Abstract
Acrylonitrile (ACN), a prevalent environmental pollutant, is associated with disruptions in male reproductive health. Recently, Portulaca oleracea polysaccharide (POP) has garnered considerable attention for its ability to modulate gut microbiota and demonstrate antioxidative properties. However, its the mechanisms of reproductive damage remain poorly understood. To investigate POP’s protective effects against ACN-induced reproductive damage, we orally gavaged ACN to male mice (26 mg/kg) to induce reproductive toxicity, then intervened with POP (200 mg/kg). This study assessed the impact of ACN exposure and evaluated POP intervention on gut microbiota, Metabolic Profiles, and reproductive function. We performed 16S rRNA gene amplicon sequencing of fecal samples (n = 6 ACN, POP and healthy controls) and untargeted metabolomic profiling using LC–MS analysis in mice exposed to ACN and treated with POP. Compared with controls, ACN exposure induced significant gut microbiota dysbiosis, characterized by altered relative abundances of specific genera, including Lactobacillus, recombinant Escherichia coli HT002, Alloprevotella, Rikenella, Clostridia_UCG-014, Turicibacter, and Lactococcus. These changes were accompanied by metabolic disturbances, including a significant decrease in the metabolite 2-aminopropenoic acid (VIP > 1, p < 0.05 and fold change (FC) > 1.2), which was significantly increased following POP intervention (p < 0.05). Furthermore, POP alleviated ACN-induced testicular tissue damage, oxidative stress, disruption in glucose-lipid and energy metabolism, and improved the sperm capacitation rate. Integrative analyses revealed a strong association between gut microbiota imbalance, metabolic alterations, and reproductive dysfunction, highlighting the protective role of POP in mitigating ACN-induced adverse effects. These findings demonstrate that POP protects reproductive health by mitigating toxin-induced damage and potentially enhancing reproductive function under environmental and metabolic stress. It also holds promise for functional food and drug development.
Similar content being viewed by others
Introduction
According to the World Health Organization’s (WHO) 2023 report, the global infertility rate ranges from 15 to 20%, affecting approximately 186 million people. Male factors contribute to 40–50% of infertility cases, primarily due to declining semen quality, abnormal hormone levels, and genetic factors1,2,3,4. In recent years, the increasing prevalence of male reproductive system abnormalities has heightened concerns about male reproductive health5. Studies suggest that semen quality may serve as an indicator of the impact of modern environmental factors on male health, with both sperm quantity and quality showing a declining trend6. Numerous studies demonstrate that exposure to exogenous chemicals in daily life can impair reproductive function7,8. Despite their societal benefits, these chemicals pose significant risks to reproductive health due to their potential toxicity.
Acrylonitrile (ACN), a highly toxic organic nitrile widely used in acrylic fibers, plastics, and consumer products, can enter the body through ingestion, inhalation, or dermal absorption. Studies have demonstrated that ACN exposure induces severe male reproductive dysfunction, characterized by dose-dependent reductions in sperm viability and pregnancy rates, along with increased sperm abnormalities and stillbirths. These findings underscore the urgent need to address ACN-induced reproductive toxicity and its broader implications for human health9,10,11. Current research on ACN reproductive toxicity has primarily focused on oxidative stress and apoptosis, yet its specific mechanisms involving gut microbiota remain unclear. Emerging evidence indicates that environmental pollutants can significantly alter gut microbiota composition, contributing to a wide range of disorders, including metabolic dysregulation, immune dysfunction, neurodevelopmental impairments, and reproductive defects12,13,14. The gut microbiota plays a pivotal role in regulating physiological processes and is essential for maintaining overall host health15. This suggests a potential link between gut microbiota dysbiosis and gonadal development, warranting further investigation. However, no studies to date have specifically examined the effects of ACN exposure on gut microbiota composition. Given its environmental persistence and toxicity, ACN poses a significant threat to reproductive health, yet the mechanisms by which it modulates gut microbiota to impair reproductive function remain poorly understood. Recently, plant-derived polysaccharides have gained increasing attention for their ability to regulate gut microbiota, support immune function, and protect the intestinal mucosa16. High-molecular-weight polysaccharides have also shown therapeutic potential in mitigating reproductive damage17. Portulaca oleracea, a medicinal and edible herb rich in α-unsaturated fatty acids, β-carotene, alkaloids, and polysaccharides, exhibits antioxidant properties by modulating reactive oxygen species (ROS) and enhancing the antioxidant enzyme defense system18.
