Abstract
Lungs receive blood supply from the pulmonary and bronchial arteries. After transplantation, lungs uniquely suffer from ischemia and injury to the airways, in part due to the surgical sacrifice of the bronchial artery circulation during procurement and implantation. Ex-vivo lung perfusion (EVLP) offers a platform to rehabilitate and assess donor lungs; however, it traditionally excludes bronchial circulation. We hypothesized that bronchial artery perfusion during EVLP may enhance airway preservation. A dual-circulation EVLP platform was developed using swine donor lungs with preserved bronchial arteries. Blood was separately routed to the pulmonary artery and to the bronchial arteries. Laser speckle contrast imaging (LASCA) was performed pre- and post-initiation of bronchial perfusion, and perfusion units were compared within lungs. Endpoint lung function and tissue integrity were assessed via histology and TUNEL staining after 6 h of perfusion. Six porcine lungs were successfully mounted on the dual-circulation EVLP system. Bronchial perfusion significantly increased airway perfusion (p = 0.03), with trends toward improved parenchymal perfusion as well. Histology demonstrated preserved architecture and less necrosis in the airways of dually perfused lungs. TUNEL staining demonstrated significantly reduced bronchial epithelial apoptosis in dual-perfused lungs compared to PA-only perfusion (p < 0.001). This study demonstrates the feasibility of dual-circulation EVLP in a large animal model and reveals early protective effects of bronchial artery perfusion on airway tissues. These findings suggest that incorporating bronchial perfusion into EVLP platforms may mitigate early ischemic airway injury, a key contributor to post-transplant complications and chronic lung allograft dysfunction. Further investigation is warranted to assess functional outcomes and long-term benefits, and to evaluate integration with bronchial artery revascularization strategies.
Similar content being viewed by others
Introduction
Lung transplantation is the gold standard treatment option for end-stage pulmonary failure1. However, lungs face the lowest survival post-transplant among the major solid organ transplants, with a median survival of nearly 6 years, compared to greater than 13 for hearts, 17 for livers, and 19 for kidneys1,2. Uniquely, lungs are perfused via a dual circulation system, with the majority of deoxygenated blood flow coming from the pulmonary artery (PA), and a minority of oxygenated blood flow mostly directed to the airways via the bronchial arteries, originating from the thoracic aorta3,4,5,6,7.
During conventional lung transplantation, these bronchial arteries are sacrificed, leaving the transplanted airways hypoxic3. This is further exacerbated by protracted complement-mediated microvascular destruction driven by rejection8. The resultant aberrant circulation to the airways makes lung transplants particularly prone to airway complications in the short term such as stenosis, dehiscence, and bronchial infection, as well as bronchiolitis obliterans syndrome (BOS) in the long term—the number one reason for allograft failure in lung transplantation3.
Ex-vivo lung perfusion (EVLP) platforms have emerged as potent vehicles not only for extended lung observation, but rehabilitation and therapy9,10,11. However, given that the bronchial arteries are routinely sacrificed, these platforms are not currently equipped to perfuse them, creating an opportunity for improvement in preservation of the airways.
We sought to utilize EVLP as a method for preserving the bronchial arteries that are usually sacrificed in lung transplantation. While techniques have been described to preserve the bronchial arteries upon procurement of the lungs, there are no described methods aimed at utilizing them during EVLP12. We hypothesize that perfusion of the bronchial circulation during EVLP will better preserve the health of the airways, and thus we have developed an EVLP platform that re-establishes perfusion to the bronchial artery system.
Results
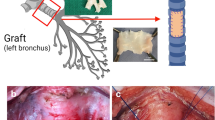
The EVLP platform (Fig. 1; Supplementary Fig. 1) consisted of a reservoir (Medtronic, Santa Clara, CA) with ¼-inch heparin-coated tubing that routes blood to an Oxygenator (Medtronic) controlled by an oxygen mixer (Sechrist Industries Inc., Anaheim, CA). Since oxygenation is performed by the lungs using a ventilator, the oxygenator is used instead for challenge gasses using a gas mixture composed primarily of nitrogen and C02 to de-oxygenate the blood prior to pulmonary perfusion, as well as temperature maintenance using a heater cooler (Stockert GmbH, Freiberg, Germany). From the oxygenator, blood was routed to two separate roller pumps (MasterFlex GmbH, Gelsenkirchen, Germany). One roller pump directed blood flow into the pulmonary artery via a cannula, and the other into a blind pouch of aorta encompassing the origin of the bronchial arteries. The lungs were mounted in an organ chamber designed to minimize the surface area of lung in contact with the basin via a sleeve, with the ability to reposition the lungs in various dependency states. The pulmonary veins drained into a collection system at the base of the basin and blood was returned to the reservoir.
