Abstract
Obesity arises from an imbalance between energy consumption and energy expenditure, and thyroid hormone levels serve as a determinant of energy expenditure. We conducted experiments at the animal and cellular levels and combined those findings with clinical data to elucidate the role of triiodothyronine (T3) in facilitating the browning of white adipose tissue (WAT) and its underlying mechanism. The results showed (i) the impaired metabolic function of local WAT and the compensatory elevation of systemic thermogenesis in obesity; (ii) T3 treatment of white adipocytes in vitro and local WAT in vivo induced a shift towards a morphologically “brown” phenotype, accompanied by upregulation of mRNA and protein expression of browning-related and mitochondrial function markers, which suggest that T3 intervention promotes the browning of WAT; and (iii) the aforementioned processes could be modulated through inhibition of the PI3K/AKT signalling pathway; however, whether T3 affects the PI3K/AKT signalling pathway by affecting insulin signalling remains to be studied and clarified. The results of our study indicate that T3 treatment promotes browning of WAT through inhibition of the PI3K/AKT signalling pathway; these findings offer novel perspectives regarding the potential of localised therapies for addressing WAT volume in individuals with obesity.
Similar content being viewed by others
Introduction
Obesity is a chronic disease caused by an imbalance between energy intake and energy expenditur1,2. In recent decades, the global prevalence of obesity has increased to pandemic proportions3. Obesity, along with its associated complications, contributes significantly to premature mortality4,5,6,7,8. Nevertheless, there remains a dearth of targeted pharmacological interventions for obesity9,10. Individuals with obesity are advised to engage in a combination of dietary modifications, physical exercise, and behavioural therapy11. However, weight reduction achieved through behavioural interventions tends to be modest and short-lived9. Obesity is characterised by the excessive accumulation of adipose tissue (AT). AT is categorised into two distinct types, namely, white adipose tissue (WAT) and brown adipose tissue (BAT), which play contrasting roles in the regulation of energy2. WAT primarily acts as a reservoir for energy in the form of triglycerides2,12. Conversely, BAT is characterised by a high abundance of mitochondria expressing uncoupling protein-1 (UCP-1), a protein crucial for thermogenesis that aids in the maintenance of body temperature by expending energy2,12. Beige fat is a distinct form of AT situated within WAT that possesses functional attributes akin to BAT13. Upon activation, beige fat exhibits elevated expression levels of UCP-1. The process of white fat browning refers to the induction of beige fat development within WAT, thereby augmenting energy expenditure, mitigating deleterious consequences associated with excessive white fat accumulation, and consequently enhancing metabolic wellbeing14,15,16.
Resting energy expenditure (REE) refers to the energy expended by an individual in a nonfasting state and represents the number of calories necessary for the body to sustain its metabolic functions over a 24-h period of inactivity17. In the context of adults, REE constitutes the predominant component of daily energy expenditure, accounting for approximately 60–75%, and serves as a crucial factor in determining overall daily energy expenditure18,19. Consequently, enhancing REE plays a pivotal role in the preservation of healthy body weight. It is well established that thyroid hormone (TH) contributes to the elevation of REE, as evidenced by weight loss in individuals with pronounced hyperthyroidism and weight gain in those with substantial hypothyroidism20. TH serves as a crucial factor in the determination of energy expenditure and REE. TH accomplishes this by governing respiration and thermogenesis, thereby influencing REE and overall energy expenditure2.
Several studies have provided preliminary evidence indicating the potential of TH to stimulate the browning of WAT. However, these studies have primarily relied on observational data obtained from animal experiments. Specifically, mice with hyperthyroidism exhibit an increase in brown fat mass, while mice with hypothyroidism display compensatory white fat browning because of reduced heat production from brown fat21,22. Johann et al. suggested that hyperthyroidism could improve glucose tolerance and induce browning of inguinal white adipose tissue, and decreases adipose tissue glucose and lipid uptake23. Moreover, the administration of thyroid hormone receptor (THR)-specific agonists to ob/ob mice leads to significant browning of subcutaneous WAT, resulting in elevated core body temperature and increased overall energy expenditure24. The classic PI3K/AKT signalling pathway plays an important role in the regulation of lipid metabolism and insulin metabolism homeostasis25. Deficiency of PI3K expression could result in increased lipolysis and browning of WAT and prevent diet-induced obesity, insulin resistance, etc.26. Excessive activation of mTOR, the primary downstream target of PI3K/AKT signalling, a crucial regulator of lipogenesis and glucose metabolism27, also contributes to diseases such as obesity and T2DM28. Several independent studies have provided evidence supporting the promotion of obesity by the PI3K/AKT signalling pathway25. Based on previous studies, the role of TH in promoting the browning of white adipose tissue and whether it is regulated by the PI3K/AKT signalling pathway are questions worth exploring. Drawing upon prior research, the objective of the present study was to examine the role and mechanism of TH, especially triiodothyronine (T3), in facilitating the browning of WAT, to identify novel targets and propose innovative approaches for addressing obesity and associated metabolic disorders.
Results
Systemic thermogenesis was elevated in obese mice, and localised energy expenditure within WAT was suppressed
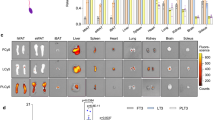
As shown in Fig. 1A, the basal surface temperature of the mice in the HFD group was greater than that of the mice in the ND group in both the dorsal and ventral aspects (p < 0.0001****) (S-Table 3, S-Fig. 5). The serum free triiodothyronine (FT3) (p < 0.0001****) and free thyroxine (FT4) (p = 0.0127*) levels were significantly greater in the mice of the HFD group than in the mice of the ND group (Fig. 1C, S-Table 3). In contrast, the basal surface temperature of the mice in the CR group was lower than that of the mice in the AL group (p < 0.0001****) (Fig. 1B, S-Table 4, S-Fig. 5). The serum FT3 (p = 0.0003***) and FT4 (p = 0.0036**) levels in the mice of the CR group were significantly lower than those in the HFD and ND groups (Fig. 1D, S-Table 4).
The systemic thermogenesis increased while the local white adipose tissue (WAT) energy expenditure was impaired in obese: (A) Infrared thermography of basal surface on ND and HFD mouse housed at 22 ℃; (B) Infrared thermography of basal surface on AL and CR mouse housed at 22 ℃; (C) Comparison of serum FT3 and FT4 levels between ND and HDF group; (D) Comparison of serum FT3 and FT4 levels between AL and CR group; (E) Comparison of FT3 and FT4 levels in WAT and BAT between ND and HDF group; (F) THR and DIO2 protein levels in WAT were measured using Western blot between ND and HDF group; (G) The brown-related proteins such as UCP-1 in WAT were measured using Western blot between ND and HDF group; (H) Comparison of serum FT3 and FT4 levels among people with Normal, Overweight and Obesity; (I) Comparison of FT3 and FT4 levels in WAT between people with Normal and people with Obesity; (J) THR and DIO2 protein levels in WAT were measured using Western blot between people with Normal and people with Obesity; (K) The brown-related proteins such as UCP-1 in WAT were measured using Western blot between people with Normal and people with Obesity; (ND: normal diet; HFD: high-fat diet; AL: ad libitum diet; CR: calorie restriction diet; Normal: 18 kg/m2 ≤ BMI < 24 kg/m2; Overweight: BMI ≥ 24 kg/m2; Obesity: BMI ≥ 30 kg/m2) The height of each bar represents the mean ± SD for the group. (Animal: *p < 0.05, **p < 0.01, ***p < 0.001 vs ND group, n = 4; Clinical: *p < 0.05 vs people with Normal, n = 3).
The tissue contents of T3 could reflect the role of T3 in different tissues29. We examined the FT3 and FT4 levels in AT. The results showed that the FT3 (p = 0.0224*) level in the WAT of the mice in the HFD group was significantly lower than that in the ND group, but there was no statistically significant difference in the FT4 level between groups (Fig. 1E, S-Table 3).
DIO2 is responsible for the conversion of TH into biologically active T3 in local tissues, thereby regulating the concentration of TH by determining the tissue content of T330. The thermogenesis of TH is mediated by THRβ. Besides, Johann et al. found that browning by TH requires THRβ23,31. The Western blot (WB) results demonstrated a significant decrease in the expression of DIO2 (p = 0.0002***) and THRβ (p = 0.0097**) in the WAT of the HFD group mice compared to that of the ND group mice (Fig. 1F), which suggests that the conversion of TH into T3 may be impaired in the WAT of the HFD group mice.
UCP-1, a marker of BAT, possesses a heat-generating function that impedes fat accumulation through the augmentation of energy expenditure32. The PPAR family of proteins is implicated in lipid metabolism and the maintenance of metabolic homeostasis. PPARγ, a prominent regulator of adipose differentiation, stimulates the expression of UCP-1 to facilitate thermogenesis33,34. PPARα, another member of the PPAR protein family, promotes lipolysis and fatty acid oxidation34. Similarly, CEBPβ, CIDEA, and FNDC5 are involved in WAT browning and significantly influence the regulation of energy expenditure. WB results revealed significant downregulation of expression of browning-related proteins, such as UCP-1 (p = 0.0039**), PPARγ (p = 0.0045**), CEBPβ (p = 0.0003***), CIDEA (p = 0.0232*), and FNDC5 (p = 0.0109*), in the WAT of the HFD group mice compared to that of the ND group mice (Fig. 1G), which indicates that the WAT browning processes of the HFD group mice were suppressed, resulting in the inhibition of local energy expenditure and subsequent worsening of obesity.
