Abstract
Fluid administration is widely used to treat hypotension in patients undergoing veno-venous extracorporeal membrane oxygenation (VV-ECMO). However, excessive fluid administration may lead to fluid overload can aggravate acute respiratory distress syndrome (ARDS) and increase patient mortality, predicting fluid responsiveness is of great significance for VV-ECMO patients. This prospective single-center study was conducted in a medical intensive care unit (ICU) and finally included 51 VV-ECMO patients with ARDS in the prone position (PP). Stroke volume index variation (ΔSVI), pulse pressure variation (PPV), stroke volume variation (SVV), baseline carotid corrected flow time (FTcBaseline), and respirophasic variation in carotid artery blood flow peak velocity (ΔVpeakCA) were taken before and after the Trendelenburg position or volume expansion. Fluid responsiveness was defined as a 15% or more increase in stroke volume index as assessed by transthoracic echocardiography after the volume expansion (VE). In our study, 33 patients (64.7%) were identified as fluid responders. Stroke volume index variation induced by the Trendelenburg position (ΔSVITrend), FTcBaseline, and ΔVpeakCA demonstrated superior predictive performance of fluid responsiveness. ΔSVITrend had an AUC of 0.89 (95% CI, 0.80–0.98) with an optimal threshold of 14.5% (95% CI, 12.5–21.5%), with the sensitivity and specificity were 82% (95% CI, 66–91%) and 83% (95% CI, 61–94%). FTcBaseline had an AUC of 0.87 (95% CI, 0.76–0.98) with an optimal threshold of 332ms (95% CI, 318-335ms), the sensitivity and specificity were 85% (95% CI, 69–93%) and 83% (95% CI, 61–94%), respectively. ΔVpeakCA showed an AUC of 0.83 (95% CI, 72–95), with a 10% optimal threshold (95% CI, 9–13%), sensitivity was 82% (95% CI, 66–91%) and specificity 78% (95% CI, 55–91%). ΔSVITrend, FTcBaseline and ΔVpeakCA could effectively predict fluid responsiveness in VV-ECMO patients with ARDS in the PP. Compared to ΔSVITrend and ΔVpeakCA, FTcBaseline is easier and more direct to acquire, and it does not require Trendelenburg position or VE, making it a more accessible and efficient option for assessing fluid responsiveness.
Similar content being viewed by others
Introduction
Veno-Venous Extracorporeal Membrane Oxygenation (VV-ECMO) serves as a potent tool for severe acute respiratory distress syndrome (ARDS) patients with poor response to conventional mechanical ventilation1,2,3,4,5. VV-ECMO serves as an artificial lung, facilitating extracorporeal gas exchange to sustain sufficient oxygenation and carbon dioxide elimination, with the objective of mitigating ventilator-induced lung injury and providing a window for primary disease treatment. Clinicians often utilize fluid administration during VV-ECMO support to bolster cardiac output (CO), thereby addressing hypotension and enhancing oxygen delivery6,7. Nevertheless, due to alterations in alveolar capillary membrane permeability, excessive fluid administration can predispose patients to complications such as pulmonary edema, thereby exacerbating the underlying condition8,9. Hence, the ability to predict fluid responsiveness is crucial for VV-ECMO patients with ARDS, as it informs fluid management strategies and mitigates the risk of exacerbating the condition10.
Commonly utilized methods for assessing volume responsiveness in critically ill patients include static indicators such as central venous pressure (CVP) and inferior vena cava (IVC), as well as dynamic indicators like stroke volume variation (SVV) or pulse pressure variation (PPV)11,12,13,14. CVP has historically been used to guide fluid therapy, but recent studies have shown that CVP cannot predict fluid responsiveness15. Additionally, in VV-ECMO patients, the placement of the venous drainage cannula limits the applicability of IVC assessment. Moreover, SVV and PPV, commonly used to assess volume responsiveness, necessitate mechanical ventilation with a tidal volume greater than 8 ml/kg predicted body weight (PBW)16,17. V-V ECMO patients typically undergo lung-protective ventilation with small tidal volumes, making them less suitable for assessing volume responsiveness in this patient population18.
Passive leg raising test has been proven to be feasible in predicting fluid responsiveness in VV-ECMO patients19. It through elevating the legs to simulate fluid expansion, primarily relying on the increased venous return when the patient is in the supine position. However, patients receiving VV-ECMO support for ARDS are often placed in the prone position (PP) to improve ventilation. In the PP, leg elevation is restricted by the body’s position. Therefore, passive leg raising is not applicable for assessing fluid responsiveness in patients in PP. In conclusion, the specificity and complexity of VV-ECMO patients in PP limits the applicability of traditional methods for assessing fluid responsiveness.
The Trendelenburg position is considered a “self” preload challenge20, similar in principle to passive leg raising, and can be applied to patients in the PP. Studies have demonstrated that the Trendelenburg position is suitable for patients in the PP and under ECMO21,22.
