Abstract
Degenerative cerebellar ataxias (DCAs) are progressive diseases that reduce quality of life (QoL). This study aimed to assess the impact of rehabilitation on QoL in patients with DCAs using structural equation modeling (SEM). This cross-sectional survey included members of a national Japanese DCAs patient association. Assessed latent variables included personal, medical, and environmental factors, impairments, activity limitations, rehabilitation (participation quantity and patient-reported quality), and QoL. SEM was used to explore causal relationships between these latent variables. Overall, 477 participants (mean age 65.4 years; 45.1% female) were included. Impairments were categorized as primary and secondary, based on preliminary analyses. The final model demonstrated acceptable-to-good fit indices, explaining 74% of the variance in QoL. The model paths showed that activity limitations, secondary impairments, and rehabilitation quality had a direct effect on QoL, in that order. The quantity of rehabilitation had an indirect effect on QoL through its direct effect on secondary impairments and quality of rehabilitation. These findings suggest that rehabilitation interventions improve QoL in the DCA population, but its effects vary depending on the quantity and quality of rehabilitation. However, the cross-sectional study design limits the ability to draw causal conclusions and longitudinal studies are needed for confirmation.
Similar content being viewed by others
Introduction
Degenerative cerebellar ataxias (DCAs) are a group of sporadic or hereditary diseases characterized by progressive degeneration that mainly affects the cerebellum1,2. Sporadic ataxia includes multiple system atrophy (MSA), whereas hereditary ataxia is classified as autosomal dominant spinocerebellar ataxia (SCA) or autosomal recessive CA3. Patients with DCAs may exhibit a pure cerebellar phenotype or a combination of systemic disorders that include extracerebellar symptoms, such as extrapyramidal movement disorders and autonomic dysfunction2,4. Recent studies suggest that DCAs lead to more extensive symptoms than previously understood, prompting a need for reassessment of the full range of symptoms2,3. Regardless of the disease subtype, quality of life (QoL) is a crucial outcome in DCAs, and is notoriously difficult to manage5,6. In fact, some studies have shown that patients with DCAs experience significantly lower QoL than does the general population, with progressive decline over time5,7,8,9,10,11,12.
Although no definitive treatments exist for DCAs, rehabilitation is essential in managing the condition13,14. Rehabilitation interventions aim to improve the activities of daily living (ADLs) and QoL in these patients15. There is evidence that rehabilitation, including physical and occupational therapy, can improve mainly ataxia, balance, and mobility, based on the integration of several clinical trials14,16,17,18. It is possible that these functional factors, and improvements in ADL, contribute to an improved QoL. However, there is currently a lack of consistent evidence showing direct improvement in QoL through rehabilitation16,18. Our study posed a simple yet vital research question: Do rehabilitation interventions improve QoL in a diverse sample of the DCA population? To answer this question, a model that clarifies the key elements of QoL and its causal relationship with rehabilitation is required19. This can be achieved using structural equation modeling (SEM) to visualize complex components that define QoL for patients with DCAs. SEM integrates multiple observed variables as latent variables, and estimates the hypothesized underlying structural relationships between them. Unlike statistical prediction, SEM is grounded in probabilistic causal modeling20. This approach is ideal for investigating the relationships between QoL and its predictors. For instance, SEM has quantified QoL effects in various populations, including those with Parkinson’s disease21, multiple sclerosis22, amyotrophic lateral sclerosis23, and spinal cord injury24. Demonstrating a causal relationship between rehabilitation and QoL in a large, diverse population of patients with DCA, rather than in a limited sample or specific subtype, will enhance our understanding of the overall effect of rehabilitation. These results can guide clinicians in identifying key variables affecting QoL, including rehabilitation factors, and inform future clinical practice.
Therefore, this study aimed to explore the framework of QoL, using multidimensional data from a diverse DCA population, and assess the overall effects of rehabilitation. The focus was on health-related QoL, encompassing aspects affected by health status and potentially influenced by medical care6. We hypothesized that rehabilitation interventions would improve QoL directly or indirectly, by optimizing function and activity domains.