The circuit was primed with 1 L of OCS Lung Solution (TransMedics, Andover, MA) and 1 L of donor pig blood. Additives to the priming solution include 500 mg of Methylprednisolone, 1 unit of multivitamins, 20 IU of insulin, 4 mg of milrinone, 40 mEq of sodium bicarbonate, 200 mg of ciprofloxacin, and 200 mg of voriconazole.
Our modified EVLP platform successfully directed perfusion into two separate outflow tracts with differential flow rates (Fig. 2A). Six swine lungs were procured with the bronchial arteries intact and mounted onto our modified EVLP platform (Fig. 2B-C). Average cold ischemia time was 30.5 ± 7.2 min from cold flush to warm reperfusion. Cold ischemic times ranged from 21 to 41 min. After flushing, and warming, PA-only circulation was commenced. First, angiography was performed to confirm successful procurement of the bronchial arteries, which were intact in all 6 lungs (Fig. 3; Supplementary Video 1).
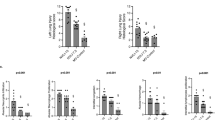
Flow was then commenced at a rate of 500mL/min in the PA only circulation, while the bronchial circulation pump was kept off. After successful venous return and flushing, the first round of LASCA was performed. Bronchial circulation was then commenced at a rate of 120 mL/min, a rate determined in a separate pilot run where perfusion was measured during isolated flow into the bronchial arteries. After 15 min of perfusion, LASCA was re-obtained (Fig. 4), demonstrating a mean 94.6 ± 32.4 perfusion unit increase (p = 0.03). The greatest improvements in perfusion were seen in the visible airways (p = 0.03), though non-statistically significantly increased parenchymal perfusion was also observed (p = 0.36). Slow stepwise increases towards target perfusion parameters, rewarming and initiation of ventilation was performed as previously described10,13.
In three swine lungs, bronchial circulation was continued in addition to standard EVLP. At six hours of perfusion, EVLP was terminated in both treatment groups, and tissue samples were taken. Endpoint PaO2/FiO2 (P/F) ratios of dual-perfused lungs versus traditional EVLP were 564.67±223.60 vs. 357.00±158.66 (p = 0.26). Dynamic compliance was 49.53±5.71 mL/mmHg vs. 54.03±24.11 mL/mmHg (p = 0.77) in dual-perfused lungs versus traditional EVLP. Representative H&E and TUNEL staining is shown in Fig. 5. In the PA-only circulation group, there appeared to be more pallor in the bronchial cartilage and signs of necrosis and loss of cell nuclei consistent with ischemic injury to the bronchi. On TUNEL staining, higher concentrations of TUNEL positivity were observed in the bronchial epithelium in the PA-only circulation, consistent with increased cell death (p < 0.001).
Discussion
We developed a dual circulation EVLP platform capable of perfusing both the PA and bronchial artery circulation. We confirmed successful harvest and perfusion of the bronchial arteries via fluoroscopy and observed significantly increased perfusion particularly in the airways after initiation of bronchial circulation in addition to PA perfusion. This increased perfusion may have a protective effect on the bronchiolar tissue, particularly in the cartilaginous regions.
Lack of bronchial circulation has been implicated in anastomotic issues after lung transplantation, namely stenosis, stricture, dehiscence, and bronchial infection5,8,14. The constellation of these airway complications occur in as many as 1 in 3 lung transplant recipients and likely lower survival after lung transplantation15,16. Repeat interventions to manage the mechanical complications of airway ischemia, including bronchial stenting or balloon dilation, and even reoperation, are expensive, morbid, and lead to prolonged length of stay and prolonged dependency on positive pressure ventilation17,18,19,20. Regeneration of the bronchial circulation is unpredictable, and takes up to 4 weeks before any significant improvements in anastomotic health are observed16.
