Abstract
Bone regeneration is a critical area of research in the treatment of bone defects within clinical practice. Molybdenum (Mo) is an essential trace element for many organisms, which plays a vital role in various cellular activities, such as maintaining energy metabolism and promoting cell proliferation. However, whether Mo regulates the osteogenic differentiation of periodontal ligament stem cells (PDLSCs) and promotes bone tissue regeneration remains unknown. This study confirmed the nourishing effect of Mo on the osteogenic differentiation of PDLSCs, as evidenced by higher expression of osteogenic-related markers (OCN, BSP, OSX and RUNX2), ALP activity and mineralization ability. Moreover, through constructing compound material GelMA + PDLSC, we proved that Mo promoted PDLSC-based bone regeneration quantity and quality in rat calvarial defects. Finally, we revealed the underlying mechanism that the JAK/STAT3 signaling pathway actively participated in the Mo-enhanced osteogenic differentiation of PDLSCs. The inhibition of STAT3 diminished the benefit of Mo. Overall, our study found that Mo facilitated PDLSC-based bone regeneration through the JAK/STAT3 signaling pathway, which may shed new light on clinical bone regeneration therapeutic strategies.
Similar content being viewed by others
Introduction
Bone regeneration is widely studied as one of the most promising approaches to address bone defects in clinical practice1 Based on the three essential elements of tissue engineering, research on various biomaterials, stem cell transplantation, and growth factors have significantly improved the speed and quality of bone regeneration. However, the apoptosis, necrosis, and dedifferentiation of stem cells in the transplanted environment often led to unsatisfactory regeneration outcomes2. Therefore, exploring strategies to stimulate stem cell differentiation potential to enhance osteogenic efficacy is of great significance.
Mounting evidence supports the role of mesenchymal stem cell (MSC) transplantation in tissue regeneration, including bone, skin, and hair regeneration3. Periodontal ligament stem cells (PDLSCs) are a unique population of MSCs found in the periodontal ligament4. PDLSCs can be easily extracted from isolated teeth with a wide range of sources. Since their first report in 2004, they have been shown to possess active multipotent differentiation capabilities5. PDLSCs, along with their exosomes and apoptotic vesicles, have been shown to promote the regeneration of bone6,7.
Molybdenum (Mo) is an essential trace element for many organisms, participating in various biochemical reactions. Mo is not only a critical component of certain enzymes in animals, but it also participates in sulfur and iron metabolism8. Moreover, it possesses antioxidant properties and contributes to maintaining cellular energy metabolism and promoting cell proliferation9. However, research on the role of Mo in bone regeneration remains limited, and its effects on the differentiation of PDLSCs have not been reported, leaving the underlying mechanisms unclear.
In the current research, we explored the influence of Mo on the osteogenic differentiation of PDLSCs. Moreover, we constructed GelMA + PDLSC and evaluated its bone regeneration potential after Mo treatment in a rat calvarial defect model. The mechanism underlying was demonstrated as well. This study provides a new theoretical foundation for PDLSC-based bone regeneration and suggests potential clinical applications.
Materials and methods
Isolation and culture of PDLSCs
PDLSCs were obtained from human teeth extracted for orthodontic reasons, with patients providing informed consent. All experiments associated with clinical samples were performed in accordance with the relevant principles and guidelines of the Declaration of Helsinki and were approved by the Ethics Committee of Tongji University. The periodontal ligament was dissected from the roots and minced into small fragments. The fragments were then enzymatically digested with 3 mg/mL collagenase I and 4 mg/mL dispase (Sigma-Aldrich) for 1 h at 37 °C. The digest was then centrifuged and resuspended in α-MEM (Hyclone) supplemented with 10% fetal bovine serum (FBS, Gibco) and 1% penicillin–streptomycin, and plated in 25 cm2 culture flasks. The cells were cultured at 37 °C in a 5% CO2 incubator.
Antibodies and reagents
Hexaammonium molybdate was purchased from Adamas (12027-67-7, Switzerland). Stattic, the inhibitor of STAT3, was purchased from MedChemExpress (HY-13818, USA, 5 μM).
