Abstract
Biomechanical analysis of crosstalk between the carotids remains poorly underexplored. This study aims to clarify the impact of severe carotid stenosis on the local hemodynamics of the opposite carotid artery, before and after treatment. Data from patients diagnosed with severe internal carotid artery stenosis (≥ 70%), treated with carotid artery stenting (CAS) or carotid endarterectomy (CEA) were analyzed. Both sides’ anatomical and hemodynamic remodeling parameters were evaluated before and after treatment. Forty-two patients underwent computed tomography angiography (CTA) and phase-contrast magnetic resonance imaging (PC-MRI) to measure carotid diameters and flow rates in the common, internal, and external carotids (CCA, ICA, and ECA, respectively) before and after CAS or CEA. Additionally, patient-specific computational fluid dynamics (CFD) simulations were performed to calculate time-averaged wall shear stress (taWSS) before and after the treatment. Post-treatment analysis revealed anatomical and hemodynamic changes in the contralateral side. Specifically, there was a decrease in blood flow and an increase in vessel diameters, particularly in the CCA, of patients who had undergone CEA (pre vs. post: flow p = .029, diameter p = .007). Additionally, there was an increase in low taWSS areas in the SOI after the operation: CEA from 15.50 (10.80) % to 18.00 (20.25) %, CAS from 17.00 (15.55) % to 19.50 (20.50) %. The study revealed new anatomical and hemodynamic parameters changes in both carotids, offering insights into the interaction between stenotic and contralateral carotid arteries in post-operative conditions.
Similar content being viewed by others
Introduction
Carotid stenosis is a major cause of stroke, which in turn is the main cause of long-term disability and the third leading cause of death1.
Usually, hemodynamically significant carotid stenosis is limited to one side, while the other side can be characterized by the absence of stenosis or the presence of mild-moderate stenosis that does not increase the risk of stroke.
Significant carotid stenosis may cause ipsilateral hemispheric stroke, and contralateral cerebral stroke is considered to be an incidental phenomenon, that however should not be neglected.
In fact, when a significant unilateral carotid stenosis is detected, hemodynamic disturbance and blood theft may occur in the contralateral hemisphere, causing poor ischemic tolerance and increasing the risk of stroke2.
Albeit the impact of stenosis treatments on cerebral perfusion has been extensively investigated from a clinical standpoint3, the biomechanical analysis of this intra-individual crosstalk between the carotids remains poorly investigated. One of the reasons is that most biomechanical studies available in the literature focus on local carotid hemodynamics of healthy subjects or isolated diseased geometries4,5.
Moreover, while it is known that both carotid endarterectomy (CEA) and carotid stenting (CAS) may induce a local remodeling on carotid morphology and hemodynamics, particularly in terms of smaller carotid bulb diameters and higher flow velocities after CAS compared to CEA-treated patients6, the assessment of the hemodynamic crosstalk between the treated and the contralateral carotid artery, after either CEA or CAS, is missing.
Based on these considerations, the present study aims to evaluate the impact of severe stenosis, before and after the vascular treatment, on the local anatomy and hemodynamics of the contralateral carotid by patient-specific data retrieved by medical image analysis and Computational Fluid Dynamics (CFD).
Results
Given the aim of the present study, five patients who did not undergo the surgical procedure were excluded. Among the 65 remaining patients, eight were excluded from preoperative computational analysis due to poor quality of CT images (n = 4), which prevented the faithful reconstruction of the vascular district of interest, or due to the absence of flow waves extracted from PC-MRI images (n = 4). Of the 57 patients analyzed in the preoperative condition, it was possible to analyze fluid dynamics through CFD simulations in only.
patients (24 CEA, 18 CAS) in the postoperative condition. Specifically, one patient died before the intervention, seven patients were lost to follow-up, and for six patients, it was impossible to analyze imaging data for geometry reconstruction or flow wave analysis. For the reasons listed, to compare the pre-and post-operative conditions between the treated stenotic side and the contralateral side, the results presented in this study considered 42 patients.
