Abstract
As a result of its high reactivity, furan-2(3H)-one derivative 2 can be selected as a versatile and suitable candidate for building of novel nitrogen heterocyclic compounds. Consequently, furan-2(3H)-one derivative 2 and some nitrogen nucleophiles were utilized as starting materials for the formation of new pyridazinone and pyrrolone derivatives bearing naphthalene moiety. The continuous buildup of insecticide resistance is the main obstacle facing pest control measures. Pyrrole-based insecticides are a favourable choice due to their unique mode of action and no cross-resistance with traditional neurotoxic insecticides. The larvicidal activities of pyrrolone derivatives were assessed against field and laboratory strains of Culex pipiens larvae in comparison with chlorfenapyr (pyrrole insecticide). Compounds 17 (21.05 µg/mL) > 9 (22.81 µg/mL) > 15 (24.39 µg/mL) > 10 (26.76 µg/mL) > 16 (32.09 µg/mL) were most effective against lab strain of C. pipiens larvae relative to chlorfenapyr (25.43 µg/mL). While in field strain, 17 and 15 were the most toxic compounds followed by 9 > 10 > 16 > 2 with LC50 of 9.87, 10.76, 11.52, 12.68, 15.32 and 18.37 µg/mL, respectively, compared with chlorfenapyr with 14.03 µg/mL. The cytochrome P-450 monooxygenase activities were significantly increased in treated larvae of lab and field strains relative to untreated. The great variations in toxicity of the synthesized compounds were interpreted by structure-activity relationship study. The pyrrolone derivatives are effective against field and insecticide-resistant strains. Therefore, they are considered promising compounds to be integrated into pest management programs.
Similar content being viewed by others
Introduction
Due to their facile ring opening by various nucleophiles, 2(3 H)furanones were readily transformed into a diversity of nitrogen heterocyclic systems of pharmacological significance particularly pyrrolones, oxadiazoles, pyridazinones and triazoles1. Several sorts of synthetic pyrrolones have a broad range of significant biological activities including antifungal2, antibacterial3, anti-inflammatory, anticonvulsant4, anticancer5, antimalarial6 and vasodilation7. In the last decades, there had been reports on many biological applications of various pyrrole derivatives either from natural resources or chemically synthesized. It was reported that some pyrrole derivatives can act as enzyme inhibitors, acaricidal and insecticidal agents8. Furthermore, pyrrolones and pyridazinones have wide applications in medicinal uses. Althiomycin is one of the most important naturally occurring pyrrolone alkaloids. It is isolated from Streptomyces althioticus and used as an antibiotic via inhibition of protein synthesis9(Fig. 1).
Mosquito species are vectors of severe infectious diseases to humans and animals such as parasites (lymphatic filariasis and malaria) and viruses (Rift Valley fever virus, Zika, chikungunya and avian arbovirus)10. Additionally, recent studies detected the transmission of Rickettsia (R.) felis bacteria by mosquitoes11. These dreadful diseases have a significant adverse impact on public health. For malaria only, more than 229 million cases and about 409,000 deaths were detected worldwide in 201912. The African regional disease represents approximately 90% of these cases and deaths detected yearly13. Also, global warming could increase the transmission risks and related threats of mosquito-borne diseases posed by mosquitoes in temperate regions14. Several mosquito species and sub-species have been recorded and identified worldwide including Culex pipiens L. (Diptera: Culicidae). Culex pipiens is a crucial nuisance and vector of severe human and animal diseases such as lymphatic filariasis and Japanese encephalitis15. Testing the susceptibility of mosquito larvae could provide crucial information for the potency assessment of new insecticides and the probability of resistance development in adults16.
Chemical application is the fastest, most reliable and effective pest control method. The intensive usage of chemical neurotoxic insecticides threatened their efficacies with the gradual rise of resistance mosquitoes17. Chlorfenapyr is a novel pyrrole insecticide class with a completely different mode of action from traditional neurotoxic insecticides used to combat mosquitoes. Pyrroles belong to class 13 of the IRAC classification, uncouplers of oxidative phosphorylation18 that exhibited a unique mode of action since it disrupts the energy production of insects. Therefore, this class doesn’t show cross-resistance with conventional insecticides used in vector control programs19.
The foregoing reports, coupled with the interest of our research group in the synthesis of biologically active heterocycles, encouraged us to report herein on the synthesis of some novel pyrrolone derivatives with anticipated insecticidal activities. The role of cytochrome P450s in the activation of tested compounds was also studied. Furthermore, the effect of different moieties inserted will be detected via the structure activity relationship study.
Pyrrole-based compounds with known biological activity.
Rational design of the work
Oriented by the insecticidal activity of chlorfenapyr; several chlorfenapyr backbone-based substances have been designed and synthesized, and numerous promising compounds have been obtained. Investigation of the primary structural features of chlorfenapyr (Fig. 2) disclosed the prominence of the following parts: (i) insertion of electron-withdrawing group (EWG) and an aryl group at positions 2- and 3 of the pyrrole ring, respectively, improved the activity of the compounds; (ii) substitution at position 5- of the pyrrole ring with aryl group substituted at para position with EWG or lipophilic group, such as Cl enhance the insecticidal activity; (iii) introducing short linear alkyl or alkoxy substitutions (small moieties) at the NH position showed substantially high contact or systemic insecticidal effectiveness, while those with longer or branched alkyl insertions (large bulking moieties) tended to decline the efficacy. From these structural features, this study planned to obtain some novel pyrrolone derivatives with anticipated insecticidal activities.
Rational design for the pyrrolone candidates as insecticidal agents based on the previously reported chlorfenapyr (pyrrole insecticide).
Results and discussion
Chemistry
As a continuation of our preceding work on the synthesis of various heterocyclic compounds with evaluation of their applications in biological and medicinal fields20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39, the present investigation focuses on the synthesis of new furanone, pyridazinone and pyrrolone derivatives bearing naphthalene moiety with estimation of their insecticidal activities. The required 5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)furan-2(3 H)-one (2) was prepared by a single step reaction under Perkin reaction conditions. Thus, condensation of 1-naphthaldehyde with 3-aroyl propionic acid 1 in the presence of freshly distilled acetic anhydride and fused sodium acetate resulted in formation of furan-2(3 H)-one derivative 2 in a good yield via anhydrous conditions (Fig. 3). The IR spectrum of furanone derivative 2 showed the absorption band for carbonyl group of γ-lactone at υ 1771 cm− 1.
Synthesis of furanone derivative 2 under conventional conditions.
The functionality in furan-2(3 H)-one derivative 2 renders it valuable candidate for building of novel nitrogen heterocyclic compounds bearing naphthalene moiety as the carbonyl function of γ-lactone is properly situated to allow reactions with common reagents. Thereby, the reaction of furan-2(3 H)-one derivative 2 with formamide under reflux gave the corresponding 3-arylidene-5-(4-chlorophenyl)-2H-pyrrol-2-one derivative 3 (Fig. 4). The IR spectrum of compound 3 showed the disappearance of the carbonyl group of γ-lactone absorption band and the presence of cyclic imide carbonyl absorption at υ 1695 cm− 1, alongside the NH absorption at υ 3154 cm− 1. In addition, its 1H NMR spectrum displayed an exchangeable singlet signal for NH proton at δ 10.71 ppm. Moreover, the interaction between the furanone derivative 2 and cyclohexylamine or benzylamine in boiling dioxane led to ring opening with the formation of the open-chain amide analogs 4 and 5, respectively (Fig. 4). The latter products 4 and 5 were subjected to ring closure to their pyrrolone derivatives 6 and 7 by refluxing in HCl/AcOH mixture (Fig. 4). The chemical structures of the amide analogs 4 and 5 and the pyrrolones 6 and 7 were proved from analytical, together with spectral data (cf. Experimental).
Reaction of furanone derivative 2 with some N-nucleophiles.
Meanwhile, reacting the furanone derivative 2 with some acid hydrazides such as ethyl carbazate and/or thiosemicarbazide under reflux in dioxane/AcOH mixture gave the desired pyrrolone derivatives 8 and 9, respectively as presented in Fig. 5. The structures of pyrrolone derivatives 8 and 9 were illustrated by IR, 1H NMR and 13C NMR spectroscopy, as well as elemental analysis. On the other hand, the acid hydrazide 10 was generated by stirring the furan-2(3 H)-one derivative 2 with hydrazine hydrate in dioxane at room temperature (Fig. 5). IR spectrum of the hydrazide 10 revealed the presence of absorption bands for NH2, NH groups at υ 3312, 3210, 3195 cm− 1, and C = O group of amide at υ 1691 cm− 1 whereas the absorption band of carbonyl group of γ-lactone was disappeared. Also, 1H NMR spectrum of 10 showed two exchangeable signals for NH and NH2 protons in addition to the methylene protons. The 13C NMR spectrum provided sufficient evidence for the structure of hydrazide 10 (cf. Experimental).
Treatment of furanone derivative 2 with some acid hydrazides and hydrazine hydrate.