Bronchial infection in particular remains a devastating component of airway complications20. In a study of 400 bronchial anastomoses in lung transplantation without any measures for bronchial artery revascularization, Muñoz-Fos et al. demonstrated a 12.5% incidence of mechanical airway complications, of which over half required further intervention. They noted that in patients who developed bronchial infection at the anastomosis, a mechanical complication became over two times more likely20. Because compromised bronchial circulation itself is a risk factor for bronchial infection, airway complications act synergistically with each other and threaten anastomotic health3,5.
Additionally, mounting evidence has tied airway ischemia with the initiation/acceleration of chronic lung allograft disease (CLAD)8. The two hits of surgical sacrifice of the bronchial arteries, followed by complement mediated microvascular destruction of the remaining bronchial vessels appears to be linked to the development of bronchiolitis obliterans syndrome (BOS) according to insights derived from an murine orthotopic tracheal transplant model employed by the Nicolls group3,4,5,8,21,22,23. Additionally, Luckraz et al. demonstrated that on autopsy of 11 lung transplant recipients whose grafts failed due to BOS, there were significantly fewer identifiable micro-vessels compared to lung transplant recipients whose cause of death was unrelated to BOS24,25.
This work builds upon the foundation set by Tanaka et al. who demonstrated successful dual circulation of the bronchial arteries and pulmonary arteries in rat EVLP26. Perfusion of the bronchial arteries was achieved in rat heart-lung blocs via cannulation of the descending thoracic aorta, similar to our method. Rat heart-lung blocs undergoing dual circulation had improved P/F ratios, dynamic compliance, and pulmonary vascular resistance compared to PA-only perfusion blocs26. Additionally, there was evidence of decreased inflammatory burden in dually circulated blocs26. Our present study advances this foundation by translating the concept of a dual perfusion EVLP platform to a clinically relevant large animal model. This work is primarily a feasibility study that demonstrates the technical steps in performing dual circulation in porcine lungs and provides evidence of improved perfusion, which, with further study, may pave the way for human translation.
Our work adds to the growing understanding that bronchial circulation is important to the health of lung allografts in revealing that even after 6 h of lack of bronchial circulation, there is a significant increase in markers of cell death specifically at the bronchial epithelium and compromised health of the cartilaginous airway as well, given our histological and immunohistochemistry results. Perfusing the bronchial circulation during EVLP appears to have a protective cellular-level effect on the health of the airways, in a time course that is shorter than described in previous studies which note that ischemic changes usually manifest days after transplantation5,27. While there was a trend towards improved P/F ratios in the lungs receiving dual circulation, further study is needed to confirm functional benefits to perfusing the bronchial circulation on EVLP.
Furthermore, the benefits of bronchial artery revascularization (BAR) via sewing of a bypass graft onto the origin of the bronchial vessels has been described by Yun et al.28 In 131 lung transplants with successful BAR, the group reports 5-year survival at 80% compared to 50% in a contemporaneous International Society of Heart and Lung Transplantation Registry study.28 Additionally, they show reduced numbers of BOS compared to registry data as well, hypothesizing that improved bronchial circulation allows for better pharmaceutical and protective immune cell components to localize to the transplanted airways. Despite this reported success however, BAR is not a widely adopted component of lung transplantation, with the vast majority of BAR being performed at the Cleveland Clinic and by the Copenhagen Group12,28. This may be due to perceptions of the increased technical complexity of BAR during lung transplantation, as well as the additional time requirements, despite reports that the skin-to-skin operating time is unchanged with BAR.28 We hypothesize that modifying EVLP to include perfusion of the bronchial circulation however poses minimal additional technical challenge or operating room time, but may better preserve the airways throughout transport, without immediate ischemic signaling present upon implantation. This may be easier to adopt on a wide basis at centers already performing EVLP, if these simple modifications may be adopted and implemented by conventional EVLP platforms. However, we hypothesize that the ultimate durable benefit to bronchial circulation may be a combination of preservation of the bronchial circulation during EVLP, followed by BAR so that the bronchial circulation sees minimal interrupted perfusion, and the airways continue to receive oxygenated blood flow after implantation.
Beyond clinical adoption of bronchial circulation during EVLP, this platform may serve as a model to study the impact of bronchial ischemia on airway health. Our EVLP platform is uniquely equipped with advanced imaging, tissue acquisition, perfusion, and functional testing capabilities that could be used to fully characterize the time course and impact of airway ischemia prior to transplantation of the lungs.