Carotid ultrasound measurements, such as baseline carotid corrected flow time (FTcBaseline), and respirophasic variation in carotid artery blood flow peak velocity (ΔVpeakCA), have recently been recognized as reliable indicators to predictive fluid responsiveness23,24,25. The measurements of these parameters offer advantages such as rapidity, non-invasiveness, and simplicity. However, it is worth noting that there are currently few studies in patients on VV-ECMO with ARDS in PP. The efficacy of these parameters in assessing fluid responsiveness in these patients remains not enough clear at present.
Methods
Study aim
The primary aim of this study was to evaluate the value of stroke volume index variation induced by the Trendelenburg position (ΔSVITrend), FTcBaseline, and ΔVpeakCA to predict fluid responsiveness in patients on VV-ECMO with ARDS in the PP. Secondary objectives were to evaluate the diagnostic performance of PPV and SVV to predict fluid responsiveness in these patients.
Study design
This prospective, single-center study was conducted in the intensive care unit (ICU) between May 2022 and February 2024. It was approved by the Institutional Review Board of Quzhou People’s Hospital (Quzhou, China: Number B 2022-083) and performed in accordance with the Declaration of Helsinki. All research was performed in accordance with relevant guidelines/regulations. Informed consent was obtained from all participants and/or their legal guardians.
Patients
VV-ECMO patients admitted to intensive care unit (ICU) were recruited in this study. The Inclusion criteria including: (1) adults (≥ 18 years) diagnosed with ARDS; (2) received prone ventilation therapy during VV-ECMO; (3) monitoring with the pulse indicator continuous cardiac output (PiCCO)® device (Pulsion Medical Systems, Feldkirchen, Germany); (4) patients who presented with at least one clinical sign of inadequate tissue perfusion for less than 24 h in the absence of a contraindication for fluids: Hypotension (systolic blood pressure < 90 mmHg or mean arterial pressure (MAP) < 65 mmHg), organ hypoperfusion (evidenced by mottled skin or delayed capillary refill), oliguria (urine output < 0.5 mL/kg/h), elevated lactate levels (> 2 mmol/L).
Exclusion criteria were patients with contraindications for volume expansion (VE) (severe cardiac dysfunction, pulmonary edema etc.), severe arrhythmias, age less than 18 years old, carotid artery stenosis > 50%, a poor ultrasonographic window for carotid doppler measurements, any contraindications to Trendelenburg position (head injury or intracranial hypertension, intraabdominal hypertension).
Study protocol
Throughout the study, patients were sedated with a combination of remifentanil and midazolam, with the aim of achieving a Ramsay score of 626, they were ventilated in volume-controlled mode with a tidal volume (VT) 4 to 6 ml/kg−1 PBW. This protocol included four sequential steps (Figs. 1 and 2):
-
(1)
Baseline-1: patient in a PP, with a 15° upward bed angulation;
-
(2)
Trendelenburg position: 15° downward bed angulation27.
-
(3)
Baseline-2: recover to the same position as baseline-1;
-
(4)
VE test: administration of 500 ml crystalloids over 15 min without postural change.
After each step is completed and stabilized for 1 min, record the hemodynamic parameters. For enhanced accuracy of the acquired data, measurements were taken three times for each patient, and the average was calculated to determine the final parameters.
The study flowchart.
The study protocols. (a) An outline of each step; (b) a clinical scenery of VV-ECMO patient in the prone position; (c) the 15° upward bed angulation for measurements at baselines and volume expansion test; (d) The 15° downward bed angulation for measurements in the Trendelenburg position.
Measurements
PiCCO
The PiCCO® catheter was inserted into the patient’s femoral artery. Using the GE B850 monitor equipped with a PiCCO® module (GE Healthcare, Chicago, USA), the following hemodynamic parameters were monitored: PPV, SVV.
Transthoracic echocardiography
The transthoracic echocardiography (TTE) (Vivid i, GE Healthcare, Chicago, IL, USA) is performed by a doctor with specialized ultrasound training for PP. Patient was placed in the left PP, left shoulder elevation with a pillow forms a space to put the transducer28. 1.5–4.5 MHZ phased array probe was the used for stroke volume examination. The diameter of the left ventricular outflow tract at the systolic aortic apex was measured by parasternal long axis echocardiography. The area of left ventricular outflow tract was calculated as π × (the square of the left ventricular outflow tract radius)29. Aortic blood flow velocity time integral (VTI) was calculated from the area under the pulse-wave Doppler signal envelope obtained from the apical five-chamber cutting surface at the level of the aortic ring and was determined by the average value of five consecutive pulses in a complete respiratory cycle. SVI was calculated as (left ventricular outflow tract area x aortic flow VTI)/body surface area (BSA), and BSA was calculated as BSA(m2) = 0.0061×body length (cm) + 0.0128×body weight (kg)-0.152930.
