Abstract
Metabolic dysfunction associated steatotic liver disease (MASLD) presents a growing global health problem. Disease progression is promoted not only by hepatic leukocyte accumulation but also by inflammatory signals from adipose tissue and an altered gut microbiome. To determine the contribution of intercellular adhesion molecule-1 (ICAM-1) to MASLD pathogenesis, male mice with an ICAM-1 mutation (Icam1tmBay) and wild type (WT) mice were compared in 12 and 24-week feeding experiments with a Western-style diet (WD) containing 40 kcal% fat, 20 kcal% fructose, and 2% cholesterol. WD-induced MASLD was accompanied by increased ICAM-1 expression in liver, epididymal white adipose tissue (EWAT), and intestine in WT mice. WD-fed Icam1tmBay mice exhibited increased circulating neutrophils, higher frequencies of inflammatory leukocytes in EWAT, and a worsened glucose tolerance when compared to WT mice. In contrast, the mutation resulted in reduced WD-induced liver disease activity and less accumulation of intrahepatic leukocytes. WD-feeding caused substantial changes in fecal microbiota with decreased microbial diversity that differed between the mouse strains. In conclusion, ICAM-1 positively regulates adipose tissue homeostasis and protects from insulin resistance but promotes liver damage in diet-induced obesity. This points to organ-specific roles for ICAM-1 and the potential of liver-specific targeting of ICAM-1 for treatment of MASLD.
Similar content being viewed by others
Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD) is strongly linked to obesity, hypertension, dyslipidemia, and insulin resistance and is considered to be the hepatic component of the metabolic syndrome1. It represents a spectrum of liver diseases ranging from simple fat accumulation and metabolic dysfunction-associated steatohepatitis (MASH) to fibrosis and hepatocellular carcinoma. Steatosis-associated liver injury is now becoming recognized as the major cause of liver damage in Western societies2. Inflammation of the liver is caused and maintained by an accumulation of leukocytes that are organized into inflammatory infiltrates, especially around damaged hepatocytes3,4. This process is brought about by the interaction of the activated hepatic endothelium with chemokine receptors and cell adhesion molecules expressed on circulating immune cells5.
The cell adhesion molecule intercellular adhesion molecule-1 (ICAM-1 or CD54) is a transmembrane glycoprotein member of the immunoglobulin (Ig) super-gene family expressed at a low level on leukocytes, endothelial-, epithelial-, and other cells6. ICAM-1 binds to multiple ligands, including the β2 integrins LFA-1 (lymphocyte function-associated antigen-1, αLβ2), MAC-1 (macrophage-1 antigen, αMβ2), and p150,95 (αXβ2). Full-length membrane bound ICAM-1 consists of five extracellular Ig domains, a transmembrane domain, and a short cytoplasmic tail6. The ligand LFA-1 binds to the first Ig domain, whereas Mac-1 and p150,95 have been shown to bind Ig domains 3 and 3/4, respectively. In addition to the full-length protein, six splice variants, varying in their combination and number of Ig domains, have been reported, which may also contribute to additional soluble (s) ICAM-1 variants. All the isoforms contain Ig domains 1 and 56,7. Their expression seems to be cell-type specific and may depend on the inflammatory status and differentially impact disease outcome8. Different results from experimental disease models using mice that lack different domains of the ICAM-1 protein support this hypothesis7,9.
Leukocyte infiltration and low-grade inflammation are important characteristics of diet-induced metabolic dysfunction10. ICAM-1 expression is upregulated in response to inflammatory stimulation, particularly on endothelium. It is best known for regulating the recruitment of circulating leukocytes to inflammatory sites and contributes to the onset but also the resolution of pathogenesis6. Increased hepatic ICAM-staining and circulating ICAM-1 levels have previously been observed in MASH patients11. In addition, elevated sICAM-1 levels have been detected in patients with metabolically unhealthy obesity and correlate with insulin resistance12,13. Moreover, in high fat-treated mice, circulating sICAM-1 levels correlate with body and abdominal fat pad weight and male mice showed an upregulation of Icam-1 mRNA in adipose tissue14.
To better define the impact of ICAM-1 in manifestations of metabolic dysfunction, we compared the outcome of western diet (WD)-induced MASLD in wild type (WT mice) and mice carrying a dysfunctional ICAM-1 gene (Icam1tmBay)9. These mice have a deletion of exon 5 which encodes the Ig domain 4 9. They are not able to express the full-length protein, but due to alternative splicing they do express low amounts of the smaller ICAM-1 isoforms (isoforms containing domain 1 + 5, 1 + 2 + 5, and 1 + 2 + 3 + 5)7. They show only residual membrane-bound ICAM-1 staining in thymus, lung, and spleen tissue but none at all in gut and liver15 which makes them a suitable model for our investigations.
Results
Dysfunction of ICAM-1 has no impact on Western diet-induced overall body fat deposition
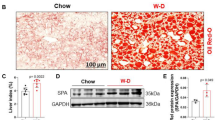
To address the importance of ICAM-1 in manifestations of metabolic dysfunction, we analyzed the expression of ICAM-1 in experimentally induced MASLD. MASLD was induced by subjecting wild type (WT) mice for 24 weeks to a specific western diet (WD). Immunohistochemical expression of ICAM-1 was strongly increased in liver, epididymal white adipose tissue (EWAT), small intestine, and colon of WD-fed mice in comparison to the chow diet (CD)-fed controls (Fig. 1A). In addition, WD treatment caused significantly increased Icam-1 mRNA levels in tissue homogenates of liver, EWAT and small intestine. (Fig. 1B-D).
The ICAM-1 mutation has no impact on western diet-induced weight gain. Wild type (WT) mice (shown in black) and ICAM-1-mutant (Icam1tmBay) mice (shown in purple) were fed for 12 or 24 weeks with Western diet (WD). (A) Representative pictures of sections from the indicated organs (liver, epididymal white adipose tissue (EWAT), small intestine and colon) of WT mice that have been fed for 24 weeks with chow diet (CD) or WD. Sections have been stained with anti-ICAM-1 antibody and ICAM-1 protein is visualized as brown deposits developed with 3,3′-Diaminobenzidin reagent. Cellular nuclei are counterstained with hematoxylin (blue) (original magnification X 20, scale bar = 50 μm). (B-D) mRNA levels of Icam1 were measured in (B) liver tissue (CD-fed (n = 6), WD-fed (n = 7) mice) (C) EWAT (CD-fed (n = 11), WD-fed (n = 12) and (D) small intestine tissue (CD-fed (n = 6), WD-fed (n = 7) of WT mice after 24 weeks of the indicated feeding. Values are expressed as fold increase over the mean value obtained for the respective CD-treated tissue. Statistical significance was calculated by non-parametric t-test. Values are represented as mean ± SD. * p ≤ 0.05 and ** p ≤ 0.01. (E) Weight gain of WT (n = 8) and Icam1tmBay (n = 8) mice represented as a percentage of weekly weight change, and (F) corresponding cumulative food intake. (G) Representative three-dimensional volume renderings of segmented bones (white), lungs (pink), visceral fat (green), and subcutaneous fat (blue) upon in vivo µCT imaging of 12 weeks WD-fed WT and Icam1tmBay mice and 2D cross-sectional µCT images in transversal planes of the abdomen of the respective mice. (H-K) Quantification of the body composition of WT (n = 3) and Icam1tmBay (n = 4) mice using µCT imaging after 12 weeks WD feeding (G) body volume, (H) liver volume, (I) visceral fat, and (J) subcutaneous fat. Statistical significance was calculated by t-test. Values are represented as mean ± SD.
