Abstract
Mast cells (MCs) are tissue-resident cells of hematopoietic origin that play an important role in host’s defense mechanism against nematodes. However, excessive activation of these cells contributes to the development of certain allergic diseases. Immunoglobin E (IgE) is one of the well-known molecules that activate MCs. Even in the absence of specific antigens, the binding of highly cytokinergic IgE to FcεRI on MCs prolongs their survival and induces cytokine production without enhancing their degranulation. In the present study, we examined the effects of the members of the interleukin-10 (IL-10) family of cytokines on IgE-mediated MCs functions. The receptors including Il10r1, Il10r2, and Il20r2, but not Il20r1, Il22r1 or Il28r1, were constitutively expressed in mouse bone marrow cell-derived cultured MCs (BMCMCs), suggesting that IL-10 may influence MCs function. Indeed, we found that only IL-10 could influence upon BMCMCs function; IL-10 enhanced prolongation of survival, promoted IL-6 and/or IL-13 production dependently of STAT1 and STAT3, and suppressed tumor necrosis factor production independently of STAT1 and STAT3 on IgE-stimulated BMCMCs. Moreover, the IL-10-mediated enhancement of IL-6 production by IgE-stimulated BMCMCs promotes Th17 cell expansion. These results suggest that IL-10 has a dual role as an anti-inflammatory and pro-inflammatory cytokine in MCs functions.
Similar content being viewed by others
Introduction
Mast cells (MCs) play a pivotal role in both innate and acquired immunities1. Crosslinking of Immunoglobulin E (IgE) bound to the high-affinity IgE receptor (FcεRI) on MCs by antigens results in their degranulation and activation, including the secretion of a variety of pro-inflammatory mediators, contributing to host’s defense mechanism against certain parasites2. In contrast, inappropriate and/or excessive activation of MCs also contribute to development of certain allergic diseases such as asthma and rhinitis3.
Members of the interleukin-10 (IL-10) family of cytokines include IL-10 and eight other related cytokines (IL-19, IL-20, IL-22, IL-24, IL-26, IL-28 A, IL-28B, and IL-29)4. IL-10 was initially described as a T helper 2 (Th2)-type cytokine that inhibited cytokine production in Th1 cells5. In addition, further evidence has suggested that regulatory T (Treg) cells are the main producers of IL-10, which plays an important immunoregulatory role by suppressing functions of various types of cells6. IL-10 binds to the heterodimeric IL-10 receptor (IL-10R), which comprises IL-10R1 and IL-10R2 (IL-10R1/IL-10R2). IL-19 binds to one receptor (IL-20R1/L-20R2), IL-20 binds to two receptors (IL-20R1/IL-20R2 and IL-20R2/IL-22R1), IL-22 binds to one receptor (IL-10R2/IL-22R1), IL-24 binds to two receptors (IL-20R1/IL-20R2 and IL-20R2/IL-22R1), IL-26 binds to one receptor (IL-10R2/IL-20R1) and, IL-28 A, IL-28B and IL-29 bind to one receptor (IL-10R2/IL-28R1)4.
Notably, IL-10 can enhance the induction of apoptosis via cooperation with IL-3 and IL-47 and suppress the degranulation8,9 of IgE/antigen (Ag)-stimulated mouse bone marrow cell-derived cultured MCs (BMCMCs). In addition, IL-10 suppresses the production of tumor necrosis factor (TNF) and/or granulocyte macrophage colony stimulation factor (GM-CSF)8,9, but enhances the production of IL-6, IL-9, and IL-13 in IgE/Ag-stimulated mouse BMCMCs10,11. Therefore, IL-10 is a cytokine with both anti-inflammatory and pro-inflammatory properties in certain settings. With respect to IL-10-related cytokines, IL-29 is known to induce the secretion of IL-4, IL-6, and IL-13, but not IL-10 and IL-12, from P815 mastocytoma cells12; however, the role of other cytokines, except IL-10 and IL-29, on MCs has not been elucidated.
Even in the absence of specific Ags, the binding of IgE (monomeric IgE or highly cytokinergic IgE [hcIgE]) to FcεRI in human and mouse MCs prolongs survival and induces cytokine production without enhancing their degranulation2,13,14. However, the roles of IL-10 and other IL-10-related cytokines remain unclear. Therefore, in the present study, we elucidated the effects of members of the IL-10 family of cytokines (IL-10, IL-19, IL-20, IL-22, IL-24, IL-26, IL-28 A, IL-28B, and IL-29) on mouse BMCMCs function during IgE stimulation in the absence of specific Ags.