Carotid ultrasonography
ΔVpeakCA and FTc were measured by a doctor with specialized ultrasound training for PP. A variable-frequency linear array transducer with a range of 6-13 MHz was longitudinally positioned on the right side of the neck, with the transducer marker oriented towards the patient’s head. In the short-axis plane below the thyroid cartilage, the right carotid artery was located, and real-time B-mode images of the long axis of the right carotid artery were obtained. The ultrasound beam angle with the direction of blood flow was maintained below 60°. Pulse wave (PW) mode was selected, and after displaying the pulse Doppler spectrum, the optimal sampling volume and angle were adjusted to obtain a satisfactory spectral freeze-frame image. Measurements of carotid blood flow, including FTc (flow time corrected) and VpeakCA were taken. ΔVpeak-CA was calculated using the formula: ΔVpeakCA = (Max peak - Min peak) / [(Max peak + Min peak) / 2] × 100%. The cycle time (flow time, FT) from the onset of systole to the dual rotation trace was measured, and the FTc in the common carotid artery was calculated according to Wodey equation31: FTc = FT + [1.29 × (HR − 60)], where FT represents the cycle time from the onset of systole to the dual rotation trace, and heart rate (HR) is obtained by measuring the heartbeat interval at the onset of Doppler blood flow.
Data collection
Upon patient inclusion, demographic information, APACHE II scores, and etiology of ARDS were collected. Supportive treatments included VV-ECMO, mechanical ventilation (MV). We documented all data related to these parameters. Follow-up was conducted for all study patients until discharge or death, recording clinical outcomes such as the duration of MV, ECMO duration, ICU length of stay, and in-hospital mortality rate.
Statistical analysis
SVI measured before and after VE was used to distinguish responders from non-responders with ΔSVI of > 15% and < 15%, respectively32,33. The data were statistically analyzed using SPSS (version 26; IBM Corp., Armonk, NY, USA) and R software (version 4.4.1; R Foundation for Statistical Computing, Vienna, Austria), and figures were created using GraphPad Prism (version 10; GraphPad Software, San Diego, CA, USA). Continuous variables were expressed as mean ± SD or median (interquartile range). Compared between groups using the Wilcoxon or Friedman rank sum tests. For pairwise multiple comparisons, we used the Nemenyi post-hoc test. Categorical variables were expressed as numbers (and percentages) and compared using the Fisher’s exact test. Linear regression analysis was used to demonstrate relationships between percent change of stroke volume index variation induced by the Trendelenburg position (ΔSVITrend), and volume expansion. The receiver operating characteristic (ROC) curve analysis was performed to assess the ability of the five hemodynamic parameters (FTcBaseline, PPV, SVV, ΔVpeakCA and ΔSVITrend), to predict fluid responsiveness. Sensitivity, specificity, positive likelihood ratio (PLR) and negative likelihood ratio (NLR), and associated 95% confidence intervals (CI) were calculated based on the cutoff value as determined by the Youden Index (specificity + sensitivity − 1). To evaluate the variation of best threshold, we conducted a gray-zone analysis, as described by Joël Coste34. A value of P < 0.05 (two-tailed test) was considered statistically significant.
Results
Patient characteristics
From May 2022 to February 2024, we enrolled 71 VV-ECMO patients with ARDS in the PP to our study, 20 were excluded because of age<18years (n = 3), carotid artery stenosis > 50% (n = 6), intraabdominal hypertension (n = 4), head injury (n = 2), or poor ultrasonographic window (n = 5). Ultimately, 51 patients were included in this study (Fig. 1). Patient characteristics were comparable between responders (n = 33) and non-responders (n = 18) (Table 1).
Effect of Trendelenburg position and volume expansion
Fluid responsiveness was observed in 33 of the 51 measurements (64.7%). The hemodynamic variables are shown in are shown (Table 2). The SVI and FTc increase between responders and non-responders after Trendelenburg position or VE, whereas an increasing trend was more apparent in responders. In responders, Trendelenburg position or VE significantly decreased the PPV, SVV or ΔVpeakCA. However, there was no evident change in SVI, ΔVpeakCA, PPV or SVV in non-responders.
Prediction of fluid responsiveness
Data on fluid responsiveness predictions are shown (Figs. 3, 4, 5 and 6; Table 3). Median ΔSVITrend amounted to 20% (95% CI, 11–25%) and was significantly greater in responders than in non-responders (23% vs. 10.5%, P < 0.001) (Fig. 3). ΔSVITrend was significantly correlated with change in stroke volume index variation induced by the VE (ΔSVIVE) related to volume expansion (R2 = 0.39, P < 0.001). In comparison, the R2 of FTcBaseline, ΔVpeakCA, PPV and SVV were (R2 = 0.31, P < 0.05), (R2 = 0.19, P < 0.05), (R2 = 0.07, P < 0.05) and (R2 = 0.09, P < 0.05), respectively (Fig. 4). The area under ROC curve (AUC) of ΔSVITrend to predict fluid responsiveness was 0.89 (95% CI 0.80–0.98), with a sensitivity of 82% (95% CI 66–91%), and specificity of 83% (95% CI 61–94%), at a best threshold of 14.5%. The corresponding gray zone was 12.5–21.5%, covering 29% of patients (Figs. 5 and 6; Table 3).