Next, we compared the outcome of WD-induced MASLD in WT mice and mice carrying the dysfunctional ICAM-1 gene (Icam1tmBay)9. Weight gain in the ICAM-1 mutant was indistinguishable to that of WT mice (Fig. 1E) and both mouse strains showed the same cumulative food consumption (Fig. 1F). Furthermore, micro-computed tomography (µCT) after 12 weeks of WD feeding demonstrated similar body composition, quantified as body volume, liver volume, and visceral and subcutaneous fat volume (Fig. 1G-K), for both mouse strains.
WD fed Icam1tmBay mice display worsened glucose tolerance and inflammatory tone
The ICAM-1 mouse mutant displayed a higher percentage of circulating neutrophils when compared with WT mice (Fig. 2A)9which was further increased by treatment with WD. Increased circulating neutrophils in the ICAM-1 mutant did not, however, translate into a generally increased inflammatory activity of the myeloid blood cell population. Instead, the respective cell populations of both mouse strains displayed comparable phagocytic activity and ROS production (Supplementary Fig. S2). On regular CD, we found no difference in glucose homeostasis between WT mice and the ICAM-1 mutant (Fig. 2B-C). However, after 24 weeks of WD feeding Icam1tmBay mice exhibited more pronounced features of the metabolic syndrome, as demonstrated by slower falls in glucose levels after glucose injection (Fig. 2D-E). Blood glucose levels of WD treated mice were independent of the genotype and slightly increased in comparison to CD-fed mice (Fig. 2F) While serum triglyceride levels were unaffected by either diet or genotype, WD treatment caused increased levels of serum cholesterol, which did not vary between the mouse strains (Fig. 2G-H). Interestingly, TNFα and IL6 were significantly increased upon WD-feeding in Icam1tmBay mice compared to WT mice. MCP-1 was slightly increased due to the diet in both mouse strains, but more in WT mice, and IL1B levels were not affected by diet or genotype (Fig. 2I).
The ICAM-1 mutation aggravates glucose tolerance and systemic inflammation. Wild type (WT) mice (shown in black) and ICAM-1 mutant (Icam1tmBay) mice (shown in purple) were fed for 24 weeks with chow diet (CD, WT (n = 6–13), Icam1tmBay (n = 6)) or Western diet (WD, WT (n = 8–15), Icam1tmBay (n = 8)). (A) Comparative immune cell analysis of circulating neutrophils by flow cytometry. Representative FACS dot plots illustrating the gating strategy are shown in Supplementary Fig. S1. Depicted are the percentages of CD45+ cells of CD45+CD11b+Ly6G+ neutrophils. (B) Endpoint glucose tolerance CD-treatment (WT (n = 10), Icam1tmBay (n = 6)), and (C) corresponding quantification as area under curve. (D) Endpoint glucose tolerance test WD-treatment (WT (n = 8), Icam1tmBay (n = 8)), and (E) corresponding quantification as area under curve. (F) Serum glucose, (G) serum triglycerides, and (H) serum cholesterol. (I) Serum pro-inflammatory cytokines tumor necrosis factor alpha (TNFA), monocyte chemoattractant protein-1 (MCP1), interleukin-1beta (IL1B), and interleukin-6 (IL6). Statistical significance was calculated either by the one-way ANOVA (A-E) or two-way ANOVA (F-I). Values are represented either as mean ± SD (A,F-I) or mean ± SEM (B-E). * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001. Only differences between genotypes (WT and Icam1tmBay mice) within a diet (CD or WD) and between diets (CD and WD) within a genotype (WT and Icam1tmBay mice) were calculated.
Together, these data demonstrate that upon WD-feeding Icam1tmBay mice are more resistant to glucose tolerance and display a higher inflammatory tone than respectively treated WT mice, however, no other metabolic feature was significantly altered due to the mutation.
Increased accumulation of neutrophils and macrophages in adipose tissue of WD-fed Icam1tmBay mice
In both mouse strains, WD feeding caused an increased EWAT to body weight ratio when compared to respective CD-fed mice, with no difference between the two mouse strains (Fig. 3A). Histological staining of macrophages in EWAT using an antibody against CD68+ showed the arrangement of macrophages in characteristic crown-like structures (CLS) in both groups of obese WD-fed mice but not in lean CD-fed mice (Fig. 3B, CLS are marked with black arrows). CLS around dead adipocytes are indicators of pathological changes in fatty tissue16 and positively correlate with metabolic disorders. CLS in adipose tissue of the WD-fed ICAM-1 mutant were slightly increased in comparison to comparably treated WT mice. This was supported by flow cytometric analysis where we observed significantly increased frequencies of CD11b+ Ly6G+ neutrophils and CD11b+ F4/80+ macrophages in adipose tissue of the WD-treated Icam1tmBay mice (Fig. 3C-D). The frequencies of CD4+ or CD8+ T cells remained unchanged by diet and genotype (not shown).
Epidydimal white adipose tissue (EWAT) of WD-fed Icam1tmBay mice exhibits increased inflammation of immune cells. Wild type (WT) mice (shown in black) and Icam1tmBay mice (shown in purple) were fed for 24 weeks with chow diet (CD) or Western diet (WD). Depicted are the endpoint values of WT CD-fed (n = 6–13), WT WD-fed (n = 8–15), Icam1tmBay CD-fed (n = 6), and Icam1tmBay WD-fed (n = 8). (A) EWAT/body weight ratio. (B) Representative images of CD68 stained (EWAT sections of the indicated mice strains (original magnification X 20, scale bar = 50 μm). Areas with CD68+ crown-like structures are labeled with black arrows. (C-D) Comparative immune cell analysis of EWAT by flow cytometry. Representative FACS dot plots illustrating the gating strategy are shown in Supplementary Fig. S1. Depicted are the percentages of CD45+ cells of (C) neutrophils (CD45+CD11b+F4/80-Ly6G+), and (D) macrophages (CD45+Ly6G-CD11b+F4/80+). Statistical significance was calculated by the two-way ANOVA. Values are represented as mean ± SD (A, C-D). * p ≤ 0.05, *** p ≤ 0.001, **** p ≤ 0.0001. Only differences between genotypes (WT and Icam1tmBay mice) within a diet (CD or WD) and between diets (CD and WD) within a genotype (WT and Icam1tmBay mice) were calculated.
Improved NAFLD activity score (NAS) in WD-fed Icam1tm1Bay mice
Under basal conditions of chow feeding, WT mice and mice with the dysfunctional ICAM-1 gene displayed no signs of hepatocyte injury, as manifested in low serum values of AST, ALT, and ALP (Fig. 4A-C). However, WD feeding led to significantly increased levels of serum transaminases and ALP in WT mice, which was not observed in the respectively treated ICAM-1 mutant. This may suggest that the ICAM-1 mutation protects mice to some extent from liver damage.
