Abstract
Hemodialysis patients often face significant challenges related to their quality of life, which can be influenced by various dietary factors. Among these, dietary minerals and the phosphorus to protein ratio may play important roles in maintaining overall quality of life and its subscales. Despite the critical nature of this topic, there remains a dearth of studies examining these associations. Therefore, we aimed to investigate the relationship between dietary intake of phosphorus, calcium, magnesium, and the phosphorus to protein ratio with quality of life and its subscales in hemodialysis patients. The present multi-center cross-sectional study was performed on 323 maintenance hemodialysis patients. Dietary intake was obtained through a validated 168-item semi-quantitative food frequency questionnaire. The Kidney Disease Quality of Life Short Form (KDQOL-SF) was used to assess quality of life and its subscales. Exclusion criteria were applied to individuals whose daily caloric intake exceeded 4200 kcal or fell below 800 kcal. Nutritionist IV software was used to calculate overall energy and nutrient intake and SPSS software version 20 was utilized for data analysis. The results showed that lower phosphorus intake increased the odds of symptoms (OR = 2.65, 95% CI: 1.07–6.59), sleep disturbances (OR = 2.82, 95% CI: 1.18–6.71), and low emotional well-being (OR = 2.63, 95% CI: 1.09–6.31) in fully adjusted model, adjusted for age, sex, BMI, energy intake, Kt-V, and URR. Low calcium intake was also associated with higher odds of low role-physical component (OR = 1.88, 95% CI: 1.13–3.15) in age and sex adjusted model. However, high dietary calcium decreased odds of low QoL in general health subscale in fully adjusted model (OR = 0.28, 95% CI: 0.09–0.88). Low magnesium intake increased odds of low QoL in work status subscale, and physical and mental composite in age and sex adjusted model, while improved symptom/problem subscale in fully adjusted model. High phosphorus to protein ratio associated with low patient satisfaction in fully adjusted model (p-value < 0.05). The study indicated a relationship between the intakes of certain minerals with low QoL in HD patients. Phosphorus, calcium, and magnesium intake were positively associated with increased overall QoL scores, while an increased phosphorus-to-protein ratio was associated with a decline in QoL. Larger sample sizes and the inclusion of healthy control groups should be pursued in future research on this topic.
Similar content being viewed by others
Introduction
Chronic kidney disease (CKD) is a prevalent public health problem worldwide, with hemodialysis (HD) being a common treatment for end-stage renal disease (ESRD)1,2. Despite advancements in dialysis technology and adherence to clinical guidelines, morbidity and mortality rates among HD patients remain alarmingly high compared to other chronic conditions3,4. It is crucial to investigate the influence of quality of life (QoL) in order to address these disparities and bridge the existing gap. According to previous studies HD patients exhibit significantly lower QoL in comparison to the general population, which is correlated with inadequate dietary intake3,5,6,7.
Poor nutritional status significantly impacts the QoL in HD patients, driven by factors like inadequate dietary intake, reduced appetite, low quality of diet, high monotony index of diet, nutrient depletion during the dialysis process, and heightened catabolism8,9,10. Among dietary factors, understanding the potential link between dietary mineral intake, phosphorus to protein ratio, and QoL remains critical. The study by Brown-Tortorici reported that individuals with reduced dietary phosphorus consumption and dietary phosphorus to protein ratio exhibited an elevated risk of mortality11. However, Experimental investigations indicate that a diet rich in phosphorus is associated with an increase in vascular calcification, a decrease in bone mass, a reduction in glomerular filtration rate, and an elevation in mortality rates12,13. Meanwhile, there is currently no randomized controlled trial that substantiates the assertion that lowering dietary phosphate intake improves cardiovascular and renal outcomes in CKD and the latest KDIGO guidelines suggest dietary restriction solely for the management of hyperphosphatemia14. Dietary restrictions can adversely impact patients’ QoL, potentially resulting in unfavorable clinical outcomes and challenges in social interactions, which may contribute to anxiety and depression. The prescribed dietary limitations often lack flexibility, making it difficult for patients to identify acceptable food options7,9,15.
The association between dietary calcium intake and the progression of CKD appears to be marginal. Although excess intake (causing hypercalcemia/hypercalciuria) may promote the development of kidney stones, hypertension, and renal impairment; Both elevated and reduced serum levels have been linked to increased mortality and the progression of CKD16,17. Meanwhile, a positive association between calcium levels and QoL was demonstrated18. A prior investigation indicated that the dietary consumption of magnesium in HD patients fell below the recommended values19,20. Additionally, reduced dietary magnesium intake was associated with an increased risk of developing CKD and a decline in kidney function, which was subsequently linked to a higher probability of diminished QoL21,22.
