Abstract
Neutrophils are known to be able to recruit von Willebrand factor (VWF) through Neutrophil Extracellular Traps (NETs) during inflammation. However, the interaction between neutrophils with plasma VWF at physiological condition remains poorly understood. In this study, we demonstrate that peripheral neutrophils constitutively carry surface-bound VWF in a NETs-independent manner. Depletion of peripheral neutrophils in mice elevated plasma VWF levels without affecting its synthesis or secretion. A similar interaction in human blood was also observed as demonstrated from the analysis of 23 healthy human donors. Human VWF binding to neutrophils is independent of gender and age; however, individuals with non-O blood types exhibit significantly lower VWF levels on neutrophils compared to those with type O blood. Furthermore, we found that the isolated human neutrophils bind stronger to the neuraminidase-treated VWF than untreated VWF. PSGL-1 and β2-integrins, two neutrophil membrane receptors, seemingly mediate this interaction since antibodies targeting both receptors showed a significant attenuating role here. These findings suggest a constitutive interaction between neutrophils and VWF in peripheral blood, in which the desialylation VWF seems play a major role here. Both PSGL-1 and β2-integrins receptors on neutrophil contribute to this interaction. Our study reveals a function of neutrophils beyond their established antimicrobial roles.
Similar content being viewed by others
Introduction
Neutrophils are highly effective immune cells that circulate alongside other leukocytes in the blood1. They are the most abundant immune cells, with counts ranging from 1,500 to 7,000/µL in adults, constituting 60–70% of the white blood cell population2,3,4. Alterations in neutrophil count, known clinically as neutropenia (low count) or neutrophilia (high count), can lead to opportunistic infections or excessive immune responses5,6. Neutrophils typically have a lifespan of 12 to 14 h in peripheral blood, with constant replenishment from the bone marrow7,8. Increasing evidence suggests that neutrophils play a significant role in thrombosis development9,10. Inflammatory responses can lead to a hypercoagulable state known as immunothrombosis, driven by the crosstalk between neutrophils and coagulation elements10. Neutrophils interact with platelets through P-selectin glycoprotein ligand-1 (PSGL-1) binding to P-selectin on platelets, forming neutrophil-platelet aggregates that are crucial in arterial thrombosis, such as myocardial infarction and stroke11,12. In venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), large quantities of neutrophils and neutrophil extracellular traps (NETs) are found within thrombi13,14. These NETs, composed of DNA, histones, and antimicrobial proteins, provoke the coagulation cascade by interacting with tissue factor and von Willebrand Factor (VWF)14.
VWF is a plasma glycoprotein produced exclusively by endothelial cells and megakaryocytes15,16. In normal biophysical blood circulation, VWF remains inactive until elevated shear forces trigger its role in inducing platelet adhesion and aggregation17. Due to its critical role in hemostasis, VWF plasma levels are clinically indicative of bleeding or thrombotic risks18. For example, reduced VWF levels, as seen in von Willebrand Disease or autoimmune conditions like systemic lupus erythematosus, lead to an increased risk of bleeding19,20,21. Conversely, elevated VWF levels, often observed in patients with cancer and COVID-19, are associated with an increased risk of thromboembolism22,23. Beyond platelets, VWF can also interact with endothelial cells, monocytes, dendritic cells, and certain cancer cells through its distinct domains24,25,26. Additionally, VWF desialylation—a natural glycan modification that serves as a crucial marker for the clearance of aged proteins—plays a key role in its recognition by macrophages and other scavenger cells27. The interaction between neutrophils and VWF, as aforementioned, is implicated in immune responses, inflammation, and thrombosis. According to recent study, neutrophils and VWF can also interact under static conditions, where VWF serves as an adhesive surface for neutrophils28. But the nature of the neutrophil-VWF interaction under non-inflammatory conditions remains poorly understood compared to their pathological interactions.
In this study, we aim to investigate the nature of the neutrophil-VWF interaction under non-inflammatory conditions. The findings will contribute to a broader understanding of the role of neutrophils beyond their involvement in inflammatory disorders.
Materials and methods
KEY material TABLE.
Reagent or resource | Source | Identifier |
|---|---|---|
Antibodies | ||
PE conjugated anti-mouse CD45 antibody | Invitrogen | Cat #12-0451-82 |
PE/Cyanine7 conjugated anti-mouse Ly6G antibody | Biolegend | Cat #127618 |
BV421 conjugated anti-human CD45 antibody | Invitrogen | Cat #404-0459-42 |
Rat anti-mouse Ly6G antibody | Bio X cell | Cat #BE0075-1 |
Rabbit anti-mouse MPO antibody | ABclonal | Cat #A24531 |
Rabbit anti-mouse citrulline-Histone H3 antibody | ABclonal | Cat #A18298 |
Alexa Fluor 488 labeled goat anti-rabbit IgG | Abcam | Cat #ab150077 |
Alexa Fluor 647 labeled goat anti-rabbit IgG | Abcam | Cat #ab150079 |
DyLight 800 4X PEG conjugate goat anti-rabbit IgG | CST | Cat #5151 |
Mouse anti-human CD16 antibody | Proteintech | Cat #PE-65090 |
Mouse anti-human β2-integrin antibody | Proteintech | Cat #65190-1-lg |
Mouse anti-human PSGL-1 antibody | SinoBiological | Cat #13863-MM07 |
Rabbit anti-human VWF antibody | Custom from Genscript | N/A |
Rabbit anti-mouse VWFpp antibody | Custom from Genscript | N/A |
Chemicals, peptides, and recombinant proteins | ||
