in the case of miR-195-5p.
Differential Expression pattern of miR-128-3p & miR-195-5p: (a) Expression levels of miR-128-3p in CAD patients (n = 89) in comparison to control subjects (n = 35) in terms of average delta Ct values. (b) Expression levels of miR-195-5p in CAD patients (n = 89) vs. control subjects (n = 35) in terms of average delta Ct values. Results were measured as Mean ± SD. (c) Relative Fold change of miR-128-3p in CAD patients as opposed to control subjects. It was significantly decreased with a p value of 0.0053. Results are (Mean ± SEM) (d) Relative fold change of miR-195-5p in CAD patients when compared to control subjects. miR-195-5p levels did not show any significant changes (p value = 0.122) Results are (Mean ± SEM).
Assessment of circulating miR-128-3p and miR-195-5p for their diagnostic potential
We conducted receiver-operating-characteristic (ROC) curve analysis to further explore the potential and application of these circulatory miRNAs (miR-128-3p and miR-195-5p) as novel and possible diagnostic biomarkers of CAD (Fig. 3). A reasonably high predictive value/area under the curve (AUC) was obtained in CAD patients in the case of miR-128-3p.
Receiver operating Characteristic (ROC) curve: diagnostic potential of miR-128-3p and miR-195-5p in coronary artery disease depicted by Receiver operating characteristic curve. (a) Area under Curve (AUC) of miR-128-3p is 0.6602, 95% confidence interval is 0.5421 to 0.7782 while the p value is 0.0056. (b) ROC curve of miR-195-5p with an AUC of 0.5841, 95% confidence interval is 0.4668 to 0.7014 and p Value is 0.1458.
The area under the curve of miR-128-3p was 0.6602 with a standard error of 0.06023 (p value 0.0056) and 95% confidence interval was between 0.5421 to 0.7782 (Fig. 3a). This implies that miR-128-3p has a reasonable predictive value (66%) for differentiating between CAD patients and healthy controls. In the case of miR-195-5p the area under the curve was 0.5841 with a standard error of 0.05986 (p value 0.1458, non-significant) and 95% confidence interval was between 0.4668 to 0.7014 (Fig. 3b). The optimal cut-off point for each miRNA was determined using the largest sum of sensitivity and specificity which was 14.81 for miR-128-3p and 13.94 for miR-195-5p24. The detail of this analysis for both miRNAs is provided in Table 3.
As a result, our findings imply that serum levels of miR-128-3p may be useful as a prospective diagnostic biomarker for the identification and prediction of CAD patients.
Multivariate analysis of miR-128-3p
To study the interrelationship between miRNA expression and classical CAD risk factors we did Pearson’s correlation analysis of these factors with miR-128-3p delta ct- values. No significant correlation was found between miR-128 and gender, BMI, hypertension, Diabetes Mellitus, LDL, VLDL, Triglycerides, Total Cholesterol. Poor correlation was found for age (R squared 0.06, p-value 0.005) and HDL (R squared 0.07, p-value 0.04).
MiR-128-3p was further evaluated as a potential independent predictor of coronary artery disease (CAD) using multiple logistic regression. The model adjusted for confounding factors, including the use of aspirin, statins, Beta-blockers, Ace-Inhibitors and angiotensin receptor blockers (ARBs), which are commonly prescribed in the patient cohort. MiR-128-3p demonstrated a statistically significant association with the presence of CAD (Log-likelihood ratio (G squared) test, p-value 0.034), suggesting its potential role as an independent biomarker. The odds ratio (OR 1.241, 95% CI 0.8566 to 1.901) adjusted for the confounding medications indicated that an increase in miR-128 delta-ct values was correlated with an elevated risk of CAD, independent of the drug regimen. Figure 4 presents a comparative analysis of the Receiver Operating Characteristic (ROC) curves for two multivariate regression models. The inclusion of miR-128-3p alongside other potential confounders in the model1 results in a superior predictive performance, as indicated by a higher Area Under the Curve (AUC) value of 0.9019, compared to the model 2 that accounts only for the confounding factors, which has an AUC of 0.8212.
