Abstract
The combination of immune checkpoint inhibitors (ICIs), such as ipilimumab and nivolumab, has revolutionized cancer treatment, particularly for advanced melanoma and other solid tumors. However, rare adverse events (AEs) associated with this combination are often underreported in clinical trials. This study aimed to investigate the safety profile of these drugs using the FDA Adverse Event Reporting System (FAERS) database. A retrospective analysis of data from 2015 to the first quarter of 2024 was conducted, focusing on AEs submitted by healthcare professionals. Disproportionality analysis, using algorithms like Reporting Odds Ratio (ROR) and Bayesian Confidence Propagation Neural Network (BCPNN), identified AE signals. A total of 19,462 reports related to the combination therapy were retrieved, spanning 20 System Organ Classes (SOCs), with endocrine and hepatobiliary disorders being most affected. Additionally, 125 unexpected significant AEs were identified, including thyrotoxic crisis, diabetes insipidus, immune-mediated optic neuritis, cystitis, eyelid ptosis, complete atrioventricular block, etc. The highest frequency of AEs occurred within the first 30 days of treatment (38.1%), with a decrease in incidence over time, as indicated by the Weibull Shape Parameter (WSP) test. These findings highlight the importance of continuous monitoring of ICIs in clinical practice.
Similar content being viewed by others
Introduction
The application of immune checkpoint inhibitors (ICIs) in cancer treatment has significantly increased, achieving substantial progress particularly in the management of advanced melanoma and other solid tumors such as renal cell carcinoma1,2. Ipilimumab and nivolumab, inhibitors of the CTLA-4 and PD-1 pathways respectively, activate the immune system to combat cancer cells. They received approval from the U.S. Food and Drug Administration (FDA) in 2011 and 2014, respectively, for the treatment of advanced melanoma3. The combination of these two drugs is believed to offer more significant clinical benefits compared to monotherapy, and thus was approved by the FDA as a combination therapy in 2015 4.
A growing body of clinical research supports the use of this combination in various malignancies such as non-small cell lung cancer5esophageal cancer6pleural mesothelioma7and recurrent or metastatic head and neck squamous cell carcinoma8demonstrating a remarkable advantage in overall survival (OS). However, the incidence and variety of immune-related adverse events (irAEs) are higher with combination therapy compared to monotherapy 9. For example, a Phase III clinical trial (CheckMate 067) for untreated advanced melanoma showed that the combination therapy group had superior progression-free survival (PFS) and OS compared to the monotherapy groups10. Additionally, 96% of patients in the combination therapy group experienced treatment-related adverse events (AEs), compared to 86% in both the nivolumab and ipilimumab monotherapy groups10. These AEs not only affect the patients’ quality of life but can also lead to treatment interruption or modification. Common AEs reported with the combination therapy include liver dysfunction, gastrointestinal issues (e.g., diarrhea, nausea, colitis), skin reactions (e.g., rash, pruritus), and endocrine abnormalities (e.g., hypothyroidism, adrenal insufficiency)2.
Due to limitations such as small sample sizes, short study durations, and stringent patient selection criteria, previous clinical trials, while identifying a higher incidence of AEs with combination therapy, have not been able to uncover new safety signals2,11. These trials often fail to reveal some potential rare AEs. For example, eosinophilic enteritis reported by Yang et al.12 and multiple mononeuritis reported by Sakai et al.13 are AEs previously unexplored. Additionally, the timing of these combination therapy-related AEs remains unclear.
This study retrospectively analyzes AEs related to the combined use of ipilimumab and nivolumab from 2015 to the first quarter of 2024, using data from the FDA Adverse Event Reporting System (FAERS). The primary rationale for this analysis is routine pharmacovigilance, aiming to support the ongoing safety evaluation of this immune checkpoint inhibitor combination in real-world clinical settings. While previous research has predominantly focused on monotherapies, our study addresses a critical knowledge gap by characterizing the safety profile of the combination therapy. By analyzing post-marketing real-world data, we aim to gain a deeper understanding of these drugs’ safety in clinical practice, with a particular focus on identifying new or rare AE signals. To further investigate AE patterns, we applied Weibull Shape Parameter (WSP) test to assess the time-to-onset of AEs, revealing trends in their incidence over different time periods. Additionally, we conducted subgroup analyses based on gender and body weight to assess AE occurrence in various patient groups. We also explored the off-label use of the combination therapy. Unlike the limitations of clinical trials, our study captures long-term and rare AEs, addressing gaps in the current literature. This study provides a comprehensive view of the safety and clinical application of cancer immunotherapy, helping to optimize treatment strategies and offering important insights for future drug safety monitoring and research.