Results
Expression of the IL-10R family on BMCMCs
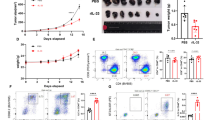
The expression of the IL-10R family of molecules in BMCMCs was examined by qPCR. A human mast cell line (HMC-1) was used as the negative control. Constitutive expression of Il10r1, Il10r2, and Il20r2 was observed in BMCMCs, whereas the expression of Il20r1, Il22r1, and Il28r1 was barely detectable (Fig. 1A). The performance of qPCR for the expression of Il20r1, Il22r1, and Il28r1 was ensured using fibroblasts (Fig. 1B). The expression levels of Il10r1 and Il20r2 remained unchanged, but those of Il10r2 were reduced in BMCMCs after IgE (SPE-7) stimulation (Fig. 1A). However, by FACS analysis, the expression of IL-10R1 was hardly detectable on the cell surface of BMCMCs irrespective of IgE (SPE-7) stimulation (data not shown). These results suggest that BMCMCs express receptors only for IL-10, and not for the other members of the IL-10 family of cytokines; thus, implying that only IL-10 may influence MCs function.
Expression of IL-10R family members in BMCMCs. Expression levels of Il10r1, Il10r2, Il20r1, Il20r2, Il22r1, and/or Il28r1 mRNA in BMCMCs (n = 3, derived from distinct mice) in the presence or absence of IgE (SPE-7) (A) and NIH3T3 (B) determined by qPCR. HMC-1 mRNA was used as a negative control (A). Data show the mean ± SEM and the representative result of two independent experiments. *p < 0.001, HMC-1 vs. BMCMCs, and †p< 0.05, medium vs. SPE-7. NS not significant.
IL-10 affects survival and cytokine production, but not degranulation, in IgE-stimulated BMCMCs
The BMCMCs were stimulated with or without IgE (SPE-7) in the presence or absence of IL-10, IL-19, IL-20, IL-22, IL-24, IL-26, IL-28 A, IL-28B, and IL-29. None of the cytokines has an influence upon degranulation of BMCMCs assessed by β-hexosaminidase release irrespective of IgE stimulation (Fig. 2A). In contrast, IL-10, but not other cytokines, enhanced the survival of BMCMCs during IgE stimulation (Fig. 2B). Moreover, IL-10, but not the other cytokines, enhanced IL-6 and IL-13 production but suppressed TNF production in BMCMCs (Fig. 2C). These results suggest that IL-10, but not other members of the IL-10 family of cytokines, affects certain MC functions.
IL-10 affects survival and cytokine production but not degranulation in IgE-stimulated BMCMCs. BMCMCs (n = 5–10 derived from distinct mice) were stimulated with or without IgE (SPE-7) in the presence or absence of 1–100 ng/mL IL-10 and 100 ng/mL IL-19, IL-20, IL-22, IL-24, IL-26, IL-28 A, IL-28B, or IL-29 for 1 h (A), 3 days (B), and 24 h (C). (A) The levels of β-hexosaminidase release. P/I = PMA + Ionomycin, “-” = no cytokine. (B) Cell survival assessed in PI- and annexin V-negative cells by FACS analysis. *p < 0.05, no cytokine (-) vs. SPE-7. (C) Levels of IL-6, IL-13, and TNF in culture supernatants, as determined by ELISA. *p < 0.05, vs. no cytokine “0”. Data show the mean ± SEM and the representative result of two independent experiments.