In Western blotting, the primary antibodies against osteocalcin (OCN, sc390877, Santa Cruz), bone sialoprotein (BSP, DF7738, Affinity), osterix (OSX, ab209484, Abcam), runt-related transcription factor 2 (RUNX2, ab236639, Abcam), phospho-STAT3-Y705 (AP0070, Abclonal), STAT3 (A1192, Abclonal) and β-actin (AC026, Abclonal) were applied. In immunofluorescence staining, the primary antibodies against OCN (23418-1-AP, Proteintech) and BSP (DF7738, Affinity) were used.
Characterization of PDLSCs
To evaluate the proliferation ability of PDLSCs, a colony forming assay was conducted. Colonies expanded from single cells were stained with 0.5% crystal violet and observed. Additionally, multiple differentiation potential was assessed by inducing the osteogenic, adipogenic and chondrogenic differentiation of PDLSCs, which were evaluated by specific staining (Alkaline phosphatase and Alizarin red for osteogenesis, Oil Red O for adipogenesis, and Alcian blue for chondrogenesis). Cytometry analysis was performed to assess the expression of stem cell surface markers CD73, CD90, and CD105.
Cell viability assay
Cell viability was tested by the Cell Counting Kit − 8 (BS350B, Biosharp). PDLSCs were plated in a 96-well plate at a density of 5 × 103 cells per well. Following Mo treatment for 24, 48, 72, and 96 h, the medium was refreshed by 90% culture medium plus 10% CCK-8 reagent. After incubation for 1 h at 37 °C, the absorbance at 450 nm was measured by a microplate reader (BioRad, USA).
Alkaline phosphatase (ALP) staining
PDLSCs were adjusted to a density of 3 × 105 cells per well and seeded into a 6-well plate. Following a 7-day osteogenic induction period, cells were fixed in 4% paraformaldehyde and subsequently stained using the ALP staining kit (C3206, Beyotime) in accordance with the manufacturer’s guidelines. Images were taken with a microscopy. The relative ALP activity was analyzed with ImageJ.
Alizarin red staining
Following a 14-day osteogenic induction, the cells were fixed and stained with Alizarin red S solution (ALIR-10001, Oricell) according to the manufacturer’s instructions. After observation under a microscopy, the mineralization nodules were dissolved in 10% hexadecylpyridinium chloride monohydrate (S24536, Yuanye biotechnology). The OD value at 562 nm was assessed with a microplate reader to represent the mineralization nodule intensity.
Preparation of GelMA + PDLSC
GelMA (EFL-GM-30, Engineering For life) was prepared in accordance with the user’s instructions. PDLSCs were harvested and resuspended at a density of 5 × 106 cell / mL in the liquid hydrogel. In each well of a 96-well plate, 100 μL of gel was added, and the plate was placed still at 37 °C for 30 min for cells to stretch. Subsequently, the hydrogel underwent light curing by exposure to 405 nm UV light for 20 s. The resulting mixture was then placed in osteogenic culture medium (with or without Mo) and incubated for 7 days prior to transplantation.
Rat calvarial defect model
All animal experimental protocols were approved by the Ethics Committee of Animal Research of Tongji University. All methods were performed in accordance with the relevant guidelines and regulations. All methods are reported in accordance with ARRIVE guidelines for the reporting of animal experiments. All euthanasia was performed by CO2 asphyxiation followed by cervical dislocation. Male Sprague–Dawley rats (200–250 g, n = 6) were anesthetized with intraperitoneal injection of 1.3% sodium pentobarbital, and the soft tissues were incised to expose the skull. Using a trephine drill with a fixed 5 mm inner diameter, two defects were created in each skull, one on the left side as the GelMA + PDLSC group and the other on the right side for GelMA + PDLSC + Mo group. The materials were then filled into the defects. The rats were housed separately in sterile conditions and euthanized 8 weeks post-surgery. Skull samples were then collected for analysis.