Results
Baseline characteristics of the study population
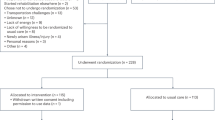
Of the 1000 individuals surveyed, 591 responded, and 477 met the eligibility criteria (Fig. 1). Table 1 summarizes the characteristics of the final participants (additional details in Supplementary Table S1). The mean age was 65.4 years (standard deviation [SD] 12.4), and 214 (45.1%) were women. The DCA type was identified in 322 participants (69.1%). The median disease duration was 10.8 years (interquartile range [IQR] 6.0–18.2), and the average number of types of medication per day was 4.4 (SD 3.5). Falls in the past year, categorized as none, once, 2–9 times, or more than 10 times, were reported by 365 participants (79.7%). Regarding residence type, 444 participants (93.5%) lived at home, 58 (12.7%) lived alone, and 62 (11.9%) had no caregiver. The severity of impairments was widely reported, with the most frequent being imbalance (median 3 [IQR 2–3]), ataxia (median 3 [IQR 2–3]), weakness (median 2 [IQR 1–3]), fatigue (median 2 [IQR 1–2]), and speech dysarthria (median 2 [IQR 1–3]). The median modified Rankin scale (mRS) score was 4 (IQR 3–4), and 110 participants (23.9%) were fully independent or had modified independence. Independent or modified independent walking was reported by 165 (35.8%) participants for indoor mobility and 149 (31.8%) for outdoor mobility. The mean Barthel index (BI) was 70.1 (SD 36.6), and the mean Life-Space Assessment (LSA) score was 36.7 (SD 28.4).
Flowchart of participant enrollment. Abbreviations: SCD, spinocerebellar degeneration; MSA, multiple system atrophy; DCA: degenerative cerebellar ataxia.
Regarding rehabilitation, 368 participants (77.1%) received professional rehabilitation, including physical therapy (84.2%), speech-language-hearing therapy (45.4%), and occupational therapy (42.4%). The median total rehabilitation time was 320 min per month (IQR 160–720), and the median duration of rehabilitation was 30 months (IQR 2–71.5). Among those who received rehabilitation, the mean scores for effects of rehabilitation, self-efficacy of rehabilitation, and satisfaction with rehabilitation ranged from 6.1 to 6.5 (SD 2.4–2.5), whereas the mean score for motivation for rehabilitation was 7.2 (SD 2.4). For QoL, the mean EQ index was 0.49 (SD 0.23), and the mean EuroQoL Visual Analogue Scale (EQ-VAS) score was 57.9 (SD 23.9). The mean life satisfaction score was 5.1 (SD 2.3).
SEM
The exploratory correlation analysis did not indicate significant multicollinearity (Supplementary Table S2). Based on the results of the preliminary confirmatory factor analysis (CFA), the impairments were dichotomized and the latent variables for primary and secondary impairments were redefined. After confirming that all local CFA models fit well, a modified model was generated (Model 2; Supplementary Fig. S1 and Supplementary Table S3). The SEM measurement model was validated using the modified Model 2. After adjusting variables and paths, the final model was established (Model 3; Table 2, Fig. 2, and Supplementary Fig. S2). The fit indices for the final model indicated acceptable-to-good fit (root mean square error of approximation [RMSEA] 0.043, 90% confidence interval [CI]: 0.040–0.046; probability of close fit 1.00; comparative fit index [CFI] 0.920; Tucker–Lewis index [TLI] 0.914; standardized root mean square residual [SRMR] 0.074).
Final model (Model 3). The numbers along the paths represent standardized path coefficients, synonymous with standardized direct effects. The numbers in the upper right corners of the endogenous latent variables indicate explanatory rates (R2). Details of the model and regression coefficients are presented in Table 3. Complete details of all variables in the final model, including observed variables and error correlations, are provided in Supplementary Fig. S2.
Table 3 summarizes the standardized direct and indirect effects on QoL. Only three latent variables had direct effects on QoL: secondary impairments (β = − 0.536, 95% CI: − 0.614 to − 0.457), activity limitations (β = − 0.391, 95% CI: − 0.478 to − 0.304), and quality of rehabilitation (β = 0.061, 95% CI: 0.012–0.109). Primary impairments, quantity of rehabilitation, and background factors did not have a direct path to QoL. The quantity of rehabilitation had an indirect effect (β = 0.106, 95% CI: 0.066–0.147) on QoL by directly affecting secondary impairments (β = − 0.121, 95% CI: − 0.109 to − 0.012) and the quality of rehabilitation (β = 0.680, 95% CI: 0.620–0.741). The final model explained 74% of the variance in QoL, indicating a strong fit between the model and the data.
Sub-analysis
In the final basic model, the path from primary impairments to activity limitations was removed and replaced with a path from environmental factors, which had a higher coefficient (Supplementary Fig. S3). Despite this change, the fit indices for the basic model remained acceptable to good (RMSEA = 0.046, 90% CI: 0.043–0.050; probability of close fit = 0.952; CFI = 0.936; TLI = 0.931; SRMR = 0.070). The explanation rate for the variance in QoL increased slightly to 76%, remaining consistent with the main model. Detailed results for this basic model are presented in Table 3 and Supplementary Table S4.