Materials and methods
Study design and data source
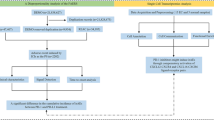
This retrospective pharmacovigilance disproportionality analysis is based on the FAERS database, a large, publicly available Individual Case Safety Report (ICSR) repository. As a post-marketing safety surveillance system, FAERS contains millions of real-world spontaneous AE reports submitted by healthcare professionals, patients, legal representatives, and drug manufacturers. It is particularly valuable for routine pharmacovigilance because it enables the detection of rare, serious, and long-latency AEs that may not be observed in pre-marketing clinical trials. The FAERS data files include seven types of datasets: patient demographic and administrative information (DEMO), drug information (DRUG), adverse event coding (REAC), patient outcomes (OUTC), report sources (RPSR), therapy start and end dates (THER), and indications for drug administration (INDI)14. Each submitted report classifies the role of each drug in the AEs: primary suspect (PS), secondary suspect (SS), interacting (I), or concomitant (C). This study extracted AEs associated with the combined use of ipilimumab and nivolumab, submitted by healthcare professionals (including doctors, pharmacists, and other health-professional) from the first quarter of 2015 to the first quarter of 2024. Considering that FDA data submissions come from various sources, we addressed potential duplicate reports following FDA recommendations: when CASEID is identical, we selected the latest FDA_DT and the higher PRIMARYID. This study adhered to the READUS-PV guidelines (see Supplementary Material 1 for details)15. Figure 1 provides a comprehensive flowchart detailing the data extraction, processing, and analysis process.
The process of selecting AEs associated with the combination of ipilimumab and nivolumab from FAERS and the research workflow.
Adverse events and drug identification
All reported AEs were meticulously coded according to the Medical Dictionary for Regulatory Activities (MedDRA) classification system. MedDRA’s hierarchical structure comprises five levels: System Organ Class (SOC), High-Level Group Term (HLGT), High-Level Term (HLT), Preferred Term (PT), and Lowest Level Term (LLT)16. In this study, we standardized and categorized the SOC and PT levels, focusing on these as primary study objects. To ensure data comprehensiveness, we identified cases using both the generic and brand names of the drugs: Ipilimumab (Yervoy) and Nivolumab (Opdivo). Combination therapy was defined in two ways: Ipilimumab (Yervoy) as PS with Nivolumab (Opdivo) as SS/C/I, and vice versa. At the SOC level, we excluded AEs unrelated to drugs, such as “injury, poisoning, and procedural complications,” “product issues,” “surgical and medical procedures,” and “social circumstances”17. At the PT level, we excluded AEs influenced by the underlying disease, such as “tumor progression” and “cancer pain.” Key patient outcomes were categorized as hospitalization, death, disability, permanent defect, life-threatening, congenital anomaly, or other important medical events.
Statistical analysis
To evaluate potential safety signals, we applied disproportionality analysis, a well-established pharmacovigilance method used to detect statistical associations between drugs and reported AEs. This study used the Reporting Odds Ratio (ROR), Proportional Reporting Ratio (PRR), Bayesian Confidence Propagation Neural Network (BCPNN), and Multi-Item Gamma Poisson Shrinker (MGPS) algorithms to detect AE signals associated with drug combinations. Generally, the higher the values of these parameters, when the incidence of a specific AE significantly exceeds the background frequency in the database, the stronger the signal18. Table 1 details the calculation equations and criteria for these four algorithms. In these calculations, “a” represents the number of specific AEs related to the target drug combination, “b” represents the number of other AEs related to the target drug, “c” represents the number of AEs related to other drugs involving the target AE, and “d” represents the number of other AEs unrelated to the target drug. Specifically, the criteria for identifying positive signals are as follows: (1) For ROR, the report count threshold is ≥ 3, and the 95% confidence interval (CI) lower limit of ROR is > 1; (2) For PRR, the report count threshold is ≥ 3, and the lower limit of the 95% CI for PRR is ≥ 2; (3) For BCPNN, the lower limit of the 95% CI for the information component (IC025) is > 0; (4) For MGPS, the empirical Bayesian geometric mean lower 95% CI (EBGM05) is > 2. In this study, we considered an AE signal to be statistically significant only if all four signal detection methods were positive. All analyses were performed using R software version 4.2.3, utilizing the “dplyr,” “data.table,” and “ggplot2” packages.
Time-to-onset analysis
Time-to-onset is defined as the time from the therapy start date (START_DT in the THER file) to the AE occurrence date (EVENT_DT in the DEMO file). Reports with input errors (EVENT_DT earlier than START_DT), inaccurate dates, and duplicates were excluded. In our study, the median, quartiles, and WSP test were used to evaluate the time to onset of AEs. The WSP test can determine changes in AE risk over time. The scale parameter α of the Weibull distribution determines the scale: a larger α stretches the distribution, while a smaller α contracts it. The shape parameter β determines the shape: a larger β produces a left-skewed curve, while a smaller β produces a right-skewed curve. When the shape parameter β < 1 and its 95% CI < 1, the risk decreases over time (early failure type); β equal to or close to 1 with its 95% CI containing 1 indicates a constant risk (random failure type); β > 1 with its 95% CI > 1 indicates an increasing risk over time (wear-out failure type)19.
