Abstract
This study aimed to assess the association between liver fibrosis scores (LFSs) and cardiovascular disease (CVD) in older adults, investigating the mediating effect of total bilirubin (TBil) between LFSs and CVD. Using data from the National Health and Nutrition Examination Survey (NHANES) between 1999 and 2018, this cross-sectional study analyzed Americans aged 60 and older. Four LFSs (Forns Score, FIB-4, NAFLD fibrosis score, and BARD Score) were utilized. Weighted logistic regression and smoothed curve fitting explored the relationship between LFSs and CVD. Subgroup analysis assessed this relationship among different populations. Mediation effect analysis examined the role of TBil. This study included 18,902 subjects, with a mean age of 69.64 ± 7.00 years and CVD prevalence of 22.33%. The fully adjusted weighted logistic regression models showed that FS, NFS, FIB-4 and BARD Score were positively associated with CVD with ORs of 1.50 (95% CI:1.37–1.64), 1.50 (95% CI:1.30–1.73), 1.39 (95% CI:1.18–1.64), and 1.54 (95% CI:1.29–1.85). Smoothed curve fitting indicated a nonlinear correlation between LFSs and CVD. Subgroup analysis indicated that the positive associations between FS, NFS, FIB-4, and CVD were consistent with respect to hepatitis B surface antigen, and the positive correlation between FIB-4, BARD Score, and CVD was coherent for the prevalence of hypertension. Mediation analysis revealed TBil mediated 12.61% of the association between FIB-4 and CVD. There exists a positive association between LFSs and CVD in older adults, and TBil mediates the relationship between FIB-4, and CVD. LFSs may serve as a tool for identifying older adults at high risk for cardiovascular disease.
Similar content being viewed by others
Background
CVD is the leading cause of death and disability worldwide, accounting for 50% of global chronic non-communicable disease-related deaths1. Globally, the burden of CVD continues to increase, with the number of CVD cases increasing from 271 million in 1990 to 523 million in 2019, according to the Global Burden of Disease (GBD) 2019 study2. Therefore, early detection and treatment of CVD is critical for patient survival and long-term health. Liver fibrosis is the most prominent prognostic factor associated with the development of cirrhosis, liver failure, NAFLD liver-related complications, and other chronic liver diseases3,4. Previous studies have reported that liver fibrosis was associated with CVD risk, and evaluating liver fibrosis can help identify people at high risk for CVD5,6.
Non-invasive LFSs consisting of routine clinical data and laboratory variables have traditionally been frequently performed to assess the presence and severity of liver injury and fibrosis due to the advantages of applicability, wide availability, and low cost7,8,9. Some of the commonly used non-invasive liver fibrosis scoring methods are FS, FIB-4, NFS, BARD Score, etc. Prior studies have reported that LFSs have potential predictive value for the risk of developing CVD in different populations10,11,12,13. The promotion of CVD pathogenesis by liver fibrosis can be explained by increased inflammation, abnormalities in lipid metabolism, coagulation disorders, and insulin resistance14,15,16. Also, the components of LFSs reflect systemic insulin resistance, obesity, and metabolic syndrome17. It has been demonstrated that mildly elevated concentrations of TBil, an indicator of liver function, is associated with a reduced risk of CVD18,19,20,21. Since CVD is highly prevalent in the elderly population, the aim of this study was to further explore the relationship between LFSs and CVD in the elderly and the mediating effect of TBil between LFSs and CVD using NHANES data. Early identification of potential CVD in the elderly population may play an essential role in improving prognosis and therapeutic management.
Methods
Data source
Data were derived from the 1999–2018 National Health and Nutrition Examination Survey (NHANES). NHANES is a nationally representative cross-sectional survey of all noninstitutionalized populations in the United States. It was designed to assess the health and nutritional status of the U.S. population through the collection of demographic, dietary, physical examination, and laboratory data, as well as to determine the prevalence of major diseases and associated risk factors. The NHANES survey has been conducted annually since 1999, with data released every two years. These data are available from the Centers for Disease Control and Prevention (CDC) Website (http://www.cdc.gov/nchs/nhanes.htm). The study protocol was approved by the National Center for Health Statistics (NCHS) Institutional Review Board, and all participants signed written informed consent form at the time of recruitment. In terms of ethics, the study followed the relevant provisions of the Helsinki Declaration to ensure participants’ privacy and data security. During data use, the principle of confidentiality was strictly adhered to, no personally identifiable information was disclosed, and the research process complied with ethical standards and legal requirements.
