Abstract
The immunomodulatory properties of mesenchymal stem/stromal cells (MSCs) are strongly influenced by IFN-γ priming and may vary depending on their tissue of origin. This study investigates whether the immunoregulatory responses of human amniotic fluid-derived MSCs (hAF-MSCs) to IFN-γ are altered when derived from individuals with unexplained recurrent pregnancy loss (RPL). We analyzed 10 well-characterized hAF-MSC clones, equally divided into non-RPL and RPL groups, to assess IFN-γ-induced expression of immunomodulatory genes. Additionally, we evaluated the paracrine effects of MSC-conditioned medium on T regulatory (Treg) cell induction and macrophage polarization. Significant differences were observed between the two groups in the expression of immunomodulatory markers following IFN-γ priming, as well as in their capacity to induce Treg cells and modulate M1/M2 macrophage polarization. These findings support the hypothesis that dysregulated immune pathways contribute to unexplained RPL by revealing impaired IFN-γ responsiveness in hAF-MSCs from fetuses derived from couples with the history of RPL.
Similar content being viewed by others
Introduction
As defined in the literature, RPL or recurrent miscarriage is defined by three or more consecutive spontaneous abortions before 20 weeks of gestation1. Despite advancements in understanding its etiology, approximately 50% of RPL cases remain unexplained or idiopathic, with no identifiable causative factors. Potential contributors to idiopathic miscarriages include ovarian aging, environmental influences, and immunological dysfunctions2. Among these, immunological mechanisms at the fetal-maternal interface have emerged as critical areas of investigation, with implications for both understanding idiopathic miscarriages and developing novel therapeutic approaches, particularly for treating autoimmune diseases3,4,5,6.
Research into the immunological underpinning of RPL has predominantly focused on analyzing autoantibodies, natural killer cells (NKCs) in blood and decidua, cytokines in circulation or secreted by decidual cells, and classical and non-classical HLA polymorphisms, as well as HLA expression on trophoblastic cells2,5,7,8,9. Recent studies, including our own, have extended this focus to explore the interplay between immunological and genomic factors in RPL, examining maternal, paternal, and combined parental genetic contributions10. This has given rise to the hypothesis of “unfavorable genomes,” suggesting that specific combinations of parental genetic profiles may increase the risk of fetal abnormalities and recurrent miscarriage. Unfavorable variants of uncertain significance (VUS), which are not definitively linked to the disease11,12,13, may nonetheless contribute to genomic instability in embryonic cells, reducing the likelihood of achieving a healthy pregnancy in RPL cases10,11. Furthermore, evidence suggests that the genomes of products of conception (POCs) in RPL families may harbor genetic predispositions to developmental disorders during pregnancy12. Though this hypothesis requires further substantiation, early findings point to potential deficiencies in key elements essential for embryonic cell growth and differentiation in RPL cases13.
MSCs are increasingly recognized as pivotal regulators of immunological mechanisms, capable of switching between pro-inflammatory (MSC1) and immunomodulatory (MSC2) phenotypes in response to pro-inflammatory cytokines such as IFN-γ, TNF-α, IL-1β, and IL-1714,15. Inflammatory microenvironments activate tissue-resident MSCs and recruit circulating MSCs to sites of tissue damage, where they promote regeneration and restore homeostasis16,17. The dynamic regulation of immune responses by MSCs is a critical aspect of tissue recovery, mediated through innate immune activation, chemokine upregulation, adaptive immune cell recruitment, and the licensing of MSCs to acquire an immunomodulatory phenotype (MSC2)17,18,19,20,21. Disruptions in this intricate process, potentially influenced by genomic variations, may impair immunoregulation and possibly contribute to RPL pathogenesis22.
Given this context, we hypothesize that the source of MSCs influences their immunomodulatory capacity in response to IFN-γ. Amniotic fluid contains a heterogeneous population of differentiated, progenitor, and multipotent fetal cells derived from various fetal and extra-embryonic tissues23,24,25. Numerous studies have investigated AF-MSCs for diverse therapeutic and research applications26,27. As AF-MSCs are representative of fetal cells that reflect the parental genomic background, an intriguing question arises: could the genomic background of couples with RPL result in fetuses with AF-MSCs that exhibit altered immunomodulatory responses to IFN-γ compared to those from unaffected pregnancies?
Materials and methods
Sampling and experimental design
Human amniotic fluid samples were collected by ultrasound through the procedure of amniocentesis at the second trimester, ranging from 16th to 18th weeks of pregnancy. The application of specimens in this study did not interfere with the prenatal diagnosis (PND) process; nonetheless, informed consent was acquired from participants, particularly due to the inclusion of vulnerable groups, i.e. women who have suffered from recurrent miscarriages. It should be noted that all methods were conducted in accordance with the strict biological ethics guidelines of the Yazd Reproductive Sciences Institute.
The clinical characteristics of mothers and their fetuses are presented in the Supplementary Table 1. These samples were included in the study based on the normal karyotype of fetus and maternal age of 27–34 years. Several specimens were excluded from the study due to high-risk pregnancies that could have influenced the results. These included cases involving tobacco and alcohol use, as well as maternal diseases such as autoimmune disorders, preeclampsia, and gestational diabetes.
The study included two groups. The first group, which served as a healthy control, was comprised of five pregnant women who had not experienced any miscarriages and had at least one full-term pregnancy (non-RPL group). The second group consisted of five pregnant women who had a history of idiopathic recurrent miscarriage (RPL group). The AF-MSC samples in both non-RPL and RPL groups were evaluated for their immunoregulatory properties under five different conditions. These included a basic culture without stimulation with pro-inflammatory cytokine IFN-γ as the control. Additionally, the IFN-γ stimulated state was evaluated at two concentrations of 20 and 100 international units per milliliter (IU/ml) of culture medium, in two time-intervals of 24 and 72 h.
Derivation of mesenchymal stem/stromal cells from human amniotic fluid
With the aim of not interfering with the PND process, we implemented a two-step protocol for obtaining AF-MSCs. Our previous findings demonstrated that this method is the most convenient and effective option with the least impact on PND28. To derive AF-MSCs, amniocytes were sequentially cultured in a T25 flasks with a density of 104 cells/cm2 at 37 ℃ and under 5% CO2. For this purpose, a modified medium composed of 2:1 v/v Dulbecco’s modified Eagle’s medium (DMEM)-AmnioMAX-II was used as a superior medium compared to either DMEM or AmnioMAX-II25. The cells were harvested at the third passage for characterization by flow cytometry analysis and mesodermal differentiation capacity, as well as experimental cultures in different stimulatory mimetic situations.
Cytogenetic analysis
To ensure that the AF-MSCs are chromosomally stable, the clones were screened for any numerical or structural abnormalities. The cells were harvested at P3 using Carnoy fixative solution, within 24 to 48 h after the sub-culturing process, depending on the growth rate. The slides were prepared, followed by banding and staining with Giemsa (Sigma-Aldrich Co., Germany), and then used for standard chromosome G-banding analysis.