α2–3,6,8,9 Neuraminidase | Hoffmann-La Roche AG | Cat #P0722L |
4′,6-diamidino- 2-phenylindole | Beyotime | Cat #C1005 |
Cell lysis buffer | Beyotime | Cat #P0013 |
Heparin | Biofer SpA | Cat #9041-08-1 |
Phorbol 12-myristate 13-acetate | MedchemExpress | Cat #16561-29-8 |
TRIzol | ThermoFisher | Cat #15596026CN |
Power SYBR green PCR master mix | ThermoFisher | Cat #4309155 |
Oligonucleotides | ||
Primer: Actin; Forward: GTGCTATGTTGCTCTAGACTTCG; Reverse: ATGCCACAGGATTCCATACC; | Genevize | N/A |
Primer: Asgr-1; Forward: GTGGAGTATGAAGGCAGCTGCT; Reverse: CGTCAGACACTAACTGCTTCACG; | Genevize | N/A |
Primer: Asgr-2; Forward: GCTCTTTCACCTGAAGCACTTCC Reverse: CCCGAGAAAACCAGTAGCAGCT; | Genevize | N/A |
Primer: Mgl-1; Forward: GAGACTTGAGCCAGAAGGTGA; Reverse: TCCGAGCCGTTGTTCTTGAGG; | Genevize | N/A |
Primer: Mgl-2; Forward: GAGCATGAAGGCAGCTGCTAT; Reverse: TTGGCTAAGCGATTCTGTAGA; | Genevize | N/A |
Primer: Lrp1; Forward: CGAGAGCCTTTGTGCTGGATGA; Reverse: CGGATGTCCTTCTCAATGAGGG; | Genevize | N/A |
Primer: Sra1; Forward: CGCACGTTCAATGACAGCATCC; Reverse: GCAAACACAAGGAGGTAGAGAGC; | Genevize | N/A |
Primer: Stab2; Forward: ATCCAGATGGCAAAGCCTCGTG; Reverse: ATCCAGATGGCAAAGCCTCGTG; | Genevize | N/A |
Primer: vWF; Forward: CTTCTGTACGCCTCAGCTATG; Reverse: GCCGTTGTAATTCCCACACAAG; | Genevize | N/A |
Software and algorithms | ||
GraphPad PRISM Version 7 | GraphPad Software | RRID: SCR_002798 |
FlowJo_v10.8.1 | BD | RRID: SCR_008520 |
Other | ||
HiPrep 26/60 Sephacryl S-400 h | Cytiva | Cat #28935605 |
Animals
Experiments involving mice were performed in accordance with experimental protocols approved by the Institutional Animal Care and Use Committee of Soochow University, based on the recommendations of the US National Institutes of Health and the ARRIVE guidelines (https://arriveguidelines.org). C57BL/6 N mice were used as the wildtype mice. The transgenic GFP-VWF knock-in mice have been characterized previously. Both female and male mice, aged 6 to 8 weeks, from the homozygous lineage were used in the experiments. The mice were fed standard rodent chow and water ad libitum and were maintained under climate-controlled conditions in a 12-hour light/dark cycle in a pathogen-free facility.
Human subjects
Human plasma derived VWF (Pd-VWF) was purified to homogeneity according to the published protocol29. Citrated whole blood was collected from healthy volunteers following an approved protocol from the Ethics Committee of Soochow University, confirming that informed consent was obtained from all participants and/or their legal guardians. The research involving human participants was performed in accordance with the Declaration of Helsinki and adhered to the ethical standards of the institution.
Isolation of leukocytes from peripheral blood
Murine blood was collected from the abdominal aorta of mice using sodium citrate as an anticoagulant. Human peripheral blood samples were obtained from healthy adult volunteers, drawn by venipuncture into sodium citrate tubes. Samples were processed within 0.5 h to maintain cell viability. Whole blood samples (both murine and human) were diluted 1:1 with sterile phosphate-buffered saline (PBS) and gently layered over a density gradient medium (e.g., Histopaque-1077 for human samples and Histopaque-1083 for murine samples) in a 1:1 ratio. Tubes were centrifuged at 400 × g for 30 min at room temperature without braking to allow optimal separation of blood cell layers. After centrifugation, the mononuclear cell layer was carefully collected using a pipette. Cells were transferred to fresh tubes, washed twice with PBS by centrifuging at 300 × g for 5 min, and resuspended in FACS buffer (PBS with 2% fetal bovine serum and 1 mM EDTA). Any residual red blood cells in the mononuclear cell suspension were removed using an ammonium chloride-based lysis buffer. The purity of the isolated leukocytes was verified by flow cytometry. Murine leukocytes were stained with anti-CD45 and anti-Ly6G antibodies, while human leukocytes were stained with anti-CD45. A minimum of 90% purity for leukocytes was confirmed before downstream analysis.
Neutrophil sorting from isolated leukocytes
Leukocytes were aliquoted into 500 µL samples at a concentration of approximately 1 × 10^6 cells per 100 µL in FACS buffer. For murine samples, cells were incubated with fluorescently conjugated anti-Ly6G and anti-CD45 antibodies, specific to neutrophil populations. For human samples, anti-CD45 and anti-CD16 antibodies were used. Stained cells were sorted on a BD FACSAria equipped with appropriate lasers for the selected fluorophores. Forward and side scatter gates were set to exclude debris and doublets. Neutrophils were identified based on their size and granularity, confirmed by Ly6G (mouse) and CD16 (human) gating. Sorting purity was set to ≥ 95%, and a minimum of 1 × 10^6 neutrophils was sorted per sample to ensure sufficient yield for downstream assays.
Laser scanning confocal fluorescence imaging (LSCFI) analysis
LSCFI analysis was performed as previously described30. Briefly, tissue isolated from GFP-VWF mice was fixed immediately in 4% paraformaldehyde at 4 °C overnight, followed by decalcification in 500 mM EDTA (pH 7.2–7.4) for one week at room temperature. Following decalcification, samples were dehydrated in 20% sucrose solution for 24 h. The tissues were then embedded in Tissue-Tek O.C.T., frozen at -20 °C, and sectioned at 8 μm thickness onto adhesion-coated microscopy slides. After washing and blocking, sections were incubated with an anti-Ly6G primary antibody at a 1:200 dilution, followed by incubation with an Alexa Fluor 647-conjugated secondary antibody at a 1:500 dilution. Nuclei were stained with DAPI. Images were acquired using an Olympus IX-81 confocal microscope.
