Abstract
Brain-specific angiogenesis inhibitor-1 (BAI1) is an endocytic scavenger receptor that mediates macrophage clearance of apoptotic or phosphatidylserine-expressing cells. Previous studies have shown that BAI1 was localized in the cellular cytoplasm and membrane. However, currently there is no available information regarding the distribution of BAI1 expression in alveolar macrophages (AMs) or its specific role in acute lung injury (ALI). The aim of this study was to preliminarily investigate BAI1 expression in AMs and changes in BAI1 expression levels in an ALI model. A mouse model of ALI was established through intratracheal instillation of a 2 mg/kg LPS. The expression of BAI1 in lung tissues was quantified using immunohistochemistry staining, while the expression of BAI1 in AMs in bronchoalveolar lavage fluid (BALF) was examined through immunofluorescence. Additionally, the MH-S cells were treated with LPS to investigate the distribution and changes in BAI1 expression levels, which were detected using immunofluorescence and Western Blot. Results showed that BAI1 protein was found to be localized in the cellular nuclei in AMs. Moreover, the BAI1 expression level was observed to escalate with the rising concentration of LPS in MH-S cells and in AMs of lungs from ALI mice. Finally, the upregulation of BAI1 expression in MH-S cells induced by LPS was associated with a decrease in the efferocytosis of MH-S cells. The discovery of BAI1 expression in the nuclei of AMs in ALI mice is a novel finding. It is plausible that BAI1 protein may participate in efferocytosis of AMs during ALI through novel signaling pathways.
Similar content being viewed by others
Introduction
In animals, ALI is a high-permeability pulmonary edema caused by various intra - and extra - pulmonary pathogenic factors other than cardiogenic factors, which then leads to acute, progressive, hypoxic respiratory failure1. In humans, it is defined as acute respiratory distress syndrome (ARDS)2. In recent multicenter prospective study carried out in 459 intensive care units (ICUs) in 50 countries in 5 continents, ARDS represented 10.4% of total admission to ICUs. The prevalence was 30.0% for mild ARDS, 46.6% for moderate ARDS, and 23.4% for severe ARDS, and overall, unadjusted ICU and hospital mortality was 35.3 and 40.0%, respectively and both augmented with increased ARDS severity3. ALI is a critical clinical illness characterized by neutrophil infiltration4. In the subsiding inflammatory stage of ALI, AMs directly recognize and engulf dying cells (particularly apoptotic neutrophils) to repair damaged tissues, a process called efferocytosis, which has profound consequences on the recovery of lung epithelial and endothelial functions and the reconstruction of lung tissue structure as apoptotic cells released many intracellular danger signaling molecules, the most fundamental of which is neutrophil extracellular traps (NETs), which further hinders tissue repair if they are not cleared in time5. ARDS patients have decreased alveolar macrophage efferocytosis, which is associated with increased alveolar inflammation, and may contribute to worse clinical outcomes, including mortality6. However, little is known about the molecular mechanisms underlying the decline in the efferocytosis function of AMs in ARDS.
These events require the exposure of phagocytic signals such as phosphatidylserine (PtdSer) on the apoptotic cell surface7. Brain-specific angiogenesis inhibitor-1 (BAI1), together with BAI2 and BAI3, forms the adhesion G protein-coupled receptors (aGPCRs) subfamily8. A characteristic feature of BAI1 is its ability to undergo autocleavage at the G protein–coupled receptor proteolytic site (GPS) and release a 120 kDa extracellular domain with angiogenic inhibitory activity9, which has led to an increasing interest in studies such as its ability to inhibit angiogenesis and tumor formation. Meanwhile, as a phosphatidylserine receptor, BAI1 binds apoptotic cell membranes and triggers activation of the apoptotic engulfment pathway by leading to the activation of the small GTPase Rac1 which is essential for actin remodeling and membrane trafficking during engulfment10. BAI1 has been proved to be a transmembrane protein highly expressed in central nervous system (CNS)11. BAI1 is localized to focal adhesions and show a speckled distribution in astrocytes and is involved in their clearance of apoptotic cells12. This prompted us to wonder if BAI1 is expressed on lung AMs? If so, how does its expression change in AMs of ALI mice and LPS-stimulate MH-S cells? What is the relationship between BAI1 and alveolar macrophage efferocytosis?
Since AMs play equally important functions in ALI, we sought to characterize the regional, cellular and subcellular expression of BAI1 in AMs of the mature mouse lung and in culture systems. Furthermore, the changes of BAI1 expression on AMs in ALI mice and LPS-stimulated MH-S cells were further investigated, hoping to reveal the preliminary relationship between BAI1 and lung injury and efferocytosis of AMs in ALI mice. Moreover, as resident macrophages of the CNS, BV2 cells (microglial cells) were selected because BAI1 was originally characterized as a brain-specific angiogenesis inhibitor11,13. By comparing BAI1 expression in BV2 (CNS macrophages) and MH-S, we aimed to test whether BAI1’s expression extends beyond the CNS to peripheral macrophages. Given that airway epithelium is a primary barrier in ALI pathogenesis14, 16HBE (human bronchial epithelial cells) served to determine if BAI1 expression is macrophage-specific or shared by structural lung cells.
Materials and methods
Anti-BAI1 antibody
We chose two primary antibodies: polyclonal rabbit antibody against extracellular epitope corresponding to amino acids 711–760 of human BAI1 (Thermo Fisher Scientific, cat#: PA5-75348) and polyclonal rabbit antibody against C-terminal (intracellular epitope, within residues 1500–1584, Novus biologicals, cat#: NB110-81586). Antibody NB110-81586 was used in Western blotting, and both of them were used in immunoreactivity analysis.