Results
Clinical characteristics
Between 2015 and the first quarter of 2024, we retrieved 13,038,441 reports from the FAERS database after removing duplicates. Among these, 19,462 reports involved AEs related to the combined use of ipilimumab and nivolumab, submitted by healthcare professionals (Fig. 1). The clinical characteristics of these events are detailed in Table 2. Over recent years, the number of AE reports for the combination therapy has shown an overall upward trend (Fig. 2), with the highest number of reports in 2023 (3,419 reports, accounting for 17.6%). The proportion of AEs was higher in male patients (58.5%) compared to female patients (30.4%). In terms of age distribution, the majority of reports were from the 65–85 age group, making up 39.4% of the total. Regarding the outcomes of the AEs, 39% of the reports indicated hospitalization, 21.8% reported death, and other severe outcomes included life-threatening conditions and disabilities. Most AE reports originated from the United States (32.8%), followed by Japan (30%). The combination therapy was most commonly used for malignant melanoma (33.6%), followed by renal cell carcinoma (20.9%), non-small cell lung cancer (17.9%), malignant mesothelioma (3.5%), esophageal cancer (1.8%), and colorectal cancer (1.6%).
The trend in the number of AEs reports for combined Ipilimumab and Nivolumab therapy per year from 2015 to the first quarter of 2024.
Signal detection
The combined use of ipilimumab and nivolumab was associated with AEs involving 20 organ systems. Table 3 shows the signal strength at the SOC level. The most prominent SOC was “endocrine disorders,” followed by “hepatobiliary disorders,” both showing positive signals in all four detection methods. Additionally, except for the BCPNN (IC) algorithm, the other three methods indicated positive signals for “metabolism and nutrition disorders,” suggesting that AEs in this system are also significant and frequent.
At the PT level, 435 AEs related to the combined use of ipilimumab and nivolumab were identified using all four signal detection methods (Supplementary Material 2: Table S1). Notably, AEs with occurrences greater than 100 have been mentioned in the package inserts of either ipilimumab or nivolumab. Furthermore, 125 new significant AE signals were unexpectedly identified (Supplementary Material 2: Table S1), with those having occurrences greater than 10 shown in Table 4. The most prominent AEs included immune-mediated optic neuritis (ROR = 725.55, IC025 = 6.74), immune-mediated cystitis (ROR = 262.05, IC025 = 5.98), acquired achromotrichia (ROR = 218.46, IC025 = 5.75), oral lichenoid reaction (ROR = 60.46, IC025 = 4.12), thyrotoxic crisis (ROR = 31.18, IC025 = 3.25), Bell’s palsy (ROR = 10.76, IC025 = 1.74), bilateral deafness (ROR = 7.42, IC025 = 1.21), complete atrioventricular block (ROR = 5.68, IC025 = 0.83), eyelid ptosis (ROR = 4.87, IC025 = 0.61), and inappropriate antidiuretic hormone secretion (SIADH, ROR = 3.43, IC025 = 0.11).
Subgroup analyses
We conducted detailed subgroup analyses to explore differences in AEs associated with the combined use of ipilimumab and nivolumab among various populations (Supplementary Material 2: Table S2). Specifically, the most prominent AEs in males were immune-mediated endocrine diseases, uveitis, hypophysitis, adrenal insufficiency, and rhabdomyolysis. In females, the most significant AEs were immune-mediated uveitis, esophagitis, hypophysitis, thyroiditis, and encephalopathy. Among patients weighing less than 50 kg, immune-mediated uveitis, asymptomatic thyroiditis, fulminant type 1 diabetes, and liver diseases were notable. In the 50–100 kg weight group, the most significant AEs were salivary gland inflammation, thyroid-stimulating hormone deficiency, hypophysitis, and autoimmune colitis. For patients weighing over 100 kg, prominent AEs included autoimmune nephritis, hypophysitis, colitis, atrial fibrillation, and ventricular tachycardia.
Off-label use
According to the package inserts, the combined use of ipilimumab and nivolumab is indicated for renal cell carcinoma, non-small cell lung cancer, malignant pleural mesothelioma, and esophageal cancer as first-line treatment, and is also approved for malignant melanoma, colorectal cancer, and liver cancer. Our study identified off-label use in the following conditions: small cell lung cancer (n = 314), urothelial carcinoma (n = 215), gastric cancer (n = 147), prostate cancer (n = 142), head and neck cancer (n = 113), pancreatic cancer (n = 98), glioma (n = 75), breast cancer (n = 73), ovarian cancer (n = 64), cervical cancer (n = 49), leukemia (n = 41), cholangiocarcinoma (n = 35), and thyroid cancer (n = 33).
Onset time of events
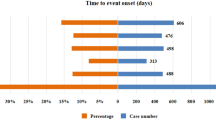
The onset times of AEs related to the combined use of ipilimumab and nivolumab were carefully collected from the database. After removing duplicates and erroneous reports, 8,870 reports provided onset time data. The median onset time was 45 days [interquartile range (IQR): 20–104 days]. The study found that the highest frequency of AEs occurred within the first 30 days of treatment (n = 3,383, 38.1%), followed by a gradual decline, with a small resurgence between 91 and 180 days (n = 1,343, 15.1%). AEs could still occur more than a year after treatment initiation (n = 429, 4.8%), as shown in Fig. 3. In the WSP test, the scale parameter was 80.7 (95% CI: 78.5–82.9), and the shape parameter was 0.80 (95% CI: 0.79–0.82). The upper limit of the 95% CI for the shape parameter being less than 1 indicates an early failure type, meaning the overall incidence of AEs decreases over time.