Neutrophil depletion
Neutrophils in peripheral blood were depleted using an antibody-mediated clearance method following a published protocol31. Briefly, an anti-mouse Ly6G antibody (0.5 mg/mL in 1× PBS) was administered via tail vein injection at a dose of 250 µg/kg. Mouse IgG at the same dose was used as a control. Neutrophil depletion was confirmed by analyzing the neutrophil population in peripheral blood using flow cytometry.
VWF desialylation
α2–3,6,8,9 neuraminidase was utilized to remove sialic acid from the glycosylation profile of VWF. A total of 2.85 mg of purified human plasma-derived VWF was treated with 15 mU of α2–3,6,8,9 neuraminidase for 45 min at 37ºC. The desialylation of VWF was confirmed by a band shift on SDS-PAGE. Desialylated VWF was then diluted to various concentrations and incubated with freshly isolated neutrophils at room temperature for 3 min. The samples were immediately fixed with 4% PFA before flow cytometry analysis. Untreated VWF at the same concentrations were processed in parallel as a control. The effect of trace amounts of neuraminidase in the final samples was considered negligible.
Detection of neutrophil extracellular traps (NETs)
The NETs signal in the isolated leukocyte samples was detected using flow cytometry. For the positive control, cells stimulated with 100 nM phorbol 12-myristate 13-acetate (PMA) for 3 h at 37 °C in a 5% CO2 incubator to induce NETs formation. Unstimulated cells were incubated under the same condition. Samples were incubated with anti-citrullinated histone H3 (Cit-H3) and anti-myeloperoxidase (MPO) primary antibodies, which are markers specific to NETs, at dilutions of 1:200 and 1:100, respectively, for 30 min on ice. After incubation, cells were washed with PBS and incubated with secondary antibodies conjugated to fluorescent dyes (e.g., Alexa Fluor 488 or 647) at a 1:500 dilution for 30 min on ice in the dark. Samples were then analyzed on a flow cytometer and isotype control antibodies were included to verify specificity. NETs formation was quantified by comparing the percentage of Cit-H3+ or MPO+ cells between stimulated and unstimulated samples.
Flow cytometry (FCM)
The FCM analysis of murine and human peripheral blood samples was performed on a flow cytometer (BD FACS Canto II) equipped with lasers suitable for the selected fluorophores. For neutrophil surface VWF analysis, isolated leukocytes were aliquoted into 100 µL samples at a concentration of approximately 1 × 10^6 cells per sample. Cells were incubated with the following fluorescently conjugated antibodies: mouse anti-CD45, anti-Ly6G, and anti-VWF, and for human samples, anti-CD45, anti-CD16, and anti-VWF. All antibodies were used at optimized concentrations as per the manufacturer’s recommendations. Samples were incubated at 4 °C for 30 min in the dark, followed by washing twice with FACS buffer to remove unbound antibodies. Forward and side scatter gates were set to exclude debris and doublets. A minimum of 10,000 neutrophils was recorded per sample. Data were analyzed using FlowJo software (version 10.8.1). Median fluorescence intensity (MFI) was used to quantify VWF signals on neutrophils, and gating strategies were standardized across all samples.
Multimeric VWF analysis
Multimeric VWF was analyzed using a 1.7% agarose gel (1.7% agarose, 0.5 M Tris-Base, 0.1% SDS, pH 8.8) as previously described32. Briefly, cells were lysed in the presence of protease inhibitors (2 mM AEBSF, 0.3 µM Aprotinin, 130 µM Bestatin, 14 µM E64, 10 µM Leupeptin, 10 mM PMSF, and 100 µM 3,4-Dichloroisocoumarin) and mixed with the running buffer (0.01 M Tris-Base, 0.001 M EDTA, 2% SDS, 8 M urea, pH 8.0 with 0.075% bromophenol blue) at a 2:8 volume ratio at 60 °C for 20 min before loading onto the gel. Electrophoresis was performed at a constant voltage of 15 V at room temperature in 25 mM Tris, 192 mM glycine, 0.1% SDS buffer. The gel was then transferred onto a GelBond Film (Lonza), dried at room temperature, stained with an anti-VWF polyclonal antibody, and analyzed using a double-color infrared imaging system.
Statistical analysis
All experimental data and statistical analyses were conducted using GraphPad Prism software (version 7; GraphPad Software, Inc., San Diego, CA). Data are presented as mean ± standard deviation (SD). Statistical differences were assessed using Student’s unpaired two-tailed t-test with Welch’s correction (assuming unequal variances) or one-way ANOVA for comparisons among three or more groups. Correlation analyses were performed using standard Pearson correlation coefficients, with two-tailed p-values and a 95% confidence interval. For all analyses, p-values < 0.05 were considered statistically significant.
Results
Constitutive association between neutrophils and VWF in mouse peripheral blood
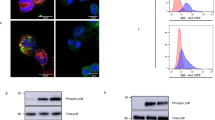
To investigate the interaction between neutrophils and VWF under non-inflammatory conditions, we labeled fresh neutrophils from wildtype mouse whole blood with an anti-Ly6G antibody and assessed the presence of surface VWF signals using flow cytometry with a custom-made polyclonal anti-VWF antibody, the effectiveness of which was showed in Supplementary Fig. S1. An IgG isotype served as the negative control. To minimize the neutrophil apoptosis, the whole process was restrained within 0.5 h after isolation. Data were analyzed by first gating on CD45⁺ leukocytes, followed by identification of Ly6G⁺ neutrophils within this population using flow cytometry. As showed, neutrophils carry significant amount of VWF signals according to the flow cytometry data (Fig. 1A,B). To further validate this, we separated leukocytes via fluorescence-activated cell sorting (FACS) and used SDS-agarose gel electrophoresis to directly analyze the presence of VWF multimer in the cell lysis. As showed, the neutrophils exhibited classical VWF multimers in comparison to other leukocytes (Fig. 1C). These results indicate that neutrophils carry significant amount of VWF on their surface. To further examine this conclusion, we utilized our established GFP-VWF transgenic mouse model, which enables tracking of VWF distribution through its intrinsic GFP fluorescence signal32. Indeed, the neutrophils in this GFP-VWF chimeric mice showed distinctive GFP-VWF signals compared to other peripheral blood cells (Fig. 1D). Furthermore, the intrinsic GFP-VWF signal enabled a direct comparison of total VWF levels between neutrophils and platelets, which are known to carry VWF in their granules. The results showed that VWF levels in neutrophils were approximately 15 times higher than those in platelets (Fig. 1E,F). In the mouse blood circulation, neutrophils can also bound by platelet. To test whether platelets contribute to the VWF binding to neutrophils, we probed the CD42b signals on the fresh neutrophils using flow cytometry. The result showed that approximately 8.14 ± 1.6% of neutrophils were associated with platelets in our system (Supplementary Fig. S2). This is in stark contrast to the 60–90% of neutrophils that were VWF-positive, as observed in our study. Based on this discrepancy, we conclude that platelet involvement in VWF-neutrophil association is limited.