Study population
There is no international consensus definition for elderly. The World Health Organization defines an elderly person as anyone over the age of 6022,23. Meanwhile, the peak at age 60 showed molecular changes related to carbohydrates, caffeine metabolism, cardiovascular disease, skin, muscle, immune regulation, and kidney function. At the molecular level, the expression of genes related to oxidative stress, messenger RNA stability, and autophagy also changed significantly after age 60. These changes are closely related to the occurrence of a variety of geriatric diseases, such as cardiovascular disease, diabetes, and kidney function decline. Furthermore, there are also many articles that have utilized the data from NHANES and have defined people over 60 years old as the elderly population24. Based on the above references, this study also selected people over 60 years old as the elderly25,26.
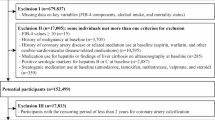
The study included 111,072 participants extracted from the NHANES database from 1999 to 2018. We excluded patients who were undiagnosed, unable to calculate the liver fibrosis index, with missing covariates, aged under 60 years, and fasting weights. A total of 189,02 participants were ultimately included in the final analysis of this study. A detailed description of the participant flow was shown in Fig. 1.
The flow chart of our study.
Liver fibrosis scores
We selected FS, FIB-4, NFS, and the BARD Score as representative non-invasive liver fibrosis scores based on their widespread use and clinical relevance. These scores incorporate routinely available clinical variables such as age, BMI, liver enzymes, and platelet count, making them feasible to apply in population-based settings like NHANES. Additionally, prior studies have demonstrated their validity in predicting hepatic and extra-hepatic outcomes, including metabolic and cardiovascular risks. Although some of these scores were originally developed in middle-aged or broader adult populations, they have been applied and evaluated in older populations in recent literature27,28,29,30 supporting their relevance to the current study cohort.
We adopted four methods to calculate the liver fibrosis score separately: FS (Forns Score), FIB-4, NFS (NAFLD Fibrosis Score), and BARD Score (BMI, AST/ALT Ratio and Diabetes).
FS: 7.811-3.131ln (platelet count) + 0.781ln (GGT) + 3.467ln (Age)-0.014 (cholesterol)31.
FIB-4: Age[years]×AST[U/L])/ ((PLT [109/L]) × (ALT [U/L])1/232.
NFS:-1.675 + 0.037×Age(years) + 0.094×BMI(kg/m2) + 1.13×IFG/diabetes(yes = 1, no = 0) + 0.99×AST/ALT ratio-0.013×PLT(×109/L)-0.66×albumin(g/dL)33.
BARD Score: BMI ≥ 28 kg/m2(1 point) + AST/ALT ratio ≥ 0.8(2 points) + presence of diabetes(1 point)34.
Cardiovascular disease
Participants were asked if they had ever been diagnosed with congestive heart failure, coronary heart disease, angina, heart attack or stroke by a doctor or other health professional. An affirmative answer to any of these conditions was defined as having prevalent CVD35.
Total bilirubin measurement
The LX20 and dx800 measured the concentration of total bilirubin in serum or plasma using a time-endpoint diazotization method (Jendrassik-Grof). In this reaction, bilirubin reacted with a diazo reagent in the presence of caffeine, benzoate and acetate as accelerators to form azobilirubin. The system monitored the change in absorbance at 520 nm at regular intervals. The change in absorbance was proportional to the concentration of total bilirubin in the sample. Total bilirubin measurements were used in the diagnosis and treatment of liver and hematologic disorders.
Assessment of covariates
We incorporated sociodemographic variables, health-related behaviors, and some confounders as covariates. Sociodemographic variables included sex (male and female), age (continuous variable), race (Mexican American, non-Hispanic Black, non-Hispanic White, other race), household composition (living alone and living with others), education level (less than high school, high school graduation or equivalent, some college education or college graduation and above), poverty-to-income ratio (PIR) for households, and BMI (continuous variables). Health-related behaviors consisted of smoking and drinking status, with smoking stratified into three groups (former smokers, never smokers, and current smokers) and drinking being categorized into two groups (yes, no) based on drinking at least 12 glasses of wine per year. Biochemical detection indicators included alkaline phosphatase, triglycerides, total bilirubin (TBil), all of which were continuous variables. There are two classifications for whether your doctor said you have hypertension (yes, no), and hepatitis B surface antigen (positive, negative).
Statistical analysis
To obtain nationally representative estimates, all analyses were weighted to account for the complex sampling design, including the use of 4-year and 2-year NHANES fasting weights36. In the baseline analysis, continuous variables were expressed as mean (standard deviation), and categorical variables were expressed as frequency (percentage). The t-test, Wilcoxon rank sum test, and Pearson’s chi-square test were employed to compare differences in variables for the presence or absence of CVD. Directed acyclic graphs (DAGs) to determine the covariates to include in our multivariate models. This approach helps to simplify the number of covariates and identify more relevant covariates, and improve the overall robustness of our model. We adopted DAGs to guide our covariate selection process. The selection criteria for included covariates were determined by a comprehensive approach that considered the results of univariate logistic regression analysis, multicollinearity test results, relevant literature reports, and the effect of covariates on the association between exposure and outcome, exceeding a threshold of 10%. The following covariates were selected: sex, race, household composition, education, PIR, hypertension, Alkaline phosphotase, Triglycerides, eGFR. After using directed acyclic graphs (DAGs), the final model considered the following covariates: Sex, PIR, Household composition, hypertension, Alkaline phosphotase, Triglycerides. As shown in the figure S1 and Table S19.
