Abstract
Alcohol-based fuels have shown high compatibility with spark-ignition (SI) engines, which require improvements in fuel efficiency and emissions reduction to meet modern environmental standards. While extensive research has been conducted on ethanol and other lower-order alcohols, there has been comparatively limited investigation into higher-order alcohols like butanol and pentanol as fuel alternatives. Previous studies on pentanol-gasoline blends in SI engines have demonstrated improved engine performance and reduced emissions. Building on this, the present study focuses on analyzing the flame characteristics—specifically speed and distribution—of pentanol-gasoline blends within the engine. In this study, pentanol was blended with gasoline by the volume of 10%, 20%, and 30%, namely 1-PNL10, 1-PNL20, and 1-PNL30, and tested in a twin-cylinder gasoline engine with an MPFI system at various load conditions. The study has focused on investigating the flame propagation of gasoline-pentanol blends by examining the in-cylinder flame image. The in-cylinder combustion evolution was visualized and captured by using an AVL Visio scope camera. Flame characteristics such as spatial flame distribution and flame speed were evaluated from the captured flame images for pentanol–gasoline blends and compared with sole gasoline. The flame study indicates that the addition of pentanol favored to increase in the flame speed, which in turn improved the combustion rate. The flame intensity and distribution area increased with the addition of pentanol in gasoline, demonstrating improved in-cylinder combustion with increased peak in-cylinder pressure and heat release rate. The insights on the flame characteristics of pentanol–gasoline blends were used to rationalize the discussion on engine performance and emissions. The performance of the engine was enhanced while increasing the proportion of Pentanol in the gasoline. The 30% Pentanol gasoline blend showed 5.71% higher BTE than gasoline at full load condition. Emissions like CO and HC also decreased at the same time, and NO emission increased. From the test results, it can be concluded that Pentanol can be blended with gasoline up to 30% without any engine modifications.
Similar content being viewed by others
Introduction
The increased number of motor vehicles and stringent emission regulations push to search for alternative fuels for internal combustion engines (ICE). The battery electric vehicles (BEV’s) could overcome the dependence on fossil-based fuels, but the penetration of BEV’s into the market is very slow, especially in highly populated developing nations. Furthermore, unless or until the electricity used for charging batteries is renewable, BEV’s are not carbon neutral. Before the infrastructure is adopted for the full uptake of BEV, ICEs would continue to evolve with different other measures to combat the emissions1. One such measure is using renewable low-carbon fuels with and without inherent oxygen, which has the advantage of reducing both the carbon intensity and air pollutants2. Biofuels play a major role not only in reducing emissions but also in increasing the performance of the engine, and they support to maintenance of the ecological balance3,4. Alcohol fuels are strongly recommended alternative fuels for gasoline ICE engines because of their favourable fuel properties and octane rating5,6. Alcohols can be blended with gasoline without any difficulties: the gasoline-alcohol blend can enhance the resistance of knocking and increase the performance of the engine. Alcohols have low molecular weight and high oxygen content, which results in clean combustion and reduced emissions such as carbon monoxide and hydrocarbon7,8.
Ethanol, methanol, and butanol are first-order alcohols with suitable chemical structures for use in the engine9. A lot of engine studies are carried out on the above fuels in the gasoline engines, particularly the ethanol was already commercialized10. Bioethanol, also referred to as ethyl alcohol, is derived from biomass feedstocks and is classified as a renewable, oxygenated fuel additive for gasoline. It has the potential to reduce harmful emissions by promoting cleaner combustion and contributing to the overall reduction of greenhouse gases11. An experimental study12 was conducted to evaluate the performance of ethanol-premium gasoline blends in a four-stroke, four-cylinder MPFI spark-ignition engine. The tests used pure regular gasoline (E0R) and ethanol blends (E6.25P, E10P, and E20P) at engine speeds of 2200, 3200, and 4200 rpm under varying loads. Results showed that BMEP, BT, BTE, and BSFC improved with increasing speed up to 3200 rpm, while BP increased with higher loads up to 4200 rpm.
Mohammed et al.13 studied the performance and emission characteristics of single-cylinder SI engines fuelled by gasoline-ethanol blends, and the experimental result showed a 28.5% increase in brake thermal efficiency (BTE) compared to sole gasoline. Ethanol is a highly oxygenated biofuel, and the physical and chemical properties of ethanol with improved octane rating enable it to emit low emissions and enhance the performance of the SI engine 14,15. Butanol is also a suitable alternative fuel for use in gasoline engines because its physical and chemical properties are more relevant to gasoline than the likes of ethanol and methanol16. Feng et al.17 investigated on single-cylinder motorcycle engine fuelled by pure gasoline and 35% butanol (1% volume of water added with butanol) under different load conditions. The 35% butanol-gasoline blend showed more positive results than sole gasoline with higher brake torque and lower HC emission.