Interaction between mouse peripheral neutrophils and VWF. (A) Representative flow cytometry histogram illustrating VWF presence on the surface of neutrophils from wildtype mice. (B) Statistical analysis of mean fluorescence intensity (MFI) for VWF on neutrophil surfaces relative to control (n = 11–12). (C) SDS-agarose gel electrophoresis comparing VWF multimer in neutrophils (Ly6G+) versus other leukocytes isolated from wildtype mouse peripheral blood. Full-length gel is included in supplementary figure S5A. (D) Flow cytometry scatter plot displaying leukocyte populations from GFP-VWF transgenic mice stained with anti-Ly6G antibody. (E) Overlay of flow cytometry histograms comparing VWF signal intensity between platelets and neutrophils in GFP-VWF transgenic mice. (F) Statistical summary (n = 4) corresponding to data in preceding panels. Statistical significance was assessed using t-tests for pairwise comparisons and one-way ANOVA for multiple groups, with significance denoted as ****p < 0.0001, *p < 0.05.
The neutrophil-VWF association is absent in bone marrow
Since neutrophils are produced in bone marrow, we asked whether the neutrophil-VWF association exists in bone marrow33. We performed LSCFI analysis to determine whether bone marrow neutrophils exhibit GFP-VWF signals. The neutrophils in bone marrow sections were labeled by an anti-Ly6G. The LSCFI result showed that in bone marrow sections the GFP-VWF signals are well-separated with the anti-Ly6G signals, suggesting that there is little neutrophils-VWF association in bone marrow (Fig. 2). This observation indicates that the neutrophil-VWF association is restricted to peripheral blood and does not occur in the bone marrow.
The neutrophil-VWF association is absent in bone marrow. Representative confocal fluorescence microscopy images of bone marrow from GFP-VWF transgenic mice, showing intrinsic GFP-VWF fluorescence in green, anti-mouse Ly6G staining labeling neutrophils in red, and nuclei stained with DAPI in blue. Scale bars represent magnifications of 100 μm and 10 μm, respectively.
Depletion of neutrophils increases the VWF level in blood
Depleting neutrophils should release bound VWF into the circulation raising plasma VWF levels if neutrophils constitutively associate with plasma VWF. To deplete neutrophils in peripheral blood, we administered anti-Ly6G antibody to the wild-type mice via tail vein injection. As showed, the anti-Ly6G antibody efficiently removed neutrophils from circulation compared to IgG control within 2 h after injection (Fig. 3A). Simultaneously, plasma VWF levels were significantly elevated compared to control mice (Fig. 3B). To rule out the elevated VWF level is contributed by VWF synthesis and secretion, we examined the VWF propeptide levels in peripheral blood and VWF mRNA levels in the aorta and lung. The results showed that during this treatment, there was no difference in VWF propeptide levels and VWF mRNA levels between control and anti-Ly6G-treated mice (Fig. 3C,D). Additionally, we also examined the change in mRNA levels of reported cellular scavengers of VWF. The result showed that all of these cellular scavengers except for MGL2 stay constant during this treatment. This lack of change in established VWF clearance pathways suggests that the elevated plasma VWF levels observed are unlikely to result from reduced elimination (Fig. 3D). Collectively, these results strongly support that the constitutive association between neutrophils and VWF significantly influences plasma VWF levels.
Depletion of neutrophils increases the VWF level in plasma. (A) Representative flow cytometry profiles showing the effect of neutrophil depletion via anti-Ly6G antibody. The IgG isotype serves as control. (B) SDS-agarose gel electrophoresis image showing VWF multimers before and after neutrophil depletion. Full-length gel is included in supplementary figure S5B. The averaged fold change in plasma VWF levels in mice post-neutrophil depletion compared to control is shown on the right (n = 6). (C) Quantitative assessment of plasma VWF propeptide (VWFpp) levels in mice following anti-Ly6G antibody injection at 0 and 12 h, with comparative analysis of VWFpp changes between time points and control groups, as measured by immunoblotting. (D) Comparison of VWF mRNA levels in lung and aorta tissues of anti-Ly6G antibody-treated versus control IgG-treated mice (n = 6, Left) and statistical analysis of mRNA levels of liver genes (asgr1, asgr2, lrp1, sra1, stab2, mgl1, mgl2) in the same groups (n = 6, Right). Statistical analyses were performed using t-tests for pairwise comparisons, with significance denoted as*p < 0.05, and “n.s.” for not significant.
The neutrophil-VWF association is NETs-Independent
To determine whether the observed neutrophil-VWF association is related to NETs formation, which typically occurs during inflammation, we assessed Cit-H3 and MPO signals as NETs markers in our neutrophil samples. As shown in Fig. 4A, both Cit-H3 and MPO signals were low in neutrophils isolated from mouse leukocyte samples. As a positive control, treatment with 100 nM PMA, a known NETs inducer, resulted in a significant increase in Cit-H3 and MPO signals. These results indicate that most neutrophils in our experiments were NETs-negative (Fig. 4A). To directly investigate whether the neutrophil-VWF interaction is NETs-related, we designed an in vitro assay in which leukocytes isolated from wild-type mice were incubated with platelet-poor plasma from GFP-VWF mice. The interaction of GFP-VWF with leukocytes was analyzed by flow cytometry. As shown in Fig. 4B, neutrophils (Ly6G-positive) exhibited a distinct binding affinity for GFP-VWF compared to Ly6G-negative cells. Using this system, we tested whether heparin, a known inhibitor of VWF-NETs interactions, could disrupt the in vitro association between neutrophils and GFP-VWF. As shown in Fig. 4C, heparin had no inhibitory effect on this interaction, even at concentrations up to 100 IU/mL. Altogether, these results demonstrated that the observed neutrophil-VWF association in our test is NETs-independent.
