Abstract
We aimed to investigate the preventive effect of vitamin D2 on COVID-19 and the improvement of symptoms after COVID-19 infection. The study recruited 228 health care workers who tested negative PCR or antigen for COVID-19. Subjects were randomly allocated to vitamin D2 or non-intervention at a ratio 1:1. Subjects recorded PCR or antigen tests and the symptoms of COVID-19 twice a week during the follow-up visit. The concentration of serum 25-hydroxyvitamin D (25(OH)D), C-reaction protein (CRP), complement component C1q and inflammatory cytokines were measured. The rates of COVID-19 infection were 50.5% in the vitamin D2 group and 52.4% in the non-intervention group (P = 0.785). There was no difference in the COVID-19 symptoms between the two groups. The mean 25(OH)D level significantly increased from 14.1 to 31.1 ng/mL after administration (P < 0.001). The difference between the two groups was not significant for the concentrations of CRP, C1q and inflammatory cytokines on the thirtieth day of the trial. According to the second level of vitamin D, there was a 14.3% difference in positive infection rates between the vitamin D adequate (> 30 ng/mL) and deficient groups (< 20 ng/mL). Adequate vitamin D had a tendency to prevent COVID-19.
Trial registration: ClinicalTrials.gov NCT05673980, dated: 12/2022.
Similar content being viewed by others
Introduction
The coronavirus disease 2019 (COVID-19) pandemic spread rapidly worldwide, affecting the quality of life of hundreds of millions of people and causing the deaths of hundreds of thousands of people. COVID-19 has already had a huge impact on human health and the economy around the world1. At present, there is currently a lack of effective measures to prevent COVID-19, and even vaccines cannot completely protect against rapidly mutating virus variants. The results of a systematic review showed that vaccine effectiveness against COVID-19 infection decreased by 21% from 1 to 6 months after full vaccination among people of all ages2. During the COVID-19 pandemic, health care workers have been highly exposed, and the infection rate in hospitals is extremely high3,4. In addition, health care workers may have also faced high work pressure, physical exhaustion, and other threats during the COVID-19 pandemic5. It is crucial to prioritize the prevention of COVID-19 among health care workers.
Vitamin D is a nutrient element that is important for bone health, and it acts by promoting the absorption of calcium by the small intestinal mucosa and the deposition of calcium at the site of new bone formation6,7. Vitamin D also plays an important role in regulating immune function8,9. Vitamin D can modulate immunity; on the one hand, vitamin D can activate non-specific immunity and enhance the body's antiviral ability10; on the other hand, vitamin D can inhibit the inflammatory response and prevent the occurrence of cytokine storms11,12. At present, there is still a great deal of controversy about the preventive and treatment effect of vitamin D on COVID-19. A randomized controlled clinical trial (RCT) showed that high-dose vitamin D reduced the proportion of hospitalized COVID-19 patients admitted to the ICU13. In an observational study of 4599 US military veterans, the hospitalization rate due to COVID-19 infection decreased from 24.1 to 18.7% (P = 0.009) when the patients' serum 25(OH)D levels increased from 15 to 60 ng/ml, and the death rate decreased from 10.4 to 5.7% (P = 0.001)14. Vitamin D deficiency and insufficiency were associated with increased severity and unfavorable outcome after 28 days15. However, some clinical trials have shown that vitamin D does not prevent or treat COVID-19. A trial showed that a single high dose of vitamin D did not significantly reduce the length of hospital stay for COVID-19 patients16.
Whether vitamin D can prevent COVID-19 and improve symptomatic COVID-19 remains unknown. There is still a need for more RCT studies to support the role of vitamin D in the prevention and treatment of COVID-19. The objective of this multicenter randomized clinical trial was to investigate the preventive effect of vitamin D2 on COVID-19 and the improvement of symptoms after COVID-19 infection in health care workers.
Methods
Study objectives
To investigate the preventive effect of vitamin D2 on COVID-19 and the improvement of symptoms after COVID-19 infection in health care workers.
Study design
This was a multicenter, open-label, randomized controlled trial. The subjects provided written informed consent before participation. They were randomly assigned to receive 200,000 IU vitamin D2 fortnightly or no intervention. This study was conducted on December 23, 2022 and was closed to recruitment on December 26, 2022, and a total of 262 participants were enrolled. The trial protocol is available in See supplement file 2.