Cell lines and cultures
MH-S mouse alveolar macrophage cell line, Jurkat T-cell line (Human T-lymphocytic leukemia cells) and 16HBE cells (Human bronchial epithelial cells) were purchased from Procell Life Science and Technology Co., Ltd. (Hubei, Wuhan, China). BV2 cells (Mouse microglia) were purchased from American Type Culture Collection (Manassas, VA, ATCC). They were maintained in Roswell Park Memorial Institute Medium 1640 (Thermo Fisher Scientific, cat#: A1049101) supplemented with 10% fetal bovine serum (FBS, Thermo Fisher Scientific, cat#: 10099158), an antibiotics combination of 1% Penicillin-Streptomycin Liquid (Solarbio, cat#: P1400) and 0.05 mM β-mercaptoethanol (Solarbio, cat#: M8210), and incubated at 37℃ in a humidified atmosphere 5% CO2.
To detect BAI1, MH-S cells were collected at 24, 48 and 72 h after 1000 ng/mL lipopolysaccharide obtained from Escherichia coli O111:B4 (LPS, Millipore Sigma, cat#: L2630) treatment when grown to 70–80% confluence, or treated with 0 ng/mL, 10 ng/mL, 100 ng/mL and 1000 ng/mL LPS for 72 h.
Western blotting analysis
Cells were washed twice with 1× cold-PBS and then were lysed with RIPA lysis buffer containing 1× proteases/ phosphatase inhibitors cocktail (Solarbio, cat#: P6730, cat#: P1260). Cell lysates were incubated for 15 min on ice followed by breaking the DNA molecules with an ultrasonic crusher to reduce the viscosity of the sample. The lysates were then centrifuged in a cryogenic centrifuge at 14,000 rpm and 4 °C for 10 min. Proteins concentration was measured using Bioepitope® Bicinchoninic Acid Protein Assay Kit (Bioworld, cat#: BD0028). The cell lysates were diluted in 5× SDS-PAGE loading buffer (Solarbio, cat#: P1040) and RIPA Lysate (volume of lysate and protein sample: volume of 5× SDS-PAGE loading buffer = 4 to 1) and then boiled at 95 °C for 5 min for protein denaturation. 20 µg of protein sample was separated on 6% SDS-PAGE gels followed by transfer to Hydrophobic PVDF Transfer Membrane (Millipore Sigma, cat#: IPVH00010). Blotted membranes were blocked with QuickBlock™ Blocking Buffer for Western Blot (Beyotime, cat#: P0252), shaking at room temperature for 30 min, then incubated overnight at 4℃ with the following primary antibodies: Rabbit anti-BAI1 (diluted 1:1000, Novus biologicals, cat#: NB110-81586) and rabbit anti-vinculin (diluted 1:10000, Proteintech, cat#: 26520-1-AP). Following primary antibodies incubation, blotted membranes were washed with 1×TBST buffer for four times (each time for 5 min) and incubated at room temperature for 1 h with the following suitable secondary-HRP conjugated antibodies: Goat anti-rabbit IgG (H + P) HRP (diluted 1:5000, Bioss, cat#: bs-0295G-HRP). After washing with 1×TBST buffer four times, Immobilon Western HRP Substrate (Merck, cat#: WBKLS0100-1) was used to detect immunoreactivity of HRP. Exposures were performed using Amersham™ Imager 680 (Gene Ray Electric company, USA), and results were quantitatively analyzed using Image J software.
Immunofluorescence analysis
MH-S, 16HBE and BV2 cells were fixed with 4% paraformaldehyde (GenXion, cat#: JX0100). They were then blocked for 30 min with QuickBlock™ Blocking Buffer for Immunol Staining (Beyotime, cat#: P0260), and incubated overnight with Rabbit anti-BAI1 (diluted 1:200, Thermo Fisher Scientific, cat#: PA5-75348) and Mouse anti-alpha Tubulin (diluted 1:1000, Servicebio, cat#: GB15200). After washing with PBS, they were incubated for 1 h at RT with FITC conjugated Goat Anti-Rabbit IgG (H + L) (diluted 1:200, Servicebio, cat#: GB22303) and Cy3 conjugated Goat Anti-Mouse IgG (H + L) (diluted 1:500, Servicebio, cat#: GB21301). After nuclear staining with DAPI (Servicebio, cat#: G1012) and washing with PBS, the cells were checked under a confocal laser microscopy (SP8, Leica, Germany).
Determination of the efferocytosis of MH-S cells
Jurkat T cells were exposed to UV light at a wavelength of 254 nm for 30 min and then returned to the incubator for 4 h to allow spontaneous apoptosis to occur, and more than 80% of the cells were observed to undergo apoptosis under the microscope using Trypan blue staining. MH-S alveolar macrophages treated according to the Control and LPS (1000 ng/mL, 72 h) groups were inoculated onto cell crawls in 12-well cell culture plates. Apoptotic Jurkat T cells were labeled with PKH26 using PKH26 Red fluorescent cell membrane staining kit (Solarbio, cat#: D0030). Apoptotic Jurkat T cells were added to each well at a ratio of 10:1 (apoptotic Jurkat T cells: MH-S alveolar macrophages) in 600 µl of complete culture medium and co-cultured with MH-S alveolar macrophages for 4 h. They were then fixed with 4% paraformaldehyde, and fluorescence of unendocytosed apoptotic Jurkat T cells was quenched by incubating for 2 min with 1 ml of 0.04% Trypan Blue Stain Solution (Solarbio, cat#: C0040). MH-S cells were then blocked for 30 min with QuickBlock™ Blocking Buffer, and incubated overnight with Rabbit anti-BAI1 (diluted 1:200, Thermo Fisher Scientific, cat#: PA5-75348). After washing with PBS, they were incubated for 1 h at RT with FITC conjugated Goat Anti-Rabbit IgG (H + L) (diluted 1:200, Servicebio, cat#: GB22303). After nuclear staining with DAPI and washing with PBS, the cells were checked under a confocal laser microscopy (SP8, Leica, Germany).