The neutrophil-VWF association is NETs-independent. (A) Statistical analysis showing levels of neutrophil extracellular traps (NETs) in neutrophil samples under test, with NETs visualized via Cit-H3 and MPO staining; Phorbol 12-myristate 13-acetate (PMA) stimulation served as a positive control for NETs formation. (B) Representative flow cytometry profiles showing the GFP-VWF signals on CD45 + cells when the isolated leukocytes from wild-type mice were inoculated with platelet poor plasma from the GFP-VWF transgenic mice. The MFI of GFP-VWF signals was quantified to assess the binding levels of VWF on these cells. (C) Histogram depicting VWF binding strength to neutrophils from wildtype mice at different heparin concentrations (n = 3). Statistical analyses were performed using t-tests for pairwise comparisons, with significance denoted as ***p < 0.001, and “n.s.” for not significant.
Association of human neutrophils and VWF in peripheral blood
To extend our investigation of neutrophil-VWF interactions to humans, we analyzed VWF binding on neutrophils in a cohort of 23 healthy donors using a method similar to that applied in mice. Consistent with our murine findings, human neutrophils exhibited significant surface-bound VWF (Fig. 5A). However, the proportion of VWF-positive neutrophils varied widely among individuals, ranging from less than 10% to over 80%. Compared to mice, human samples showed a lower percentage of VWF-positive neutrophils (Fig. 5B), possibly due to the larger neutrophil count in humans. We further examined potential factors influencing VWF-neutrophil interactions, including gender, age, and blood type. The results showed no significant gender-based differences in VWF-neutrophil association (Fig. 5C). A weak inverse correlation was observed between VWF-positive neutrophils and age (Pearson correlation coefficient: -0.2431) (Fig. 5D). Further analysis revealed no correlation between the percentage of VWF-bound neutrophils and the total neutrophil count in individual donors (Fig. 5E). Notably, blood type significantly affected VWF-neutrophil interactions, with blood type O exhibiting substantially stronger VWF binding than non-O blood types (Fig. 5F). In conclusion, human neutrophils also constitutively interact with VWF in peripheral blood, with blood type O displaying stronger VWF binding than non-O blood types.
Association of human neutrophils and VWF in peripheral blood. (A) Representative flow cytometry histogram showing VWF presence on the surface of human neutrophils. (B) Statistical comparison of VWF-positive neutrophil percentages between human and mouse peripheral blood samples. (C) Statistical analysis of VWF levels on human neutrophils from male and female participants. (D) Correlation analysis of the relationship between VWF levels (mean fluorescence intensity, MFI) on human neutrophils and participant age (years); each data point representing an individual, with the solid line shows the line of best fit. Pearson correlation coefficient (r) = 0.2431, n = 23. (E) Correlation analysis between the percentage of VWF-bound neutrophils and total neutrophil count. Each data point represents an individual, with the solid line indicating the line of best fit. Pearson correlation coefficient (r) = 0.0286; n = 23. (F)Statistical comparison of VWF levels on neutrophils from individuals with blood types O and non-O. Statistical analyses were conducted using t-tests for pairwise comparisons, with significance indicated as ****p < 0.0001, **p < 0.01, and “n.s.” for not significant.
Desialylation of VWF and PSGL-1/ β2-integrins on neutrophils mediate the constitutive neutrophil-VWF association
To explore the mechanism underlying the observed constitutive neutrophil-VWF interaction, we performed in vitro binding assays using freshly isolated human neutrophils. These neutrophils were isolated from healthy human donors with over 93% purity (supplementary Figure S3). Given the lower percentage of VWF-positive neutrophils in humans despite their large overall neutrophil population, we hypothesized that neutrophils may selectively bind specific VWF molecules rather than all circulating VWF. To test this, we investigated whether VWF desialylation, a critical step in VWF aging34influences the VWF-neutrophil interaction. Desialylated VWF, confirmed by its mobility shift on SDS-PAGE (Supplementary Fig. S4), was incubated with freshly isolated neutrophils at various concentrations. Untreated VWF at the same concentrations was processed in parallel as a control. The potential influence of residual neuraminidase in the final samples was considered negligible. The results showed that desialylated VWF exhibited significantly higher binding affinity to neutrophils compared to normal VWF at equivalent concentrations (Fig. 6A), with maximum binding occurring at approximately 50 µg/mL for both forms. This finding suggests that desialylation may play a key role in regulating neutrophil-VWF interactions as part of a constitutive mechanism during VWF circulation.
Desialylation of VWF and PSGL-1/ β2-integrins on neutrophils mediate the constitutive neutrophil-VWF association. (A) Flow cytometry analysis of binding affinity of native VWF (solid circles, n = 4) and desialylated VWF (hollow frames, n = 4) to human neutrophils, with binding curves fitted using a ‘One Site-Total Binding’ model. (B) Flow cytometry analysis of the inhibition of desialylated VWF binding to neutrophils by anti-PSGL-1 and anti-β2 integrin antibodies, normalized to isotype IgG control (n = 4). Statistical analyses were conducted using t-tests for pairwise comparisons, with significance indicated as ***p < 0.001, **p < 0.01, *p < 0.05.
In previous studies, PSGL-1 and β2-integrins were implicated in cooperatively mediating VWF-leukocyte interactions in vitro through various VWF domains28. To test the involvement of these receptors in neutrophil binding to desialylated VWF, we recruited antibodies against PSGL-1 and β2-integrins in the binding of 50 µM desialylated VWF with neutrophils. The results indicated that applying each antibody individually reduced VWF binding by approximately 40–60%, and combined antibody treatment reduced binding to ~ 25% (Fig. 6B). These results indicate that both PSGL-1 and β2-integrins play a role in mediating neutrophil interactions with desialylated VWF in peripheral blood.