Most studies in the literature have primarily explored the use of methanol or ethanol blended with gasoline. However, pentanol, also referred to as pentan-1-ol, n-amyl alcohol, or n-pentanol, emerges as a prominent higher-order alcohol gaining attention as a potential alternative fuel for spark ignition (SI) engines18. Additionally, pentanol, a straight-chain alcohol, is gaining increased attention due to its advantages over other alcohols. Despite its longer molecular structure, pentanol requires less energy to produce than shorter-chain alcohols19,20. The use of higher carbon chain alcohols was previously discouraged due to factors such as high production costs, limited availability of non-petroleum sources, and their restricted practical applications in the food industry21,22. In recent years, there has been a resurgence of interest in higher-order alcohols as renewable and sustainable fuels for engines, driving increased production. Advances in fermentation processes, including the use of new Clostridium strains, and biosynthesis from glucose through genetically engineered microorganisms like Saccharomyces cerevisiae, have enabled the production of alcohols like butanol and pentanol from cellulose23,24. During the synthesis of higher-order alcohols, various types of biomass can be steam reformed, gasified, or partially oxidized, producing syngas components such as CO, CO2, and H2. These gases are then catalytically converted into higher-order alcohols with longer carbon chains. Additionally, alternative methods like photosynthetic CO2 recycling or direct electro-microbial conversion have been employed, allowing the transformation of CO2, a greenhouse gas, into higher-order alcohols without the need for biomass breakdown. Furthermore, biofuel producers are optimizing existing biochemical pathways to lower costs and enable large-scale commercial production of these alcohols23,25.
A study reported that higher-order alcohols performed better with gasoline engines in an SI engine than lower-order alcohols26. Pentanol is a higher-order alcohol produced from agricultural renewable raw materials, and its energy content is higher than lower carbon alcohols27. Pentanol is more suitable for SI engines, as engine combustion and performance are improved with the increase in the proportion of pentanol in gasoline, as pentanol has superior physical and chemical properties than ethanol 28,29. The flame speed of pentanol is faster, resulting in faster combustion and increased BTE30. Pentanol is a promising fuel because of its long carbon chain compared to other alcohols and OH content supported for better combustion31. Gravalos et al.32 reported the emission characteristics of ethanol, methanol, propanol, butanol, and pentanol blended with gasoline in various proportions in a single-cylinder SI engine loaded, and the test result showed reduced HC and CO emissions compared to neat gasoline. Yaman et al.20 examined the performance and emission behaviours of single-cylinder port fuel injection SI engine fuelled by Pentanol and gasoline blends. Pentanol was blended with gasoline by volume of 5%, 10%, 15%, and 20%, and the engine was operated at a constant speed (1600 rpm) at different load conditions. The results showed that a 20% blend of pentanol in gasoline showed 10.5% higher BTE than gasoline. The presence of oxygen in pentanol was reported to reduce the CO and HC emissions for all Pentanol gasoline blend fuels.
Several studies reported using endoscopic techniques for spatial visualization of combustion evolution in engines. The visualization technique is also used to investigate the spray characteristics in both SI and CI engines. The endoscope is installed on top of the engine head with small modifications to provide a direct view of the combustion chamber. A light source is typically used to capture the fuel spray, and the flame propagation is captured using a suitable camera. Karimi33 studied the spray characteristics of ethanol-gasoline blend and also evaluated the flame propagation speed in a gasoline direct engine from the images captured through the endoscopic visualization technique. Agarwal et al.34 stated that the optical visualization technique is the most important method to resolve the combustion and spatial visualization and obtain valuable information about the combustion phenomenon of the engine. Alam et al.35 compared the endoscopic combination images of oxygenated fuel blends with 100% diesel, and the study reported the finding of decreased flame luminosity with higher oxygen fuels. Vikneswaran et al.36 evaluated the combustion flame propagation of gasoline additive with 1,4 Dioxane and reported notable improvement in the combustion flame with the addition of 1,4 dioxane. Ravikumar et al.37 studied the flame speed of gasoline-2 butoxyethanol blend by using a visioscope system and reported that the luminosity of the blend was increased with the addition of 2 butoxyethanol in the blend and flame speed of 97.5% gasoline + 2.5% butoxyethanol blend was reportedly 1.4% higher flame speed than sole gasoline.
Based on the above discussion, a lot of studies were carried out to explore the engine characteristics of gasoline–pentanol blends in an SI engine. While there has been extensive research on gasoline-pentanol blends, focusing on engine characteristics such as performance, emissions, and combustion, this study introduces a novel approach by concentrating primarily on combustion aspects. Specifically, it focuses on flame propagation, spatial flame distribution within the engine cylinder at different stages of the combustion phase, and the derivation of temperature distribution from flame distribution images. These characteristics were analyzed using the endoscopic visualization technique, providing a deeper and more comprehensive understanding of combustion dynamics in this study.