Discussion
The key findings of this study are as follows. First, ICF-based models of a QoL framework in the DCA population were successfully identified, using SEM to provide robust statistical evidence that builds on previous models. Second, the relative importance of impairments and activity limitations on QoL was demonstrated, with secondary impairments showing a more direct impact on QoL than primary impairments. Third, that the quantity of rehabilitation, measured in time and variety of therapies, had a direct effect on secondary impairments was confirmed. Conversely, the quality of rehabilitation, reflecting patient engagement and perceived effectiveness, had a direct influence on QoL. These findings support our hypothesis that general rehabilitation interventions contribute directly and indirectly to the QoL of patients with DCAs, across various clinical subtypes.
Several large-scale studies have evaluated QoL in patients with SCA and MSA7,8,25. The mean EQ index of 0.49 and the mean EQ-VAS score of 57.9 in this study align with previous findings, confirming the diminished QoL in the DCA population. Factors that have been reported to lower QoL in patients with DCAs include the severity of ataxia, motor dysfunction, depression, anxiety, pain, fatigue, autonomic symptoms, and MSA subtypes7,26,27,28,29,30. The SEM model in the current study included the individual variables investigated in these studies. Factors reported to cause the greatest difficulties for patients, validated in other DCA populations, involved balance, coordination, mobility, posture/gait, speech, feeding/swallowing, and oculomotor/vision items5,31. These factors are consistent with the observed variables that had high factor loadings in the final model. Significant variables identified in this study also included the Patient Health Questionnaire-2 score, fatigue, and urinary and bowel impairments, which have been acknowledged as important symptoms in patients with DCA3,32,33,34. Other notable variables that contributed to impairments were weakness, rigidity, and sensory disturbances. Although many of these are secondary impairment variables, their direct impact on QoL was not fully investigated in prior studies, emphasizing the need for further research. The activity limitation variables in this study had higher factor loadings for indoor basic ADLs, compared to those of more extensive performance activities. This underscores the importance of interventions focusing on indoor basic ADLs from medical and environmental perspectives.
Although two final models were presented in this study, with and without the rehabilitation component, the basic structure of the models generally remained the same. Both models confirmed that impairment and activity limitation factors significantly contributed to QoL, although they portrayed the relationships between these factors differently. Specifically, the rehabilitation effects model demonstrated a clear path from primary impairments to activity limitations, whereas the basic model suggested that impairments and activity limitations were independent of each other. The basic model reflects a simpler relationship in which medical factors influence impairments and environmental factors impact activity limitations35. The background component, encompassing personal, medical, and environmental factors, contributed complexly to basic components like impairments and activity limitations, with medical factors having a strong influence on QoL. However, an important finding is that the background component did not directly affect QoL. Personal and medical factors influenced primary impairments independently, without interrelationships. Unsurprisingly, primary impairments had the highest effect size on QoL. Since secondary impairments are significantly influenced by primary impairments and directly impact QoL, they play a crucial role in improving patient outcomes. Although activity limitation factors decrease partially from a decline in primary impairments, the pathway from environmental factors is stronger; therefore, it can be inferred that the environmental approach can support activity levels. Indeed, patients with DCAs may be more sensitive to their caregiving environment than patients with other neurological diseases36.
An important finding of this study is that rehabilitation interventions are effective in patients with DCAs who present with diverse clinical features, addressing a crucial research question. Although the quantity of rehabilitation influenced secondary impairments, it did not significantly affect primary impairments or activity limitations. This aligns with a core concept of rehabilitation, which aims to prevent and/or ameliorate secondary complications14. Thus, rehabilitation contributes to improved QoL by mitigating manageable secondary impairments. In terms of the quantity of rehabilitation by factor loadings, time and duration are important, but incorporating a greater variety of professional rehabilitation types appears to be more effective for enhancing QoL, rather than simply engaging in many rehabilitation programs. However, this study does not provide insight into which specific intervention content is most effective. A prior study found that the aspects of QoL most patients with ataxia wished to improve were fitness/physical recreation and mobility31. Another study reported that approximately half of respondents experienced improvement with various medical approaches, including physical therapy, where exercise and physical activity were key elements in symptom management or overall well-being5. In this study, most participants reported engaging in physical therapy, with exercises such as gait and balance training being the most common, consistent with standard intervention strategies37,38,39. This suggests that rehabilitation interventions are generally aligned with patient needs. However, the fact that the quantity of rehabilitation does not necessarily reflect the quality of rehabilitation highlights that not only is quantitative enrichment needed, but so are outcomes that patients perceive as meaningful40. Improving the quality of rehabilitation in patients with DCAs, given the progressive nature of the impairment, remains challenging37,38. Nevertheless, even limited improvements in impairment and activity limitation factors may explain how rehabilitation can enhance QoL through comprehensive care, including environmental modifications, psychological support, and family guidance. Finally, the low path coefficient from rehabilitation factors to QoL should not be interpreted as diminishing the value of rehabilitation, but rather as a cautious reflection of the complexity of the causal relationship between rehabilitation interventions and QoL improvements in degenerative diseases.