Discussion
Neutrophils primarily act as the first line of defense in the immune system. Emerging evidence indicates that neutrophils may interact with plasma VWF through NETs binding to the A1 domain of VWF, particularly in the context of inflammatory thrombosis35,36,37. In this study, we demonstrate a constitutive association between neutrophils and VWF in normal peripheral blood environment, likely revealing a novel function of neutrophils beyond their established antimicrobial role. Several key aspects of our findings warrant further discussion.
The distinguishable anti-VWF antibody signals on neutrophils observed in our study represent the initial evidence of neutrophil-VWF binding in normal peripheral blood. This finding is further supported by more definitive evidence from agarose gel electrophoresis analysis of VWF multimers on neutrophils. Initially, we were unable to detect VWF bands on the gel when lysing cells on ice for 30 min with NP40 lysis buffer. VWF bands were only detected after rapid cell lysis at 60 ºC, supplemented with an excess of protease inhibitors. This suggests that neutrophils likely release potent proteases that degrade VWF during lysis, which could explain why this interaction was not recognized in previous studies. The neutrophil-VWF binding in normal peripheral blood was also directly observed in our established transgenic mouse model expressing chimeric GFP-VWF, which avoids potential false positives from antibody use. Importantly, this GFP-VWF model closely resembles all features of normal VWF, except for its lower plasma levels. This similarity between GFP-VWF and normal VWF was further strengthened by their binding to neutrophils.
It is worth noting that not all VWF in peripheral blood interacts with neutrophils. Our data show that, on average, about 30% of human neutrophils and 80% of mouse neutrophils in peripheral blood are associated with VWF. Human neutrophils constitute 60–70% of the white blood cell population, while mouse neutrophils generally comprise 20–30%. This difference in neutrophil population may explain the varying percentages of VWF-bound neutrophils between humans and mice. Mechanistically, the role of glycan structures in mediating the neutrophil-VWF interaction is highlighted by multiple pieces of evidence. Our analysis of VWF-bound neutrophils between individuals in the blood O-type group compared to non-O groups showed that blood type O exhibited a significantly higher percentage of VWF-bound neutrophils. Considering the relatively lower plasma VWF levels in blood type O individuals compared to those with non-O blood types38,39,40this observation suggests that the neutrophil-VWF interaction may contribute to the different plasma VWF levels seen among individuals with varying blood types. Furthermore, the observation that desialylated VWF binds more strongly to neutrophils provides additional direct evidence for the importance of glycans in neutrophil-VWF association, further implicating VWF aging in this process. Previous studies have shown that desialylated VWF binds more avidly to residual macrophages in the liver and spleen. This raises the possibility that neutrophils in peripheral blood might also contribute to VWF clearance through a similar “clear me” signal. Consistent with this idea, neutrophil depletion resulted in a significant increase in plasma VWF levels, reinforcing the concept that neutrophil-VWF binding is crucial for maintaining plasma VWF levels. Notably, neutrophil depletion had minimal impact on VWF secretion.
We also examined the role of neutrophil receptors PSGL-1 and β2 integrins, which have been shown to mediate the static in vitro interaction between VWF and leukocytes cooperatively28. Our study extends this understanding by showing that these receptors may specifically recognize desialylated VWF, as antibodies targeting PSGL-1 and β2 integrins significantly blocked the interaction between neutrophils and desialylated VWF. These results imply that neutrophils in peripheral blood may selectively recognize desialylated VWF rather than intact VWF through these receptors. However, additional in vivo evidence is required to fully support this conclusion. Notably, monocytes also express PSGL-1 and β2 integrins; however, they were excluded from the current study due to their relatively low abundance compared to neutrophils.
Previous research has suggested that during inflammatory responses, neutrophils interact with VWF through a NETs-dependent mechanism37,41,42,43,44,45. In contrast, the neutrophils assessed in our study were predominantly NET-negative. Therefore, the neutrophil-VWF association observed in our study is novel and may provide new insights into the diverse biological roles of neutrophils. We also found that VWF signals on neutrophils were distinctly stronger compared to platelets, though we do not yet know whether this enrichment of VWF on neutrophils affects the NETs-dependent interactions between VWF and neutrophils. Notably, in VWF-deficient mice, NET formation was significantly reduced in brain tissue during cerebral ischemia-reperfusion injury46. Thus, the constitutive association of VWF with neutrophils may, to some extent, contribute to the neutrophil response to thromboinflammation, warranting future investigation.
It is important to acknowledge several limitations of our study. We assessed VWF clearance receptor expression using liver RNA; however, mRNA levels do not always correlate with protein expression due to post-transcriptional and translational regulation. Additionally, VWF clearance is a multi-organ process that involves not only the liver but also other organs such as the spleen. Future studies incorporating protein-level analyses and examining multiple tissues would provide a more comprehensive understanding of VWF clearance mechanisms.
In conclusion, our study reveals a novel function of neutrophils in normal conditions. The constitutive interaction between neutrophils and VWF in peripheral blood suggests that neutrophils may play a role in regulating plasma glycoprotein levels. This discovery opens new avenues for exploring a broader spectrum of neutrophil functions beyond their well-established antimicrobial roles.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Liew, P. X. & Kubes, P. The neutrophil’s role during health and disease. Physiol. Rev. 99, 1223–1248. https://doi.org/10.1152/physrev.00012.2018 (2019).
Mestas, J. & Hughes, C. C. Of mice and not men: differences between mouse and human immunology. J. Immunol. 172, 2731–2738. https://doi.org/10.4049/jimmunol.172.5.2731 (2004).
Ng, L. G., Ostuni, R. & Hidalgo, A. Heterogeneity of neutrophils. Nat. Rev. Immunol. 19, 255–265. https://doi.org/10.1038/s41577-019-0141-8 (2019).