Flow cytometry analysis
Based on the International Society for Cell & Gene Therapy (ISCT) minimal criteria, MSCs are classified as clonal cells expressing mesenchymal markers, including CD44, CD73, CD90, and CD105, while they lack the expression of hematopoietic markers, such as CD31, CD34 and CD4529. For this purpose, the expanded clones were characterized by flow cytometry regarding the presence of mesenchymal surface markers, i.e., CD44, CD90, and CD105. The vascular endothelial marker CD31 was also analyzed as a negative indicator. To do so, at least 105 cells for each assay were detached from T25 flasks by trypsin-EDTA treatment and the cell pellet was washed twice in phosphate-buffered saline (PBS) containing 0.2% fetal bovine serum (FBS). Cells were then incubated with the following antibodies in PBS supplemented with 1% bovine serum albumin (BSA), including FITC-conjugated anti-human CD44 and CD90, as well as PE-conjugated anti-human CD105 and CD31 (all from Immunostep).
In order to detect induced Treg cells and macrophage polarization, human peripheral blood mononuclear cells (PBMCs) cultured doubly in MSC-conditioned medium (MSC-CM) were analyzed by flow cytometry regarding the target markers. For this purpose, PBMCs were incubated with Treg-specific conjugates in PBS supplemented with 1% BSA. These markers included PerCP-conjugated anti-human CD4, APC-conjugated anti-human CD25, and PE-conjugated anti-human FOXP3 antibodies. The target markers for macrophage polarization were examined using PE.Cy7-conjugated anti-human CD68, FITC-conjugated anti-human CD80, and PE-conjugated anti-human CD206 antibodies. The conjugates were purchased from Immunostep, BioLegend, and Exbio. The results were analyzed on a BD FACSCalibur and the graphs were generated in FlowJo (v 10.1, Tree Star, Inc.) software.
Mesodermal differentiation
Differentiation ability of clonal cells into mesodermal lineages was examined by assessing adipogenic and osteogenic differentiation. The expanded cells were detached from T25 flasks in third passage, counted and then seeded in 6-well plates at a density of 5 × 103 cells/cm2 in DMEM containing 10% FBS and 1% penicillin-streptomycin (Pen-Strep) as basal medium. At 70% confluency, the cultures were incubated under differentiation media for 3 weeks (refreshing medium every 3 days). For adipogenic induction medium, the basal medium was supplemented with 50 µg/ml indomethacin, 50 µg/ml ascorbic acid-2-phosphate, 10 IU/ml insulin and 100 nM dexamethasone. Further, for osteogenic differentiation, 10 mM β-glycerol phosphate, 50 µg/ml ascorbic acid-2-phosphate and 10 nM dexamethasone were added to the basal medium (all supplements were from Sigma-Aldrich). To confirm adipogenesis in the cells, intracellular lipid vacuoles were stained with oil red O (Sigma-Aldrich). Validation of osteogenesis was performed via visualization of extracellular matrix mineralization by alizarin red S30.
Molecular analysis
The quantitative RT-PCR analysis was used to evaluate the response of AF-MSCs to IFN-γ under five priming treatments. For this purpose, AF-MSCs from non-RPL and RPL groups were assessed for the expression levels of immunoregulatory genes, including indoleamine 2,3-dioxygenase 1 and 2 (IDO1 and IDO2), leukemia inhibitory factor (LIF), transforming growth factor-beta (TGF-β), interleukin-6 and − 10 (IL-6 and IL-10), cyclooxygenase 1 and 2 (COX1 and COX2), Toll-like receptor 4 (TLR-4), and vascular cell adhesion molecule 1 (VCAM1)24,31. 18 S rRNA was used as a reference gene in all RT-PCR reactions. All human-specific primers were designed for intron-spanning regions. Primer sequences, their melting temperatures (Tm) and the size of amplified products are listed in Table 1.
Following qualitative assessment of total RNA extracted from AF-MSCs by TRIzol reagent (Roche), 100 ng of total RNA was used to synthesize cDNA using the RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific). Applied Biosystems® SYBR® Green PCR Master Mix was used for preparation of RT-PCR reactions. Quantitative analysis was carried out by means of Applied Biosystems StepOnePlus Real-Time PCR System. To do this, real-time system was set on initial denaturation step at 95 ℃ for 10 min, followed by 40 cycles of 95 ℃ for 20 s, 58–60 ℃ (based on different annealing temperatures of primers) for 20 s and 72 ℃ for 30 s.
Induction of Treg cells by MSC-conditioned medium
To evaluate the effects of MSC-CM on Treg cell induction, peripheral blood (PB) was collected from two healthy male donors aged 23 and 25 years. Donors were selected based on their general health status, including the absence of severe pathological conditions such as autoimmune diseases, and were screened for current infections using routine blood tests. These tests included a complete blood count (CBC) and additional indicators such as red and white blood cell counts, platelet counts, hemoglobin, hematocrit, lipid panel (LDL, HDL), metabolic panel (calcium, glucose, sodium, potassium), thyroid panel (T3, T4, TSH), and C-reactive protein (CRP) levels. The donors were included in the study due to their normal blood test results.
PBMCs were isolated from 15 ml of PB using a density gradient method with Lymphocyte Separation Solution (Serana), following the manufacturer’s protocol. The isolated PBMCs were cultured at a concentration of 106 cells/ml in Roswell Park Memorial Institute (RPMI) 1640 medium supplemented with 10% FBS, 1% Pen-Strep, and 1 µg/ml phytohemagglutinin (PHA; Gibco) for 48 h to stimulate mitogenic activation. After culture, PHA-stimulated PBMCs were harvested by centrifugation, washed with 5 ml PBS, and centrifuged again. The resulting cell pellet was resuspended and cultured in MSC-CM (106 cells/ml) for 72 h to assess Treg cell induction.
For MSC-CM preparation, the culture media from AF-MSC clones were collected separately and stored at − 80 °C. MSC-CM from clones of non-RPL and RPL groups were pooled into five distinct treatment conditions. To serve as a control for Treg induction efficiency, PHA-stimulated PBMCs were cultured in a modified medium comprising DMEM-AmnioMAX-II (2:1 v/v) supplemented with recombinant human TGF-β1 (R&D Systems) at a final concentration of 10 ng/ml, as previously described32.
Induction of M2 macrophages by MSC-conditioned medium
PHA-stimulated PBMCs from the same healthy donors were used to evaluate the effects of MSC-CM on macrophage polarization. The PHA-stimulated PBMCs (106 cells/ml) were cultured in MSC-CM for 72 h to assess its impact on macrophage phenotype. To serve as a control for inducing M2 macrophages, PHA-stimulated PBMCs were cultured in a modified medium consisting of DMEM-AmnioMAX-II (2:1 v/v), supplemented with recombinant human TGF-β1 at a final concentration of 10 ng/ml, based on previously reported protocols33.
The impact of MSC-CM on both Treg induction and macrophage polarization was analyzed using flow cytometry, as detailed in section “2.4. Flow cytometry analysis”.
Statistical analysis
The RT-PCR results were quantitatively assessed using the 2−∆∆CT method. This method involves analyzing the difference between the reference and target CT values of each sample. The expression levels of five mimetic states were analyzed using two-way analysis of variance (ANOVA) to evaluate all treatments together. Additionally, the non-parametric Mann-Whitney test was used for dual analysis of treatments. Pearson’s coefficients were calculated to identify possible differences in the correlation of target genes between the non-RPL and RPL groups. In all statistical analyses, significant level was set at p-value < 0.05.