Multivariate Analysis: Comparison between ROCs of two models used for multivariate analysis. Model 1 (AUC 0.9019) has miR-128-3p and other confounding factors such as age, gender, statin, aspirin, ARB, beta blocker, ACE inhibitors. Model 2 (AUC 0.8212) contains all the confounding factors of Model 1 except miR-128. Model 1 shows a better predictive value than model 2.
Function and pathway enrichment analysis of miR-128-3p
Using target prediction tools such as TargetScan, miRDB, miRTarBase we explored the top pathways regulated by miR-128-3p. Comparing different databases we found a total of 136 common predicted targets of miR-128-3p (Fig. 5a, Supplementary Table 1). The common targets were further uploaded to ShinyGO database (http://bioinformatics.sdstate.edu/go/) for pathway analysis. Figure 5b shows a list of the top 20 KEGG pathways as mentioned in Table 4. Several pathways directly related to CAD such as Insulin signaling pathway, diabetic cardiomyopathy pathway, lipid and atherosclerosis pathway were observed to be regulated by miR-128-3p. In order to further understand the role of these genes in the pathophysiology of CAD we also performed GO Enrichment analysis for top 50 biological processes (Fig. 5c). The common target genes were highly enriched in lipid homeostasis. The target genes of these pathways have been mentioned in Table 5. Some of the proteins such as LDLR, ABCA1, RXRA have been previously shown to be the direct target of miR-128-3p by our lab15 (Table 5).
Bioinformatic analysis of predicted targets of miR-128-3p: (a) Venny tool was used to create a Venn diagram of common targets of miR-128-3p from three different target prediction tools TargetScan8.0, miRDB6.0, miRTarBase. (b) KEGG pathway analysis of the overlapping genes done using ShinyGO 0.77 shows Lipid and Atherosclerosis as a significant pathway with an FDR of 0.013. (c) Gene Ontology (GO)enrichment analysis of top 50 Biological Processes of common targets was also done using ShinyGO 0.77.
In our study miR-128-3p has been shown to be significantly decreased in CAD patients as compared to healthy controls. Our current findings strongly suggest that miR-128-3p could be a potential biomarker for CAD diagnosis. However, the sensitivity and specificity of our current circulating miR-128-3p need to be further investigated in a larger cohort.
MiR-128 is involved in the regulation of cholesterol efflux from macrophages
It is evident from the bioinformatic analysis that miR-128 plays an important role in the regulation of cholesterol homeostasis by targeting key genes involved in cholesterol transport. Cholesterol homeostasis regulates the rate of lipid uptake and accumulation by macrophages to form foam cells which is the preliminary major event in the development of coronary artery disease1. Reverse Cholesterol Transport (RCT) or cholesterol efflux in macrophages attenuates the formation of foam cells and thus reverses the condition of CAD1. Therefore, we decided to investigate the role of miR-128 in modulating cholesterol efflux in macrophage cells (THP-1 cells) in vitro (n = 3). From the results we can see that overexpression of miR-128 significantly reduces the cholesterol efflux by 15.27 percent as compared to control (pSilencer) (p value 0.04). Inhibition of miR-128 using antimiR increased the efflux by 20.10% as compared to negative control (NC) (p value 0.15) (Fig. 6).
Regulation of Cholesterol Efflux in macrophage cells by miR-128-3p: Figure represents cholesterol efflux % of differentiated THP-1 macrophage cells in during overexpression of miR-128-3p (miR-128) and Inhibition using anti-miR-128-3p (AM). Overexpression of miR-128-3p significantly decreases cholesterol efflux by 15.27% as compared to pSil (pSilencer4.1) control. Inhibition of miR-128-3p increased the efflux by 20.10% in comparison to the negative control.
Discussion
Due to their small size and protection from endogenous RNase activity provided by their association with certain lipid-based carriers or their encapsulation within micro vesicles, circulating miRNAs are extremely resistant to destruction25. Circulatory miRNAs have become a major and intriguing prospect for the discovery of novel regulators of cardiovascular illnesses, including CAD, thanks to their benefits over other traditional methods of diagnostics25,26.
This pilot study based on the Indian cohort was aimed at identification of novel microRNA-based biomarkers in coronary artery disease. To summarize our findings, miR-128-3p was found to be significantly decreased in CAD patients as compared to control whereas no significant difference was observed in case of miR-195-5p. The diagnostic potential evaluated using the ROC curve has shown miR-128-3p to have 66% accuracy (AUC 0.6602) in distinguishing CAD patients from control.