Statistical analysis
The results are expressed as the mean ± standard deviation (SD) from three independent experiments. Statistical analyses were conducted using GraphPad Prism 9. The Shapiro–Wilk test assessed normal distribution. The Student’s t-test was employed to compare two groups, while one-way ANOVA was utilized for multiple group comparisons, followed by Bonferroni correction. Statistical significance was indicated as * for P < 0.05, ** for P < 0.01, and *** for P < 0.001, with P > 0.05 deemed not significant (ns).
Results
Isolation and characterization of PDLSCs
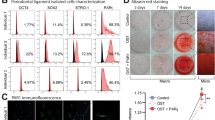
PDLSCs were isolated from extracted teeth and exhibited fibroblast-like morphology (Fig. 1A). When cultured at low density, they could form separate colony clusters, which indicated that they possess strong self-replicating ability (Fig. 1A). Moreover, PDLSCs showed the potential of multidirectional differentiation in vitro. After osteogenic induction, the ALP staining and Alizarin red staining displayed alkaline phosphatase activity and mineralization nodules, respectively (Fig. 1B). When induced for chondrogenic differentiation, cartilaginous matrix was observed through the Alcian Blue staining (Fig. 1B). When induced for adipogenic differentiation, lipid droplets were observed by the Oil Red O staining (Fig. 1B). Further, PDLSCs highly expressed stem cell surface markers such as CD105, CD73 and CD90, while scarcely expressed hematopoietic markers such as CD45 and CD34 (Fig. 1C). In summary, we successfully isolated and characterized the stemness of PDLSCs.
Characterization of PDLSCs. (A) The colony forming ability of PDLSCs were examined by crystal violet staining. Scale bar, 500 μm. (B) The osteogenic, chondrogenic and adipogenic potentials of PDLSCs were identified by ALP staining, Alizarin red staining, Alcian Blue staining and Oil Red O staining. Scale bars, 200 μm. (C) The expression of surface markers of PDLSCs (CD105, CD73, CD90, CD45 and CD34) were verified by flow cytometry analysis.
Mo promoted the osteogenic differentiation of PDLSCs in a concentration-dependent manner
We wondered whether Mo played a role in the osteogenic differentiation of PDLSCs. First, we confirmed that Mo had no cytotoxicity and did not affect PDLSC proliferation by CCK-8 assay (Fig. 2A). PDLCSs were then cultured in the osteogenic medium supplemented with hexaammonium molybdate (4 μg/mL for Mo-4 and 8 μg/mL for Mo-8) for 4 days. The CON group served as a negative control, which is complete medium instead of osteogenic medium. We found an increase in the mRNA levels of the osteogenic markers (OCN, BSP, OSX, RUNX2) in the Mo-4 and Mo-8 group in a concentration dependent manner (Fig. 2B), which is also supported by the WB results on day 4 (Fig. 2C). Further, ALP staining on day 7 (Fig. 2D,E) and Alizarin red staining on day 12 (Fig. 2F,G) validated the facilitating effect of Mo in the osteogenic differentiation of PDLSCs.
Mo promoted the osteogenic differentiation of PDLSCs in a concentration-dependent manner. (A) The effect of Mo on the proliferation of PDLSCs measured by the CCK-8 assay. (B) The mRNA levels of osteogenic markers in PDLSCs after Mo stimulation for 4 days. (C) The protein levels of osteogenic markers in PDLSCs after Mo stimulation for 4 days. (D, E) The ALP activity of PDLSCs after Mo stimulation for 7 days was assessed by ALP staining. Scale bars, 100 μm. (F, G) The mineralization nodules of PDLSCs after Mo stimulation for 12 days were assessed by Alizarin red staining. Scale bars, 100 μm.
Mo facilitated PDLSC-based bone regeneration in a rat calvarial defect model
To assess whether Mo enhances bone regeneration in vivo, we utilized a rat calvarial defect model. First, a composite material, GelMA + PDLSC, was constructed through 3D cell culture (Fig. 3A). Bilateral cranial defects, each 5 mm in diameter, were created, and GelMA + PDLSC or Mo-pretreated PDLSCs embedded in GelMA (GelMA + PDLSC + Mo) was implanted into the defect sites (Fig. 3B). The micro-CT analysis showed significantly more new bone formation in the skulls of rats transplanted with GelMA + PDLSC + Mo, compared to GelMA + PDLSC (Fig. 3C). Quantitative analysis demonstrated that the GelMA + PDLSC + Mo group exhibited superior bone structure and quality (Fig. 3D). Additionally, H&E and Masson’s staining revealed the formation of thick new bone tissue in this group (Fig. 3E,F). Immunofluorescence staining for the osteogenic markers OCN and BSP further confirmed enhanced osteogenic activity in the GelMA + PDLSC + Mo group (Fig. 3G,H). Taken together, wo conclude that Mo efficiently enhanced PDLSC-based bone regeneration.