Several studies have shown that POP shows unique potential in regulating the balance of gut microbiota, enhancing gut barrier function, reducing inflammatory response, and improving the status of gut-related diseases19,20,21. Nevertheless, there is currently no published research on how POP regulate gut flora to alleviate reproductive impairment. In view of the potential reproductive risk of ACN environmental exposure and the unique potential of POP in regulating intestinal flora, this study used untargeted metabolomics and intestinal microbial detection methods to deeply analyze the effects of ACN on intestinal-testis axis, and explain part of POP in maintaining intestinal health and improving ACN. This research aims to provide a scientific foundation for the further processing of Portulaca oleracea and the development of functional foods with potential health benefits.
Methods
Animals and experimental design
Sixty adult male ICR mice of (SPF grade, weighing between 16 and 18 grams, and thirty-six female ICR mice, (weighing between 22 and 25 grams, were procured from the Experimental Animal Center of Ningxia Medical University (License No.: SCXK (Ning) 2020-0001). The mice were accommodated in a specific pathogen-free ((SPF) environment, with unrestricted access to water and standard feed (provided by Jiangsu Cooperative Medicine & Bioengineering Co., Ltd. The environmental conditions encompassed a temperature range of 21–25°C, a relative humidity of 40–70%, and a 12-h alternating light-dark cycle. All animal experimentation adhered to the ARRIVE guidelines and was sanctioned by the Animal Ethics Committee of Ningxia Medical University (Approval Number: IACUC-NYLAC-2023-001). The conduct of all animal studies was in accordance with the American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals (2020) and the Guide for the Care and Use of Laboratory Animals. All methodologies were executed in strict accordance with the ARRIVE guidelines 2.0.
ACN was purchased from Energy Chemical and dissolved in purified water at a concentration of 100 mg/mL. Vitamin E dissolved in peanut oil at a concentration of 100 mg/mL. The mice were randomly divided into six groups based on body weight. Control, ACN (26 mg/kg), this dose was selected to ensure 100% mouse survival throughout the experimental period. ACN (26 mg/kg) + Vitamin E (100 mg/kg), and three POP intervention groups with ten mice per group (n=10). All groups had ad libitum access to food and water. After three days of acclimatization, mice in the control group were administered physiological saline by oral gavage, while all other groups received ACN (26 mg/kg) once every morning for six consecutive days, followed by one rest day. This dosage was determined from a preliminary experiment to be safe (ensuring no mortality). Two hours after ACN administration, the intervention groups were given 50 mg/kg, 100 mg/kg, and 200 mg/kg doses of POP and 100mg/kg doses of Vitamin E. After eight weeks of gavage exposure, three mice from each group were randomly selected for mating experiments (male: female=1:2). The remaining mice were fasted but given free access to water for 13–15 h, and sacrificed the following day for various measurements.
After three days of acclimation, mice were weighed to record the initial body weight. During the 8-week modeling period, food intake and body weight were recorded weekly, and the final body weight was measured before the experiment ended to calculate weight gain rate. Weekly feed weight was also measured to assess food consumption. At the end of the experiment, fecal samples from each mouse were collected and stored at − 80 °C for further analysis. Blood samples were also collected and stored at − 80 °C for biochemical analyses. All mice were euthanized by cervical dislocation, after which the testes and epididymides were immediately removed. Epididymal sperm samples were collected, and the testes were weighed. The testes were then flash-frozen in liquid nitrogen and stored at − 80 °C for further processing. Data on body and testicular weights are provided in Table S1.