Results
Characterization of mesenchymal stem/stromal cells derived from amniotic fluid
Adherent cells derived from amniotic fluid were isolated, expanded to the third passage, and analyzed to confirm their MSC characteristics and chromosomal stability. Flow cytometry revealed that more than 90% of the cells in both the non-RPL (healthy control) and RPL (case) groups expressed the MSC markers CD44, CD90, and CD105. Additionally, over 97% of the cells were negative for CD31, indicating the absence of endothelial or hematopoietic contamination. The flow cytometry results for non-RPL and RPL clones are detailed in Supplementary Figs. 1 and 2, respectively.
To confirm the differentiation capacity of the derived MSCs, all clones were cultured under adipogenic and osteogenic conditions. Cells from both groups successfully differentiated into adipocytes and osteocytes, as shown in Supplementary Fig. 3 (Panels A and B). Cytogenetic evaluations further confirmed the normal karyotype of all 10 clones included in the study, with representative karyograms provided in Supplementary Fig. 4. Two clones, one from each group, were excluded from the study due to trisomy 21 chromosomal abnormalities observed during the initial screening.
Expression of immunoregulatory genes in response to IFN-γ
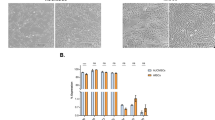
The study assessed the relative expression of immunomodulatory genes (IDO1, IDO2, IL-10, and LIF) under various inflammatory-mimetic conditions (Fig. 1). ANOVA analysis revealed significant differences among treatments for IDO1 (p = 0.023), IL-10 (p = 0.002), and LIF (p = 0.006), while differences for IDO2 were not statistically significant (p = 0.12).
Expression of target genes in response to IFN-γ. The expression level of immunomodulatory genes, IDO1, IDO2, IL-10, LIF, IL-6, TGF-β, COX1, COX2, TLR4, and VCAM-1 in AF-MSCs derived from samples of non-RPL and RPL groups under five different treatments; control (non-preconditioned by IFN-γ), preconditioned with two different concentrations of IFN-γ (20 IU/ml and 100 IU/ml) after two time-intervals (24 h and 72 h). The averages of five clones were analyzed using the Mann-Whitney test as dual comparisons. The p-values of non-significant differences (close to the significant level, i.e., < 0.1) are written above the compared column bars. The significant levels are marked as p-values < 0.05 (*) and < 0.01 (**).
In the RPL group, IDO1 expression significantly increased following treatments with 20 IU/ml-24 h and 100 IU/ml (both 24 h and 72 h) IFN-γ, compared to the control group (p < 0.01). However, these treatments had no significant impact on IDO1 expression in the non-RPL group. A similar trend was observed for IDO2, IL-10, and LIF: non-RPL clones responded moderately to 20 IU/mL-24 h IFN-γ treatment, whereas RPL clones showed significant upregulation with increasing IFN-γ doses or prolonged exposure (p < 0.05).
Expression of pleiotropic cytokines IL-6 and TGF-β also displayed distinct patterns between the groups. Non-RPL clones exhibited higher baseline expression levels and moderate responses to 20 IU/mL-24 h IFN-γ treatment, followed by reductions at higher doses and prolonged exposures. In contrast, RPL clones displayed minimal baseline expression but showed substantial upregulation with higher IFN-γ doses (100 IU/mL-24 h) or extended treatment times (72 h).
As illustrated in Fig. 1, the relative expression of COX2, TLR4, and VCAM-1 in response to IFN-γ exhibited a consistent trend, showing a significant increase from the control to the 20 IU/mL-24 h treatment in the non-RPL group. Similarly, in the RPL group, the expression levels were significantly upregulated in the 100 IU/mL-24 h treatment compared to the controls (p < 0.05). However, COX1 displayed a distinct behavior compared to these three genes—COX2, TLR4, and VCAM-1. Specifically, the response to IFN-γ was similar in both non-RPL and RPL groups. Despite this similarity, the high baseline expression of COX1 in the controls of the non-RPL group resulted in a non-significant increase in expression after the 20 IU/mL-24 h treatment. Conversely, the absence of COX1 expression in the controls of the RPL group led to a significant upregulation from the control to the 20 IU/mL-24 h treatment (p < 0.05).
Overview of expression profiles and correlations among genes
Heatmaps (Fig. 2) were generated to visualize the relative expression of target genes across individual clones (C1–C5) and treatments in both non-RPL (Fig. 2A) and RPL (Fig. 2B) groups. Positive correlations were represented by Pearson’s coefficient (r) of “0.9 to 1”, while values of “-0.9 to -1” were indicated as negative correlations, provided they were within the significant range (p-values < 0.05). Data normalization ensured that the lowest and highest values were represented as 0 and 1, respectively. The heatmaps revealed clear differences between groups: non-RPL clones exhibited moderate responses to varying IFN-γ doses and exposure times (Fig. 2A; C1-C5), while RPL clones showed more pronounced and dose-specific induction of target genes (Fig. 2B; C1-C5). For instance, non-RPL clones maintained COX1 and COX2 expression under control conditions, whereas RPL clones exhibited minimal baseline expression, with strong induction observed only after specific IFN-γ treatments.
Overview of expression profiles and correlations among target genes. The heatmaps represent a descriptive overview of the expression level of target genes across different treatments in both non-RPL (A) and RPL (B) groups, in which the data have been normalized so that the lowest and highest values are represented as 0 and 1, respectively. To show any possible relationships between the levels of target genes and their responses to different treatments, in non-RPL (C) and RPL (D) groups, Pearson correlation analysis was used based on the Pearson coefficient within the significant range (p-values < 0.05). Correlations were classified based on the Pearson coefficient, with highly positive ranging from 0.95 to 1, possibly positive ranging from 0.90 to < 0.95, possibly negative ranging from >-0.95 to -0.90 and highly negative ranging from − 1 to -0.95. Non-significant correlations (Pearson coefficient: >-0.90 and < 0.90) are excluded in the plots as white cells.
Pearson correlation analyses further highlighted group-specific relationships among gene expression profiles (Fig. 2C and D). In the RPL group, strong positive correlations were observed between IL-10 and IDO2 (r = 0.96), as well as between IL-10 and VCAM-1 (r = 0.97). Similarly, LIF correlated positively with IL-6 (r = 0.95), while moderate correlations were noted between TGF-β and both LIF and COX2 (r = 0.90–0.95). In contrast, the non-RPL group showed no significant correlations among the genes.
These findings suggest a distinct, dysregulated response to IFN-γ in RPL clones, potentially driven by genomic or immunological abnormalities. The observed correlations in the RPL group may reflect underlying mechanisms contributing to altered immunoregulation in these cases.
Comparison of the Immunomodulatory capacity in paracrine mode
Treg cell induction in PBMCs under MSC-conditioned medium
PBMCs were isolated from two healthy young donor to investigate the induction of regulatory T cells (Treg; CD4⁺CD25⁺FOXP3⁺) under the influence of MSC-CM. Figure 3 displays the Treg induction across different conditions. The Treg induction by the basic medium (RPMI 1640 supplemented with 10 ng/ml TGF-β1) and pooled MSC-CMs from five clones in the non-RPL (Fig. 3-Panel A) and RPL (Fig. 3-Panel B) groups is illustrated. The percentage of CD4⁺CD25⁺FOXP3⁺ Treg cells was determined using a three-step gating strategy. First, PBMCs were gated based on lymphocyte size and granularity using forward scatter (FSC) and side scatter (SSC) parameters. Next, CD4⁺ lymphocytes were identified within this gated population, represented in the trapezoid region of the left dot plot in each binary plot. Finally, CD25⁺FOXP3⁺ cells were selected from the CD4⁺ population, represented in the square region of the right dot plot in each binary plot (Fig. 3).