Mo facilitated PDLSC-based bone regeneration in a rat calvarial defect model. (A) The well-stretched cell morphology of GelMA + PDLSC. Scale bar, 100 μm. (B) The illustration of rat calvarial defect transplantation experiments. (C) 3D reconstructed Micro-CT images of the calvarial defects. (D) Quantitative analysis based on Micro-CT data: bone tissue volume (BV/TV), bone surface density (BS/TV), bone mineral density (BMD), trabecular thickness (Tb.Th), trabecular number (Tb.N) and structure model index (SMI). Representative microscope images of H&E staining (E) and Masson staining (F) of the calvarial defect areas. Scale bars, 1 mm (low magnification) and 500 μm (high magnification). (G, H) The immunofluorescence staining of osteogenic markers of the calvarial defect areas. Scale bars, 100 μm.
Mo promoted the osteogenic differentiation of PDLSCs partially via the JAK/STAT3 signaling pathway
We hypothesized that JAK/STAT3 pathway was majorly involved in Mo-enhanced osteogenic differentiation of PDLSCs, which was validated by Western blotting results. Mo stimulation induced a strong increase in p-STAT3 level (Fig. 4A). To confirm whether Mo promoted the osteogenic differentiation of PDLSCs via JAK/STAT3 pathway, we applied the specific inhibitor stattic. As shown in Fig. 4B,C, the mRNA and protein levels of the osteogenic markers decreased in the (Mo-8 + st) group, compared to the Mo-8 group. ALP activity (Fig. 4D,E) and Alizarin red staining (Fig. 4F,G) further confirmed the tendency. Collectively, these results demonstrated that Mo promoted the osteogenic differentiation of PDLSCs partially via the JAK/STAT3 signaling pathway (Fig. 4H).
Mo promoted the osteogenic differentiation of PDLSCs partially via the JAK/STAT3 signaling pathway. (A) The protein levels of p-STAT3 and total-STAT3 of PDLSCs after Mo stimulation for 4 days. (B) The protein levels of osteogenic markers in PDLSCs treated with Mo-8 or (Mo-8 + st) for 4 days. (C) The mRNA levels of osteogenic markers in PDLSCs treated with Mo-8 or (Mo-8 + st) for 4 days. (D, E) The ALP activity of PDLSCs treated with Mo-8 or (Mo-8 + st) for 7 days was assessed by ALP staining. Scale bars, 100 μm. (F, G) The mineralization nodules of PDLSCs treated with Mo-8 or (Mo-8 + st) for 12 days were assessed by Alizarin red staining. Scale bars, 100 μm. (H) The schematic summary showing the role of Mo and JAK/STAT3 signaling pathway in promoting the osteogenic differentiation of PDLSCs.
Discussion
This study investigates the role of Mo in regulating the osteogenic differentiation of PDLSCs. After confirming the differentiation-accelerating effect of Mo on PDLSCs, we further assessed the outcomes of bone regeneration in vivo. Our results indicated that Mo promotes PDLSC-based bone regeneration in rat calvarial defects. Moreover, we verified that Mo facilitates the osteogenic differentiation of PDLSCs partially via the JAK/STAT3 signaling pathway. These findings shed new light on bone tissue regeneration.