Individual values from five parameters to detect fluid responsiveness in fluid responders and non-responders. Black dotted line displays optimal thresholds for each diagnostic test computed by ROC curve analysis. (a) ΔSVITrend, Stroke volume index variation induced by the Trendelenburg position; (b) FTcBaseline, baseline carotid artery corrected flow time; (c) ΔVpeakCA, respirophasic variation in carotid artery blood flow peak velocity; (d) PPV, pulse pressure variation; (e) SVV, stroke volume variation.
The relationship between ΔSVIVE and the five parameters.The grey line is the regression line. The gray dotted line area is the 95% confidence interval of the regression line. (a) ΔSVITrend, Stroke volume index variation induced by the Trendelenburg position; (b) FTcBaseline, baseline carotid artery corrected flow time; (c) ΔVpeakCA, respirophasic variation in carotid artery blood flow peak velocity; (d) PPV, pulse pressure variation; (e) SVV, stroke volume variation.
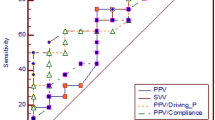
Receiver operating characteristics curves from five diagnostics tests to detect fluid responsiveness. (a) ΔSVITrend, Stroke volume index variation induced by the Trendelenburg position; (b) FTcBaseline, baseline carotid artery corrected flow time; (c) ΔVpeakCA, respirophasic variation in carotid artery blood flow peak velocity; (d) PPV, pulse pressure variation; (e) SVV, stroke volume variation.
Grey zone analysis. (a) ΔSVITrend, Stroke volume index variation induced by the Trendelenburg position; (b) FTcBaseline, baseline carotid artery corrected flow time; (c) ΔVpeakCA, respirophasic variation in carotid artery blood flow peak velocity; (d) PPV, pulse pressure variation; (e) SVV, stroke volume variation.
FTcBaseline had an AUC of 0.87 (95% CI, 0.76–0.98) with an optimal threshold of 332ms, the sensitivity and specificity were 85% (95% CI, 69–93%) and 83% (95% CI, 61–94%), respectively. The corresponding gray zone was 318–335ms, covering 29% of patients. ΔVpeakCA showed an AUC of 0.83 (95% CI, 72–95), with a 10% optimal threshold, sensitivity was 82% (95% CI, 66–91%) and specificity 78% (95% CI, 55–91%), the corresponding gray zone was 9–13%, covering 43% of patients.
PPV and SVV displayed lower predictive performance. The AUC were 0.75 (95% CI 0.61–0.89) and 0.76 (95% CI 0.61–0.90). The gray zone of optimal thresholds was 7.5% (95% CI 6.5–9.5%) and 11.5% (95%CI 10.5–14.5%), and included more patients in grey zone (39% and 41%), than ΔSVITrend and FTcBaseline.
Discussion
Our study evaluates the diagnostic value of several parameters to predict fluid responsiveness in Patients on VV-ECMO with ARDS in the PP under protective ventilation (VT 4–6 ml/kg−1 PBW). The main findings are that: (1) ΔSVITrend is a highly reliable parameter to predict fluid responsiveness, the AUC is 0.89 (95% CI 0.80–0.98), with sensitivity of 82% (95% CI 66–91%) and specificity of 83% (95% CI 61–94%); (2) FTcBaseline also is a highly reliable parameter to predict fluid responsiveness, which had an AUC is 0.87 (95% CI 0.76–0.98), with a higher sensitivity of 85% (95% CI 69–93%) than ΔSVITrend, and specificity of 83% (95% CI 61–94%) (3) While ΔVpeakCA also had acceptable predictive performance for fluid responsiveness, its AUC [0.83 (95% CI 0.72–0.95)], sensitivity [82% (95% CI 66–91%)], and specificity [78% (95% CI 55–91%)] are all inferior to those of ΔSVITrend and FTcBaseline (4) PPV and SVV is moderate sensitivity to predict fluid responsiveness, but with low specificity, and their AUC are also relatively low. This study enables clinicians to predict fluid responsiveness in such patients non-invasively, facilitating the development of appropriate fluid management strategies.
VV-ECMO is a valuable therapeutic option for severe ARDS with suboptimal response to protective lung ventilation1,35. During this support, clinicians typically administer appropriate fluid infusion to stabilize ECMO flow and correct hypotension, thereby increasing oxygen delivery36. However, Positive fluid balance is associated with a higher risk of death in ECMO patients9, excessive fluid administration can cause pulmonary edema and heart failure, exacerbating the condition of ARDS patients and ultimately leading to increased mortality37.Therefore, fluid management in VV-ECMO patients is closely intertwined with their condition. Accurate prediction of fluid responsiveness is crucial to improve prognosis for these patients.