Due to the lack of studies on dietary minerals (phosphorus, calcium, magnesium) and phosphorus-to-protein ratio in relation to QoL subscales, this study examines their associations in HD patients.
Materials and methods
Study population and protocol
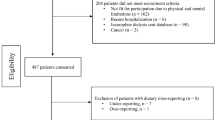
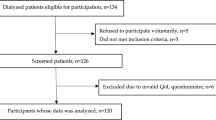
This cross-sectional study was conducted among 323 patients receiving maintenance HD from September 2021 to march 2022. Participants were recruited from five governmental and charitable hemodialysis facilities in Isfahan, Iran. Inclusion criteria required participants to be at least 18 years of age, have undergone maintenance hemodialysis for a minimum duration of three months, and possess both the capacity and willingness to participate in the study. Exclusion criteria were applied to individuals whose daily caloric intake exceeded 4200 kcal or fell below 800 kcal. To ensure that participants understood the significance, methodology, objectives, and timeline of the study, a concise explanation was provided to each individual. Subsequently, informed consent forms were completed by all volunteers prior to their involvement in the research. The study was conducted according to the declaration of Helsinki and the STROBE checklist. This study received ethical approval from The Research Ethics Committee of Isfahan University of Medical Sciences, Isfahan, Iran (IR.MUI.RESEARCH.REC.1399.605).
Dietary intake assessment
A validated semi-quantitative food frequency questionnaire (FFQ) consisting of 168 items was employed to assess the typical dietary intake of the study population over the past year23. Participants received instructions from a trained dietitian on how to complete this self-administered dietary assessment according to established protocols. They were prompted to indicate the frequency of consumption for various food items using six categorical options: ‘seldom/never’, ‘2–3 times per month’, ‘once per week’, ‘2–3 times per week’, ‘4–6 times per week’, and ‘daily’. Additionally, participants were required to specify the portion sizes for each food and beverage item. Consumption frequencies were converted to daily intake values, and portion sizes were quantified in grams using household measurements. Subsequently, the total food intake (g/day) was analyzed using Nutritionist IV software (Version 7; N-squared computing, OR, USA) to calculate overall energy and nutrient intake.
Quality of life assessment
The Kidney Disease Quality of Life Short Form (KDQOL-SF 1/3) was used to evaluate the QoL among patients undergoing hemodialysis. This instrument encompasses various kidney disease-related dimensions, including Symptom and problem, the effects of kidney disease on daily activities, the burden of kidney disease, work status, cognitive abilities, quality of social interactions, sexual health, sleep quality and duration, social support, encouragement from dialysis staff, and patient satisfaction. The questionnaire is structured into eight sections along with two summary scores: the physiological component summary and the physical component summary. It addresses aspects such as physical functioning, physical roles, bodily pain, general health perceptions, emotional well-being, psychological roles, social functioning, and energy/fatigue. Responses are scored on a scale ranging from 0 to 100, where higher scores reflect a better quality of life24. The reliability and validity of this questionnaire have been previously assessed and deemed satisfactory in Iranian patients undergoing hemodialysis25.
Anthropometric assessments
Dry weight was determined by adjusting post-dialysis weight to prevent hypovolemia or hypervolemia26. Participants, wearing light clothing and no shoes, were weighed using a calibrated digital scale (0.1 kg accuracy). Height was measured with a non-stretchable tape (0.1 cm precision) while participants stood barefoot against a wall. Body composition was analyzed using the Tanita MC-780MA analyzer (0.01 kg accuracy), assessing total body water, body mass index (BMI), body fat percentage (BFP), fat mass (FM), and fat-free mass (FFM). Furthermore, waist circumference (WC) was measured at the midpoint between the lowest rib and iliac crest with participants standing upright and relaxed.
Assessment of hemodialysis quality
To assess the hemodialysis quality, the Urea Reduction Ratio (URR) and Kt/V were applied. The URR was calculated as follows:
Additionally, Kt/V (t) was determined by multiplying the dialyzer’s urea clearance (K) by the time of dialysis (t), and then dividing this product by the subject’s urea distribution volume (V)27.