In order to evaluate the relationship between LFSs and CVD more effectively, we analyzed LFSs as continuous and categorical variables, respectively, and established three weighted logistic regression models to investigate the correlation between LFSs and CVD. Model 1 did not adjust for any factors. Model 2 adjusted for sex, PIR, household composition. Model 3 further adjusted for hypertension, alkaline phosphatase, triglycerides, Smoothed curve fitting examined nonlinear relationships. Given the variation in descriptive characteristics observed in previous studies, possible confounding covariates (including sex, household composition, alcohol consumption, hypertension, Diabetes, Hepatitis B surface antigen, Severe obesity37, Renal insufficiency(defined as estimated GFR < 60 mL/min/1.73 m²38)) were estimated by subgroup analysis, and interaction tests were performed to assess whether the effect of the intervention was affected by a subgroup factors. Bonferroni method was used to correct for multiple testing. In addition, a sensitivity analysis was performed on people without liver problems to assess the potential impact of other liver problems and the stability of the results in the link between LFSs and CVD. Based on the R “cutoff " software package, we determined the optimal cut-off value for LFSs in predicting prevalent CVD. To evaluate the robustness of our findings to potential unmeasured confounding, we conducted a sensitivity analysis using the E-value method. Also, we have now performed propensity score matching (PSM) to adjust for potential confounding variables and to better estimate the association between Liver Fibrosis Scores and Cardiovascular Disease. Specifically, we used a 1:1 nearest-neighbor matching algorithm without replacement, based on key covariates including sex, age, ace, household composition, education level, poverty-to-income ratio, BMI, smoking, drinking, hypertension, diabetes, cholesterol. After matching, we reanalyzed the primary outcomes, thereby reinforcing the robustness of our conclusions. Finally, mediation effect analysis was applied to explore the indirect effect of LFSs on CVD through TBil. Briefly, LFSs acted as a predictor variable (X), TBil as a mediator variable (M), and CVD as an outcome variable (Y). All variables in the model with VIF > 5 deleted. A significance level of 0.05 (two-tailed) was applied. All statistical analyses were performed using R version 4.2.1, the mediation package was used for mediation analysis (R Foundation for Statistical Computing, Vienna, Austria; http://www.r-project.org).
Results
Baseline characters
This study comprised18,902 subjects, of which 50% were males and 50% were females. The mean age of the participants was 69.64 ± 7.00 years. The prevalence of CVD was 22.33%. Clinical characteristics associated with CVD included sex, age, race, household composition, education level, PIR, BMI, smoking, hypertension, diabetes, platelet count, ALT, AST, GGT, alkaline phosphatase, cholesterol, triglycerides, total bilirubin, eGFR, FS, NFS, FIB-4, and BARD Score (P < 0.001). Compared to the group without CVD, patients with CVD tended to be older, male, non-Hispanic White, living with others, have less than a high school education, lower PIR, higher BMI, be former smokers, hypertensive, non-diabetic, with higher ALT, AST, GGT, alkaline phosphatase, triglycerides, and total bilirubin. Platelet counts, cholesterol, and eGFR were lower. Moreover, patients with CVD had higher LFSs, including FS, NFS, FIB-4, and BARD Score (Table 1 and Table S1).
Association between LFSs and CVD
Weighted logistic regression revealed a positive correlation between LFSs and CVD. When FS, NFS, FIB-4, and BARD Score were continuous variables, the ORs were 1.50 (95% CI:1.37–1.64), 1.50 (95% CI:1.30–1.73), 1.39 (95% CI:1.18–1.64), and 1.54 (95% CI:1.29–1.85), respectively, in Model 3. This trend continued when FS, NFS, FIB-4, and BARD Score were categorical variables, with ORs of 4.63 (95% CI:3.04–7.04), 2.74 (95% CI: 2.45–5.71), 2.60 (95% CI: 1.71–3.98), 2.19 (95% CI: 1.53–3.13) (Tables 2, 3, 4 and 5). Smooth curve fitting confirmed the nonlinear positive correlation between FS, NFS, FIB-4, BARD Score, and CVD (P < 0.001) (Fig. 2).
The nonlinear association between the Liver Fibrosis Scores and Cardiovascular Disease. Solid red line represents the smooth curve fit between variables. Dotted red line represents the 95% confidence interval for the fit. The graphs in (A–D) represented the Forns Score (FS), FIB-4, NAFLD fibrosis score (NFS), and BARD Score, respectively. Legend of the histogram: Displays the distribution of liver fibrosis index.