Effects of IL-10 on the production of IL-6, but not of TNF, by BMCMCs are dependent of STAT1 and STAT3
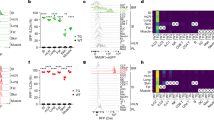
STAT1 and STAT3 are important transcription factors for signal transduction of IL-10R after IL-10 binding4. IL-10 enhanced IL-6 production in Stat1+/+ BMCMCs, but not in Stat1−/− BMCMCs, during IgE stimulation (Fig. 3A). Treatment with a STAT3 inhibitor (cucurbitacin I) partially inhibited the enhanced IL-6 production by BMCMCs in the presence of low doses of IL-10 (1 or 10 ng/ml) during IgE stimulation (Fig. 3B). However, such inhibitory effects of the STAT3 inhibitor were not observed in BMCMCs treated with a high dose of IL-10 (100 ng/mL) (Fig. 3B). Similar results were also observed using Cell-permeable STAT3 inhibitor peptides (Supplementary Fig. 1). Additionally, IL-6 production was comparable between STAT3 inhibitor- and vehicle-treated Stat1−/− BMCMCs in the presence of IL-10 during IgE stimulation (Fig. 3C). These observations suggest that both STAT1 and STAT3 are important for the enhancement of IL-6 production by IgE-stimulated BMCMCs in response to the optimal dose of IL-10; however, STAT1, rather than STAT3, is more crucial for these settings. In contrast, neither STAT1 nor STAT3 was important for the suppression of TNF-production by BMCMCs in the presence of various doses of IL-10 during IgE stimulation (Fig. 3A-C).
Effects of IL-10 on the production of IL-6, but not on TNF, by BMCMCs depend on STAT1 and STAT3. BMCMCs (A) or 0.5 µM cucurbitacin I-treated BMCMCs (B,C) (n = 5–10 derived from distinct mice) were stimulated with or without IgE (SPE-7) in the presence or absence of 1–100 ng/mL IL-10 for 24 h. The levels of IL-6 and TNF in the culture supernatants were determined using ELISA. (A) The cultivation using Stat1+/+ and Stat1−/− BMCMCs. *p < 0.05, Stat1+/+ BMCMCs vs. Stat1−/− BMCMCs. (B) Culture of cucurbitacin-I-treated wild-type BMCMCs. *p < 0.05, vehicle-treated BMCMCs vs. cucurbitacin I-treated BMCMCs. (C) The cultivation using cucurbitacin I-treated Stat1−/− BMCMCs. Data show the mean ± SEM and the representative result of two independent experiments.
IL-10 promotes Th17 cell differentiation by enhancing STAT1-dependent IL-6 production by IgE-stimulated MCs
Notably, IL-6 is a key cytokine for Th17 cell differentiation15, and MCs can enhance various types of Th cell responses16,17. Thus, we examined the effect of IL-10 on MC-derived IL-6-mediated Th17 cell differentiation in a co-culture of naïve CD4+ T cells and BMCMCs. Proportion of IL-17+ and Foxp3+ CD4+ T cells were below 0.1% in the culture of CD4+ T cells alone irrespective of stimuli such as plate-coated anti-CD3 mAb and/or rmIL-10 (Fig. 4A). When naïve CD4+ T cells were co-cultured with wild-type BMCMCs and stimulated with anti-mouse CD3 mAb and IgE (SPE-7) in the presence or absence of IL-10, the proportion of Th17 cells in the IL-10-treated condition was approximately two-fold higher than that in the untreated condition (Fig. 4A, B). These effects of IL-10 were not observed in the co-culture of naïve CD4+ T cells and Il6−/− BMCMCs or Stat1−/− BMCMCs (Fig. 4A, B). In contrast, IL-10 did not influence the proportion of Tregs, irrespective of the genotype of BMCMCs (Fig. 4A, B). These observations suggest that IL-10 promotes IL-6 production by IgE-stimulated MCs via STAT1, followed by the enhancement of IL-6-mediated Th17 cell differentiation.
IL-10 promotes Th17 cell differentiation by enhancing STAT1-dependent IL-6 production in IgE-stimulated mast cells. Naïve CD4+ T cells were co-cultured with wild-type and Il6−/− BMCMCs (n = 4–5 derived from distinct mice) or with Stat1+/+ and Stat1−/− BMCMCs (n = 4–5 derived from distinct mice) in the presence of anti-mouse CD3 mAb and IgE (SPE-7), with or without 100 ng/mL rmIL-10, for 4 days. The cells were then stimulated with PMA + ionomycin in the presence of monensin for 6 h. Intracellular IL-17 and Foxp3 expression levels were determined by FACS analysis. (A) Representative FACS results. (B) Data show the mean ± SEM from (A), and the representative results of two independent experiments. *p < 0.05, medium vs. rmIL-10, and †p < 0.05, wild-type BMCMCs vs. Il6−/− BMCMCs.