Limitations
This is a preliminary feasibility study meant to describe the technical steps and improvements in perfusion when performing dual bronchial and PA circulation EVLP. Though there are small animal numbers in the present study, the cohort sizes were chosen based on power calculations derived from preliminary data on this subject. Previous trial runs of dual circulation EVLP were performed in n = 6 porcine lungs, ranging in size from 15kg to 75kg, testing various cannulation methods, including direct cannulation of the bronchial arteries and trialing in-house designed dispersion cannula tips (Supplementary Fig. 2) before settling on standard aortic cannulation. Using this preliminary data, we determined that a two-sample t-test with 4 animals provided 98% power to detect a 150-unit difference in perfusion units assuming a pooled standard deviation of 50 units (Cohen’s d = 3.0), with α = 0.05 (two-tailed), and opted for n = 6 pigs for the study. While the feasibility of perfusing the bronchial circulation on EVLP was confirmed, functional assessments as well as long term implantation studies are needed to confirm whether these effects durably improve allograft health. Furthermore, this study utilized healthy porcine lungs. Future studies would be necessary to examine the impact of bronchial circulation on rehabilitation of injured lungs. Key insights included that the modified procurement strategy reliably preserves the bronchial arteries, and they are amenable to perfusion at 120 cc/min. Additionally, we uncovered a cellular protective characteristic to bronchial circulation during EVLP with early differences in bronchial airway health, albeit in a small sample size. Furthermore, the modifications made to EVLP were made to a device designed at our institution, commercial adoption of bronchial perfusion during EVLP will require further study. It is yet unclear if there is a benefit to perfusing the bronchial circulation during EVLP, without subsequent BAR. However, as transport time and distance continue to increase in lung transplantation, particularly in the composite allocation score era29,30we hypothesize this method may decrease ischemia to the airways during organ preservation, and have immediate and long-term benefit.
Conclusion
Bronchial artery devascularization during lung transplantation has lasting negative effects on the health of lung allografts, which currently have the lowest survival among conventional solid organ transplants. Measures like BAR have been introduced to mitigate this ischemic injury but face limited utilization. We introduced a simple modification to an EVLP platform that facilitated perfusion of the bronchial arteries during EVLP, which resulted in an early protective cellular effect localized to the airways. This platform serves as a clinically translatable method of preserving the bronchial circulation during procurement, with minimal added time and complexity.
Methods
Ex-Vivo lung perfusion platform
We developed an open drainage ex-vivo lung perfusion (EVLP) platform utilizing a common reservoir and oxygenator with two roller-head pumps directed towards two separate cannulas. The first cannula was directed to the pulmonary artery. The second was directed to the aorta. This configuration was used to perfuse the pulmonary artery and the bronchial arteries simultaneously.
Porcine lungs
All experimental protocols were approved by the Stanford University Administrative Panel on Lab Animal Care (APLAC), and all methods were carried out in accordance with the USDA Animal Welfare Act, and the Stanford Assurance of Compliance with PHS Policy on Humane Care and Use of Laboratory Animals. Adult, male and female Yorkshire swine weighing 50–70 kg were anesthetized using inhaled isoflurane 1–5% before undergoing humane euthanasia with intravenous potassium chloride. Upon confirmation of death, lungs were procured and mounted on EVLP. The end point for the lungs were six hours post-EVLP perfusion.
A modified procurement strategy was used to preserve the entire descending thoracic aorta, as well as the esophagus, to preserve the bronchial arteries as described by Unai et al.12 3.1 L of Perfadex (XVIVO, Goeteborg, Sweden) was perfused through the lungs, 1 L antegrade through the pulmonary artery, and 500 cc retrograde through each pulmonary vein prior to harvest. Additionally, 100 cc was perfused through the aorta.
Once explanted, the pulmonary artery was cannulated with an 18 French cannula, and the aorta was then cannulated with a 16 French aortic cannula after stapling off the distal and proximal portions and suture ligating the spinal arteries. The pulmonary artery and aortic cannulas were then connected to their respective tubing from the EVLP circuit. Perfusion was initially initiated only through the pulmonary artery circuit at an initial rate of 500–750 mL/min and then gradually increased to 35 mL/min/kg, which is 50% of the estimated cardiac output of a pig and is concordance with a modification of the Lund protocol for open-drainage EVLP31,32.