Ethical approval
This study has been registered in a clinical trial registry (clinicaltrial.gov, ID: NNCT05673980 on 22/12/2022). This study protocol was reviewed and approved by the Peking University Third Hospital Medical Science Research Ethics Committee, approval number was M2022828. This manuscript was written in accordance with the CONSORT 2010 guidelines for reporting randomized controlled trials. Signed informed consent must be obtained from participants before any operational procedures related to clinical trials. The informed consent was dated and signed by the subjects.
Patient and public involvement
Patient or the public did not be involved in the design, conduct, reporting, or dissemination plans of this study.
Participants
The study included 248 health care workers from Peking University Third Hospital, Beijing Haidian Hospital, Chongli Hospital and Yanqing Hospital. All subjects were off vitamin D or received only preventive doses before signing the informed consent. The inclusion criteria consisted of having a negative PCR or antigen test and being asymptomatic for COVID-19. The exclusion criteria consisted of pregnant and lactating women, long-term vegetarians, and a history of serious concomitant diseases, chronic diarrhea, subtotal gastrectomy, biliary obstruction, and pancreatic disease.
All the subjects followed up regularly by PCR or antigen test for COVID-19 infection and completed seven electronic questionnaires during this trial. The information collected included the name, age, height, weight, vaccine, antigen or PCR test results, the time of positive PCR test or antigen test, the time of positive to negative PCR test or antigen test, the extent and duration of COVID-19 symptoms (fever, cough, sore throat, headache, joint pain, diarrhea, and vomiting), concomitant diseases, and smoking history of subjects.
Randomization and study interventions
The subjects were randomly allocated into different groups by local investigators using a randomization envelope, which concealed the allocation sequence. The random sequence was generated with the SAS 9.4 procedure using a size block of 4, and the ratio between the two groups was 1:1.
Subjects in the vitamin D2 group were given 200,000 IU containing 5 mg of calciferol orally on the first day and the fourteenth day after enrollment, while the other group subjects received no intervention during the whole trial.
Outcome assessment
The primary outcome was the incidence of COVID-19 infection as detected by laboratory PCR or antigen test within one month. The secondary outcomes were the duration of a positive to negative PCR or antigen test after COVID-19 infection, the duration of fever, cough, runny nose, stuffy nose, sore throat, headache, fatigue, muscle aches, joint aches, loss of taste or smell, vomiting, diarrhea and other symptoms after COVID-19 infection, the proportion of subjects whose COVID-19 infection symptoms were confirmed by PCR or antigen testing for more than 7 days, the proportion of subjects who tested positive by PCR or antigen test but had no symptoms of COVID-19, the proportion of subjects with mild, moderate and severe COVID-19 symptoms, and the change in 25(OH)D concentration.
At baseline, serum 25(OH)D, CRP, complement component C1q and inflammatory cytokines (IL-1β, IFN-α, IFN-γ, TNF-α, MCP-1, IL-6, IL-8, IL-10, IL-12, IL-17A, IL-18, IL-23, and IL-33) were measured in a group of 214 subjects, and subsequently, on the thirtieth day of the study, the subjects completed the second laboratory assessment. All the blood samples were analyzed at the Peking University Third Hospital. Seven follow-up visits were conducted during the trial.
Blinding
This was a open-label controlled trial, the blinding method was not applicable.
Sample size calculation and power
The primary objective of the study was to compare the incidences of COVID-19 infection in the two groups. According to our preliminary study, the expected incidence of COVID-19 infection was 60.0% in the treatment group and 70.0% in the control group. Given an α of 0.05 and β of 0.2 (i.e., statistical power of 80%), a total of 103 subjects per group (total sample size: 206 subjects) was needed. Assuming a dropout rate of 5%, 218 patients were required for screening to provide an adequate sample size.
Statistical analysis
The statistician who conducted the analysis was blinded to group allocation. Primary and secondary outcomes were analyzed according to an intention-to-treat (ITT) principle in the ITT dataset. The per-protocol (PP) sets were used for sensitivity analysis. The safety analysis sets included all enrolled subjects who received treatment and were evaluated for safety. Primary analyses were conducted using multiple imputation methods for missing observations based on the baseline, post-intervention and follow-up, assuming that missing data were missing at random.
For the primary outcome and categorical secondary outcomes, the chi-square test or Fisher’s exact test was used to detect the differences between groups. The Kaplan–Meier survival curve and log-rank test were used to analyze the primary outcome as supplements. The generalized linear mixed model (GLMM) was used to control the baseline imbalance and central effect, and the different centers were set as fixed effects.