Murine acute lung injury model
The 6–8-week-old SPF grade male BALB/c mice were obtained from Guangdong Medical Experimental Animal Center (license number SCXK [YUE] 2022-0002). They were housed individually in pathogen-free cages at the Experimental Animal Center of Guangzhou Medical University. The housing conditions were standardized with a room temperature of 20–26 °C, humidity of 40–70%, and a 12-hour light/dark cycle. The mice had free access to water and standard rodent chow throughout the protocols. The experimental protocol was approved by the Animal Care and Use Committee of Guangzhou Medical University. After one week of acclimatization, a total of 20 male BALB/c mice were randomly assigned to control (n = 10) and LPS-induced ALI groups (n = 10). Control mice received intratracheal instillation of sterile PBS (equal volume to LPS), while ALI groups received 2 mg/kg LPS dissolved in PBS, as described in other groups’ protocols15. All mice underwent intraperitoneal anesthesia with 3% sodium pentobarbital (30 mg/kg) prior to intratracheal procedures. Mice were euthanized 48 h post-instillation via cervical dislocation under deep anesthesia. Post-mortem, lungs were harvested for histopathology (H&E), immunohistochemistry, and immunofluorescence analysis; BALF was concurrently collected for alveolar macrophage isolation and BAI1 expression assessment. Ensure that the entire process complies with animal ethics regulations and minimizes the pain of the mice as much as possible.
Hematoxylin-eosin staining analysis
For histopathological assessment, mouse lungs were dissected and fixed in paraformaldehyde for 24 h. The lung tissues were then sectioned, embedded in paraffin, and cut into 4 μm sections. Subsequently, the sections underwent routine dewaxing using xylene I and xylene II solutions, followed by hydration with an ethanol concentration gradient. Hematoxylin and eosin staining (Servicebio, cat#: G1076) was performed to visualize nuclei and cytoplasmic structures in the sections. Bright-field images were acquired using a microscope (DM68, Leica, Germany).
Immunohistochemistry analysis
The lung tissue sections of mice were cut into 4 μm slices after being embedded in paraffin. After antigen retrieval and washing, endogenous peroxidase activity was blocked with a 3% H2O2 solution in methanol for 20 min at room temperature in the absence of light, followed by washing with 1×PBS. The sections were then incubated with QuickBlock™ Blocking Buffer for Immunol Staining (Beyotime, cat#: P0260) for 15 min and stained overnight at 4 ℃ with a primary antibody against BAI1 (diluted 1:200, Thermo Fisher Scientific, cat#: PA5-75348). Subsequently, an HRP-conjugated goat anti-Rabbit secondary antibody (diluted 1:400, Servicebio, cat#: GB27303) was added to incubate the slices for one hour at room temperature. Next, a color reaction was developed using diaminobenzidine (Servicebio, cat#: G1212), and nuclei were counterstained with hematoxylin (Servicebio, cat#: G1004).
Immunofluorescence analysis of BAI1 expression in AMs from BALF
The cells within the alveoli were isolated from BALB/c mice lungs by cannulating the trachea with a blunt 20-gauge needle and then lavaging the lungs ten times with 0.8 ml of chilled PBS/EDTA (5 mM). The AMs from BALF were collected in a 15 mL tube, immediately placed on ice, filtered through a 200-mesh sieve, and then centrifuged at 450 × g for 5 min. The resulting pellets were washed twice with cold PBS. Erythrocyte lysis buffer (1 mL, Solarbio, cat#: R1010) was added to the cell pellet to remove red blood cells, followed by another centrifugation at 450 × g for 5 min. Finally, the BALF cell pellets were resuspended in 1 mL of phosphate-buffered saline (PBS). The BALF cells were manually counted using a hemocytometer and used for subsequent immunofluorescence staining.
The BALF cells were fixed with 4% paraformaldehyde (GenXion, cat#: JX0100). They were then blocked for 30 min with QuickBlock™ Blocking Buffer for Immunol Staining (Beyotime, cat#: P0260), and incubated overnight with Rabbit anti-BAI1 (diluted 1:200, Thermo Fisher Scientific, cat#: PA5-75348) and Mouse anti-CD11c (diluted 1:200, Signalway Antibody, cat#: 48280). After being washed with PBS, they were incubated for 1 h at RT with FITC conjugated Goat Anti-Rabbit IgG (H + L) (diluted 1:200, Servicebio, cat#: GB22303) and Cy3 conjugated Goat Anti-Mouse IgG (H + L) (diluted 1:500, Servicebio, cat#: GB21301). After being washed with PBS, the cells were checked under a confocal laser microscopy (SP8, Leica, Germany).