WD-fed Icam1tmBay mice exhibit ameliorated MASH progression. Wild type (WT) mice (shown in black) and Icam1tmBay mice (shown in purple) were fed for 24 weeks with chow diet (CD) or Western diet (WD). Depicted are the endpoint values of WT CD-fed (n = 4–13), WT WD-fed (n = 7–15), Icam1tmBay CD-fed (n = 6), and Icam1tmBay WD-fed (n = 6–8). (A) Quantification of serum aspartate aminotransferase (AST). (B) Quantification of serum alanine aminotransferase (ALT). (C) Quantification of alkaline phosphatase (ALP). (D) Representative images of H&E-stained liver sections of the indicated mice strains (original magnification X 20, scale bar = 50 μm). (E) Non-alcoholic fatty liver disease (NAFLD) activity score (NAS). (F) Representative images of Sirius red-stained liver sections of the indicated mice strains (original magnification X 20, scale bar = 50 μm). (G) Quantification of Sirius red-positive area expressed as a percentage of the tissue area. (H) mRNA levels of collagen 1alpha (col1a) in liver tissue homogenates. For quantification, values are expressed as fold increase over the mean values obtained for liver tissue from CD-fed WT mice. (I) Hepatic triglycerides in liver tissue homogenates. (J) Free fatty acids (FFA) in liver tissue homogenates. Statistical significance was calculated by the two-way ANOVA. Values are represented as mean ± SD (A-C, E, G, H). * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001. Only differences between genotypes (WT and Icam1tmBay mice) within a diet (CD or WD) and between diets (CD and WD) within a genotype (WT and Icam1tmBay mice) were calculated.
Histological evaluation of H&E-stained liver sections revealed a marked increase in hepatocyte ballooning with micro- and macrosteatosis in both mouse strains after 24 weeks of WD (Fig. 4D). However, this was significantly less pronounced in the Icam1tmBay mice, as can be seen in the respective NAFLD activity score (NAS) (Fig. 4E). Moreover, Sirius red staining of liver sections revealed a marked increase in hepatic fibrosis in WT mice post WD feeding, whereas comparably treated mutant mice were less affected (Fig. 4F-G). In line with our observations of an increased WD-induced fibrosis, both mouse strains showed increased Col1a mRNA post WD feeding in liver tissue homogenates. This increase was less pronounced in the ICAM-1 mutant (Fig. 4H). No differences were observed in levels of hepatic triglycerides or free-fatty acids between the four experimental mice groups (Fig. 4I, J). Increased intrahepatic inflammatory immune cell proportions are a characteristic feature of progressing MASLD. Therefore, we next performed a detailed flow cytometric analysis of the major hepatic leukocyte groups. (Fig. 5A). Feeding with WD caused an overall increase in the frequencies of CD4+ T cells and macrophages, that was independent of the genotype, whereas the frequency of B cells was decreased. Under chow diet conditions, we observed lower proportions of hepatic CD8+ T cells in the ICAM-1 mutant. However, 24 weeks post WD feeding, both mouse strains showed the same frequencies of hepatic CD8+ T cells. The WD-induced increase in macrophages, which are central players in MASLD progression, was less pronounced in Icam1tmBay mice. This result is in line with a decreased expression of Mcp-1 mRNA as detected by RT-PCR on liver tissue homogenates. (Fig. 5B).
Increased WD-induced hepatic neutrophil frequencies in Icam1tmBay mice do not correlate with hepatic myeloperoxidase expression. Wild type (WT) mice (shown in black) and mice with dysfunctional ICAM-1 gene (Icam1tmBay) mice (shown in purple) were fed for 24 weeks with chow diet (CD) or Western diet (WD). Depicted are the endpoint values of WT CD-fed (n = 4–6), WT WD-fed (n = 7–8), Icam1tmBayCD-fed (n = 5–6), and Icam1tmBayWD-fed (n = 7–8). (A) Comparative immune cell analysis of liver by flow cytometry. Representative FACS dot plots illustrating the gating strategy are shown in Supplementary Fig. S1. Depicted are the percentages of CD45+ cells of CD4+ T cells (CD45+CD3+CD8-CD4+), CD8+ T cells (CD45+CD3+CD4-CD8+), B cells (CD45+CD3-CD19+), NK cells (CD45+NK1.1+), neutrophils (CD45+CD11b+Ly6G+), and macrophages (CD45+Ly6G-CD11b+F4/80+). (B) mRNA levels of monocyte chemoattractant protein 1 (Mcp1) in liver tissue homogenates. For quantification, values are expressed as fold increase over the mean values obtained for liver tissue from CD-fed WT mice. (C) Representative images of myeloperoxidase (MPO) stained liver sections of the indicated mice strains (original magnification X 20, scale bar = 50 μm). (D) Quantification of MPO positive area expressed as a percentage of the tissue area. Statistical significance was calculated by two-way ANOVA. Values are represented as mean ± SD (B, C). * p ≤ 0.05, ** p ≤ 0.01, **** p ≤ 0.0001. Only differences between genotypes (WT and Icam1tmBay mice) within a diet (CD or WD) and between diets (CD and WD) within a genotype (WT and Icam1tmBay mice) were calculated.
Interestingly, a WD induced increase in hepatic neutrophil frequencies, was only observed in the ICAM-1 mutant. To assess neutrophil-mediated liver damage, we performed immunohistochemical staining for MPO on liver sections (Fig. 5C). Most interestingly, the increased WD-induced neutrophil frequencies in Icam1tmBay mice did not result in an increased MPO signal intensity. In fact, the MPO positive area in liver sections from the WD-treated ICAM mutant was smaller than the area in respectively treated WT mice (Fig. 5D). Although this finding was not statistically significant, it correlated with the overall MASLD amelioration observed in mice with dysfunctional ICAM-1 expression.
Fecal microbiota profiles are altered by WD-feeding
To compare WD-induced changes in the gut microbiota of WT and ICAM-1tmbay mice, we used 16 S rRNA gene amplicon sequencing. Fecal samples were collected from WD-fed WT and ICAM-1tmbay mice at the start (0 weeks, 16 samples) and end (24 weeks, 14 samples) of the experimental feeding. Across the 30 samples sequenced, 897,246 high-quality, chimera-checked sequences (29,908 ± 15,279 per sample) were analyzed, representing 222 operational taxonomic units (OTUs) (135 ± 69 OTUs per sample). Sufficient sequencing depth for all samples was estimated based on the plateauing of the rarefaction curves (Supplemental Fig. S3). First, we assessed differences in bacterial profiles between the experimental groups at the start and end point of WD-treatment by meta non-metric multidimensional scaling (NMDS) visualization of Unifrac distances (Fig. 6A). WD feeding led to significantly altered microbiota profiles in both mouse strains. Alpha-diversity was then evaluated by calculating bacterial richness (i.e., number of observed OTUs) (Fig. 6B) and Shannon effective counts (Fig. 6C). At the starting point a higher diversity was observed in ICAM-1tmBay mutant mice, which decreased significantly after 24 weeks of WD feeding. Next, the relative abundance of major bacterial phyla was calculated (Fig. 6D-G). Whilst the proportions of Bacillota (formerly Firmicutes), Actinomycetota (formerly Actinobacteria) and Pseudomonadota (formerly Proteobacteria) were significantly increased and that of Bacteroidota (formerly Bacteroidetes) significantly decreased in feces of WD-fed WT mice, their relative abundance seemed to be unaffected by WD feeding in feces of the ICAM-1tmBay mutant mice. Both mouse strains exhibited equal relative abundances of Bacillota and Bacteroidota after WD feeding. In contrast, the relative abundances of Actinomycetota and Pseudomonadota were substantially higher in feces of the WT mice. The most obvious WD-induced differences at the family level (Fig. 6H-L) were an increase in the relative abundance of Bacteroidaceae and Peptostreptococcaceae and a decrease in the relative abundance of Muribaculaceaea which was unaffected by the mouse genotype. Interestingly, increased proportions of Clostridiaceae were only observed in WT mice, while these were practically absent in feces of ICAM-1tmBay mutants. In contrast, an increased relative abundance of Prevotellaceae, which was absent in the WT, was only seen in the feces of ICAM-1tmBay mutants.