The strength of this study is the development of an SEM-based QoL model using a relatively large sample of patients with DCAs. This modeling approach also enabled the incorporation of observed and unobserved variables, offering a more comprehensive and robust analysis compared to simpler regression methods or factor analysis. Therefore, SEM allows for the analysis of complex relationships between latent variables like QoL and rehabilitation, and it provides a clearer understanding of direct and indirect effects within a multidimensional framework. This perspective is particularly valuable, as future randomized controlled trials may face challenges in validating these findings due to the complexity and heterogeneity of the DCA population.
However, this study has some limitations. First, all data were collected using self-assessment methods, which introduces the potential for information bias due to the study participants underestimating or overestimating their responses. Nevertheless, the use of patient-reported outcomes (PROs) in research has become increasingly valuable in recent years. Although the standardized PROs of the DCAs were not available at the time of this study, it is worth noting that the Patient-Reported Outcome Measure of Ataxia (PROM-Ataxia)41 has become available in recent years. To include the multiple factors that are the objective of this study, some of the questions were developed by the researchers, but to improve the quality of the methodology, it would be more appropriate to narrow it down to established variables only. Second, the outcomes investigated in this study do not cover all the necessary elements. For example, regarding effective rehabilitation interventions, rehabilitation methods, costs, access to rehabilitation facilities, and individual or group interventions require investigation. However, the limitations of questionnaire surveys mean that they are limited to feasible survey items and to one-sided information from patients. Comprehensive information from both the medical facility and the patient may help to create a more extensive intervention framework. Third, the cross-sectional nature of the data limits our ability to infer clear causal relationships. However, the final model constructed with a relatively large sample has a reasonable goodness-of-fit index and high explanatory power; thus, it fulfills the significance of cross-cutting clarification of the QoL framework. Further longitudinal studies are necessary to strengthen the generalizability of our findings and ascertain causal relationships.
Our results suggest the need for sufficient quantity and quality of rehabilitation for patients with DCAs. The finding that disease duration was not significantly related to QoL is consistent with previous reports7,27 and we believe that rehabilitation is crucial at all stages of disease progression. In clinical practice, the visualized QoL framework for patients with DCAs is useful for demonstrating patient understanding and intervention efficacy at the individual level. On a broader scale, this framework is essential for promoting the development of a healthcare system that delivers enhanced and professional rehabilitation services.
In conclusion, the SEM of QoL in patients with DCAs conducted in this study confirms that all dimensions of patient-reported background, impairments, activity limitations, and rehabilitation factors significantly impact QoL in this population. These findings contribute novel insights into the effects of standard rehabilitation in patients with DCAs. Despite the varied impairments associated with DCAs, this study highlights the pivotal role of quantity and quality of rehabilitation in improving QoL, suggesting that tailored, comprehensive rehabilitation programs can positively influence patient outcomes.
Methods
Study design
This study was a cross-sectional, observational, nationwide mail survey of Japanese patients with DCAs, conducted from October to December 2020. Details of the study population and procedures using the same dataset were previously described42. The study is reported in line with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines.
This study was approved by the Ethics Committee of the Faculty of Health Science, Juntendo University (approval number: 20-012) and was performed in accordance with the ethical standards laid down in 1964 Declaration of Helsinki and its later amendments.
Participants
The study participants were people with Spinocerebellar Degeneration (SCD) and MSA. The eligibility criterion was membership of the Japanese SCD and MSA Patient Association. Exclusion criteria were the absence of a DCA diagnosis, age under 18 years, lack of consent for data use, and incomplete responses for more than 20% of the survey. Eligibility questions ensured that participants met the criteria for SCD or MSA, based on the diagnostic criteria defined by the Ministry of Health, Labor, and Welfare and the Japanese Society of Neurology for SCD (https://www.nanbyou.or.jp/entry/4880) and the Second Consensus Statement for MSA43.
The survey was anonymous, ensuring confidentiality. Informed consent was obtained through a written explanation accompanying the survey, with consent implied by the return of the questionnaire.
Study procedure and variables
The questionnaires were sent to all participants by post using the DCA association’s address database. Participants were asked to complete the self-report questionnaire and were allowed to seek assistance from a third party for reading or writing, if necessary. The questionnaire consisted of items selected by our research group and was written entirely in Japanese, with explanations provided for technical terms. It covered various aspects relevant to QoL, aligned with the study’s research goals, and included sociodemographic and medical information and PROs regarding clinical findings and rehabilitation. For variables for which there was no appropriate existing scale, we used simpler questions that had only two options (yes/no) or a total count. In contrast, the variables related to social support and quality of rehabilitation, which are PROs, were unified using a 0–10 Likert scale, as with the Short Form of the General Life Satisfaction Scale, which is described later, so that the participants could easily understand them.