The acid hydrazide 10 was utilized as a principal starting material for the synthesis of some pyridazinone and pyrrolone derivatives bearing a naphthalene moiety. Therefore, the pyridazinone derivative 11 was obtained via refluxing the hydrazide derivative 10 in a mixture of glacial acetic acid and hydrochloric acid solution (Fig. 6). Condensation of compound 10 with isatin in refluxing EtOH/AcOH mixture yielded the desired pyrrolone derivative 12 (Fig. 6). Contrarily, condensation of hydrazide 10 with p-methoxybenzaldehyde under various conditions was also examined. Thus, conducting the reaction in boiling ethanol yielded the hydrazone derivative 13. Whereas carrying out the reaction in ethanol/glacial acetic acid mixture under reflux motivated the cyclization step to generate the pyrrolone derivative 14. The same compound 14 was also achieved by ring closure of the hydrazone 13 in refluxing HCl/AcOH mixture (Fig. 6). The chemical structures of obtained compounds 13 and 14 were verified from their spectral and analytical data. Their IR spectra lacked the carbonyl group of γ-lactone absorption band and displayed the amide carbonyl absorption band for the hydrazone derivative 13 at υ 1677 cm− 1 and the pyrrolone derivative 14 at υ 1703 cm− 1 in addition to the NH absorption at υ 3167 cm− 1 for the hydrazone derivative 13.
Cyclization of hydrazide derivative 10 to generate pyridazinone and pyrrolone derivatives.
Additionally, refluxing a solution of the hydrazide 10 in ethanol with chloroacetyl chloride on a water-bath furnished the pyrrolone derivative 15 (Fig. 7). The IR spectrum of pyrrolone derivative 15 displayed υNH (br.) at 3242 cm− 1 along with υCO at 1736 and 1676 cm− 1. Moreover, the existence of one singlet signal for CH2 protons of CH2Cl at δ 4.25 ppm together with one singlet signal for one NH proton at δ 11.12 ppm beside the aromatic protons in the 1H NMR spectrum asserts the structure 15.
Reaction of hydrazide 10 with one C-donors.
Meanwhile, acylation of the hydrazide 10 with acetic anhydride at room temperature provided the N-acetyl hydrazide derivative 16 as yellow crystals, while carrying out the reaction under reflux conditions, was simply cyclized into the pyrrolone derivative 17. Compound 17 was also obtained via refluxing compound 16 in a mixture of CH3COOH/HCl (1:1) (Fig. 7). The structures of the products 16 and 17 were verified by spectroscopic data (cf. Experimental).
Biological activity
Insecticidal activity
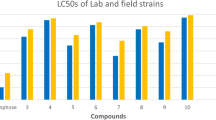
The potency of pyrrolone derivatives and chlorfenapyr was assessed at different concentrations against the field and lab strains of the third larval instar of C. pipiens and the mortalities were recorded 72 h post-treatment. The LC values and other criteria were estimated and summarized in Tables 1 and 2. In the lab strain, the pyrrolone derivatives showed various activities. Most of the compounds demonstrated significant activities, which exhibited 6.77, 6.24, 5.84, 5.60, 5.32, 4.44 and 3.74 folds than 11, respectively, against C. pipiens larvae, the activity order is as follows: 17 > 9 > 15 > chlorfenapyr > 10 > 16 > 5. Hence 17 and 9 were the most effective compounds with LC50 of 21.05 and 22.81 µg/mL, respectively, while 11 was the least active with LC50 of 142.56 µg/mL. Also, the larvae were highly susceptible to 15, chlorfenapyr, 10, 16 and 5 with LC50 values of 24.39, 25.43, 26.76, 32.09 and 38.07 µg/mL, respectively, against C. pipiens larvae. It was noted that compounds 17, 9 and 15 were more toxic than chlorfenapyr. On the other hand, some compounds showed low potency, and their order as follows: 13 < 12 < 7 < 14 with LC50 values of 122.49, 107.53, 97.74 and 88.65 µg/mL, respectively. In comparison, other compounds showed moderate activities as follows: 2 > 4 > 8 > 6 and 3 with LC50 of 44.96, 51.94, 59.87, 67.70, and 79.30 µg/mL, respectively. Moreover, the larval sensitivity to 2, 4, 8, 6, and 3 were 3.17, 2.74, 2.38, 2.10 and 1.79 times than 11, Table 1. The chi-square values of all tested compounds were significant at P < 0.05. The slope values of tested compounds were low, revealing the homogeneity of the tested strain in its response to the pyrrolone derivatives and chlorfenapyr.
After 72 h of exposure, the activity of the tested compounds increased against the field strain of C. pipiens larvae than the lab strain. 17 and 15 were the most toxic compounds followed by 9 > 10 > chlorfenapyr > 16 > 2 with LC50 of 9.87, 10.76, 11.52, 12.68, 14.03, 15.32 and 18.37 µg/mL, respectively. It was noticed that 17, 15, 9 and 10 were more potent than chlorfenapyr against the field strain of C. pipiens larvae. On the other hand, 13 was the least potent compound against the field strain with LC50 of 76.61 µg/mL. So, the most efficient compounds 17, 15, 9, 10, 16 and 2 showed high relative toxicities of 7.76, 7.11, 6.65, 6.04, 5.00 and 4.17 folds than 13, respectively. On the other hand, 13, 11, 12 and 14 were the least efficient compounds with LC50 of 76.61, 64.83, 56.28 and 50.65 µg/mL, respectively Table 2. While 5, 4, 6, 8, 3 and 7 exhibited moderate activity with LC50 of 21.94, 25.65, 29.84, 34.10, 40.46 and 45.61 µg/mL, respectively.
Biochemical analysis
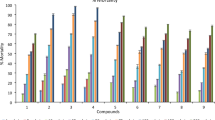
The results of cytochrome P-450 monooxygenase analysis against C. pipiens larvae indicated that the activities were significantly influenced (P < 0.01) after treatment with 17, 9, 15, 10, 16, 5 and chlorfenapyr compared with untreated for lab strain and 17, 15, 9, 10, 16, 2 and chlorfenapyr for field strain.
In lab strain, the cytochrome P-450 activity significantly increased after exposure to all tested compounds, where 17 and 9 greatly enhanced the cytochrome P-450 activity relative to untreated, followed by chlorfenapyr and 15 which showed similar effects. While 16 and 5 showed the lowest changes in cytochrome P-450 activity as shown in Fig. 8.
Similarly, in field strain, the tested compounds significantly increased (P < 0.01) the activity in the treated larvae relative to untreated. Maximum significant elevation of cytochrome P-450 activity was achieved by 17. Both 15 and 9 showed similar influence of enzyme activity with a remarkable increase than untreated. As well, the treated larvae exhibited convergent elevation rates post-treatment with 16 and 10 compared to untreated. Also, a significant enhancement in enzyme activity was detected post-treatment with chlorfenapyr and 2, respectively, relative to untreated Fig. 9.
Effect of most toxic pyrrolone derivatives and chlorfenapyr on cytochrome P-450 monooxygenase activity of treated and untreated lab strain of C. pipiens larvae. Means with different letters are significantly different (P > 0.01) Duncan`s multiple range test.
Effect of most toxic pyrrolone derivatives and chlorfenapyr on cytochrome P-450 monooxygenase activity of treated and untreated field strain of C. pipiens larvae. Means with different letters are significantly different (P > 0.01) Duncan`s multiple range test.
Discussion
Pyrrole insecticides impair mitochondrial activity and have been widely employed in urban and agricultural pest control. Chlorfenapyr is considered one of a new generation of malaria-preventative insecticides40. This pyrrole insecticide is a highly efficient compound with high residual toxicity, making it an attractive option to reduce insect resistance41.
Chlorfenapyr is a pro-insecticide activated and becomes toxic by cytochrome P450s, resulting in improved activity. Chlorfenapyr metabolized by P450s of An. Gambiae and Ae. aegyptia and a single NADPH was produced. Tralopyril is the main toxic metabolite produced by removing the N-dealkylated group, through P450-mediated oxidation42,43,44. Tralopyril mode of action is a mitochondrial electron transport uncoupler that disrupts the proton gradient across the mitochondrial membranes causing damage to mitochondria [44]. Moreover, it obstructs the production of ATP through oxidative phosphorylation resulting in energy production disorders that ultimately lead to insect death41,42.
Unexpectedly, the toxicity of tested compounds revealed higher activity rates in the field than the lab strain of C. pipiens larvae which agrees with Oxborough, et al.,47 who stated that the application of chlorfenapyr in field trials induced high mortality rates of host-seeking mosquitoes while the in-laboratory bioassay, it exhibited slow-acting mortality levels. Two hypotheses could explain the obtained results, firstly the pyrrole’s mode of action differs significantly from the neurotoxic insecticides, which makes it improbable to show cross-resistance to currently used insecticides48. Secondly, metabolic resistance is one of the primary mechanisms of insecticide resistance detected in insects. Neuro-insecticides are used broadly for mosquito larval control or agricultural application and cytochrome P450 monooxygenase production could be increased as one of the metabolic enzymes that break down these insecticides. This detoxification or metabolism process could activate pro-insecticides such as pyrroles and chlorfenapyr in the field rather than the lab strain49. Moreover, this potency may increase in nature, mosquito larvae will encounter pyrroles while actively swimming to the water surface to obtain oxygen or search for food when their metabolic enzymes are upregulated. Consequently, it might be exploited to control the resistant insect populations. Generally, a unique mode of action of chlorfenapyr leads to the absence of cross-resistance to conventional insecticides rendering it a potential candidate for inclusion in vector control efforts, especially in areas with high or emergent resistance to other insecticide classes50. Chlorfenapyr had a significant insecticidal potency against all developmental stages of Bradysia odoriphaga and there were no significant variations in sensitivity between the laboratory and field populations51. Raghavendra, et al.,52 observed the absence of cross-resistance against chlorfenapyr in multiple insecticides- resistant strains of An. stephensi was reared in the laboratory beside field-collected multiple insecticides-resistant An. culicifacies. Additionally, Anopheline larvae collected from several locations in rural and urban areas of Cameroon were more susceptible to chlorfenapyr than emerging female adults16.