WD feeding causes important shifts in gut microbiota diversity and composition. Fecal samples were collected from wild type (WT) mice (black) and mice with dysfunctional ICAM-1 gene (Icam1tmBay) (purple) before (0 weeks (0w)) and after 24 weeks (24w) of western diet (WD) feeding. Number of mice: WT 0w (n = 7), WT 24w (n = 7), Icam1tmBay 0w (n = 9), and Icam1tmBay 24w (n = 7). The diversity of operational taxonomic units (OTUs) within a given sample (alpha-diversity) and between samples (beta-diversity) was calculated in R using Rhea52. (A) Beta-diversity of experimental groups shown as meta non-metric multidimensional scaling (NMDS) plot of microbiota profiles based on generalized UniFrac distances from mice at the indicated time points. Individual time points that were considered for calculations have been marked within their respective circles. Alpha-diversity of experimental groups shown as (B) number of observed OTUs (richness), or (C) Shannon effective index. (D-G). Relative abundance at the phylum level. (H-L) Relative abundance at the family level. Statistical significance was calculated by PERMANOVA (A) or two-way ANOVA (B-L). Values are represented as mean ± SD (B-L). * p ≤ 0.05, ** p ≤ 0.01, ***p ≤ 0.001, **** p ≤ 0.0001. Only differences between genotypes (WT and Icam1tmBay mice) within a diet (CD or WD) and between diets (CD and WD) within a genotype (WT and Icam1tmBay mice) were calculated.
Discussion
MASLD progression involves a complex interplay of imbalanced inflammatory cell populations and inflammatory signals derived from several organs including liver, adipose tissue, gut and its enteric microbiota, making MASLD rather to a systemic disease1. The progression from steatosis to cirrhosis is driven by chronic inflammation of the injured liver, involving adhesion molecule-mediated immigration of inflammatory immune cells into the liver parenchyma3. However, these processes have not yet been sufficiently elucidated, which is prerequisite for identifying therapeutic targets.
We set up experiments to better define the role of the adhesion molecule ICAM-1 in WD-induced MASLD. Previously, increased ICAM-1 expression in adipose tissue has been shown in mice fed for 2, 3 or 6 months on a high-fat or high-fat/high-sugar diet14,17. Here we show that WD treatment also leads to increased ICAM-1 expression in liver, adipose tissue, colon, and small intestine of WT mice, pointing to an obesity-induced inflammatory status within those tissues and supporting the notion of their joint involvement in MASLD progression. ICAM-1 does not appear to be involved in WD-induced food intake and body fat gain, as there were no differences in overall body fat deposition between WD-fed WT mice and Icam1tmBay mice carrying the dysfunctional ICAM-1 gene. These observations corroborate the results of earlier studies where no differences in the weight of fat pads were detected in WD-fed WT-mice and ICAM-1null-mice18. Moreover, mice deficient for the ICAM-1 ligand Mac-1 and mice lacking β2 integrin function exhibited identical WD-induced weight gain and expanded fat mass18,19. WD-fed Icam1tmBay mice exhibited an increased insulin resistance and systemic inflammation, as evidenced by worsened glucose tolerance and increased circulating neutrophils and TNFα, suggesting a protective effect of ICAM-1 against metabolic dysregulation. In contrast, an overall improvement in glucose tolerance has been observed in WD-treated mice lacking beta2-integrin function19suggesting that other ICAM-1 ligands may counteract the negative impact of beta2-integrin ligands on glucose metabolism. Systemic glucose regulation is complex and involves several organs, including adipose tissue, where inflammatory status directly impacts on insulin sensitivity. Obesity is accompanied by the progressive recruitment of pro-inflammatory immune cells to adipose tissue, which contribute to the development of metabolic dysregulation20. Inflammatory macrophages, which differentiate from monocytes accumulate in adipose tissue and are thought to play a key role in adipose tissue inflammation21. In lean mice and humans, macrophages constitute 5% of cells in adipose tissue depots, but in obese rodents and humans macrophages are increased by up to 50% 21. The higher frequencies of macrophages in the adipose tissue of ICAM-1 mutants observed here could therefore be causally involved in the impaired glucose tolerance.
In contrast, to the worsened WD-induced metabolic phenotype upon ICAM-1 dysfunction, we observed a slower MASH progression in Icam1tmBay mice, reflected in lower serum liver damage levels, a lower NAS score and reduced fibrosis compared to respectively treated WT animals, suggesting that ICAM-1 contributes to disease progression. Amelioration of disease was accompanied by a smaller increase in the frequency of hepatic macrophages and decreased expression of MCP-1, the chemokine ligand known to be responsible for augmented infiltration of inflammatory monocytes22. As bone marrow-derived hepatic macrophages reportedly display an inflammatory phenotype and contribute to hepatocellular damage in experimental models of chronic liver injury and obesity23,24it would be reasonable to attribute reduced inflammatory macrophages in livers of Icam1tmBay mice to the observed amelioration of disease.
Neutrophils are the first immune cells to accumulate in insulin-sensitive tissues during obesity and they seem to contribute to diet-induced peripheral tissue insulin resistance25. In addition to the finding of increased circulating neutrophils in the ICAM-1 mutant in homeostasis, which corroborates earlier results9we observed higher percentages of neutrophils in adipose tissue and liver tissue of the WD-fed mutant, a phenotype which has also been seen in mice lacking beta2-integrin function19. We thereby conclude that WD-induced neutrophil migration to liver and fat can also occur independently of ICAM-1 and that increased neutrophil numbers in these tissues might result from the strongly increased levels of circulating neutrophils. This hypothesis fits with previously published data showing that acute LPS-induced neutrophil infiltration in the cremaster muscle and peritonitis model does not require ICAM-1 26. Expression of Myeloperoxidase (MPO), an enzyme predominantly released by neutrophils, correlates with the presence of MASH in patients and murine livers fibrosis, and MPO deficiency or pharmacological inhibition protects against MASH-induced liver injuries in mice27. Interestingly, higher frequencies of neutrophils in WD-fed Icam1tmBay mice did not translate into an increased inflammatory activity of these cells, as shown by a rather lower MPO expression in liver tissue in comparison to the WT situation. This is in line with the improved liver phenotype. The finding points to a reduced activation potential of neutrophils with dysfunctional ICAM-1, which would also explain why phagocytic capacity and ROS production of leukocytes in blood from mutant mice remained stable despite increased numbers of circulating neutrophils. Of note, previously it was shown that LPS induced neutrophil ICAM-1 expression and ICAM-1-mediated intracellular signaling caused increased phagocytosis and ROS generation26.