Multiple immunofluorescence staining of paraffin sections
4 μm paraffin sections are dewaxed and rehydrated through an eco-friendly dewaxing solution and graded ethanol series, followed by antigen retrieval (citrate buffer, Servicebio, cat#: G1202) and PBS rinsing. After hydrophobic barrier drawing and blocking, three identical cycles of core staining are performed sequentially: (1) Serum blocking (rabbit serum or BSA selected per primary antibody source), (2) Primary antibody incubation at 4 °C overnight, (3) PBS rinse and HRP-conjugated secondary antibody (species-matched) incubation (50 min, RT), (4) PBS rinse and TSA incubation (10 min, RT, dark), (5) TBST rinse. After each cycle, microwave-mediated elution in citrate buffer removes antibody complexes (prevent drying), followed by repeated serum blocking. Post three cycles, sections undergo DAPI (Servicebio, cat#: G1012) nuclear counterstaining, PBS rinsing, autofluorescence quenching, running water wash, final PBS rinse, and mounting with anti-fade medium for fluorescence imaging. The primary antibodies used are Rabbit anti-BAI1 (diluted 1:200, Thermo Fisher Scientific, cat#: PA5-75348), Rabbit anti-CD11c (diluted 1:500, Servicebio, cat#: GB11059), and Rabbit anti-CD68 (diluted 1:500, Servicebio, cat#: GB113109), and immunofluorescence detection was per-formed using iF488-Tyramide (TSA, Servicebio, cat#: G1231), iF555-Tyramide (TSA, Servicebio, cat#: G1233) and iF647-Tyramide (TSA, Servicebio, cat#: G1232), respectively. Whole section imaging was carried using a slide scanner (PhenoImager Fusion, Akoya Biosciences, USA). Confocal microscopy was conducted using a Leica DLS microscope with a 40× oil immersion objective lens.
Statistical analysis
The statistical analysis was conducted using GraphPad Prism software version 9.0.0 (GraphPad Software, San Diego, CA). Comparisons between groups were analyzed using one-way ANOVA with Bonferroni posttest when there are more than 2 groups, as indicated in the figure legends. The independent samples T-Test is utilized to compare the difference in means between two separate sets of data. All experiments were repeated at least three times, and the data were presented as mean ± standard error (Mean ± SE). A P-value less than 0.05 was considered statistically significant.
Results
Constitutive expression characterization of BAI1 protein on MH-S cells, BV2 cells, 16HBE cells and lung tissue
BAI1 expression has been reported to be present in macrophages7,16. To examine whether BAI1 is expressed in AMs, we collected lysate from MH-S cells. Western blotting using antibody NB110-81586 revealed a single band between the 90–130 kDa range (Fig. 1A). It has been reported that BAI1 has many cleavage forms or isoforms17, which explains the possibility of observing this band. The antigenic target of antibody NB110-81586 is “C-terminal portion of the human BAI1 protein (within residues 1500–1584)”, which can only detect the C-terminus, thus excluding the possibility of detecting Vasculostatin-120, a cleaved form of BAI1 extracellular domain (corresponding to amino acid sequence 34–926 aa, which often appears as a 120 kDa band due to glycosylation and other post-translational modifications)9. Therefore, it is highly likely that the antibody detected a different isoform expressed in MH-S cells. Next, we further consider whether similar bands will occur in the lysates of mouse microglia and human airway epithelial cells. Using BV2 cells and 16HBE cells, we observed that a single band of approximately 110 kDa was also detected in the lysate of BV2 cells and a single band between the 55–65 kDa range was detected in the lysate of 16HBE cells (Fig. 1A). Additionally, tissue lysates from normal mouse brain showed a band around 165 kDa, which is extremely close to the full-length BAI1 protein with the expected 170 kDa size. Weaker bands were also observed, and some might be isoforms or non-specific signals. Nevertheless, no significant BAI1 expression was observed in normal mouse lung tissue (Fig. 1A).
BAI1 is located in the alveolar macrophage nucleus
BAI1 belongs to the adhesion G-protein-coupled receptors (GPCR), which are typical transmembrane proteins18. However, the results of our recent experiments are slightly different from previous reports in the literature12,16,19. We performed immunofluorescence staining on several cultured cell lines with antibody NB110-81586 and antibody PA5-75348, and found that their staining patterns were identical. MH-S cells, BV2 cells and 16HBE cells were positively stained for the anti-BAI1 antibody. Clusters of BAI1 protein were observed in the nuclei of MH-S cells under a confocal laser microscope (Fig. 1C), much of it is probably anchored to the nuclear membrane of the nucleus (Fig. 1B). And it was also localized in the nuclei of BV2 cells (Fig. 1C). It is worth further determining whether BAI1 is also localized in the nucleolus of the nucleus, since when antibody NB110-81586 was utilized, BAI1 was distributed in the nucleus in single or multiple spots (Fig. 1C). BAI1 was observed to be localized in the cytoplasm of 16HBE cells and distributed tightly around the nucleus and occasionally in the nuclei (Fig. 1C), and extremely few 16HBE cells stained positively with antibody PA5-75348.
BAI1 isoforms are expressed in alveolar macrophages, microglia, and bronchial epithelial cells and normal tissues. (A) Western blot analysis of the cell lysates and the tissue lysates. (B) Representative pictures taken with a Nikon-Structured Illumination Microscopy. Under high magnification (100× objective lens), a continuous and bright fluorescent signal is presented at the nuclear membrane, forming a ring around the nucleus with uniform fluorescence intensity. The relatively weak fluorescence intensity inside the nucleus contrasts with the strong fluorescence at the nuclear membrane. (C) The results of immunocytochemical staining of BAI1 with antibody PA5-75348 and antibody NB110-81586 show that BAI1 is expressed in the cytoplasm of 16HBE cells but in the nuclei of MH-S and BV2 cells. Data were obtained from at least three independent experiments.