Time to onset of ipilimumab and nivolumab combination-related AEs.
Discussion
The combination therapy of ipilimumab and nivolumab has garnered significant attention and application in treating various malignancies, particularly advanced melanoma, renal cell carcinoma, and non-small cell lung cancer. Our retrospective pharmacovigilance analysis, based on the FAERS database and covering reports submitted by healthcare professionals from the first quarter of 2015 to the first quarter of 2024, provides new insights into the safety of this combination therapy in real-world clinical settings. We observed that the number of AE reports for the combined use of ipilimumab and nivolumab has increased yearly (Fig. 2), peaking in 2023. These reports indicate a high hospitalization rate (39%) and death rate (21.8%), reflecting the growing clinical application of this drug combination and underscoring the necessity for vigilant safety monitoring.
Excluding cases with unknown gender, the rate of AEs reported by male patients (58.5%) was higher than that by female patients (30.4%). This may be related to the higher incidence of the top three indications for this drug combination—malignant melanoma, renal cell carcinoma, and non-small cell lung cancer—among men20,21,22. We also observed the highest AE reporting rate among individuals aged 65–85, likely because this age group represents the peak incidence of adult cancers23. Clinicians should pay particular attention to these high-risk groups, ensuring closer monitoring and follow-up.
While the combination therapy of ipilimumab and nivolumab offers better clinical efficacy compared to monotherapy, it also comes with a higher incidence of AEs24. Most AEs were irAEs, caused by the activation of T-cells through CTLA-4 and PD-1 pathway inhibition, which are crucial for maintaining immune homeostasis and preventing autoimmunity25. This immune activation can attack normal tissues, causing autoimmune reactions. Through FAERS data mining, we identified common AEs with more than 100 cases, such as pyrexia, colitis, pneumonitis, hypophysitis, hepatitis, myocarditis, hyperthyroidism, and hyponatremia, all of which are mentioned in the drug package inserts, further validating our findings.
Based on disproportionality analysis at the SOC level, “endocrine disorders” showed the strongest AE signals for the combination therapy of ipilimumab and nivolumab, consistent with current research26,27. In addition to high-frequency AEs such as hypophysitis, adrenal insufficiency, and thyroiditis, we also identified unexpected findings, including thyrotoxic crisis, SIADH, and diabetes insipidus (DI), which are not mentioned in the drug package inserts. Thyrotoxic crisis, a life-threatening complication of hyperthyroidism, was not reported in previous clinical trials but was identified in 26 cases in our FAERS analysis. The CTLA-4 and PD-1 pathways play a critical role in maintaining immune tolerance28. Combination therapy may trigger thyrotoxic crisis through synergistic immune activation, increased production of autoantibodies, and the dynamic progression of thyroiditis, ultimately leading to excessive release of thyroid hormones29. Yonezaki et al. described a severe case in an 85-year-old melanoma patient30highlighting the need for high vigilance and immediate treatment with β-blockers, iodide, and glucocorticoids. SIADH, caused by excessive ADH secretion, leads to water retention and hyponatremia. Our FAERS analysis identified 30 SIADH reports, with ICIs potentially inducing it through CNS lesions or immune responses31. We also found 11 reports of DI, which can result from immune-mediated hypophysitis or renal inflammation32,33. Both SIADH and DI are rare AEs with ICIs, requiring routine monitoring of serum sodium, urine output, and osmolality to detect early signs during treatment.
Hepatobiliary disorders also showed strong signal strength. Common AEs such as immune-mediated hepatitis, drug-induced hepatitis, and liver failure are documented in the package inserts. Interestingly, we discovered new AEs like cholecystitis (n = 54, ROR = 5.17, IC025 = 0.7) and biliary obstruction (n = 13, ROR = 4.73, IC025 = 0.57), not previously reported in package inserts. Some studies support our findings. A study on 4,253 patients using ICIs reported cholecystitis in 1.6% of anti-CTLA-4 monotherapy patients, 0.4% of anti-PD-1/L1 monotherapy patients, and 0.9% of combination therapy patients34. Kashima et al. reported a case of cholecystitis, cholangitis, and secondary biliary obstruction in a patient treated with nivolumab35. The underlying mechanism may primarily involve excessive T-cell activation targeting the gallbladder and bile duct epithelium, leading to inflammation, biliary strictures, or dilation36. Histopathological findings may include inflammatory cell infiltration of the bile duct epithelium and noncentrilobular diffuse fibrosis of the extrahepatic bile ducts36. Given these findings, clinicians should be alert to biliary system AEs in patients receiving ICIs, especially those on combination therapy, presenting with abdominal pain and jaundice, and consider imaging and liver function tests for early diagnosis.