Several micro RNAs have been reported over the years which regulate coronary artery disease27,28,29,30,31,32. Some of these micro RNAs are present in circulation and few have been reported to be differentially expressed in serum/plasma30,31,32,33. To report a few, Ling et al. found that the exosomal miR-126 levels were positively correlated in cases of unstable angina (n = 31) as well as acute myocardial infarction (n = 34)34. In another study by O′Sullivan et al. on a sample set of 150 patients, miR-93-5p was reported to be significantly upregulated in stable CAD when compared to the control group35. However, most of these studies have been carried out in Caucasian population. In this study we have reported two miRNAs miR-128-3p and miR-195-5p in Indian population. These miRNAs have a known role in metabolic disorders as described previously. As per our results only miR-128-3p can clearly differentiate between CAD and control population. MiR-128-3p serum levels has shown negative correlation with respect to risk of CAD. Similar to our findings, few microRNAs have also been reported in Indian population such as Reddy et al. in 2019 reported miR-33 to be overexpressed by 2.9 folds in CAD patients than in control group (n = 60)36. Kumar et al. in 2020 reported that miR-133b & miR-21 as a predictive biomarker for coronary artery disease (AUC 0.8 & 0.79 respectively) in a cohort of 147 subjects37.
To further elucidate the role of miR-128-3p in coronary artery disease we investigated its potential gene targets and major affected signaling pathways using several bioinformatic tools. Our results showed that miR-128-3p was significantly associated with lipid homeostasis. In silico analysis using publicly available databases identified several genes related to cholesterol metabolism and efflux that were inversely correlated with miR-128-3p expression. These results indicate that miR-128-3p may have a broader impact on cholesterol homeostasis.
Cholesterol efflux is a key process for maintaining cellular cholesterol homeostasis and preventing atherosclerosis38. Perturbation of homeostasis leads to increased accumulation of cholesterol resulting in foam cell development and exacerbation of a pro-inflammatory state that in turn promotes atherosclerosis development. Cholesterol efflux is mediated by ATP-binding cassette transporters (ABCA1 and ABCG1) that transfer cholesterol to apolipoprotein A-I (apoA-I) and high-density lipoprotein (HDL) particles, respectively39. The expression of ABCA1 and ABCG1 is regulated by nuclear receptors such as liver X receptor (LXR) and retinoid X receptor (RXR)39. Previous studies have shown that microRNAs (miRNAs) can modulate the expression of genes involved in cholesterol metabolism and efflux27,40. In a study done by Rayner et al., miR-33 was shown to inhibit the expression of ABCA1 and ABCG1, thereby repressing cholesterol efflux to ApoA1 and HDL, respectively41. Jin et al. showed that miR-19a-3p regulates apoptosis and cholesterol efflux through down-regulation of SDC-1, ABCA1, ABCG1, TGF-β1 and p-Smad3 proteins in ox-LDL-induced HAECs42.
In our previous lab findings, it was reported that miR-128-3p directly targets ABCA1, ABCG1 and RXRα, and reduces their expression levels in different cell types15. We hypothesized that miR-128-3p may modulate cholesterol efflux and affect the development of atherosclerosis in CAD patients. To validate the functional role of miR-128-3p in cholesterol efflux, we performed an efflux assay using THP-1 macrophages transfected with either miR-128 plasmid or inhibitor. We found that overexpression of miR-128 significantly decreased the cholesterol efflux to cyclodextrin, whereas inhibition of miR-128 increased the efflux. Together our findings confirmed that miR-128 negatively regulates cholesterol efflux in macrophages.
In this study, we found that overexpression of miR-128 inhibited the expression of genes involved in regulation of cellular cholesterol homeostasis in macrophages. However, we also observed that miR-128-3p was decreased in the extracellular secretion in serum of patients with CAD. These contrasting results may be due to miR-128’s varied regulatory effects across distinct cell types implicated in CAD. Previous studies have shown that miR-128-3p can modulate the function of endothelial cells and Vascular smooth muscle cells (VSMCs) which are also involved in CAD development and progression43. For instance, miR-128-3p can inhibit endothelial cell proliferation and migration by targeting VEGF1 and can regulate proliferation, differentiation, and migration in VSMCs43,44. Therefore, the net effect of miR-128-3p on CAD may depend on the balance between its actions on different cell types.