Baseline laser contrast speckle imaging (LCSI) was obtained using the RFLSI-ZW Laser Speckle Contrast Imaging System (RWD Systems, Sugar Land, TX) with isolated pulmonary artery flow. Then, bronchial circulation was initiated, and after 15 additional minutes of perfusion, repeat imaging on LCSI was obtained. To remove motion artifact from imaging, Valsalva maneuvers were performed just prior to image acquisition.
Laser contrast speckle imaging involves the combination of thermal imaging with refractor laser patterns to sense fluid movement. Using an artificial intelligence algorithm, an angiogram is recreated with the ability to overlay thermal data on it to produce dynamic visualization of blood flow. Based on a prespecified color scale, perfusion units are calculated by the software associated with the camera, and comparisons may be made among groups. Perfusion units are standardized and dimensionless but can provide information on relative perfusion changes33,34.
In three pigs, bronchial perfusion was then continued for a total perfusion time of 6 h. In the remaining three, bronchial circulation was terminated after the initial images, and only PA perfusion was maintained until the end of the experiment. At the end of 6 h of perfusion, a repeat image was obtained, and then the lungs were removed from EVLP. The pigs that received both bronchial and PA perfusion were chosen randomly to avoid any learning curve bias in performing bronchial EVLP.
Ventilator settings
Ventilation was provided by a clinical-grade mechanical ventilator (Draeger, Lubeck, Germany) with a positive end expiratory pressure of 5 mmHg, tidal volumes of 6–8 mL/kg, and a respiratory rate of 12–14 respirations/minute. The FiO2 was 21% (room air) as these were healthy uninjured lungs.
Functional assessment
Nitrogen challenge gases with full deoxygenation of perfusate were used to assess function, with titrations made to ventilator settings based on findings to optimize the blood gases in both groups. From challenge gasses, P/F ratios were obtained. Additionally, endpoint dynamic compliance was measured by the following formula: \(\:\frac{Tidal\:Volume}{Peak\:Inspiratory\:Pressure-Positive\:End\:Expiratory\:Pressure}\).
Tissue fixation and sectioning
Lung parenchymal samples were taken upon the completion of EVLP from each treatment arm. Samples were preserved in 4% paraformaldehyde (PFA) (Sigma-Aldrich, St. Louis, MO) before being transferred to 70% ethanol 24 h after initial preservation. Samples were then embedded in paraffin and sliced to 8 μm sections and stored for staining.
Epredia’s Shandon Rapid-Chrome H&E Frozen Section Staining Kit (Epredia, Kalamazoo, MI) was utilized to perform hematoxylin & eosin (H&E) staining. After deparaffinization, slides were fixed in Rapid-Fixx for 5–7 s and then rinsed in distilled water for 5–7 dips with each dip lasting 2–3 s. Each slide was then immersed in hematoxylin for 1 min before rinsing again in distilled water for another 5–7 dips. The slides were dipped 3 times in Bluing Reagent for 2–3 s each dip. Slides were then washed in 95% Alcohol for 5–7 dips before being immersed in Eosin-Y for 13 s. The slides were then dipped in a separate 95% ethanol, 100% ethanol, and another 100% ethanol each 5–7 times for 2–3 s per dip. Slides were then dipped in two separate washes of Xylene 5–7 times each for 2–3 s per dip. All slides were mounted with Shandon-Mount (Epredia, Kalamazoo, MI) and sealed with glass coverslips. The stained sections were imaged on the Keyence All-in-One Fluorescence Microscope BZ-X810 series (Keyence, Cupertino, CA).