The density dot plots from flow cytometry analysis representing Treg gates. The results are depicted in 3 boxes: (A) non-RPL and (B) RPL Panels (five upper and lower double-dot plots, respectively) for induction of Treg cells by MSC-CM in different treatments, including control (CM from unstimulated MSCs), 20 and 100 IU/ml of IFN-γ during 24 h and 72 h. One double-dot plot depicted in a single box in the middle for induction of Treg cells by the basic medium (TGF-β1-supplemented RPMI 1640). PBMCs were gated based on lymphocyte size and granularity using forward scatter (FSC) and side scatter (SSC) parameters (not presented here). Next, CD4⁺ lymphocytes were identified within this gated population, represented in the trapezoid region of the left dot plot in each binary plot. Finally, CD25⁺FOXP3⁺ cells were selected from the CD4⁺ population, represented in the square region of the right dot plot in each binary plot.
Figure 5 further quantifies Treg induction, presenting the percentages of Treg cells (Fig. 5A) and Treg/CD4⁺ T cell ratios (Fig. 5B) as bar graphs. The percentage of Treg cells was highest in the basic medium and lower in both MSC-CM groups. While Treg induction levels were comparable between non-RPL and RPL groups under most conditions (24 h treatments with both IFN-γ concentrations and 72 h treatment with 20 IU/ml IFN-γ), a significant reduction in Treg induction was observed in the MSC-CM derived from the control (p = 0.012) and 100 IU/mL-72 h (p = 0.011) treatments of the RPL group compared to the non-RPL group (Fig. 5A). Similarly, the Treg/CD4⁺ T cell ratios followed the same trend. Ratios were significantly lower in the MSC-CM derived from the control (p = 0.012) and 100 IU/mL-72 h (p = 0.004) treatments of the RPL group compared to the non-RPL group (Fig. 5B).
Macrophage polarization in PBMCs under MSC-conditioned medium
The study aimed to evaluate the effect of MSC-CM on macrophage polarization. To determine the proportions of M1 and M2 macrophage phenotypes, CD68, CD80, and CD206 markers were analyzed. The polarization data, derived from pooled MSC-CM related to five clones, are presented in dot plots in Fig. 4, with separate panels for non-RPL (Fig. 4A) and RPL (Fig. 4B) groups. For comparison, macrophage polarization in the basic medium (TGF-β1-supplemented RPMI 1640) was used as a control. The summarized results from these markers are depicted as bar graphs in Fig. 5 (C-E) for clarity. The analysis focused on CD68⁺ cells (total macrophages/monocytes) to assess the proportions of CD80⁺ (M1 macrophages) and CD206⁺ (M2 macrophages) populations.
The density dot plots from flow cytometry analysis representing macrophage gates. The results are depicted in 3 boxes: (A) non-RPL and (B) RPL Panels (5 upper and lower double-dot plot, respectively) for polarization of macrophages by MSC-CM in different treatments, including control (CM from unstimulated MSCs), 20 and 100 IU/ml of IFN-γ during 24 h and 72 h. One double-dot plot depicted in a single box in the middle represents the polarization of macrophages by the basic medium (TGF-β1-supplemented RPMI 1640). The results of each double-dot plot depict the gating of main population based on the markers CD68 and CD80 on the left and CD80 and CD206 on the right. The main population is gated based on FSC and SSC parameters, which their plots are not presented here. FSC: forward scatter; SSC: side scatter.
The bar graphs representing the results of flow cytometry analysis. The results of Treg cell induction and macrophage polarization by MSC-CM are presented in different treatments, including basic medium (BM) TGF-β1-supplemented RPMI 1640, control (CM from unstimulated MSCs), 20 and 100 IU/ml of IFN-γ during 24 h and 72 h. (A) The proportion of Treg cells as the fraction of CD25⁺FOXP3⁺ cells in the CD4⁺ cell population, as described in the legend of Fig. 3; (B) The ratio of CD25⁺FOXP3⁺ Treg cells to CD4⁺ cells; (C) The proportion of M1 macrophages in the main population based on the fraction of CD68⁺CD80⁺ cells; (D) The proportion of M2 macrophages in the main population based on the fraction of CD68⁺CD206⁺ cells; (E) The proportion of mixed M1/M2 macrophages in the main population based on the fraction of CD68⁺CD80⁺CD206⁺ cells.
The MSC-CM derived from the control treatment significantly increased the proportion of CD68⁺CD80⁺ (M1) cells in the non-RPL group compared to the RPL group (p = 0.004, Fig. 5C). Conversely, the treatments with 20 IU/ml for 72 h (p = 0.020) and 100 IU/ml for both 24 h (p = 0.021) and 72 h (p = 0.001) showed significantly higher M1 polarization in the RPL group compared to the non-RPL group. For M2 polarization, the proportion of CD68⁺CD206⁺ cells significantly increased in the control (p = 0.007) and the 20 IU/ml 72 h (p = 0.033) treatments of the RPL group compared to the non-RPL group (Fig. 5D). However, in the 100 IU/ml 72 h treatment, M2 polarization was significantly higher in the non-RPL group compared to the RPL group (p = 0.016). To investigate the mixed M1/M2 phenotype, the co-expression of CD80 and CD206 was analyzed. The proportion of CD80⁺CD206⁺ cells was significantly higher in the control (p = 0.045) and in the 72 h treatments of both 20 IU/ml (p = 0.024) and 100 IU/ml (p = 0.002) in the RPL group compared to the non-RPL group (Fig. 5E). However, the 100 IU/ml 24 h treatment showed a significantly higher mixed phenotype in the non-RPL group compared to the RPL group (p = 0.016).
Discussion
Unexplained or idiopathic miscarriages are largely attributed to defective mechanisms in the acceptance of the semi-allograft fetus by the maternal uterine environment, potentially associated with immunological processes2,34. While studies have focused on maternal immune dysfunction and tolerogenic mechanisms, the potential role of fetal factors in RPL remains underexplored. Emerging evidence suggests that RPL may stem from genetic contributions, particularly those associated with immunoregulatory processes10. The “unfavorable genomes” hypothesis proposes that genetic instability resulting from maternal and paternal genome interactions may impair embryonic viability and compromise immunoregulatory functions of fetal cells10,11,12. As a result, modifying the immunoregulatory functions of fetal cells may be a potential outcome of unfavorable genetic compositions.
Developmental abnormalities have been consistently observed in aborted fetuses from RPL cases, even in the absence of chromosomal anomalies, indicating severe growth and morphogenesis disturbances35. Furthermore, studies have linked maternal RPL history to increased risks of birth defects (4.2% vs. 2.5%)36, as well as long-term neurological, developmental, and movement disorders in offspring37. Similar patterns have been reported in other conditions, such as respiratory morbidity, suggesting a shared immunological etiology38. These findings inspired our hypothesis that cellular-level abnormalities, particularly in fetal stem cells, may also be present in RPL cases. Accordingly, we focused on AF-MSCs as representative models of fetal MSCs to explore potential differences between RPL and non-RPL groups.