Personal factors
Personal data, including age, sex, height, weight, and educational background, were collected. Body mass index (kg/m2) was calculated from height and weight.
Medical factors
Medical data included the name of the diagnosed disease, disease duration, common and disease-related complications, medication use, drugs for cerebellar symptoms and Parkinsonism, and history of falls in the past year. Participants first selected either the SCD or MSA category, then specified their diagnosis based on their physician’s assessment. Diagnostic categories included SCD, SCA, MSA, and unknown subtypes. Hereditary ataxia was classified as either pure (pure or relatively pure ataxias) or complex (complex ataxias), following the MDS Task Force classification4. MSA was categorized as a complex type. If only SCA and SCD diagnostic information was available, participants reporting symptoms for more than half of the 22 impairment items (rating > 1) were classified as complex, and the remaining participants were classified as pure. The number of medications used was defined by the number of different medications taken daily, regardless of dosage or the number of pills consumed. The use of medications for cerebellar symptoms and Parkinsonism was also recorded. Commonly diagnosed complications included cancer, cardiovascular disease, respiratory disease, gastrointestinal disease, kidney disease, cerebrovascular disease, diabetes mellitus, hypertension, and hyperlipidemia. Disease-related complications included orthostatic hypotension, postprandial hypotension, sleep apnea, and sudomotor dysfunction, based on physician diagnoses. Falls in the past year were categorized into four ordinal categories: none, once, 2–9 times, and more than 10 times.
Environmental factors
Environmental and social data included residence type, cohabitation, caregiver support, work status, and social support needs and satisfaction. For residence type, participants were asked whether they lived at home, in a hospital, or in an institution, and were categorized either as living at home or not. If the participant was temporarily hospitalized, they were still classified as living at home. Participants were also asked whether they lived with someone or had a caregiver (yes/no). Work status was categorized on a four-level ordinal scale: (1) not required to work (e.g., full-time housewives, retirees, students)/working without issues, (2) working with manageable health issues, (3) work adjustments due to health issues, or (4) temporary or permanent withdrawal from work due to health issues. Social support was assessed from two perspectives: actual conditions and perceived need. The need for social support was rated on an 11-point Likert scale, from “not needed at all” to “maximum need.” Satisfaction with social support was rated similarly, with responses ranging from “completely unfulfilling” to “completely fulfilling.”
Impairment factors
Symptom and impairment data included ataxia, muscle weakness/atrophy, muscle rigidity, spasticity, imbalance, malalignment, fatigue, pain, numbness, sensory disturbances, tremor, involuntary movements, orthostatic hypotension, poor sleep, respiratory disorder, speech/dysarthria, dysphagia, and visual, urinary, bowel, and cognitive impairments42. The severity score of each symptom was rated on a 4-point Likert scale, from 0 (no impairment) to 3 (severe impairment). Decreased endurance was not assessed, as participants found it difficult to distinguish this from fatigue. Depression was evaluated using the Patient Health Questionnaire-2, excluding the motor dysfunction items44.
Activity limitation factors
Activity limitations and participation restrictions were assessed using the mRS, BI, LSA, and indoor and outdoor mobility. The mRS was used to measure functional independence45. The BI was employed to assess basic ADLs via a modified Japanese self-rating version46. The LSA score was used to evaluate the degree of participation in one’s living space47. Additional information on mobility was collected. Indoor mobility was defined as the primary means of movement indoors, categorized as (1) bedridden, (2) wheelchair-bound, (3) crawling, (4) needing walking aids/assisted ambulation, or (5) independent ambulation. Outdoor mobility, defined as the primary means of movement outdoors, was categorized as (1) unable or totally dependent, (2) powered mobility devices, (3) manual mobility devices, (4) needing walking aids/assisted ambulation, or (5) independent ambulation.
Rehabilitation factors
Data on the following rehabilitation items was collected: types of rehabilitation implemented, types of therapy, total rehabilitation time, duration of rehabilitation, types of rehabilitation programs, self-rehabilitation, effects of rehabilitation, self-efficacy of rehabilitation, satisfaction with rehabilitation, and motivation for rehabilitation. Rehabilitation was defined as professional therapy administered by a qualified therapist. Self-rehabilitation, defined as a voluntary program, was included if performed independently, regardless of professional guidance. Types of rehabilitation implemented were defined as the total number of all implemented types and modes of professional rehabilitation. Types of therapy were selected from physical, occupational, and speech-language-hearing therapy (maximum of three). Total rehabilitation time was calculated as minutes per month based on the time and frequency of rehabilitation. The duration of rehabilitation was defined as the longest continuous intervention period, measured in months. The types of rehabilitation programs were counted based on the number of rehabilitation programs selected from 28 options (Supplementary Table S1). These variables were grouped under the latent variable “quantity of rehabilitation.” Rehabilitation-related PROs were assessed; they included the effects of rehabilitation, self-efficacy of rehabilitation, satisfaction with rehabilitation, and motivation for rehabilitation, using an 11-point Likert scale (0 = not at all, 10 = very much). These PRO variables were grouped under the latent variable “quality of rehabilitation,” as perceived by participants. The research team assumed a one-way causal relationship in the model, from quantity to quality of rehabilitation.