Localised energy expenditure within WAT was suppressed in the people with obesity
A cross-sectional study indicated a slight elevation in serum FT3 levels between overweight individuals and normal individuals, although this difference did not reach statistical significance. Conversely, obese individuals exhibited significantly lower serum FT3 levels than did normal individuals (p = 0.0175*) (Fig. 1H, S-Table 5).
Five normal and 5 obese individuals whose WAT was collected were randomly selected to measure the WAT FT3 and FT4 levels and protein expression. As shown in Fig. 1I, the FT3 (p = 0.0129*) level in the WAT of the people with obesity was significantly lower than that in the WAT of the people with normal (S-Table 6). The expression of DIO2 (p = 0.0336*) in the WAT of the people with obesity was significantly lower than that in the WAT of the people with normal (Fig. 1J). Compared with that in the people with normal, the expression of UCP-1 (p = 0.0320*), PPARγ (p = 0.0368*) and CEBPβ (p = 0.0103*) was significantly downregulated in the people with obesity (Fig. 1K).
T3 promoted browning of white adipocytes
Following the successful construction of the mature white adipocyte model derived from mouse, a physiological concentration of 10 pmol/L T3 in the WAT was used as the control, whereas the intervention groups underwent tenfold gradient increases in the physiological concentrations of T3, resulting in concentrations of T3 of 1 × 102 pmol/L, 1 × 103 pmol/L, 1 × 104 pmol/L, 1 × 105 pmol/L and 1 × 106 pmol/L for 72 h. Our findings revealed significant upregulation of gene expression related to browning and mitochondrial energy production in white adipocytes after 72 h of T3 treatment (Fig. 2A). In particular, a concentration of 100 pmol/L led to the greatest number of browning-related genes with significantly upregulated expression. Compared with those in the control group, the mRNA expression levels of brown genes such as UCP-1 (p = 0.0052**), PPARγ (p = 0.0144*), PPARα (p = 0.0423*), CEBPβ (p = 0.0003***), CIDEA (p = 0.0164*), FNDC5 (p = 0.0009***), Tmem26 (p = 0.0166*), Tbx1 (p = 0.0012**) and Ebf3 (p = 0.0269*) were significantly increased in white adipocytes after T3 intervention at 100 pmol/L. Consistently, the mRNA expression of genes related to mitochondrial productivity, such as PGC1α (p = 0.0173*), Cox7a1 (p = 0.0124*) and Cox8b (p = 0.0231*), was also significantly increased after T3 intervention at 100 pmol/l (Fig. 2B). Consequently, we selected 100 pmol/l as the T3 intervention concentration for the subsequent phase of our experiment.
T3 promoted browning of white adipocytes may through inhibiting of the PI3K/AKT signalling pathways and PI3K/AKT signaling pathway was activated in local WAT of obese: (A) The brown-related mRNA expression after 72 h of intervention in white adipocytes using different concentrations of T3; (B) The brown-related mRNA expression after intervention in white adipocytes using 100 pmol/l of T3 as the intervening concentration; (C) The expression of THR and DIO2 protein after 100 pmol/l of T3 as intervention concentration in white adipocytes; (D) The expression level of brown-related proteins after 100 pmol/l of T3 as intervention concentration in white adipocytes; (E) The morphology of adipocytes highlighted on an oil red O stain (× 100 and × 200); (F) The protein expression of UCP-1 and CIDEA were detected by immunofluorescence after 100 pmol/l of T3 as intervention concentration in white adipocytes; (G) Volcano diagrams of differential mRNA. The abscissa and ordinate represent X = log2 (fold changes) and Y = − log10 (P-value), the red dots represent up-regulated differential genes, the blue dots represent down-regulated differential genes, and the black dots represent genes with no significant difference; (H) The clustering heatmaps of the differential mRNA, A1, A2, A3: Control group, B1, B2, B3: 100 pmol/l of T3 treated white adipocytes as intervention group; (I) GO analysis; (J) KEGG pathway analysis of the target gene; (K) The qRT-PCR validated mRNA expression of PI3K/AKT signalling pathway related genes in the high-throughput mRNA sequencing; (L) The Western blot validated relative expression of PI3K/AKT signalling pathway related proteins in the high-throughput mRNA sequencing; (M) The expression of PI3K/AKT signalling pathway related proteins in WAT of ND and HFD groups; (N) The expression of PI3K/AKT signalling pathway related mRNA in WAT among people with Normal, Overweight and Obesity. (ND: normal diet; HFD: high-fat diet; Normal: 18 kg/m2 ≤ BMI < 24 kg/m2; Overweight: BMI ≥ 24 kg/m2; Obesity: BMI ≥ 30 kg/m2). The height of each bar represents the mean ± SD for the group. (Cell culture: *p < 0.05, **p < 0.01, ***p < 0.001 vs Control, n = 3; Animal: *p < 0.05, **p < 0.01 vs ND group, n = 4; Clinical: *p < 0.05 vs people with Normal, n = 5).
Furthermore, WB revealed significant downregulation of DIO2 expression (p = 0.0312*) and significant upregulation of UCP-1 (p = 0.0102*), PPARγ (p = 0.0039**), CEBPβ (p = 0.0255*), CIDEA (p = 0.0131*) and FNDC5 (p = 0.0171*) protein expression in white adipocytes following T3 intervention (Fig. 2C,D). Moreover, as shown in Fig. 2F, immunofluorescence staining revealed an intensified fluorescence signal for UCP-1 and CIDEA following T3 intervention, which indicated an increase in their protein expression levels. Additionally, the morphologies of adipocytes were evaluated using oil red O staining. The lipid droplet content within adipocytes decreased, and the number of lumens increased after T3 intervention (Fig. 2E).
Browning of white adipocytes induced by T3 may be facilitated through inhibition of the PI3K/AKT signalling pathway
High-throughput mRNA sequencing was performed on white adipocytes from the control and 100 pmol/L T3 intervention groups to investigate the underlying mechanism by which T3 facilitates the browning process of white adipocytes. The volcano map, heatmap, GO analysis and KEGG pathway analysis results35,36,37 are shown in Fig. 2G–J, and the details are described in the Supplementary Appendix. Figure 2J shows the top 20 pathway terms enriched in the DEGs between the two groups. The PI3K/AKT pathway was the most significantly enriched pathway in white adipocytes following T3 intervention compared with that in the control group, and this pathway was inhibited (NES = − 1.0308), suggesting that the promotion of white adipocyte browning by T3 may be mediated through the inhibition of the PI3K/AKT pathway.
The PI3K/AKT signalling pathway is closely linked to the insulin signalling pathway within the endocrine system25, and the BAT marker UCP-1 is also affected by the insulin signalling pathway. Furthermore, the PI3K/AKT and PPAR signalling pathways play crucial roles in the regulation of adipocyte differentiation and lipid metabolism. The findings of this study indicate that T3 may promote white adipocyte browning by inhibiting the PI3K/AKT signalling pathway.
The mRNA expression levels of IRS-1 (p = 0.0210*), PI3K (p = 0.0228*), AKT (p = 0.0034**) and mTOR (p = 0.0056**) were significantly lower in the T3 intervention group than in the control group, which was consistent with the high-throughput mRNA sequencing results (Fig. 2K). The relative expression levels of p-IRS-1/IRS-1 (p = 0.0088**), p-PI3K/PI3K (p = 0.0127*), p-AKT/AKT (p = 0.0040**), and p-mTOR/mTOR (p = 0.0405*) significantly decreased after T3 intervention, which was consistent with the changes in the mRNA expression levels (Fig. 2L).
The WB results revealed elevated expression levels of p-IRS-1/IRS-1 (p = 0.0234*), p-PI3K/PI3K (p = 0.0016**), p-AKT/AKT (p = 0.0047**), and p-mTOR/mTOR (p = 0.0006***) in the WAT of HFD-fed mice compared to that of ND-fed mice, suggesting that activation of this pathway might be involved in the development of obesity (Fig. 2M).
In the clinical study, significantly higher mRNA expression levels of PI3K (P = 0.0299*), AKT (P = 0.0490*), and mTOR (P = 0.0464*) were observed in the people with obesity than in people with normal, which was consistent with alterations observed in the model animals (Fig. 2N).
The promotion of white adipocyte browning by T3 may be mediated through the PI3K/AKT pathway
We used an IRS-1/PI3K/AKT agonist (licarin B) in conjunction with T3 to treat WAT. Regarding the role of T3 in promoting the browning of white adipocytes, the mRNA expression levels of UCP-1 (p = 0.0004***, p < 0.0001****), PPARγ (p = 0.0006***, p = 0.0005***), PPARα (p = 0.0109*, p = 0.0117*), CEBPβ (p = 0.0105*, p = 0.0037**), CIDEA (p = 0.0027**, p = 0.0134*) and PRDM16 (p = 0.0137*, p = 0.0481*) were significantly decreased after T3 combined with PI3K/AKT agonist intervention, regardless of whether they was compared with the control or T3 group (Fig. 3A). Additionally, the mRNA expression levels of Tbx1 (p = 0.0280*), Ebf3 (p = 0.0053**), Cox7a1 (p = 0.0021**) and Cox8b (p = 0.0043**) were also significantly downregulated after T3 combined with PI3K/AKT agonist intervention compared with those in the T3 group (Fig. 3A). The protein expression levels of UCP-1 (p = 0.0043**, p < 0.0001****) and CIDEA (p = 0.0013**, p < 0.0001****) were significantly downregulated after T3 combined with PI3K/AKT agonist intervention, regardless of whether they were compared with those in the control or T3 group (Fig. 3B).