MSCs exhibit remarkable plasticity, dynamically adapting their immunoregulatory functions in response to environmental cues. These cues polarize MSCs into either a pro-inflammatory (MSC1) or anti-inflammatory (MSC2) phenotype39. This adaptability allows MSCs to function as “sensors and switchers,” amplifying inflammatory responses in hypoactive immune states or mitigating excessive inflammation to restore immune homeostasis in hyperactive systems40. The polarization process is orchestrated by a sophisticated network of cytokines, chemokines, and cell-surface receptors, enabling MSCs to effectively modulate immune responses and contribute to tissue repair14. A recent comprehensive review by Tan et al.. underscores the impact of IFN-γ dosing on MSC polarization41. The study highlights that priming MSCs with IFN-γ at concentrations between 10 and 100 ng/mL for 24 to 48 h markedly enhances their immunosuppressive capabilities. Specifically, this priming induces the expression of IDO through the Janus kinase (JAK)-signal transducer and activator of transcription 1 (STAT1) pathway. As a result, MSCs exhibit increased suppression of T-cell proliferation and a shift toward the anti-inflammatory MSC2 phenotype. However, insufficient IFN-γ concentrations fail to elicit this immunosuppressive state, while excessive levels may compromise MSC viability and functionality. This dose-dependent response emphasizes the critical role of cytokine signaling in modulating MSC therapeutic potential.
In inflammatory contexts, interactions between tissue-resident cells, immune cells, and MSCs play a critical role in determining the inflammatory state. Dysregulated cytokine levels can skew MSC polarization, contributing to autoimmune diseases42. For instance, type 1 diabetes mellitus (T1DM) patients and their relatives exhibit heightened baseline inflammation, leading to amplified responses to inflammatory stimuli43. Our previous work demonstrated that AF-MSCs co-cultured with PBMCs from T1DM patients showed altered chemokine expression and Treg induction compared to PBMCs from healthy donors. RPL patients may also experience a similar dysregulated inflammatory microenvironment that impairs the immunomodulatory capacity of MSCs, which play a key role in regulating immune responses at inflamed sites. Our study supports this notion by analyzing gene expression levels under different treatments, revealing distinct patterns between RPL and non-RPL clones.
Specifically, most target genes in RPL clones showed a pronounced response to only one treatment, while remaining unresponsive to others. In contrast, non-RPL clones exhibited a more consistent, mild expression of these genes across various treatments. A comparison of target gene expression between non-RPL and RPL clones demonstrated significant differences, likely stemming from the varied responses of AF-MSCs to different concentrations and exposure durations of IFN-γ. Notably, RPL clones required higher doses of IFN-γ to induce expression of most target genes, resulting in markedly elevated expression levels compared to non-RPL clones. This included genes such as IDO2, IL-10, LIF, IL-6, TGF-β, COX2, TLR4 and VCAM-1, as illustrated in Fig. 1. Moreover, the duration of IFN-γ exposure significantly influenced gene expression patterns between the two groups. In the non-RPL group, genes such as IDO2, LIF, IL-6, TGF-β, IL-10, COX1, and COX2 were downregulated when exposure time increased from 24 to 72 h at a dosage of 20 IU/mL. In contrast, these genes in the RPL group showed either stable or increased expression levels under the same conditions. These findings highlight the altered responsiveness of AF-MSCs in RPL patients, suggesting a disrupted capacity to adapt to inflammatory cues, which may contribute to their impaired immunoregulatory function.
The heatmap and correlation analysis revealed notable differences in gene expression and regulatory patterns between non-RPL and RPL clones, highlighting a distinct response to IFN-γ in the RPL group (Fig. 2A and B). Non-RPL clones displayed moderate and consistent responses across varying doses and exposure times, leading to variable levels of target genes among treatments. In contrast, RPL clones exhibited minimal baseline expression of key genes, such as COX1 and COX2, with sharp induction under specific IFN-γ treatments. These variable responses to IFN-γ observed in RPL clones resulted in modified correlations of some target genes, as evidenced by Pearson’s coefficient analysis (Fig. 2C and D). Strong positive correlations, particularly between IL-10 and IDO2, and IL-10 and VCAM-1, may imply a dysregulated network of immunomodulatory genes in RPL clones. This altered gene expression profile may reflect genomic or immunological abnormalities that disrupt the normal interplay of cytokines and regulatory molecules, contributing to the immune niches associated with RPL. While these findings suggest possibly impaired sensitivity of RPL clones to IFN-γ priming compared to non-RPL clones, the underlying mechanisms remain unclear. A comprehensive understanding of MSC-mediated immunomodulation44 and the pathophysiology of idiopathic miscarriages45 is essential to determine whether the genomic background of the fetus influences the immunomodulatory capacity of fetal MSCs. This underscores the importance of further exploring the genomic and epigenetic underpinnings of immunoregulatory mechanisms in RPL to better understand its pathophysiology and identify potential therapeutic targets.
Our study focused on the paracrine functions of AF-MSCs, particularly their role in shaping the immune microenvironment and influencing key mediators of T helper 1 (Th1) and T helper 2 (Th2) responses, such as Treg cells and macrophage subsets. We observed significant differences in Treg proportions between the RPL and non-RPL groups, particularly under the control condition and the 100 IU/mL-72 h IFN-γ treatment. Notably, MSC-CM derived from the RPL group induced fewer Treg cells compared to the non-RPL group (Fig. 5A). This suggests that the RPL-derived MSC secretome may have a reduced capacity for Treg induction, likely reflecting deficiencies or dysfunction in active mediators essential for immune tolerance in this group.
Interestingly, Treg generation was higher in the TGF-β1-supplemented basic medium (BM) compared to the MSC-CM control group. Given that TGF-β1 is a potent inducer of Treg differentiation from PBMCs46, this observation highlights the dominant role of TGF-β1 in driving Treg expansion independent of MSC-derived signals. A similar trend was evident in the Treg/CD4⁺ ratio (Fig. 5B), where the RPL group consistently showed lower ratios under both the control and IFN-γ-treated conditions. Since an elevated Treg/CD4⁺ ratio reflects enhanced immune tolerance and suppression of effector T cell proliferation, these findings may indicate an intrinsic immune dysregulation in RPL patients, as reported in reproductive and tumor immunology contexts47,48,49,50.
AF-MSC-derived secretomes, including extracellular vesicles (EVs), cytokines, and microRNAs, are well-documented for their immunomodulatory capabilities51. Recent evidence shows that MSC-derived EVs, particularly from IFN-γ-primed cells, can enhance FOXP3 expression in Tregs via microRNAs such as miR-139-5p and miR-214-5p52. The discrepancy in Treg induction between the RPL and non-RPL groups observed here could reflect differences in the bioactive composition of their respective MSC-CM.
Beyond Tregs, macrophage polarization was another key focus of our study. Circulating monocytes respond to microenvironmental cues by differentiating into pro-inflammatory M1 macrophages or anti-inflammatory M2 macrophages. M1 macrophages (CD68⁺CD80⁺) are typically involved in Th1-driven immune responses, while M2 macrophages (CD68⁺CD206⁺) promote Th2-mediated immune regulation and tissue repair53,54. MSCs are widely recognized for their capacity to modulate macrophage phenotypes, frequently inducing a shift toward the M2 phenotype through secreted immunoregulatory mediators such as TGF-β, IL-10, and PGE255,56. Rather than isolating macrophages prior to analysis, we assessed polarization directly within the PBMC population. This approach allowed us to preserve the natural cellular interactions that occur between monocytes, T cells, and MSC-derived signals, thereby providing a more physiologically relevant model. Additionally, we employed CD68, a well-established marker for monocyte-derived macrophages in vitro57, to identify macrophages within the PBMC population.