QoL factors
QoL was assessed using the EuroQoL-5 Dimension-5 Level (EQ-5D-5L), EQ-VAS, and life satisfaction. The EQ index48, ranging from 0 (lowest) to 1 (highest), was calculated from the EQ-5D-5L using the Japanese dataset49. Life satisfaction was measured using a validated 11-point Likert scale from the General Life Satisfaction Short Scale50.
Statistical analysis
The data was carefully examined to identify inconsistent or erroneous entries or impossible values and corrections were made when necessary. Descriptive statistics, such as means, SDs, and frequencies, were used to summarize all variables.
SEM was applied to explore causal relationships between latent variables and quantify the impact of rehabilitation factors on participants’ QoL. Figure 3 illustrates the theoretical model (Model 1), while Table 1 lists the observed and latent variables. The model was based on the International Classification of Functioning, Disability, and Health and incorporated QoL factors as endpoints and the rehabilitation components. Each observed variable was grouped under a corresponding latent variable. All latent variables were considered positive when they contributed positively to QoL, except impairment and activity limitation factors. Detailed SEM procedures and results are presented as supplementary information (Supplementary Methods and Supplementary Results). Before conducting SEM, an exploratory correlation analysis was performed to check for multicollinearity among the observed variables. Following this, a modified model (Model 2) was developed, where local solutions were validated using the preliminary CFA of each latent variable defined in the theoretical model (Supplementary Fig. S1). SEM was then performed for Model 2 using the mean- and variance-adjusted weighted least squares estimator. The SEM measurement model was statistically modified to establish the final model (Model 3). Model fit was assessed using the RMSEA, CFI, TLI, and SRMR51,52. From this final model, path coefficients, explanatory rates, and effect sizes (direct, indirect, and total) were calculated using the path coefficients.
Theoretical model (Model 1). Latent variables of interest are represented by ellipses. Observed variables and error terms are omitted from the figure for clarity (refer to Table 1 for their composition). The directional arrows indicate linear regression and the bidirectional arrows represent correlations between variables. Direct paths from background factors to QoL are omitted in the diagram.
Since no prior validated model evaluates QoL in patients with DCAs, the research team tested not only a comprehensive model with rehabilitation items (rehabilitation effects model) but also an additional sub-model that excluded rehabilitation factors (basic model). The analytical procedure for this basic model followed the same steps as the main SEM analysis, but excluded all variables and paths related to the quantity and quality of rehabilitation from Model 2. This basic model helps to clarify how pure impairments and activity limitations contribute to QoL and provides insights into disease characteristics. Comparing these models provides valuable insight into the contribution of rehabilitation factors to QoL outcomes in patients with DCAs.
SPSS version 29 (IBM Corp, Armonk, NY, USA) was used for descriptive analyses and Mplus version 8.1 (Muthén and Muthén, Los Angeles, CA, USA) for SEM.
Data availability
The data underlying this study, including anonymized participant responses and analytical models, will be shared upon reasonable request to the corresponding authors, subject to pre-specified criteria for confidential access.
Change history
16 September 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41598-025-18782-5
References
Gandini, J., Manto, M., Bremova-Ertl, T., Feil, K. & Strupp, M. The neurological update: therapies for cerebellar ataxias in 2020. J. Neurol. 267, 1211–1220 (2020).
Manto, M., Gandini, J., Feil, K. & Strupp, M. Cerebellar ataxias: An update. Curr. Opin. Neurol. 33, 150–160 (2020).
Malek, N., Makawita, C., Al-Sami, Y., Aslanyan, A. & de Silva, R. A systematic review of the spectrum and prevalence of non-motor symptoms in adults with hereditary cerebellar ataxias. Mov. Disord. Clin. Pract. 9, 1027–1039 (2022).
Lange, L. M. et al. Nomenclature of genetic movement disorders: Recommendations of the international Parkinson and Movement Disorder Society task force—An update. Mov. Disord. 37, 905–935 (2022).
Gorcenco, S., Karremo, C. & Puschmann, A. Patients’ perspective in hereditary ataxia. Cerebellum 23, 82–91 (2024).
Meyers, A. R., Gage, H. & Hendricks, A. Health-related quality of life in neurology. Arch. Neurol. 57, 1224–1227 (2000).
Schrag, A. et al. Health-related quality of life in multiple system atrophy. Mov. Disord. 21, 809–815 (2006).