We also identified high-signal AEs not mentioned in package inserts, such as immune-mediated optic neuritis, immune-mediated cystitis, eyelid ptosis, oral lichenoid reaction, Bell’s palsy, bilateral deafness, and complete atrioventricular block. Due to the limitations of clinical trials, some delayed and rare AEs may not be detected. Post-marketing data analysis can supplement drug safety information. Although some rare AEs have few cases, existing literature supports our findings. For instance, a 61-year-old female and a 58-year-old male experienced painless visual decline after combination therapy, diagnosed as immune-mediated optic neuritis24,37. Another case described immune-mediated cystitis in a metastatic renal cell carcinoma patient after the third cycle of combination therapy, presenting with intense urinary frequency, dysuria, and bladder spasms, partially relieved with systemic corticosteroids38. In addition, a 2024 case report described a patient with left eyelid ptosis severe enough to cover the pupil and block vision after combination therapy39. Although Bell’s palsy is not specifically mentioned in the package insert, it has been reported in a 45-year-old male who developed left facial droop40. This may be related to immune-mediated facial neuritis, for which corticosteroid therapy is often rapid and effective, although the exact mechanism remains unclear40.
Cardiovascular AEs such as myocarditis, pericarditis, and vasculitis have been identified in clinical trials. Our study found complete atrioventricular block as a significant rare AE. Logiacco et al. reported a 70-year-old female developing complete atrioventricular block after combination therapy, requiring a leadless atrioventricular pacemaker due to persistent conduction abnormalities41. The underlying mechanism primarily involves immune-related myocarditis, which can lead to inflammation and damage of the cardiac conduction system—such as the atrioventricular node and the His bundle—ultimately resulting in advanced atrioventricular block42. Therefore, early identification and intervention are essential for patients with cardiovascular symptoms and ECG abnormalities undergoing combination therapy. Lastly, although AEs like achromotrichia acquired and salivary duct inflammation are rarely reported, ICIs are known to cause a broad range of immune-mediated AEs affecting the skin, endocrine, and other organ systems. Clinicians should remain vigilant, closely monitoring symptoms and intervening as necessary.
In subgroup analysis, we found that the signal for rhabdomyolysis was significant in males receiving combination therapy but not observed in females. Previous studies indicate that excess androgens in muscle cells can lead to oxidative stress, damaging cell membranes and organelles, increasing the risk of myocyte apoptosis and lysis43. The role of sex hormone differences in this AE requires further investigation. In the > 100 kg weight subgroup, we observed atrial fibrillation and ventricular tachycardia, not seen in the 50–100 kg and < 50 kg subgroups. Obesity induces structural and functional changes in the atria and ventricles, including atrial enlargement, myocardial fibrosis, and adipose infiltration, altering conduction pathways and increasing arrhythmia risk44. Therefore, strict monitoring and management are necessary for obese patients on combination therapy to detect and address arrhythmias.
Our study identified off-label use of combination therapy in indications not included in the package inserts. These include small cell lung cancer, urothelial carcinoma, gastric cancer, prostate cancer, and head and neck cancer. Some clinical trials are exploring the efficacy and toxicity of this combination therapy in these diseases compared to other treatment regimens45,46,47but more research is needed to confirm whether this regimen offers more clinical benefits. Mining new indications from the database has the potential to expand drug application, promote drug development, and improve clinical practice.
We found that the median onset time for AEs with combination therapy was 45 days. Most AEs occurred within the first month (n = 3383, 38.1%) and the second month (n = 1827, 20.6%), with the WSP test indicating a general decline in AE incidence over time. However, AEs could still occur more than a year after treatment initiation (n = 429, 4.8%). Thus, clinicians should closely observe patients on combination therapy, especially during the first two months, and continue long-term monitoring and follow-up to ensure timely detection and management of late-onset AEs.
This study offers several key strengths. By utilizing FAERS—the world’s largest real-world pharmacovigilance database—we comprehensively assessed the safety of ipilimumab-nivolumab combination therapy and addressed limitations of clinical trials. We analyzed 19,462 AE reports from 2015 to the first quarter of 2024, a sample size far larger than most clinical studies, allowing us to detect long-term and rare AEs (e.g., thyrotoxic crisis, immune-mediated cystitis) that are often missed in trials due to limited follow-up and strict inclusion criteria. To ensure methodological rigor, we applied four disproportionality algorithms (ROR, PRR, BCPNN, MGPS) to improve signal reliability and reduce false positives. We also conducted time-to-onset analysis using the WSP test, revealing that 38.1% of AEs occurred within 30 days, which helps inform early clinical monitoring. Notably, we identified 125 unexpected AEs—such as immune-mediated optic neuritis and complete atrioventricular block—that are not included in the current drug labels. Some of these adverse events have previously been reported only in isolated cases, such as ptosis39 and complete atrioventricular block41and our findings provide the first population-level evidence. We also highlighted key organ-specific risks, with endocrine (ROR: 28.79) and hepatobiliary (ROR: 6.21) disorders being the most prominent, expanding upon prior meta-analyses with greater detail26. Together, these findings offer valuable insights for post-marketing safety monitoring and clinical decision-making in cancer immunotherapy.