In this study, we aimed to elucidate the paracrine effects of MSC-CM on macrophage polarization, particularly focusing on the immunomodulatory influence of both unstimulated and IFN-γ-preconditioned MSCs on PBMCs. To establish a consistent baseline for macrophage differentiation, all MSC-CM experimental groups were compared to cultures in basic medium (BM, RPMI 1640) supplemented with TGF-β1. TGF-β1 is known to enhance monocyte survival and plasticity, promoting the differentiation of non-activated macrophages into the M2 phenotype46. By using TGF-β1-supplemented medium as a control, we aimed to compare MSC-CM-induced effects on M2 polarization against a standardized M2-favoring environment.
Our results revealed a distinct pattern in macrophage polarization influenced by both the source of MSCs (RPL vs. non-RPL) and the treatment condition. Under control conditions, MSC-CM from non-RPL donors primarily induced an M1 phenotype, while RPL-derived MSC-CM favored M2 polarization (Fig. 5C and D). In contrast, following IFN-γ preconditioning (100 IU/mL-72 h), non-RPL MSC-CM promoted M2 polarization, while RPL MSC-CM shifted toward an M1 phenotype. These findings suggest that AF-MSCs respond to IFN-γ in a source-dependent manner, and that the immune background of the donor may shape the MSC secretome’s immunomodulatory capacity. Interestingly, the pattern of Treg induction partially mirrored the M2 macrophage trend: both were enhanced by non-RPL MSC-CM under IFN-γ preconditioning, suggesting the existence of shared or complementary regulatory pathways. However, the inverse relationship in the control condition (higher Treg levels in non-RPL but higher M2 macrophages in RPL) underlines the complexity of immune network interactions and the distinct pathways that govern Treg expansion and macrophage polarization58. We also observed time-dependent differences in the induction of a mixed M1/M2 phenotype (CD80⁺CD206⁺). This intermediate state, which reflects the inherent plasticity of macrophages in response to fluctuating environmental cues54,59, was more pronounced at 24 h in the non-RPL group, but shifted toward the RPL group at 72 h (Fig. 5E). This temporal variability further emphasizes the dynamic nature of MSC-mediated immune regulation.
Another key observation was that IFN-γ preconditioning did not consistently enhance M2 polarization, as might be expected from prior reports showing IFN-γ primes MSCs to produce anti-inflammatory mediators60. In our study, IFN-γ-treated MSC-CM led to an increased proportion of CD68⁺CD206⁺ M2 macrophages compared to control. Conversely, in the RPL group, M2 polarization decreased under IFN-γ treatment, except for the 20 IU/ml 72 h condition. This divergence suggests that the immunomodulatory effect of IFN-γ-primed MSCs may be altered in the context of RPL, potentially reflecting patient-specific immune backgrounds rather than MSC conditioning alone. It is also important to note that Tregs and M2 macrophages, while often functionally linked in maintaining immune tolerance, do not always correlate proportionally, as their induction is governed by partially overlapping but distinct regulatory pathways50,58,61. These findings underscore the importance of considering both MSC source and patient-specific factors when evaluating MSC-mediated immunomodulation. The unexpected results highlight the complexity of MSC-mediated immunoregulation, influenced not only by external stimuli like IFN-γ but also by the source of MSCs and the immune background of the patient samples.
This study has several limitations that should be acknowledged. The absence of additional control conditions — such as PBMC cultures in cytokine-free medium — limits our ability to fully distinguish the direct effects of MSC-CM from background immune plasticity. Moreover, while IFN-γ levels were standardized across all MSC-CM conditions, its well-established role in promoting M1 polarization and modulating Treg function62,63 cannot be entirely excluded as a confounding factor influencing the observed immune profiles. Additionally, although immune cell phenotypes were assessed using surface markers, secreted proteins and EVs cargo were not directly quantified, which limits the mechanistic depth of our conclusions. Functional validation assays, including T cell proliferation suppression tests, were also not performed, which would have clarified the biological relevance of the immunomodulatory shifts observed. Finally, the exploratory nature of the study and limited sample size necessitates cautious interpretation, and future research using larger cohorts, comprehensive molecular profiling, and broader experimental models will be essential to confirm and expand these findings.
Despite these limitations, our study addresses a novel and previously unexplored question. The findings underscore the need for further investigation into whether developmental defects observed in children born to couples with a history of RPL are reflected in AF-MSC characteristics and other cell types. Future research employing high-throughput techniques, such as RNA-seq and proteomics, along with rigorous functional assays, will be essential for validating and expanding these findings. By acknowledging these constraints, we aim to provide a transparent interpretation of our results and encourage further exploration of this promising research avenue.
Conclusion
This study investigated the hypothesis that the immunomodulatory response of AF-MSCs to IFN-γ priming is influenced by their source, specifically comparing AF-MSC clones derived from healthy pregnancies and those with a history of idiopathic RPL. Our findings revealed significant differences between the two groups in their response to IFN-γ stimulation. AF-MSCs from the RPL group required higher doses of IFN-γ to induce immunomodulatory markers compared to the control condition. Conversely, AF-MSCs from the non-RPL group showed upregulation of these markers at lower doses but displayed decreased responsiveness or no significant changes at higher doses. In addition, the MSC-CM from the two groups exhibited distinct effects on immune cell modulation. Specifically, non-RPL-derived MSC-CM showed a greater capacity to induce Treg cells and anti-inflammatory M2 macrophages under specific treatment conditions, whereas RPL-derived MSC-CM demonstrated a stronger induction of pro-inflammatory M1 macrophages under comparable conditions. These observations underscore the differential immunomodulatory capacities of AF-MSC clones, highlighting the influence of their origin on the secretion of bioactive mediators and the subsequent modulation of immune responses. Our findings support the hypothesis that dysregulated immune pathways may play a critical role in the pathophysiology of idiopathic RPL. However, further studies are required to validate these results and elucidate the underlying mechanisms. High-throughput sequencing and functional analyses with larger sample sizes and more rigorous experimental designs will be essential to uncover the specific immune pathways and mediators involved. Such efforts could pave the way for developing targeted therapeutic strategies to address immune dysregulation in idiopathic RPL.
Data availability
All data will be available on request from the corresponding author.
References
Karami, N. et al. Aberrant expression of microRNAs 16 and 21 and gene targets in women with unexplained recurrent miscarriage: A case-control study. Int. J. Reprod. Biomed. 16(10), 617–622 (2018).
Krieg, S. & Westphal, L. (eds) Immune Function and Recurrent Pregnancy Loss. Seminars in Reproductive Medicine (Thieme Medical, 2015).
Chen, S. J., Liu, Y. L. & Sytwu, H. K. Immunologic regulation in pregnancy: from mechanism to therapeutic strategy for immunomodulation. Clin. Dev. Immunol. 2012, 258391 (2012).
Luo, X., Miller, S. D. & Shea, L. D. Immune tolerance for autoimmune disease and cell transplantation. Annu. Rev. Biomed. Eng. 18, 181–205 (2016).
Deshmukh, H. & Way, S. S. Immunological basis for recurrent fetal loss and pregnancy complications. Annu. Rev. Pathol. 14, 185–210 (2019).
Genest, G. et al. Immunotherapy for recurrent pregnancy loss: a reappraisal. F&S Reviews. 3(1), 24–41 (2022).
Mehta, S. & Gupta, B. Recurrent Pregnancy Loss (Springer, 2018).