Comprehensive patient demographics and disease characteristics are reported in Table 1. Patients’ median (IQR) age was 71.00(8.20) years old. Males represented a significant majority, constituting 74% of the whole cohort of patients. They presented a median (IQR) SOI stenosis of 45.00(17.5)%, while the stenotic side showed a percentage of stenosis of 75.00(10.00)% and a median (IQR) VPS of 237.00(81.00) cm/s.
In Table 2, an overview of patient demographics and disease characteristics about the type of administered treatment is reported. Specifically, 24 patients underwent CEA, while 18 were treated with CAS. Notably, both groups exhibit comparability in terms of disease characteristics and age. However, an imbalance emerges within the CEA group concerning the gender variable, with 21 male and three female patients, whereas the CAS group comprises ten male and eight female patients. We also conducted statistical analysis between CAS and CEA groups on all demographic variables. Except for the gender variable (p =.02), all remaining demographic variables do not reach statistical significance.
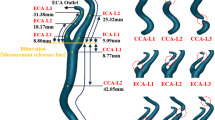
The comparison between the two sides of CCA and ICA parameters considering the same treatment is reported in Figs. 1, 2, 3, 4 and 5, while the value of each parameter is reported in Table 3.
Diameter of common carotid artery (CCA) is reported for each side and condition analysed for both type of treatment. Panel A shows the boxplot related to patients treated with carotid endarterectomy (CEA), while panel B illustrates the value related to subjects that performed stenting (CAS). P-value related to the comparison between SOI and STENOSIS are also reported. SOI = side of interest; PRE = pre-operative; POST = post-operative. P refers to Wilcoxon ranked-sign test.
Flow rate [mL/s] of common carotid artery (CCA) is reported for each side, and the condition analysed for both types of treatment. Panel A shows the boxplot related to patients treated with carotid endarterectomy (CEA), while panel B illustrates the value related to subjects that performed stenting (CAS). P-values related to the comparison between SOI and STENOSIS are also reported. SOI = side of interest; PRE = pre-operative; POST = post-operative. P refers to Wilcoxon ranked-sign test.
The diameter of internal carotid artery (ICA) is reported for each side, and condition analysed for both types of treatment. Panel A shows the boxplot related to patients treated with carotid endarterectomy (CEA), while panel B illustrates the value related to subjects that performed stenting (CAS). P-value related to the comparison between SOI and STENOSIS are also reported. SOI = side of interest; PRE = pre-operative; POST = post-operative. P refers to Wilcoxon ranked-sign test.
The flow rate [mL/s] of internal carotid artery (ICA) is reported for each side and the condition analysed for both types of treatment. Panel A shows the boxplot related to patients treated with carotid endarterectomy (CEA), while panel B illustrates the value related to subjects that performed stenting (CAS). P-value related to the comparison between SOI and STENOSIS are also reported. SOI = side of interest; PRE = pre-operative; POST = post-operative. P refers to Wilcoxon ranked-sign test.
The percentage of area exposed to low time average wall shear stress(taWSS) is reported for each side, and condition analysed for both types of treatment. Panel A shows the boxplot related to patients treated with carotid endarterectomy (CEA), while panel B illustrates the value related to subjects that performed stenting (CAS). P-values related to the comparison between SOI and STENOSIS are also reported. SOI = side of interest; PRE = pre-operative; POST = post-operative. P refers to Wilcoxon ranked-sign test.
Regarding the CEA group (see Figs. 1, 2, 3 and 4 A), our preoperative assessment involved a comparative analysis of the diameter and flow of CCA and ICA on the SOI and stenotic side. The results revealed disparities, with higher values for the diameter and flow of ICA and flow of CCA in the SOI compared to the stenotic side. As reported in Fig. 4A a significant difference was identified only for ICA flow (p <.001) with values of 3.33(0.93) m/s for the SOI and 2.18(1.22) ml/s for the stenotic side. No other statistically significant differences emerged among the remaining parameters, as also reported in Figs. 1A, 2A and 3A. Concerning the percentage of area exposed to low taWSS, the stenotic side seems to reach a higher value when compared to the SOI. However, statistical significance is not reached, as illustrated in Fig. 5A.