Subgroup analysis
Subgroup analysis and interaction tests demonstrated that the positive associations between FS, NFS and CVD were consistent with respect to hepatitis B surface antigen (P for interaction > 0.05). The ORs of FS, Q2: 1.46 (95% CI: 1.3–1.64), Q3: 2.31 (95% CI: 2.07–2.58), Q4: 4.21 (95% CI: 3.79–4.68), The ORs of NFS: Q2: 1.22 (95% CI: 1.09–1.37), Q3: 2.28 (95% CI: 2.05–2.54), Q4: 3.79 (95% CI: 3.42–4.21). The positive correlation between FIB-4 and CVD was coherent for the prevalence of hypertension (P for interaction > 0.05) (P for interaction > 0.05), the ORs of FIB-4, Q2: 1.08 (95% CI: 0.95–1.22), Q3: 1.76 (95% CI: 1.56–1.98), Q4: 2.39 (95% CI: 2.13–2.68). And the positive relationship between BARD and CVD was in agreement for the prevalence of hypertension (P for interaction > 0.05), the ORs of FIB-4, Q2: 1.23 (95% CI: 1.08–1.4), Q3: 1.37 (95% CI: 1.21–1.55), Q4: 2.22 (95% CI: 1.97–2.5). Differences in gender, household composition, BMI, Renal insufficiency and drinking status were statistically significant (interaction P < 0.001). CVD was more prevalent in males, living with others, drinking alcohol, hypertensive, non-diabetic, and hepatitis B surface antigen-negative individuals (Tables S2-S5).
Sensitivity analysis
We conducted supplementary sensitivity analysis to assess the potential influence of liver disease as a confounding factor in the relationship between LFSs and CVD. As shown in Tables S6-S9, the association between LFSs and CVD remained consistent after excluding patients with liver problems. Using the R “cutoff " package, we identified optimal cutoffs for predicting prevalent CVD: FIB-4 ≥ 1.685 (AUC = 0.689), NFS ≥–0.800 (AUC = 0.687), FS ≥ 4.866 (AUC = 0.693), and BARD ≥ 1.152 (AUC = 0.675), indicating moderate discriminative performance (Tables S10-13 and Figure S2). Moreover, the E values were 8.72, 4.92, 4.63, and 3.80, indicating that to explain the observed ORs of 4.63, 2.74, 2.60, and 2.19, there would need to be unmeasured confounders associated with both LFSs and CVD with ORs of at least 8.72, 4.92, 4.63, and 3.80 for both (Table S18). This suggests that our findings are relatively robust to unmeasured confounding. Furthermore, after matching, we reanalyzed the primary outcomes, and the results remained largely consistent with our initial findings, thereby reinforcing the robustness of our conclusions (Table S14-17).
Mediating role of total bilirubin between LFSs and CVD
Figure 3 summarizes the potential mediating role of TBil reduction between LFSs and CVD. In the mediating effect models between FIB-4, and CVD, the proportion of the mediating effect of TBil was 12.61%.
Path diagram of the mediation analysis of TBil on the relationship between between LFSs and CVD. The graphs in (A–D) represented the Forns Score (FS), NAFLD fibrosis score (NFS), FIB-4, and BARD Score, respectively.
Discussion
In this nationally representative cross-sectional study involving 18,902 individuals aged 60 years or older, we observed a positive association between FS, NFS, FIB-4, BARD Score and CVD, even after stratifying FS, NFS, FIB-4, and BARD Score into quartiles. This implies that the association between LFSs and CVD actually represents a higher prevalence of CVD among participants with high LFSs. Sensitivity Analysis Using E-value: this suggests that our findings are relatively robust to unmeasured confounding. In addition, using propensity matching scores, we found that the results were generally consistent with previous results, thus strengthening the stability of our conclusions. Mediation effect analysis suggested that TBil partially mediated the correlation between FIB-4 and CVD. These findings indicated that elevated LFSs may be a risk factor for CVD, emphasizing the value of LFSs in predicting CVD.