T3 promoted browning of white adipocytes through inhibiting of the PI3K/AKT signalling pathways : (A) The brown-related mRNA expression after intervention in white adipocytes using different intervene: Control, T3, Agonist and T3 + Agonist; (B) Western blot validated the expression levels of UCP-1 and CIDEA proteins in white adipocytes after interventions with Control, T3, Agonist and T3 + Agonist; (C) The mRNA expression of PI3K/AKT pathway related genes after intervention in white adipocytes using different intervene: Control, T3, Agonist and T3 + Agonist; (D) Western blot validated the expression levels of PI3K/AKT pathway related proteins in white adipocytes after interventions with Control, T3, Agonist and T3 + Agonist; (E) The mRNA expression of brown-related genes after insulin intervention in white adipocytes in blank and insulin groups; (F) Western blot validated the expression levels of UCP-1 and CIDEA proteins in white adipocytes after interventions with insulin; (G) The mRNA expression of PI3K/AKT pathway related genes after insulin intervention in white adipocytes in blank and insulin groups; (H) Western blot validated the expression levels of PI3K/AKT pathway related proteins in white adipocytes after interventions with insulin; (I) The mRNA expression of UCP-1 and PI3K/AKT pathway related genes after intervention in white adipocytes using different intervene: Control, T3, Insulin and T3 + Insulin; (J) Western blot validated the expression of UCP-1 and CIDEA proteins in white adipocytes after interventions with Control, T3, Insulin and T3 + Insulin; (K) Western blot validated the expression levels of PI3K/AKT pathway related proteins in white adipocytes after interventions with Control, T3, Insulin and T3 + Insulin. The height of each bar represents the mean ± SD for the group. (A–D: *p < 0.05, **p < 0.01, ***p < 0.001,****p < 0.0001 vs Control, #p < 0.05, ##p < 0.01, ###p < 0.001, ####p < 0.0001 vs T3-treated, n = 3; E–H: *p < 0.05, **p < 0.01, ***p < 0.001 vs Blank, n = 3; I–K: **p < 0.01 vs Control, #p < 0.05, ##p < 0.01, ####p < 0.0001 vs T3-treated, n = 3).
As shown in Fig. 3C, the mRNA expression levels of AKT (p = 0.0086**, p = 0.0016**) and mTOR (p = 0.0016**, p = 0.0005***) were notably upregulated after treatment with T3 combined with the PI3K/AKT agonist, regardless of the treatment comparison (control or T3). Compared with the T3 group, T3 combined with the PI3K/AKT agonist significantly increased the relative expression levels of p-IRS-1/IRS-1 (p = 0.0005***), p-PI3K/PI3K (p = 0.0044**), p-AKT/AKT (p < 0.0001****), and p-mTOR/mTOR (p < 0.0001****) (Fig. 3D), which aligns with the observed mRNA expression levels. This indicates that the inhibitory effects of T3 on this pathway could be counteracted through the use of a PI3K/AKT agonist. The above data suggest that the use of PI3K/AKT agonists could inhibit T3-promoted browning of WAT and that T3-promoted browning of WAT may be mediated through the PI3K/AKT pathway.
Because IRS-1 serves as an upstream marker gene for PI3K/AKT signalling activation, we chose insulin as another agonist to intervene in WAT. Initially, we treated WAT with insulin and monitored alterations in the expression of PI3K/AKT pathway components, as well as relevant indices associated with browning of WAT. The mRNA expression levels of UCP-1 (p = 0.0099**), CEBPβ (p = 0.0049**) and Cox8b (p = 0.0004***) were downregulated after insulin intervention (Fig. 3E), and the protein expression levels of UCP-1 (p = 0.0092**) and CIDEA (p = 0.0096**) were also downregulated (Fig. 3F). Furthermore, the mRNA expression levels of IRS-1 (p = 0.0408*) and PI3K (p = 0.0275*) in WAT were upregulated after insulin intervention (Fig. 3G). Consistently, the relative protein expression levels of p-IRS-1/IRS-1 (p = 0.0001***) and p-PI3K/PI3K (p = 0.0266*) were also significantly increased, indicating the activation of IRS-1 and PI3K (Fig. 3H). This outcome implies that insulin potentially facilitates the reversal of white adipocyte browning.
Subsequently, we used insulin combined with T3 to treat WAT. The results showed that the mRNA expression levels of IRS-1 (p = 0.0115*), AKT (p = 0.0045**) and mTOR (p = 0.0110*) and the relative protein expression levels of p-IRS-1/IRS-1 (p = 0.0115*), p-PI3K/PI3K (p < 0.0001****) and p-AKT/AKT (p = 0.0143*) were significantly upregulated in cells treated with T3 combined with insulin compared to those treated with T3 (Fig. 3I,K), which suggests that the use of insulin may counteract the inhibitory effect of T3 on the PI3K/AKT pathway. However, in relation to the promotion of WAT browning, the impact of T3 in combination with insulin resulted in decreased protein expression levels of UCP-1 (p < 0.0001****) and CIDEA (p = 0.0002***) (Fig. 3J), while there was no notable difference in the mRNA expression of UCP-1 (Fig. 3I). However, whether T3 promotes browning of WAT by inhibiting the PI3K/AKT pathway via the insulin signalling pathway remains to be confirmed in future studies.
Local T3 injection increased food intake in obese mice but did not significantly change body weight
Following the establishment of the obese mouse model, hyaluronic acid (HA) or an HA + T3 complex were administered locally into the subcutaneous AT of the mouse groin. The daily trends in body weights and food intake during the initial intervention cycle (first 7 days) are depicted in Fig. 4A,B. Statistical analysis revealed no significant difference in body weights between the two groups prior to intervention (p = 0.6263) or after the first intervention cycle (p = 0.2980). Notably, following the initiation of the intervention, the food consumption of the mice in the HA + T3 group significantly increased compared to that of the HA group (p < 0.05*). After the initial intervention cycle, the average food intake of the HA + T3 group remained significantly greater than that of the HA group (p < 0.0001****). Consequently, additional investigations categorised the mice into a free diet (FD) group, an FD + T3 group, a control diet (CD) group, and a CD + T3 group based on the regulation of food intake. Notably, the food consumption of the mice in the CD group and the CD + T3 group was maintained at 3.00 ± 0.20 g/day, which was consistent with the average food intake of the mice in the HA group. The daily trends in body weight after establishing the intervention groups are shown in Fig. 4C,D, S-Table 8. Following local T3 intervention, there remained no significant difference in weight after intervene between the FD group and the FD + T3 group (p = 0.1514), no significant difference in intake between the FD group and the CD group (p = 0.1061), and no significant difference in weight after intervene (p = 0.9768). Thus, it appears reasonable to maintain food intake at a normal level (3.00 ± 0.20 g/day), which had no significant impact on T3 intervention results.
T3 combined with control diet (CD) promoted browning of white adipose tissue (WAT) may through inhibiting of the PI3K/AKT signalling pathways: (A) Weight changes of HFD mice in first period (7 days) after HA or HA + T3 intervention; (B) Intake of HFD mice in first period (7 days) after HA or HA + T3 intervention; (C) Weight change of HFD mice in 8–21 days after HA or HA + T3 intervention under the condition of free diet (FD); (D) Weight change of HFD mice in 8–21 days after HA or HA + T3 intervention under the condition of CD; (E) Infrared thermography of basal surface on FD and FD + T3 mouse housed at 22 ℃; (F) Infrared thermography of basal surface on CD and CD + T3 mouse housed at 22 ℃; (G) HE staining of the liver in FD and FD + T3 groups; (H) HE staining of the liver in CD and CD + T3 groups; (I) The brown-related mRNA expression in WAT between FD and FD + T3 group; (J) THR and DIO2 protein levels in WAT between FD and FD + T3 group; (K) The brown-related proteins in WAT between FD and FD + T3 group; (L) HE staining of WAT in FD and FD + T3 groups; (M) The protein expression levels of UCP-1 of WAT were detected by immunofluorescence in FD and FD + T3 groups; (N) The brown-related mRNA expression in WAT between CD and CD + T3 group; (O) THR and DIO2 protein in WAT between CD and CD + T3 group; (P) The brown-related proteins in WAT between CD and CD + T3 group; (Q) HE staining of WAT in CD and CD + T3 groups; (R) The protein expression of UCP-1 of WAT were detected by immunofluorescence in CD and CD + T3 groups; (S) The expression levels of PI3K/AKT signalling pathway related proteins in WAT of CD and CD + T3 groups were measured using Western blot. (FD: free diet; CD: control diet) The height of each bar represents the mean ± SD for the group (B: *p < 0.05, **p < 0.01, ***p < 0.001 vs HA group, n = 10; K: ***p < 0.001 vs FD group, n = 3; J–N,S: *p < 0.05, **p < 0.01, ***p < 0.001 vs CD group, n = 3).