Discussion
IL-10 is a well-known anti-inflammatory cytokine. In support of this, Il10−/− mice spontaneously develop autoimmune-like colitis18 and exhibit increased allergic airway eosinophilia19. In contrast, Il10−/− mice show attenuated allergen-induced airway hyperresponsiveness19, suggesting that IL-10 has pro-inflammatory properties. Similarly, IL-10 can suppress TNF and/or GM-CSF production8,9 but enhance IL-6, IL-9, and/or IL-13 production by IgE/Ag-stimulated mouse BMCMCs10,11. In the present study, we demonstrated that IL-10 plays dual anti-inflammatory and pro-inflammatory roles in hcIgE-mediated MC function.
Even in the absence of specific Ags, the binding of IgE (monomeric IgE or hcIgE) to FcεRI on human and mouse mast cells prolongs survival and induces cytokine production without enhancing their degranulation2,13,14. In these settings, we demonstrated that IL-10 could enhance the prolongation of survival and production of IL-6 and IL-13, but suppress the production of TNF in mouse BMCMCs (Fig. 2B, C). Similar effects of IL-10 were also observed in BMCMCs stimulated with LPS or IL-33 (data not shown). In addition, the members of the IL-10 family of cytokines, except IL-10, did not affect MCs functions due to the lack of receptor mRNA expression (Figs. 1 and 2B, C).
We also demonstrated that both STAT1 and STAT3 are important for the enhancement of IL-6 production by IgE-stimulated BMCMCs in response to IL-10; however, STAT1, rather than STAT3, was more crucial in these settings (Fig. 3). In contrast, neither STAT1 nor STAT3 were involved in the suppression of TNF production by IgE-stimulated BMCMCs in response to IL-10 (Fig. 3). In addition to STAT1 and STAT3, STAT5 is also involved in signal transduction of IL-10R after IL-10 binding4. Therefore, STAT5 may be involved in the IL-10-mediated suppression of TNF production by BMCMCs under these conditions.
Notably, IL-10 suppresses IL-6- and TGF-β1-mediated Th17 cell differentiation in vitro20 and Th17 cell activation in vivo21. In contrast, we demonstrated that the IL-10-mediated enhancement of IL-6 production by IgE-stimulated MCs promoted Th17 cell expansion (Fig. 4). These observations indicate that IL-10 can directly inhibit Th17 cell differentiation and activation, while promoting Th17 cell differentiation and activation via mast cell activation.
In summary, IL-10 has a dual role as an anti-inflammatory and pro-inflammatory cytokine in MCs. Our results contribute to a better understanding of the mechanisms by which IL-10 exerts different effects, such as the suppression of allergic eosinophilic airway inflammation while promoting airway hyperresponsiveness.
Methods
Mice
C57BL/6J-wild-type mice (males, 6–10 weeks) were purchased from CLEA Japan (Tokyo, Japan). C57BL/6-Il6-/- and 129xB6-Stat1-/- mice were obtained from Jackson Laboratory (Harbor, ME, USA) and Taconic Farms (Germantown, NY, USA), respectively. Mice were euthanized by inhalation anesthesia with sevoflurane. All the mice were housed under specific pathogen-free conditions at individual universities and institutes. All experiments including animals were approved by and performed in accordance with the Animal Care and Use Committee of each university and institute (Approval No. C23-28, C23-26-2 and S-290069) and with ARRIVE guidelines (https://arriveguidelines.org).
Cells
A human mast cell line (HMC-1), kindly provided by Dr. Butterfield (Mayo Clinic, Rochester, MN, USA), was cultured in RPMI culture medium (RPMI1640 medium [SIGMA-Aldrich, St. Louis, MO, USA] containing 10% fetal bovine serum [Thermo Fisher Scientidic, Waltham, MA, USA], 50 µg/mL penicillin + 50 U/mL streptomycin [Thermo Fisher Scientidic] and 2-mercaptoethanol [Thermo Fisher Scientidic]). The mouse BMCMCs were differentiated from the bone marrow cells, as previously described22,23. Briefly, bone marrow cells were cultured for 4–8 weeks in culture medium containing 20 ng/mL mouse recombinant mouse (rm) IL-3 (PeproTech, Rocky Hill, NJ, USA) at 37 °C in a humidified 5% CO2 incubator. The culture medium was changed once a week (> 95% c-kithi FcεRIαhi population by FACS). A mouse fibroblast cell line, NIH3T3 (obtained from ATCC), was cultured in Dulbecco’s modified Eagle medium (DMEM) culture medium (DMEM [Thermo Fisher Scientific] containing 10% fetal bovine serum [Thermo Fisher Scientific] and 50 µg/mL penicillin + 50 U/mL streptomycin [Thermo Fisher Scientific]).