Our study found a significant association between LFSs and CVD, and prior studies have demonstrated the diagnostic and predictive value of LFSs for CVD and diabetes in different populations11,39,40,41,42. FS, NFS, FIB-4, and BARD Score were independent predictors of major adverse cardiac events (MAEs), cardiovascular, and all-cause mortality12. These are in accordance with our findings. Weighted logistic regression and smoothed curve fitting confirmed that FS, NFS, FIB-4, and BARD Score were nonlinearly and positively associated with CVD in the U.S. elderly population aged 60 years or older, and that individuals with high LFSs were more prone to CVD than those with low LFSs. There are several potential mechanisms that could explain the correlation between liver fibrosis and CVD. Liver fibrosis is associated with an increased inflammatory state and increased circulating inflammatory markers, such as interleukin-6 and tumor necrosis factor-α, which further drive atherosclerosis and cardiovascular outcomes (CVOs)10. The liver is the central regulator of lipid metabolism, and liver fibrosis causes lipid disturbances, such as elevated TG and LDL-C levels, resulting in more CVOs43. Liver fibrosis facilitates the formation of blood clots by increasing clotting factors and the risk of ischemic cardiovascular disease44. Insulin resistance is prevalent in patients with liver fibrosis, which can contribute to the development of CVD by causing imbalances in glucose and lipid metabolism45. These findings support the use of LFSs as an indicator of CVD risk and contribute to the prevention and management of CVD in older adults, while our findings provide new perspectives for screening populations at high risk for CVD.
The mediation analysis revealed that total bilirubin accounted for 12.61% of the association between liver fibrosis scores and CVD. While statistically significant, this mediated proportion is modest, suggesting that TBil may serve as a partial pathway linking hepatic dysfunction to cardiovascular risk. This finding highlights the multifactorial nature of the liver-heart axis46 and suggests that other mechanisms—such as systemic inflammation, insulin resistance, or oxidative stress—may also play important roles47. Indicators of liver function, especially TBil, are strongly related to CVD48,49. TBil, composed of both unconjugated and conjugated forms, is a potent indicator of the severity of impaired liver function. Elevated bilirubin levels above the reference range usually reflect disease or hereditary disorders21. Recent evidence supports that TBil is a robust endogenous antioxidant in the normal physiological range, with endocrine properties such as anti-inflammatory, immunomodulatory, anti-thrombotic, and hypolipidemic properties50,51 which may contribute to the protective effects against oxidative stress disorders, including diabetes, stroke, heart failure, CVD, and others52,53,54,55,56. Some research has shown that slightly elevated levels of physiological concentrations of TBil are associated with a reduced risk of CVD20. Thus, LFSs may influence the occurrence of CVD in older adults through TBil. The present study validated these results by confirming the findings through mediation effect analysis. In the mediation effect analysis of LFSs and CVD, TBil partially mediated the association between FIB-4, and CVD. Our findings suggest that the protective effect of elevated TBil can be utilized to minimize the risk of CVD and enhance a validated intervention against CVD.
Although our current study is observational and lacks molecular-level data, several plausible biological mechanisms may underlie the association between liver fibrosis, total bilirubin (TBil), and cardiovascular disease (CVD). One hypothesis is that elevated TBil, within a certain range, may serve as a marker of increased oxidative stress or impaired hepatic clearance, both of which can contribute to vascular endothelial dysfunction—a known precursor of atherosclerosis57. Additionally, liver fibrosis is often accompanied by chronic low-grade inflammation and altered lipid metabolism, including dysregulation of triglycerides and LDL cholesterol, which are established risk factors for CVD58. Fibrotic liver tissue may also produce pro-inflammatory cytokines (e.g., IL-6, TNF-α), further promoting systemic inflammation and vascular injury59. Future studies are needed to investigate these pathways more directly. Incorporating inflammatory biomarkers (such as CRP, IL-6), lipidomic profiling, and endothelial function tests into prospective cohort designs could provide more definitive insights. Experimental studies using animal models or in vitro systems could also help delineate the cellular and molecular mechanisms linking hepatic fibrosis to cardiovascular outcomes. These directions will be critical to translating our epidemiological findings into clinical and therapeutic implications.
Our study has several limitations. Firstly, the cross-sectional nature of the NHANES data precludes clear causal inferences. For this reason, the observed associations and mediation effects should be interpreted with caution and do not imply temporal order or causality. Future longitudinal or intervention studies are needed to confirm and extend our findings. We acknowledge that the operational definition of CVD in NHANES is based on participants’ self-reports of prior physician diagnoses, which may not always reflect clinical verification through imaging, laboratory results, or medical records. We note that this may have led to misclassification of disease status, potentially attenuating the observed associations. future studies incorporate clinically verified diagnostic criteria—such as imaging findings, laboratory biomarkers, or electronic health records—whenever possible, to improve diagnostic accuracy and reduce bias.
Secondly, our study is based on data derived from a U.S. population, which may limit the generalizability of our findings to other regions. Elderly populations in different countries may vary in genetic predisposition, chronic disease patterns, healthcare access, and lifestyle factors. Therefore, future studies involving more diverse, multi-ethnic, and international cohorts are needed to validate the observed associations and strengthen the external applicability of our findings.
Thirdly, the specific mechanism leading to elevated LFSs is uncertain because liver biopsies were not performed to assess liver pathology. However, liver biopsies cannot be performed on the general population due to their invasive nature60. In addition, LFSs include nonspecific indicators such as age, BMI, platelet count, cholesterol, and diabetes, therefore, the positive association between LFSs and CVD is not particularly surprising. Finally, despite adjusting for many potential confounders in the weighted logistic regression analysis, there may still be residual confounding effects. And although the mediation proportion for the other three scores appears small, it is valuable to mention these findings as they may indicate distinct biological pathways captured by these three scores.