Study limitations
Our study highlights promising data on the dysregulation of miR-128-3p in CAD and its potential role in cholesterol regulation. However, there are several limitations to consider. The research was based on a small group of CAD patients and healthy individuals, necessitating further validation of hsa-miR-128-3p’s diagnostic accuracy in a broader sample. Moreover, the lack of longitudinal data calls for additional studies to explore the prognostic implications of hsa-miR-128-3p. To ensure the findings are generalizable, it’s crucial to replicate the study across diverse cohorts in various regions of India.
CAD is a complex disease influenced by genetic and environmental factors. Understanding miR-128-3p’s role in CAD is crucial for validating its impact on the disease’s progression. The effects of epigenetic changes, especially those caused by related conditions like hypertension and diabetes or medications, are not fully understood44. Additionally, CAD’s complexity, involving interactions among proteins in different cellular pathways, underscores the need to confirm our findings across various cell types (VSMCs, endothelial cells) involved in CAD development and pathogenesis.
Conclusion
In our study miR-128-3p has been shown to be decreased in CAD patients as compared to healthy individuals. Our study, which included 124 subjects, provides evidence that miR-128-3p is significantly correlated to the chance of developing CAD. We have shown miR-128-3p to regulate cholesterol efflux in macrophages which is an important event in the development of coronary artery disease. Examination of exosomal miR-128-3p and validation of the status of miR-128-3p in patient cohorts stratified by severity of atherosclerosis could be explored for better understanding in patients with CAD in further studies.
Data availability
The qRT-PCR data generated in the current study is available from the corresponding author on reasonable request.
Abbreviations
- CAD:
-
Coronary artery disease
- ECG:
-
Electrocardiogram
- cTnT:
-
Cardiac troponin
- CK-MB:
-
Creatine kinase
- CRP:
-
C-reactive protein
- ROC:
-
Receiver’s operating characteristic
- AUC:
-
Area under curve
- TC:
-
Total cholesterol
- TG:
-
Triglyceride
- HDL:
-
High density lipoproteins
- LDL:
-
Low density lipoproteins
- VLDL:
-
Very low-density lipoproteins
References
Libby, P. & Theroux, P. Pathophysiology of coronary artery disease. Circulation 111, 3481–3488 (2005).
Prabhakaran, D., Jeemon, P. & Roy, A. Cardiovascular diseases in India: Current epidemiology and future directions. Circulation 133, 1605–1620 (2016).
Malakar, A. K. et al. A review on coronary artery disease, its risk factors, and therapeutics. J. Cell Physiol. 234, 16812–16823 (2019).
Ralapanawa, U. & Sivakanesan, R. Epidemiology and the magnitude of coronary artery disease and acute coronary syndrome: A narrative review. J. Epidemiol. Glob. Health 11, 169–177 (2021).
Dhingra, R. & Vasan, R. S. Biomarkers in cardiovascular disease: Statistical assessment and section on key novel heart failure biomarkers. Trends Cardiovasc. Med. 27, 123 (2017).
Hata, A. Functions of microRNAs in cardiovascular biology and disease. Annu. Rev. Physiol. 75, 69–93 (2013).
Ji, J. et al. MicroRNA expression, survival, and response to interferon in liver cancer. N. Engl. J. Med. 361, 1437–1447 (2009).
Schetter, A. J. et al. MicroRNA expression profiles associated with prognosis and therapeutic outcome in colon adenocarcinoma. JAMA 299, 425–436 (2008).
Arroyo, J. D. et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA 108, 5003–5008 (2011).
O’Brien, K., Breyne, K., Ughetto, S., Laurent, L. C. & Breakefield, X. O. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 21, 585–606 (2020).
Zhou, M. et al. Circulating organ-specific microRNAs serve as biomarkers in organ-specific diseases: Implications for organ allo- and xeno-transplantation. Int. J. Mol. Sci. 17, 1232 (2016).