Quantitative real-time polymerase chain reaction (PCR)
The expression levels of mRNA were determined by quantitative real-time PCR, as described previously24. Briefly, total RNA was extracted from mast cells (MCs) and NIH3T3 using Sepasol-RNA I Super G (09379; Nacalai Tesque, Inc., Kyoto, Japan). cDNA was synthesized, and contaminating DNA was removed using ReverTra Ace® qPCR RT Master Mix with gDNA Remover (FSQ-301; TOYOBO CO., LTD, OSAKA, Japan). Quantitative real-time PCR was performed with SYBR Premix Ex Taq (TAKARA BIO Inc., Shiga, Japan) using a CFX384TM Touch Real-time PCR Detection System (BioRad Laboratories Inc, Hercules, CA). Relative gene expression was determined based on Gapdh mRNA expression. The PCR primers were designed as shown in Supplemental Table 1.
β-hexosaminidase release assay
The mouse BMCMCs (1 × 105 cells/well), placed in a 96-well V-bottom plate, were cultured in 100 µL/well of Tyrode’s buffer with or without 100 ng/mL recombinant mouse (rm) IL-10 (Peprotech), rmIL-19 (R&D Systems, Minnesota, MN, USA), rmIL-22 (R&D Systems), rmIL-24 (R&D Systems), recombinant human (rh) IL-26 (R&D Systems), rmIL-28 A (R&D Systems), rmIL-28B (R&D Systems), and rhIL-29 (R&D Systems) in the presence or absence of 1 µg/mL IgE (SPE-7) or with 0.1 µg/mL PMA and 1 µg/mL Ionomycin (as a positive control) at 37 °C for 1 h. In addition, for 100% degranulation, BMCMCs lysates were prepared by treated with TritonX-100. The levels of degranulation in BMCMCs were measured by the release of β-hexosaminidase in the granule component into the supernatant as described elsewhere13. Data show the percent release of β-hexosaminidase under various stimulation conditions relative to the total amount of β-hexosaminidase in the cells, as measured in the supernatants of Triton X-100-treated cells.
Cell survival
BMCMCs (5 × 105 cells/well), placed in a 24 well plate, were cultured in culture medium (0.5 mL/well) with or without 100 ng/mL cytokines in the presence or absence of 1 µg/mL IgE (SPE-7) at 37 °C for 3 days. Propidium iodide (PI)- and annexin V-negative live cells were determined by FACS analysis (MACSQuant Analyzer; Miltenyi Biotec, Bergisch Gladbach, Germany) using the MACSQuantify software (Miltenyi Biotec) and FlowJo software (BD Biosciences, San Jose, CA, USA), as previously described13.
ELISA for cytokines in culture supernatants
BMCMCs (2 × 105 cells/well), placed in a 96 well flat-bottom plate, were cultured in 200 µL /well of culture medium with 0, 1, 10, or 100 ng/mL cytokines in the presence or absence of 1 µg/mL IgE (SPE-7) at 37 °C for 24 h. For STAT3 inhibition, 0.5 µM cucurbitacin I (Merck KGaA, Darmstadt, Germany) or STAT3 inhibitor peptides (cell-permeable or inactive control; Merck) was added 30 min before stimulation with cytokines or IgE. After cultivation, the supernatants were collected, and the concentrations of IL-6, IL-13, and TNF in the supernatants were determined using mouse IL-6, IL-13, and TNF ELISA kits (BioLegend, San Diego, CA).