Regarding the CAS group (see Figs. 1, 2, 3, 4 and 5B), all parameters were found to be statistically insignificant between the two sides, except for the percentage of area exposed to low taWSS, which had a lower median value (p =.029) for the percentage of the area exposed to low taWSS (SOI: 17.00(15.55)% vs. STENOSIS: 11.05(14.05)%). However, there was also a noticeable trend for the diameter and flow of ICA and CCA of SOI, which were higher than those on the stenotic side.
No differences were identified in comparing the diameter and percentage of area exposed to low taWSS between the CEA and CAS groups in preoperative conditions. However, a significant difference was observed in the flow between the two procedures; details are reported in Table 4. The CEA group exhibited higher flows of ICA (Fig. 4) and CCA (Fig. 2), and a significant difference was highlighted for CCA flow (p =.020) and ICA flow (p =.002) on the SOI side.
The same tests were conducted on the postoperative data. In both groups, the differences in diameter and percentage of area exposed to low taWSS observed between the two sides during preoperative conditions were not present anymore. It was observed that the CCA flow and ICA flow on the SOI side in the CEA group were significantly higher than in the CAS group (CCA p =.010, ICA p <.001). A significant difference was found also on the stenotic side of CCA in the CEA group (p =.042). Conversely, no statistically significant difference was noted on the stenotic side among the CAS group for ICA flow (p =.102).
Furthermore, we comprehensively analysed pre- and post-operative results, categorising them by the type of surgery (also reported in Table 4).
We found a general trend within the CEA group, which showed a reduction in flow rate, an increase in diameter, and a nearly consistent percentage of area exposed to low taWSS. However, we could only identify statistically significant differences in the SOI. The diameter of CCA increased from 6.21(1.24) mm to 6.56(1.14) mm (p =.007), and the flow rate of CCA decreased from 6.47(2.77) ml/s to 5.33(2.29) ml/s (p =.029) after the treatment. The percentage of area exposed to low taWSS in SOI increased from 15.50(10.80) % to 18.00 (20.25) % after the treatment, but this increase was not statistically significant (p =.148).
Similarly, we conducted the same analysis for the CAS group, revealing a comparable trend but no significant changes were observed. Additionally, we found that the stenotic side showed a non-significant (p =.194) increase in the percentage of area exposed to low taWSS after surgery, transitioning from a preoperative value of 11.05 (14.05) % to 18.50 (12.30) %.
Discussion
The treatment of carotid stenosis involves local anatomical and hemodynamic remodelling, which is well-known. However, it is usually only analysed on the treated side. Examining the contralateral side and comparing pre- and post-operative conditions can help us understand the changes that occur locally on the untreated side. This approach enhances our understanding of the crosstalk mechanisms between the two sides. We conducted a comprehensive analysis examining imaging data and implementing patient-specific CFD simulations to achieve this. First, we compared the results by looking at the sides (SOI vs. stenosis) in pre- and post-operative conditions, stratifying by treatment type (CEA and CAS). Then, we studied the treatment (CEA vs. CAS) in pre- and post-operative conditions, stratifying by side. Finally, we compared the pre-and post-operative results (preop vs. postop) by stratifying them by side and treatment type.
The analyses showed that flow rates were consistently higher on the SOI before surgery, regardless of the type of surgery used to treat stenosis. The stenotic side had a larger area exposed to low values of taWSS. After treatment, hemodynamic and anatomical remodelling increased diameters on the untreated side and reduced flow rates. At the same time, the area exposed to low taWSS increased. Severe stenosis can cause blood flow to favour the healthier side for adequate cerebral perfusion4. Our findings confirm this, showing uneven flow distribution between the region of interest and the stenotic side. Typically, localised hemodynamic assessments of carotid arteries focus only on the treated area, overlooking the hemodynamic aspects of the contralateral side. Our clinical and CFD analyses reveal that the treatment of the pathologic segment facilitated a hemodynamic and anatomical reconfiguration on the contralateral side, showing the existence of a cross-linking phenomenon. This mechanism, which restores normal perfusion by removing calcified plaque, is statistically significant in the CEA group. The CAS procedure restores proper perfusion but may lead to small residual stenosis that may moderate the extent of anatomical and hemodynamic remodelling on the contralateral side, explaining the differences in results between the two groups under investigation.