Dong, J. et al. Potential role of lipometabolism-related microRNAs in peripheral blood mononuclear cells as biomarkers for coronary artery disease. J. Atheroscler. Thromb. 24, 430–441 (2017).
Bergami, M., Fabin, N., Cenko, E., Bugiardini, R. & Manfrini, O. MicroRNAs as potential biomarkers in coronary artery disease. Curr. Top. Med. Chem. 23, 454–469 (2023).
Singh, R., Yadav, V., Kumar, S. & Saini, N. MicroRNA-195 inhibits proliferation, invasion and metastasis in breast cancer cells by targeting FASN, HMGCR, ACACA and CYP27B1. Sci. Rep. 5, 17454 (2015).
Adlakha, Y. K. et al. Pro-apoptotic miRNA-128-2 modulates ABCA1, ABCG1 and RXRα expression and cholesterol homeostasis. Cell Death Dis. 4, e780 (2013).
Chandra, A., Sharma, K., Pratap, K., Singh, V. & Saini, N. Inhibition of microRNA-128-3p attenuates hypercholesterolemia in mouse model. Life Sci. 264, 118633 (2021).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 28, 1947–1951 (2019).
Kanehisa, M. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Kanehisa, M., Furumichi, M., Sato, Y., Kawashima, M. & Ishiguro-Watanabe, M. KEGG for taxonomy-based analysis of pathways and genomes. Nucleic Acids Res. 51, D587–D592 (2023).
Adlakha, Y. K. & Saini, N. MicroRNA-128 downregulates Bax and induces apoptosis in human embryonic kidney cells. Cell Mol. Life Sci. 68, 1415–1428 (2011).
Bras, J. P. et al. miR-195 inhibits macrophages pro-inflammatory profile and impacts the crosstalk with smooth muscle cells. PLoS ONE 12, e0188530 (2017).
Shen, Z. & Li, H. Long non-coding RNA GAS5 knockdown facilitates proliferation and impedes apoptosis by regulating miR-128-3p/FBLN2 axis in ox-LDL-induced THP-1 cells. Clin. Hemorheol. Microcirc. 77, 153–164 (2021).
Zheng, H., Yu, Z., Wang, H., Liu, H. & Chen, X. MicroRNA-195-5p facilitates endothelial dysfunction by inhibiting vascular endothelial growth factor A in gestational diabetes mellitus. Reprod. Biol. 22, 100605 (2022).
Habibzadeh, F., Habibzadeh, P. & Yadollahie, M. On determining the most appropriate test cut-off value: The case of tests with continuous results. Biochem. Med. https://doi.org/10.11613/BM.2016.034 (2016).
Momen-Heravi, F., Getting, S. J. & Moschos, S. A. Extracellular vesicles and their nucleic acids for biomarker discovery. Pharmacol. Ther. 192, 170–187 (2018).
Heo, J. & Kang, H. Exosome-based treatment for atherosclerosis. Int. J. Mol. Sci. 23, 1002 (2022).
Lu, X. et al. MicroRNA-320b modulates cholesterol efflux and atherosclerosis. J. Atheroscler. Thromb. 29, 200–220 (2022).
Duan, L. et al. miR194 hypomethylation regulates coronary artery disease pathogenesis. BMC Med. Genomics 15, 264 (2022).
Ma, C., Zheng, X., Wu, X., Cheng, J. & Zhang, K. microRNA-181c-5p stimulates the development of coronary artery disease by targeting SIRT1. Hellenic J. Cardiol. 69, 31–40 (2023).
Han, J. et al. Plasma-derived exosomal let-7c-5p, miR-335-3p, and miR-652-3p as potential diagnostic biomarkers for stable coronary artery disease. Front. Physiol. 14, 1161612 (2023).
Vancheri, C. et al. Downregulation of circulating Hsa-miR-200c-3p correlates with dyslipidemia in patients with stable coronary artery disease. Int. J. Mol. Sci. 24, 1112 (2023).
Li, L., He, X., Liu, M., Yun, L. & Cong, B. Diagnostic value of cardiac miR-126-5p, miR-134-5p, and miR-499a-5p in coronary artery disease-induced sudden cardiac death. Front. Cardiovasc. Med. 9, 944317 (2022).