LPS treatment induces the upregulation of BAI1 expression in MH-S cells cultured in vitro and BAI1 is highly expressed in the lungs of ALI mice
It has been reported that BAI1 is among several identified phosphatidylserine receptors that function in apoptotic cell engulfment7,10, and it has been demonstrated to mediate macrophage recognition and clearance of both apoptotic cells and Gram-negative bacteria10,16. AMs are important lung phagocytes and play an important role in lung natural immunity. In ALI, inflammatory conditions are accompanied by a reduced activity of efferocytosis in macrophages5. Therefore, after determining the presence of BAI1 expression in alveolar macrophages, we further investigated whether the expression level of BAI1 in AMs would be altered under simulated lung injury in vitro. MH-S cells were treated with 1000 ng/mL of LPS for 24 h, 48 h and 72 h or treated with 10 ng/mL, 100 ng/mL and 1000 ng/mL of LPS for 72 h. Western blotting using the antibody NB110-81586 demonstrated that BAI1 was upregulated in cultured MH-S cells after LPS stimulation for 24 h, 48 h and 72 h respectively in a time-dependent manner (Fig. 2A). Furthermore, after 72 h of LPS stimulation at concentrations of 10 ng/mL, 100 ng/mL, and 1000 ng/mL respectively, the BAI1 expression was significantly augmented with no dose-dependent effect (Fig. 2B).
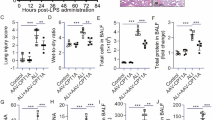
To determine the subcellular localization of BAI1 in the lung more definitively, we also performed immunohistochemistry and immunofluorescence on 4 μm paraffin sections. ALI mice displayed thickened alveolar septa, collapsed alveoli, and increased inflammatory cells infiltration within interstitial lung tissue and alveoli (Fig. 3A and B). A careful examination of the alveolar cavities and terminal bronchioles of adult mice revealed that BAI1 was mainly expressed in the nuclei of AMs (Fig. 3C, D and E), while its expression was relatively weak in the cytoplasm of airway epithelial cells (Fig. 3C). Moreover, immunohistochemical staining revealed a substantial upregulation of BAI1 expression on lung slices from ALI mice with exacerbated pulmonary damage, particularly on AMs compared to control mice lungs (Fig. 3D and E). And the immunofluorescence results show that alveolar macrophages with nuclear-expressed BAI1 accumulate in the lesion (Fig. 3F and G).
To understand the cell populations in BALF of ALI mice, fresh cells were collected and isolated from BALF and subjected to immunofluorescence analysis using a confocal microscope to assess BAI1 expression. We observed that the BAI1-expressing cell population in the alveolar lumen consisted mainly of alveolar macrophages (Fig. 4A and B).
Levels of BAI1 protein expression are elevated in LPS-treated MH-S cells using western blot. (A) MH-S cells were stimulated with LPS (1000 ng/mL) for 0, 24, 48, and 72 h. (B) MH-S cells were stimulated with different concentrations of LPS (0 ng/mL, 10 ng/mL, 100 ng/mL, 1000 ng/mL) for 72 h. Western blot analysis was performed to detect the total protein levels of BAI1. A significant increase in BAI1 (~ 110 kDa) was observed at 72 h after LPS (10 ng/mL) stimulation. The quantitative analysis of BAI1 expression were presented as Mean ± SE from three independent experiments (ns: not significant, * P < 0.05, ** P < 0.01, *** P < 0.001).
BAI1 is immunoreactive in adult mouse lungs and elevated in ALI mouse lungs. (A, B) Representative photomicrographs of lung tissues stained with hematoxylin and eosin (H&E) were obtained, and a large inflammatory cell infiltrate was observed in the lung of ALI mice. (C, D, E) Immunohistochemistry on adjacent paraffin sections using the antibody PA5-75348. In the lungs of normal adult mice, BAI1 was mainly expressed in the nuclei of AMs, but was relatively weak in the cytoplasm of airway epithelial cells. Moreover, BAI1 expression increased with the elevation of AMs in the lung tissues of mice with ALI. (F, G) Immunofluorescence staining of lung tissues showed BAI1 immunoreactivity mainly originated from a small number of AMs in the alveolar cavity. Scale bars: A, B, C, D, E, F = 100 μm.
The BAI1-expressing cell population in the alveolar lumen consisted mainly of AMs (A, B). Cells in BALF were separated from each group of mice for immunofluorescence staining, and the expression of BAI1 was visualized using laser confocal microscopy. Blue fluorescence corresponds to DAPI-stained nuclei; Red fluorescence represents CD11c stained with anti-CD11c antibody, marking AMs; Green fluorescence represents BAI1 stained with antibody PA5-75348. Merge: Overlap of three fluorescence spectra at the same position. Scale bars: A, B = 50 μm.
LPS treatment led to a decrease in the efferocytosis ability of MH-S cells
To examine impacts of LPS on MH-S cells efferocytosis, MH-S cells were treated with LPS (1000 ng/mL) for 72 h, and then cocultured with apoptotic Jurkat T-cell labeled with PKH26 to determine efferocytosis. Confocal microscopy with z-sectioning showed that the debris (identified by PKH26 staining) and apoptotic nuclear debris (identified by condensed DAPI staining) were within the confines of the cytoplasm of BAI1-positive macrophages (Fig. 5A and B). Compared with the control group, LPS treatment significantly reduced the percentage of ingested apoptotic cells assessed by immunofluorescence analysis (Fig. 5C).