Other variables assessment
Data concerning patients’ age, gender, residential location, marital status, employment status, educational attainment, family income, smoking history, menopausal status, and alcohol consumption were obtained through patient interviews. Additionally, medical records provided information on medications, duration of dialysis treatment, frequency of dialysis sessions, total dialysis duration, comorbid conditions, and the underlying causes of renal failure.
Statistical analysis
The Kolmogorov–Smirnov test was applied to evaluate the normality of quantitative variables. The quantitative variables were illustrated as mean ± SD and qualitative variables as percentage. To compare the quantitative variables among three levels of phosphorus and calcium intake, one-way analysis of variance (ANOVA) was used. Quantitative variables between two levels of magnesium intake and phosphorus to protein ratio were examined by independent t-test. For categorical variables chi-square test was applied. The correlation of phosphorus, calcium, magnesium intake, and phosphorus to protein ratio with quality of life and its sub-scales was evaluated by Pearson correlation coefficient. To determine the association between phosphorus, calcium, magnesium intake, and phosphorus to protein ratio and quality of life as well as its sub-scales, multivariable logistic regression was applied. The odds ratios (ORs) and their 95% confidence intervals (95% CIs) were calculated in crude and adjusted models. The SPSS software version 20 (IBM, Chicago, IL) was used to perform analysis and p-value < 0.05 (two-sided) was considered statistically significant.
Results
In total, 323 HD patients were included in this study. The mean age of the patients with phosphorus intake < 700 mg/day and > 1000/day mg was 59.86 and 56.59 years, respectively, representing a statistically significant difference (p-value < 0.05). In addition, participants with magnesium intake below the Recommended Dietary Allowance (RDA) demonstrated a significantly higher mean age compared to those exceeding it (P = 0.03). However, no significant age differences were noted among groups with varying calcium, and phosphorus/protein intake. Phosphorus, calcium, and magnesium intake also differed significantly between sexes, with the majority of individuals with higher intake being male (p-value < 0.05) (Table 1).
Anthropometric indices revealed that fat mass significantly varied among individuals with different calcium and phosphorus intake levels. Specifically, an increase in calcium and phosphorus intake was associated with a decrease in fat mass (p-value < 0.05). No significant differences in HD indices were observed between patients with respect to calcium, phosphorus, magnesium, and phosphorus/protein intake.
A statistically significant but weak positive correlation was observed between calcium (r = 0.22), magnesium (r = 0.26), and phosphorus (r = 0.24) intake and total quality of life (QoL). Conversely, a Statistically significant yet weak negative correlation was found between phosphorus/protein intake (r= −0.12) and total QoL (P-value < 0.05) (Table 2). Slight significant positive correlations were also observed between SF-36 components and calcium, magnesium, and phosphorus intake, while a weak, albeit statistically significant negative correlation was observed with phosphorus/protein intake (p-value < 0.05). Additionally, a significant association was observed between the intake levels of phosphorus, calcium, magnesium, and phosphorus/protein with certain components of ESRD-targeted Areas, such as symptoms, the impact of kidney disease, the occurrence of kidney disease, and dialysis staff encouragement (p-value < 0.05) (Table 2).
Multivariate adjusted odds ratio and 95% confidence interval for the association of the dietary intake of phosphorus, calcium, magnesium, and phosphorus to protein ratio with low quality of life and its sub-scales are demonstrated in Table 3. Strikingly, lower phosphorus intake (< 700 mg/day) was robustly associated with reduced scores in symptoms/problems (OR = 2.42, 95% CI: 1.16–5.02), burden of kidney disease (OR = 2.37, 95% CI: 1.16–4.86), sleep quality (OR = 2.71, 95% CI: 1.32–5.56), pain (OR = 2.90, 95% CI: 1.40–5.98), and other components such as emotional well-being (OR = 3.33, 95% CI: 1.60–6.92), energy/fatigue (OR = 2.58, 95% CI: 1.24–5.36), and the SF-12 physical (OR = 2.60, 95% CI: 1.24–5.46), and mental composites (OR = 2.57, 95% CI: 1.23–5.37). These associations exhibited remarkable consistency across sequential adjustments: age and sex adjustments preserved links to sleep quality (OR = 2.46, 95% CI: 1.16–5.20), pain (OR = 2.29, 1.07–4.90), emotional well-being (OR = 3.01, 1.41–6.41), energy levels (OR = 2.38, 95% CI: 1.11–5.08), and the SF-12 physical (OR = 2.22, 95% CI: 1.01–4.91), and mental composites (OR = 2.35, 95% CI: 1.09–5.04); meanwhile further adjustments for BMI and energy intake upheld decreased symptoms/problems (OR = 2.45, 1.01–6.01), sleep quality (OR = 2.89, 1.22–6.84), and emotional well-being (OR = 2.74, 1.15–6.52). Even after accounting for dialysis adequacy (Kt-V and URR), lower phosphorus intake remained a significant predictor of symptoms/problems (OR = 2.65, 95% CI:1.07–6.59), sleep disturbances (OR = 2.82, 95% CI: 1.18–6.71), and emotional well-being (OR = 2.63, 95% CI:1.09–6.31).