Major reagents and instruments
Portulaca oleracea polysaccharide (Batch No: QZ220717, purity 50%, Xi’an Qingzhi Biotechnology Co., Ltd.), Acrylonitrile (Batch No: D9RDREDQ, Energy Chemical), Corn oil (Golden Dragon Fish), Vitamin E (TF541-100mL, Shanghai Ron Chemical Technology Co., Ltd), Physiological saline, Liquid nitrogen, Absolute ethanol, Xylene (Beijing Beilian Fine Chemicals Development Co., Ltd.).
SAS-II Sperm Quality Analyzer (Saize Medical Technology, Beijing, China), AX224ZH Precision Electronic Balance (Ohaus Instruments, Shanghai, China), ASP200S Advanced Smart Tissue Processor, HistoCore Arcadia Paraffin Embedding Station, RM2245 Rotary Microtome, ST5020 Fully Automatic Multifunctional Stainer (Leica Microsystems, Germany), MDF-U74V Ultra-Low Temperature Freezer, HYC-260 Medical Refrigerator (Haier), JCA-BM6010C Fully Automatic Biochemical Analyzer (Sysmex), DK-600S Three-Purpose Thermostatic Water Bat (Shanghai Jinghong Experimental Equipment Co., Ltd.), PMC-60 Low-Temperature Centrifuge (Eppendorf), NIKON Ni-U Upright Fluorescence Microscope (Nikon, Japan), PTY-C2200 Animal Weighing Scale (Fuzhou Huazhi Scientific Instruments Co., Ltd.).
Number of offspring and organ coefficient measurement
After ACN administration, male mice (three per group) were housed with untreated female mice in a 1:2 ratio for mating for 3 days. The presence of vaginal plugs was checked twice daily. After cohabitation, female mice were housed individually, and the number of offspring was recorded. At the end of the intervention, mice were weighed, euthanized by cervical dislocation after orbital blood collection, and dissected. Both testes and epididymides were carefully isolated, weighed, and organ coefficients were calculated.
Sperm characteristics analysis
The cauda epididymis was separated and placed into a dish containing 2 mL of physiological saline at 37 °C. The tissue was minced with scissors to fully release sperm, followed by a 20 min water bath. The suspension was filtered through a 400-mesh sieve, and 20 μL was placed on a glass slide and covered with a coverslip22. Sperm counts and viability were evaluated using a SAS-II sperm quality analyzer. A total of 200 sperm were analyzed for viability and other indicators to assess sperm quality.
Sperm Malformation Rate: One drop of semen was smeared, air-dried, fixed with methanol, and stained with 1% eosin for 1 h. After drying, 1000 sperm were examined under a microscope. The calculation of sperm malformation rate followed the reference method18.
Histopathology
The testes were fixed in paraformaldehyde for 24 h, bisected, and placed in embedding cassettes for a 2-h rinse under running water. Tissues were dehydrated using an automatic tissue processor (Leica TP1020, Germany), embedded in paraffin, and sectioned at 5 μm using a rotary microtome (Leica RM2135, Germany). Slides were baked at 60 °C for 2 h, then deparaffinized, rehydrated, and stained with hematoxylin–eosin (HE).
Biochemical analysis
The reproductive hormone levels and oxidative stress markers in mouse serum were measured. The reproductive hormones included testosterone(T), estradiol(E2), follicle-stimulating hormone (FSH), and luteinizing hormone (LH). Oxidative stress indicators included malondialdehyde(MDA), catalase(CAT), superoxide dismutase(SOD), and glutathione peroxidase (GSH-Px). All measurements were performed using enzyme-linked immunosorbent assay (ELISA) kits for mice (Wuhan Saier Biotechnology Co., Ltd., supplied by Youersheng) following the manufacturer’s instructions.