Diet-induced gut microbiota changes and a leaky barrier contribute to the systemic inflammation and disease progression of MASLD28. In addition to alterations in gut permeability, WD-induced gut microbiota changes can affect hepatic lipid and carbohydrate metabolism, inflammatory processes in liver and adipose tissue through activation of innate immunity, gut hormone production, and bile acid metabolism29. Moreover, a contribution of the enteric microflora to the constitutive expression of ICAM-1 in intestinal and liver tissue has been reported30. In future, gut microbiota manipulations could become a useful tool for the treatment of obesity-related diseases while potential influences of the gut microbiota on ICAM-1-expression and vice versa could be of interest. At the family level, fecal microbial composition of both mouse strains exhibited a substantial increase in the relative abundance of Bacteroidaceae and Peptostreptococcaceae after 24 weeks of WD. Increased relative abundance of Peptostreptococcaceae (phylum Bacillota) after WD feeding has already been demonstrated in previous studies and might present a marker of disease risk31. According to Iwamoto et al., Peptostreptococcaceae contribute to cholesterol conversion and might contribute to WD-induced hypercholesterolemia32. The WD-induced increase in relative abundance of Bacteroidaceae (phylum Bacteroidota) observed here also fits with previous publications reporting increased Bacteroidaceae in high-fat diet treated mice and a positive correlation of the genus Bacteroides (family Bacteroidaceae) with disease severity in patients with biopsy proven NASH33,34. Bacteroides-associated hepatic pathology is associated with changes in bacterial metabolites such as a decrease in short chain fatty acids or increases in deoxycholic acid and carbohydrates which can promote liver inflammation35. Independent of the genetic background, WD caused a drastic decrease in the Muribaculaceae, which represent a large proportion of the mouse gut Bacteroidota. This corroborates former studies showing a decreased relative abundance of Muribaculaceae upon WD feeding and a negative correlation with obesity36. Interestingly, a positive correlation of these bacteria with life span has been observed in mice. Furthermore, feces of WD-fed WT mice, but not of ICAM-1tmbay mutant mice, showed a significantly higher relative abundance of Clostridiaceae. A WD-induced expansion of Clostridaceae (phylum Bacillota) has been reported previously37. Clostridium species contribute to bile acid metabolism and, according to Xiao et al.38expansion of these bacteria might contribute to an altered bile acid metabolism upon WD feeding. Unexpectedly, we observed an increased relative abundance of Prevotellaceae (phylum Bacteroidota) in feces of WD fed ICAM-1tmbay mice, which was not seen in WT mice. According to the literature, the occurrence of Prevotellaceae is negatively related to a high-fat diet in mice, pigs and humans39. In addition, Segatella copri (formerly Prevotella copri)40 has been reported to improve the abnormal glucose metabolism in diabetic mice and ameliorated histological damage of pancreas, liver and colon41. An increased relative abundance in the Prevotellaceae might therefore have contributed to improvement of liver damage in the ICAM-1tmbay mutant.
Collectively, our study reveals marked changes in the gut microbiota of WT and ICAM-1tmbay mice due to WD feeding, and differences in the microbiota between both mouse strains, which might have contributed to the different phenotypes.
In summary, we have shown that ICAM-1 has no impact on WD-induced body fat gain, protects from systemic glucose tolerance and differentially alters local tissue inflammation by ameliorating adipose tissue inflammation and partially aggravating MASH progression. This points to a variety of organ-specific roles for ICAM-1 and the potential of liver-specific targeting of ICAM-1 for treatment of MASLD.
Methods
Housing, mice, and dietary treatments
Animals were bred at RWTH Aachen University and housed in specific pathogen-free conditions with 12 h light/dark cycles and water and food available ad libitum. Western diet (WD) treatments were performed with 8 to 12-week-old male only, as female mice are more resistant to diet-induced metabolic disorders42. ICAM-1 mutant (Icam1tm1Bay) mice9 were originally derived from The Jackson Laboratory, strain #:002127 and wild type (WT) mice on C57BL/6J background were originally derived from Janvier. Initially Icam1tm1Bay mice were rederived by in-vitro fertilization of C57BL/6J mice. The absence of ICAM-1 was verified by Western blots on liver and adipose tissue (Supplementary Fig. S4). After mating to homozygosity, the animals were kept in groups. Mice were fed chow diet (CD) (9 kcal % fat, 24 kcal % protein, 67 kcal % carbohydrates) (ssniff, Soest, Germany, rat/mouse–maintenance) or WD (40 kcal % fat (vegetable fats, 20 kcal % fructose, 2% cholesterol) (Brogaarden, Lynge, Denmark; cat. no. D16022301). The ingredients of the WD are listed in the Supplementary materials Table S1. To reduce unwanted variability only mice from the same barrier and room weighing at least 25 g were included in the study and were kept in individual cages for the duration of the experiment. Experimental mice were kept in single cages, which were changed weekly and provided with fresh food and water. Food intake was assessed weekly over the duration of 12 or 24 weeks of WD treatment by calculating the difference between the grams of food added and the grams of food remaining in the cage.
At the end of the experiment, the animals were sacrificed by CO2 inhalation and subsequent cervical dislocation. Afterwards, blood was taken by cardiac puncture and organs were removed. All WD experiments except the µCT imaging were repeated in at least two independent experimental setups.
Serum and liver biochemical measurements
Serum aspartate aminotransferase (AST), serum alanine aminotransferase (ALT), alkaline phosphatase (ALP), glucose, triglyceride, and cholesterol levels in serum were measured by the Central Laboratory Facility of the University Hospital, RWTH Aachen.
Determination of the intrahepatic triglyceride concentration was performed in accordance with the manufacturer’s instructions of the Instruchemie LiquiColor mono Kit (Instruchemie, Delfzijl, the Netherlands). Intrahepatic free fatty acid quantification was measured with the FFA quantification kit (Abcam, Cambridge, UK, cat. no ab65345) as instructed by the manufacturer.
Serum cytokine concentrations were measured by a bead-based immunoassay technique using the LegendPlex Mouse Inflammation Panel (13-plex) (Biolegend, San Diego, cat. no 740446) following the manufacturer’s instructions. Samples were acquired on a Canto-II flow cytometer (BD Biosciences) and data were analyzed using the LegendPlex data analysis software.
Glucose tolerance test (GTT)
GTT were performed on the penultimate day of the 24-week WD. Mice were fasted for 6 h and blood glucose was measured using an Accu-Check® Aviva meter (Roche, Basel, Switzerland), via one drop of blood taken from the animal’s tail, every 15 min for 2 h following intraperitoneal administration of 2 g/kg glucose.
Micro-Computed tomography (µCT)
In vivo µCT imaging was performed using a hybrid µCT-Fluorescence Tomography system (MILabs B.V., Houten, the Netherlands) in the ultra-focus fast scan mode. The X-ray tubes of the µCT were operated at a voltage of 65 kV with a current of 0.13 mA. To cover the entire mouse, a continuous rotation scan was performed with one full rotation, exposure time of 75 ms, total scan duration of 27 s, and dose estimation of 35 mGy. After acquisition, volumetric data sets were reconstructed at an isotropic voxel size of 140 μm using MILabs Auto Rec 12.00. The fat-containing tissue regions, which appear hypo-intense in the µCT data, were segmented using an automated segmentation method with interactive correction of segmentation errors using the Imalytics Preclinical 3.0.2.5 software (Gremse-IT GmbH, Aachen, Germany)43.