Higher phosphorus intake (> 1000 mg/day) was related to reduced scores in cognitive function (OR = 2.22, 95% CI: 1.22–4.05), and sleep quality (OR = 1.81, 95% CI: 1.01–3.26). Adjustments for age and sex preserved this relation with cognitive function (OR = 2.44, 95% CI: 1.30–4.57), and sleep quality (OR = 2.45, 95% CI: 1.19–4.20). Further adjustments for BMI, and energy intake also intensified the association with sleep quality (OR = 3.20, 95% CI: 1.46–7.02), overall health deterioration (OR = 2.34, 95% CI: 1.08–5.02), patient dissatisfaction (OR = 2.30, 95% CI: 1.06–5.02), and physical functioning limitation (OR = 2.33, 95% CI: 1.02–5.31). Controlling for dialysis adequacy in addition to previous factors also intensified the relation in a way that elevated the odds of low sleep quality (OR = 3.20, 95% CI: 1.46–7.02), overall health deterioration (OR = 2.33, 95% CI: 1.08–5.03), patient dissatisfaction (OR = 2.31, 95% CI: 1.05–5.05), and reduced physical functioning (OR = 2.33, 95% CI: 1.02–5.30) was observed. Intriguingly, higher phosphorus intake exhibited a protective association with physical role functioning before (OR = 0.35, 0.19–0.64) and after age and sex adjustments (OR = 0.42, 0.22–0.78).
Moreover, our analysis revealed striking associations between calcium intake and multidimensional health outcomes. Participants with low calcium intake (< 1000 mg/day) exhibited a nearly twofold increase in the likelihood of reduced total QoL (OR = 1.86, 95% CI: 1.16–2.98) and a 2.27-fold higher odds of impaired physical role (OR = 2.27, 95% CI: 1.40–3.68). Notably, the detrimental association between low calcium intake and role-physical limitations remained robust after adjusting for age and sex (OR = 1.88, 95% CI: 1.13–3.15).
Conversely, higher calcium consumption (> 2000 mg/day) demonstrated protective associations across multiple domains. These intake levels were associated with a 61% improvement in social interaction quality (OR = 0.39, 95% CI: 0.16–0.99), and significant enhancements in physical role (OR = 0.37, 95% CI: 0.14–0.93), emotional role (OR = 0.33, 95% CI: 0.13–0.84), and increased energy levels accompanied by reduced fatigue (OR = 0.26, 95% CI: 0.10–0.70). Higher intakes were also related to better general health (OR = 0.42, 95% CI: 0.18–0.98) and the SF-12 mental composite (OR = 0.35, 95% CI: 0.13–0.87). Meanwhile, higher calcium intake was associated with reduced odds of kidney disease effects (OR = 0.42, 95% CI: 0.17–0.99).
These relationships retained statistical significance after controlling for confounders. Age and sex adjusted models preserved associations with general health (OR = 0.40, 95% CI: 0.17–0.97), emotional role (OR = 0.37, 95% CI: 0.14–0.95), and energy levels (OR = 0.29, 95% CI: 0.11–0.78). Remarkably, the relationship between higher calcium intake and improved general health persisted even after stringent adjustments for BMI, energy intake (Model 2: OR = 0.28, 95% CI: 0.09–0.88), and dialysis adequacy (Model 3: OR = 0.28, 95% CI: 0.09–0.88).