Fluid responsiveness refers to the physiological response of patients to fluid loading12. The evaluation of fluid responsiveness has always been a hot and challenging area of research. Clinical indicators are diverse, ranging from static parameters such as CVP and IVC to dynamic parameters such as SVV and PPV. Evaluation indicators have undergone a transition from static to dynamic and from invasive to non-invasive. The purpose of our study of volume responsiveness was to improve organ tissue ischemia and hypoxia while avoiding volume overload, increasing mortality and hospitalization time.
CVP serving as a pressure indicator, can reflect preload depending on cardiac compliance. However, various factors such as heart failure, volume expansion, the use of vasopressors, or mechanical ventilation can alter cardiac compliance, leading to inaccuracies in preload assessment via CVP38. The study by Marik PE et al.15 also indicates that central venous pressure should not be used to guide fluid therapy in clinical practice.
PPV and SVV are dynamic parameters widely used to predict fluid responsiveness39,40. Nowadays, optimization strategies for fluid management are often based on these parameters, which can be easily obtained through invasive arterial pressure monitoring. However, some studies suggest that factors such as low VT (< 8 ml/kg−1 PBW) may influence the performance of PPV and SVV, especially in patients undergoing thoracoabdominal closure or positioned in the PP41,42. During the treatment of ARDS patients, mechanical ventilation can induce ventilator-induced lung injury (VILI) through various mechanisms, including volutrauma, barotrauma, and atelectrauma43. Therefore, guidelines from the Extracorporeal Life Support Organization (ELSO) recommend limiting tidal volume to less than 6 ml/kg to reduce the occurrence of VILI6, and supplementing with prone positioning ventilation during ECMO to treat moderate to severe ARDS44. Multiple studies45,46,47,48 have demonstrated that prone positioning during ECMO is feasible, safe, and can enhance ECMO weaning and improve outcomes. In our study, patients in the PP, combined with reduced chest wall compliance49 and low VT (4–6 ml/kg−1 PBW), resulted in lower sensitivity, specificity, and AUC values for PPV and SVV compared to other parameters. These findings align with previous research results21,24,50,51. PPV and SVV may not applicable for predicting fluid responsiveness in such patients.
In contrast, the application of FTcBaseline and ΔVpeakCA derived from carotid ultrasound has multiple advantages. Firstly, this technique is entirely non-invasive, allowing for easy assessment of a patient’s hemodynamic status through carotid artery ultrasound. Secondly, these parameters are applicable to patients with low VT (≤ 6 ml/kg−1 PBW) and cardiac arrhythmias41, unlike other dynamic markers such as PPV and SVV, which require higher tidal volumes and regular heart rhythms. Additionally, the measurement of FTcBaseline is not affected by changes in intrathoracic pressure during respiration52.
Trendelenburg position or passive leg raising are used either as a diagnostic tool to assess fluid responsiveness20,53. The physiological mechanisms of both methods are similar, involving changes in body position or elevation of the lower limbs to promote blood flow toward the heart, resulting in “autotransfusion,” increasing venous return to the heart, enhancing CO, and ultimately increasing organ perfusion. However, the passive leg raising test requires patients to remain in the supine position, whereas our study subjects were VV-ECMO patients in the prone position, making it fundamentally unsuitable. Additionally, there were some concerns about the safety of passive leg raising among ECMO patients. Previous studies19 have indicated that patients undergoing passive leg raising did not experience any adverse effects on the ECMO system, it is still important to consider that leg elevation may pose risks of catheter displacement or deformation, as well as vascular injury. Therefore, the Trendelenburg position may be more suitable for assessing fluid responsiveness in our study population. In our study, all patients underwent Trendelenburg position in the PP without any adverse impact on the ECMO system.
The Trendelenburg position and PP can significantly affect chest wall compliance, as well as respiratory system mechanics54, which may impact the assessment of fluid responsiveness using ultrasound. However, Yonis H et al.21 reported that the Trendelenburg maneuver is reliable to predict fluid responsiveness in patients with ARDS under protective ventilation in the prone position. Based on the results of our study, we have also drawn similar conclusions. The Trendelenburg position induced a 64.7% change in SVI, induced by VE, indicating a similar physiological effect to passive leg raising. The high correlation between ΔSVITrend and ΔSVIVE, along with the high sensitivity, specificity, AUC, and narrow gray zone of ΔSVITrend, suggests that the Trendelenburg position can be used to assess fluid responsiveness in patients undergoing VV-ECMO in the PP.
Conclusions
Our study provides robust evidence supporting ΔSVITrend, FTcBaseline and ΔVpeakCA are able to predict fluid responsiveness in patients on VV-ECMO with ARDS in the PP, while neither PPV and SVV reached acceptable predictive performance to predict fluid responsiveness. FTcBaseline can be directly obtained using neck ultrasound, eliminating the need Trendelenburg position or VE. This characteristic enhances its practicality in clinical settings, making it a more accessible and efficient option for assessing fluid responsiveness.