Terminal deoxynucleotidyl transferase-dUTP Nick end labeling (TUNEL)
The Invitrogen Click-iT Plus TUNEL Assay Kit with Alexa Fluor 594 dye (Thermo Fisher Scientific, Waltham, MA) was used to perform a terminal deoxynucleotidyl transferase-dUTP nick end labeling (TUNEL) stain. This assay works to detect in situ apoptosis by staining damaged 3’-OH ends of DNA with terminal deoxynucleotidyl transferase (TdT). Slides were washed in PBS for 5 min. In a humidified chamber, slides were permeabilized in Proteinase K for 15 min and then washed again in PBS for 5 min. Slides were then covered with 4% PFA for 5 min followed by PBS and Deionized water washes. The slides were covered with TdT Reaction Buffer and incubated at 37ºC for 10 min. TdT reaction mixtures were added to experimental slides and more TdT Reaction Buffer was added to control slides to be incubated in a humidified chamber at 37ºC for 60 min. Slides were washed with 3% Bovine Serum Albumin (BSA) and 0.1% Triton X-100 for 5 min. Click-iT Plus TUNEL reaction cocktail was added to slides and incubated away from light at 37ºC for 30 min. Slides were rinsed in PBS and then mounted with VectaShield Antifade Mounting Medium with DAPI (VectorLabs, Newark, CA). Coverslips were placed on slides and sealed with clear nail polish. Slides were stored at 4ºC until imaged on the Keyence microscope (Keyence, Cupertino, CA). For quantification, two bronchi from each sample were randomly chosen, and staining was quantified within those bronchi. Parenchymal staining was not considered.
Fluoroscopy
Angiographic assessment of pulmonary and bronchial circulation was performed with a mobile fluoroscopic C-arm (Phillips, Amsterdam, Netherlands) via cine capture during hand injection of 50 cc of a 50/50 mixture of normal saline and iodinated contrast. The pulmonary artery circulation was imaged first, then the bronchial circulation in isolation with the EVLP circuit paused.
Statistical analysis
Descriptive statistics are used to characterize means and standard deviations. When comparing LASCA perfusion differences, paired T-testing was performed given that means were compared within samples pre- and post-initiation of dual bronchial and pulmonary artery circulation. Unpaired T-tests are used to quantify differences in TUNEL staining. Quantification of stain positivity was performed in ImageJ 1.54k (National Institutes of Health, Bethesda, MD). Analysis of histology and immunohistochemistry was performed by two reviewers in a blinded fashion. All analyses were performed in Stata Version 18 (StataCorp, College Station, TX). All methods are reported in accordance with ARRIVE guidelines.
Ex-Vivo Lung Perfusion Set-Up and Capabilities. (A) Ex-Vivo Lung Perfusion (EVLP) constructed with the capability for multiple pumps, carbon-dioxide saturation for challenge gas acquisition, built in mount for camera, bronchoscopy mount and pressure/flow monitoring on all lines. Utilizes an open drainage system. Lung mount is mobile magnetic resonance imaging compatible. (B) Demonstration of fluoroscopy incorporation, utilizing the mobile lung mount to obtain impeded images from all angles. (C) Set-up incorporating laser speckle contrast imaging platform to obtain real-time perfusion imaging.
Modifications to Perfuse Bronchial Arteries. (A) Schematic of EVLP modified to include additional antegrade perfusion to a cannula placed in the aorta, procured with the lungs to preserve the bronchial circulation. (B) View of lungs procured to include intact bronchial circulation with aorta procured along with lungs. Esophagus is taken as well and then dissected off on the back table. (C) The modified set-up fits within traditional EVLP specifications. PA: Pulmonary Artery; PV: Pulmonary Vein.
Fluoroscopic angiography performed while on EVLP using iodinated contrast perfused into the (A) PA only, and (B) into the aortic cannula directed towards the bronchial arteries (highlighted with yellow arrows).
Representative Region-Specific LASCA Images. Top Row: Trachea with Perfusion Mapping Overlaid (left) : PA only perfusion, (right): dual circulation. Bottom Row: Parenchyma with Perfusion Mapping Overlaid (left): PA only perfusion, (right): dual circulation Graph: Quantification of perfusion differences in key regions. *p < 0.05. N = 6 pigs represented in each group.
TUNEL Staining. (A) Representative H&E and TUNEL staining from n = 3 control pigs that received only PA perfusion (Top row), versus n = 3 pigs that received dual circulation (Bottom Row). (B) Comparison of peri-bronchial TUNEL expression after 6 h of EVLP, n = 3 each arm. Blue: DAPI, Red: TUNEL ***: p < 0.001. Scale bar = 1 mm.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Valapour, M. et al. OPTN/SRTR 2021 annual data report: Lung. Am. J. Transplant. 23 (2), S379–S379. https://doi.org/10.1016/J.AJT.2023.02.009 (2023).
Rana, A. et al. Survival benefit of solid-organ transplant in the united States. JAMA Surg. 150 (3), 252–259. https://doi.org/10.1001/jamasurg.2014.2038 (2015).