Given that, the activation process depends mainly on insect metabolism. It may take some time to start, but as soon as the conversion begins the insect’s respiration increases, which influences additional conversion of more parent compounds53. So, the mortality results were recorded 72 h post-treatment because the toxic efficiency of chlorfenapyr and pyrrole derivatives was slower than that of traditional neurotoxic insecticides. Most mortalities occurred after 24 h to 72 h19, Moreover, the chlorfenapyr toxicity increased gradually with time against Aedes aegypti larvae, low mortality was observed after 24-h exposure and increased at 72 h of exposure54. Chlorfenapyr exerts physiological effects contributing to the significant and extended acute and delayed mortalities detected. Its enzymatic transformation could be quite variable and slow. The uncoupling-oxidative phosphorylation could be affected by cuticular penetrations, food-seeking behaviour, degree of metabolic activity, physical movement of targeted insects, temperature and concentrations of pyrroles55. The sulfanyl 5 H-dihydropyrrole derivatives showed good insecticidal activity against larvae of Ephestia kuehniella and Tribolium confusum. Their residual potencies were influenced by temperature and relative humidity56. Six new pyrrole derivatives were revealed as grain protectants against T. confusum, S. oryzae, E. kuehniella and R. dominica. The insecticidal efficiency of tested derivatives was moderate and slow acting, where they showed the highest mortality rates 21 days post-treatment. Additionally, the adult population was suppressed, and progeny production was reduced41.
Two series of 2-phenylpyrroles exerted moderate larvicidal potency against oriental armyworms and 6b indicated a good acaricidal potency, so 4-cyanopyrrole-2-carboxylate derivatives were promising to be lead compounds for novel pesticides57.
The biochemical analyses revealed that the cytochrome P-450 monooxygenase activities were significantly increased in treated larvae of lab and field strains relative to untreated. The populations of insecticide-resistant mosquitoes showed overexpression of P450s that may promote the activation of chlorfenapyr, resulting in improved susceptibility to this insecticide. Furthermore, the pyrethroid-resistant pests such as the tobacco budworm and the cattle horn fly were more susceptible to chlorfenapyr44. The increased toxicity of tested compounds caused by increased metabolic activity of cytochrome P-450 leads to the activation of the toxins and increased mortalities of mosquito larvae55. Chlorfenapyr was the most toxic tested insecticide against house fly larvae, followed by abamectin and lambda-cyhalothrin, respectively. Chlorfenapyr greatly increased the cytochrome P450 activity relative to other insecticides58.
Structure-activity relationship
The synthesized pyrrolone derivatives resemble chlorfenapyr in the presence of a chlorobenzene ring in the same position at the pyrrole ring, this lipophilic group strengthens the acaricidal and insecticidal activity [46]. In general, the compounds with short linear alkyl or alkoxy substitutions (small bulking moieties) showed substantially high contact or systemic insecticidal effectiveness, while those with longer or branched alkyl insertions (large bulking moieties) tended to decline the efficacy59. The insertion of small moieties at the NH position to pyrrolones showed high activity as acetamido group in 17 and chloroacetamido group in 15, where the acetamide derivatives were widely used as insecticidal agents60,61. Also, the insertion of thiourea group in 9 and acetyl, 4-oxobutane hydrazide groups in 16 enhanced the derivatives potency. These results were interpreted by Huang, et al.,46 who found that the alkyl or alkoxy substitutions are the most suitable at position N of chlorfenapyr, additionally weak acidic amino groups are desired for the uncoupling action.
Although the good toxicity of 16 with N-acetyl hydrazide moiety, the cyclization of this moiety greatly enhanced the activity of compound 17 which showed high toxicity to both field and lab strains. These results are similar to the study of Abdelhamid, et al.,62 which revealed that the pyrrole compounds containing the acetohydrazide group showed insecticidal toxicity against the 2nd and 4th larval instars of S. littoralis (Boisd.). The insertion of chloroacetylchloride to hydrazide 10 and the formation of pyrrolone with N-acetylchloride in 15 remarkably improved the toxicity to C. pipiens larvae where the presence of electron-withdrawing Cl promotes the binding with the target site63. The insertion of thiosemicarbazide into pyrrolone significantly increased the activity of 9 due to the presence of electron-withdrawing S increasing the positivity of the amino group64.
On the other hand, the compounds with aromatic and long insertions showed moderate to low toxicity according to the bulking of inserted substitutions as methoxybenzylidene in 13, imino indolinone in 12, benzyl in 7 and amino methoxybenzylidene in 14 which showed the least toxicity. These results are consistent with Zhao, et al.,65 who observed that the aromatic-substituted compounds exerted lower insecticidal potencies against oriental armyworms than short alkyl-substituted compounds. As well as the conversion of the pyrrolone ring to the pyridazine ring in 11 completely changed its toxic characteristics and diminished its potency. All these observations approved the hypothesis of the rational design study.
Experimental
Chemistry
All melting points were measured on a Griffin and George melting-point apparatus (Griffin & Georgy Ltd., Wembley, Middlesex, UK) and are uncorrected. IR spectra were recorded on Pye Unicam SP1200 spectrophotometer (Pye Unicam Ltd., Cambridge, UK) by using the KBr wafer technique. 1H-NMR spectra were determined on a Varian Gemini 300 MHz on Bruker Avance III using tetramethylsilane as an internal standard (chemical shifts in δ scale), while 13C NMR spectra were run at 75 MHz. Elemental analyses were carried out at the Microanalytical Unit, Faculty of Science, Ain Shams University, using a Perkin-Elmer 2400 CHN elemental analyzer (Waltham, MA), and satisfactory analytical data (± 0.4) were obtained for all compounds. The homogeneity of the synthesized compounds was controlled by thin layer chromatography (TLC), using aluminum sheet silica gel F254 (Merck). The structures of the compounds in (Figs. 1, 2, 3, 4, 5, 6 and 7) were drawn in Chem Office Ultra 2004 software, version 8, https://chemoffice-ultra-2004.software.informer.com.
(E)-5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)furan-2(3 H)-one 2:
A mixture of 4-(4-chlorophenyl)-4-oxobutanoic acid 1 (2.12 g, 10 mmol) and 1-naphthaldehyde (1.3 mL, 10 mmol) was fused in the presence of sodium acetate (0.8 g, 10 mmol) and acetic anhydride (2 mL) on a hot plate then heated on a water bath for 1 h. The crude material was filtered off, washed with ethanol and then recrystallized from dioxane to give 2 as orange crystals; mp 258–260 °C, yield 60%. IR (υ/cm− 1): 3049 (aryl-H), 1771 (C = Olactone), 1618 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 7.49 (s, 1H, CH Furanone), 7.55–8.23 (m, 12 H, 11 Ar-H + CH=). Anal. calcd. for C21H13ClO2 (332.78): C, 75.79; H, 3.94; Cl, 10.65. Found: C, 75.66; H, 3.74; Cl, 10.73.
(E)-5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)-1,3-dihydro-2 H-pyrrol-2-one 3:
A solution of furanone derivative 2 (3.3 g, 10 mmol) in formamide (10 mL) was heated under reflux for 1 h. The obtained solid was collected, washed with diethyl ether, and then recrystallized from EtOH to give 3 as red-orange crystals; mp 294–296 °C, yield 65%. IR (υ/cm1): 3154 (NH), 1695 (C = Olactam), 1614 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 6.80 (s, 1H, CHpyrrolone), 7.50–7.65 (m, 5 H, Ar-H), 7.86–8.02 (m, 6 H, Ar-H), 8.18 (d, 1H, Ar-H, J = 8.4 Hz), 10.71 (s, 1H, NH, exchangeable with D2O). 13C-NMR (DMSO-d6) δ (ppm): 98.1, 123.3, 125.9, 126(2), 127(2), 128(4), 129.9, 131(2), 132.8, 133.3, 134.2, 145.1, 170.4. Anal. calcd. for C21H14ClNO (331.80): C, 76.02; H, 4.25; Cl, 10.68; N, 4.22. Found: C, 76.21; H, 4.16; Cl, 10.55; N, 4.29.
(E)-4-(4-chlorophenyl)-N-cyclohexyl-2-(naphthalen-1-ylmethylene)-4-oxobutanamide 4:
Cyclohexyl amine (1 mL, 10 mmol) was added dropwise to a solution of furanone derivative 2 (3.3 g, 10 mmol) in dioxane (20 mL) at ambient temperature and then refluxed for 10 h. After cooling, the deposited solid was collected by filtration, dried and then recrystallize from EtOH/dioxane to give 4 as white crystals; mp 266–268 °C, yield 75%. IR (υ/cm− 1): 3161 (NH), 2938, 2848 (C-H, aliphatic), 1669 (C = O), 1640 (C = C).1H-NMR (DMSO-d6) δ (ppm): 0.85–1.81 (m, 10Hcyclohexyl), 3.25 (s, 2 H, CH2), 3.71 (m, 1H, NH-CHcyclohexyl), 6.81 (br s, 1H, NH, exchangeable with D2O), 7.41–8.15 (m, 11 H, Ar-H + 1Holefinic).13C-NMR (DMSO-d6) δ (ppm): 24.3, 25(3), 29(2), 31.5, 32.5, 38.7, 53.0, 123.5, 124(2), 125(2), 126(3), 127(2), 128(4), 130(2), 131(3), 132.4, 133(3), 134.4, 135.2, 138.1, 143.2, 166(2), 196.7. Anal. calcd. for C27H26ClNO2 (431.96): C, 75.08; H, 6.07; Cl, 8.21; N, 3.24. Found: C, 75.18; H, 6.00; Cl, 8.39; N, 3.16.