Furthermore, based on our findings magnesium intake below the Recommended Dietary Allowance (RDA) was associated with decreased total QoL (OR = 1.89, 95% CI: 1.18–3.04), reduced effects of kidney disease (OR = 1.73, 95% CI: 1.07–2.79), and reduced perceived encouragement from dialysis staff (OR = 2.20, 95% CI: 1.34–3.61). Furthermore, magnesium insufficiency demonstrated significant adverse associations across multiple SF-36 domains. Specifically, magnesium deficiency was related to diminished physical functioning (OR = 1.91, 95% CI: 1.18–3.10), greater limitations in both physical role (OR = 3.11, 95% CI: 1.89–5.10), and emotional role components (OR = 2.12, 95% CI: 1.30–3.44), impaired social functioning (OR = 1.67, 95% CI: 1.03–2.71), and reduced energy levels accompanied by heightened fatigue (OR = 1.95, 95% CI: 1.20–3.17). These associations extended to the SF-12 composite scores, with marked reductions in physical (OR = 1.81, 95% CI: 1.11–2.96) and mental health (OR = 1.93, 95% CI: 1.18–3.15) dimensions. Notably, after rigorous adjustment for age, and sex (Model 1), the link between inadequate magnesium intake and adverse outcomes persisted robustly. The association with diminished dialysis staff encouragement (OR = 1.99, 95% CI: 1.19–3.31), impaired work status (OR = 1.75, 95% CI: 1.02–3.01), both physical (OR = 2.62, 95% CI: 1.15–4.37) and emotional role limitations (OR = 1.81, 95% CI: 1.10–2.99), reduced energy levels (OR = 1.64, 95% CI: 1.01–2.71) and worse SF-12 mental composite scores (OR = 1.72, 95% CI: 1.04–2.86), remained statistically significant. Intriguingly, our findings revealed a robust association between lower magnesium intake and symptoms/problems, after rigorous adjustment for age, sex, BMI, and total energy intake (OR = 0.30, 95% CI: 0.13–0.70). Strikingly, this association persisted consistently after further controlling for dialysis adequacy (OR = 0.30, 95% CI: 0.13–0.69).
On the other hand, we identified significant associations between higher than median phosphorus/protein intake (median: 17.92 mg/g) and outcomes related to QoL and its sub-scales. Specifically, consumption exceeding the median was associated with low effects of kidney disease (OR = 1.58, 95% CI: 1.01–2.48), worsened work status (OR = 1.96, 95% CI: 1.22–3.17), lower encouragement from dialysis staff (OR = 2.42, 95% CI: 1.53–3.81), decreased patient satisfaction (OR = 1.92, 95% CI: 1.21–3.06), impaired physical role functioning (OR = 1.68, 95% CI: 1.07–2.63), and reduced SF-12 physical composite scores (OR = 1.76, 95% CI: 1.12–2.78). Furthermore, after adjusting for age, sex in Model 1, the association between greater than median phosphorus/protein intake and worsened work status (OR = 2.08, 95% CI: 1.27–3.43), diminished dialysis staff encouragement (OR = 2.42, 95% CI: 1.52–3.86), reduced patient satisfaction (OR = 1.88, 95% CI: 1.18–3.01), impaired physical role functioning (OR = 1.59, 95% CI: 1.01–2.52), and decreased SF-12 physical composite scores (OR = 1.65, 95% CI: 1.02–2.66) remained robust. In addition, the relationship between higher phosphorus/protein intake and diminished dialysis staff encouragement (OR = 2.21, 95% CI: 1.31–3.72) and reduced patient satisfaction (OR = 2.06, 95% CI: 1.23–3.46) persisted even after stringent adjustments for BMI, energy intake. Remarkably, these associations persisted resolutely when further controlling for dialysis adequacy, retaining their statistical relevance for both reduced staff encouragement (OR = 2.14, 95% CI: 1.27–3.63) and lower satisfaction (OR = 2.10, 95% CI: 1.25–3.53).
Discussion
Hemodialysis is recognized as a high-tech treatment for ESRD. Despite its effectiveness, patients receiving HD face multiple physical, psychological, and social stressors that are yet to be addressed. The cumulative impact of these stressors leads to reduced QoL in patients28,29. The present study aimed to examine the relationship between dietary mineral intake and the phosphorus-to-protein ratio with QoL and its subscales in HD patients. The results clearly indicated a significant relationship between the intake of phosphorus, calcium, magnesium, and the phosphorus-to-protein ratio with QoL. Specifically, as the intake of phosphorus, calcium, and magnesium increased, the overall QoL score improved in these patients, whereas an increase in the phosphorus-to-protein ratio was associated with a decrease in the total QoL score.