Detection of intracellular cytokines by FACS
CD4+ T cells and BMCMCs were co-cultured as previously described17. Briefly, Splenic CD4+ T cells (2 × 106 cells/well) and BMCMCs (5 × 105 cells/well), placed in a 24-well plate, were cultured in 0.5 mL/well culture medium with or without 100 ng/mL rmIL-10 in the presence of 1 µg/mL anti-mouse CD3ε mAb (145-2C11; eBioscience) and 1 µg/mL IgE (SPE-7) at 37 °C for 3 days. Thereafter, the cells were stimulated with 0.1 µg/mL phorbol 12-myristate 13-acetate (PMA; Sigma-Aldrich) plus 1 µg/mL Ionomycin (Sigma-Aldrich) in the presence of 1 µM monensin (Sigma-Aldrich) for 6 h. After cultivation, intracellular IL-17 A and Foxp3 expression in c-kit-negative and CD4-positive cells were analyzed by FACSCanto II (BD Biosciences) using FlowJo software (BD Biosciences). Purified anti-mouse CD16/CD32 (2.4G2), fluorescently labeled anti-mouse CD4 (GK1.5), IL-17 (TC11-18H10.1), c-kit (2B8), and Foxp3 (FJK-16s) mAbs were purchased from BD Biosciences (San Jose USA, CA).
Statistics
Data are represented as the mean ± SEM. A two-tailed unpaired t-test was performed to determine the statistical significance using GraphPad Prism software (San Diego, CA, USA). P-values (p < 0.05) was considered statistically significant.
Data availability
All data generated or analyzed during this study are included in this article. Further details are available from the corresponding author upon request.
References
Numata, T., Harada, K. & Nakae, S. Roles of mast cells in cutaneous diseases. Front. Immunol. 13, 923495. https://doi.org/10.3389/fimmu.2022.923495 (2022).
Kalesnikoff, J. et al. Monomeric IgE stimulates signaling pathways in mast cells that lead to cytokine production and cell survival. Immunity 14, 801–811 (2001).
Lu-Kuo, J. M., Austen, K. F. & Katz, H. R. Post-transcriptional stabilization by interleukin-1beta of interleukin-6 mRNA induced by c-kit ligand and interleukin-10 in mouse bone marrow-derived mast cells. J. Biol. Chem. 271, 22169–22174. https://doi.org/10.1074/jbc.271.36.22169 (1996).
Pestka, S. et al. Interleukin-10 and related cytokines and receptors. Annu. Rev. Immunol. 22, 929–979. https://doi.org/10.1146/annurev.immunol.22.012703.104622 (2004).
Fiorentino, D. F., Bond, M. W. & Mosmann, T. R. Two types of mouse T helper cell. IV. Th2 clones secrete a factor that inhibits cytokine production by Th1 clones. J. Exp. Med. 170, 2081–2095 (1989).
Saraiva, M. & O’Garra, A. The regulation of IL-10 production by immune cells. Nat. Rev. Immunol. 10, 170–181. https://doi.org/10.1038/nri2711 (2010).
Yeatman, C. F. 2 et al. Combined stimulation with the T helper cell type 2 cytokines interleukin (IL)-4 and IL-10 induces mouse mast cell apoptosis. J. Exp. Med. 192, 1093–1103 (2000).
Polukort, S. H. et al. IL-10 enhances IgE-Mediated mast cell responses and is essential for the development of experimental Food Allergy in IL-10-Deficient mice. J. Immunol. 196, 4865–4876. https://doi.org/10.4049/jimmunol.1600066 (2016).
Arock, M., Zuany-Amorim, C., Singer, M., Benhamou, M. & Pretolani, M. Interleukin-10 inhibits cytokine generation from mast cells. Eur. J. Immunol. 26, 166–170. https://doi.org/10.1002/eji.1830260126 (1996).
Stassen, M. et al. Murine bone marrow-derived mast cells as potent producers of IL-9: costimulatory function of IL-10 and kit ligand in the presence of IL-1. J. Immunol. 164, 5549–5555. (2000).
Qayum, A. A. et al. IL-10-Induced miR-155 Targets SOCS1 To Enhance IgE-Mediated Mast Cell Function. J. Immunol. 196, 4457–4467. https://doi.org/10.4049/jimmunol.1502240 (2000).
He, S. et al. Expression and release of IL-29 by mast cells and modulation of mast cell behavior by IL-29. Allergy. 65, 1234–1241. https://doi.org/10.1111/j.1398-9995.2010.02349.x (2010).