Pasnupneti, S. & Nicolls, M. R. Airway hypoxia in lung transplantation. Curr Opin. Physiol Feb. 7, 21–26. https://doi.org/10.1016/j.cophys.2018.12.002 (2019).
Jiang, X. et al. Promotion of airway anastomotic microvascular regeneration and alleviation of airway ischemia by deferoxamine nanoparticles. Biomaterials 35 (2), 803–813. https://doi.org/10.1016/j.biomaterials.2013.09.092 (2014).
Nicolls, M. R. & Zamora, M. R. Bronchial blood supply after lung transplantation without bronchial artery revascularization. Curr. Opin. Organ. Transplant. 15 (5), 563–567. https://doi.org/10.1097/MOT.0b013e32833deca9 (2010).
Tane, S., Noda, K., Toyoda, Y. & Shigemura, N. Bronchial-arterial-circulation-sparing Lung Preservation: A New Organ Protection Approach for Lung Transplantation. Transplantation 104 (3), 490–499. https://doi.org/10.1097/TP.0000000000002984 (2020).
Shigemura, N., Tane, S. & Noda, K. The bronchial arterial circulation in lung transplantation: bedside to bench to bedside, and beyond. Transplantation August. 102 (8). https://doi.org/10.1097/TP.0000000000002180 (2018).
Khan, M. A. & Nicolls, M. R. Complement-mediated microvascular injury leads to chronic rejection. Adv. Exp. Med. Biol. 735, 233–246. https://doi.org/10.1007/978-1-4614-4118-2_16 (2013).
Divithotawela, C. et al. Long-term outcomes of lung transplant with ex vivo lung perfusion. JAMA Surg. 154 (12), 1143–1150. https://doi.org/10.1001/JAMASURG.2019.4079 (2019).
Cypel, M. et al. NOrmothermic ex vivo lung perfusion in clinical lung transplantation. N. Engl. J. Med. 364 (15), 1431–1440. https://doi.org/10.1056/NEJMoa1014597 (2011).
Cypel, M., Yeung, J. C. & Keshavjee, S. Novel approaches to expanding the lung donor pool: donation after cardiac death and ex vivo conditioning. Clin. Chest. Med. 32 (2), 233–244. https://doi.org/10.1016/j.ccm.2011.02.003 (2011).
Unai, S., Yun, J. & Pettersson, G. B. Bronchial artery revascularization: Surgical technique. Operative Techniques Thorac. Cardiovasc. Surg. 28 (2), 139–153. https://doi.org/10.1053/j.optechstcvs.2022.11.004 (2023).
Iyer, V. N. Ex vivo lung perfusion in lung transplantation. The New England journal of medicine. ;365(1):91 – 2; author reply 92. (2011). https://doi.org/10.1056/NEJMc1105429
Crespo, M. M. et al. ISHLT consensus statement on adult and pediatric airway complications after lung transplantation: definitions, grading system, and therapeutics. J. Heart Lung Transplant. 37 (5), 548–563. https://doi.org/10.1016/j.healun.2018.01.1309 (2018).
Crespo, M. M. Airway complications in lung transplantation. J. Thorac. Disease. (11). /11;13 (2021).
Mendogni, P. et al. Usefulness of autofluorescence bronchoscopy in early diagnosis of airway complications after lung transplantation. Sci. Rep. 10 (1). https://doi.org/10.1038/s41598-020-79442-4 (2020).
Herrera, J. M. et al. Airway complications after lung transplantation: Treatment and long-term outcome. Ann. Thorac. Surg. 71 (3), 989–993. https://doi.org/10.1016/s0003-4975(00)02127-5 (2001).
Higgins, R. et al. Airway stenoses after lung transplantation: management with expanding metal stents. J Heart Lung Transplant Sep-Oct. 13 (5), 774–778 (1994).
Thistlethwaite, P. A. et al. Airway stenoses after lung transplantation: incidence, management, and outcome. J. Thorac. Cardiovasc. Surg. 136 (6), 1569–1575. https://doi.org/10.1016/j.jtcvs.2008.08.021 (2008).
Muñoz-Fos, A. et al. Airway complications after lung Transplantation-A contemporary series of 400 bronchial anastomoses from a single center. J Clin. Med Apr. 23 (9). https://doi.org/10.3390/jcm12093061 (2023).