LPS inhibits phagocytosis of apoptotic cells by MH-S cells. (A) BAI1-positive MH-S cell that had engulfed apoptotic nuclear and cytoplasmic debris were observed after co-culture with a large number of apoptotic cells for 4 h. Confocal microscopy with z-sectioning showed the DAPI-positive nuclear debris was located within the cytoplasm of macrophages (as indicated by the arrow). (B) LPS-treated MH-S cells engulfed very few apoptotic cells. T-PMT is an open field. The scale bar is 100 μm. All laser confocal microscope photos were taken using a 40× objective lens. (C) Efferocytosis index of MH-S cells under confocal microscopy. The results are presented as Mean ± SE. N = 3, **** P < 0.0001.
Discussion
Our present results showed that MH-S cells and AMs from LPS-induced ALI mice expressed BAI1 or its isoforms. Moreover, LPS treatment promoted its expression while accompanied by a decline in efferocytosis function. Additionally, immunohistochemical and immunofluorescence labeling experiments in mouse lung and cultured MH-S cells revealed that BAI1 was expressed in the nuclei of AMs, which has implications for its likely regulatory functions. These new findings regarding AMs suggest a potentially unique role for BAI1 within these cells.
The development of ALI is triggered by excessive and unregulated systemic inflammatory responses to direct or indirect lung damage. AMs play a crucial role in the pathogenesis of ALI, serving as the first line of defense against pathogenic microorganisms and lung injury20,21. BAI1 is a phosphatidylserine recognition receptor that can directly mediate the phagocytosis of apoptotic cells by macrophages10 and a pattern recognition receptor that mediates the nonopsonic phagocytosis of Gram-negative bacteria by macrophages16,22. It is important to determine whether AMs express BAI1 for further analysis of its function in ALI.
Firstly, we collected lysate from MH-S cells and performed western blotting studies using rabbit polyclonal anti-BAI1 antibody (NB110-81586), as well as in other different cells and tissues for comparison. Our results revealed a non-full-length BAI1 that migrates at an apparent size of 90–130 kDa on immunoblots, which is different from previously known proteolytic events that can occur in the BAI1 protein9,23: cleavage at a site between TSR1 and TSR2 by matrix metalloproteinase 14 (MMP14) yields an anti-angiogenic N-terminal fragment (NTF) of BAI1 of ~ 40 kDa containing the RGD motif and the first TSR, and a remaining truncated membrane receptor (predicted size of 138.8 kDa) migrating at ~ 160 kDa23, and autoproteolytic cleavage at the GPS site generates a low abundance 120 kDa fragment (Vasculostatin-120) containing all five TSRs, which leaves a membrane-bound CTF containing 7TM domain and C-terminal domain (predicted size of 72.2/71.8 kDa) migrating at ~ 75 kDa9. Tissue lysates from the mouse brain showed a band of the size of about 165 kDa, which is very close to the theoretical molecular weight of 170 kDa. This can help us rule out the effects of experimental manipulation as well as excessive protein binding and electrical loading in SDS-PAGE. A new undiscovered cleavage site of BAI1 protein in AMs may explain this event, or we can also explain it in terms of transcriptional isoforms. The BAI1 gene, containing thirty exons and at least one functional intron with a p53 binding site13, is characterized by the generation of multiple heterogeneous mRNA transcripts and translations17. The BAI1 isoforms being produced arise from the combination of multiple transcription initiation sites and alternate splicing, these different mRNAs are translated to distinct local isoforms17. We believe further experiments are needed to determine if the single band we observed is a new isoform.
Through immunohistochemistry and immunofluorescence studies on paraffin sections, we also observed that BAI1 was localized in the nuclei of AMs. And we carried out immunofluorescence staining on cultured MH-S cell lines with two antibodies against different antigenic determinants and found that their staining patterns were identical. The nuclear localization of BAI1 in AMs may be due to the presence of nuclear localization sequences (NLS)24, which makes nuclear localization of BAI1 theoretically possible. The nuclear G-protein-coupled receptors (nGPCRs), which have been identified as having important biological implications and are gradually emerging as prospective new therapeutic targets due to their role in regulating gene transcription in physiology and disease25. It is currently believed that nGPCRs are generated from two transporter events26,27: mechanisms of cell surface-derived nGPCRs translocation rely on NLS to mediate action, and mechanisms of nGPCRs transport from non-cell surface sources refers to the transport of this fraction of nGPCRs directly to the nucleus after synthesis. It is currently hypothesised that such nGPCRs may be synthesised to diffuse rapidly along the outer nuclear membrane to the nuclear pore complex before crossing the peripheral channel between the nuclear pore complex and the nuclear membrane28. Future research could employ a combination of techniques like proteomics identifying the proteins that interact with the NLS of BAI1 in the nucleus could provide insights into its nuclear transporter mechanisms. The presence of NLS in BAI1 indicates that the intracellular domains (ICDs) of BAI1 may potentially act as regulatory elements of gene expression in the nucleus. For example, it has been reported that the ICDs of BAI1 interact with the PAF1 complex, which is involved in RNA polymerase II transcription elongation and transcription-coupled histone modifications24. Further investigation of transcriptional changes associated with BAI1 expression in AMs could help to elucidate its role in regulating the immune response in ALI.