DNA extraction and sequencing of fecal bacterial 16S rRNA genes
On the day of sacrifice, fresh feces of mice were collected and quickly freeze them in liquid nitrogen, and transfer them to -80° C freezer within 3 h after sampling. The total fecal microbial DNA was obtained through the Fecal Genome DNA Extraction Kit(AU46111-96, BioTeke,China) according to the manufacturer’sinstruction manual. The DNA was quantified by Qubit (Invitrogen, USA). Total DNA was amplified by PCR using the universal primer341F/805R(341F: 5′-CCT ACGGGNGGCWGCAG-3′,805R: 5′-GACTACHVGGGTATCT AATCC-3′). The PCR product was purifed using AMPure XP Beads (Beckman Coulte and quantifed using Qubit (Invitrogen USA). Qualifed PCR products were evaluated using an Agilent 2100 Bioanalyzer (Agilent, USA) and Illumina library quantitative kits (Kapa Biosciences, Woburn, MA, USA), which were further pooled together and sequenced on an Illumina NovaSeq 6000 (PE250 ), provided by LC-Bio Technology Co., Ltd., Hangzhou, China.
Sequencing primer were removed from de-multiplexed raw sequences using cutadapt (v1.9). Then, Pairedend reads were merged using FLASH(v1.2.8). The low-quality reads (quality scores<20), short reads (<100 bp), and containing more than 5% “N” records were trimmed by using the sliding-window algorithm method in fqtrim (v 0.94). Quality fltering was performed to obtain high-quality clean tags according to fqtrim. Chimeric sequences were fltered using Vsearch software (v2.3.4). DADA2 was applied for denoising and generating amplicon sequence variants (ASVs). The sequence alignment of species annotation was performed by QIIME2 plugin feature-classifier, and the alignment database was SILVA and NT-16S. Alpha and beta diversities werecalculated using QIIME2, Relative abundance was used in bacteria taxonomy.
Fecal untargeted metabolomics analysis and data preprocessing
LC–MS/MS analyses were performed using an UHPLC system (Vanquish, Thermo Fisher Scientific) with a Waters ACQUITY UPLC BEH Amide (2. 1 mm × 50 mm, 1.7 μm) coupled to Orbitrap Exploris 120 mass spectrometer (Orbitrap MS, Thermo). The mobile phase consisted of 25 mmol/L ammonium acetate and 25 mmol/L ammonia hydroxide in water (pH = 9.75) (A) and acetonitrile (B). The auto-sampler temperature was 4 ℃, and the sample was randomly injected with 2 μL. The quality control (QC) sample was prepared by mixing an equal aliquot of the supernatant of samples.
Instrument calibration is performed Weekly.The accuracy of the instrument is controlled by the external deviation of positive and negative ions 0.16, 0.15 ppm, and the internal deviation of positive and negative ions 0.09 and 0.16 ppm. The relative standard deviation (RSD) of total ionic current (TIC) of positive and negative ions was 1.9% and 11.9%. All parameters meet the predefined acceptance criteria (quality accuracy ≤ 3 ppm, stability RSD ≤ 15%).
Prior to fecal metabolome analysis, raw metabolomic data underwent preprocessing including logarithmic transformation (base 2) and unit variance scaling. Multivariate analysis was initiated by principal component analysis (PCA) to exclude outlier samples falling outside the 95% confidence interval. Subsequently, orthogonal partial least squares discriminant analysis (OPLS-DA) and two-way orthogonal PLS-DA (O2PLS-DA, for tri-group comparisons) were conducted using SIMCA software (version 18.0.1, Umetrics AB, Sweden). Model robustness was assessed through sevenfold cross-validation, with permutation testing (n=200 iterations) confirming overfitting absence (Q2 intercept <0.05). The raw data were converted to mzXML format with Proteo Wizard and then processed with an in-house program (developed using R for automatic data analysis) for peak detection, extraction, alignment, and integration.
Statistics
All experimental data were displayed as m ± sd (mean ± standard deviation). GraphPad Prism 8.4.3 software were used for statistical analysis. Student t-test was used to analyse the two groups of measurement data, while comparisons between three or more groups were made using the Kruskal–Wallis test. If the data does not satisfy the normal distribution or had significant heterogenicity, Mann–Whitney U test was applied for data analysis. P < 0.05 was considered statistically significant.Multiple hypothesis tests were adjusted using Benjamini and Hochberg false discovery rate (FDR), and FDR of < 0.05 was considered significant.