This study intends to investigate the impact of pentanol blending with gasoline on the spatial combustion flame characteristics in a multipoint fuel injection spark ignition engine. The captured flame images were processed to determine the flame distribution and flame speed. These fundamental results are then used to rationalize the findings from the engine tests for gasoline–pentanol blends. The fundamental flame characterization results are coupled with engine test results to provide more insights into pentanol–gasoline combustion characteristics.
Experimental setup and procedure
Research engine
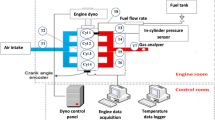
A twin cylinder four-stroke spark ignition engine with multi-point port fuel injection system was used for this research work. The SI engine has a compression ratio of 9.5 with a naturally aspirated airflow system. The engine is installed with an eddy current dynamometer, namely Schenck Avery ASE model 50. An S-type load cell was attached to one side of the dynamometer, which is cooled by water. The engine has a small port on the cylinder head for enclosing the endoscope. The injector nozzle has 3 holes, and the port fuel injection system injects fuel at 5 bar pressure. Electronic control unit (ECU) was used for controlling and determining the quantity of fuel injected at each load. Engine in-cylinder pressure was measured using a pressure transducer with the help of a crank angle encoder; the crank angle encoder was coupled with a crankshaft with a resolution of 0.5°. The pressure measurement was taken for 100 consecutive cycles and recorded by AVL Indicom software. The pressure data is processed to determine the heat release rate and other derived combustion parameters. AVL di gas analyzer was used to measure the exhaust emissions such as HC, CO, and NO after the catalytic converter available in the engine setup. Figure 1 shows the schematic view of the engine setup with an endoscopic setup. The detailed engine specifications are mentioned in Table 1.
Schematic view of test engine with endoscopic system.
Blend preparation
The n-Pentanol was blended with gasoline in the ratio of 10%, 20%, and 30% by volume. The prepared blends were stirred by using a tech magnetic steering machine to ensure the homogeneity of the blends. To check the stability of the blends, it was kept for a few days at room temperature and were observed with no separation in the blends. The Pentanol was bought from the local chemical industry. The designation of the prepared blends is given in Table 2.
Engine operating procedure
The engine was operating in ideal condition fuelled by neat gasoline for removing previous test fuels, which were in the fuel line, and ensuring the steady state operating condition. After ensuring the engine condition, the test was started by using pentanol – gasoline blended fuels. During experiments, the energy input for each cycle is made equal, not the fuel injection quantity. Since different fuels have varying lower calorific values, the fuel injection quantity is adjusted accordingly to ensure that the energy input remains constant. This adjustment is managed through the ECU (Engine Control Unit) settings, which regulate the fuel injection rate to compensate for differences in energy content between the fuels used. Every test has been taken with an interval of two hours for the cooling purposes of the engine and vision scope window. The flame propagation was captured at full load condition. The engine was operated at five different load conditions such as 1.6, 3.2, 4.8, 6.4, and 8 kW brake power. The engine load was varied by the eddy current dynamo meter. For each and every load, the engine speed was adjusted by throttle to maintain the constant engine speed of 2500 rpm. Fuel consumption was noted down for 1 min at each load. AVL digas analyzer displayed the amount of emission was exhausted from tailpipe. All measurements were repeated three times, and the average value was used for the calculation to ensure the accuracy of the results.
Uncertainty analysis enhances the precision of experimental results by assessing errors originating from instrumentation, human factors, and environmental conditions. Table 3 outlines the uncertainties associated with the measuring devices utilized in the study. Prior to data collection, the engine was operated for 10–15 min to achieve a steady-state condition, minimizing potential errors. Each fuel test was conducted three times, and the mean values were calculated. The errors were quantified using the standard deviation (SD) formula, as detailed by Vikneswaran et al.38. The standard deviations for gasoline and pentanol blends across various engine brake powers are presented as error bars in Figs. 3, 5, 6, 7, 8, 9 and 10.
Fuel characteristics
Properties of the fuel blends were measured as per the standards of ASTM and listed in Table 4. Pentanol has physical and chemical properties that are favourable for blending with gasoline. While the viscosity is higher than gasoline, the lower boiling point of pentanol improves the evaporation characteristics of pentanol–gasoline blends. Pentanol has a slightly lower octane rating and calorific valve compared to gasoline. Unlike methanol or ethanol, pentanol does not suffer from the higher latent heat of vaporization and would not produce a cooling effect to affect the volumetric efficiency of the engine. The presence of oxygen is an added benefit, and the fraction of hydrogen increases with the increase in the proportion of pentanol in the gasoline blend, as evident from CNHS analysis in Table 5.