From the clinical point of view, a better understanding of the mechanism underlying the cross-talking between the SOI and the contralateral side may help for a better stratification of patients who should undergo carotid revascularization, especially in case of asymptomatic lesions.
Furthermore, our study aimed to also increase the knowledge about possible different impacts of CEA and CAS on the postoperative vascular remodelling.
Stenosis treatment increases low taWSS on the SOI. Vessel remodelling and flow reduction induce lower taWSS with atherogenic potential. The presence of stenosis can lead to an unfavorable hemodynamic environment and SOI overload.
To analyse WSS distribution, we used fully patient-specific CFD analysis. Our study proposes CFD simulation of a large cohort of patients using CT and MRI data before and after surgery, which is a novelty. CFD simulations have been widely used to study atherosclerosis since Milner et al.‘s pioneering study7, but our approach is unique in using both CTA and MRI data. Other studies have shown the feasibility of computing hemodynamics in stenotic carotids given in-vivo measurements8,9,10,11. However, CFD simulations have often been used to integrate flow measurements performed by (idealised) in vitro stenosed carotid replicas12,13,14,15 or with a relatively small cohort of patients16.
At the same time, from the first report about time-resolved WSS vectors estimated directly from 3D-MRI data17, several studies have proved that in-vivo carotid hemodynamics, including WSS, can be measured non-invasively by 4D flow MRI18,19,20. However, there are still several technical limitations to tackle when using this approach, mostly dealing with space and time resolution of the methods21,22.
Our work has some limitations, which will be the object of future developments for further improvements. One of these limitations is using literature-based thresholds to identify low taWSS. To overcome this, we suggest calculating the mean taWSS in the preoperative case and using it as a threshold. This approach would show a patient-specific and relative difference in the percentage of the area subjected to low taWSS. A patient-specific approach is crucial in defining the pathology, and computational simulation can be an effective supplementary tool for stenosis treatment.
In addition, in this work, only the taWSS was considered as near-wall hemodynamics descriptors. Other descriptors accounting for pulsatility and multidirectionality could be analysed. Additionally, an analysis of the topological skeleton of the wall shear stress vector field could be conducted, further extending the hemodynamic characterization. Furthermore, in this study the percent area exposed to low taWSS was calculated in the entire carotid bifurcation model, in future it could be interesting considering stratifying the analysis according to specific locations.
Although some analyzed parameters result to be significantly different, others do not reach a statistically significant difference. While the anatomical and flow-related data obtained from CTA and PC-MRI showed statistically significant differences, the CFD-derived results did not reach statistical significance in either treatment group. The lack of significance in our study could be due to the small sample size of CAS and CEA groups. For this reason, we plan to expand our work by involving more patients.
Another limitation could be the imbalance of having more female CAS, given the smaller diameter of female patients compared to males. Other studies assess the significance of gender in determining CAS procedures’ benefit and clinical outcome showing no differences in immediate, and long-term CAS outcomes between gender23 or conforming no association with higher rates of stroke after CAS24. Future investigations are needed to explore if female gender influences the hemodynamic crosstalk between carotid arteries, as there is currently no compelling evidence available to suggest this. However, our study was not planned to analyse gender differences, and the enrolled patients were initially randomized to perform either CEA or CAS.
Finally, in the presented study the cerebral perfusion and its relationship with the stenosis presence and treatment were not elucidated, thus the analysis presented lacks information regarding the vertebral arteries and the Circle of Willis.
Methods
For this study, the treated carotid artery is labeled as stenosis, while the opposite side is defined as the side of interest (SOI).
Study cohort
All included patients were referred for a subgroup of patients enrolled in a randomized study named BAROX (ClinicalTrials.gov registration number: NCT03493971). BAROX study was approved by the local ethical committee (record number 62/int/2017, 8 June 2017). Inclusion criteria of this randomized trial were patients with ≥ 70% symptomatic or ≥ 80% asymptomatic internal carotid artery (ICA) stenosis according to the European Carotid Surgery Trial (ECST) method25, deemed fit for the treatment with either CAS or CEA, according to the international guidelines26. Exclusion criteria were age > 75 years, previous disabling stroke, contralateral carotid occlusion or > 70% stenosis, and other systemic diseases judged non-compatible with the procedures or randomization.