Schmutz, N., Henry, E., Jopling, J. & Christensen, R. D. Expected ranges for blood neutrophil concentrations of neonates: the Manroe and Mouzinho charts revisited. J. Perinatol. 28, 275–281. https://doi.org/10.1038/sj.jp.7211916 (2008).
Frater, J. L. How I investigate neutropenia. Int. J. Lab. Hematol. 42(Suppl 1), 121–132. https://doi.org/10.1111/ijlh.13210 (2020).
Tahir, N. & Zahra, F. In StatPearls (StatPearls PublishingCopyright © 2024 (StatPearls Publishing LLC., 2024).
Tak, T., Tesselaar, K., Pillay, J., Borghans, J. A. & Koenderman, L. What’s your age again? Determination of human neutrophil half-lives revisited. J. Leukoc. Biol. 94, 595–601. https://doi.org/10.1189/jlb.1112571 (2013).
Lawrence, S. M., Corriden, R. & Nizet, V. The ontogeny of a neutrophil: mechanisms of granulopoiesis and homeostasis. Microbiol. Mol. Biol. Rev. 82 https://doi.org/10.1128/mmbr.00057-17 (2018).
Zhou, Y., Xu, Z. & Liu, Z. Impact of neutrophil extracellular traps on thrombosis formation: new findings and future perspective. Front. Cell. Infect. Microbiol. 12, 910908. https://doi.org/10.3389/fcimb.2022.910908 (2022).
Blanch-Ruiz, M. A., Ortega-Luna, R., Martínez-Cuesta, M. & Álvarez, Á. The neutrophil secretome as a crucial link between inflammation and thrombosis. Int. J. Mol. Sci. 22 https://doi.org/10.3390/ijms22084170 (2021).
Roldán, V. et al. Short alleles of P-selectin glycoprotein ligand-1 protect against premature myocardial infarction. Am. Heart J. 148, 602–605. https://doi.org/10.1016/j.ahj.2004.04.020 (2004).
Yu, M. et al. QiShen YiQi and its components attenuate acute thromboembolic stroke and carotid thrombosis by Inhibition of CD62P/PSGL-1-mediated platelet-leukocyte aggregate formation. Biomed. Pharmacother. 160, 114323. https://doi.org/10.1016/j.biopha.2023.114323 (2023).
Alkarithi, G., Duval, C., Shi, Y., Macrae, F. L. & Ariëns, R. A. S. Thrombus structural composition in cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 41, 2370–2383. https://doi.org/10.1161/atvbaha.120.315754 (2021).
Fuchs, T. A. et al. Extracellular DNA traps promote thrombosis. Proc. Natl. Acad. Sci. U.S.A. 107, 15880–15885. https://doi.org/10.1073/pnas.1005743107 (2010).
Sporn, L. A., Marder, V. J. & Wagner, D. D. Inducible secretion of large, biologically potent von Willebrand factor multimers. Cell 46, 185–190. https://doi.org/10.1016/0092-8674(86)90735-x (1986).
El-Mansi, S. & Nightingale, T. D. Emerging mechanisms to modulate VWF release from endothelial cells. Int. J. Biochem. Cell Biol. 131, 105900. https://doi.org/10.1016/j.biocel.2020.105900 (2021).
von Springer, T. A. Willebrand factor, jedi knight of the bloodstream. Blood 124, 1412–1425. https://doi.org/10.1182/blood-2014-05-378638 (2014).
Lazzari, M. A., Sanchez-Luceros, A., Woods, A. I., Alberto, M. F. & Meschengieser, S. S. Von Willebrand factor (VWF) as a risk factor for bleeding and thrombosis. Hematology 17(Suppl 1), S150–152. https://doi.org/10.1179/102453312x13336169156618 (2012).
Leebeek, F. W. & Eikenboom, J. C. Von willebrand’s disease. N. Engl. J. Med. 375, 2067–2080. https://doi.org/10.1056/NEJMra1601561 (2016).
Budde, U., Bergmann, F. & Michiels, J. J. Acquired von Willebrand syndrome: experience from 2 years in a single laboratory compared with data from the literature and an international registry. Semin. Thromb. Hemost. 28, 227–238. https://doi.org/10.1055/s-2002-27824 (2002).
Garg, A., Gupta, G., Gupta, R. & Mishra, R. K. Converging pathways: acquired von Willebrand disease in systemic lupus erythematosus with antiphospholipid antibodies presenting with persistent menstrual bleeding. BMJ Case Rep. https://doi.org/10.1136/bcr-2024-260824 (2024).
Comerford, C., Glavey, S., Quinn, J. & O’Sullivan, J. M. The role of VWF/FVIII in thrombosis and cancer progression in multiple myeloma and other hematological malignancies. J. Thromb. Haemostas. 20, 1766–1777. https://doi.org/10.1111/jth.15773 (2022).
Favaloro, E. J., Henry, B. M., Lippi, G., Increased, V. W. F., Decreased & and ADAMTS-13 in COVID-19: creating a milieu for (Micro)Thrombosis. Semin. Thromb. Hemost. 47, 400–418. https://doi.org/10.1055/s-0041-1727282 (2021).
Arisz, R. A., de Vries, J. J., Schols, S. E. M., Eikenboom, J. C. J. & de Maat, M. P. M. Interaction of von Willebrand factor with blood cells in flow models: a systematic review. Blood Adv. 6, 3979–3990. https://doi.org/10.1182/bloodadvances.2021006405 (2022).
Ruggeri, Z. M. Von Willebrand factor, platelets and endothelial cell interactions. J. Thromb. Haemostasis: JTH. 1, 1335–1342. https://doi.org/10.1046/j.1538-7836.2003.00260.x (2003).
Wang, Y. et al. Heparan sulfate dependent binding of plasmatic von Willebrand factor to blood Circulating melanoma cells attenuates metastasis. Matrix Biol. 111, 76–94. https://doi.org/10.1016/j.matbio.2022.06.002 (2022).