Endoscope
The in-cylinder visualization of combustion was performed by AVL Visio scope setup, and the flame propagation was captured at the end of the full load condition. An endoscope was mounted on the head of the cylinder in 800 inclined position, and a camera was fitted at another end of the endoscope. The camera is held by a flexible steel stand, and the camera lens is adjusted to focus point for required light conditions. The endoscope is covered by a steel sleeve, and the endoscope temperature is controlled by a cooled air supply through the steel sleeve at a pressure of 5 bar37. The inserted endoscope was covered by a quartz glass window flush mount at the cylinder head end to provide the field of view for capturing the flame propagation. The quartz window also protects the endoscope from high combustion temperatures. The spatial flame propagation was recorded at various crank angle positions for different pentanol–gasoline blends. Combustion events were recorded five times for each operating condition and the camera captured the image between 1400 K and 2800 K temperature ranges. The camera records combustion events by video, which is then converted into bitmap images by using inbuilt software provided in the visioscope software39,40,41.
Results and discussion
Flame propagation
The flame propagation was captured only at full load condition in the temperature range between 1400 K and 2400 K only39. The flame region was rounded by a white circle, and the remaining places were painted by black color to erase the reflection available in the captured images. The flames can be characterized from the color of the image as a premixed flame or diffusion flame. The image was processed by using MATLAB software40. In this study, the image was captured between the start and end of combustion. Figure 2 shows the flame propagation pattern observed with pentanol-gasoline blends. The flame luminosity increased by increasing the pentanol fraction in gasoline fuel, and flame speed increased for all the fuel blends, for the reason that pentanol has a higher carbon chain and oxygen content. In addition, the percentage of hydrogen in the blend increased with the increase in the proportion of pentanol (Table 5). Given that the flame speed of hydrogen is faster, the pentanol-gasoline blends showed improved flame speed. When pentanol is blended with gasoline, the increased oxygen level for each blend promotes combustion and shortens the burn rate than sole gasoline. The improved combustion with a faster burn rate for the aforementioned reasons resulted in increased flame distribution, as shown in the processed images.
Photo graphic view of flame propagation of pentanol gasoline fuel blend for various crank angle (oCA).
Figure 3 shows the flame propagation region of gasoline and pentanol-gasoline blends. 1-PNL10, 1-PNL20, and PNL-30 blends occupied more flame area at CA 22o compared to gasoline. In particular, 1-PNL30 showed a higher flame distribution area than gasoline, as well as 1-PNL10 and 1-PNL20. After CA 22o, the combustion region was reduced because the fuel blends were burned near the spark plug region. As seen from the figure, the flame distribution decreases progressively with the increase in the proportion of pentanol in blends. This is because more amount of fuel mixture burnt ahead with an increase in pentanol ratio in the blend, resulting in lower combustion region at later stages.
Region occupied flame within endoscope widow by pentanol-gasoline blends at various crank angle.
The bright white-yellow color flame highlighted by the red color boundary in Fig. 2, represents the diffusion flame in the image. The diffusion flame was identified based on the Hue (H) number. Hue number varies between 0 and 80 for diffusion flames42. Firstly, the RGB (Red, Green, Blue) color scheme was converted into an HSV (Hue, Saturation, value) model. Hue (H) value is unique, corresponding to each RGB combination. RGB values obtained from each pixel in the flame image were converted into one H value by the equations given in the literature43. The diffusion flame fraction decreased with the increase in the fraction of pentanol in the blend compared to 100% gasoline. This indicates that the mixture formation is comparatively better with pentanol-gasoline blends due to the presence of inherent oxygen and better evaporation characteristics of pentanol compared to gasoline.
Figure 4 represents the temperature distribution contour for pentanol-gasoline blends with respect to various crank angle positions. The contour was obtained from the flame images given in Fig. 2 using a method called the correlated color temperature method. The complete details about the CCT method and equation for calculating temperature are given in the literature43. It is observed from the contour that for gasoline fuel, the region is dominated by the distribution of temperature ranges between 2600 and 2400 K. Whereas, for pentanol blends, the dominated by the temperature distribution ranges between 2300 and 2100 K, which is lesser by 100–300 K compared to that of gasoline. This is because of better distribution of air-fuel mixture and higher latent heat of vaporization of the pentanol blends compared to sole gasoline fuel. The presence of inherent oxygen in pentanol improved the mixture distribution, i.e., making the air-fuel ratio closer to the stoichiometric ratio. For a mixture closer to a stoichiometric ratio, the temperature ranges between 1800 and 2200 K. Further, comparatively higher latent heat of pentanol induces a cooling effect, which in turn improves the volumetric efficiency and thereby contributes to better mixture formation and, at the same time, reduces the overall combustion temperature. As a result of this, the temperature distribution seems comparatively lesser for pentanol blends than gasoline. The diffusion flame regions showed a peak temperature of 2600 K for all test fuels.
Temperature distribution contour of pentanol gasoline fuel blends for various proportions with respect tooCA.