Conclusion
Cross-sectional study finds correlation between liver fibrosis index and CVD. The findings demonstrate that FS, NFS, FIB-4, and BARD Score may be associated with an increased risk of CVD in older adults. TBil mediates the association between FIB-4 and CVD. These LFSs may assist in identifying older adults at high risk of CVD, provide new insights into secondary prevention of CVD, and suggest that the protective effect of elevated TBil can be exploited to lessen the risk of CVD in older adults.
The findings indicate an association between FS, NFS, FIB-4, and BARD Score with an increased risk of CVD in older adults. Furthermore, TBil appears to mediate the association between FIB-4 and CVD. These LFSs may have the potential to aid in the identification of older adults at high risk of CVD, providing valuable insights for secondary prevention strategies. Additionally, the observed protective effect of elevated TBil suggests the possibility of utilizing this factor to mitigate the risk of CVD in older adults.
Data availability
Publicly available datasets were analyzed in this study. These data can be found here: https://wwwn.cdc.gov/nchs/nhanes/default.aspx.
References
Jagannathan, R., Patel, S. A., Ali, M. K. & Narayan, K. M. V. Global Updates on Cardiovascular Disease Mortality Trends and Attribution of Traditional Risk Factors. J. Current diabetes reports. 19(7), 44 (2019).
Roth, G. A. et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. J Journal Am. Coll. Cardiology. 76 (25), 2982–3021 (2020).
Dulai, P. S. et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. J Hepatology (Baltimore Md). 65 (5), 1557–1565 (2017).
Yasui, Y. et al. Elastin Fiber accumulation in liver correlates with the development of hepatocellular carcinoma. J PloS One. 11 (4), e0154558 (2016).
Tamaki, N. et al. Liver fibrosis and fatty liver as independent risk factors for cardiovascular disease. J Journal Gastroenterol. Hepatology. 36 (10), 2960–2966 (2021).
Baratta, F. et al. Nonalcoholic Fatty Liver Disease and Fibrosis Associated With Increased Risk of Cardiovascular Events in a Prospective Study. J. Clinical gastroenterology and hepatology: the official clinical practice journal of the American Gastroenterological Association. 18(10), 2324–2331.e2324 (2020).
Easl-Aleh Clinical Practice Guidelines. Non-invasive tests for evaluation of liver disease severity and prognosis. J Journal Hepatology. 63 (1), 237–264 (2015).
Lee, J. et al. Prognostic accuracy of FIB-4, NAFLD fibrosis score and APRI for NAFLD-related events: A systematic review. J Liver International: Official J. Int. Association Study Liver. 41 (2), 261–270 (2021).
Siddiqui, M. S. et al. Diagnostic accuracy of noninvasive fibrosis models to detect change in fibrosis stage. J Clinical Gastroenterol. Hepatology: Official Clin. Pract. J. Am. Gastroenterological Association. 17 (9), 1877–1885e1875 (2019).
Francque, S. M., van der Graaff, D. & Kwanten, W. J. Non-alcoholic fatty liver disease and cardiovascular risk: pathophysiological mechanisms and implications. J Journal Hepatology. 65 (2), 425–443 (2016).
Jin, J. L. et al. Liver fibrosis scores and coronary atherosclerosis: novel findings in patients with stable coronary artery disease. J Hepatology International. 15 (2), 413–423 (2021).
Cao, Y. X. et al. Impact of liver fibrosis score on prognosis in patients with previous myocardial infarction: A prospective cohort study. J Liver International: Official J. Int. Association Study Liver. 41 (6), 1294–1304 (2021).
Wen, W. et al. Linear relationship between hepatic steatosis index and major adverse cardiovascular events in hypertensive patients with obstructive sleep apnea: A Real-World cohort study from China. J Reviews Cardiovasc. Medicine. 24 (10), 280 (2023).
Clarembeau, F., Bale, G. & Lanthier, N. Cirrhosis and insulin resistance: current knowledge, pathophysiological mechanisms, complications and potential treatments. J. Clin. Sci. (London England: 1979). 134 (16), 2117–2135 (2020).
Ogresta, D. et al. Coagulation and endothelial dysfunction associated with NAFLD: current status and therapeutic implications. J Journal Clin. Translational Hepatology. 10 (2), 339–355 (2022).
Wilde, B. & Katsounas, A. Immune Dysfunction and Albumin-Related Immunity in Liver Cirrhosis. J. Mediators of inflammation. 7537649(2019). (2019).