All enrolled patients were then randomized using a dedicated software to perform either CAS or CEA with patch closure. From March 2018 to March 2021, 70 patients were recruited.
Before the intervention, all patients underwent a Carotid Duplex ultrasound, computed tomography angiography (CTA), and phase-contrast magnetic resonance imaging (PC-MRI). After the operation, patients were followed up with Duplex ultrasound at 6, 12, and 24 months, and CTA and MRI at 12 and 24 months. According to the study protocol, all patients also performed a mini-mental test evaluation and a clinical evaluation before the procedure and at 6, 12, and 24 months after.
From these data of the BAROX study, the subanalysis of the present study included data from 42 patients (24 CEA and 18 CAS) that completed the follow-up with CTA and MRI and had high-quality images.
Imaging modalities
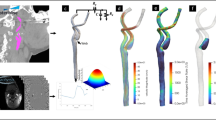
CTA of the supra-aortic trunks was performed (Siemens SOMATOM Definition AS, Erlangen, Germany, 64 slices). Acquisition parameters were 120 kV, 100 mAs, pixel size 0.45 mmx0.45 mm, and acquisition matrix 512 × 512. The images were reconstructed using a slice-thickness of 1 mm with an incremental factor of 0.5 mm. MRI was performed using a 1.5-T unit with 45-mT/m gradient power (Magnetom Aera Maestro Class, Siemens, Erlangen, Germany) and a head coil. Electrocardiographically triggered free-breathing through-plane phase-contrast (PC) sequences were performed for phase-velocity mapping with the following parameters: TR/TE = 4/3.2 ms, thickness 5 mm, and temporal resolution 41 ms. The flow was measured with PC sequences in the three vessels of interest CCA, ECA, and ICA. To this aim, two axial planes were acquired, one 20 mm below the bifurcation on the CCA, and one 30 mm above the bifurcation, including both the ICA and ECA lumens27. Velocity encoding (VENC) has been varied in different cases from 80 cm/s to 120 cm/s to exploit the maximum dynamic range of the velocity field and minimize image artifacts such as aliasing. All data were processed according to the workflow shown in Fig. 6 and described in the following paragraph.
Workflow is presented. All data were processed to obtain the anatomical and hemodynamic parameters of interest. CTA scans were segmented to reconstruct the 3D models of stenotic and controlateral carotid arteries. The models were used to extract patient-specific diameters. The phase contrast magnetic resonance images were processed to extract patient-specific flow waves of the common carotid artery (CCA), internal carotid artery (ICA), and external carotid artery (ECA); flow waves were used to analyze patient-specific flow rates and define the BCs. A surface mesh was defined to import the fluid domain into Simvascular to perform CFD simulations. The simulations were run, and the results were post-processed to evaluate the percentage of the luminal area exposed to low-time average wall shear stress (taWSS).
Imaging and data processing
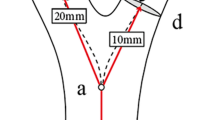
CTA scans were segmented to obtain right and left carotids using the Vascular Modeling Toolkit 8 (VMTK toolbox v1.4). The segmentation starts from the CCAs origin up to the base of the skull for ICAs and up to their first branch for ECAs, which usually takes off between 20 and 30 mm downstream of the bifurcation. CCAs were clipped proximally at a minimum distance of 7 diameters below the bifurcation, according to Hoi et al.27.
For both sides, luminal diameters were measured 20 mm below the bifurcation in the CCA, 30 mm above the bifurcation in the ICA, and 10 mm above the bifurcation in the ECA, as referred in Markl M18., to ensure a consistent comparison among patients and to avoid confounding factors related to the presence of the plaque.
PC-MRI data were analyzed with Medis software (Medis medical imaging systems BV, Leiden, Netherlands) to measure velocity and time-averaged flow waves.