Gupta, M. Immunology and Pregnancy Loss 53–65 (Springer, 2018).
Srivastava, M. & Srivastava, A. Immunotherapy for Recurrent Miscarriages 327–335 (Springer, 2018).
Montazeri, F., Tajamolian, M., Hosseini, E. S. & Hoseini, S. M. Immunologic factors and genomic considerations in recurrent pregnancy loss: A review. Int. J. Med. Lab. 10(4), 279–305 (2023).
Liu, P. et al. An organismal CNV mutator phenotype restricted to early human development. Cell 168(5), 830–42e7 (2017).
Kasak, L., Rull, K., Sõber, S. & Laan, M. Copy number variation profile in the placental and parental genomes of recurrent pregnancy loss families. Sci. Rep. 7, 45327 (2017).
Sõber, S. et al. RNA sequencing of chorionic villi from recurrent pregnancy loss patients reveals impaired function of basic nuclear and cellular machinery. Sci. Rep. 6(1), 38439 (2016).
Le Blanc, K. & Davies, L. C. Mesenchymal stromal cells and the innate immune response. Immunol. Lett. 168(2), 140–146 (2015).
Haghighitalab, A. et al. Investigating the effects of IDO1, PTGS2, and TGF-β1 overexpression on Immunomodulatory properties of hTERT-MSCs and their extracellular vesicles. Sci. Rep. 11(1), 7825 (2021).
Carvalho, A. E. S., Sousa, M. R. R., Alencar-Silva, T., Carvalho, J. L. & Saldanha-Araujo, F. Mesenchymal stem cells Immunomodulation: the road to IFN-gamma licensing and the path ahead. Cytokine Growth Factor. Rev. 47, 32–42 (2019).
López-García, L. & Castro-Manrreza, M. E. TNF-α and IFN-γ participate in improving the immunoregulatory capacity of mesenchymal stem/stromal cells: importance of Cell-Cell contact and extracellular vesicles. Int. J. Mol. Sci. ;22(17). (2021).
Fallahi, P. et al. CXCR3, CXCR5, CXCR6, and CXCR7 in diabetes. Curr. Drug Targets. 17(5), 515–519 (2016).
Burke, S. J. & Collier, J. J. Transcriptional regulation of chemokine genes: a link to pancreatic islet inflammation? Biomolecules 5(2), 1020–1034 (2015).
Szűcs, D. et al. Effect of inflammatory microenvironment on the regenerative capacity of Adipose-Derived mesenchymal stem cells. Cells ;12(15). (2023).
Kurte, M. et al. Time-dependent LPS exposure commands MSC immunoplasticity through TLR4 activation leading to opposite therapeutic outcome in EAE. Stem Cell Res. Ther. 11(1), 416 (2020).
Hoseini, S., Hosseini, E., Abessi, P. & Montazeri, F. Paracrine secretions and immunological activities of human mesenchymal stem cells: the key regenerative factors of microenvironment. J. Stem Cell. Res. Ther. 14. (2024).
Hoseini, S. M., Moghaddam-Matin, M., Bahrami, A. R., Montazeri, F. & Kalantar, S. M. Human amniotic fluid stem cells: general characteristics and potential therapeutic applications. J. Shahid Sadoughi Univ. Med. Sci. 28(12), 3252–3275 (2021).
Hoseini, S. M. et al. A comparative analysis of Immunomodulatory genes in two clonal subpopulations of CD90 + amniocytes isolated from human amniotic fluid. Placenta 101, 234–241 (2020).
Hoseini, S. M., Kalantar, S. M., Bahrami, A. R. & Matin, M. M. Human amniocytes: a comprehensive study on morphology, frequency and growth properties of subpopulations from a single clone to the senescence. Cell. Tissue Biology. 14(2), 102–112 (2020).
Mu, X-P. et al. Enhanced differentiation of human amniotic fluid-derived stem cells into insulin-producing cells in vitro. J. Diabetes Invest. 8(1), 34–43 (2017).
Murphy, S. V. & Atala, A. Amniotic fluid and placental membranes: unexpected sources of highly multipotent cells. Semin Reprod. Med. 31(1), 62–68 (2013).
Hoseini, S. et al. Efficient isolation of mesenchymal stem cells from amniotic fluid via a two-step culture method. Biomedical Res. Therapy. 11(8), 6683–6697 (2024).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy 8(4), 315–317 (2006).
Kornicka, K., Marycz, K., Tomaszewski, K. A., Marędziak, M. & Śmieszek, A. The effect of age on osteogenic and adipogenic differentiation potential of human adipose derived stromal stem cells (hASCs) and the impact of stress factors in the course of the differentiation process. Oxid. Med. Cell. Longev. 2015. (2015).
Bulati, M. et al. The Immunomodulatory properties of the human amnion-derived mesenchymal stromal/stem cells are induced by INF-γ produced by activated lymphomonocytes and are mediated by Cell-To-Cell contact and soluble factors. Front. Immunol. 11, 54 (2020).
Schmidt, A., Eriksson, M., Shang, M. M., Weyd, H. & Tegnér, J. Comparative analysis of protocols to induce human CD4 + Foxp3 + Regulatory T cells by combinations of IL-2, TGF-beta, retinoic acid, rapamycin and butyrate. PLoS One. 11(2), e0148474 (2016).
Mia, S., Warnecke, A., Zhang, X. M., Malmström, V. & Harris, R. A. An optimized protocol for human M2 macrophages using M-CSF and IL-4/IL-10/TGF-β yields a dominant immunosuppressive phenotype. Scand. J. Immunol. 79(5), 305–314 (2014).
Saini, V., Arora, S., Yadav, A. & Bhattacharjee, J. Cytokines in recurrent pregnancy loss. Clin. Chim. Acta. 412(9–10), 702–708 (2011).
Feichtinger, M., Wallner, E., Hartmann, B., Reiner, A. & Philipp, T. Transcervical embryoscopic and cytogenetic findings reveal distinctive differences in primary and secondary recurrent pregnancy loss. Fertil. Steril. 107(1), 144–149 (2017).
Khoury, M. J. & Erickson, J. D. Recurrent pregnancy loss as an indicator for increased risk of birth defects: a population-based case-control study. Paediatr. Perinat. Epidemiol. 7(4), 404–416 (1993).
Walfisch, A., Wainstock, T., Segal, I., Landau, D. & Sheiner, E. 828: maternal history of recurrent pregnancy loss increases the risk for long term paediatric neurological morbidity of the offspring. Am. J. Obstet. Gynecol. 216(1), S474–S5 (2017).
Davidesko, S., Wainstock, T., Sheiner, E., Landau, D. & Walfisch, A. Maternal history of recurrent pregnancy loss increases the risk for long-term pediatric respiratory morbidity of the offspring. Pediatr. Pulmonol. 55(7), 1765–1770 (2020).
Bernardo, M. E. & Fibbe, W. E. Mesenchymal stromal cells: sensors and switchers of inflammation. Cell. Stem Cell. 13(4), 392–402 (2013).
Jiang, W. & Xu, J. Immune modulation by mesenchymal stem cells. Cell Prolif. 53(1), e12712 (2020).
Tan, Y. L. et al. Therapeutic efficacy of Interferon-Gamma and Hypoxia-Primed mesenchymal stromal cells and their extracellular vesicles: underlying mechanisms and potentials in clinical translation. Biomedicines ;12(6). (2024).