T3 local intervention combined with dietary control improves overall thermogenesis
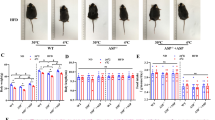
Infrared thermal imaging analysis was conducted on mice in the FD and FD + T3 groups. The results depicted in Fig. 4E indicate that the overall body temperatures of the mice in each group were roughly similar (p = 0.0921) (S-Table 9, S-Fig. 5). However, mice in the FD + T3 group exhibited a significant increase in inguinal body surface temperature compared to those in the FD group (p < 0.0001****). There were no significant differences in subcutaneous fat content or serum levels of FT3 or FT4 between the two groups (S-Table 9).
Insulin overload may promote reverse browning of local white adipose tissue(WAT): (A) Weight changes of HFD mice after Insulin intervention; (B) Infrared thermography of basal surface on PBS and Det Ins mouse housed at 22 ℃; (C) Comparison of serum FT3 and FT4 levels between PBS and Det Ins group; (D) Comparison of FT3 and FT4 levels in WAT between PBS and Det Ins group; (E) The UCP-1 and CIDEA proteins in WAT between PBS and Det Ins group; (F) The expression levels of PI3K/AKT signalling pathway related proteins in WAT of PBS and Det Ins groups. The height of each bar represents the mean ± SD for the group. (*p < 0.05 vs PBS group, n = 3).
Infrared thermal imaging analysis was conducted on mice in the CD and CD + T3 groups. Figure 4F shows a slight decrease in the overall body temperatures of mice in the CD + T3 group compared to that in the CD group (p = 0133*) (S-Table 10, S-Fig. 5). Additionally, the inguinal body surface temperature of mice in the CD + T3 group significantly increased (p < 0.0001****), while the subcutaneous fat content did not significantly differ between groups. Furthermore, the serum FT3 level in the CD + T3 group was significantly decreased compared to that in the CD group (p = 0.0229*), whereas there was no significant difference in the serum FT4 level between the two groups (p = 0.3855) (S-Table 10).
As all interventions were conducted using obese mouse models, the pathological manifestations of nonalcoholic fatty liver disease (NAFLD) were already present in obese mice. HE staining of liver tissues from the FD and CD groups revealed fat infiltration characterised by prominent lipid droplet vacuoles surrounding the majority of hepatocytes and an absence of inflammatory cell infiltration. The histological analysis of liver tissues from the FD + T3 group showed findings comparable to those of the FD group, indicating no significant improvement in the observed pathological changes (Fig. 4G). However, HE staining of liver tissue from mice in the CD + T3 group revealed only a minor portion of liver cells displaying an adipose phenotype and a significant decrease in lipid droplet vacuolation (Fig. 4H). These findings suggest that local T3 intervention in conjunction with dietary control may be effective in ameliorating NAFLD in diet-induced obese mice.
Local T3 intervention combined with dietary control promotes browning of WAT through inhibition of the PI3K/AKT signalling pathway
There was no significant difference in the expression of browning-related mRNAs or proteins in the WAT of mice in the FD group or FD + T3 group (Fig. 4I,K). The protein expression of DIO2 in the WAT of the FD + T3 group was significantly upregulated (p = 0.0479*) (Fig. 4J). HE staining of WAT in the FD + T3 group was comparable to that in the FD group (Fig. 4L). Immunohistochemistry analysis indicated that the UCP-1 protein expression in WAT in both the FD and FD + T3 groups was similar (Fig. 4M).
Compared with those in the CD group, the mRNA expression levels of UCP-1 (p = 0.0322*), PPARγ (p = 0.0003***), CEBP-β (p = 0.0178*) and Ebf3 (p = 0.0168) in the WAT of mice in the CD + T3 group were significantly greater, and the mRNA expression of Cox7a1 (p = 0.0125*), which represents mitochondrial productivity, was also significantly greater (Fig. 4N). Moreover, UCP-1 (p = 0.0164*), PPARγ (p = 0.0408*) and CEBPβ (p = 0.0357*) protein expression was also significantly upregulated in the CD + T3 group compared to the CD group (Fig. 4P). The protein expression of DIO2 in WAT in the CD + T3 group was significantly upregulated compared to that n the CD group (p = 0.0445*), while there was no significant difference in THRβ protein expression between the two groups (Fig. 4O). HE staining of WAT from mice in the CD group revealed a high number and volume of adipocytes with large intracellular lipid droplets. Conversely, mice in the CD + T3 group exhibited a significant reduction in adipocyte volume, smaller cytoplasmic lipid droplets, and the formation of smaller, multilumen adipocytes with a darker colour resembling brown adipocytes. These findings suggest that local intervention with T3 in combination with dietary control may facilitate the browning of WAT, as illustrated in Fig. 4Q. Immunohistochemistry showed that UCP-1 protein expression in the WAT of mice in the CD + T3 group was significantly upregulated compared with that in the CD group (Fig. 4R).
Compared with those in the CD group, the relative expression levels of p-IRS-1/IRS-1 (p = 0.0001***), p-PI3K/PI3K (p = 0.0304*), and p-Akt/Akt (p = 0.0025**) were significantly lower in the CD + T3 group. There was no notable difference in the relative expression of p-mTOR/mTOR (Fig. 4S). In summary, the data suggest that local T3 intervention combined with dietary control promotes browning of WAT through inhibition of the PI3K/AKT signalling pathway.
Elevated insulin levels may contribute to a decrease in overall thermogenesis and abnormal local thermogenesis in WAT
The weight change trend of mice fed an HFD combined with insulin treatment on a weekly basis is illustrated in Fig. 5A. There was no statistically significant difference in the weights of the mice before intervention (p = 0.1343) or after intervention (p = 0.1099) in the two groups. Infrared thermal imaging analysis was conducted on the mice in the PBS group and the Det Ins group (p < 0.0001****), as depicted in Fig. 5B, S-Table 12, S-Fig. 5. The overall body temperatures of the mice in the Det Ins group decreased significantly. The serum levels of FT3 (p = 0.0100*) and FT4 (p = 0.0259*) in the Det Ins group were found to be significantly lower (Fig. 5C). Additionally, the levels of FT3 and FT4 in the WAT of the mice in each group was analysed, revealing a significant reduction in FT4 levels in the WAT of the Det Ins group (p = 0.0400*), while there was no statistically significant difference in FT3 levels (p = 0.2100) (Fig. 5D).
The protein expression of UCP-1 in the WAT of mice in the Det Ins group was significantly decreased (p = 0.0151*) (Fig. 5E). Additionally, the relative expression levels of p-IRS-1/IRS-1, p-PI3K/PI3K, and p-AKT/AKT in the Det Ins group tended to increase (Fig. 5F). The results of this experiment suggest that elevated insulin levels may contribute to a decrease in overall thermogenesis and abnormal local thermogenesis of WAT, potentially exacerbating obesity. However, further research is needed to elucidate the underlying mechanisms involved.
Discussion
Overall, we have provided evidence that T3 intervention promotes browning of WAT through inhibition of the PI3K/AKT signalling pathway.
Obesity is caused by the excessive accumulation of WAT in the body, leading to detrimental effects on the development of chronic diseases. Therefore, reducing WAT volume has emerged as a promising strategy for preventing obesity at its core. Given that the total WAT cell number remains relatively stable in adults, contemporary investigations into obesity treatment have predominantly concentrated on decreasing the WAT volume. Within the reported research thus far, the browning of WAT has garnered increasing attention as a prospective avenue for combating obesity. Considering the thermogenic properties of BAT, the browning of WAT is a viable approach for enhancing WAT utilisation and expediting metabolism. In this study, we observed that obesity adversely affects metabolism in individuals with obesity, with T3 resistance in local WAT and reduced energy expenditure within the local WAT, although compensatory increases in the systemic metabolic rate may occur. These results offer novel insights into the potential for targeted localised treatments aimed at WAT in obese individuals.
Previous research by Michalaki et al. demonstrated significantly elevated serum FT3 and FT4 levels in obese individuals, potentially attributable to heightened central thyroid function38. Additionally, other clinical studies have indicated that serum T3 levels are significantly elevated in obese individuals, which may be associated with cellular resistance39. In this study, the overall metabolism of obese mice and obese individuals was also significantly elevated, corroborating previous findings. The generation of TH is tightly regulated by classical negative feedback loops involving the hypothalamic-pituitary-thyroid axis40. When necessary, T3 and T4 are released into the blood circulation to exert their physiological effects. TH could modulate the expression of genes associated with mitochondrial biogenesis, oxidative phosphorylation, the tricarboxylic acid cycle, lipogenesis, and fatty acid catabolism41,42,43, augment the number of mitochondria within the cytoplasm, thereby enhancing the production of ATP and the oxygen necessary for metabolic processes to influences energy expenditure by regulating respiration and thermogenesis2.