For continuous secondary outcomes, normally distributed data were presented as the means with standard deviations and compared between groups using the independent t test, while non-normally distributed variables were presented as medians with interquartile ranges (IQRs) and compared using the Mann–Whitney U test. For secondary outcomes and subgroup analyses, the analyses were performed as exploratory tests.
All the statistical analyses were conducted using Statistical Product and Service Solutions 26.0 (SPSS 26.0, IBM, Armonk, NY, USA, http://www.ibm.com/spss) and R 4.2.2 software (http://www.r-project.org). The statistical significance was defined as P < 0.05 with two-sided testing.
Results
Baseline characteristics and follow-up
Of a total of 214 subjects who signed informed consent forms, 99 subjects were randomly assigned to receive vitamin D2, 103 subjects were assigned to the non-intervention, and 193 subjects completed the trial. The follow-up visit occurred one month later. A total of 4.5% of subjects were lost to follow-up, and the loss rate of the vitamin D2 group was lower than that of the non-intervention group (1% in the vitamin D2 group vs. 7.6% in the no intervention group). All the subjects in the vitamin D2 group received 200,000 IU vitamin D2 fortnightly. The subjects in the non-intervention group who took additional vitamin D2 or vitamin D3 were recorded using electronic questionnaires during the trial. Efficacy analyses were performed on the ITT population (202 subjects). The flow chart for this trial is shown in Fig. 1.
Flow of subjects in the study on the role of high-dose vitamin D2 for preventing COVID-19 in health care workers, including screening, randomization, and follow-up during the whole trial.
The baseline demographic and clinical characteristics were similar in the two groups, with 17.3% of the subjects having 25(OH)D concentrations greater than 20 ng/mL and only 4.9% of the subjects having adequate 25(OH)D concentrations (greater than 30 ng/mL). The median age was 36.5 years old in this population. There were few concomitant diseases in this population. The results are presented in Table 1. A total of 1518 electronic questionnaires were received. The questions on the self-assessment of COVID-19 symptoms are shown in See supplementary file1 S1 Table 1.
Primary outcome
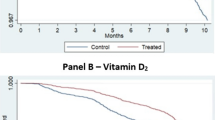
Since there were differences between the two groups in the ages and BMIs of the subjects at baseline, we adopted GLMM to regulate the age and BMI as covariables. A total of 104 subjects tested positive for COVID-19 during the follow-up period. The infection rates are shown in See supplementary file1 S1 Table 2. Vitamin D2 was associated with a COVID-19 infection rate of 50.5% (95% CI, 40.7 to 60.4) compared with 52.4% (95% CI, 42.8 to 62.1) in the no intervention group according to the ITT dataset (P = 0.785). The primary outcome was subgroup-analyzed by age, sex and 25(OH)D level, and no statistical significance was found for the COVID-19 infection rate. The COVID-19 infection rate was very similar in the ITT and PP subjects. The Kaplan–Meier survival curve of COVID-19 infection is shown in Fig. 2.
The Kaplan–Meier survival curve of COVID-19 infection between the vitamin D2 group and the non-intervention group (a) intention-to-treat (ITT) dataset and (b) per-protocol (PP) dataset; the shadow of the above curve represents the confidence interval).
Secondary outcomes
No statistically significant effect between the two groups was observed on the proportion of subjects whose COVID-19 infection symptoms were confirmed by PCR or antigen testing for more than 7 days (17.1% in the vitamin D2 group vs. 16.5% in the non-intervention group). There were no significant differences between the vitamin D2 group and the non-intervention group in the duration of COVID-19 symptoms. The rates of asymptomatic, mild, moderate and severe symptoms (fever, cough, runny nose, stuffy nose, sore throat, headache, fatigue, muscle aches, joint aches, loss of taste or smell, vomiting, diarrhea and other symptoms) of COVID-19 were not significantly different between the two groups. The duration of PCR or antigen testing from positive to negative after COVID-19 infection was similar over the two groups (6.6 days in the vitamin D2 group vs. 7.4 days in the non-intervention group). No subjects required hospitalization or died in either group. The results are shown in Table 2.