Histological stainings
All stainings were performed on 4 μm paraffin sections. Images were acquired using an Axioplan2 microscope (Carl Zeiss Microscopy, Oberkochen, Germany) and Zen lite 3.2 software (Carl Zeiss Microscopy, Oberkochen, Germany). Histopathological scoring of hematoxylin and eosin stained sections from liver and their validation was performed blinded via a non-alcoholic fatty liver disease (NAFLD) activity score (NAS)44. The total hepatic parenchymal area and the Sirius red positive area were estimated by means of a size marker using ImageJ software (version 1.50; National Institutes of Health Bethesda, MD)45. Briefly, Sirius red-stained liver sections were imaged by tile scanning at 10x magnification to capture the entire tissue section. Images were converted to TIFF format and TIFF images were split into Red, Green, and Blue channels. Sirius red positive areas were manually thresholded in the green channel since it shows the best separation. The thresholded region was calculated as a fraction of the whole tissue area. Blood vessels were not excluded from the thresholding analysis (Reference: https://imagej.net/ij/docs/examples/stained-sections/index.html).
For immunohistochemical staining of ICAM-1, CD68, and MPO, formalin-fixed tissue sections were first permeabilized and heat-induced epitope retrieval in citrate buffer was performed. Subsequently, the sections were blocked with 2% goat serum, and incubated with a rabbit-anti-ICAM-1 or -MPO antibody overnight at 4 °C. Then, the sections were treated with biotinylated goat-anti-rabbit IgG for 30 min at room temperature. Antibody binding was visualized with the ABC kit and diaminobenzidine reagent as a chromogen (both from Vector, Burlingame, USA). Nuclei were stained with hematoxylin. Antibodies are listed in the Supplementary materials Table S2.
Western blot analysis
Protein isolation and western blot analysis was performed as described previously46. Briefly, tissues were homogenized in radioimmunoprecipitation assay (RIPA) buffer containing protease inhibitors (order no 3755.1, Carl Roth GmbH + Co KG, Karlsruhe, Germany) using the protein_01.01 setting on the gentle MACS dissociator (Miltenyi Biotec, Bergisch Gladbach, Germany). Cell lysates were separated on SDS-10% polyacrylamide gels and electrophoretically transferred onto polyvinylidene difluoride microporous membranes (Immobilon-P, Millipore Corp., Bedford, Mass.). After blocking, blots were incubated with primary antibodies (either ICAM-1 recombinant rabbit monoclonal antibody (order no MA5-43105, Life Technologies GmbH Darmstadt, Germany) or β-actin mouse monoclonal antibody (order no sc-47778, Santa Cruz Biotechnology, Dallas Texas)) at a 1:2000 dilution. Subsequently blots were washed and incubated with secondary goat anti-rabbit (order no P0448) or goat anti-mouse (order no P0161) immunoglobulin antibody conjugated with horseradish peroxidase (both from Agilent Dako, Santa Clara, California) at 1:2000 dilution. Visualization was performed with the Super Signal West Dura Kit (order no 34075, Life Technologies GmbH, Darmstadt, Germany) on a LAS 3000 imager (Fujifilm, Düsseldorf, Germany). Molecular weights of proteins were determined according to Spectra™ Multicolor Broad Range Protein ladder (order no 26634, Life Technologies GmbH Darmstadt, Germany).
Flow cytometry
All flow cytometric measurements were performed on a Canto-II cytometer (BD Biosciences). Single cell suspensions were stained directly using combinations of the monoclonal antibodies listed in Supplementary materials Table S. Representative FACS dot plots illustrating the gating strategy are shown in Supplementary Fig. S1. Data were analyzed by FlowJo 10.2 software (Tree Star, Ashland, OR, USA).
RNA isolation and cDNA synthesis
Tissues were stored in RNA-Later® at −20 °C until processing. Total RNA was extracted from samples using the RNeasy kit including a DNA digestion step with the RNase Free DNAse set (both from Qiagen GmbH, Hilden, Germany) according to the manufacturer’s description. The tissue was homogenized using the RNA_02.01 setting on the gentle MACS dissociator (Miltenyi Biotec, Bergisch Gladbach, Germany). RNA concentration was photometrically measured and integrity was checked on a Nanodrop ND-100 (Thermo Fisher Scientific, USA). cDNA was synthesized from 1 µg RNA using the Transcriptor FS cDNA S-Kit (Roche Diagnostic GmbH, Mannheim, Germany) following the manufacturer’s instructions.
Gene expression analysis by Real-Time PCR
Real-time polymerase chain reactions (RT-PCR) were performed in duplicate on a 7300 RT-PCR system with 7000 System SDS Software Version 1.2.3 (Applied Bioscience, Darmstadt, Germany) using the quantitative (q)PCR Master Mix for SYBR Green I (Eurogentec, Cologne, Germany). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH was used as endogenous control for normalization). Primer sequences are listed in Supplementary materials Table S4.
DNA isolation from feces, 16 S rRNA gene amplicon sequencing, and data analysis
Stool samples were processed by the Functional Microbiome Research Group at the Faculty of Medicine, RWTH Aachen University. Metagenomic DNA was isolated from samples and negative controls using a modified protocol according to Godon et al.47 16 S rRNA gene amplicons were generated with primers 341 F-785R48 and a two-step PCR (15 + 10 cycles)49 using a pipetting robot. Sequencing was performed as described in detail previously50. Individual libraries were diluted to a final concentration of 4 nM and 5 µl of each library were added to the final pool. The 16 S rRNA gene amplicon libraries were sequenced with dual barcodes in paired-end mode (2 × 300 nt) using a MiSeq sequencer (Illumina, Inc.). Data were analyzed with an updated version of a previously described workflow50. Raw reads were processed using an in-house developed pipeline (www.imngs.org)51,52 based on UPARSE. In brief, sequences were demultiplexed and trimmed to the first base with a quality score < 3. The pairing, chimaera filtering and OTU clustering (97% identity) was done using USEARCH 11.053. Sequences with less than 350 and more than 500 nucleotides and paired reads with an expected error > 3 were excluded from the analysis. The remaining reads were trimmed by ten nucleotides on each end to avoid GC bias and non-random base composition. Operational taxonomic units (OTUs) were clustered at 97% sequence similarity and only those with a relative abundance > 0.25% in at least one sample were kept. Sequence alignment and taxonomic classification were conducted with SINA 1.6.1, using the taxonomy of SILVA release 12854. Downstream analysis was performed in the R programming environment using Rhea (https://lagkouvardos.github.io/Rhea/)52. OTU tables were normalized to account for differences in sequence depth. Βeta-diversity was computed based on generalized UniFrac distances55. Αlpha-diversity was assessed based on species richness and Shannon effective diversity56.
Statistical analysis
Unless otherwise indicated, statistical analysis was performed with GraphPad Prism software (version 10 GraphPad, La Jolla, CA, USA). Data are presented as mean ± Standard Deviation (SD). Significance values were calculated using the Student’s t-test when comparing two groups or two-way analysis of variance (ANOVA) and Tukey post-test. Values of p < 0.05 were considered significant (* p < 0.05, ** p < 0.01, *** p < 0.001, and **** p ≤ 0.0001). Tissues for downstream analysis were chosen randomly and based on availability and quality. The ROUT method in the GraphPad Prism software was used to identify outliers.
Data availability
The microbiota sequencing data presented in this study were submitted to the Sequence Read Archive and are openly available under the accession number PRJEB80543.