TH serves as a fundamental regulator of metabolism, mitochondrial function, and thermogenesis31,44. Previous research have shown that the T3 could increase glucose uptake by heart cells, diaphragm cells, and fat cells by mediating the PI3K/AKT signalling pathway45,46. In addition, in cardiomyocytes or neuronal cells, the binding of thyroid hormones to cytoplasmic THR could promote cardiomyocyte growth or affect central system regulation through interactions with PI3K/AKT45,46,47. Notably, it exerts control over molecular pathways such as the PI3K, AKT, and mTOR signalling pathways through interactions between proteins and further governs mRNA transcription, such as uncoupling proteins, acetylamino esters, and apolipoproteins, which are related to various processes, including mitochondrial biogenesis, oxidative phosphorylation, the tricarboxylic acid cycle, adipogenesis, and fatty acid catabolism48. As previously stated, previous animal research has been conducted to support the notion that TH might induce the browning of WAT. In the current investigation, active T3 was selected as an intervention to directly target WAT, with an HA/T3 complex utilised to target local WAT in obese mice. It is worth mentioning that while the food intake of the mice increased following T3 intervention, their weight remained largely unchanged. The increased food intake may account for the lack of significant weight change in the mice. From an alternative perspective, it could be inferred that the weight of mice subjected to T3 intervention did not exhibit a significant increase despite an elevated food intake. This phenomenon may be attributed to T3's role in enhancing energy expenditure, which subsequently triggered a compensatory increase in food consumption. Consequently, the augmented metabolic rate offset the increased caloric intake, resulting in no substantial change in body weight. Based on these observations, the mice were grouped according to the average food intake of control mice with HA. The findings revealed that T3 stimulated the browning of WAT cells, and local HA/T3 complex intervention combined with dietary control enhanced systemic thermogenesis and facilitated the browning of WAT in obese mice. Both in vitro and in vivo studies have shown that T3 promotes the browning of WAT, leading to increased mitochondrial heat production and energy expenditure.
Next, we explored the potential mechanisms by which T3 promotes the browning of WAT. The PI3K/AKT pathway is a crucial regulatory pathway involved in various cellular functions, including the control of lipid metabolism, maintenance of glucose balance, regulation of protein synthesis and modulation of cell growth, proliferation, autophagy and apoptosis49,50,51. The PI3K/AKT signalling pathway plays an important role in maintaining insulin metabolic homeostasis, and the anabolic effects of insulin rely on its activation31. Increased concentrations of free fatty acids, glucose, and amino acids stimulate insulin signalling52,53, resulting in the direct or indirect binding of PI3K to IRS-1 receptors, which in turn modulates lipid and glucose metabolism through the recruitment of various molecules via the PI3K/AKT pathway. In adipocytes, there is antagonism between IRS-1/PI3K/AKT signalling and cAMP/PKA-driven lipolysis, which governs lipid catabolism54. The main downstream target of the PI3K/AKT pathway is mTOR, which is activated upon AKT binding to PI3K, followed by activation of mTOR and interaction with diverse downstream substrates. mTOR acts as a pivotal regulator of lipogenesis and glucose metabolism across various tissues, with its overactivation contributing to various structural growth disorders, such as obesity and T2DM27,55. In WAT, mTOR activation augments triglyceride synthesis and deposition and thus facilitates energy storage28. Research has demonstrated the role of the insulin/PI3K/AKT signalling pathway in promoting obesity. β-Adrenergic signalling is the most effective signalling pathway for inducing lipolysis in white adipocytes, and sustained β-adrenergic/cAMP/PKA signalling has been found to promote the formation and activation of thermogenic beige adipocytes. Conversely, the insulin/PI3K/AKT signalling pathway has been determined to be the most potent negative regulator of β-adrenergic-driven cAMP/PKA signalling and lipolysis25. Thus, insulin plays a crucial role in counteracting the mobilisation of lipids in AT, which potentially contributes to the development of obesity56. Multiple independent studies have demonstrated that reducing PI3K/AKT signalling through PI3K inhibition effectively safeguards mice against excessive obesity. In a model of obesity in mice and monkeys, the inhibition of pan-PI3K has been shown to ameliorate obesity and insulin resistance57. Additionally, mice with deletion of PI3Kγ expression (PI3Kγ-ko) exhibited protection against diet-induced obesity, insulin resistance, fatty liver and metabolic inflammation26,58. Further investigations have indicated that the deletion of PI3Kγ expression in nonhaematopoietic regions induces phenotypic thinning, reduced adipose tissue mass, heightened lipolysis, and browning of WAT in mice lacking catalytically active PI3Kβ and PI3Kγ26.
Previous studies have shown that TH stimulates WAT browning; but these studies have mainly relied on observational data obtained from animal experiments. Our study also confirmed that the thyroid hormone T3 promotes browning of WAT at the cellular and animal levels. However, until now, the mechanism by which T3 promotes the browning of WAT has not been clarified. The innovation of our study is as follows: First, the potential mechanism by which T3 promotes the browning of WAT, which may be mediated by inhibiting the PI3K/AKT signalling pathway, was investigated. Second, previous studies have been conducted under conditions in which circulating T3 levels increase (citation added), which can cause many side effects, such as arrhythmias, muscle weakness, weight loss, and irritable mood. In this study, the main purpose of combining T3 with cross-linked HA was to overcome the side effects caused by increasing the circulating T3 concentration in previous animal studies. HA/T3 conjugates target local AT, improve bioavailability, reduce systemic side effects caused by elevated circulating T3 levels, and prolong therapeutic duration and efficacy by regulating the rate of drug release.
This study utilised both in vivo and in vitro methodologies to demonstrate that T3 promoted browning of WAT by inhibiting the PI3K/AKT signalling pathway. Moreover, activation of the PI3K/AKT pathway was observed in both obese mice and obese people, suggesting its involvement in the pathogenesis of obesity, consistent with prior research. Nonetheless, the precise mechanism by which T3 modulates the PI3K/AKT signalling pathway through the insulin signalling pathway warrants further investigation and elucidation.
Materials and methods
All experimental procedures were performed in accordance with the specified guidelines and regulations.
Reagents
A high-fat diet (60 kcal%, PD6001) was purchased from SYSE Biotechnology Co. LTD. Normal feed was purchased from Beijing HFK Bioscience. Cell culture-related DMEM, FBS, antibiotic solution and pancreatin were purchased from Gibco. Collagenase was purchased from Sigma. BSA was purchased from Meilunbio. T3, insulin, dexamethasone, IBMX and an IRS-1/PI3K/AKT agonist (licarin B) were purchased from MedChemExpress. Mouse TSH ELISA kits were purchased from Jianglai. Mouse insulin ELISA kits, mouse C-peptide ELISA kits and mouse leptin ELISA kits were purchased from Elabscience. RNAeasy™ animal RNA isolation kits with spin columns were purchased from Beyotime. PrimeScript™ RT reagent kits and SYBR Green PCR kits were purchased from Takara. RIPA lysis buffer, protease inhibitor, phosphatase inhibitor and primary antibody diluent were purchased from Beyotime. A BCA protein assay kit was purchased from Biosharp. An SDS‒PAGE gel preparation kit, 10× Tris-Gly-SDS buffer, 5× loading buffer, 20× Western rapid transfer buffer, 10× TBST washes and skim milk powder were purchased from ZOMANBIO. Antibodies against UCP-1, CIDEA, PPARγ, PPATα, CEBPβ, FNDC5, DIO2, THRβ, IRS-1, p-IRS-1, PI3K, p-PI3K, AKT, p-AKT, mTOR, p-mTOR and β-actin were obtained from Affinity. Secondary antibodies and ECL reagents were also purchased from Affinity. A primary antibody against UCP-1 for immunofluorescence was purchased from Cell Signaling Technology. A PDVF membrane was purchased from Millipore. 4% paraformaldehyde, 0.5% Triton-100, PBS and DEPC were purchased from Biosharp. Oil red O working solution was purchased from OriCell. Crosslinked hyaluronic acid was purchased from Bloomage Biotech. Dete Insulin was purchased from Novo Nordisk Pharmaceutical Co., Ltd. Haematoxylin and eosin were purchased from Beyotime. Other supplies were purchased from Nest or LabSelect.
Animals
All animal experiments were conducted in accordance with the regulations and guidelines for the care and use of laboratory animals and approved by the Laboratory Animal Center of Quanzhou Medical College (SYXK (Min) 2023-0012). C57BL/6 mice were purchased from Shanghai Animal Laboratory Center (Shanghai SLAC Laboratory Animal Co., Ltd., China) and raised in the Laboratory Animal Center of Quanzhou Medical College. The animals were housed at 22 ± 2 °C and 55 ± 10% humidity under a 12 h light/dark cycle. Body weights were recorded every week. Each group had at least 3 independent samples.
Mouse model of obesity
Four-week-old male C57BL/6 mice (SPF grade, 16 ± 2 g) were randomly distributed into two groups: the normal diet (ND) group and the high-fat diet (HFD) group59,60,61. The mice were given free access to food and water. The HFD-fed mice were fed chow including 60 kcal% fat for 12 weeks, and the ND-fed mice fed a normal-fat diet were used as the control group. The weight of the mice in the HFD group was 20% greater than that of the ND group, which was considered to indicate successful modelling of obese mice.
Caloric restriction (CR) mice
Eight-week-old male C57BL/6 mice (SPF grade, 22 ± 2 g) were randomly distributed into two groups: the ad libitum (AL) group and the calorie restriction (CR) group62,63. CR mice were fed a diet restricted to 40% of the caloric intake of the ad libitum group for 6 weeks, while AL mice were fed a normal diet. The mice were given free access to water. The weight of the mice in the CR group was 30% lower than that of the mice in the AL group, which was considered to indicate successful establishment of ta calorie-restricted group.