Computational fluid dynamics simulations
CFD simulations were carried out in every patient’s stenotic and SOI carotids to find the luminal area exposed to atherogenic biomechanical stimuli, considering the near-wall hemodynamics through wall shear stress-based descriptors. After the lumen segmentation, non-structured uniform tetrahedral meshes were created with TetGen v1.513, with a target element size of 0.01–0.02 cm27, which yielded meshes between 0.7 and 2 million elements28, resolving satisfactorily the WSS field29. Pulsatile flow simulations were run with the software SimVascular v2019.08.0915 to solve the incompressible Navier-Stokes equations in a rigid-wall domain. Simulations were run for three cardiac cycles to damp transitory states, and the last cycle was used to inform the results26,30. Variations in the quantities of interest were < 5% along all the carotid bifurcation between the second and third cardiac cycles. Blood density was set at 1.06 g/cm3, viscosity 0.04 Poise, and a no-slip condition was imposed at the vessel wall31,32. For each patient, the cardiac cycle duration was based on the measurements at the PC-MRI scan; the time step was imposed as one-thousandth of the cycle period; thus 3000 time steps were run to simulate three cardiac cycles, as mentioned before.
We used patient-specific inlet and outlet flow boundary conditions (BC) following the strategy used by Cibis and colleagues19, where PC-MRI velocity is directly imposed as a flat profile at the origin of the CCA as inflow. Then, to fulfill the conservation of mass law on the outflow BC, both PC-MRI derived flows of ICA, and ECA were corrected by a constant. In particular, in the branch of the carotid bifurcation with the highest flow, we imposed a Dirichlet BC with a parabolic velocity profile. In contrast, a stress-free BC was used in the branch with a lower flow.
CFD temporally discretized results were analyzed. The time-averaged wall shear stress (taWSS) in the cardiac cycle was evaluated and processed according to cut-off values reported in the literature28,33 to identify three regions along the lumen: 1) low taWSS, defined as 0 < taWSS < 4 dyn/cm² and associated with atherosclerosis-prone regions; (2) physiological taWSS (10 < taWSS < 70 dyn/cm²); and (3) high-shear thrombogenic regions, where taWSS > 70 dyn/cm². The results are reported as a percentage of the total lumen area exposed to low taWSS, considering 20 mm below the bifurcation of the CCA, 30 mm above the bifurcation of the ICA, and 10 mm above the bifurcation of the ECA.
Statistical analysis
According to the type of treatment (CAS or CEA), the population was divided into two groups. Geometric and hemodynamic data between the two carotids were compared in preoperative and postoperative conditions. The Shapiro-Wilk test tested the normality of the distribution of values, and the variables sample was defined as non-normally distributed. The Wilcoxon signed-rank test compared CCA and ICA parameters, considering the same treatment. Concurrently, the Mann-Whitney test and Chi-Square test were used to analyze the differences between the two groups (CEA vs. CAS), used respectively for continuous and categorical variables. Accordingly, the percentage of areas exposed to low taWSS was also included in the analyzed parameters. All results are presented as median and interquartile range. Statistical significance was assumed at p <.050.
Data availability
Data will be provided on request to the corresponding author.
References
van der Bos, D. et al. Intracranial carotid artery atherosclerosis: prevalence and risk factors in the general population. Stroke 43 (7), 1878–1884. https://doi.org/10.1161/STROKEAHA.111.648667 (2012).
Li, M. et al. Lesion patterns and mechanism analysis of acute contralateral ischemic stroke accompanying stenosis of unilateral extracranial internal carotid artery. Brain Behav. 13 (9), e3111. https://doi.org/10.1002/brb3.3111 (2023).
AbuRahma, A. F. et al. Correlation of intraoperative collateral perfusion pressure during carotid endarterectomy and status of the contralateral carotid artery and collateral cerebral blood flow. Ann. Vasc Surg. 25 (6), 830–836 (2011).
Zhao, S. Z. et al. Inter-individual variations in wall shear stress and mechanical stress distributions at the carotid artery bifurcation of healthy humans. J. Biomech. 35 (10), 1367–1377 (2002).