Xie, T. et al. Clinical characteristics and construction of a predictive model for patients with sepsis related liver injury. J Clinica Chim. Acta; Int. J. Clin. Chemistry. 537, 80–86 (2022).
Suh, S. et al. Relationship between serum bilirubin levels and cardiovascular disease. J PloS One. 13 (2), e0193041 (2018).
Kimm, H., Yun, J. E., Jo, J. & Jee, S. H. Low serum bilirubin level as an independent predictor of stroke incidence: a prospective study in Korean men and women. J Stroke. 40 (11), 3422–3427 (2009).
Kunutsor, S. K., Bakker, S. J., Gansevoort, R. T., Chowdhury, R. & Dullaart, R. P. Circulating total bilirubin and risk of incident cardiovascular disease in the general population. J Arteriosclerosis Thromb. Vascular Biology. 35 (3), 716–724 (2015).
Nilsen, D. W. T. et al. Total Bilirubin Yields Prognostic Information Following a Myocardial Infarction in the Elderly. J. Antioxidants (Basel, Switzerland). 12(6), (2023).
Atramont, A. et al. Association of age with short-term and long-term mortality among patients discharged from intensive care units in France. J. JAMA Network Open. 2(5), e193215-e193215 (2019).
Guidet, B. et al. The trajectory of very old critically ill patients. J Intensive Care Medicine. 50 (2), 181–194 (2024).
Shen, X. et al. Nonlinear dynamics of multi-omics profiles during human aging. J Nature Aging 1–16 (2024).
Zheng, Z. et al. Masticatory function and cognition in older adults: A population-based study. J the J. Prosthet. Dentistry (2025).
Du, L. et al. Independent and joint role of inflammatory diet and physical activity with cognitive function in aging: evidence from a population-based survey. J General Hosp. Psychiatry. 91, 52–59 (2024).
Salles, N., Dussarat, P., Foucher, J., Villars, S. & de Lédinghen, V. Non-invasive evaluation of liver fibrosis by transient elastography and biochemical markers in elderly inpatients. J Gastroentérologie Clinique Et Biologique. 33 (2), 126–132 (2009).
Kurosaki, M. & Izumi, N. External validation of FIB-4: diagnostic accuracy is limited in elderly populations. J Hepatology. 47 (1), 352–352 (2008).
Hartleb, M., Barański, K., Zejda, J., Chudek, J. & Więcek, A. Non-alcoholic fatty liver and advanced fibrosis in the elderly: results from a community‐based Polish survey. J Liver International. 37 (11), 1706–1714 (2017).
Bai, Q. et al. Advanced prediction of heart failure risk in elderly diabetic and hypertensive patients using nine machine learning models and novel composite indices: insights from NHANES 2003–2016. J European J. Prev. Cardiology zwaf081 (2025).
Forns, X. et al. Identification of chronic hepatitis C patients without hepatic fibrosis by a simple predictive model. J Hepatology (Baltimore Md). 36 (4 Pt 1), 986–992 (2002).
Sterling, R. K. et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. J. Hepatology (Baltimore, Md). 43(6), 1317–1325 (2006).
Angulo, P. et al. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. J Hepatology (Baltimore Md). 45 (4), 846–854 (2007).
Harrison, S. A., Oliver, D., Arnold, H. L., Gogia, S. & Neuschwander-Tetri, B. A. Development and validation of a simple NAFLD clinical scoring system for identifying patients without advanced disease. J Gut. 57 (10), 1441–1447 (2008).
Shi, Z. et al. The circadian syndrome is a significant and stronger predictor for cardiovascular disease than the metabolic syndrome-The NHANES survey during 2005–2016. J Nutrients 14(24), (2022).
Johnson, C. L. et al. National health and nutrition examination survey: analytic guidelines, 1999–2010. J. Vital and health statistics Series 2, Data evaluation and methods research. (161), 1–24 (2013).
Organization, W. H. Obesity: preventing and managing the global epidemic: report of a WHO consultation. J., (2000).
Care, D. Chronic Kidney Disease and Risk Management. J. Medications for the Treatment of Diabetes Mellitus. 47(1), 376 (2024).
Colak, E. et al. Evaluation of the non-alcoholic fatty liver fibrosis score in predicting short-term outcomes and severe coronary artery disease in patients undergoing coronary computed tomography angiography. J Postepy W Kardiologii interwencyjnej = Adv. Interventional Cardiology. 20 (1), 45–52 (2024).
Chen, Q. et al. Association between liver fibrosis scores and the risk of mortality among patients with coronary artery disease. J Atherosclerosis. 299, 45–52 (2020).
Cai, X. et al. Hepatic Steatosis Index and the Risk of Type 2 Diabetes Mellitus in China: Insights from a General Population-Based Cohort Study. J. Disease markers. 3150380(2022). (2022).