Wenning, G. K. et al. The natural history of multiple system atrophy: a prospective European cohort study. Lancet Neurol. 12, 264–274 (2013).
Menon, P. J. et al. Health-related quality of life in patients with inherited ataxia in Ireland. Cerebellum 23, 1328–1337 (2024).
Winter, Y. et al. Health-related quality of life in multiple system atrophy and progressive supranuclear palsy. Neurodegener. Dis. 8, 438–446 (2011).
Low, P. A. et al. Natural history of multiple system atrophy in the USA: A prospective cohort study. Lancet Neurol. 14, 710–719 (2015).
Jacobi, H. et al. Evolution of disability in spinocerebellar ataxias type 1, 2, 3, and 6. Ann. Clin. Transl. Neurol. 9, 286–295 (2022).
Cassidy, E., Naylor, S. & Reynolds, F. The meanings of physiotherapy and exercise for people living with progressive cerebellar ataxia: An interpretative phenomenological analysis. Disabil. Rehabil. 40, 894–904 (2018).
Fonteyn, E. M. R. et al. The effectiveness of allied health care in patients with ataxia: A systematic review. J. Neurol. 261, 251–258 (2014).
Ilg, W. et al. Consensus paper: Management of degenerative cerebellar disorders. Cerebellum 13, 248–268 (2014).
Milne, S. C., Corben, L. A., Georgiou-Karistianis, N., Delatycki, M. B. & Yiu, E. M. Rehabilitation for individuals with genetic degenerative ataxia: A systematic review. Neurorehabil. Neural Repair 31, 609–622 (2017).
He, M., Zhang, H. N., Tang, Z. C. & Gao, S. G. Balance and coordination training for patients with genetic degenerative ataxia: A systematic review. J. Neurol. 268, 3690–3705 (2021).
Matsugi, A. et al. Effects of physiotherapy on degenerative cerebellar ataxia: A systematic review and meta-analysis. Front. Neurol. 15, 1491142 (2025).
Haraldstad, K. et al. A systematic review of quality of life research in medicine and health sciences. Qual. Life Res. 28, 2641–2650 (2019).
Kline, R. B. Principles and Practices of Structural Equation Modelling 4th edn. (Guilford Press, 2016).
He, L. et al. The key determinants to quality of life in Parkinson’s disease patients: Results from the Parkinson’s Disease Biomarker Program (PDBP). J. Parkinsons Dis. 6, 523–532 (2016).
Kasser, S. L. & Zia, A. Mediating role of resilience on quality of life in individuals with multiple sclerosis: A structural equation modeling approach. Arch. Phys. Med. Rehabil. 101, 1152–1161 (2020).
Lapin, B., Mate, K., Li, Y. & Thakore, N. Subjective health perception prioritizes psychological well-being over physical function in advanced ALS: A multigroup structural equation modeling analysis. J. Neurol. Sci. 442, 120442 (2022).
Zwecker, M. et al. Determinants of quality of life in individuals with spinal cord injury using structural equation modeling. Arch. Phys. Med. Rehabil. 103, 2375–2382 (2022).
Buchholz, M. et al. Health-related quality of life in patients with spinocerebellar ataxia: A validation study of the EQ-5D-3L. Cerebellum 23, 1020–1030 (2024).
Schrag, A. et al. A comparison of depression, anxiety, and health status in patients with progressive supranuclear palsy and multiple system atrophy. Mov. Disord. 25, 1077–1081 (2010).
Tamaš, O. et al. The impact of demographic and clinical factors on the quality of life in patients with neurodegenerative cerebellar ataxias. Brain Sci. 14, 1 (2023).
Jacobi, H. et al. Long-term evolution of patient-reported outcome measures in spinocerebellar ataxias. J. Neurol. 265, 2040–2051 (2018).
Schmitz-Hübsch, T. et al. Self-rated health status in spinocerebellar ataxia—Results from a European multicenter study. Mov. Disord. 25, 587–595 (2010).
Lo, R. Y. et al. Depression and clinical progression in spinocerebellar ataxias. Parkinsonism Relat. Disord. 22, 87–92 (2016).
Joyce, M. R. et al. Quality of life changes following the onset of cerebellar ataxia: Symptoms and concerns self-reported by ataxia patients and informants. Cerebellum 21, 592–605 (2022).
Schmitz-Hübsch, T. et al. Depression comorbidity in spinocerebellar ataxia. Mov. Disord. 26, 870–876 (2011).
Lai, R. Y. et al. Fatigue impacts quality of life in people with spinocerebellar ataxias. Mov. Disord. Clin. Pract. 11, 496–503 (2024).
Afonso Ribeiro, J. et al. Lower urinary tract and bowel dysfunction in spinocerebellar ataxias. Ann. Clin. Transl. Neurol. 8, 321–331 (2021).