The level of mean serum 25(OH)D was significantly increased from baseline to after two doses of vitamin D2 administration (from 14.1 to 31.1 ng/mL, P < 0.001) compared with the non-intervention group (from 14.4 to 15.3 ng/mL). After receiving 200,000 IU vitamin D2, 87 of 99 subjects (87.9%) had serum 25(OH)D levels greater than 20 ng/mL in the vitamin D2 group, compared with 21 of 102 subjects (20.6%) in the non-intervention group. The result is shown in Fig. 3.
Serum 25(OH)D concentration in the vitamin D2 group and non-intervention group. Serum 25(OH)D concentrations were measured at baseline (first) and on the thirtieth day of the trial (second).
There was no significant difference in the levels of inflammatory cytokines at baseline and on the thirtieth day of the trial in the vitamin D2 and non-intervention groups. The result is shown in Supplementary Fig. 2.
Subgroup analysis
According to the second level of vitamin D, subjects with serum 25(OH)D group with less than 20 ng/mL (group A, n = 85) had a positive infection rate of 58.9% , the serum 25(OH)D group between 20 and 30 ng/mL (group B, n = 44) had a positive infection rate of 52.4%, and the serum 25(OH)D greater than 30 ng/mL (group C, n = 44) had a positive infection rate of 44.6%. The results of the survival analysis are shown in Supplementary Fig. 1. There was a 14.3% difference in positive infection rates between the vitamin D-adequate and deficient groups. During the first ten days of the trial, the infection rates of subjects in the three groups were rapid. However, group A and B tended to be stable after the tenth day of the trial. The infection rate of subjects with 25(OH)D greater than 20 ng/mL was still more serious, and the infection rate was still increasing on the twentieth day during the trial period. The subgroup analysis showed that the 25(OH)D level was not significantly associated with COVID-19 symptoms, including the period of symptoms, the number of symptoms and the extent of the symptoms.
Adverse events
The administration of 200,000 IU vitamin D2 was well tolerated by the subjects, and no severe adverse events occurred during the trial, with only 1 subject who experienced nausea, 3 subjects who vomited and 1 subject who was constipated after vitamin D2 administration. During the follow-up visit, the nausea and vomiting symptoms in the above subjects occurred after the emergence of COVID-19 symptoms and disappeared with the negative PCR or antigen test. Therefore, the above nausea and vomiting were not considered to be adverse events caused by vitamin D. No subjects stopped the trial because of adverse events in the vitamin D2 group.
Discussion
Here, we found that 200,000 IU vitamin D2 administration did not significantly prevent COVID-19 infection or improve COVID-19 symptoms among health care workers. Some benefits were also found that the positive infection rate of VD-sufficient (less than 20 ng/mL) subjects was 14.3% lower than that of VD-deficient (greater than 30 ng/mL) subjects.
Vitamin D deficiency has become an "epidemic" and is common in all age groups17. In 2011, the US Endocrine Society together with other scientific societies upgraded their standards to indicate that thresholds of vitamin D and serum 25(OH)D levels of less than 20 ng/ml were defined as vitamin D deficiency, 20 ng/mL to 30 ng/mL were insufficient, and greater than 30 ng/ml was adequate18. Our study showed that the total proportion of serum 25(OH)D deficiency (82.7%) and insufficiency (12.4%) in 202 subjects was 92.0%, the ratio of vitamin D sufficiency was only 4.9%, and severe vitamin D deficiency with serum 25(OH)D of less than 10 ng/ml accounted for 13.9%. The heavy workload of the majority of health care workers in first-class hospitals, especially the high frequency of night work and long-term indoor work, leads to a general lack of exercise and sunlight exposure, and night work also changes their normal eating habits, resulting in insufficient vitamin D synthesis. Current studies have found that vitamin D deficiency is closely related to bone diseases19, immune diseases20, cardiovascular diseases21, tumors22, diabetes23, obesity24, chronic kidney disease25 and others. The benefit-risk ratio of vitamin D supplementation is higher, and vitamin D safety is better. Vitamin D supplementation at 100,000 IU/month did not increase kidney stone risk or serum calcium in 5110 participants26. Therefore, it is necessary to increase the level of active vitamin D through sunshine, dietary and drug supplementation.