However, several limitations exist. The data were spontaneously submitted to the FAERS database, which may introduce bias. To ensure accuracy, only reports submitted by healthcare professionals were analyzed. Additionally, the FAERS database does not include information on the total number of patients treated, preventing the estimation of AE incidence rates. Incomplete patient data also limited the ability to control for confounding factors, meaning the study can only indicate an association between the drug combination and AEs, without establishing causality. Further clinical investigation is needed to confirm these findings.
Conclusion
This study utilized the FAERS database to conduct a safety analysis of the combination therapy of ipilimumab and nivolumab, scientifically elucidating the true safety profile of this drug combination. New significant AEs were identified. These findings address the limitations of clinical trials, provide a basis for safety monitoring of the combined use of ipilimumab and nivolumab, and offer valuable evidence for further research and clinical application of this drug combination.
Data availability
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-adverse-event-reporting-system-faers-database.
Abbreviations
- FAERS:
-
FDA adverse event reporting system
- ICSR:
-
Individual case safety report
- irAEs:
-
Immune-related adverse events
- AEs:
-
Adverse events
- PFS:
-
Progression-free survival
- OS:
-
Overall survival
- ROR:
-
Reporting odds ratio
- PRR:
-
Proportional reporting ratio
- BCPNN:
-
Bayesian confidence propagation neural network
- MGPS:
-
Multi-Item gamma poisson shrinker
- WSP:
-
Weibull shape parameter
- ICIs:
-
Immune checkpoint inhibitors
- MedDRA:
-
Medical dictionary for regulatory activities
- SOC:
-
System organ class
- HLGT:
-
High-level group term
- HLT:
-
High-level term
- PT:
-
Preferred term
- LLT:
-
Lowest level term
- DEMO:
-
Patient demographic and administrative information
- DRUG:
-
Drug information
- REAC:
-
Adverse event coding
- OUTC:
-
Patient outcomes
- RPSR:
-
Report sources
- THER:
-
Therapy start and end dates
- INDI:
-
Indications for drug administration
- IC:
-
Information component
- SIADH:
-
Syndrome of inappropriate antidiuretic hormone secretion
- DI:
-
Diabetes insipidus
- IQR:
-
Interquartile range
References
Stoff, R. et al. Efficacy and toxicity of Ipilimumab-Nivolumab combination therapy in elderly metastatic melanoma patients. Front. Oncol. 12, 1020058. https://doi.org/10.3389/fonc.2022.1020058 (2022).
Albiges, L. et al. Nivolumab plus ipilimumab versus Sunitinib for first-line treatment of advanced renal cell carcinoma: extended 4-year follow-up of the phase III checkmate 214 trial. ESMO Open. 5, e001079. https://doi.org/10.1136/esmoopen-2020-001079 (2020).
Almutairi, A. R., McBride, A., Slack, M., Erstad, B. L. & Abraham, I. Potential Immune-Related adverse events associated with monotherapy and combination therapy of ipilimumab, nivolumab, and pembrolizumab for advanced melanoma: A systematic review and Meta-Analysis. Front. Oncol. 10, 91. https://doi.org/10.3389/fonc.2020.00091 (2020).
Carlino, M. S. & Long, G. V. Ipilimumab combined with nivolumab: A standard of care for the treatment of advanced melanoma?? Clin. Cancer Res. 22, 3992–3998. https://doi.org/10.1158/1078-0432.Ccr-15-2944 (2016).
Hellmann, M. D. et al. Nivolumab plus ipilimumab in advanced Non-Small-Cell lung Cancer. N Engl. J. Med. 381, 2020–2031. https://doi.org/10.1056/NEJMoa1910231 (2019).
Doki, Y. et al. Nivolumab combination therapy in advanced esophageal Squamous-Cell carcinoma. N Engl. J. Med. 386, 449–462. https://doi.org/10.1056/NEJMoa2111380 (2022).
Peters, S. et al. First-line nivolumab plus ipilimumab versus chemotherapy in patients with unresectable malignant pleural mesothelioma: 3-year outcomes from checkmate 743. Ann. Oncol. 33, 488–499. https://doi.org/10.1016/j.annonc.2022.01.074 (2022).
Harrington, K. J. et al. Efficacy and safety of nivolumab plus ipilimumab vs nivolumab alone for treatment of recurrent or metastatic squamous cell carcinoma of the head and neck: the phase 2 checkmate 714 randomized clinical trial. JAMA Oncol. 9, 779–789. https://doi.org/10.1001/jamaoncol.2023.0147 (2023).
Janssen, J. C. et al. Safe stop IPI-NIVO trial: early discontinuation of nivolumab upon achieving a complete or partial response in patients with irresectable stage III or metastatic melanoma treated with first-line ipilimumab-nivolumab - study protocol. BMC Cancer. 24, 632. https://doi.org/10.1186/s12885-024-12336-0 (2024).
Hodi, F. S. et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 19, 1480–1492. https://doi.org/10.1016/s1470-2045(18)30700-9 (2018).