Ho, L. H. et al. IL-33 induces IL-13 production by mouse mast cells independently of IgE-FcepsilonRI signals. J. Leukoc. Biol. 82, 1481–1490. https://doi.org/10.1189/jlb.0407200 (2007).
Matsuda, K. et al. Monomeric IgE enhances human mast cell chemokine production: IL-4 augments and dexamethasone suppresses the response. J. Allergy Clin. Immunol. 116, 1357–1363. https://doi.org/10.1016/j.jaci.2005.08.042 (2005).
McGeachy, M. J. et al. TGF-beta and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain T(H)-17 cell-mediated pathology. Nat. Immunol. 8, 1390–1397 (2007).
Nakae, S. et al. Mast cells enhance T cell activation: importance of mast cell-derived TNF. Proc. Natl. Acad. Sci. U S A. 102, 6467–6472 (2005).
Morita, H. et al. An interleukin-33-Mast cell-Interleukin-2 Axis suppresses Papain-Induced allergic inflammation by Promoting Regulatory T Cell Numbers. Immunity. 43, 175–186. https://doi.org/10.1016/j.immuni.2015.06.021 (2015).
Berg, D. J. et al. Enterocolitis and colon cancer in interleukin-10-deficient mice are associated with aberrant cytokine production and CD4(+) TH1-like responses. J. Clin. Invest. 98, 1010–1020. https://doi.org/10.1172/jci118861 (1996).
Mäkelä, M. J. et al. IL-10 is necessary for the expression of airway hyperresponsiveness but not pulmonary inflammation after allergic sensitization. Proc. Natl. Acad. Sci. U S A. 97, 6007–6012. https://doi.org/10.1073/pnas.100118997 (2000).
Heo, Y. J. et al. IL-10 suppresses Th17 cells and promotes regulatory T cells in the CD4 + T cell population of rheumatoid arthritis patients. Immunol. Lett. 127, 150–156. https://doi.org/10.1016/j.imlet.2009.10.006 (2010).
Huber, S. et al. Th17 cells express interleukin-10 receptor and are controlled by Foxp3⁻ and Foxp3 + regulatory CD4 + T cells in an interleukin-10-dependent manner. Immunity. 34, 554–565. https://doi.org/10.1016/j.immuni.2011.01.020 (2011).
Numata, T., Ito, T., Maeda, T., Egusa, C. & Tsuboi, R. IL-33 promotes ICAM-1 expression via NF-kB in murine mast cells. Allergol. Int. 65, 158–165. https://doi.org/10.1016/j.alit.2015.10.004 (2016).
Meurer, S. K. et al. Isolation of mature (Peritoneum-Derived) mast cells and immature (bone Marrow-Derived) mast cell precursors from mice. PloS One. 11, e0158104. https://doi.org/10.1371/journal.pone.0158104 (2016).
Numata, T. et al. IL-36alpha is involved in hapten-specific T-cell induction, but not local inflammation, during contact hypersensitivity. Biochem. Biophys. Res. Commun. 506, 429–436. https://doi.org/10.1016/j.bbrc.2018.10.104 (2018).
Acknowledgements
We thank Keisuke Oboki, Naoki Kajiwara, Michiko Yamada, and Noriko Hashimoto for technical assistance.This work was supported by Grants-in-Aid for Scientific Research (B) (18H02847, 21H02963 to SN) and Scientific Research (C) (22K08439 to HS; 24K11583 to KA; 23K07795 to KH) from the Japan Society for the Promotion of Science. and National Institute of Biomedical Innovation (ID 05-24; HS).
Author information
Authors and Affiliations
Contributions
TN, MI and SN designed the experiments. TN, MI, KA, TO, K. Okada, and SN performed the experiments. TY, KS, HS, K. Okumura, HS and KH provided resources. TN, MI and SN wrote the manuscript. All authors reviewed the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Numata, T., Ikutani, M., Arae, K. et al. IL-10 promotes Th17 cell differentiation by enhancing STAT1-dependent IL-6 production via IgE-stimulated mast cells. Sci Rep 14, 26706 (2024). https://doi.org/10.1038/s41598-024-77929-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-77929-y
Keywords
This article is cited by
-
Multiomic Integration Reveals Taxonomic Shifts Correlate to Serum Cytokines in an Antibiotics Model of Gut Microbiome Disruption
Cellular and Molecular Bioengineering (2025)