Limitations
Our study had several limitations. Firstly, since all patients were treated for ARDS, they required low tidal volume ventilation (4–6 ml/kg) as part of lung-protective strategies; thus, our findings may not extend to patients receiving higher tidal volumes (6–8 ml/kg or > 8 ml/kg). Secondly, we did not conduct comparisons with findings from non-prone positions, which may limit the generalizability of our results to other positional settings. Additionally, this study did not include other tests for fluid responsiveness, such as the Mini Fluid Challenge or the End-Expiratory Occlusion test. In future research, we aim to further explore these aspects to enhance fluid responsiveness assessment.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Combes, A. et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N. Engl. J. Med. 378, 1965–1975. https://doi.org/10.1056/NEJMoa1800385 (2018).
Bellani, G. et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 315, 788. https://doi.org/10.1001/jama.2016.0291 (2016).
Sud, S. et al. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: Systematic review and meta-analysis. Intensive Care Med. 36, 585–599. https://doi.org/10.1007/s00134-009-1748-1 (2010).
Balk, R. A. Positive end-expiratory pressure setting in adults with acute lung injury and acute respiratory distress syndrome: A randomized controlled trial. Yearbook Crit. Care Med. 2009, 6–8. https://doi.org/10.1016/S0734-3299(08)79045-2 (2009).
Schmid, C. et al. Venovenous extracorporeal membrane oxygenation for acute lung failure in adults. J. Heart Lung Transplantation. 31, 9–15. https://doi.org/10.1016/j.healun.2011.07.013 (2012).
Tonna, J. E. et al. Management of adult patients supported with venovenous extracorporeal membrane oxygenation (VV ECMO): Guideline from the extracorporeal life support organization (ELSO). ASAIO J. 67, 601–610. https://doi.org/10.1097/MAT.0000000000001432 (2021).
Kim, H. et al. Permissive fluid volume in adult patients undergoing extracorporeal membrane oxygenation treatment. Crit. Care. 22, 270. https://doi.org/10.1186/s13054-018-2211-x (2018).
Messmer, A. S. et al. Fluid overload and mortality in adult critical care patients—A systematic review and meta-analysis of observational studies*. Crit. Care Med. 48, 1862–1870. https://doi.org/10.1097/CCM.0000000000004617 (2020).
Schmidt, M. et al. Impact of fluid balance on outcome of adult patients treated with extracorporeal membrane oxygenation. Intensive Care Med. 40, 1256–1266. https://doi.org/10.1007/s00134-014-3360-2 (2014).
Douglas, I. S. et al. Fluid Response Evaluation in Sepsis Hypotension and Shock. Chest 158, 1431–1445. https://doi.org/10.1016/j.chest.2020.04.025 (2020).
Monnet, X., Marik, P. E. & Teboul, J-L. Prediction of fluid responsiveness: An update. Ann. Intensive Care. 6, 111. https://doi.org/10.1186/s13613-016-0216-7 (2016).
Jozwiak, M., Monnet, X. & Teboul, J-L. Prediction of fluid responsiveness in ventilated patients. Ann. Transl Med. 6, 352–352. https://doi.org/10.21037/atm.2018.05.03 (2018).
Monnet, X. & Teboul, J-L. Assessment of fluid responsiveness: Recent advances. Curr. Opin. Crit. Care. 24, 190–195. https://doi.org/10.1097/MCC.0000000000000501 (2018).
Huang, H. et al. Value of variation index of inferior vena cava diameter in predicting fluid responsiveness in patients with circulatory shock receiving mechanical ventilation: A systematic review and meta-analysis. Crit. Care. 22, 204. https://doi.org/10.1186/s13054-018-2063-4 (2018).
Marik, P. E. & Cavallazzi, R. Does the central venous pressure predict fluid responsiveness? An updated meta-analysis and a plea for some common sense*. Crit. Care Med. 41, 1774–1781. https://doi.org/10.1097/CCM.0b013e31828a25fd (2013).
Yang, X. & Du, B. Does pulse pressure variation predict fluid responsiveness in critically ill patients? A systematic review and meta-analysis. Crit. Care. 18, 650. https://doi.org/10.1186/s13054-014-0650-6 (2014).
Backer, D. D. & Vincent, J-L. Infuence of respiratory rate on stroke volume variation in mechanically ventilated patients. J. Am. Soc. Anesthesiol. 110(5), 1092–1097. https://doi.org/10.1097/ALN.0b013e31819db2a1 (2009).
Michard, F., Chemla, D. & Teboul, J-L. Applicability of pulse pressure variation: How many shades of grey? Crit. Care 19, 144. https://doi.org/10.1186/s13054-015-0869-x (2015).
Guinot, P-G. et al. Passive leg raising can predict fluid responsiveness in patients placed on venovenous extracorporeal membrane oxygenation. Crit. Care. 15, R216. https://doi.org/10.1186/cc10451 (2011).