(E)-N-benzyl-4-(4-chlorophenyl)-2-(naphthalen-1-ylmethylene)-4-oxobutanamide 5:
A mixture of furanone derivative 2 (3.3 g, 10 mmol) and benzyl amine (1.1 mL, 10 mmol) in dioxane (20 mL) was heated under reflux for 12 h. The obtained oil after concentration of the reaction mixture was solidified by boiling with ethanol. The precipitated solid was filtered off, dried and recrystallized from dioxane to give 5 as white crystals; mp 240–242 °C, yield 60%. IR (υ/cm1): 3315 (br NH), 3051 (C-H, aromatic), 2925, 2851 (C-H, aliphatic), 1679 (C = O), 1649 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 3.35 (s, 2 H, =C-CH2-CO), 4.26, 4.36 (d, d, 2 H, NH-CH2-, J = 15.3 Hz), 6.92 (s, 1H, NH, exchangeable with D2O), 7.18–8.16 (m, 16 H, Ar-H + 1Holefinic). 13C-NMR (DMSO-d6) δ (ppm): 38(2), 43(2), 123.4, 125(2), 126(4), 127(2), 128(2), 129.0, 131(2), 132(2), 133.2, 138.1, 142.1, 167.7. Anal. calcd. for C28H22ClNO2 (439.94): C, 76.44; H, 5.04; Cl, 8.06; N, 3.18. Found: C, 76.52; H, 5.17; Cl, 8.21; N, 3.11.
Synthesis of compounds 6 and 7
A mixture of acetic acid (10 mL) and concentrated hydrochloric acid (1 mL) was added to compound 4 and/or 5 (10 mmol) then refluxed for 15 min. The deposited solid while heating was collected, washed with diethyl ether, dried and recrystallized from appropriate solvent to give 6 and 7, respectively.
(E)-5-(4-chlorophenyl)-1-cyclohexyl-3-(naphthalen-1-ylmethylene)-1,3-dihydro-2 H-pyrrol-2-one 6:
Recrystallized from EtOH as orange crystals; mp 208–210 °C, yield 70%. IR (υ/cm− 1): 3050 (C-H, aromatic), 2954, 2926, 2864 (C-H, aliphatic), 1685 (C = O), 1608 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 1.11 (m, 3 H, CHcyclohexyl), 1.56–1.75 (m, 5 H, CHcyclohexyl), 2.26 (m, 2 H, CH2cyclohexyl), 3.4 (m, 1H, NH-CHcyclohexyl), 6.20 (s, 1H pyrrolone ring), 7.50–7.66 (m, 8 H, Ar-H), 7.89–8.17 (m, 4 H, 3 Ar-H + CH=), 13C-NMR (DMSO-d6) δ (ppm): 24.9, 25.7, 28.6, 29(2), 63.4, 101.2, 120(2), 123.5, 126(2), 127(3), 128(2), 129(3), 131.6, 133.4, 134.3, 143.9, 149.7, 169.2. Anal. calcd. for C27H24ClNO (413.95): C, 78.34; H, 5.84; Cl, 8.56; N, 3.38. Found: C, 78.23; H, 5.92; Cl, 8.70; N, 3.18.
(E)-1-benzyl-5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)-1,3-dihydro-2 H-pyrrol-2-one 7:
Recrystallized from EtOH as orange crystals; mp 220–222 °C, yield 80%. IR (υ/cm− 1): 3057 (C-H, aromatic), 2911 (C-H, aliphatic) 1701 (C = O pyrrolone). 1H-NMR (DMSO-d6) δ (ppm): 4.89 (s, 2 H, CH2-Ph), 6.41 (s, 1H, =CH pyrrolone), 7.02–7.05 (m, 2 H, Ar-H), 7.17–7.29 (m, 3 H, Ar-H), 7.34–7.50 (m, 4 H, Ar-H), 7.57–7.67 (m, 3 H, Ar-H), 7.97–8.03 (m, 3 H, Ar-H), 8.13 (s, 1H, CHolefinic), 8.20 (d, 1H, Ar-H, J = 8.1 Hz).13C-NMR (DMSO-d6) δ (ppm): 43.8, 101.3, 120.7, 123.4, 125.9, 126.4, 127(2), 128(4), 129.4, 130.2, 131(2), 134.2, 137.5, 148.3, 169.0. Anal. calcd. for C28H20ClNO (421.92): C, 79.71; H, 4.78; Cl, 8.40; N, 3.32. Found: C, 79.80; H, 4.69; Cl, 8.53; N, 3.12.
Ethyl (E)-(5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)-2-oxo-2,3-dihydro-1 H-pyrrol-1-yl)carbamate 8:
A mixture of furanone derivative 2 (3.3 g, 10 mmol), ethyl carbazate (1 g, 10 mmol) in dioxane (20 mL) and glacial acetic acid (10 mL) was heated under reflux for 15 h. After evaporation of the excess solvent, the precipitated solid was collected by filtration, washed with diethyl ether, dried and then recrystallized from EtOH to give 8 as yellow crystals; mp 184–186 °C, yield 67%. IR (υ/cm1): 3260 (NH), 1736 (C = Oester), 1704 (C = O pyrrolone), 1621 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 1.17 (t, 3 H, CH2CH3), 4.07 (q, 2 H, CH2CH3), 6.60 (s, 1H, CHpyrrolone), 7.53–7.60 (m, 7 H, Ar-H), 8.00-8.18 (m, 5 H, 4 Ar-H + 1CH=), 10.15 (br s, 1H, NH, exchangeable with D2O). 13C-NMR (DMSO-d6) δ (ppm): 14.3, 61.4, 99.8, 123.4, 125.9, 126.5, 127(2), 128(3), 129(2), 130.4, 131(2), 133.3, 134.5, 146.8, 155.4, 168.4. Anal. calcd. for C24H19ClN2O3 (418.88): C, 68.82; H, 4.57; Cl, 8.46; N, 6.69. Found: C, 68.74; H, 4.42; Cl, 8.39; N, 6.79.
(E)-1-(5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)-2-oxo-2,3-dihydro-1 H-pyrrol-1-yl)thiourea 9:
A mixture of furanone derivative 2 (3.3 g, 10 mmol) and thiosemicarbazide (0.9 g, 10 mmol) in dioxane (20 mL) and glacial acetic acid (10 mL) was refluxed for 14 h. After evaporation of the excess solvent, the precipitated solid was collected by filtration, washed with diethyl ether, dried and then recrystallized from EtOH to give 9 as red-orange crystals; mp 235–237 °C, yield 60%. IR (υ/cm1): 3450, 3239, 3191 (NH2, NH), 1701 (C = O pyrrolone). 1H-NMR (DMSO-d6) δ (ppm): 6.61 (s, 1H CHpyrrolone), 7.51–8.30 (m, 11 H, Ar-H + 1H, CHolefinic+ 2 H, NH2, exchangeable with D2O), 10.66 (br s, 1H, NH, exchangeable with D2O). 13C-NMR (DMSO-d6) δ (ppm): 100.5, 123.4, 125.9, 126.5, 127.3, 128(4), 129.0, 131(2), 132.7, 133.3, 134.25, 134.4, 146.0, 147.2, 168.3, 182.5. Anal. calcd. For C22H16ClN3OS (405.90): C, 65.10; H, 3.97; Cl, 8.73; N, 10.35; S, 7.90. Found: C, 65.16; H, 3.86; Cl, 8.56; N, 10.49; S, 8.12.
(E)-4-(4-Chlorophenyl)-2-(naphthalen-1-ylmethylene)-4-oxobutanehydrazide 10:
Hydrazine hydrate (1 mL, 20 mmol) was added dropwise to a stirred solution of furanone derivative 2 (3.3 g, 10 mmol) in dioxane (30 mL) at ambient temperature. The reaction mixture was further stirred for 1 h, then poured into water. The deposited solid was filtered off, dried and recrystallized from EtOH to give 10 as white crystals; mp 178–180 °C, yield 72%. IR (υ/cm1): 3312, 3210, 3195 (NH2, NH), 1691 (C = O). 1H-NMR (DMSO-d6) δ (ppm): 3.16 (s, 2 H, CH2), 4.52 (br s, 2 H, NH2, exchangeable with D2O), 6.75 (s, 1H, NH, exchangeable with D2O), 7.40–8.11 (m, 11 H, Ar-H + 1Holefinic). 13C-NMR (DMSO-d6) δ (ppm): 42.6, 123.4, 125(2), 126(3), 127.7, 128(3), 130.8, 131(2), 132.2, 133.2, 142.4, 166.2. Anal. calcd. for C21H17ClN2O2 (364.83): C, 69.14; H, 4.70; Cl, 9.72; N, 7.68. Found: C, 69.23; H, 4.79; Cl, 9.85; N, 7.49.