Brake thermal efficiency
Brake thermal efficiency (BTE) is plotted with respect to different brake powers, as shown in Fig. 5. The experimental results showed the BTE of the engine slightly increased while increasing the volume of Pentanol in the fuel blend. The brake thermal efficiency was 26.2%, 26.8%, 27.2%, and 27.8% for gasoline and 1-PNL10 1-PNL20 1-PNL30, respectively at 8 kW brake power. It is found that the BTE is increased by 2.2%, 3.8%, and 6.1%, respectively, for 1-PNL10, 1-PNL20 and 1-PNL30 when compared to gasoline fuel at 8 kW. Pentanol’s physio-chemical properties are conducive to operation in SI engines. In particular, Pentanol has a higher calorific value comparatively than other higher-order alcohols44. Pentanol is an oxygenated fuel with a long carbon chain, which enhances the combustion rate and improves engine performance. Its molecular structure includes an -OH group, contributing to faster flame propagation, which is evident from the flame distribution images given in Fig. 2, as the blend ratio in the fuel increases. The presence of the -OH group promotes the formation of H free radicals, further accelerating the combustion process and boosting overall combustion efficiency45.
Brake thermal efficiency of pentanol-gasoline blends.
Heat release rate
Figure 6 shows the heat release rate of various pentanol-gasoline fuel blends and sole gasoline with respect to crank angle at maximum load condition. HRR values are derived by using the first law of thermodynamic values taken at full load condition. Compared to the gasoline, the 1-PNL30 showed a higher heat release rate. The addition of pentanol in gasoline efficiently improved the combustion rate due to increased flame prorogation and faster flame speed, as demonstrated in the flame characterization study. N-pentanol with long carbon chain alcohol needs less activation energy to break the chain and release the energy. The highly oxygenated fuel burned closer to the spark plug and emitted more heat that spread faster throughout the combustion chamber than sole gasoline. The presence of excess oxygen molecules improved the combustion of fuel blends to increase the heat release rate, as also explained in other previous studies20. Combustion can be advanced for gasoline-pentanol blend than sole gasoline with the help of the OH group present to enhance the speed of the combustion. From the CHNS analysis, the hydrogen level ahead in all fuel blends helped to increase the rate of combustion. Thus, the combustion phasing is advanced to improve the burn rate and shorten the combustion duration when pentanol is blended with gasoline. The pentanol-gasoline fuel blends have higher calorific value than other lower alcohol–gasoline blends, and thus heat release is not compromised. The net energy release is increased for pentanol–gasoline blends, as evident from the late diffusion combustion phase for the pentanol blended gasoline fuels. Correlating the flame images after 22°CA with the heat release rate, the flame distribution decreases due to more pronounced diffusion burn with increased cumulative energy release. Given the fact that pentanol has a higher viscosity, the atomization and air/fuel mixing is slightly affected by the increase in the proportion of pentanol in the blend despite the comparable boiling point of pentanol with gasoline. This resulted in late diffusion burn as evident from the flame images and heat release rate.
Heat release rate of pentanol-gasoline blends with respect to crank angle.
In-cylinder pressure
The in-cylinder pressure for 1-PNL10, 1-PNL20, and 1-PNL30 is compared with sole gasoline. Figure 7 shows the pressure difference between fuel blends and gasoline with respect to crank angle at maximum load condition. It is observed that maximum cylinder pressure is 28.4, 29.9, 30, and 30.5 bar for gasoline, 1-PNL10, 1-PNL20, and 1-PNL30, respectively. The increased peak in-cylinder pressure for 1-PNL30 was due to the improved laminar speed of fuel blends than gasoline, as demonstrated in the flame characterization study. Pentanol has a lower adiabatic temperature than gasoline, which leads to decreased heat loss from the combustion, and hence the in-cylinder pressure is higher. The presence of oxygen and OH groups in pentanol also favoured accelerating the combustion. The higher the burn rate, the increased the in-cylinder pressure comparatively to gasoline, and thus higher the BTE, as explained before.
Cylinder Pressure of pentanol-gasoline blends with respect to crank angle.
HC emissions
Figure 8 shows the level of hydro-carbon emission emitted from the engine with respect to various brake power. All pentanol-gasoline blends showed lower HC emission than sole gasoline, with the PNL 30% blend showing the lowest HC emissions than other fuels. The Hydrocarbon emission of the gasoline and 1-PNL10, 1-PNL20, and 1-PNL30 are 48, 39, 35, and 31 ppm. The HC emission of 1-PNL30 is 35% lower than gasoline. Native oxygen and the faster flame speed of pentanol played a vital role in reducing the HC by improving the combustion efficiency. The major source of HC emission is incomplete combustion due to less homogeneity of the fuel, which leads to partially reacted particles31. The presence of oxygen content in the pentanol could increase the homogeneity of the fuel blend to increase the combustion rate and reduce the HC emission than sole gasoline. Finally, the increased peak in-cylinder pressure and temperature for pentanol blended gasoline fuels might reduce the incomplete air-fuel mixture quenched on the cylinder walls and escaped through the crankcase to lower the HC emission.