As an immunomodulator, vitamin D can inhibit the excessive production of reactive oxygen species in vivo, increase the glutathione level in cells, and thus inhibit the expression of the NF-kB and p38 MAPK kinases to inhibit the expression of the proinflammatory factors TNF-α, IL-6, MCP-1, IL-12, IL-18, IL-33, and others27. In this study, the levels of inflammatory cytokines, including IL-1β, IFN-α, IFN-γ, TNF-α, MCP-1, IL-6, IL-8, IL-10, IL-12, IL-17A, IL-18, IL-23, and IL-33, were measured at baseline and on the thirtieth day of the trial in the vitamin D2 and non-intervention groups. The baseline inflammatory cytokines are shown in See supplementary file1 S1 Table 3. The second level of inflammatory cytokines is shown in Supplementary Fig. 2. The results did not show that 200,000 IU vitamin D2 reduced inflammatory cytokines. The half-life of most inflammatory cytokines is short. When inflammation disappears, inflammatory cytokines quickly return to normal and maintain the homeostasis of the body. Our study measured levels of inflammatory cytokines at baseline and one month after enrollment, whereas the majority of positive subjects in this study developed infections within 15 days of the trial. Therefore, we believe that we may have missed the peak in inflammatory cytokines. We ultimately failed to see that high doses of vitamin D reduced inflammation.
Limitations of the study
There were some limitations in this study. Based on the previous literature, we calculated the sample size in this trial using the assumption of a 10% positive infection rate in the vitamin D2 group and the non-intervention group, whereas this conclusion was not reached in our trial. In addition, there was a large difference in the ratio of males to females in our study, primarily because the enrolled subjects were mostly nursing staff, which resulted in an imbalance in the gender ratio. Lastly, the time interval between the two blood samples was long, and the change in inflammatory cytokines in the two groups of subjects could not be observed during the trial.
Conclusions
Our findings indicated that the administration of vitamin D2 did not significantly reduce the rate of COVID-19, and it was not related to COVID-19 symptoms in health care workers. Adequate vitamin D had a tendency to prevent COVID-19. Vitamin D deficiency (82.7%) was widespread among health care workers, and the percentage of vitamin D sufficiency was only 4.9% at baseline, and it is necessary for workers to take vitamin D supplements.
Data availability
The data underlying this article will be shared upon reasonable request to the corresponding author.
Change history
31 December 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41598-024-83804-7
References
Dhama, K. et al. Global emerging Omicron variant of SARS-CoV-2: Impacts, challenges and strategies. J. Infect. Public Health. 16, 4–14 (2023).
Feikin, D. R. et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: Results of a systematic review and meta-regression. Lancet. 399, 924–944 (2022).
Cegolon, L., Ronchese, F., Ricci, F., Negro, C. & Larese-Filon, F. SARS-CoV-2 infection in health care workers of Trieste (North-Eastern Italy), 1 October 2020–7 February 2022: Occupational risk and the impact of the omicron variant. Viruses 14(8), 1663. https://doi.org/10.3390/v14081663 (2022).
Wong, S. C. et al. Infection of healthcare workers despite a high vaccination rate during the fifth wave of COVID-19 due to Omicron variant in Hong Kong. Infect. Prev. Pract. 5, 100261 (2023).
Eftekhar Ardebili, M. et al. Healthcare providers experience of working during the COVID-19 pandemic: A qualitative study. Am. J. Infect. Control. 49, 547–554 (2021).
Goltzman, D. Functions of vitamin D in bone. Histochem. Cell Biol. 149, 305–312 (2018).
Khazai, N., Judd, S. E. & Tangpricha, V. Calcium and vitamin D: Skeletal and extraskeletal health. Curr. Rheumatol. Rep. 10, 110–117 (2008).
Campbell, G. R. & Spector, S. A. Autophagy induction by vitamin D inhibits both Mycobacterium tuberculosis and human immunodeficiency virus type 1. Autophagy. 8, 1523–1525 (2012).
L Bishop, E., Ismailova, A., Dimeloe, S., Hewison, M. & White, J. H. Vitamin D and immune regulation: Antibacterial, antiviral, anti-inflammatory. JBMR Plus. 5, e10405 (2021).
Klug-Micu, G. M. et al. CD40 ligand and interferon-γ induce an antimicrobial response against Mycobacterium tuberculosis in human monocytes. Immunology. 139, 121–128 (2013).
Chambers, E. S. & Hawrylowicz, C. M. The impact of vitamin D on regulatory T cells. Curr. Allergy Asthma Rep. 11, 29–36 (2011).
Baeke, F., Takiishi, T., Korf, H., Gysemans, C. & Mathieu, C. Vitamin D: Modulator of the immune system. Curr. Opin. Pharmacol. 10, 482–496 (2010).