Among sequencing of fecal bacterial 16S rRNA genes, the wilcox test was used to identify the differentially abundant genus, and signifcances were declared at p < 0.05. LDA effect size (LEfSe, LDA ≥ 3.0, p < 0.05) was performed using nsegata-lefse. Other diagrams were implemented using the R package(v3.4.4).
The metabolites with VIP > 1 and p < 0.05 and fold change ≥ 2 or FC ≤ 0.5 were considered to be diferential metabolites. p < 0.05 was considered statistically signifcant and correlation plots were plotted by corrplot package in R language.
Results
POP mitigates ACN-induced reproductive damage in male mice
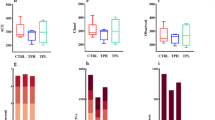
Body weight measurements and weight gain rates were compared among the experimental groups. Significant differences in body weight changes were observed among the groups (p < 0.05) (Fig. 1A). However, analysis of food intake revealed no significant differences among the four groups (Fig. 1B). Notably, female mice did not become pregnant when housed with males from the model and low-dose POP groups, demonstrating a significant difference compared to the control group. In the medium-dose POP group, some females became pregnant, but with fewer offspring, whereas the high-dose POP group showed significantly higher numbers of offspring compared to the model group (Fig. 1C). These results suggest that POP exhibits a dose-dependent effect on ACN-induced reproductive damage, particularly at high doses, albeit with limited impact at lower doses. Consequently, subsequent analyses focused solely on the high-dose POP group, referred to as the POP group.
(A) Comparison of the growth rate of body weight among all groups. (B) Comparison of the amount of food in every week among all groups. (C) Comparison of the number of childbirth among all groups. (D) Comparison of testis index among all groups. (E) Comparison of the number of sperm among all groups. (F) Comparison of rate of sperm abnormality among all groups. The unpaired t Text used to analysis date and Significance is expressed as *p < 0.05,**p < 0.01 vs ACN group; #p < 0.05, ##p < 0.01 vs Control group.
Testis weight as a percentage of body weight varied among the groups, but no statistical significance was detected (Fig. 1D). Sperm count and sperm malformation rate are presented in Fig. 1E and F. Compared to the control group, the ACN group exhibited significantly reduced sperm count and increased sperm malformation (p=0.0076, p<0.0001). However, the POP intervention group demonstrated significant decrease in these parameters relative to the model group (p=0.0417, p=0.0011), indicating a protective effect of POP against ACN-induced reproductive toxicity.
Histopathological analysis revealed that testicular tissue from the control and vitamin E (VE) groups displayed typical morphology, with seminiferous epithelia containing germ cells at various stages of development (Fig. 3A–C). In contrast, the ACN-treated group showed enlarged seminiferous tubule lumens, thinning or incomplete seminiferous epithelia, and reduced numbers of germ cells, with scarce sperm observed in most tubule lumens, indicating impaired spermatogenesis due to ACN exposure (Fig. 3B). The POP group, however, exhibited increased numbers and layers of germ cells in the seminiferous tubules, suggesting that POP mitigated ACN-induced testicular tissue damage, particularly at higher doses (Fig. 3D).
POP improves serum oxidative stress levels
The serum malondialdehyde (MDA) level in the model group was slightly higher than in the control group, but the difference was not statistically significant (p = 0.9854) (Fig. 2B). In contrast, superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) levels were significantly higher in the model group compared to the control group (p = 0.0284, p = 0.0266, p = 0.0410) (Fig. 2A, C and D). The POP intervention group showed reduced MDA levels compared to the model group, although the difference was not statistically significant (p = 0.8664) (Fig. 3B). However, SOD, CAT, and GSH-Px levels were significantly lower in the POP group compared to the model group (p = 0.0005, p = 0.0087, p = 0.0278), indicating improved oxidative stress status. Although the change of MDA level was not statistically significant, the change direction was consistent with the change trend of SOD and GSH-Px, which may be related to oxidative stress or inflammatory response of the body.