HC emission of pentanol-gasoline blends.
CO emission
Figure 9 shows the CO emission of the gasoline and test fuels with respect to the various brake power. CO emissions of 1-PNL10, 1-PNL20, and 1-PNL30 are 0.09%, 0.07%, and 0.06%, respectively. 3-PNL30 showed 57.14% lower CO emission than sole gasoline. CO emission trend is similar to the HC emission trend for pentanol–gasoline blends, with a progressive decrease in emissions with the increase in the proportion of pentanol in the blend. CO emissions are directly related to the amount of carbon available in the test fuel. Pentanol, with a longer carbon chain than other higher-order alcoholic fuels, has 18% oxygen content, which displaces some carbon with oxygen and hydrogen, thereby favoring to reduce of CO emissions. Better evaporation quality and homogeneity of fuel blend with addition oxygen ensures44. Oxygen content at high combustion temperature helped to increase the oxygenated CO into CO2. Pentanol has a lower boiling point than gasoline, which helps to improve the evaporation and air/fuel mixing inside the combustion chamber to enhance the combustion process.
CO emission of pentanol-gasoline blends.
NO emission
Combustion temperature is the turning factor of forming NO emission. Pressure and combustion duration also plays a role in forming NO emission. The addition of oxygen and a long carbon chain tends to increase the duration and advance the combustion. Figure 10 shows the formation of NO for all test fuels with respect to brake power. 1-PNL10, 1-PNL20, and 1-PNL30 has 22 ppm, 21 ppm, and 49 ppm NO emission. 100%gasoline emitted 136 ppm NO at the time of engine run at full load condition. In this study, all test fuels showed higher NO emissions than gasoline at all brake power. NO occurred when nitrogen reacted with oxygen at high in-cylinder temperature, with pentanol having high oxygen content, which increases the oxygen concentration in the fuel mixture to increase the NOX emission at higher in-cylinder temperature.
NO emission of pentanol-gasoline blends.
Conclusions
This study explored the flame characterization of pentanol – gasoline blend fuels. The flame characterization results were used to rationalize the findings from engine testing. The following are the major conclusions of this study.
-
1.
The flame propagation increased by increasing the Pentanol fraction with gasoline. The 30% pentanol-gasoline blend showed higher flame propagation than other fuel blends.
-
2.
The flame luminosity increased with increasing fraction of pentanol in gasoline. The addition of pentanol in gasoline favoured to increase in the flame speed, which improved the combustion rate, increased the peak in-cylinder pressure, advanced the combustion, and shortened the combustion duration.
-
3.
The flame distribution area increased with the increased fraction of pentanol up to a certain crank angle, and subsequently, the flame distribution area decreased.
-
4.
Pentanol gasoline blend showed higher brake thermal efficiency than sole gasoline. 30% Pentanol gasoline blend showed 5.71% higher BTE than gasoline at full load condition.
-
5.
Exhaust emissions like CO and HC were reduced by 57.14% and 35%, respectively, for the 30% Pentanol blend compared to 100% gasoline. But the NO emission increased by 19.05% more than sole gasoline.
In conclusion, the present study primarily brought out the flame characteristics such as flame distribution, flame propagation, and temperature distribution of gasoline-pentanol blends, comparing them to gasoline. It can be concluded that the pentanol-gasoline blends exhibited higher flame propagation, leading to a larger flame distribution in the initial stages of combustion than gasoline. This enhanced flame spread improved the combustion rate, resulting in higher thermal efficiency. The increased oxygen content in pentanol contributed to more complete combustion, leading to a reduction in CO and HC emissions. However, the higher in-cylinder temperatures associated with pentanol blends resulted in an increase in NO emissions. These combined effects highlight the trade-offs between improved combustion efficiency and NO formation when using pentanol as a fuel additive in gasoline engines.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Woo, S., Lee, J. & Lee, K. Experimental study on the performance of a liquefied petroleum gas engine according to the air fuel ratio. Fuel. 303, 121330 (2021).
Simsek, S. & Uslu, S. Investigation of the impacts of gasoline, biogas and LPG fuels on engine performance and exhaust emissions in di ff erent throttle positions on SI engine. Fuel. 279, 118528 (2020).
Kemal, M., Sayin, C. & Canakci, M. An evaluation of the use of alcohol fuels in SI engines in terms of performance, emission and combustion characteristics. Rev. 286, (2021).
Zhang, Z. et al. Performance and emission evaluation of a marine diesel engine fueled with natural gas ignited by biodiesel-diesel blended fuel. Energy. 256, 124662 (2022).