T3 intervention was performed following the successful establishment of the obesity model
After successful establishment of the obesity mouse model, the HFD-fed mice were randomly divided into two groups and injected subcutaneously with hyaluronic acid (HA) (150 μl/week) or an HA/T3 complex (100 pmol/l, 150 μl/week). Subsequently, the mice were further divided into different groups based on observation indicators and whether feeding was controlled.
Construction of an obesity mouse model with insulin combined with HFD
During the construction of the obesity mouse model, the HFD group was randomly divided into two groups, which were injected with PBS (0.2 ml) or Dete Insulin (0.5 IU/ml, 0.2 ml/day) and continued to be fed an HFD. After the planned duration of the intervention, all animals were fasted for 12 h and anaesthetised with pentobarbital (0.1 mg/g of body weight) administered intraperitoneally by multiple researchers according to the manufacturer’s instructions. The body surface temperatures and thermographic data of the dorsal and ventral surfaces of the mice in each group were simultaneously monitored by a thermal imaging camera 5 ± 0.5 min after anesthesia operation. Blood samples were taken from the left ventricles of the mice. After blood sampling, the animals were sacrificed by cervical dislocation. The AT, liver, heart, lungs, spleen, and kidneys were then removed, rinsed, weighed if necessary, and fixed or directly stored at − 80 °C until further analysis.
Cell culture
White adipocytes
The ADSCs were extracted by digesting the subcutaneous fat from the groin of the mice using collagenase and BSA. Isolated ADSCs were cultured in DMEM supplemented with 10% FBS and antibiotic solution and were grown in a humidified incubator at 37 °C and 5% CO2. When the cells reached 100% confluence, 8.7 µmol/L insulin, 2.5 µmol/L dexamethasone and 0.5 mmol/L IBMX were added to the fusion culture and incubated for 3 days, and then the medium was replaced with insulin (10 µmol/L) alone for 2 days64. The two media were used alternately until 95% of the lipid droplets were of appropriate size under the microscope. That is, the induction of lipogenesis was successful.
Administration of T3
The white adipocytes were treated with T3 for a period of 72 h. A physiological WAT FT3 concentration (10 pmol/l) was used to treat the control WAT group, and the tenfold gradient of T3 concentration was successively established. White adipocytes were treated with T3 at concentrations of 1 × 102 pmol/l, 1 × 103 pmol/l, 1 × 104 pmol/l, 1 × 105 pmol/l or 1 × 106 pmol/l for 72 h in the intervention groups.
Administration of T3 combined with PI3K/AKT agonists
We treated white adipocytes with T3 in combination with an IRS-1/PI3K/AKT agonist (licarin B, 15 µmol/l) for 72 ho. White adipocytes were divided into four groups: control, 100 pmol/L T3, PI3K/AKT agonist and T3 + PI3K/AKT agonist.
Administration of insulin
White adipocytes were stimulated with insulin-free medium or medium supplemented with 850 nmol/l insulin for 72 h.
Administration of T3 combined with insulin
We treated white adipocytes with T3 in combination with insulin (850 nmol/l) for 72 h. White adipocytes were divided into four groups: the control group, the 100 pmol/L T3 group, the insulin group and the T3 + insulin agonist group.
The above experiments were performed independently three times.
Clinical samples
We conducted two cross-sectional studies that adhered to the guidelines set forth in the Declaration of Helsinki. Prior to participation, all subjects were provided with detailed information regarding the study's objectives and all subjects subsequently signed informed consent forms. The collection of blood and tissue samples was authorized by the Medical Ethics Committee of the Second Affiliated Hospital of Fujian Medical University (054).
First, a cohort of 81 individuals at the Physical Examination Center of our hospital who were deemed healthy and to have normal thyroid function between September 2022 and August 2023 were included in the study. The heights and weights of all participants were initially measured, and BMIs were subsequently calculated. The participants were subsequently grouped according to BMI as follows: normal (18.5 ≤ BMI < 25), overweight (25 ≤ BMI < 30) and obese (BMI ≥ 30). Blood samples were collected in a procoagulant tube to obtain serum and sent to the clinical laboratory for measurement of FT3, FT4 and TSH levels.
Second, individuals with normal thyroid function who needed to receive gynaecologic laparoscopic surgery at the Department of Obstetrics and Gynecology of our hospital from January 2023 to October 2023 were included and were also grouped according to BMI into the following groups: normal, overweight and obese. An appropriate quantity of adipose tissue from the distal border of the greater omentum was extracted during the operation by the chief obstetrician of our hospital. The collected omental adipose tissue was quickly transferred to liquid nitrogen and stored at − 80 °C to facilitate subsequent analysis.
Assessment of the basal surface with infrared thermography
The basal surface temperatures of the mice were measured via infrared thermography (T2Pro, Airui, China). After being anaesthetised, the mice were placed in a temperature-controlled room at 22 °C and were not allowed to be exposed to outside wind or light.
Blood biochemistry
Blood samples were sent to the clinical laboratory of our hospital. Serum FT3 and FT4 levels were measured by electrochemiluminescence, and serum glucose, TC, TG, LDL-C, HDL-C, ALT and AST were measured by enzymatic colorimetric assays.
Measurement of FT3 and FT4 in tissue homogenates
WAT and BAT were taken and weighed according to tissue weight: tissue lysate = 1:3; liver, heart, lung, spleen and kidney were taken according to tissue weight: tissue lysate = 1:9. After full cleavage, FT3 and FT4 levels in tissue homogenates were measured by electrochemiluminescence.
Measurement of serum hormone concentrations by ELISA
The serum levels of TSH in mice were measured using mouse ELISA kits, which are competitive ELISA kits. Serum insulin, C-peptide and leptin levels were determined through a double-antibody sandwich ELISA in mouse insulin ELISA kits, mouse C-peptide ELISA kits and mouse leptin ELISA kits.
Evaluation of mRNA expression levels by qRT‒PCR
Total RNA was extracted from white adipocytes and WAT using RNAeasyTM animal RNA isolation kits according to the standard protocol and then reverse transcribed into cDNA using a PrimeScript™ RT reagent kit. qRT‒PCR was performed using a SYBR Green PCR Kit in a 7500 Real-Time PCR System (Thermo Fisher Scientific). The relative expression levels were analysed by the 2-ΔΔCT method. The gene-specific primers used in this study, which were designed by Shenggong Biotechnology Co., Ltd., are listed in S-Table 1.
Determination of protein expression by Western blotting (WB)
Total proteins were isolated from white adipocytes and WAT using RIPA lysis buffer supplemented with protease and phosphatase inhibitors. The protein concentration was measured using the BCA method. Subsequently, the boiled protein samples were subjected to electrophoresis and electrically transferred to PDVF membranes. The membranes were blocked with 5% skim milk solution for 2 h and incubated with different primary antibodies overnight at 4 °C. Subsequently, the membranes were washed with TBST, and the membranes were incubated with secondary antibodies for 1 h. After washing the membranes with TBST, the protein bands were detected with enhanced chemiluminescence (ECL) and visualised using X-ray film. The proteins were quantified using Image J (version 1.8.0) (https://imagej.en.softonic.com/) analysis software.
Evaluation of protein expression by immunofluorescence (IF)
Postintervention white adipocytes were fixed in 4% paraformaldehyde and then permeabilised with 0.5% Triton-100. Next, the cells were blocked with 5% BSA and incubated with different primary antibodies in a humidified chamber overnight at 4 °C. The cells were washed with PBS and then incubated with secondary antibodies for 1 h at room temperature in the dark. After washing, the cells were incubated with DAPI solution in the dark to stain the cell nuclei. The samples were photographed with a camera (KFBIO KF-PRO-120 pathology slide scanner).
Oil red O staining
White adipocytes were fixed in 4% paraformaldehyde for 30 min and then washed with PBS. Next, the sections were stained with oil red O working solution for 30 min at room temperature. After staining, the sections were washed with PBS and photographed under a light microscope.
High-throughput mRNA sequencing
Construction of the transcriptome
High-throughput mRNA sequencing was performed by Biomarker Technologies Co., Ltd. Total RNA was extracted from white adipocytes using Trizol reagent. After the samples were qualified, the randomly sheared mRNA was used as a template for cDNA purification, and the cDNA library was obtained by PCR enrichment. Finally, the PCR products were purified (AMPure XP system), and library quality was assessed on an Agilent Bioanalyzer 2100 system. The libraries were sequenced on an Illumina NovaSeq platform to generate 150 bp paired-end reads according to the manufacturer’s instructions. Differential expression analysis of both groups was performed using DESeq2. Genes with an adjusted P value < 0.01 and a fold change ≥ 2 according to DESeq2 were considered differentially expressed.
Functional enrichment analysis
GO enrichment analysis of the DEGs was performed with the clusterProfiler package-based Wallenius noncentral hypergeometric distribution. Statistical enrichment analysis of DEGs in KEGG pathways was performed using the KOBAS database and clusterProfiler software. R software (version 3.4.1) (https://www.r-project.org/) was used for visualisation of the functional enrichment analysis.