References
Targher, G., Byrne, C. D. & Tilg, H. MASLD: a systemic metabolic disorder with cardiovascular and malignant complications. Gut 73, 691–702. https://doi.org/10.1136/gutjnl-2023-330595 (2024).
Younossi, Z. M. et al. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): A systematic review. Hepatology77, 1335–1347. https://doi.org/10.1097/HEP.0000000000000004 (2023).
Huby, T. & Gautier, E. L. Immune cell-mediated features of non-alcoholic steatohepatitis. Nat. Rev. Immunol.22, 429–443. https://doi.org/10.1038/s41577-021-00639-3 (2022).
Peiseler, M. et al. Immune mechanisms linking metabolic injury to inflammation and fibrosis in fatty liver disease - Novel insights into cellular communication circuits. J. Hepatol.77, 1136–1160. https://doi.org/10.1016/j.jhep.2022.06.012 (2022).
Shetty, S., Lalor, P. F. & Adams, D. H. Liver sinusoidal endothelial cells - Gatekeepers of hepatic immunity. Nat. Rev. Gastroenterol. Hepatol.15, 555–567. https://doi.org/10.1038/s41575-018-0020-y (2018).
Bui, T. M., Wiesolek, H. L. & Sumagin, R. ICAM-1: A master regulator of cellular responses in inflammation, injury resolution, and tumorigenesis. J. Leukoc. Biol. 108, 787–799. https://doi.org/10.1002/JLB.2MR0220-549R (2020).
Ramos, T. N., Bullard, D. C. & Barnum, S. R. ICAM-1: Isoforms and phenotypes. J. Immunol.192, 4469–4474. https://doi.org/10.4049/jimmunol.1400135 (2014).
Bullard, D. C. et al. Expression of a single ICAM-1 isoform on T cells is sufficient for development of experimental autoimmune encephalomyelitis. Eur. J. Immunol.44, 1194–1199. https://doi.org/10.1002/eji.201344023 (2014).
Sligh, J. E. Jr. et al. Inflammatory and immune responses are impaired in mice deficient in intercellular adhesion molecule 1. Proc. Natl. Acad. Sci. U. S. A.90, 8529–8533. https://doi.org/10.1073/pnas.90.18.8529 (1993).
Kolb, H. Obese visceral fat tissue inflammation: From protective to detrimental?. BMC Med.20, 494. https://doi.org/10.1186/s12916-022-02672-y (2022).
Sookoian, S. et al. Circulating levels and hepatic expression of molecular mediators of atherosclerosis in nonalcoholic fatty liver disease. Atherosclerosis 209, 585–591. https://doi.org/10.1016/j.atherosclerosis.2009.10.011 (2010).
Leinonen, E. et al. Insulin resistance and adiposity correlate with acute-phase reaction and soluble cell adhesion molecules in type 2 diabetes. Atherosclerosis 166, 387–394. https://doi.org/10.1016/s0021-9150(02)00371-4 (2003).
Widjaja, N. A., Caesar, L. A., Nova, S. & Ardianah, E. Beyond the scale: Investigating adiponectin, ICAM-1, and VCAM-1 as metabolic markers in obese adolescents with metabolic syndrome. J. Obes.2023, 4574042. https://doi.org/10.1155/2023/4574042 (2023).
Brake, D. K., Smith, E. O., Mersmann, H., Smith, C. W. & Robker, R. L. ICAM-1 expression in adipose tissue: effects of diet-induced obesity in mice. Am. J. Physiol. Cell. Physiol. 291, C1232–1239. https://doi.org/10.1152/ajpcell.00008.2006 (2006).
King, P. D. et al. Novel isoforms of murine intercellular adhesion molecule-1 generated by alternative RNA splicing. J. Immunol.154, 6080–6093 (1995).
Murano, I. et al. Dead adipocytes, detected as crown-like structures, are prevalent in visceral fat depots of genetically obese mice. J. Lipid Res.49, 1562–1568. https://doi.org/10.1194/jlr.M800019-JLR200 (2008).
Masi, L. N. et al. Combination of a high-fat diet with sweetened condensed milk exacerbates inflammation and insulin resistance induced by each separately in mice. Sci. Rep.7, 3937. https://doi.org/10.1038/s41598-017-04308-1 (2017).
Robker, R. L., Collins, R. G., Beaudet, A. L., Mersmann, H. J. & Smith, C. W. Leukocyte migration in adipose tissue of mice null for ICAM-1 and Mac-1 adhesion receptors. Obes. Res. 12, 936–940. https://doi.org/10.1038/oby.2004.114 (2004).
Meakin, P. J. et al. Mice lacking beta2-integrin function remain glucose tolerant in spite of insulin resistance, neutrophil infiltration and inflammation. PLoS One10, e0138872. https://doi.org/10.1371/journal.pone.0138872 (2015).
Sam, S. & Mazzone, T. Adipose tissue changes in obesity and the impact on metabolic function. Transl. Res.164, 284–292. https://doi.org/10.1016/j.trsl.2014.05.008 (2014).
Weisberg, S. P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808. https://doi.org/10.1172/JCI19246 (2003).
Preisser, L. et al. IL-34 and macrophage colony-stimulating factor are overexpressed in hepatitis C virus fibrosis and induce profibrotic macrophages that promote collagen synthesis by hepatic stellate cells. Hepatology60, 1879–1890. https://doi.org/10.1002/hep.27328 (2014).
Krenkel, O. et al. Myeloid cells in liver and bone marrow acquire a functionally distinct inflammatory phenotype during obesity-related steatohepatitis. Gut 69, 551–563. https://doi.org/10.1136/gutjnl-2019-318382 (2020).
Baeck, C. et al. Pharmacological inhibition of the chemokine C-C motif chemokine ligand 2 (monocyte chemoattractant protein 1) accelerates liver fibrosis regression by suppressing Ly-6 C(+) macrophage infiltration in mice. Hepatology59, 1060–1072. https://doi.org/10.1002/hep.26783 (2014).
D’Souza, R. F. et al. alpha1-Antitrypsin A treatment attenuates neutrophil elastase accumulation and enhances insulin sensitivity in adipose tissue of mice fed a high-fat diet. Am. J. Physiol. Endocrinol. Metab. 321, E560–E570. https://doi.org/10.1152/ajpendo.00181.2021 (2021).
Woodfin, A. et al. ICAM-1-expressing neutrophils exhibit enhanced effector functions in murine models of endotoxemia. Blood 127, 898–907. https://doi.org/10.1182/blood-2015-08-664995 (2016).
Koop, A. C. et al. Therapeutic targeting of myeloperoxidase attenuates NASH in mice. Hepatol. Commun. 4, 1441–1458. https://doi.org/10.1002/hep4.1566 (2020).
Mohammad, S. & Thiemermann, C. Role of metabolic endotoxemia in systemic inflammation and potential interventions. Front. Immunol. 11, 594150. https://doi.org/10.3389/fimmu.2020.594150 (2020).
Tran, H. Q. et al. Western diet-induced adipose inflammation requires a complex gut microbiota. Cell. Mol. Gastroenterol. Hepatol.9, 313–333. https://doi.org/10.1016/j.jcmgh.2019.09.009 (2020).
Komatsu, S., Berg, R. D., Russell, J. M., Nimura, Y. & Granger, D. N. Enteric microflora contribute to constitutive ICAM-1 expression on vascular endothelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 279, G186–191. https://doi.org/10.1152/ajpgi.2000.279.1.G186 (2000).