Numerous studies have been conducted on the impact of nutritional factors on QoL in HD patients, with evidence suggesting that nutritional status significantly impacting QoL components in these patients3,9,30. Visiedo et al. demonstrated that the nutritional status of HD patients was significantly associated with QoL components. They identified malnutrition as the most important predictor of QoL for the patients9. Our findings were consistent with previous studies, showing that the intake of certain minerals, such as phosphorus, calcium, magnesium, and the phosphorus-to-protein ratio, were significantly associated with to QoL in HD patients31,32. Malnutrition, particularly protein-energy malnutrition, is a common problem among patients undergoing HD and is associated with increased risk of mortality and morbidity33,34. Due to its association with infections and cardiovascular diseases, malnutrition also leads to a reduction in life expectancy and a decline in QoL for these patients35,36. Multiple causes can contribute to malnutrition in HD patients, including dietary restrictions, metabolic acidosis, gastroparesis, medications that suppress appetite, an acute or chronic systemic disease that triggers an inflammatory response, the catabolic process of dialysis, and elevated serum leptin levels8,37. The systematic review by Ouirdani et al. indicates that nutritional education positively influences several factors in patients undergoing hemodialysis. These factors include biological markers, quality of life, healthcare costs, adherence to dietary guidelines, knowledge acquisition, malnutrition-related inflammation, dietary intake, weight change, and behavioral modifications38.
The findings of our study also suggest that nutritional components, such as mineral intake, can account for the variation in QoL among HD patients. Guo et al. examined the association between various nutrient intakes and health-related quality of life in patients undergoing dialysis. demonstrated. This study demonstrated a positive association between the intake of total calories, fat, niacin, and vitamin E and HRQoL in HD patients, indicating the significant relationship between nutritional status and HRQoL32.
Depression and cognitive impairment are prevalent in patients undergoing HD. A decline in appetite often serves as an initial indicator of these issues, which could contribute to reduced dietary intake and subsequent micronutrient deficiencies in these patients34,39. In a study by Mekki et al., which followed patients for 9 years, time on dialysis was associated with decreased intake of milk, dairy products, meat, fish, eggs, fruits, vegetables, and fats. Additionally, the intake of calcium and iron decreased with more time spent on HD. The reduction in the intake of vitamins A, C, and E in cases of prolonged HD may be attributed to altered taste, increased thirst, dietary restrictions, nausea, and vomiting associated with uremia40.
Overall, the effects of these indicators on QoL are complex and difficult to predict, but there is good reason to believe that nutritional interventions may confer significant clinical benefits. The dialysis diet is among the most restrictive, which may lead to frustration, impose a significant burden, and result in suboptimal adherence to treatment, which in turn could worsen QoL and patient satisfaction. Adherence to evidence-based dietary guidelines, particularly for micronutrient intake, is critical to ensure optimal nutrient sufficiency and mitigate risks associated with both under- and overconsumption. Dietary regimens must be carefully tailored to individual clinical profiles and metabolic demands, guided by standardized protocols, to prevent nutritional imbalances that may adversely impact patient quality of life.
As a novel contribution, the current study is a cross-sectional investigation and, to the best of our knowledge, is the first to examine the relationship between dietary mineral intake, the phosphorus to protein ratio, quality of life, and its subscales among patients undergoing maintenance hemodialysis in Iran. A strength of our study is the relatively large and representative sample, which includes patients from five governmental and charitable hemodialysis centers in Isfahan, Iran. However, ethnic and geographic homogeneity, limits the generalizability of the findings. Caution is warranted when applying these results to ethnically diverse populations or other regions. Furthermore, the use of validated questionnaires and the adjustment for potential confounding variables enhance the robustness of our findings. Nonetheless, several limitations warrant consideration. We employed a self-reported semi-quantitative FFQ to assess dietary intake, which may introduce measurement errors and misclassification of participants. Additionally, recall bias is another limitation associated with this type of questionnaire that should be acknowledged. Furthermore, unknown and residual confounders must also be considered in the interpretation of our results. Finally, this study has a cross-sectional design, which, due to the nature of such studies, limits our ability to determine causal relationships.
Conclusion
The results of this study clearly indicated a relationship between nutritional status and the intake of certain minerals with QoL improvements in HD patients. Phosphorus, calcium, and magnesium intake were positively associated with increased overall QoL scores, while an increased phosphorus-to-protein ratio was associated with a decline in QoL. Larger sample sizes and the inclusion of healthy control groups should be pursued in future research on this topic.
Data availability
Data will be available on reasonable request from the corresponding author.
References
Sanyaolu, A. et al. Epidemiology and management of chronic renal failure: a global public health problem. Biostatistics Epidemiol. Int. J. 1 (1), 11–16 (2018).