Furthermore, we investigated the changes in BAI1 expression on cultured LPS-treated MH-S cells and AMs in mice with LPS-induced ALI. Our western blotting analysis of the lysate of the MH-S cells with antibody NB110-81586 demonstrated an upregulation in BAI1 expression in MH-S cells treated with LPS, and immunohistochemistry and immunofluorescence analysis revealed that BAI1 was highly expressed in the lungs of ALI mice. These findings indicate that BAI1 may be involved in the pathophysiological process of ALI. The upregulation of BAI1 expression could potentially be a response to LPS-induced inflammation or a compensatory mechanism in the context of lung injury. Further studies are needed to elucidate the exact mechanisms underlying this increased expression and its significance in ALI. Combined with our observation that BAI1 is localized in the nuclei of AMs, further investigation of transcriptional changes associated with BAI1 expression in AMs could help to clarify its role in regulating the immune response in ALI.
Finally, we observed a decrease in the clearance ability of apoptotic cells by LPS-treated AMs. This phenomenon may be related to the changes in BAI1 expression. It is possible that the altered expression or localization of BAI1 in AMs after LPS treatment disrupts the normal phagocytic function of these cells. For instance, the increased expression of BAI1 could lead to the activation of other inflammatory pathways that indirectly affect the phagocytic ability of AMs. Further investigations are required to determine the exact relationship between BAI1 and the impaired clearance of apoptotic cells in LPS-treated AMs.
Additionally, the same isoforms and nuclear localization were also observed in microglia, a finding that was not mentioned in previous studies11,12,29. Bronchial epithelial cells also express BAI1. Moreover, there may be autoproteolytic cleavage of BAI1 since we observed a single band in the 55–65 kDa range in the lysate of 16HBE cells. It would be intriguing to investigate whether the isoforms and nuclear localization of BAI1 are specific to AMs or if they are also seen in other cell types.
In conclusion, our study has provided new insights into the expression and role of BAI1 in AMs. The abnormal molecular weight and nuclear localization of BAI1, as well as the increased expression in response to LPS and its potential association with the decreased clearance of apoptotic cells, suggest that BAI1 may play a crucial role in the pathogenesis of ALI. Future studies should concentrate on clarifying the detailed mechanisms through which BAI1 affects the function of AMs and the development of ALI, which may lead to the development of novel therapeutic strategies for this inflammatory disease.
Data availability
All data generated or analyzed during this study are included in this published article.
References
Matute-Bello, G. et al. An official American thoracic society workshop report: Features and measurements of experimental acute lung injury in animals. Am. J. Respir Cell. Mol. Biol. 44, 725–738. https://doi.org/10.1165/rcmb.2009-0210ST (2011).
Thille, A. W. et al. Comparison of the Berlin definition for acute respiratory distress syndrome with autopsy. Am. J. Respir Crit. Care Med. 187, 761–767. https://doi.org/10.1164/rccm.201211-1981OC (2013).
Bellani, G. et al. Patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. Jama 315, 788–800. https://doi.org/10.1001/jama.2016.0291 (2016). Epidemiology.
Wang, J. F. et al. Upregulated PD-L1 delays human neutrophil apoptosis and promotes lung injury in an experimental mouse model of sepsis. Blood 138, 806–810. https://doi.org/10.1182/blood.2020009417 (2021).
Grégoire, M. et al. Impaired efferocytosis and neutrophil extracellular trap clearance by macrophages in ARDS. Eur. Respir. J. 52, 1702590. https://doi.org/10.1183/13993003.02590-2017 (2018).
Mahida, R. Y. et al. Acute respiratory distress syndrome is associated with impaired alveolar macrophage efferocytosis. Eur. Respir. J. 58 https://doi.org/10.1183/13993003.00829-2021 (2021).
Lemke, G. How macrophages deal with death. Nat. Rev. Immunol. 19, 539–549. https://doi.org/10.1038/s41577-019-0167-y (2019).
Stephenson, J. R., Purcell, R. H. & Hall, R. A. The BAI subfamily of adhesion gpcrs: Synaptic regulation and beyond. Trends Pharmacol. Sci. 35, 208–215. https://doi.org/10.1016/j.tips.2014.02.002 (2014).
Kaur, B., Brat, D. J., Devi, N. S. & Van Meir, E. G. Vasculostatin, a proteolytic fragment of brain angiogenesis inhibitor 1, is an antiangiogenic and antitumorigenic factor. Oncogene 24, 3632–3642. https://doi.org/10.1038/sj.onc.1208317 (2005).
Park, D. et al. BAI1 is an engulfment receptor for apoptotic cells upstream of the ELMO/Dock180/Rac module. Nature 450, 430–434. https://doi.org/10.1038/nature06329 (2007).
Mori, K. et al. Brain-specific angiogenesis inhibitor 1 (BAI1) is expressed in human cerebral neuronal cells. Neurosci. Res. 43, 69–74. https://doi.org/10.1016/s0168-0102(02)00018-4 (2002).
Sokolowski, J. D. et al. Brain-specific angiogenesis inhibitor-1 expression in astrocytes and neurons: Implications for its dual function as an apoptotic engulfment receptor. Brain. Behav. Immun. 25, 915–921. https://doi.org/10.1016/j.bbi.2010.09.021 (2011).
Nishimori, H. et al. A novel brain-specific p53-target gene, BAI1, containing thrombospondin type 1 repeats inhibits experimental angiogenesis. Oncogene 15, 2145–2150. https://doi.org/10.1038/sj.onc.1201542 (1997).
Bos, L. D. J. & Ware, L. B. Acute respiratory distress syndrome: Causes, pathophysiology, and phenotypes. Lancet 400, 1145–1156. https://doi.org/10.1016/s0140-6736(22)01485-4 (2022).