Volynets, V. et al. Intestinal barrier function and the gut microbiome are differentially affected in mice fed a Western-style diet or drinking water supplemented with fructose. J. Nutr.147, 770–780. https://doi.org/10.3945/jn.116.242859 (2017).
Iwamoto, J. et al. Western diet changes gut microbiota and ameliorates liver injury in a mouse model with human-like bile acid composition. Hepatol. Commun.5, 2052–2067. https://doi.org/10.1002/hep4.1778 (2021).
Boursier, J. et al. The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology 63, 764–775. https://doi.org/10.1002/hep.28356 (2016).
Singh, R. P., Halaka, D. A., Hayouka, Z. & Tirosh, O. High-fat diet induced alteration of mice microbiota and the functional ability to utilize fructooligosaccharide for ethanol production. Front. Cell. Infect. Microbiol.10, 376. https://doi.org/10.3389/fcimb.2020.00376 (2020).
den Besten, G. et al. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 54, 2325–2340. https://doi.org/10.1194/jlr.R036012 (2013).
Yang, M. et al. Western diet contributes to the pathogenesis of non-alcoholic steatohepatitis in male mice via remodeling gut microbiota and increasing production of 2-oleoylglycerol. Nat. Commun.14, 228. https://doi.org/10.1038/s41467-023-35861-1 (2023).
Jena, K. et al. Intestinal microbiota remodeling protects mice from western diet-induced brain inflammation and cognitive decline. Cells11, https://doi.org/10.3390/cells11030504 (2022).
Xiao, L. et al. High-fat feeding rather than obesity drives taxonomical and functional changes in the gut microbiota in mice. Microbiome 5, 43. https://doi.org/10.1186/s40168-017-0258-6 (2017).
Qiao, F. et al. Alteration and the function of intestinal microbiota in high-fat-diet- or genetics-induced lipid accumulation. Front. Microbiol.12, 741616. https://doi.org/10.3389/fmicb.2021.741616 (2021).
Hitch, T. C. A. et al. A taxonomic note on the genus prevotella: Description of four novel genera and emended description of the genera hallella and xylanibacter. Syst. Appl. Microbiol.45, 126354. https://doi.org/10.1016/j.syapm.2022.126354 (2022).
Yang, C. et al. Prevotella copri alleviates hyperglycemia and regulates gut microbiota and metabolic profiles in mice. mSystems9, e0053224. https://doi.org/10.1128/msystems.00532-24 (2024).
Oraha, J., Enriquez, R. F., Herzog, H. & Lee, N. J. Sex-specific changes in metabolism during the transition from Chow to high-fat diet feeding are abolished in response to dieting in C57BL/6J mice. Int. J. Obes. (Lond). 46, 1749–1758. https://doi.org/10.1038/s41366-022-01174-4 (2022).
Gremse, F. et al. Imalytics preclinical: Interactive analysis of biomedical volume data. Theranostics6, 328–341. https://doi.org/10.7150/thno.13624 (2016).
Kleiner, D. E. et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology41, 1313–1321. https://doi.org/10.1002/hep.20701 (2005).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH image to ImageJ: 25 years of image analysis. Nat. Methods9, 671–675. https://doi.org/10.1038/nmeth.2089 (2012).
Hollnagel, A., Grund, C., Franke, W. W. & Arnold, H. H. The cell adhesion molecule M-cadherin is not essential for muscle development and regeneration. Mol. Cell. Biol.22, 4760–4770. https://doi.org/10.1128/MCB.22.13.4760-4770.2002 (2002).
Godon, J. J., Zumstein, E., Dabert, P., Habouzit, F. & Moletta, R. Molecular microbial diversity of an anaerobic digestor as determined by small-subunit rDNA sequence analysis. Appl. Environ. Microbiol. 63, 2802–2813. https://doi.org/10.1128/aem.63.7.2802-2813.1997 (1997).
Klindworth, A. et al. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res.41, e1. https://doi.org/10.1093/nar/gks808 (2013).
Berry, D., Ben Mahfoudh, K., Wagner, M. & Loy, A. Barcoded primers used in multiplex amplicon pyrosequencing bias amplification. Appl. Environ. Microbiol. 77, 7846–7849. https://doi.org/10.1128/AEM.05220-11 (2011).
Lagkouvardos, I. et al. Gut metabolites and bacterial community networks during a pilot intervention study with flaxseeds in healthy adult men. Mol. Nutr. Food Res.59, 1614–1628. https://doi.org/10.1002/mnfr.201500125 (2015).
Lagkouvardos, I. et al. A comprehensive open resource of processed 16S rRNA microbial profiles for ecology and diversity studies. Sci. Rep. 6, 33721. https://doi.org/10.1038/srep33721 (2016).
Lagkouvardos, I., Fischer, S., Kumar, N. & Clavel, T. Rhea: A transparent and modular R pipeline for microbial profiling based on 16S rRNA gene amplicons. PeerJ5, e2836. https://doi.org/10.7717/peerj.2836 (2017).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461. https://doi.org/10.1093/bioinformatics/btq461 (2010).
Pruesse, E., Peplies, J. & Glockner, F. O. SINA: Accurate high-throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics28, 1823–1829. https://doi.org/10.1093/bioinformatics/bts252 (2012).
Chen, J. et al. Associating microbiome composition with environmental covariates using generalized UniFrac distances. Bioinformatics28, 2106–2113. https://doi.org/10.1093/bioinformatics/bts342 (2012).
Jost, L. Partitioning diversity into independent alpha and beta components. Ecology 88, 2427–2439. https://doi.org/10.1890/06-1736.1 (2007).
Acknowledgements
This research was funded by the DFG (German Research Foundation)–Project-ID 403224013–SFB 1382 to T.C., N.W., F.K., and A.S. This work was supported by the “Immunohistochemistry facility”, a core facility of the Interdisciplinary Center for Clinical Research (IZKF) Aachen within the Faculty of Medicine at RWTH Aachen University.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization: S.E., and A.S.; methodology: S.E., L.G., S.H., S.D., and A.Se.; software: S.E., S.S., S.H., N.T.; investigation: S.E., L.G., S.H., T.R.; resources: N.W., F.K., T.C.; funding acquisition: A.S., N.W., F.K., and T.C.; supervision: A.S. and N.W.; original draft preparation: A.S., S.E., N.T., N.W., and T.C.; review and editing: A.Se., L.G., S.S., S.H., T.R., S.D., and F.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declarations
All efforts were made to minimize the number of animals used and their suffering. The study was approved by the regional authorities for nature, environmental, and consumer protection of North Rhine-Westphalia (LANUV, Recklinghausen, Germany) and the respective Committee (Permit Number: 81-02.04.2017.A429). All experiments were performed in accordance with the relevant guidelines and regulations laid down by the national authorities of the LANUV and treatments fulfilled the criteria of the German administrative panel on laboratory animal care. Authors complied with the ARRIVE guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Eswaran, S., Gebert, L., Schraven, S. et al. Intercellular adhesion molecule-1 protects against adipose tissue inflammation and insulin resistance but promotes liver disease activity in western-diet fed mice. Sci Rep 15, 25884 (2025). https://doi.org/10.1038/s41598-025-11555-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-11555-0