AN, B. & JM, T. M. Microvascular destruction identifies murine allografts that cannot be rescued from airway fibrosis - PubMed. J. Clin. Investig. 117 (12). https://doi.org/10.1172/JCI32311 (2007).
Jiang, X. et al. Tie2-dependent VHL knockdown promotes airway microvascular regeneration and attenuates invasive growth of Aspergillus fumigatus. J. Mol. Med. 91 (9), 1081–1093. https://doi.org/10.1007/S00109-013-1063-8 (2013).
Lama, V. N. et al. Models of lung transplant research: A consensus statement from the National heart, lung, and blood Institute workshop. JCI Insight. https://doi.org/10.1172/jci.insight.93121 (2017).
Luckraz, H. et al. Microvascular changes in small airways predispose to obliterative bronchiolitis after lung transplantation. J. Heart Lung Transplant. 23 (5), 527–531. https://doi.org/10.1016/j.healun.2003.07.003 (2004).
Luckraz, H. et al. Is obliterative bronchiolitis in lung transplantation associated with microvascular damage to small airways?? Ann. Thorac. Surg. 82 (4), 1212–1218. https://doi.org/10.1016/j.athoracsur.2006.03.070 (2006).
Tanaka, Y. et al. A novel dual ex vivo lung perfusion technique improves immediate outcomes in an experimental model of lung transplantation. Am. J. Transplant. (5). /05/01;15 (2015).
Crespo, M. M. Airway complications in lung transplantation. J Thorac. Dis Nov. 13 (11), 6717–6724. https://doi.org/10.21037/jtd-20-2696 (2021).
Yun, J. J., Unai, S. & Pettersson, G. Lung transplant with bronchial arterial revascularization: review of surgical technique and clinical outcomes. J. Thorac. Disease. 11 (S14), S1821–S1828. https://doi.org/10.21037/jtd.2019.09.09 (2019).
Krishnan, A. et al. Donor profile changes with the lung composite allocation score. J. Heart Lung Transplantation. 43 (4), S420. https://doi.org/10.1016/j.healun.2024.02.1334 (2024).
Li, S. S. et al. Going the distance: geographic effects of the lung transplant composite allocation score. JHLT Open. 6, 100128. https://doi.org/10.1016/j.jhlto.2024.100128 (2024).
C, A. W. & AA, H. S. S. A simplified preservation method for lungs donated after cardiac death - PubMed. J. Heart Lung Transplantation: Official Publication Int. Soc. Heart Transplantation. 33 (5). https://doi.org/10.1016/j.healun.2014.01.854 (2014).
Buttar, S. N. et al. Porcine lungs perfused with three different flows using the 8-h open-atrium cellular ex vivo lung perfusion technique. Front. Bioeng. Biotechnol. 12doi https://doi.org/10.3389/fbioe.2024.1357182 (2024).
Li, D-Y. et al. Transmissive-detected laser speckle contrast imaging for blood flow monitoring in thick tissue: from Monte Carlo simulation to experimental demonstration. Light: Science & Applications 2021 (2021). https://doi.org/10.1038/s41377-021-00682-8
Laser Speckle Contrast Imaging. theory, instrumentation and applications - PubMed. IEEE Rev. Biomed. Eng. 6doi https://doi.org/10.1109/RBME.2013.2243140 (2013).
Funding
and Disclosures: AK is funded by NHLBI 1T32HL098049, the remainder of this work was funded by institutional funding. AK, MF, SE, and BAG have filed a patent application for material appearing in this manuscript.
This work was originally presented at the International Society of Heart and Lung Transplantation Meeting in Prague, Czech Republic on April 10th, 2024.
Author information
Authors and Affiliations
Contributions
AK and BAG wrote the main manuscript text. AK, MF, SE, EH, AG, AYC, CR, and BAG procured and handled the tissue used in this study. AK, MF, JS, JPM, and BC ran the Ex Vivo Lung Perfusion platform. AK, JWM, YJW, and BAG designed the experimental conditions. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Krishnan, A., Fawad, M., Elde, S. et al. Preservation of bronchial artery circulation on ex-vivo lung perfusion. Sci Rep 15, 23354 (2025). https://doi.org/10.1038/s41598-025-06174-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-06174-8