Stem cell-based tissue engineering provides a new therapeutic method for the repair of cranio-maxillofacial bone defects caused by trauma, infection, deformity and tumor10. The selection of stem cells for transplantation is the key of successful tissue regeneration. PDLSCs are a unique group of cells because they can be isolated from a patient’s naturally lost or extracted teeth by non-invasive means without additional surgery (compared to bone marrow-derived stem cells). Also, they have been proved to promote bone regeneration through self-renewal, differentiation and immune modulation4. Our study strengthened the osteogenic differentiation capability of PDLSCs through Mo stimulation to enhance bone tissue regeneration. This not only revealed a new physiological function of Mo, but also explored its application potential.
Mo is an essential element for almost all living organisms. After being absorbed by cells in the form of molybdate, molybdenum is integrated into the molybdenum cofactor, which serves as the catalytic center for various molybdenum-dependent enzymes, thereby contributing to numerous biological processes9. Few studies focused on the relationship between molybdenum and human diseases, with only some reporting inflammatory diseases and tumors11,12. In the field of tissue regeneration, molybdenum disulfide and the derived compound biomaterials (nanosheets, microspheres, etc.) have been explored to enhance osteogenesis13,14,15. However, these studies focused on the antimicrobial and physicochemical properties of the scaffold materials, attempting to developing an implant material with osteointegration and osteoinduction properties. Also, the underlying mechanisms remains largely uncharted. It is reported though that GelMA/molybdenum (Mo)-based polyoxometalate nanoclusters hydrogel scavenges ROS and activates the PI3K/Akt signaling pathway to enhance osteogenic differentiation and bone regeneration16. We report that molybdate can be used as a bioactive molecule in tissue engineering to promote stem cell differentiation and reveal relevant signaling pathway, which complement the understanding of the function of molybdenum.
The JAK/STAT3 signaling pathway is a key intracellular cascade that regulates cell proliferation, differentiation and immune responses17. Dysregulation of JAK/STAT3 signaling is linked to various diseases, including cancer, autoimmune disorders, and chronic inflammation, making it a significant therapeutic target18,19,20. Previous work has revealed its role in promoting adipogenic and osteogenic differentiation21,22. Although direct evidence linking Mo to the JAK/STAT3 signaling pathway remains limited, several studies have reported that Mo-containing compounds or molybdate-based biomaterials could enhance the expression of inflammatory cytokines such as IL-6 and IL-10, both of which are classical upstream activators of the JAK/STAT3 pathway23,24. Therefore, we hypothesized and validated that the activation of JAK/STAT3 signaling pathway is responsible for the osteogenic promoting effect of Mo (Fig. 4H).
Collectively, our study demonstrated the nourishing role of Mo in the osteogenic differentiation of PDLSCs. Also, we confirmed that Mo accelerates bone regeneration in vivo with a rat calvarial defect model. In terms of mechanism, we revealed that Mo facilitates PDLSC-based bone regeneration through the JAK/STAT3 signaling pathway. We believe that our results help to provide new directions for clinical bone regeneration therapeutic strategies.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Tan, B. et al. Biomaterial-based strategies for maxillofacial tumour therapy and bone defect regeneration. Int. J. Oral Sci. 13, 9 (2021).
Iwasaki, K., Peng, Y., Kanda, R., Umeda, M. & Ishikawa, I. Stem cell transplantation and cell-free treatment for periodontal regeneration. Int. J. Mol. Sci. 23, 1011 (2022).
Margiana, R. et al. Clinical application of mesenchymal stem cell in regenerative medicine: A narrative review. Stem Cell Res. Ther. 13, 366 (2022).
Zhai, Q., Dong, Z., Wang, W., Li, B. & Jin, Y. Dental stem cell and dental tissue regeneration. Front. Med. 13, 152–159 (2019).
Seo, B.-M. et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 364, 149–155 (2004).
Zhao, Y. et al. The experimental study of periodontal ligament stem cells derived exosomes with hydrogel accelerating bone regeneration on alveolar bone defect. Pharmaceutics 14, 2189 (2022).
Deng, Y. et al. VDAC1-enriched apoptotic extracellular vesicles emerge as an autophagy activator orchestrating PDLSC-based bone regeneration. Chem. Eng. J. 497, 154625 (2024).
Mendel, R. R. The history of the molybdenum cofactor-a personal view. Molecules 27, 4934 (2022).