Xue, Y., Gao, P., Lin, Y. & Dai, C. Preliminary study of hemodynamics in human carotid bifurcation by computational fluid dynamics combined with magnetic resonance angiography. Acta Radiol. 48 (7), 788–797 (2007).
Morales, M. M. et al. Morphological and hemodynamic patterns of carotid stenosis treated by endarterectomy with patch closure versus stenting: a duplex ultrasound study. Clinics 65 (12), 1315–1323 (2010).
Milner, J. S., Moore, J. A., Rutt, B. K. & Steinman, D. A. Hemodynamics of human carotid artery bifurcations: computational studies with models reconstructed from magnetic resonance imaging of normal subjects. J. Vasc Surg. 28 (1), 143–156 (1998).
Birchall, D., Zaman, A., Hacker, J., Davies, G. & Mendelow, D. Analysis of haemodynamic disturbance in the atherosclerotic carotid artery using computational fluid dynamics. Eur. Radiol. 16 (5), 1074–1083 (2006).
Xue, Y. J., Gao, P. Y., Duan, Q., Lin, Y. & Dai, C. B. Preliminary study of hemodynamic distribution in patient-specific stenotic carotid bifurcation by image-based computational fluid dynamics. Acta Radiol. 49 (5), 558–565 (2008).
LaDisa, J. F. et al. Time-efficient patient-specific quantification of regional carotid artery fluid dynamics and Spatial correlation with plaque burden. Med. Phys. 37 (2), 784–792 (2010).
Dong, J., Inthavong, K. & Tu, J. Image-based computational hemodynamics evaluation of atherosclerotic carotid bifurcation models. Comput. Biol. Med. 43 (10), 1353–1362 (2013).
Steinman, D. A., Poepping, T. L., Tambasco, M., Rankin, R. N. & Holdsworth, D. W. Flow patterns at the stenosed carotid bifurcation: effect of concentric versus eccentric stenosis. Ann. Biomed. Eng. 28 (4), 415–423 (2000).
Cheung, S. C. et al. Experimental and numerical study on the hemodynamics of stenosed carotid bifurcation. Australas Phys. Eng. Sci. Med. 33 (4), 319–328 (2010).
Buchmann, N. A., Yamamoto, M., Jermy, M. & David, T. Particle image velocimetry (PIV) and computational fluid dynamics (CFD) modelling of carotid artery haemodynamics under steady flow: a validation study. J. Biomech. Sci. Eng. 5 (4), 421–436 (2010).
Kefayati, S., Milner, J. S., Holdsworth, D. W. & Poepping, T. L. In vitro shear stress measurements using particle image velocimetry in a family of carotid artery models: effect of stenosis severity, plaque eccentricity, and ulceration. PLoS One. 9 (7), e98209 (2014).
Domanin, M. et al. Prediction of long term restenosis risk after surgery in the carotid bifurcation by hemodynamic and geometric analysis. Ann. Biomed. Eng. 47, 1129–1140 (2019).
Papathanasopoulou, P. et al. MRI measurement of time-resolved wall shear stress vectors in a carotid bifurcation model, and comparison with CFD predictions. J. Magn. Reson. Imaging. 17 (2), 153–162 (2003).
Markl, M. et al. In-vivo wall shear stress distribution in the carotid artery: effect of bifurcation geometry, internal carotid artery stenosis and recanalization therapy. Circ Cardiovasc Imaging 2010;CIRCIMAGING-110.
Cibis, M. et al. Relation between wall shear stress and carotid artery wall thickening MRI versus CFD. J. Biomech. 49, 735–741 (2016).
Harloff, A. et al. 3D blood flow characteristics in the carotid artery bifurcation assessed by flow-sensitive 4D MRI at 3T. Magn. Reson. Med. 61 (1), 65–74 (2009).
Szajer, J. & Ho-Shon, K. A comparison of 4D flow MRI-derived wall shear stress with computational fluid dynamics methods for intracranial aneurysms and carotid bifurcations—A review. Magn. Reson. Imaging. 48, 62–69 (2018).
Secchi, F. et al. Carotid Phase-Contrast magnetic resonance before treatment: 4D-Flow versus standard 2D imaging. Tomography 7 (4), 513–522 (2021).