6-(4-Chlorophenyl)-4-(naphthalen-1-ylmethyl)pyridazin-3(2 H)-one 11
A solution of hydrazide derivative 10 (3.6 g, 10 mmol) in sodium hydroxide (10%, 30 mL) was heated under reflux for 10 h. After cooling, the reaction mixture was acidified with cold dilute hydrochloric acid and the deposited solid was collected then recrystallized from EtOH/dioxane to give 11 as white crystals; mp 290–292 °C, yield 65%. IR (υ/cm1): 3200, 3129 (NH), 1653 (C = O), 1604 (C = N). 1H-NMR (DMSO-d6) δ (ppm): 4.32 (s, 2 H, CH2), 7.39–8.07 (m, 11 H, Ar-H + 1H pyridazine), 13.32 (br. s, 1H, NH, exchangeable with D2O). 13C-NMR (DMSO-d6) δ (ppm): 31.7, 123.9, 125(2), 126.4, 127(3), 128(2), 131.5, 133(3), 134.1, 142(2), 160.7. Anal. calcd. for C21H15ClN2O (346.81): C, 72.73; H, 4.36; Cl, 10.22; N, 8.08. Found: C, 72.59; H, 4.19; Cl, 10.33; N, 7.95.
(E)-3-(((E)-5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)-2-oxo-2,3-dihydro-1 H-pyrrol-1-yl)imino)indolin-2-one 12:
A mixture of the hydrazide 10 (3.6 g, 10 mmol) and isatin (1.4 g, 10 mmol) in absolute ethanol (20 mL) containing glacial acetic acid (10 mL) was refluxed for 2 h. The obtained solid while heating was filtered off, dried and then recrystallized from dioxane to give 12 as orange crystals; mp 292–294 °C, yield 60%. IR (υ/cm1): 3165 (NH), 1723 (C = O). 1H-NMR (DMSO-d6) δ (ppm): 6.91–8.22 (m, 17 H, 1H pyrrolone ring + 1Holefinic + 15Ar-H), 11.04 (s, 1H, NH, exchangeable with D2O). 13C-NMR (DMSO-d6) δ (ppm): 102.5, 103.5, 104.2, 111.0, 116.8, 117.7, 120(2), 122(2), 123(2), 126.1, 127.5, 128(2), 129(3), 130.9, 131(2), 133.4. 134(2) 140.7, 144.2, 145.3, 147(2), 152.4, 153.0, 155.6, 163.5, 164.1. Anal. calcd. for C29H18ClN3O2 (475.93): C, 73.19; H, 3.81; Cl, 7.45; N, 8.83. Found: C, 73.06; H, 3.59; Cl, 7.53; N, 8.77.
(E)-4-(4-Chlorophenyl)-N’-((Z)-4-methoxybenzylidene)-2-(naphthalen-1-ylmethylene)-4-oxobutanehydrazide 13:
A mixture of the hydrazide 10 (3.6 g, 10 mmol) and p-methoxybenzaldehyde (1.2 mL, 10 mmol) in absolute ethanol (20 mL) was refluxed for 6 h. The deposited solid on hot was collected, dried and then recrystallized from dioxane to give 13 as pale-yellow crystals; mp 222–224 °C, yield 70%. IR (υ/cm1): 3167 (NH), 3075 (C-H, aromatic), 2934, 2836 (C-H, aliphatic), 1677 (C = O), 1638 (C = N), 1611 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 3.15 (s, 2 H, CH2), 3.76 (s, 3 H, OCH3), 6.78 (s, 1H, NH, exchangeable with D2O), 6.96–8.12 (m, 15 H, Ar-H), 9.06 (s, 1H, N = CH). 13C-NMR (DMSO-d6) δ (ppm): 43.2, 55.4, 112.1, 123.5, 125.7, 126.4, 127(2), 128(4), 129(2), 130.5, 131(2), 132.5, 133.3, 140.5, 142.6, 145.9, 153.8, 161.4, 164.4. Anal. calcd. for C29H23ClN2O3 (482.96): C, 72.12; H, 4.80; Cl, 7.34; N, 5.80. Found: C, 72.04; H, 4.64; Cl, 7.48; N, 5.87.
(E)-5-(4-Chlorophenyl)-1-(((E)-4-methoxybenzylidene)amino)-3-(naphthalen-1-ylmethylene)-1,3-dihydro-2 H-pyrrol-2-one 14:
Method A: A mixture of the hydrazide 10 (3.6 g, 10 mmol) and p-methoxybenzaldehyde (1.2 ml, 10 mmol) in absolute ethanol (20 mL) in the presence of glacial acetic acid (10 mL) was heated under reflux for 30 min. The precipitated solid while heating was filtered off, dried and recrystallized from EtOH to give 14 as orange crystals; mp 242–244 °C, yield 82%. IR (υ/cm1): 3047 (C-H, aromatic), 2932, 2836 (C-H, aliphatic), 1703 (C = O), 1622 (C = N), 1608 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 3.81 (s, 3 H, CH3), 6.68 (s, 1H pyrrolone ring), 7.05–8.20 (m, 15 H, Ar-H + 1Holefinic), 9.39 (s, 1H, N = CH). Anal. calcd. for C29H21ClN2O2 (464.95): C, 74.92; H, 4.55; Cl, 7.62; N, 6.03. Found: C, 74.78; H, 4.51; Cl, 7.83; N, 6.13.
Method B: A solution of compound 13 (4.8 g, 10 mmol) in glacial acetic acid (20 mL) containing concentrated hydrochloric acid (1 mL) was heated under reflux for 30 min. The precipitated solid while heating was filtered off, dried and recrystallized from EtOH to give 14 as orange crystals; yield 85%.
(E)-2-Chloro-N-(5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)-2-oxo-2,3-dihydro-1H-pyrrol
To a solution of the hydrazide 10 (3.6 g, 10 mmol) in ethanol (20 mL), chloroacetyl chloride (0.8 mL, 10 mmol) was added dropwise and then refluxed on water-bath for 2 h. The reaction mixture was then poured into water and the obtained solid was collected, dried and recrystallized from petroleum ether 80–100 °C to give 15 as red crystals; mp 218–220 °C, yield 64%. IR (υ/cm1): 3242 (NH), 3043 (C-H, aromatic), 1736 (C = Opyrrolone ring), 1676 (C = Oamide), 1612 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 4.25 (s, 2 H, CH2Cl), 6.64 (s, 1Hpyrrolone ring), 7.50–8.23 (m, 11 H, Ar-H + 1Holefinic), 11.12 (s, 1H, NH, exchangeable with D2O).13C-NMR (DMSO-d6) δ (ppm): 40.4, 100.1, 123.4, 125.8, 126.5, 127(2), 128 (3), 129.6, 130.4, 131(2), 133.3, 134.5, 146.7, 165.8, 167.9. Anal. calcd. for C23H16Cl2N2O2 (423.29): C, 65.26; H, 3.81; Cl, 16.75; N, 6.62. Found: C, 65.35; H, 3.70; Cl, 16.68; N, 6.41.
(E)-N’-Acetyl-4-(4-chlorophenyl)-2-(naphthalen-1-ylmethylene)-4-oxobutanehydrazide 16:
The hydrazide derivative 10 (3.6 g, 10 mmol) in acetic anhydride (30 mL) was stirred at room temperature for 3 h. The separated yellow solid was filtered off, wash with diethyl ether, dried and then recrystallized from EtOH to give 16 as yellow crystals; mp 230–232 °C, yield 65%. IR (υ/cm1): 3276, 3189 (NH), 3022 (C-H, aromatic), 2912, 2808 (C-H, aliphatic), 1713, 1687, 1667 (C = O), 1644 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 1.85 (s, 3 H, CH3), 3.35 (s, 2 H, CH2), 6.88 (s, 1H, NH-NH-COCH3, exchangeable with D2O), 7.38–8.13 (m, 11 H, Ar-H + 1Holefinic), 9.86 (s, 1H, NH-NH-COCH3, exchangeable with D2O). 13C-NMR (DMSO-d6) δ (ppm): 20.5, 42.7, 123.4, 125.5, 126 (3), 127 (2), 128(2), 129(2), 131(2), 132.5, 133.2, 141.2, 165.3, 168.6. Anal. calcd. for C23H19ClN2O3 (406.87): C, 67.90; H, 4.71; Cl, 8.71; N, 6.89. Found: C, 67.96; H, 4.62; Cl, 8.86; N, 7.06.
(E)-N-(5-(4-chlorophenyl)-3-(naphthalen-1-ylmethylene)-2-oxo-2,3-dihydro-1 H-pyrrol-1-yl)acetamide 17:
Method A: The hydrazide derivative 10 (3.6 gm, 10 mmol) in acetic anhydride (30 mL) was heated under reflux for 30 min. The precipitated solid while heating was filtered off, dried and recrystallized from EtOH to give 17 as yellow crystals; mp 264–266 °C, yield 68%. IR (υ/cm1): 3230 (NH), 3018 (C-H, aromatic), 2962, 2885 (C-H, aliphatic), 1728 (C = Opyrrolone ring), 1674 (C = Oamide), 1655 (C = C). 1H-NMR (DMSO-d6) δ (ppm): 1.91 (s, 3 H, CH3), 6.60 (s, 1Hpyrrolone ring), 7.42–8.08 (m, 11 H, Ar-H + 1Holefinic), 8.16 (d, 1H, Ar-H, J = 8.1 Hz), 10.69 (s, 1H, NH, exchangeable with D2O). 13C-NMR (DMSO-d6) δ (ppm): 20.3, 99.8, 123(2), 125(2), 126(3), 127.4, 128(5), 129(2), 130.5, 131(2), 133(3), 134(3), 142(2), 147.3, 160.7, 168.3, 169.0. Anal. calcd. for C23H17ClN2O2 (388.85): C, 71.04; H, 4.41; Cl, 9.12; N, 7.20. Found: C, 70.92; H, 4.32; Cl, 8.98; N, 7.28.