In vitro drug release
The release curve of the HA/T3 complex was studied by the dialysis bag method using PBS as the release medium at a pH of 7.2 and a temperature of 37 ℃. A dialysis bag containing 1.0 ml of the HA/T3 complex was immersed in a vial containing 100 ml of PBS at 37 °C and incubated in a temperature-controlled magnetic stirring device at a stirring speed of 100 rpm. Samples of 200 μl were taken at the specified time points for detection, and the content of T3 in the filtrate was determined; that is, the content of T3 released from the sample during this period was measured. The same volume of PBS solution was added to maintain an unchanged total volume of the release system. The release curve of the drug was plotted using the cumulative amount and time of T3 release per day.
Haematoxylin and eosin (HE) staining
After fixation and embedding, the tissues and organs were dewaxed and rehydrated, stained with haematoxylin solution for 10 min followed by 5 s in 1% acid ethanol, rinsed in distilled water and dehydrated with an alcohol gradient. Next, the sections were stained with eosin solution for 3 min, dehydrated again using an alcohol gradient and cleared with xylene. Finally, the sections were photographed with a camera (KFBIO KF-PRO-120 pathology slide scanner).
Evaluation of UCP-1 protein expression by immunohistochemistry (IHC)
WAT was fixed with 4% paraformaldehyde in phosphate-buffered saline, dehydrated, embedded in paraffin, and sectioned. After dewaxing and rehydration, the sections were stained with anti-UCP-1 (1:200), haematoxylin was added, the sections were dehydrated with an alcohol gradient, and xylene was used for clearing. Finally, the sections were photographed with a camera (KFBIO KF-PRO-120 pathology slide scanner).
Statistical analysis
The data are presented as the means ± standard deviations (x̅ ± s), and all analyses were performed using GraphPad Prism (version 8.0)(https://www.graphpad.com/). Continuous outcomes were analysed using independent t tests for two groups. One-way ANOVA was used for multiple comparisons. All reported p values were two-tailed, and a value of p < 0.05 was considered to indicate statistical significance.
Guideline statement
The study is reported in accordance with the ARRIVE guidelines.
Conclusions
The results of our study indicated that T3 promotes browning of WAT through inhibition of the PI3K/AKT signalling pathway. This finding suggests that enhancing the concentration of T3 in WAT enhances its local metabolic activity, leading to increased energy expenditure, which offers novel perspectives on the potential of localised therapies for addressing WAT volume in individuals with obesity.
Data availability
The datasets generated and/or analyzed during the current study are available in the Sequence Read Archive(SRA) repository, PRJNA1097497. (https://dataview.ncbi.nlm.nih.gov/object/PRJNA1097497?reviewer=9p0m274lm7qpk60ji54vv9he8d).
References
National Institutes of Health. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults—the evidence report. Obes. Res. 6(Suppl 2), 51s–209s (1998).
Walczak, K. & Sieminska, L. Obesity and thyroid axis. Int. J. Environ. Res. Public Health https://doi.org/10.3390/ijerph18189434 (2021).
Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 894, i-xii, 1–253 (2000).
Jiang, L. et al. The relationship between body mass index and hip osteoarthritis: A systematic review and meta-analysis. Joint Bone Spine 78, 150–155. https://doi.org/10.1016/j.jbspin.2010.04.011 (2011).
Lauby-Secretan, B. et al. Body fatness and cancer-viewpoint of the IARC working group. N. Engl. J. Med. 375, 794–798. https://doi.org/10.1056/NEJMsr1606602 (2016).
Singh, G. M. et al. The age-specific quantitative effects of metabolic risk factors on cardiovascular diseases and diabetes: A pooled analysis. PLoS ONE 8, e65174. https://doi.org/10.1371/journal.pone.0065174 (2013).
Finucane, M. M. et al. National, regional, and global trends in body-mass index since 1980: Systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9·1 million participants. Lancet 377, 557–567. https://doi.org/10.1016/s0140-6736(10)62037-5 (2011).
Sadeghian, M., Hosseini, S. A., Zare Javid, A., Ahmadi Angali, K. & Mashkournia, A. Effect of fasting-mimicking diet or continuous energy restriction on weight loss, body composition, and appetite-regulating hormones among metabolically healthy women with obesity: A randomized controlled. Parallel Trial. Obes. Surg. 31, 2030–2039. https://doi.org/10.1007/s11695-020-05202-y (2021).
Fogarasi, A., Gonzalez, K., Dalamaga, M. & Magkos, F. The impact of the rate of weight loss on body composition and metabolism. Curr. Obes. Rep. 11, 33–44. https://doi.org/10.1007/s13679-022-00470-4 (2022).
Franz, M. J. et al. Weight-loss outcomes: A systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J. Am. Diet Assoc. 107, 1755–1767. https://doi.org/10.1016/j.jada.2007.07.017 (2007).
Lamarca, F., Melendez-Araújo, M. S., Porto de Toledo, I., Dutra, E. S. & de Carvalho, K. M. B. Relative energy expenditure decreases during the first year after bariatric surgery: A systematic review and meta-analysis. Obes. Surg. 29, 2648–2659. https://doi.org/10.1007/s11695-019-03934-0 (2019).
van Marken Lichtenbelt, W. Brown adipose tissue and the regulation of nonshivering thermogenesis. Curr. Opin. Clin. Nutr. Metab. Care 15, 547–552. https://doi.org/10.1097/MCO.0b013e3283599184 (2012).
Shao, M. et al. Cellular origins of beige fat cells revisited. Diabetes 68, 1874–1885. https://doi.org/10.2337/db19-0308 (2019).
Zhang, J. et al. TSH promotes adiposity by inhibiting the browning of white fat. Adipocyte 9, 264–278. https://doi.org/10.1080/21623945.2020.1783101 (2020).
Bartelt, A. & Heeren, J. Adipose tissue browning and metabolic health. Nat. Rev. Endocrinol. 10, 24–36. https://doi.org/10.1038/nrendo.2013.204 (2014).
Yoneshiro, T. et al. Recruited brown adipose tissue as an antiobesity agent in humans. J. Clin. Investig. 123, 3404–3408. https://doi.org/10.1172/jci67803 (2013).
Tur, J. A. & Bibiloni, M. D. M. Anthropometry, body composition and resting energy expenditure in human. Nutrients https://doi.org/10.3390/nu11081891 (2019).
Leibel, R. L., Rosenbaum, M. & Hirsch, J. Changes in energy expenditure resulting from altered body weight. N. Engl. J. Med. 332, 621–628. https://doi.org/10.1056/nejm199503093321001 (1995).
Doucet, E. et al. Evidence for the existence of adaptive thermogenesis during weight loss. Br. J. Nutr. 85, 715–723. https://doi.org/10.1079/bjn2001348 (2001).
Johnstone, A. M., Murison, S. D., Duncan, J. S., Rance, K. A. & Speakman, J. R. Factors influencing variation in basal metabolic rate include fat-free mass, fat mass, age, and circulating thyroxine but not sex, circulating leptin, or triiodothyronine. Am. J. Clin. Nutr. 82, 941–948. https://doi.org/10.1093/ajcn/82.5.941 (2005).
Weiner, J. et al. Thyroid hormone status defines brown adipose tissue activity and browning of white adipose tissues in mice. Sci. Rep. 6, 38124. https://doi.org/10.1038/srep38124 (2016).
Schulz, T. J. et al. Brown-fat paucity due to impaired BMP signalling induces compensatory browning of white fat. Nature 495, 379–383. https://doi.org/10.1038/nature11943 (2013).
Johann, K. et al. Thyroid-hormone-induced browning of white adipose tissue does not contribute to thermogenesis and glucose consumption. Cell Rep. 27, 3385-3400.e3383. https://doi.org/10.1016/j.celrep.2019.05.054 (2019).
Villicev, C. M. et al. Thyroid hormone receptor beta-specific agonist GC-1 increases energy expenditure and prevents fat-mass accumulation in rats. J. Endocrinol. 193, 21–29. https://doi.org/10.1677/joe.1.07066 (2007).
Solinas, G. & Becattini, B. PI3K and AKT at the interface of signaling and metabolism. Curr. Top. Microbiol. Immunol. 436, 311–336. https://doi.org/10.1007/978-3-031-06566-8_13 (2022).
Becattini, B. et al. PI3Kγ within a nonhematopoietic cell type negatively regulates diet-induced thermogenesis and promotes obesity and insulin resistance. Proc. Natl. Acad. Sci. USA 108, E854-863. https://doi.org/10.1073/pnas.1106698108 (2011).
Huang, S. mTOR signaling in metabolism and cancer. Cells https://doi.org/10.3390/cells9102278 (2020).
Liu, G. Y. & Sabatini, D. M. mTOR at the nexus of nutrition, growth, ageing and disease. Nat. Rev. Mol. Cell Biol. 21, 183–203. https://doi.org/10.1038/s41580-019-0199-y (2020).
Salas-Lucia, F. & Bianco, A. C. T3 levels and thyroid hormone signaling. Front. Endocrinol. 13, 1044691. https://doi.org/10.3389/fendo.2022.1044691 (2022).
Mendoza, A. & Hollenberg, A. N. New insights into thyroid hormone action. Pharmacol. Ther. 173, 135–145. https://doi.org/10.1016/j.pharmthera.2017.02.012 (2017).