Duan, X. et al. E ff ects of natural gas composition and compression ratio on the thermodynamic and combustion characteristics of a heavy-duty lean-burn SI engine fueled with lique fi ed natural gas. Fuel. 254, 115733 (2019).
Yusri, I. M. et al. Alcohol based automotive fuels from first four alcohol family in compression and spark ignition engine: a review on engine performance and exhaust emissions. Renewable and Sustainable Energy Reviews. 77, 169–181 (2017).
Aghahossein, S. et al. Effects of dual-alcohol gasoline blends on physiochemical properties and volatility behavior. Fuel. 252, 542–552 (2019).
Rao, V., Kumar, A., Kozinski, J. & Applied Catalysis, A. General Alcohols as alternative fuels : an overview. Appl. Catal. Gen. 404, 1–11 (2011).
Wei, J. et al. Physicochemical properties and oxidation reactivity of exhaust soot from a modern diesel engine: Effect of oxyfuel type. Combust. Flame. 238, 111940 (2022).
Broustail, G., Seers, P., Halter, F. & Moréac, G. Mounaim-rousselle, C. Experimental determination of laminar burning velocity for butanol and ethanol iso-octane blends. Fuel. 90, 1–6 (2011).
Taghavifar, H., Koozegar, B. & Kheyrollahi, J. Application of composite TNA nanoparticle with bio-ethanol blend on gasoline fueled SI engine at di ff erent lambda ratios. Fuel. 277, 118218 (2020).
Verma, A., Dugala, N. S. & Singh, S. Experimental investigations on the performance of SI engine with ethanol-Premium gasoline blends. Mater. Today Proc. 48, 1224–1231 (2022).
Mohammed, M. K., Balla, H. H., Al-dulaimi, Z. M. H., Kareem, Z. S. & Al-zuhairy, M. S. Case studies in Thermal Engineering Effect of ethanol-gasoline blends on SI engine performance and emissions. Case Stud. Therm. Eng. 25, 100891 (2021).
Verma, A., Dugala, N. S. & Singh, S. Materials Today: Proceedings Experimental investigations on the performance of SI engine with Ethanol-Premium gasoline blends. Mater Today Proc. (2021). https://doi.org/10.1016/j.matpr.2021.08.255
Yakın, A. & Behc, R. Effect of different types of fuels tested in a gasoline engine on engine performance and emissions. International Journal of Hydrogen Energy. 46(66), 33325-33338 (2021).
Yusoff, M. N. A. M. et al. Performance and emission characteristics of a spark ignition engine fuelled with butanol isomer-gasoline blends. Transp. Res. D Transp. Environ. 57, 23–38 (2017).
Feng, R. et al. Experimental study on SI engine fuelled with butanol-gasoline blend and H2O addition. Energy Convers. Manag. 74, 192–200 (2013).
Kothare, C. B. et al. Performance improvement and CO and HC emission reduction of variable compression ratio spark-ignition engine using n-pentanol as a fuel additive. Alexandria Eng. J. 74, 107–119 (2023).
Şahin, S. Comparison of machine learning algorithms for predicting diesel/biodiesel/iso-pentanol blend engine performance and emissions. Heliyon. 9, (2023).
Yaman, H. & Yesilyurt, M. K. The influence of n-pentanol blending with gasoline on performance, combustion, and emission behaviors of an SI engine. Eng. Sci. Technol. Int. J. 24, 1329–1346 (2021).
Sabu, V. R., Thomas, J. J. & Nagarajan, G. Experimental investigation on the effects of multiple injections and EGR on: N -pentanol-biodiesel fuelled RCCI engine. RSC Adv. 10, 29498–29509 (2020).
Karabektas, M. & Yilancilar, M. S. Investigation of the effects of high-carbon alcohol addition to camelina oil methyl ester on the performance parameters and soot emission of a diesel engine. Int. J. Low-Carbon Technol. 17, 206–213 (2022).
Shirazi, S. A., Foust, T. D. & Reardon, K. F. Identification of promising alternative mono-alcohol fuel blend components for spark ignition engines. Energies (Basel). 13, 1–16 (2020).
Kothare, C. B., Kongre, S. & Bhope, D. V. Experimental investigation of effect of Gasoline-higher alcohol blend on performance characteristic of four stroke Spark Ignition engine at variable compression ratio. International Conference on Electrical, Electronics, and Optimization Techniques, ICEEOT 2016 27–33 (2016). https://doi.org/10.1109/ICEEOT.2016.7755182
Nguyen, X. P. et al. Biomass-derived 2,5-dimethylfuran as a promising alternative fuel: an application review on the compression and spark ignition engine. Fuel Process. Technol. 214, (2021).
Vinod Babu, V. B. M. & Murthy, M. Amba Prasad Rao, G. Butanol and pentanol: the promising biofuels for CI engines – A review. Renew. Sustain. Energy Rev. 78, 1068–1088 (2017).