Geerts, B. F. et al. Comprehensive review: Is it better to use the Trendelenburg position or passive leg raising for the initial treatment of hypovolemia? J. Clin. Anesth. 24, 668–674. https://doi.org/10.1016/j.jclinane.2012.06.003 (2012).
Yonis, H. et al. Change in cardiac output during Trendelenburg maneuver is a reliable predictor of fluid responsiveness in patients with acute respiratory distress syndrome in the prone position under protective ventilation. Crit. Care. 21, 295. https://doi.org/10.1186/s13054-017-1881-0 (2017).
Monnet, X., Shi, R. & Teboul, J-L. Prediction of fluid responsiveness. What’s new? Ann. Intensive Care. 12, 46. https://doi.org/10.1186/s13613-022-01022-8 (2022).
Wang, H. et al. Value of corrected flow time in common carotid artery in predicting volume responsiveness under mechanical ventilation. Shock 58, 28–33. https://doi.org/10.1097/SHK.0000000000001959 (2022).
Song, Y. et al. Respirophasic carotid artery peak velocity variation as a predictor of fluid responsiveness in mechanically ventilated patients with coronary artery disease. Br. J. Anaesth. 113, 61–66. https://doi.org/10.1093/bja/aeu057 (2014).
Pace, R. et al. Carotid vs. aortic velocity time integral and peak velocity to predict fluid responsiveness in mechanically ventilated patients. A comparative study. Minerva Anestesiol. 88, 352–360. https://doi.org/10.23736/S0375-9393.21.16035-3 (2022).
Ramsay, M. A. E. et al. Controlled Sedation with Alphaxalone-Alphadolone. BMJ 2, 656–659. https://doi.org/10.1136/bmj.2.5920.656 (1974).
Nassar, B. et al. Trendelenburg position does not increase cross-sectional area of the internal jugular vein predictably. Chest 144, 177–182. https://doi.org/10.1378/chest.11-2462 (2013).
Ugalde, D. et al. Transthoracic cardiac ultrasound in prone position: A technique variation description. Intensive Care Med. 44, 986–987. https://doi.org/10.1007/s00134-018-5049-4 (2018).
Peachey, T. et al. The assessment of circulating volume using inferior vena cava collapse index and carotid Doppler velocity time integral in healthy volunteers: A pilot study. Scand. J. Trauma. Resusc. Emerg. Med. 24, 108. https://doi.org/10.1186/s13049-016-0298-0 (2016).
Wang, Y. et al. Assessment of left atrial function by full volume real-time three-dimensional echocardiography and left atrial tracking in essential hypertension patients with different patterns of left ventricular geometric models. Chin. Med. Sci. J. 28, 152–158. https://doi.org/10.1016/S1001-9294(13)60041-3 (2013).
Mohammadinejad, P. & Hossein-Nejad, H. Calculation of corrected flow time: Wodey’s formula vs. Bazett’s formula. J. Crit. Care. 44, 154–155. https://doi.org/10.1016/j.jcrc.2017.10.046 (2018).
Michard, F. et al. Global end-diastolic volume as an indicator of cardiac preload in patients with septic shock*. Chest 124, 1900–1908. https://doi.org/10.1378/chest.124.5.1900 (2003).
Kim, D-H. et al. Carotid ultrasound measurements for assessing fluid responsiveness in spontaneously breathing patients: Corrected flow time and respirophasic variation in blood flow peak velocity. Br. J. Anaesth. 121, 541–549. https://doi.org/10.1016/j.bja.2017.12.047 (2018).
Coste, J. & Pouchot, J. A grey zone for quantitative diagnostic and screening tests. Int. J. Epidemiol. 32, 304–313. https://doi.org/10.1093/ije/dyg054 (2003).
Combes, A. et al. What is the niche for extracorporeal membrane oxygenation in severe acute respiratory distress syndrome? Curr. Opin. Crit. Care. 18, 527–532. https://doi.org/10.1097/MCC.0b013e328357f090 (2012).
Hutchens, M., Song, H. & Ibsen, L. Extracorporeal membrane oxygenation for ARDS in adults. N. Engl. J. Med. 366(6), 575–576. https://doi.org/10.1056/NEJMc1114604 (2012).
Thompson, B. T., Chambers, R. C. & Liu, K. D. Acute respiratory distress syndrome. N. Engl. J. Med. 377, 562–572. https://doi.org/10.1056/NEJMra1608077 (2017).
Hamzaoui, O. & Teboul, J-L. Central venous pressure (CVP). Intensive Care Med. 48, 1498–1500. https://doi.org/10.1007/s00134-022-06835-6 (2022).
Teboul, J-L. et al. Arterial pulse pressure variation with mechanical ventilation. Am. J. Respir Crit. Care Med. 199, 22–31. https://doi.org/10.1164/rccm.201801-0088CI (2019).