Method B: A solution of compound 16 (4 g, 10 mmol) in glacial acetic acid (20 mL) containing concentrated hydrochloric acid (1 mL) was heated under reflux for 30 min. The precipitated solid while heating was filtered off, dried and recrystallized from EtOH to give 14 as orange crystals; yield 72%.
Biological evaluation
Insect rearing
A field population was collected from breeding sites in Abou Rawash, Giza, Egypt. The laboratory strain of C. pipiens larvae used in larvicidal activity was reared at the insectary of the Entomology Department, Faculty of Science, Ain Shams University, Cairo, Egypt, for several generations. This strain was reared in optimized conditions of temperature (27 ± 2 °C), relative humidity (75 ± 5%) and light and dark circadian (14 h: 10 h) according to Kufman, et al.,66 with some modifications. The adults were reared in a wooden cage (40 × 40 × 40 cm) for feeding, mating and oviposition so it was provided with deionized water-filled glass jars for adult emergence and then egg laying. Adults were fed on sugar solution (10%) and only females were fed on a blood meal from live pigeons for egg production. The egg rafts were transferred to enamel containers for larval hatching. The larvae were fed on a freshly prepared larval diet (biscuits dried: yeasts (3: 1))67. The pupae were collected in emergency jars daily.
Larvicidal activity
The potency of sixteen tested pyrrolone derivatives and chlorfenapyr as reference insecticide was assessed against the C. pipiens (third larval instar) following68 with some modifications. The stock solutions were prepared by dissolving the synthesized compounds in DMSO, while the chlorfenapyr (Challenger 24% EC) was obtained from BASF Chemicals Co. Egypt and dissolved in deionized water. Six concentrations were prepared in deionized water and Triton X-100 for synthesized compounds and in deionized water only for chlorfenapyr as follows (10, 25, 50, 100, 200 and 300 µg/mL) for lab strain, and (5, 10, 25, 50, 100 and 200 µg/mL) for field strain. The control was conducted by application of solvents only without exposure to tested compounds. Each concentration of the tested compounds, reference insecticide and control were replicated three times. A batch of 20 early third larval instar, was transferred to each prepared concentration (80 mL)69. The treatments and control were examined after 72 h and the mortality rates were estimated.
Biochemical analysis
To evaluate the biochemical effects of pyrrolone derivatives, the 3rd instar of C. pipiens larvae was treated with LC50 of the six most effective pyrrolone derivatives for 72 h. The treated and untreated larvae were kept at freezing conditions at −20˚C. Then the larval bodies were homogenized in a chilled glass Teflon tissue grinder for 5 min, according to70. Homogenates were centrifuged at 8000 r.p.m in a refrigerated centrifuge for 15 min. The supernatants were stored at 5 ˚C until used for biochemical analysis71. For quantitative determination of the cytochrome P-450 monooxygenase activity, the p-nitro anisole o-demethylation was used according to the method of Hansen and Hodgson72 with some modifications.
Statistical analysis
By using LdP-Line© package software (Ehabsoft, Egypt), different lethal concentrations were determined at the 95% fiducial limits, in addition, slope ± standard error and Chi-square test were calculated for the goodness of fit according to Finney73. The relative potency was calculated to compare the effectiveness of tested compounds74. The biochemical analysis results were tested by one-way analysis of variance (ANOVA) by using the CoStat system for Windows, (Version 6.311, (CoHort software, Berkeley, CA 94701 https://www.cohortsoftware.com/costat.html)). The mean values were compared by Duncan’s multiple range test75 ANOVA statistics were significant (P < 0.01).
Conclusion
A series of new pyrrolone and pyridazinone derivatives bearing naphthalene moiety were synthesized. The chemical structures of the synthesized compounds were assured by spectroscopic techniques. All these compounds were evaluated against field and laboratory strains of Culex pipiens larvae in comparison with reference pyrrole insecticide (chlorfenapyr). In the lab strain, the pyrrolone derivatives showed a great variation of activities, hence the compounds 17 > 9 > 15 > 10 > 16 > 5 were the most effective derivatives against C. pipiens larvae, which exhibited 6.77, 6.24, 5.84, 5.32, 4.44 and 3.74 folds than 11, respectively. While in field strain, 17 > 15 > 9 > 10 > 16 > 2 showed toxicity 7.76, 7.11, 6.65, 6.04, 5.00 and 4.17 folds than 13, respectively. The structure-activity relationship study illustrated the variations observed in the toxicity of synthesized compounds. The cytochrome P-450 monooxygenase activities were significantly increased in treated larvae of lab and field strains in comparison to untreated which is remarkably responsible for tested compounds activation. Pyrrolone derivatives are promising compounds with novel mode of action in pest management programs and more studies should be prepared to create more active compounds.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Change history
03 December 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41598-024-79443-7
References
Hashem, A. I., Youssef, A. S. A., Kandeel, K. A. & Abou-Elmagd, W. S. I. Conversion of some 2(3H)-furanones bearing a pyrazolyl group into other heterocyclic systems with a study of their antiviral activity. Eur. J. Med. Chem. 42, 934–939. https://doi.org/10.1016/j.ejmech.2006.12.032 (2007).
Husain, A., Khan, M. S. Y., Hasan, S. M. & Alam, M. M. 2-Arylidene-4-(4-phenoxy-phenyl)but-3-en-4-olides: synthesis, reactions and biological activity Eur. J. Med. Chem. 40, 1394–1404. https://doi.org/10.1016/j.ejmech.2005.03.012 (2005).
Alam, M. M. et al. Synthesis of quinoline attached-furan-2(3H)-ones having anti-inflammatory and antibacterial properties with reduced gastro-intestinal toxicity and lipid peroxidation. J. Serb. Chem. Soc. 26, 1617–1626. https://doi.org/10.2298/JSC110131142A (2011).
Kumar, A., Ahmed, B., Srivastawa, B. & Vaishali COX-2 inhibitory and GABAergic activity of newly synthesized 2(3H)-furanone. Der Pharm. Chem. 4 383–391. (2012).
Chen, D., Song, Y., Lu, Y. & Xue, X. Synthesis and in vitro cytotoxicity of andrographolide-19-oic acid analogues as anti-cancer agents. Bioorg. Med. Chem. Lett. 23, 3166–3169. https://doi.org/10.1016/j.bmcl.2013.04.010 (2013).
Murugesan, D. et al. Discovery and structure – activity relationships of Pyrrolone Antimalarials. J. Med. Chem. 8, 1537–1544. https://doi.org/10.1021/jm400009c (2013).
Husain, A., Alam, M. M., Hasan, S. M. & Yar, M. S. 2(3H)-furanones and 2(3H)-pyrrolones: synthesis and antimycobacterial evaluation. Acta Pol. Pharm. Drug Res. 66, 173–180 (2009).
Gholap, S. S. Pyrrole: an emerging scaffold for construction of valuable therapeutic agents. Eur. J. Med. Chem. 110, 13–31. https://doi.org/10.1016/j.ejmech.2015.12.017 (2016).
Zarantonello, P., Leslie, C. P., Ferritto, R. & Kazmierski, W. M. Total synthesis and semi-synthetic approaches to analogues of antibacterial natural product althiomycin. Bioorg. Med. Chem. Lett. 12 (4), 561–565. https://doi.org/10.1016/S0960-894X(01)00802-2 (2002).
Weaver, S. C., Charlier, C., Vasilakis, N. & Lecuit, M. Zika, Chikungunya, and other Emerging Vector-Borne viral diseases. Annu. Rev. Med. 69, 395–408. https://doi.org/10.1146/annurev-med-050715-105122 (2018).
Parola, P., Musso, D. & Raoult, D. Rickettsia felis: the next mosquito-borne outbreak? Lancet Infect. Dis. 16, 1112–1113. https://doi.org/10.1016/S1473-3099(16)30331-0 (2016).
World Malaria Report. Available online: (2020). https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2020. (accessed on 1 September 2022).
Nebbak, A., Almeras, L., Parola, P. & Bitam, I. Mosquito vectors (Diptera: Culicidae) and Mosquito-Borne diseases in North Africa. Insects. 13 (10), 962. https://doi.org/10.3390/insects13100962 (2022).
Blagrove, M. S. C. et al. Co-occurrence of viruses and mosquitoes at the vectors’ optimal climate range: an underestimated risk to temperate regions? PLoS Negl. Trop. Dis. 11, e0005604. https://doi.org/10.1371/journal.pntd.0005604 (2017).
Yuan, J. Z., Li, Q. F., Huang, J. B. & Gao, J. F. Effect of chlorfenapyr on cypermethrin-resistant Culex pipiens pallens Coq mosquitoes. Acta Trop. 143, 13–17. https://doi.org/10.1016/j.actatropica.2014.12.002 (2015).
Bouaka, C., Ambadiang, M., Ashu, F., Fouet, C. & Kamdem, C. Testing Anopheles larvae and adults using standard bioassays reveals susceptibility to chlorfenapyr (pyrrole) while highlighting variability between species. bioRxiv. https://doi.org/10.1101/2024.03.24.586483
Nkya, T. E. et al. Insecticide resistance mechanisms associated with different environments in the malaria vector Anopheles gambiae: a case study in Tanzania. Malar. J. 13, 28. https://doi.org/10.1186/1475-2875-13-28 (2014).