Fazmin, I. T., Achercouk, Z., Edling, C. E., Said, A. & Jeevaratnam, K. Circulating microRNA as a biomarker for coronary artery disease. Biomolecules 10, 1354 (2020).
Ling, H., Guo, Z., Shi, Y., Zhang, L. & Song, C. Serum exosomal microRNA-21, microRNA-126, and PTEN are novel biomarkers for diagnosis of acute coronary syndrome. Front. Physiol. 11, 654 (2020).
O’Sullivan, J. F., Neylon, A., McGorrian, C. & Blake, G. J. miRNA-93–5p and other miRNAs as predictors of coronary artery disease and STEMI. Int. J. Cardiol. 224, 310–316 (2016).
Reddy, L. L., Shah, S. A. V., Ponde, C. K., Rajani, R. M. & Ashavaid, T. F. Circulating miRNA-33: A potential biomarker in patients with coronary artery disease. Biomarkers 24, 36–42 (2019).
Kumar, D. et al. Circulatory miR-133b and miR-21 as novel biomarkers in early prediction and diagnosis of coronary artery disease. Genes 11, 164 (2020).
Ouimet, M., Barrett, T. J. & Fisher, E. A. HDL and reverse cholesterol transport. Circ. Res. 124, 1505–1518 (2019).
Dergunov, A. D. & Baserova, V. B. Different pathways of cellular cholesterol efflux. Cell Biochem. Biophys. 80, 471–481 (2022).
Xu, Y. et al. Macrophage miR-34a is a key regulator of cholesterol efflux and atherosclerosis. Mol. Ther. 28, 202–216 (2020).
Rayner, K. J. et al. MiR-33 contributes to the regulation of cholesterol homeostasis. Science 328, 1570–1573 (2010).
Jin, Q., Deng, Y., Li, L., Chen, R. & Jiang, L. miR-19a-3p affected ox-LDL-induced SDC-1/TGF-β1/Smad3 pathway in atherosclerosis. Cell Mol. Biol. 69, 75–81 (2023).
Zhou, J. et al. MiR-128-3p directly targets VEGFC/VEGFR3 to modulate the proliferation of lymphatic endothelial cells through Ca2+ signaling. Int. J. Biochem. Cell Biol. 102, 51–58 (2018).
Farina, F. M. et al. miR-128-3p is a novel regulator of vascular smooth muscle cell phenotypic switch and vascular diseases. Circ. Res. 126, e120–e135 (2020).
Acknowledgements
This work was supported by research grants from Indian Council of Medical Research (Project—“Identification and functional validation of candidate microRNA biomarkers in coronary artery diseases- An exploratory study”, 5/4/1-12/2020-NCD-I, GAP241). We acknowledge the Council of Scientific and Industrial Research and Academy of Scientific and Innovative Research for research facilities. We also acknowledge Saumya Johri, CSIR-IGIB for her help with transfection reagents and protocol; Dolly Rathore, All India Institute of Medical Sciences, Delhi for providing us with valuable patient biological samples and relevant data.
Author information
Authors and Affiliations
Contributions
R.R.C.: Conception, Methodology, Data analysis, Data curation, Investigation, Writing-original draft, review & editing; H.G.: Methodology, Data curation, Investigation, review & editing; S.B.: Sample Collection, Biochemical analysis, Data curation; A.S.: Sample Collection, Biochemical analysis, Draft review & editing; A.R.: Sample Collection, Biochemical analysis, Draft review & editing, Funding acquisition; N.S.: (corresponding author): Conception, Methodology, Data analysis, Investigation, Supervision; Writing-review & editing, Funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Choudhury, R.R., Gupta, H., Bhushan, S. et al. Role of miR-128-3p and miR-195-5p as biomarkers of coronary artery disease in Indians: a pilot study. Sci Rep 14, 11881 (2024). https://doi.org/10.1038/s41598-024-61077-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-61077-4
Keywords
This article is cited by
-
MicroRNA linked to coronary heart disease
Nature India (2024)
-
Exploring the versatility of miRNA-128: a comprehensive review on its role as a biomarker and therapeutic target in clinical pathways
Molecular Biology Reports (2024)