Huang, X.-Y., Hu, D.-W. & Zhao, F.-J. Molybdenum: More than an essential element. J. Exp. Bot. 73, 1766–1774 (2022).
Gaihre, B., Uswatta, S. & Jayasuriya, A. C. Reconstruction of craniomaxillofacial bone defects using tissue-engineering strategies with injectable and non-injectable scaffolds. J. Funct. Biomater. 8, 49 (2017).
Zhang, C. et al. Oral zero-valent-molybdenum nanodots for inflammatory bowel disease therapy. Sci. Adv. 8, eabp9882 (2022).
Dhas, N. et al. Molybdenum-based hetero-nanocomposites for cancer therapy, diagnosis and biosensing application: Current advancement and future breakthroughs. J. Control. Release 330, 257–283 (2021).
Kaewmanee, R. et al. Molybdenum disulfide nanosheet/polyimide composites with improved tribological performances, surface properties, antibacterial effects and osteogenesis for facilitating osseointegration. J. Mater. Chem. B 10, 5058–5070 (2022).
Kaewmanee, R. et al. Microporous surface containing flower-like molybdenum disulfide submicro-spheres of sulfonated polyimide with antibacterial effect and promoting bone regeneration and osteointegration. Biomater. Sci. 10, 4243–4256 (2022).
Wu, Z. et al. Quercetin-loaded porous biocomposite of polyimide and molybdenum disulfide nanosheets with antibacterial capability for boosting osteoblastic differentiation and bone-bonding. Biomater. Adv. 154, 213585 (2023).
Liao, X. et al. Combined molybdenum gelatine methacrylate injectable nano-hydrogel effective against diabetic bone regeneration. Int. J. Nanomed. 18, 5925–5942 (2023).
Hillmer, E. J., Zhang, H., Li, H. S. & Watowich, S. S. STAT3 signaling in immunity. Cytokine Growth Factor Rev. 31, 1–15 (2016).
Yu, H., Lee, H., Herrmann, A., Buettner, R. & Jove, R. Revisiting STAT3 signalling in cancer: New and unexpected biological functions. Nat. Rev. Cancer 14, 736–746 (2014).
Gharibi, T. et al. Targeting STAT3 in cancer and autoimmune diseases. Eur. J. Pharmacol. 878, 173107 (2020).
He, G. & Karin, M. NF-κB and STAT3—Key players in liver inflammation and cancer. Cell Res. 21, 159–168 (2011).
Qin, A. et al. Knockout of NOS2 promotes adipogenic differentiation of rat MSCs by enhancing activation of JAK/STAT3 signaling. Front. Cell Dev. Biol. 9, 638518 (2021).
Xiong, W., Guo, X. & Cai, X. SDF-1/CXCR4 axis promotes osteogenic differentiation of BMSCs through the JAK2/STAT3 pathway. Folia Histochem. Cytobiol. 59, 187–194 (2021).
Lin, H. et al. Effects of industrially produced 2-dimensional molybdenum disulfide materials in primary human basophils. NanoImpact 29, 100451 (2023).
Huang, X. et al. Controllable adaptive molybdate-oligosaccharide nanoparticles regulate M2 macrophage mitochondrial function and promote angiogenesis via PI3K/HIF-1α/VEGF pathway to accelerate diabetic wound healing. Adv. Healthc Mater. 13, e2302256 (2024).
Acknowledgements
We thank Figdraw (www.figdraw.com) and Servier Medical Art for the assistance in creating schematic diagram.
Author information
Authors and Affiliations
Contributions
Conceptualization, J.X., J.Y., W.S. and Y.L.; methodology, J.X. and J.Y.; data curation, J.X. and W.S.; writing—original draft preparation, J.X. and Y.L.; writing—review and editing, Y.L. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xia, J., Yu, J., Shi, W. et al. Molybdenum facilitates PDLSC-based bone regeneration through the JAK/STAT3 signaling pathway. Sci Rep 15, 22204 (2025). https://doi.org/10.1038/s41598-025-07298-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07298-7
Keywords
This article is cited by
-
Current state of bioceramic bone repair materials in immune regulation: a review
Frontiers of Medicine (2025)