Zhao, J. et al. J-Shaped Relationship Between Weight-Adjusted-Waist Index and Cardiovascular Disease Risk in Hypertensive Patients with Obstructive Sleep Apnea: A Cohort Study. J. Diabetes, metabolic syndrome and obesity: targets and therapy. 17, 2671–2681 (2024).
Alkhouri, N. et al. The inflamed liver and atherosclerosis: a link between histologic severity of nonalcoholic fatty liver disease and increased cardiovascular risk. J Digestive Dis. Sciences. 55 (9), 2644–2650 (2010).
Caldwell, S. H. et al. Coagulation disorders and hemostasis in liver disease: pathophysiology and critical assessment of current management. J Hepatology (Baltimore Md). 44 (4), 1039–1046 (2006).
Lonardo, A., Nascimbeni, F., Mantovani, A. & Targher, G. Hypertension, diabetes, atherosclerosis and nash: cause or consequence? J. J. Hepatol. 68 (2), 335–352 (2018).
Cao, Y. et al. Liver-heart cross-talk mediated by coagulation factor XI protects against heart failure. J Science. 377 (6613), 1399–1406 (2022).
Ndumele, C. E. et al. Cardiovascular-kidney-metabolic health: a presidential advisory from the American heart association. J Circulation. 148 (20), 1606–1635 (2023).
Yin, G. et al. Association between initial serum total bilirubin and clinical outcome in myocardial infarction with Non-Obstructive coronary arteries. J International J. Med. Sciences. 19 (6), 986–992 (2022).
Marconi, V. C. et al. rd. Bilirubin Is Inversely Associated With Cardiovascular Disease Among HIV-Positive and HIV-Negative Individuals in VACS (Veterans Aging Cohort Study). J. Journal of the American Heart Association. 7(10), (2018).
Bulmer, A. C. et al. Bilirubin acts as a multipotent guardian of cardiovascular integrity: more than just a radical Idea. J American J. Physiol. Heart Circ. Physiology. 315 (3), H429–h447 (2018).
Akboga, M. K. et al. Association of serum total bilirubin level with severity of coronary atherosclerosis is linked to systemic inflammation. J Atherosclerosis. 240 (1), 110–114 (2015).
Yang, M. et al. Association between serum total bilirubin levels and the risk of type 2 diabetes mellitus. J Diabetes Res. Clin. Practice. 152, 23–28 (2019).
Ouyang, Q. et al. Serum bilirubin levels are associated with poor functional outcomes in patients with acute ischemic stroke or transient ischemic attack. J BMC Neurology. 21 (1), 373 (2021).
Frikha, Z. et al. Relation of High Serum Bilirubin to Short-Term Mortality Following a Myocardial Infarction Complicated by Left Ventricular Systolic Dysfunction (from the High-Risk Myocardial Infarction Database Initiative). J. The American journal of cardiology. 121(9), 1015–1020 (2018).
Jain, V. et al. Serum bilirubin and coronary artery disease: intricate relationship, pathophysiology, and recent evidence. J Current Probl. Cardiology. 46 (3), 100431 (2021).
Cai, X. et al. Association of the trajectory of plasma aldosterone concentration with the risk of cardiovascular disease in patients with hypertension: a cohort study. J Sci Rep. 14 (1), 4906 (2024).
Luo, Y., Li, J., Zhang, J. & Xu, Y. Elevated bilirubin after acute ischemic stroke linked to the stroke severity. J International J. Dev. Neuroscience. 31 (7), 634–638 (2013).
Chen, W. et al. Destabilization of atherosclerotic plaque by bilirubin deficiency. J Circulation Research. 132 (7), 812–827 (2023).
Tanaka, M. & Miyajima, A. Liver regeneration and fibrosis after inflammation. J Inflammation Regeneration. 36, 19 (2016).
Easl-Easd-Easo. Clinical practice guidelines for the management of non-alcoholic fatty liver disease. J Diabetologia. 59 (6), 1121–1140 (2016).
Acknowledgements
We would like to thank all the participants of the NHANES study for their valuable contributions.
Funding
The authors did not receive specific funding for this study from any public, commercial, or non-profit sector funding agency.
Author information
Authors and Affiliations
Contributions
MD was primarily responsible for drafting this manuscript and contributed to its revision and refinement. YL and ST were involved in data cleansing processes. JT, JQ, WW and LF made insightful suggestions for revising the manuscript. SM conducted data analysis and assumes responsibility for the content of the article. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
The studies involving human participants were reviewed and approved by NCHS Ethics Review Board. Written informed consent to participate in this study was provided by the participants’ legal guardian/next of kin. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.


Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Du, M., Li, Y., Du, J. et al. Association between liver fibrosis scores and cardiovascular disease in elderly based on NHANES 1999–2018. Sci Rep 15, 23334 (2025). https://doi.org/10.1038/s41598-025-06871-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-06871-4