Ferrans, C. E., Zerwic, J. J., Wilbur, J. E. & Larson, J. L. Conceptual model of health-related quality of life. J. Nurs. Scholarsh. 37, 336–342 (2005).
Miyashita, M. et al. Care burden and depression in caregivers caring for patients with intractable neurological diseases at home in Japan. J. Neurol. Sci. 276, 148–152 (2009).
Ilg, W. et al. Intensive coordinative training improves motor performance in degenerative cerebellar disease. Neurology 73, 1823–1830 (2009).
Miyai, I. et al. Cerebellar ataxia rehabilitation trial in degenerative cerebellar diseases. Neurorehabil. Neural Repair 26, 515–522 (2012).
Bogaert, A., Romanò, F., Cabaraux, P., Feys, P. & Moumdjian, L. Assessment and tailored physical rehabilitation approaches in persons with cerebellar impairments targeting mobility and walking according to the International Classification of Functioning: A systematic review of case-reports and case-series. Disabil. Rehabil. 46, 3490–3512 (2024).
Fonteyn, E. M. R., Keus, S. H. J., Verstappen, C. C. P. & van de Warrenburg, B. P. C. Physiotherapy in degenerative cerebellar ataxias: Utilisation, patient satisfaction, and professional expertise. Cerebellum 12, 841–847 (2013).
Schmahmann, J. D., Pierce, S., MacMore, J. & L’Italien, G. J. Development and validation of a patient-reported outcome measure of ataxia. Mov. Disord. 36, 2367–2377 (2021).
Haruyama, K., Kawakami, M., Miyai, I., Nojiri, S. & Fujiwara, T. COVID-19 pandemic and the international classification of functioning in multiple system atrophy: A cross-sectional, nationwide survey in Japan. Sci. Rep. 12, 14163 (2022).
Gilman, S. et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 71, 670–676 (2008).
Levis, B. et al. Accuracy of the PHQ-2 alone and in combination with the PHQ-9 for screening to detect major depression: systematic review and meta-analysis. JAMA 323, 2290–2300 (2020).
van Swieten, J. C., Koudstaal, P. J., Visser, M. C., Schouten, H. J. & van Gijn, J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke 19, 604–607 (1988).
Hachisuka, K., Ogata, H., Ohkuma, H., Tanaka, S. & Dozono, K. Test-retest and inter-method reliability of the self-rating Barthel index. Clin. Rehabil. 11, 28–35 (1997).
Baker, P. S., Bodner, E. V. & Allman, R. M. Measuring life-space mobility in community-dwelling older adults. J. Am. Geriatr. Soc. 51, 1610–1614 (2003).
Herdman, M. et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual. Life Res. 20, 1727–1736 (2011).
Ikeda, S. et al. Developing a Japanese version of the EQ-5D-5L value set. J. Natl. Inst. Public Health 64, 47–55 (2015).
Nießen, D., Partsch, M. & Groskurth, K. Data for: an English-language adaptation of the General Life Satisfaction Short Scale (L-1). Data file version 1.0.0. GESIS, Cologne https://doi.org/10.7802/2079 (2020).
Hu, L. & Bentler, P. M. Cutoff criteria for fit indexes in covariance structure analysis: Conventional criteria versus new alternatives. Struct. Equ. Model. 6, 1–55 (1999).
Hooper, D., Coughlan, J. & Mullen, M. R. Structural equation modelling: Guidelines for determining model fit. Electron. J. Bus. Res. Methods. 6, 53–60 (2008).
Acknowledgements
The authors express their gratitude to all the study participants and to the Japanese SCD and MSA Patient Association for their invaluable contributions. We also thank Editage (www.editage.jp) for providing English language editing services.
Funding
This work was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (grant number: 20K11219 to K.H.) and a Grant‐in‐Aid from the Research Committee of Ataxia, Health Labour Sciences Research Grant, the Ministry of Health, Labour and Welfare, Japan (JPMH20FC1041 to I.M.).
Author information
Authors and Affiliations
Contributions
KH: conceptualization, data curation, formal analysis, investigation, methodology, writing – original draft, and funding acquisition; MK: conceptualization, methodology, writing – review & editing, and project administration; IM: methodology and writing – review & editing; SN: formal analysis, methodology, and writing – review & editing; TF: methodology, writing – review & editing, and supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained errors in Table 1 and Table 2. Full information regarding the corrections made can be found in the correction for this Article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Haruyama, K., Kawakami, M., Miyai, I. et al. Impact of rehabilitation on quality of life in patients with degenerative cerebellar ataxias using structural equation modeling. Sci Rep 15, 21997 (2025). https://doi.org/10.1038/s41598-025-01990-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-01990-4