Entrenas Castillo, M. et al. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 203, 105751 (2020).
Seal, K. H. et al. Association of vitamin D status and COVID-19-related hospitalization and mortality. J. Gen. Intern. Med. 37, 853–861 (2022).
Renieris, G. et al. Association of vitamin D with severity and outcome of COVID-19: Clinical and experimental evidence. J. Innate Immun. 16, 1–11 (2024).
Murai, I. H. et al. Effect of a single high dose of vitamin D3 on hospital length of stay in patients with moderate to severe COVID-19: A randomized clinical trial. JAMA. 325, 1053–1060 (2021).
Lips, P., de Jongh, R. T. & van Schoor, N. M. Trends in vitamin D status around the world. JBMR Plus. 5, e10585 (2021).
Holick, M. F. et al. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96, 1911–1930 (2011).
Lips, P. & van Schoor, N. M. The effect of vitamin D on bone and osteoporosis. Best Pract. Res. Clin. Endocrinol. Metab. 25, 585–591 (2011).
Ismailova, A. & White, J. H. Vitamin D, infections and immunity. Rev. Endocr. Metab. Disord. 23, 265–277 (2022).
de la Guía-Galipienso, F. et al. Vitamin D and cardiovascular health. Clin. Nutr. 40, 2946–2957 (2021).
Brunner, R. L. et al. The effect of calcium plus vitamin D on risk for invasive cancer: Results of the Women’s Health Initiative (WHI) calcium plus vitamin D randomized clinical trial. Nutr. Cancer. 63, 827–841 (2011).
Mozaffari-Khosravi, H., Hosseinzadeh-Shamsi-Anar, M., Salami, M. A., Hadinedoushan, H. & Mozayan, M. R. Effects of a single post-partum injection of a high dose of vitamin D on glucose tolerance and insulin resistance in mothers with first-time gestational diabetes mellitus. Diabet. Med. 29, 36–42 (2012).
Bassatne, A., Chakhtoura, M., Saad, R. & Fuleihan, G. E. Vitamin D supplementation in obesity and during weight loss: A review of randomized controlled trials. Metabolism. 92, 193–205 (2019).
Jean, G., Souberbielle, J. C. & Chazot, C. Vitamin D in chronic kidney disease and dialysis patients. Nutrients. 9, 328 (2017).
Malihi, Z. et al. Monthly high-dose vitamin D supplementation does not increase kidney stone risk or serum calcium: Results from a randomized controlled trial. Am. J. Clin. Nutr. 109, 1578–1587 (2019).
Mohan, M., Cherian, J. J. & Sharma, A. Exploring links between vitamin D deficiency and COVID-19. PLoS Pathog. 16, e1008874 (2020).
Acknowledgements
The authors would like to express sincere thanks to all patients who participated in the study. We would like to acknowledge Jiangxi Gannan Haixin Pharmaceutical Co., Ltd for supporting this research project.
Funding
This work was supported by the National Key Research and Development Program (Grant number. 2020YFC2009004, 2021YFC2501700) and the National Natural Science Foundation of China (Grant number. 82272554).
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript. Chunli Song, Baohua Li, Haiyan Li, Yahong Chen, Dongyang Liu, Yuping Yang, Yang Lv and Fuchun Zhang designed this research. Chunli Song directed the whole study. Drafting of the manuscript: Huan Wang and Liyuan Tao. Statistical analysis: Liyuan Tao, Xiaoxiao Wang and Huan Wang. Critical revision of the manuscript for important intellectual content: All authors. Acquisition, analysis, or interpretation of data: Huan Wang, Wanqiong Yuan, Hao Liu, Jie Huang. Obtained funding: Chunli Song. Administrative, technical, or material support: Liyan Cui, Lixiang Xue, Tiancheng Wang, Yanfang Jiang, Na Liu, Yanfang Li, Yuling Gao. Supervision: Chunli Song, Baohua Li amd Haiyan Li.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in Reference 3. It now reads: 3. Viruses. 2022 jul 28, 14(8): 1663. doi: 10.3390/v14081663.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, H., Tao, L., Cui, L. et al. Randomized trial of influence of vitamin D on the prevention and improvement of symptomatic COVID-19. Sci Rep 14, 20519 (2024). https://doi.org/10.1038/s41598-024-66267-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-66267-8