Wouk, N. End-stage renal disease: medical management. Am. Family Phys. 104 (5), 493–499 (2021).
Daniel, S. C., Azuero, A., Gutierrez, O. M. & Heaton, K. Examining the relationship between nutrition, quality of life, and depression in Hemodialysis patients. Qual. Life Res. 30, 759–768 (2021).
USRDS. The USRDS end-stage renal disease incident and prevalent quarterly update. (2018). www.usrds.org/adr.htm
Sułkowski, L., Matyja, A. & Matyja, M. Social support and quality of life in hemodialysis patients: A comparative study with healthy controls. Medicina 60(11), 1732 (2024).
Marte, J. L. C., Javier, A., Ruiz-Matuk, C. & Paulino-Ramirez, R. Quality of life and nutritional status in diabetic patients on Hemodialysis. Diabetes Metabolic Syndrome: Clin. Res. Reviews. 13 (1), 576–580 (2019).
Nagy, E., Mahmoud, M., El-kannishy, G. & Sayed‐Ahmed, N. Impact of malnutrition on health‐related quality of life in patients on maintenance Hemodialysis. Therapeutic Apheresis Dialysis. 25 (4), 467–474 (2021).
Sahathevan, S. et al. Understanding development of malnutrition in Hemodialysis patients: a narrative review. Nutrients 12 (10), 3147 (2020).
Visiedo, L. et al. The impact of nutritional status on health-related quality of life in Hemodialysis patients. Sci. Rep. 12 (1), 3029 (2022).
Viramontes-Hörner, D., Pittman, Z., Selby, N. M. & Taal, M. W. Impact of malnutrition on health-related quality of life in persons receiving dialysis: a prospective study. Br. J. Nutr. 127 (11), 1647–1655 (2022).
Brown-Tortorici, A. R. et al. The interplay between dietary phosphorus, protein intake, and mortality in a prospective Hemodialysis cohort. Nutrients 14 (15), 3070 (2022).
Cannata-Andía, J. B., Román-García, P., Carrillo-López, N. & Dusso, A. S. Clinical and Preclinical Evidence of the Skeletal and Vascular Adverse Health Effects of High Dietary Phosphorusp. 31–44 (CRC, 2017).
Rubio-Aliaga, I. & Krapf, R. Phosphate intake, hyperphosphatemia, and kidney function. Pflügers Archiv-European J. Physiol. 474 (8), 935–947 (2022).
KDIGO. Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney international supplements. 2017;7(1):1–59. (2017).
Forfang, D., Edwards, D. P. & Kalantar-Zadeh, K. The impact of phosphorus management today on quality of life: patient perspectives. Kidney Med. 4 (4), 100437 (2022).
Janmaat, C. J. et al. Lower serum calcium is independently associated with CKD progression. Sci. Rep. 8 (1), 5148 (2018).
Mazzaferro, S. et al. Focus on the possible role of dietary sodium, potassium, phosphate, magnesium, and calcium on CKD progression. J. Clin. Med. 10 (5), 958 (2021).
dos Santos, A. C., Machado Mdo, C., Pereira, L. R., Abreu, J. L. & Lyra, M. B. Association between the level of quality of life and nutritional status in patients undergoing chronic renal Hemodialysis. Jornal Brasileiro De Nefrologia. 35 (4), 279–288 (2013).
Bossola, M. et al. Dietary intake of trace elements, minerals, and vitamins of patients on chronic Hemodialysis. Int. Urol. Nephrol. 46, 809–815 (2014).
Bogacka, A., Sobczak-Czynsz, A., Kucharska, E., Madaj, M. & Stucka, K. Analysis of nutrition and nutritional status of haemodialysis patients. Roczniki Państwowego Zakładu Higieny 69(2), 165–174 (2018).
Rasyid, H., Kasim, H. & Sampebuntu, J. Quality of life in patients with renal failure undergoing Hemodialysis. Acta Med. Indones. 54 (2), 307 (2022).
Koh, H. B. et al. Association between dietary magnesium intake and incident chronic kidney disease: a prospective observational cohort study. Am. J. Clin. Nutr. 120 (4), 964–972 (2024).
Mirmiran, P., Esfahani, F. H., Mehrabi, Y., Hedayati, M. & Azizi, F. Reliability and relative validity of an FFQ for nutrients in the Tehran lipid and glucose study. Public Health. Nutr. 13 (5), 654–662 (2010).