Fornelli, F. et al. Could gender impact on immediate and Long-term carotid artery stenting outcome?? Insight from an Italian single center experience. Ann. Vasc. Surg. 76, 342–350 (2021).
Goicoechea, S. et al. Female gender increases risk of stroke and readmission after carotid endarterectomy and carotid artery stenting. J. Vasc. Surg. 75 (6), 1935–1944 (2022).
European Carotid Surgery Trialists’ Collaborative Group. Randomised trial of endarterectomy for recently symptomatic carotid stenosis: final results of the MRC European carotid surgery trial (ECST). Lancet 351, 1379–1387. https://doi.org/10.1016/S0140-6736(97)09292-1 (1998).
Naylor, R. et al. (eds) ‘s Choice - European Society for Vascular Surgery (ESVS) 2023 Clinical Practice Guidelines on the Management of Atherosclerotic Carotid and Vertebral Artery Disease. Eur J Vasc Endovasc Surg. ;65(1):7-111. (2023). https://doi.org/10.1016/j.ejvs.2022.04.011. Epub 2022 May 20. PMID: 35598721.
Hoi, Y., Wasserman, B. A., Lakatta, E. G. & Steinman, D. A. Effect of common carotid artery Inlet length on normal carotid bifurcation hemodynamics. J. Biomech. Eng. 132 (12), 121008 (2010).
Morbiducci, U. et al. Outflow conditions for image-based hemodynamic models of the carotid bifurcation: implications for indicators of abnormal flow. J. Biomech. Eng. 132 (9), 91005 (2010).
Moyle, K. R., Antiga, L. & Steinman, D. A. Inlet conditions for image-based CFD models of the carotid bifurcation: is it reasonable to assume fully developed flow? J. Biomech. Eng. 128 (3). https://doi.org/10.1115/1.2187035 (2006).
Gallo, D., Steinman, D. A. & Morbiducci, U. An insight into the mechanistic role of the common carotid artery on the hemodynamics at the carotid bifurcation. Ann. Biomed. Eng. 43, 68–81 (2014).
Khader, A. et al. Haemodynamics study in subject specific carotid bifurcation using FSI. Int. J. Mech. Aerosp. Ind. Mechatron. Manuf. Eng. 8, 1885–1890 (2014).
Kannojiya, V., Das, A. K. & Das, P. K. Simulation of blood as fluid: A review from rheological aspects. IEEE Rev. Biomed. Eng. 14, 327–341. https://doi.org/10.1109/RBME.2020.3011182 (2021).
Malek, A. M., Alper, S. L. & Izumo, S. Hemodynamic shear stress and its role in atherosclerosis. JAMA 282 (21), 2035–2042 (1999).
Acknowledgements
Valentina Ceserani acknowledges the charity #findtheorange Associazione onlus of Osborne Clarke.The authors thank Rodrigo M. Romarowski and Renato Vitale for their valuable contributions to the preliminary data collection and management.The authors also acknowledge dr. Massimiliano Marrocco-Trischitta, who participated in funding acquisition, study design, and enrollment/surgery of patients but passed away before the manuscript was completed and the final approval was given.
Funding
This study was partially supported by Ricerca Corrente funding from Italian Ministry of Health to IRCCS Policlinico San Donato.
Author information
Authors and Affiliations
Contributions
V.C.: dataset organization, development of methods, data analyses, manuscript writing and critical revision. M.C.: study conception and design, development of the proposed method, statistical analysis, manuscript critical revision. N.C.: manuscript writing and critical revision. F.S.: patient image collection, manuscript critical revision. I.B.: dataset organization, manuscript critical revision. V.B.: manuscript critical revision. P.R.: manuscript critical revision. G.N.: manuscript critical revision. D.M.: study conception and design, data collection, manuscript critical revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ceserani, V., Conti, M., Curcio, N. et al. The impact of stenosis treatment on the hemodynamic crosstalk between carotid arteries. Sci Rep 15, 20442 (2025). https://doi.org/10.1038/s41598-025-05466-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-05466-3