IRAC, The Insecticide Resistance Action Committee, Mode of Action Classification Brochure, Edition: 11.1 – January 2024. (2024). http://www.efaidnbmnnnibpcajpcglclefindmkaj/https://irac-online.org/documents/moa-brochure/.
N’Guessan, R. et al. Chlorfenapyr: a pyrrole insecticide for the control of pyrethroid or DDT resistant Anopheles gambiae (Diptera: Culicidae) mosquitoes. Acta Trop. 102, 69–78. https://doi.org/10.1016/j.actatropica.2007.03.003 (2007).
Hekal, M. H., Farag, P. S., Hemdan, M. M. & El-Sayed, W. M. New N-(1,3,4- Thiadiazol-2-yl)furan-2-carboxamide derivatives as potential inhibitors of the VEGFR-2. Bioorg. Chem. 115, 105176. https://doi.org/10.1016/j.bioorg.2021.105176 (2021).
Mahmoud, M. R., Abou-Elmagd, W. S. I., Derbala, H. A. & Hekal, M. H. Synthesis and spectral characterisation of some phthalazinone derivatives. J. Chem. Res. 36, 75–82. https://doi.org/10.3184/174751912X13274297624330 (2012).
Mahmoud, M. R., Abu El-Azm, F. S. M., Ismail, M. F., Hekal, M. H. & Ali, Y. M. Synthesis and antitumor evaluation of Novel Tetrahydrobenzo [4′,5′]Thieno[3′,2′ :5,6]Pyrimido[1,2-b]isoquinoline derivatives. Synth. Commun. 48, 428–438. https://doi.org/10.1080/00397911.2017.1406520 (2018).
Mahmoud, M. R., El-Shahawi, M. M., Abou-Elmagd, W. S. I. & Hekal, M. H. Novel synthesis of isoquinoline derivatives derived from (Z)-4-(1,3- Diphenylpyrazol-4-yl)Isochromene-1,3-Dione. Synth. Commun. 45, 1632–1641. https://doi.org/10.5155/eurjchem.1.2.134-139.71 (2015).
Hekal, M. H., El-Naggar, A. M., Abu El-Azm, F. S. M. & El-Sayed, W. M. Synthesis of New Oxadiazol-Phthalazinone derivatives with anti-proliferative activity; Molecular Docking, Pro-apoptotic, and enzyme Inhibition Profile. RSC Adv. 10, 3675–3688. https://doi.org/10.1039/C9RA09016A (2020).
Mahmoud, M. R., Abou-Elmagd, W. S. I., Derbala, H. A. & Hekal, M. H. Novel synthesis of some phthalazinone derivatives. Chin. J. Chem. 29, 1446–1450. https://doi.org/10.1002/cjoc.201180264 (2011).
Hamed, N. A., Ismail, M. F., Hekal, M. H. & Marzouk, M. I. Design, synthesis, and evaluation of some Novel Heterocycles Bearing Pyrazole Moiety as potential Anticancer agents. J. Het Chem. 56, 1771–1779. https://doi.org/10.1002/jhet.3544 (2019).
Ali, A. T. & Hekal, M. H. Convenient synthesis and anti-proliferative activity of some benzochromenes and chromenotriazolopyrimidines under classical methods and phase transfer catalysis. Synth. Commun. 49, 3498–3509. https://doi.org/10.1080/00397911.2019.1675173 (2019).
Hekal, M. H., Abu El-Azm, F. S. M. & Salla, H. A. Synthesis, spectral characterization, and in vitro Biological evaluation of some Novel Isoquinolinone- based heterocycles as potential Antitumor agents. J. Heterocycl. Chem. 56, 795–803. https://doi.org/10.1002/jhet.3448 (2019).
Abu El-Azm, F. S. M., Ali, A. T. & Hekal, M. H. Facile synthesis and anticancer activity of Novel 4-Aminothieno[2,3-d]pyrimidines and Triazolothienopyrimidines. Org. Prep. Proced. Int. 51, 507–520. https://doi.org/10.1080/00304948.2019.1666635 (2019).
Hekal, M. H., Ali, Y. M. & Abu El-Azm, F. S. M. Utilization of Cyanoacetohydrazide and 2-(1,3-Dioxoisoindolin-2-yl) acetyl chloride in the synthesis of some Novel anti-proliferative heterocyclic compounds. Synth. Commun. 50, 2839–2852. https://doi.org/10.1080/00397911.2020.1786125 (2020).
Hekal, M. H. & Abu El-Azm, F. S. M. New potential Antitumor quinazolinones derived from dynamic 2-Undecyl benzoxazinone: synthesis and cytotoxic evaluation. Synth. Commun. 48, 2391–2402. https://doi.org/10.1080/00397911.2018.1490433 (2018).
Hekal, M. H., Abu El-Azm, F. S. M. & Atta-Allah, S. R. Ecofriendly and highly efficient Microwave-Induced synthesis of Novel Quinazolinone-Undecyl hybrids with in vitro Antitumor Activity. Synth. Commun. 49, 2630–2641. https://doi.org/10.1080/00397911.2019.1637001 (2019).
Abdalha, A. A. & Hekal, M. H. An efficient synthesis and evaluation of some novel quinazolinone-pyrazole hybrids as potential Antiproliferative agents. Synth. Commun. 51, 2498–2509. https://doi.org/10.1080/00397911.2021.1939058 (2021).
Hekal, M. H., Abu El-Azm, F. S. M. & Samir, S. S. An efficient approach for the synthesis and antimicrobial evaluation of some new benzocoumarins and related compounds. Synth. Commun. 51, 2175–2186. https://doi.org/10.1080/00397911.2021.1925917 (2021).
Hamed, N. A., Marzouk, M. I., Ismail, M. F. & Hekal, M. H. N’-(1-([1,1’-biphenyl]-4-yl)ethylidene)-2- cyanoacetohydrazide as scaffold for the synthesis of diverse heterocyclic compounds as prospective antitumor and antimicrobial activities. Synth. Commun. 49, 3017–3029. https://doi.org/10.1080/00397911.2019.1655578 (2019).
Hekal, M. H. & Abu El-Azm, F. S. M. Efficient MW-Assisted synthesis of some New Isoquinolinone derivatives with in vitro Antitumor Activity. J. Het Chem. 54, 3056–3064. https://doi.org/10.1002/jhet.2916 (2017).
Hekal, M. H., Samir, S. S. & Ali, Y. M. El- Sayed, New Benzochromeno[2,3-d]pyrimidines and Benzochromenotriazolo[1,5-c]pyrimidines as potential inhibitors of the topoisomerase II. Polycycl. Arom. Compd. 42, 7644–7660. https://doi.org/10.1080/10406638.2021.2006247 (2022).
Hekal, M. H., Farag, P. S., Hemdan, M. M., El-Sayed, A. A. & Hassaballah, A. I. El-Sayed. New 1,3,4-thiadiazoles as potential anticancer agents: pro-apoptotic, cell cycle arrest, molecular modelling, and ADMET profile. RSC Adv. 13, 15810. https://doi.org/10.1039/D3RA02716C (2023).
Hekal, M. H., Ali, Y. M., Abdel-Haleem, D. R. & Abu El-Azm, F. S. M. Diversity oriented synthesis and SAR studies of new quinazolinones and related compounds as insecticidal agents against Culex pipiens L. Larvae and associated predator. Bioorg. Chem. 133, 106436. https://doi.org/10.1016/j.bioorg.2023.106436 (2023).
David, M. D. The potential of pro-insecticides for resistance management. Pest Manag Sci. 77, 3631–3636. (2021).
Boukouvala, M. C., Kavallieratos, N. G., Athanassiou, C. G., Benelli, G. & Hadjiarapoglou, L. P. Insecticidal efficacy of six new pyrrole derivatives against four stored-product pests. Environ. l Sci. Pull Res. Inter. 26 (29), 29845–29856. https://doi.org/10.1007/s11356-019-05961-x (2019).
Treacy, M. et al. Uncoupling activity and pesticidal properties of pyrroles. Biochem. Soc. Trans. 22, 244–247. https://doi.org/10.1042/bst0220244 (1994).
Hunt, D. A. & Treacy, M. F. in Pyrrole Insecticides: A New Class of Agriculturally Important Insecticides Functioning as Uncouplers of Oxidative Phosphorylation BT - Insecticides with Novel Modes of Action: Mechanisms and Application (eds. Ishaaya, I. & Degheele, D.) 138–151 (Springer Berlin Heidelberg). (1998).
Yunta, C. et al. Chlorfenapyr metabolism by mosquito P450s associated with pyrethroid resistance identifies potential activation markers. Sci. Rep. 13 (1), 14124. https://doi.org/10.1038/s41598-023-41364-2 (2023).
Zhang, L., Ou, X. M. & Pei, H. A review on pyrrole compounds with insecticidal and miticidal activity. Fine-Scale Chem. Intermed. 39, 1–6 (2009).
Huang, P. et al. A Comprehensive Review of the current knowledge of Chlorfenapyr: Synthesis, Mode of Action, Resistance, and Environmental Toxicology. Molec. 28 (22), 7673. https://doi.org/10.3390/molecules28227673 (2023).
Oxborough, R. M. et al. The activity of the pyrrole insecticide chlorfenapyr in mosquito bioassay: towards a more rational testing and screening of non-neurotoxic insecticides for malaria vector control. Malar. J. 14, 124. https://doi.org/10.1186/s12936-015-0639-x (2015).