Reuter, D. A. et al. Stroke volume variations for assessment of cardiac responsiveness to volume loading in mechanically ventilated patients after cardiac surgery. Intensive Care Med. 28, 392–398. https://doi.org/10.1007/s00134-002-1211-z (2002).
Xu, Y. et al. Efficacy of using tidal volume challenge to improve the reliability of pulse pressure variation reduced in low tidal volume ventilated critically ill patients with decreased respiratory system compliance. BMC Anesthesiol. 22, 137. https://doi.org/10.1186/s12871-022-01676-8 (2022).
Berger, K. et al. Prone position affects stroke volume variation performance in predicting fluid responsiveness in neurosurgical patients (2015).
Slutsky, A. S. & Ranieri, V. M. Ventilator-induced lung injury. N. Engl. J. Med. 369, 2126–2136. https://doi.org/10.1056/NEJMra1208707 (2013).
Guérin, C. et al. Prone positioning in severe acute respiratory distress syndrome. N. Engl. J. Med. 368, 2159–2168. https://doi.org/10.1056/NEJMoa1214103 (2013).
Zaaqoq, A. M. et al. Beneficial effect of prone positioning during venovenous extracorporeal membrane oxygenation for coronavirus disease 2019*. Crit. Care Med. 50, 275–285. https://doi.org/10.1097/CCM.0000000000005296 (2022).
Petit, M. et al. Prone-positioning for severe acute respiratory distress syndrome requiring extracorporeal membrane oxygenation. Crit. Care Med. 50, 264–274. https://doi.org/10.1097/CCM.0000000000005145 (2022).
Giani, M. et al. Timing of prone positioning during venovenous extracorporeal membrane oxygenation for acute respiratory distress syndrome. Crit. Care Med. 51, 25–35. https://doi.org/10.1097/CCM.0000000000005705 (2023).
Giani, M. et al. Prone positioning during venovenous extracorporeal membrane oxygenation for acute respiratory distress syndrome: A pooled individual patient data analysis. Crit. Care. 26, 8. https://doi.org/10.1186/s13054-021-03879-w (2022).
Pelosi, P. et al. Effects of the prone position on respiratory mechanics and gas exchange during acute lung injury. Am. J. Respir Crit. Care Med. 157, 387–393. https://doi.org/10.1164/ajrccm.157.2.97-04023 (1998).
Jung, S. et al. Ability of carotid corrected flow time to predict fluid responsiveness in patients mechanically ventilated using low tidal volume after surgery. JCM 10, 2676. https://doi.org/10.3390/jcm10122676 (2021).
Luo, J. et al. Trendelenburg maneuver predicts fluid responsiveness in patients on veno-arterial extracorporeal membrane oxygenation. Ann. Intensive Care. 11, 16. https://doi.org/10.1186/s13613-021-00811-x (2021).
Hung, K-C. et al. Diagnostic efficacy of carotid ultrasound for predicting the risk of perioperative hypotension or fluid responsiveness: A meta-analysis. Diagnostics 13, 2290. https://doi.org/10.3390/diagnostics13132290 (2023).
Monnet, X., Marik, P. & Teboul, J-L. Passive leg raising for predicting fluid responsiveness: A systematic review and meta-analysis. Intensive Care Med. 42, 1935–1947. https://doi.org/10.1007/s00134-015-4134-1 (2016).
Rezoagli, E. et al. Physical manoeuvres in patients with ARDS and low compliance: Bedside approaches to detect lung hyperinflation and optimise mechanical ventilation. Eur. Respir J. 61, 2202169. https://doi.org/10.1183/13993003.02169-2022 (2023).
Acknowledgements
The authors wish to thank all research staff and patients for participating in this study.
Funding
This work was supported in part by grants from the Project of Zhejiang Provincial Department of Health (No. 2023KY1296) and Quzhou Bureau of Science and Technology (No. 2022K71).
Author information
Authors and Affiliations
Contributions
H.F. and J.Z. conceptualized the study. J.Z. developed the methodology. J.Z. was responsible for the software. J.Z. and J.T. conducted the formal analysis. Resources were provided by H.F. and Y.S. Data curation was handled by Y.S., J.T., K.G., and K.W. J.Z. wrote the original draft. Supervision was provided by H.F. and J.Z.G. All authors read and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This prospective, single-center study was conducted in the ICU between May 2022 and February 2024. It was approved by the Institutional Review Board of Quzhou People’s Hospital (Quzhou, China: Number B 2022-083) and performed in accordance with the Declaration of Helsinki. All research was performed in accordance with relevant guidelines/regulations. Informed consent was obtained from all participants and/or their legal guardians.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, J., Sun, Y., Tang, J. et al. Predictive value of trendelenburg position and carotid ultrasound for fluid responsiveness in patients on VV-ECMO with acute respiratory distress syndrome in the prone position. Sci Rep 14, 31808 (2024). https://doi.org/10.1038/s41598-024-83038-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-83038-7