(A–D) The expression level of SOD, MDA, CAT and GSH-PX among all groups. (E–H) The expression level of FSH, E2, LH and T of sex hormones in the serum among all groups (n = 6).
(A-D) H&E staining of testiular tissue among all groups. These representative photos were taken under a magnification of × 200. A red asterisk represents abnormal curved seminiferous tubules.
POP increases serum sex hormone levels
The effects of POP on serum sex hormones were also examined. The ACN group exhibited significantly lower levels of follicle-stimulating hormone (FSH), luteinizing hormone (LH), and testosterone (T) compared to the control group (p = 0.0214, p = 0.0016, p = 0.0002)(Fig. 2E, G and H), while estradiol (E2) levels were higher, although not statistically significant (p = 0.7905)(Fig. 2F). The POP and VE groups demonstrated significantly increased levels of FSH, LH, and T compared to the model group (p = 0.0398, p = 0.0277, p = 0.0006), while E2 levels were reduced, though not significantly (p = 0.3432) (Fig. 2E–H).
POP improves gut microbiota composition following ACN exposure
We conducted 16S rRNA gene sequencing on 18 fecal samples from the control, model, and POP groups (n=6). Alpha diversity, which summarizes microbial community richness and evenness, was evaluated using the Chao1 richness index and Shannon and Simpson diversity indices. Results showed changes in richness and diversity for the model and POP groups compared to the control group, however, these changes were not statistically significant (p > 0.05) (Chao1 index p=0.88, Shannon index p = 0.42, Simpson index p=0. 13) (Fig. 4A–C). Despite the Shannon index not reaching statistical significance between groups (P = 0.42), its absolute value increased in the POP intervention group (Con: 6.19 ± 1.37, ACN: 6.58 ± 1.46, POP: 7.11 ± 0.25). This trend is consistent with previous studies23 reporting that prebiotic interventions improve reproductive function, suggesting that the current treatment duration or dose may be close to the threshold of effectiveness. Future research should aim to determine the threshold at which microbial diversity contributes to reproductive protection through dose escalation experiments.
Comparison of (A) Chao index, (B) Shannon index and (C) Simpson index among samples (n=6). (D) Weighted PCoA analysis results from different groups. (E) NMDS analysis results from different groups. (F) The heatmap of phylum level from different groups. (H) The heatmap of genus level from different groups. Composition of gut microbiota at the (G) phylum level and (I) genus level.
In addition, we compared β diversity using linear models (PCoA analysis) and non-linear models (NMDS analysis) (Fig. 4D, E). PCoA analysis based on Jaccard distance showed that the gut microbial community of the model group deviated from the control group, while the POP group had significant changes compared to the model group, indicating significant differences in β diversity among the three groups. To visualize the structural diversity of the gut microbiome in the dicovery group, we used PCoA/NMDS plots based on the unweighted UniFrac distances. The corresponding statistical significance of the beta diversity was measured separately by Adonis and ANOSIM. The results showed a significant difference in the gut microbiome in three groups (Adonis for PCoA, R2= 0.25, p=0.03, ANOSIM FOR NMDS, R=0.33, p < 0.001). To further evaluate the composition of gut microbiota among the three groups, we assessed bacterial taxonomic similarity. Relative abundance analysis at the phylum level showed that the model group had a substantial impact on gut microbiota composition, particularly affecting two dominant phyla. Firmicutes (47-58%) and Bacteroidetes (37-45%) were the main dominant phyla across the three groups (Fig. 4F, G). However, the Firmicutes/Bacteroidetes (F/B) ratio was higher in the model group (1.58) compared to the control (1.07) and POP groups (1. 12). At the genus level, significant differences were observed between the model group and the control and POP groups, with significant changes in the relative abundance of Lactobacillus(Con=8.1, ACN=18.9, POP=2.1) , recombinant Escherichia coli HT002(Con=2.3, ACN=4.7, POP=1.6), Alloprevotella(Con=2.9, ACN=1.2, POP=3.4) , Rikenella (family Rikenellaceae)(Con=2.2, ACN=1.4, POP=2.2), unclassified Clostridia_UCG−014(Con=1.7, ACN=3.0, POP=09), Turicibacter(Con=0.02, ACN=3.0, POP=0.05), and Lactococcus(Con=1.3, ACN=0, POP=0.7). Ding and colleagues found that a significant increase in Bacteroides and *Proteus mirabilis* in the mouse gut led to local inflammation and endotoxemia, and caused disruption of spermatogenesis19. In this study, we found a significant increase in the abundance of Lactobacillus and *S. aureus* in the model group (Fig. 4H, I), while the abundance of *Prevotella* decreased. Based on these key findings, the next step will be to conduct intervention experiments using sterile mice with significantly different bacterial genera to verify their reproductive relevance. Encouragingly, the study also found that POP can effectively reverse the microbial community changes induced by ACN.