Oliver, S. V. et al. Evaluation of the pyrrole insecticide chlorfenapyr against pyrethroid resistant and susceptible Anopheles funestus (Diptera: Culicidae). Trop. Med. Int. Heal. 15, 127–131. https://doi.org/10.1111/j.1365-3156.2009.02416.x (2010).
Kweyamba, P. A. et al. Sub-lethal exposure to chlorfenapyr reduces the probability of developing Plasmodium falciparum parasites in surviving Anopheles mosquitoes. Paras Vect. 16 (1), 342. https://doi.org/10.1186/s13071-023-05963-2 (2023).
Dagg, K. et al. Evaluation of toxicity of clothianidin (neonicotinoid) and chlorfenapyr (pyrrole) insecticides and cross-resistance to other public health insecticides in Anopheles arabiensis from Ethiopia. Malar. J. 18 (1), 49. https://doi.org/10.1186/s12936-019-2685-2 (2019).
Zhao, Y. et al. Chlorfenapyr, a Potent Alternative Insecticide of Phoxim to Control Bradysia odoriphaga (Diptera: Sciaridae). J. Agric. Food Chem. 65 (29), 5908–5915. https://doi.org/10.1021/acs.jafc.7b02098 (2017).
Raghavendra, K. et al. Chlorfenapyr: a new insecticide with novel mode of action can control pyrethroid resistant. Malar. Vectors Malar. J. 10, 16. https://doi.org/10.1186/1475-2875-10-16 (2011).
Black, B. C., Hollingworth, R. M., Ahammadsahib, K. I., Kukel, C. D. & Donovan, S. Insecticidal action and mitochondrial uncoupling activity of AC-303,630 and related halogenated pyrroles. Pestic Biochem. Physiol. 50, 115–128. https://doi.org/10.1006/pest.1994.1064 (1994).
Murtaza, G. et al. Toxicity of different insecticides against dengue vector aedes aegypti larvae under laboratory conditions. J. Innov. Sci. 8 (1), 08–12. https://doi.org/10.17582/journal.jis/2022/8.1.08.12 (2022).
Che-Mendoza, A. et al. Efficacy of targeted indoor residual spraying with the pyrrole insecticide chlorfenapyr against pyrethroid resistant Aedes aegypti. PLoS Negl. Trop. Dis. 15 (10), e0009822. https://doi.org/10.1371/journal.pntd.0009822 (2021).
Boukouvala, M. C. et al. Laboratory evaluation of five novel pyrrole derivatives as grain protectants against Tribolium confusum and ephestia kuehniella larvae. J. Pest Sci. 90, 569–585. https://doi.org/10.1007/s10340-016-0808-x (2017).
Li, Y. et al. The trifluoromethyl transformation synthesis, crystal structure and insecticidal activities of novel 2-pyrrolecarboxamide and 2-pyrrolecarboxlate. Bioorg. Medic Chem. Lett. 22 (22), 6858–6861. https://doi.org/10.1016/j.bmcl.2012.09.036 (2012).
El Sherif, D. F. et al. The Binary mixtures of Lambda-Cyhalothrin, Chlorfenapyr, and Abamectin, against the House fly Larvae, Musca domestica L. Molec. 27 (10), 3084. https://doi.org/10.3390/molecules27103084 (2022).
Ito, M. et al. Synthesis and insecticidal activity of novel N-oxydihydropyrroles: 4-hydroxy-3-mesityl-1-methoxymethoxy derivatives with various substituents at the 5-position. Bioorg. Medic Chem. 11 (5), 761–768. https://doi.org/10.1016/s0968-0896(02)00474-1 (2003).
Li, H., Yang, N., Xiong, L. & Wang, B. Design, synthesis and biological evaluation of Novel Thienylpyridyl- and thioether-containing acetamides and their derivatives as Pesticidal agents. Molec. 26 (18), 5649. https://doi.org/10.3390/molecules26185649 (2020).
Rashid, K. O. et al. Synthesis of novel phenoxyacetamide derivatives as potential insecticidal agents against the cotton leafworm, Spodoptera littoralis. Polycycl. Arom. Compd. 43 (1), 356–369. https://doi.org/10.1080/10406638.2021.2015400 (2023).
Abdelhamid, A. A., Salama, K. S. M., Elsayed, A. M. & Gad, M. A. El-Remaily, synthesis and Toxicological Effect of some New pyrrole derivatives as prospective Insecticidal agents against the Cotton Leafworm, Spodoptera Littoralis (Boisduval). ACS Omega. 7, 3990–4000. https://doi.org/10.1021/acsomega.1c05049 (2022).
Abdel-Haleem, D. R., Badr, E. E., Samy, A. M. & Baker, S. A. Larvicidal evaluation of two novel cationic gemini surfactants against the potential vector of West Nile virus Culex pipiens Linnaeus (Diptera: Culicidae). Med. Vet. Entomol. 37 (3), 483–490. https://doi.org/10.1111/mve.12645 (2023).
Tantawy, A. H., Farag, S. M., Abdel-Haleem, D. R. & Mohamed, H. I. Facile synthesis, larvicidal activity, biological effects, and molecular docking of sulfonamide-incorporating quaternary ammonium iodides as acetylcholinesterase inhibitors against Culex pipiens L. Bioorg. Chem. 128, 106098. https://doi.org/10.1016/j.bioorg.2022.106098 (2022).
Zhao, Y. et al. Synthesis, insecticidal, and acaricidal activities of novel 2-aryl-pyrrole derivatives containing ester groups. J. Agric. Food Chem. 56 (21), 10176–10182. https://doi.org/10.1021/jf802464d (2008).
Kauffman, E. et al. Rearing of Culex spp. and Aedes spp. mosquitoes. Bio-Protoc.. 7 (17), e2542. https://doi.org/10.21769/BioProtoc.2542 (2017).
El-Sayed, M. K. F., El-Shahawi, M. M., Ali, Y. M. & Abdel-Haleem, D. R. Abu El-Azm, Synthesis, larvicidal efficiency and molecular docking studies of novel annulated pyrano[2,3-c]pyrazoles against Culex pipiens L. and Musca domestica L. larvae. Bioorg. Chem. 130, 106258. https://doi.org/10.1016/j.bioorg.2022.106258 (2023).
WHO. Guidelines for Laboratory and Field Testing of Mosquito Larvicides (World Health & Organization https://www.who.int/publications/i/item/WHO-CDS-WHOPES-GCDPP-2005.13 (2005).
Abbass, E. M., Ali, A. K., El-Farargy, A. F., Abdel-Haleem, D. R. & Shaban, S. S. Synthesis, toxicological and in silico evaluation of novel spiro pyrimidines against Culex pipiens L. referring to chitinase enzyme. Sci. Rep. 14 (1), 1516. https://doi.org/10.1038/s41598-024-51771-8 (2024).
Amin, T. Biochemical and Physiological Studies of Some Insect Growth Regulators on the Cotton Leafworm Spodoptera littoralis (Boisd.). Cairo University. (1998).
Hadidy, D. E., El Sayed, A. M., Tantawy, M. E., El Alfy, T. & Farag, S. M. Abdel Haleem, Larvicidal and repellent potential of Ageratum houstonianum against Culex pipiens. Sci. Rep. 12, 21410. https://doi.org/10.1038/s41598-022-25939-z (2022).
Hansen, I. G. & Hodgson, E. Biochemical characteristics of insect microsomes, N-and o-demethylation. Biochem. Pharmacol. 20, 1569–1578. https://doi.org/10.1016/0006-2952(71)90285-1 (1971).
Finney, D. Quantal Responses to Mixtures, Probit Analysispp. 230–268 (third ed., Cambridge University Press, 1971).
Villeneuve, D. L., Blankenship, A. L. & Giesy, J. P. Derivation and application of relative potency estimates based on in Vitro Bioassay results. Environ. Toxicol. Chem. 19, 2835–2843. https://doi.org/10.1002/etc.5620191131 (2000).
Duncan, D. B. Multiple range, and multiple F tests. Biom. 2, 1–42. https://doi.org/10.2307/3001478 (1955).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Dr. Mohamed H. Hekal, Dr. Fatma S.M. Abu El-Azm, Dr. Ahmed I. Hashem, Dr. El-Hady Rafat and Dr. Yasmeen M. Ali, synthesized and discussed new pyrrolone derivatives and wrote the main manuscript and prepared figures. Dr. Doaa R. Abdel-Haleem made evaluation of Larvicidal Efficacy of new pyrrolone derivatives against Culex pipiens L. Larvae . All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This research was approved by the Research Ethics Committee at the Faculty of Science, Ain Shams University (Approval code: ASU-SCI/ENTO/2024/7/3).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: In the original version of the Article, Figure 6 was a duplication of Figure 5.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hekal, M.H., Hashem, A.I., El-Azm, F.S.A. et al. Rational design and synthesis of new pyrrolone candidates as prospective insecticidal agents against Culex pipiens L. Larvae. Sci Rep 14, 24467 (2024). https://doi.org/10.1038/s41598-024-74011-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-74011-5
Keywords
This article is cited by
-
Synthesis, SAR, and in silico studies of new benzochromene derivatives as insecticidal agents against Culex pipiens L. larvae and adults
Scientific Reports (2025)
-
Synthesis and in silico studies of new thiophene-isoquinolinone hybrids as potential larvicides against Culex pipiens
Scientific Reports (2025)