Gauthier, B. R. et al. Thyroid hormones in diabetes, cancer, and aging. Aging Cell 19, e13260. https://doi.org/10.1111/acel.13260 (2020).
Kang, M. C. et al. Anti-obesity effects of Sargassum thunbergii via downregulation of adipogenesis gene and upregulation of thermogenic genes in high-fat diet-induced obese mice. Nutrients https://doi.org/10.3390/nu12113325 (2020).
Festuccia, W. T., Blanchard, P. G. & Deshaies, Y. Control of brown adipose tissue glucose and lipid metabolism by PPARγ. Front. Endocrinol. 2, 84. https://doi.org/10.3389/fendo.2011.00084 (2011).
Tiefenbach, J. et al. Idebenone and coenzyme Q(10) are novel PPARα/γ ligands, with potential for treatment of fatty liver diseases. Dis. Model. Mech. https://doi.org/10.1242/dmm.034801 (2018).
Kanehisa, M. & Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30. https://doi.org/10.1093/nar/28.1.27 (2000).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 28, 1947–1951. https://doi.org/10.1002/pro.3715 (2019).
Kanehisa, M., Furumichi, M., Sato, Y., Kawashima, M. & Ishiguro-Watanabe, M. KEGG for taxonomy-based analysis of pathways and genomes. Nucleic Acids Res. 51, D587-d592. https://doi.org/10.1093/nar/gkac963 (2023).
Nie, X. et al. Characteristics of serum thyroid hormones in different metabolic phenotypes of obesity. Front. Endocrinol. 11, 68. https://doi.org/10.3389/fendo.2020.00068 (2020).
Pearce, E. N. Thyroid hormone and obesity. Curr. Opin. Endocrinol. Diabetes Obes. 19, 408–413. https://doi.org/10.1097/MED.0b013e328355cd6c (2012).
Bianco, A. C. & McAninch, E. A. The role of thyroid hormone and brown adipose tissue in energy homoeostasis. Lancet Diabetes Endocrinol. 1, 250–258. https://doi.org/10.1016/s2213-8587(13)70069-x (2013).
Flores-Morales, A. et al. Patterns of liver gene expression governed by TRbeta. Mol. Endocrinol. 16, 1257–1268. https://doi.org/10.1210/mend.16.6.0846 (2002).
Jackson-Hayes, L. et al. A thyroid hormone response unit formed between the promoter and first intron of the carnitine palmitoyltransferase-Ialpha gene mediates the liver-specific induction by thyroid hormone. J. Biol. Chem. 278, 7964–7972. https://doi.org/10.1074/jbc.M211062200 (2003).
Singh, B. K. et al. Thyroid hormone receptor and ERRα coordinately regulate mitochondrial fission, mitophagy, biogenesis, and function. Sci. Signal. https://doi.org/10.1126/scisignal.aam5855 (2018).
Sinha, R. A., Singh, B. K. & Yen, P. M. Direct effects of thyroid hormones on hepatic lipid metabolism. Nat. Rev. Endocrinol. 14, 259–269. https://doi.org/10.1038/nrendo.2018.10 (2018).
Witham, E. A. et al. Kisspeptin regulates gonadotropin genes via immediate early gene induction in pituitary gonadotropes. Mol. Endocrinol. 27, 1283–1294. https://doi.org/10.1210/me.2012-1405 (2013).
Matsui, H., Takatsu, Y., Kumano, S., Matsumoto, H. & Ohtaki, T. Peripheral administration of metastin induces marked gonadotropin release and ovulation in the rat. Biochem. Biophys. Res. Commun. 320, 383–388. https://doi.org/10.1016/j.bbrc.2004.05.185 (2004).
Uenoyama, Y., Tsukamura, H. & Maeda, K. I. Kisspeptin/metastin: A key molecule controlling two modes of gonadotrophin-releasing hormone/luteinising hormone release in female rats. J. Neuroendocrinol. 21, 299–304. https://doi.org/10.1111/j.1365-2826.2009.01853.x (2009).
Cheng, S. Y., Leonard, J. L. & Davis, P. J. Molecular aspects of thyroid hormone actions. Endocr. Rev. 31, 139–170. https://doi.org/10.1210/er.2009-0007 (2010).
Hamzehzadeh, L., Atkin, S. L., Majeed, M., Butler, A. E. & Sahebkar, A. The versatile role of curcumin in cancer prevention and treatment: A focus on PI3K/AKT pathway. J. Cell Physiol. 233, 6530–6537. https://doi.org/10.1002/jcp.26620 (2018).
Heras-Sandoval, D., Pérez-Rojas, J. M., Hernández-Damián, J. & Pedraza-Chaverri, J. The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal. 26, 2694–2701. https://doi.org/10.1016/j.cellsig.2014.08.019 (2014).
Wu, H. T. et al. MLL3 induced by luteolin causes apoptosis in tamoxifen-resistant breast cancer cells through H3K4 monomethylation and suppression of the PI3K/AKT/mTOR pathway. Am. J. Chin. Med. 48, 1221–1241. https://doi.org/10.1142/s0192415x20500603 (2020).
Czech, M. P. Insulin action and resistance in obesity and type 2 diabetes. Nat. Med. 23, 804–814. https://doi.org/10.1038/nm.4350 (2017).
Czech, M. P., Tencerova, M., Pedersen, D. J. & Aouadi, M. Insulin signalling mechanisms for triacylglycerol storage. Diabetologia 56, 949–964. https://doi.org/10.1007/s00125-013-2869-1 (2013).
Ravnskjaer, K., Madiraju, A. & Montminy, M. Role of the cAMP pathway in glucose and lipid metabolism. Handb. Exp. Pharmacol. 233, 29–49. https://doi.org/10.1007/164_2015_32 (2016).
Chang, T. J. et al. Genetic variation of SORBS1 gene is associated with glucose homeostasis and age at onset of diabetes: A SAPPHIRe Cohort Study. Sci. Rep. 8, 10574. https://doi.org/10.1038/s41598-018-28891-z (2018).
Zechner, R., Madeo, F. & Kratky, D. Cytosolic lipolysis and lipophagy: Two sides of the same coin. Nat. Rev. Mol. Cell Biol. 18, 671–684. https://doi.org/10.1038/nrm.2017.76 (2017).
Ortega-Molina, A. et al. Pharmacological inhibition of PI3K reduces adiposity and metabolic syndrome in obese mice and rhesus monkeys. Cell Metab. 21, 558–570. https://doi.org/10.1016/j.cmet.2015.02.017 (2015).
Kobayashi, N. et al. Blockade of class IB phosphoinositide-3 kinase ameliorates obesity-induced inflammation and insulin resistance. Proc. Natl. Acad. Sci. USA 108, 5753–5758. https://doi.org/10.1073/pnas.1016430108 (2011).
Griffin, T. M., Batushansky, A., Hudson, J. & Lopes, E. B. P. Correlation network analysis shows divergent effects of a long-term, high-fat diet and exercise on early stage osteoarthritis phenotypes in mice. J. Sport Health Sci. 9, 119–131. https://doi.org/10.1016/j.jshs.2019.05.008 (2020).
Fu, Z. et al. In vivo self-assembled small RNAs as a new generation of RNAi therapeutics. Cell Res. 31, 631–648. https://doi.org/10.1038/s41422-021-00491-z (2021).
Kim, S. et al. Tanycytic TSPO inhibition induces lipophagy to regulate lipid metabolism and improve energy balance. Autophagy 16, 1200–1220. https://doi.org/10.1080/15548627.2019.1659616 (2020).
Liu, X. et al. Calorie restriction and calorie dilution have different impacts on body fat, metabolism, behavior, and hypothalamic gene expression. Cell Rep. 39, 110835. https://doi.org/10.1016/j.celrep.2022.110835 (2022).
Pak, H. H. et al. Fasting drives the metabolic, molecular and geroprotective effects of a calorie-restricted diet in mice. Nat. Metab. 3, 1327–1341. https://doi.org/10.1038/s42255-021-00466-9 (2021).
Jiang, D. et al. Thyroid-stimulating hormone inhibits adipose triglyceride lipase in 3T3-L1 adipocytes through the PKA pathway. PLoS ONE 10, e0116439. https://doi.org/10.1371/journal.pone.0116439 (2015).
Funding
The Fujian Province Science and Technology Innovation Joint Funding Project (2021Y9028). The Second Affiliated Hospital of Fujian Medical University horizontal scientific research project (HX202201 and HX202202). The Second Affiliated Hospital of Fujian Medical University Doctoral Research Project (2022BD0701).
Author information
Authors and Affiliations
Contributions
LingHong Huang and HuiBin Huang conceived the project; designed, performed, and analyzed experiments; and wrote the manuscript. ZhiFeng Guo contributed to bioinformatics analysis and MingJing Huang and XiYing Zeng contributed to in vivo experiments. HuiBin Huang is the guarantor of this work and had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huang, L., Guo, Z., Huang, M. et al. Triiodothyronine (T3) promotes browning of white adipose through inhibition of the PI3K/AKT signalling pathway. Sci Rep 14, 20370 (2024). https://doi.org/10.1038/s41598-024-71591-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-71591-0
Keywords
This article is cited by
-
Obesity: pathophysiology and therapeutic interventions
Molecular Biomedicine (2025)