Wolchok, J. D. et al. Long-Term outcomes with nivolumab plus ipilimumab or nivolumab alone versus ipilimumab in patients with advanced melanoma. J. Clin. Oncol. 40, 127–137. https://doi.org/10.1200/JCO.21.02229 (2022).
Yang, J., Lagana, S. M., Saenger, Y. M. & Carvajal, R. D. Dual checkpoint inhibitor-associated eosinophilic enteritis. J. Immunother Cancer. 7, 310. https://doi.org/10.1186/s40425-019-0743-5 (2019).
Sakai, K. et al. A Case of nivolumab-induced severe mononeuropathy multiplex and rhabdomyolysis. Case Rep. Med. 1093858, (2017). https://doi.org/10.1155/2017/1093858 (2017).
Kadoyama, K., Sakaeda, T., Tamon, A. & Okuno, Y. Adverse event profile of tigecycline: data mining of the public version of the U.S. Food and drug administration adverse event reporting system. Biol. Pharm. Bull. 35, 967–970. https://doi.org/10.1248/bpb.35.967 (2012).
Fusaroli, M. et al. The reporting of a disproportionality analysis for drug safety signal detection using individual case safety reports in pharmacovigilance (READUS-PV): development and statement. Drug Saf. 47, 575–584. https://doi.org/10.1007/s40264-024-01421-9 (2024).
Mascolo, A. et al. Immune checkpoint inhibitors and cardiotoxicity: an analysis of spontaneous reports in eudravigilance. Drug Saf. 44, 957–971. https://doi.org/10.1007/s40264-021-01086-8 (2021).
Wang, J., Zhang, A., Ye, M. & Zhang, C. Examining the safety of mirabegron: an analysis of real-world pharmacovigilance data from the US FDA adverse event reporting system (FAERS) database. Front. Pharmacol. 15 https://doi.org/10.3389/fphar.2024.1376535 (2024).
Yang, Y. et al. A real-world pharmacovigilance study of FDA adverse event reporting system (FAERS) events for venetoclax. PLoS One. 17, e0278725. https://doi.org/10.1371/journal.pone.0278725 (2022).
Kinoshita, S., Hosomi, K., Yokoyama, S. & Takada, M. Time-to-onset analysis of amiodarone-associated thyroid dysfunction. J. Clin. Pharm. Ther. 45, 65–71. https://doi.org/10.1111/jcpt.13024 (2020).
Olsen, C. M., Thompson, J. F., Pandeya, N. & Whiteman, D. C. Evaluation of sex-specific incidence of melanoma. JAMA Dermatol. 156, 553–560 (2020).
Alexander, M., Kim, S. Y. & Cheng, H. Update 2020: management of Non-Small cell lung Cancer. Lung 198, 897–907. https://doi.org/10.1007/s00408-020-00407-5 (2020).
Jonasch, E., Gao, J. & Rathmell, W. K. Renal cell carcinoma. Bmj 349, g4797. https://doi.org/10.1136/bmj.g4797 (2014).
National Academies of Sciences, E. & Medicine Diagnosing and Treating Adult Cancers and Associated Impairments (National Academies, 2021).
Postow, M. A., Longo, D. L., Sidlow, R. & Hellmann, M. D. Immune-Related adverse events associated with immune checkpoint Blockade. N. Engl. J. Med. 378, 158–168. https://doi.org/10.1056/NEJMra1703481 (2018).
Caturegli, P. et al. Autoimmune hypophysitis. Endocr. Rev. 26, 599–614. https://doi.org/10.1210/er.2004-0011 (2005).
Barroso-Sousa, R. et al. Incidence of endocrine dysfunction following the use of different immune checkpoint inhibitor regimens. JAMA Oncol. 4 https://doi.org/10.1001/jamaoncol.2017.3064 (2018).
Chang, L. S. et al. Endocrine toxicity of Cancer immunotherapy targeting immune checkpoints. Endocr. Rev. 40, 17–65. https://doi.org/10.1210/er.2018-00006 (2019).
Buchbinder, E. I. & Desai, A. CTLA-4 and PD-1 pathways: similarities, differences, and implications of their Inhibition. Am. J. Clin. Oncol. 39, 98–106. https://doi.org/10.1097/coc.0000000000000239 (2016).
Sznol, M. et al. Endocrine-related adverse events associated with immune checkpoint Blockade and expert insights on their management. Cancer Treat. Rev. 58, 70–76. https://doi.org/10.1016/j.ctrv.2017.06.002 (2017).
Yonezaki, K. et al. Combination therapy of ipilimumab and nivolumab induced thyroid storm in a patient with hashimoto’s disease and diabetes mellitus: a case report. J. Med. Case Rep. 12 https://doi.org/10.1186/s13256-018-1708-x (2018).
Park, S. J. & Shin, J. I. Inflammation and hyponatremia: an underrecognized condition? Korean J. Pediatr. 56, 519–522. https://doi.org/10.3345/kjp.2013.56.12.519 (2013).
Mamlouk, O. et al. Nephrotoxicity of immune checkpoint inhibitors beyond tubulointerstitial nephritis: single-center experience. J. Immunother Cancer. 7 https://doi.org/10.1186/s40425-018-0478-8 (2019).