Ehrentraut, H., Weisheit, C. K., Frede, S. & Hilbert, T. Inducing acute lung injury in mice by direct intratracheal lipopolysaccharide instillation. J. Visual. Exp. JoVE. https://doi.org/10.3791/59999 (2019).
Das, S. et al. Brain angiogenesis inhibitor 1 (BAI1) is a pattern recognition receptor that mediates macrophage binding and engulfment of Gram-negative bacteria. Proc. Natl. Acad. Sci. U.S.A. 108, 2136–2141. https://doi.org/10.1073/pnas.1014775108 (2011).
Parag, R. R. et al. Novel isoforms of adhesion G Protein-Coupled receptor B1 (ADGRB1/BAI1) generated from an alternative promoter in intron 17. Mol. Neurobiol. https://doi.org/10.1007/s12035-024-04293-3 (2024).
Kishore, A., Purcell, R. H., Nassiri-Toosi, Z. & Hall, R. A. Stalk-dependent and Stalk-independent signaling by the adhesion G Protein-coupled receptors GPR56 (ADGRG1) and BAI1 (ADGRB1). J. Biol. Chem. 291, 3385–3394. https://doi.org/10.1074/jbc.M115.689349 (2016).
Hsiao, C. C., van der Poel, M., van Ham, T. J. & Hamann, J. Macrophages do not express the phagocytic receptor BAI1/ADGRB1. Front. Immunol. 10 https://doi.org/10.3389/fimmu.2019.00962 (2019).
Huang, X., Xiu, H., Zhang, S. & Zhang, G. The role of macrophages in the pathogenesis of ALI/ARDS. Mediat. Inflamm. 2018 (1264913). https://doi.org/10.1155/2018/1264913 (2018).
Korkmaz, F. T. & Traber, K. E. Innate immune responses in pneumonia. Pneumonia (Nathan Qld). 15, 4. https://doi.org/10.1186/s41479-023-00106-8 (2023).
Billings, E. A. et al. The adhesion GPCR BAI1 mediates macrophage ROS production and microbicidal activity against Gram-negative bacteria. Sci. Signal. 9, ra14. https://doi.org/10.1126/scisignal.aac6250 (2016).
Cork, S. M. et al. A proprotein convertase/MMP-14 proteolytic cascade releases a novel 40 kda vasculostatin from tumor suppressor BAI1. Oncogene 31, 5144–5152. https://doi.org/10.1038/onc.2012.1 (2012).
Knapp, B. et al. Affinity proteomics identifies novel functional modules related to adhesion GPCRs. Ann. N. Y. Acad. Sci. 1456, 144–167. https://doi.org/10.1111/nyas.14220 (2019).
Gonçalves-Monteiro, S. et al. Insights into nuclear G-Protein-Coupled receptors as therapeutic targets in Non-Communicable diseases. Pharmaceuticals 14 https://doi.org/10.3390/ph14050439 (2021).
Gobeil, F. et al. G-protein-coupled receptors signalling at the cell nucleus: An emerging paradigmthis paper is one of a selection of papers published in this special issue, entitled the nucleus: A cell within A cell. Can. J. Physiol. Pharmacol. 84, 287–297. https://doi.org/10.1139/y05-127 (2006).
Joyal, J. S., Bhosle, V. K. & Chemtob, S. Subcellular G-protein coupled receptor signaling hints at greater therapeutic selectivity. Expert Opin. Ther. Targets. 19, 717–721. https://doi.org/10.1517/14728222.2015.1042365 (2015).
Zuleger, N., Kerr, A. R. W. & Schirmer, E. C. Many mechanisms, one entrance: Membrane protein translocation into the nucleus. Cell. Mol. Life Sci. 69, 2205–2216. https://doi.org/10.1007/s00018-012-0929-1 (2012).
Mazaheri, F. et al. Distinct roles for BAI1 and TIM-4 in the engulfment of dying neurons by microglia. Nat. Commun. 5, 4046. https://doi.org/10.1038/ncomms5046 (2014).
Acknowledgements
Not applicable.
Funding
This work was supported by the 2023 Guangdong Provincial Department of Education Featured Innovation Projects for General Higher Education Institutions in Guangdong Province (No. 2023KTSCX105), the Research Capacity Enhancement Project of Guangzhou Medical University in 2023, the Student Innovation Capacity Enhancement Program Project of Guangzhou Medical University in 2022, the Special Funds for Science and Technology Innovation Strategy of Guangdong Province (pdjh2021b0411) and the Natural Science Foundation of Guangdong Province (2018A0303130256).
Author information
Authors and Affiliations
Contributions
All authors participated in the design, interpretation of the studies and analysis of the data and review of the manuscript; A.L.: Conducing the experiments, participation in data analysis and interpretation, drafting the manuscript and critical revision of the manuscript. X.H. and K.Z.: Conducing the experiments, participation in data analysis and interpretation. P. X. and H.W.: Conception of the work, participation in data analysis and interpretation. Y.Y. and Y.T.: Conception of the work, participation in data analysis and interpretation, critical revision of the manuscript and final approval of the version to be published.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declarations
All animal experiments were carried out in accordance with the Regulations on the Administration of Laboratory Animals and Guiding Opinions on the Kind Treatment of Laboratory Animals. This study was approved by Guangzhou Medical University Laboratory Animal Centre (No. GY2023-541). This study is reported in accordance with the ARRIVE guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, AG., Huang, XY., Zhang, KQ. et al. Nuclear localization and upregulation of BAI1 in alveolar macrophages during LPS-Induced acute lung injury. Sci Rep 15, 24574 (2025). https://doi.org/10.1038/s41598-025-08990-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-08990-4