Zhu, L., Xiao, Y., Cheung, C. S., Guan, C. & Huang, Z. Combustion, gaseous and particulate emission of a diesel engine fueled with n-pentanol (C5 alcohol) blended with waste cooking oil biodiesel. Appl. Therm. Eng. 102, 73–79 (2016).
Yesilyurt, M. K., Eryilmaz, T. & Arslan, M. A comparative analysis of the engine performance, exhaust emissions and combustion behaviors of a compression ignition engine fuelled with biodiesel/diesel/1-butanol (C4 alcohol) and biodiesel/diesel/n-pentanol (C5 alcohol) fuel blends. Energy. 165, 1332–1351 (2018).
Uslu, S. & Celik, M. B. Combustion and emission characteristics of isoamyl alcohol-gasoline blends in spark ignition engine. Fuel. 262, 116496 (2020).
Mani Sarathy, S. et al. A comprehensive experimental and modeling study of iso-pentanol combustion. Combust. Flame. 160, 2712–2728 (2013).
Duan, Y. et al. Oxidation kinetics of n-pentanol: a theoretical study of the reactivity of the 1hydroxy-1-peroxypentyl radical. Combust. Flame. 219, 20–32 (2020).
Gravalos, I. et al. Comparison and analysis of the emissions of a small non-road spark-ignition engine operating under different alcohol–gasoline blended fuels. Int. J. Sustain. Energ. 35, 258–266 (2016).
Allocca, L., Catapano, F., Montanaro, A., Sementa, P. & Vaglieco, B. M. Study of E10 and E85 effect on Air Fuel Mixing and Combustion process in Optical Multicylinder GDI Engine and in a Spray Imaging Chamber. (2018). https://doi.org/10.4271/2013-01-0249
Agarwal, A. K., Agarwal, A. & Singh, A. P. Time resolved in-situ biodiesel combustion visualization using engine endoscopy. Meas. (Lond). 69, 236–249 (2015).
Alam, M., Song, J., Zello, V. & Boehman, A. Spray and combustion visualization of a direct-injection diesel engine operated with oxygenated fuel blends. Int. J. Engine Res. 7, 503–521 (2006).
Vikneswaran, M., Saravanan, C. G. & Sasikala, J. Endoscopic visualization of combustion flame to study the effect of 1,4-dioxane as an additive on the spatial flame characteristics of spark ignition engine. Fuel. 276, 118072 (2020).
Ravikumar, V. et al. Study on the effect of 2-butoxyethanol as an additive on the combustion flame, performance and emission characteristics of a spark ignition engine. Fuel. 285, 119187 (2021).
Vikneswaran, M. et al. A study on the feasibility of bergamot peel oil-gasoline blends for spark-ignition engines. J. Clean. Prod. 130515 https://doi.org/10.1016/j.jclepro.2022.130515 (2022).
Manoj Babu, A. et al. Analysis of performance, emission, combustion and endoscopic visualization of micro-arc oxidation piston coated SI engine fuelled with low carbon biofuel blends. Fuel. 285, 119189 (2021).
Narayanamoorthy, R., Sivaprakasam, S., Saravanan, C. G., Sivaraj, P. & Vikneswaran, M. Experimental investigation of 2-methyl furan as an additive with camphor blended gasoline blend for SI engines. Fuel. 306, 121748 (2021).
Saravanan, C. G. et al. Experimental study of feasibility of orange peel oil as a partial replacement for gasoline fuel in SI engine with and without MAO coated piston. Fuel. 315, 123173 (2022).
Huang, H. W. & Zhang, Y. Dynamic application of digital image and colour processing in characterizing flame radiation features. Meas. Sci. Technol. 21, (2010).
Agarwal, A. K. et al. Spatial combustion analysis of biodiesel fueled engine using combustion chamber endoscopy and modeling. Renew. Energy. 98, 292–303 (2016).
Pan, M. et al. Effect of EGR dilution on combustion, performance and emission characteristics of a diesel engine fueled with n-pentanol and 2-ethylhexyl nitrate additive. Energy Convers. Manag. 176, 246–255 (2018).
Biswal, A. et al. An experimental and kinetic modeling study of gasoline/lemon peel oil blends for PFI engine. Fuel. 267, 117189 (2020).
Acknowledgements
This project was supported by Researchers Supporting Project number (RSP2025R5), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
S. K, C.G. S, M. V- Research Concept and DesignVR, J. S - Collection and Assembly of dataFJJS, SAA, AP-Data analysisEGV, HLA - writing the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kumaravel, S., Saravanan, C.G., Vikneswaran, M. et al. Exploration of flame characteristics of gasoline engine fuelled by gasoline-pentanol blends using combustion endoscopy. Sci Rep 14, 31692 (2024). https://doi.org/10.1038/s41598-024-81221-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-81221-4