Fayyad-Kazan, M., Najar, M., Fayyad-Kazan, H., Raicevic, G. & Lagneaux, L. Identification and evaluation of new immunoregulatory genes in mesenchymal stromal cells of different origins: comparison of normal and inflammatory conditions. Med. Sci. Monit. Basic. Res. 23, 87–96 (2017).
Cabrera, S. M., Chen, Y. G., Hagopian, W. A. & Hessner, M. J. Blood-based signatures in type 1 diabetes. Diabetologia 59(3), 414–425 (2016).
Fayyad-Kazan, H. et al. Study of the microRNA expression profile of foreskin derived mesenchymal stromal cells following inflammation priming. J. Translational Med. 15(1), 10 (2017).
Ford, H. B. & Schust, D. J. Recurrent pregnancy loss: etiology, diagnosis, and therapy. Rev. Obstet. Gynecol. 2(2), 76–83 (2009).
Zhang, F. et al. TGF-β induces M2-like macrophage polarization via SNAIL-mediated suppression of a pro-inflammatory phenotype. Oncotarget 7(32), 52294–52306 (2016).
Li, L. et al. Increased frequency of regulatory T cells in the peripheral blood of patients with endometrioid adenocarcinoma. Oncol. Lett. 18(2), 1424–1430 (2019).
Gao, W., Yang, N., Ji, S. & Zeng, Y. Frequency of CD4 + regulatory T cells and modulation of CD4 + T lymphocyte activation in pleural tuberculoma. Tuberculosis 134, 102210 (2022).
Xu, T., Lu, J. & An, H. The relative change in regulatory T cells / T helper lymphocytes ratio as parameter for prediction of therapy efficacy in metastatic colorectal cancer patients. Oncotarget 8(65), 109079–109093 (2017).
Whiteside, T. L. Regulatory T cell subsets in human cancer: are they regulating for or against tumor progression? Cancer Immunol. Immunother. 63(1), 67–72 (2014).
Hoseini, S. M., Azad, F. S., Zare, E. & Montazeri, F. Therapeutic potential of amniotic fluid and amniotic fluid-derived stem cells in regenerative medicine. Regenerative Biomed. 1(2), 85–105 (2025).
Hyland, M. et al. Extracellular vesicles derived from umbilical cord mesenchymal stromal cells show enhanced anti-inflammatory properties via upregulation of MiRNAs after pro-inflammatory priming. Stem Cell. Rev. Rep. 19(7), 2391–2406 (2023).
Modak, M. et al. CD206 + tumor-associated macrophages cross-present tumor antigen and drive antitumor immunity. JCI Insight ;7(11). (2022).
Furgiuele, S. et al. Dealing with macrophage plasticity to address therapeutic challenges in head and neck cancers. Int. J. Mol. Sci. ;23(12). (2022).
Lo Sicco, C. et al. Mesenchymal stem Cell-Derived extracellular vesicles as mediators of anti-inflammatory effects: endorsement of macrophage polarization. Stem Cells Translational Med. 6(3), 1018–1028 (2017).
Kuppa, S. S., Kim, H. K., Kang, J. Y., Lee, S. C. & Seon, J. K. Role of mesenchymal stem cells and their paracrine mediators in macrophage polarization: an approach to reduce inflammation in osteoarthritis. Int. J. Mol. Sci. 23(21) (2022).
Zhang, J., Li, S., Liu, F. & Yang, K. Role of CD68 in tumor immunity and prognosis prediction in pan-cancer. Sci. Rep. 12(1), 7844 (2022).
Guan, T., Zhou, X., Zhou, W. & Lin, H. Regulatory T cell and macrophage crosstalk in acute lung injury: future perspectives. Cell. Death Discovery. 9(1), 9 (2023).
Smith, T. D., Tse, M. J., Read, E. L. & Liu, W. F. Regulation of macrophage polarization and plasticity by complex activation signals. Integr. Biol. (Camb). 8(9), 946–955 (2016).
Hoseini, S. M., Zare, E., Azad, F. S. & Montazeri, F. The niche of the mesenchymal stem cells displays a vital determinant role of immunoregulatory dynamics. Regenerative Biomed. 1(2), 106–126 (2025).
Shaopeng, Z., Yang, Z., Yuan, F., Chen, H. & Zhengjun, Q. Regulation of regulatory T cells and tumor-associated macrophages in gastric cancer tumor microenvironment. Cancer Med. 13(2), e6959 (2024).
Chen, S. et al. Macrophages in immunoregulation and therapeutics. Signal. Transduct. Target. Therapy. 8(1), 207 (2023).
Gouirand, V., Habrylo, I. & Rosenblum, M. D. Regulatory T cells and inflammatory mediators in autoimmune disease. J. Invest. Dermatology. 142(3, Part B), 774–780 (2022).
Acknowledgements
The authors would like to express their gratitude to Miss Fateme Zarein, for her valuable technical assistance in this work, as well as Zeynab Darvishian and Sama Ghasemi, SabaBiomedicals Science-Based Company, Tehran, Iran, for their appreciated technical assistance in flow cytometry analysis.
Funding
Yazd Reproductive Sciences Institute funded this study through grant numbers of 5664 and 10001.
Author information
Authors and Affiliations
Contributions
(1) Seyed Mehdi Hoseini: managing the project, interpretation of results, and writing the manuscript draft. (2) Ahmad Reza Bahrami: conception of the study, study design. (3) Seyed Mehdi Kalantar: supervision of the study. (4) Mohammad Hasan Sheikhha: genetic counseling of the patients, collecting patients’ medical records. (5) Behrouz Aflatoonian: analysis and interpretation of data. (6) Nasrin Ghasemi: genetic counseling of the patients, collecting patients’ medical records. (7) Elham Sadat Hosseini: data acquisition, collection of samples from patients. (8) Maryam M. Matin: supervision of the study, study design. (9) Fateme Montazeri: managing the project, statistical analysis of data, manuscript revision.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
1—Title of the approved project: Investigation of the Expression Level of Indoleamine 2,3-Dioxygenase (IDO) As an Immunosuppressive-Immunomodulatory Marker in Amniotic Fluid-Derived Mesenchymal Stem/Stromal Cells from Women Who Experienced Recurrent Pregnancy Loss.
Name of the institutional approval committee: Research Ethics Committee of Shahid Sadoughi University of Medical Sciences (Specific Code: IR.SUU.REC); approval number: IR.SSU.REC.1396.169 (official committee minutes number of 255339 for project 5664); approval date: 2018, January 9.
2—Title of the approved project: Investigation of Induced T-Regulatory Cells in Peripheral Blood Mononuclear Cells Cultured Under Conditioned Medium of Mesenchymal Stem/Stromal Cells Derived from Amniotic Fluid of Pregnant Women Who Experienced Idiopathic Recurrent Pregnancy Loss.
Name of the institutional approval committee: Research Ethics Committee of Research Center for Infertility - Shahid Sadoughi University of Medical Sciences (Specific Code: IR.SUU.RSI.REC); approval number: IR.SSU.RSI.REC.1399.041 (official committee minutes number of 320711 for project 10001); approval date: 2021, February 21.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hoseini, S.M., Bahrami, A.R., Kalantar, S.M. et al. Immunomodulatory effects of primed amniotic fluid-derived mesenchymal stem/stromal cells with IFN-γ from unexplained recurrent miscarriage sources. Sci Rep 16, 281 (2026). https://doi.org/10.1038/s41598-025-01799-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-01799-1