Hays, R. D. et al. Kidney Disease Quality of Life Short Form (KDQOL-SF™), Version 1.3: a Manual for Use and Scoring7994 (RAND, 1997).
Amir, N., Tong, A., McCarthy, H. & Howell, M. Trajectories of Quality of Life in Chronic Kidney Disease: a Novel Perspective of Disease Progressionp. 1563–1565 (Oxford University Press, 2021).
Sinha, A. D. & Agarwal, R. (eds) Setting the Dry Weight and its Cardiovascular Implications. Seminars in dialysis (Wiley Online Library, 2017).
Maduell, F. et al. Urea reduction ratio considering Urea rebound. Nephron 78 (2), 143–147 (1998).
García-Martínez, P. et al. Predictive model of variables associated with health-related quality of life in patients with advanced chronic kidney disease receiving Hemodialysis. Qual. Life Res. 29, 1817–1827 (2020).
Al Salmi, I. et al. Kidney Disease-Specific quality of life among patients on Hemodialysis. Int. J. Nephrol. 2021 (1), 8876559 (2021).
Ng, H-M. et al. Is malnutrition a determining factor of health-related quality of life in hemodialysis patients? A cross-sectional design examining relationships with a comprehensive assessment of nutritional status. Qual. Life Res. 31(5), 1441–1459 (2022).
Saglimbene, V. M. et al. Dietary intake in adults on hemodialysis compared with guideline recommendations. J. Nephrol. 34(6), 1999–2007 (2021).
Guo, Y. et al. Varying association of nutrient intakes with quality of life in patients receiving different modes of dialysis. Front. Cardiovasc. Med. 11, 1407650 (2024).
Macedo, C., Amaral, T. F., Rodrigues, J., Santin, F. & Avesani, C. M. Malnutrition and sarcopenia combined increases the risk for mortality in older adults on Hemodialysis. Front. Nutr. 8, 721941 (2021).
Kanda, E. et al. The combination of malnutrition-inflammation and functional status limitations is associated with mortality in Hemodialysis patients. Sci. Rep. 11 (1), 1582 (2021).
Rashid, I. et al. Malnutrition as a potential predictor of mortality in chronic kidney disease patients on dialysis: A systematic review and meta-analysis. Clin. Nutr. 43(7), 1760–1769 (2024)
Yamada, S. et al. Malnutrition-inflammation complex syndrome and bone fractures and cardiovascular disease events in patients undergoing hemodialysis: the Q-cohort study. Kidney Med. 4 (3), 100408 (2022).
Mali, N., Ge, J., Su, F., Li, C. & Fan, W. Review of risk factors of malnutrition in maintenance Hemodialysis patients. Archives Nephrol. Urol. 5 (1), 24–33 (2022).
Ouirdani, M. et al. (eds) Impact of Nutrition Education on Various health-related Components of Hemodialysis Patients: A Systematic Review (MDPI, 2024).
Iseki, K. Nutrition and quality of life in chronic kidney disease patients: a practical approach for salt restriction. Kidney Res. Clin. Pract. 41 (6), 657 (2022).
Mekki, K., Remaoun, M., Belleville, J. & Bouchenak, M. Hemodialysis duration impairs food intake and nutritional parameters in chronic kidney disease patients. Int. Urol. Nephrol. 44 (1), 237–244 (2012).
Funding
This study was supported by Isfahan University of Medical science. The funders had no role in study design; collection, analysis, and interpretation of data; writing of the report; the decision to submit the report for publication.
Author information
Authors and Affiliations
Contributions
MH.R, F.M and M.B; conception and design of the work, S.F, F.N and F.SH; conduction of the work, F.SH and R. Z; analysis and interpretation of data, F.SH, M.B, MH.R and FS.HJ; draft the work and revise it, and all authors have approved the submitted version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was ethically approved by The Research Council and Ethical Committee of Isfahan University of Medical Sciences, Isfahan, Iran, (Code: IR.MUI.RESEARCH.REC.1399.605). Participants were provided with an information sheet explaining the study protocol, and consented to participate. Mohammed Hossein Rouhani as the lead author affirms that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned have been explained.
Consent for publication
Informed consent was obtained from all subjects and/or their legal guardian(s).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shahdadian, F., Javaheri, F.S.H., Shams, F. et al. Association of dietary minerals and phosphorus to protein ratio with quality of life in Iranian Hemodialysis patients: a cross-sectional study. Sci Rep 15, 22010 (2025). https://doi.org/10.1038/s41598-025-05637-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-05637-2