Kennedy, R., Awada, H., Vura, N., Ciltea, D. & Morocco, M. Endocrinopathies from checkpoint inhibitors: incidence, outcomes, and management. Cleve Clin. J. Med. 90, 307–317. https://doi.org/10.3949/ccjm.90a.22032 (2023).
Abu-Sbeih, H. et al. Case series of cancer patients who developed cholecystitis related to immune checkpoint inhibitor treatment. J. Immunother. Cancer. 7 https://doi.org/10.1186/s40425-019-0604-2 (2019).
Kashima, J., Okuma, Y., Shimizuguchi, R. & Chiba, K. Bile duct obstruction in a patient treated with nivolumab as second-line chemotherapy for advanced non-small-cell lung cancer: a case report. Cancer Immunol. Immunother. 67, 61–65. https://doi.org/10.1007/s00262-017-2062-3 (2017).
Pi, B. et al. Immune-related cholangitis induced by immune checkpoint inhibitors: a systematic review of clinical features and management. Eur. J. Gastroenterol. Hepatol. 33, e858–e867. https://doi.org/10.1097/meg.0000000000002280 (2021).
Kim, J. M. et al. Ophthalmic Immune-Related adverse events of immunotherapy: A Single-Site case series. Ophthalmology 126, 1058–1062. https://doi.org/10.1016/j.ophtha.2019.01.031 (2019).
Letícia Quinane, D. H., Ladislau, L. & Tarouquella, C. C. J. Autoimmune cystitis: an unusual adverse effect of the ipilimumab/nivolumab combination in metastatic renal cell Carcinoma–Case report and review of literature. Int. J. Clin. Case Stud. Rep. 2, 123–127 (2020).
Jazieh, K., Kottschade, L. & Dimou, A. Keeping an eye out for immunotherapy toxicity: A case of unilateral ptosis caused by ipilimumab/nivolumab therapy. J. Immunother Precis Oncol. 7, 126–129. https://doi.org/10.36401/jipo-23-44 (2024).
Zecchini, J. M., Kim, S., Yum, K. & Friedlander, P. Development of bell’s palsy after treatment with ipilimumab and nivolumab for metastatic melanoma: A case report. J. Immunother. 41, 39–41. https://doi.org/10.1097/cji.0000000000000184 (2018).
Logiacco, A. et al. A rare case of Ipilimumab/Nivolumab–Induced myocarditis complicated by high grade atrioventricular block in a patient with metastatic renal cell Cancer. Eur. Heart J. Supplements. 26, ii175–ii175. https://doi.org/10.1093/eurheartjsupp/suae036.425 (2024).
Kondo, H., Kirigaya, J., Matsuzawa, Y. & Hibi, K. Two cases of immune checkpoint Inhibitor-Induced myocarditis with complete atrioventricular block. Cureus 15, e36446. https://doi.org/10.7759/cureus.36446 (2023).
Stadler, K., Liu, S., Navarro, G. & Mauvais-Jarvis, F. Androgen excess produces systemic oxidative stress and predisposes to β-Cell failure in female mice. PLoS One. 5 https://doi.org/10.1371/journal.pone.0011302 (2010).
Patel, K. H. K. et al. Obesity as a risk factor for cardiac arrhythmias. BMJ Med. 1 https://doi.org/10.1136/bmjmed-2022-000308 (2022).
Ready, N. E. et al. Nivolumab monotherapy and nivolumab plus ipilimumab in recurrent small cell lung cancer: results from the checkmate 032 randomized cohort. J. Thorac. Oncol. 15, 426–435. https://doi.org/10.1016/j.jtho.2019.10.004 (2020).
Sharma, P. et al. Nivolumab alone and with ipilimumab in previously treated metastatic urothelial carcinoma: checkmate 032 nivolumab 1 mg/kg plus ipilimumab 3 mg/kg expansion cohort results. J. Clin. Oncol. 37, 1608–1616. https://doi.org/10.1200/jco.19.00538 (2019).
Sharma, P. et al. Nivolumab plus ipilimumab for metastatic Castration-Resistant prostate cancer: preliminary analysis of patients in the checkmate 650 trial. Cancer Cell. 38, 489–499e483. https://doi.org/10.1016/j.ccell.2020.08.007 (2020).
Acknowledgements
We appreciate the FAERS open policy and the provided data, as well as all participants in this study.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 52173281).
Author information
Authors and Affiliations
Contributions
XD was responsible for the conceptualization, data curation, formal analysis, software, and writing of the original draft. HC contributed to the conceptualization, data curation, and reviewed and edited the manuscript. JL was involved in the methodology and reviewed and edited the manuscript. TZ handled the visualization and reviewed and edited the manuscript. YH contributed to writing, reviewed and edited the manuscript, as well as funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dai, X., Cao, H., Li, J. et al. Disproportionality analysis of adverse events associated with ipilimumab and nivolumab combination therapy based on FAERS database. Sci Rep 15, 21055 (2025). https://doi.org/10.1038/s41598-025-07287-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07287-w