O’Sullivan, J. M. et al. N-linked glycan Truncation causes enhanced clearance of plasma-derived von Willebrand factor. J. Thromb. Haemostas. 14, 2446–2457. https://doi.org/10.1111/jth.13537 (2016).
Pendu, R. et al. P-selectin glycoprotein ligand 1 and beta2-integrins cooperate in the adhesion of leukocytes to von Willebrand factor. Blood 108, 3746–3752. https://doi.org/10.1182/blood-2006-03-010322 (2006).
Parker, E. T. & Lollar, P. Conformation of the von Willebrand factor/factor VIII complex in quasi-static flow. J. Biol. Chem. 296, 100420. https://doi.org/10.1016/j.jbc.2021.100420 (2021).
Kusumbe, A. P., Ramasamy, S. K., Starsichova, A. & Adams, R. H. Sample Preparation for high-resolution 3D confocal imaging of mouse skeletal tissue. Nat. Protoc. 10, 1904–1914. https://doi.org/10.1038/nprot.2015.125 (2015).
Boivin, G. et al. Durable and controlled depletion of neutrophils in mice. Nat. Commun. 11, 2762. https://doi.org/10.1038/s41467-020-16596-9 (2020).
Xu, L. et al. Tissue dynamics of von Willebrand factor characterized by a novel fluorescent protein-von Willebrand factor chimera. J. Thromb. Haemostasis: JTH. 20, 208–221. https://doi.org/10.1111/jth.15542 (2022).
Basu, S., Hodgson, G., Katz, M. & Dunn, A. R. Evaluation of role of G-CSF in the production, survival, and release of neutrophils from bone marrow into circulation. Blood 100, 854–861. https://doi.org/10.1182/blood.v100.3.854 (2002).
Rydz, N. et al. The C-type lectin receptor CLEC4M binds, internalizes, and clears von Willebrand factor and contributes to the variation in plasma von Willebrand factor levels. Blood 121, 5228–5237. https://doi.org/10.1182/blood-2012-10-457507 (2013).
Grässle, S. et al. Von Willebrand factor directly interacts with DNA from neutrophil extracellular traps. Arterioscler. Thromb. Vasc. Biol. 34, 1382–1389. https://doi.org/10.1161/atvbaha.113.303016 (2014).
Sandoval-Pérez, A. et al. DNA binds to a specific site of the adhesive blood-protein von Willebrand factor guided by electrostatic interactions. Nucleic Acids Res. 48, 7333–7344. https://doi.org/10.1093/nar/gkaa466 (2020).
Oklu, R. et al. Detection of extracellular genomic DNA scaffold in human thrombus: implications for the use of deoxyribonuclease enzymes in thrombolysis. J. Vasc. Interv. Radiol. 23, 712–718. https://doi.org/10.1016/j.jvir.2012.01.072 (2012).
Albanez, S. et al. Aging and ABO blood type influence von Willebrand factor and factor VIII levels through interrelated mechanisms. J. Thromb. Haemostas. 14, 953–963. https://doi.org/10.1111/jth.13294 (2016).
Franchini, M., Crestani, S., Frattini, F., Sissa, C. & Bonfanti, C. ABO blood group and von Willebrand factor: biological implications. Clin. Chem. Lab. Med. 52, 1273–1276. https://doi.org/10.1515/cclm-2014-0564 (2014).
Murray, G. P., Post, S. R. & Post, G. R. ABO blood group is a determinant of von Willebrand factor protein levels in human pulmonary endothelial cells. J. Clin. Pathol. 73, 347–349. https://doi.org/10.1136/jclinpath-2019-206182 (2020).
Sandoval-Perez, A. et al. DNA binds to a specific site of the adhesive blood-protein von Willebrand factor guided by electrostatic interactions. Nucleic Acids Res. 48, 7333–7344. https://doi.org/10.1093/nar/gkaa466 (2020).
Fukui, S., Gutch, S., Fukui, S., Chu, L. & Wagner, D. D. Anti-inflammatory protective effect of ADAMTS-13 in murine arthritis models. J. Thromb. Haemostas. 20, 2386–2393. https://doi.org/10.1111/jth.15828 (2022).
Mang, G. et al. Von Willebrand factor exacerbates heart failure through formation of neutrophil extracellular traps. Eur. Heart J. 45, 3853–3867. https://doi.org/10.1093/eurheartj/ehae517 (2024).
Yada, N. et al. Targeting neutrophil extracellular trap accumulation under flow in patients with immune-mediated thrombotic thrombocytopenic purpura. Blood Adv. 8, 2536–2551. https://doi.org/10.1182/bloodadvances.2023011617 (2024).
Yang, J. et al. Insights into immunothrombosis: the interplay among neutrophil extracellular trap, von Willebrand factor, and ADAMTS13. Front. Immunol. 11, 610696. https://doi.org/10.3389/fimmu.2020.610696 (2020).
De Wilde, M., Desender, L., Tersteeg, C., Vanhoorelbeke, K. & De Meyer, S. F. Spatiotemporal profile of neutrophil extracellular trap formation in a mouse model of ischemic stroke. Res. Pract. Thromb. Haemost. 7, 100028. https://doi.org/10.1016/j.rpth.2022.100028 (2023).
Acknowledgements
We thank the public instrument platform of Cyrus Tang Medical institution that provides great help on fluorescence microscopy experiment.
Funding
This study was supported by the National Natural Science Foundation of China (NSFC) (U23A20419, 81970125, and 82270138), the Priority Academic Program Development of Jiangsu Higher Education Institutions, the Project of Fujian Province Department of Science & Technology (2023Y9079), and the Talent Scientific Research Project of the First Affiliated Hospital of Fujian Medical University (YJRC4415).
Author information
Authors and Affiliations
Contributions
Y.W., T.Y. and W.D. designed the experiments and analyzed the data; Y.W., M.G., J.Z., Q.M., Y.P., Y.C. performed the experiments, W.D. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wan, Y., Gao, M., Zhu, J. et al. Constitutive interaction between neutrophils and von Willebrand factor in peripheral blood. Sci Rep 15, 24895 (2025). https://doi.org/10.1038/s41598-025-10321-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-10321-6