Fecal untargeted metabolomics analysis
To elucidate metabolic disturbances linked to ACN exposure, untargeted metabolomics analysis was performed on fecal samples from different groups using ultra-high-performance liquid chromatography (UHPLC) coupled with Orbitrap mass spectrometry. Following data preprocessing, 5,974 metabolite features were extracted from raw data obtained in both positive and negative ionization modes, among which 353 metabolites were identified. Principal component analysis (PCA) was employed to assess data stability and reproducibility Unit variance (UV) scaling and non-logarithmic transformation were conducted prior to multivariate statistical analysis. Quality control (QC) samples, composed of equal amounts from all test samples, were inserted every 10 samples to monitor instrument stability during the experiment. The QC samples exhibited clear clustering, indicating good stability and reproducibility in the sample analysis sequence (Fig. 5A).
(A) PCA score diagram of the used all groups and QC samples. Fecal metabolome profiles among all groups assessed by OPLS-DA (orthogonal partial least squares discriminating analysis). (B) Model group was compared with the control group and (C) POP group was compared with the model group. (D) Volcano plot of faecal metabolome changes in control group versus ACN group. (E) Volcano plot of faecal metabolome changes in ACN group versus POP group. (F) Matchstick plot of faecal metabolome changes in control group versus ACN group. (G) Matchstick plot of faecal metabolome changes in ACN group versus POP group.
Orthogonal partial least squares discriminant analysis (OPLS-DA) was subsequently used to classify fecal metabolites from the control, ACN, and POP groups. As demonstrated in the OPLS-DA score plots (Fig. 5B–C), samples from the control and ACN groups were clearly separated. In the score plot comparing the model and control groups, the cumulative R2Y and Q2 values were 0.905 and 0.619, respectively. Permutation testing confirmed the model’s stability, with an R2Y intercept of 0.82 and a Q2 intercept of − 0.66, indicating that the model was not overfitted. Similarly, when comparing the POP group with the ACN group, the cumulative R2Y and Q2 values were 0.982 and 0.813, respectively. Permutation testing further validated that this model was not overfitted (R2Y intercept of 0.78, Q2 intercept of -0.77). The model was reliable by 200 permutations (p < 0.05).
The volcano map shows the results of the differential analysis of metabolites between groups. The X-axis represents log2-fold changes (log2FC), while the Y-axis indicates the significance level (− log10(p-value)). Red and blue dots represent metabolites that are significantly upregulated or downregulated, respectively (p < 0.05, |log2FC| > 1.25), whereas gray dots indicate metabolites with no significant difference. Volcano plot analysis identified 79 differentially expressed metabolites between the control and ACN groups, of which 19 were upregulated and 60 were downregulated (Fig. 5C, D). Comparing the model and intervention groups, 184 differentially expressed metabolites were found, including 157 upregulated and 27 downregulated. Among these differentially expressed metabolites, the top 5 upregulated metabolites between the control and ACN groups were Desfesoterodine, Daidzein, Deoxycorticosterone, Asparaginyl-Alanine and [10]-Gingerol. The top 5 downregulated metabolites were 2-Aminoacrylic acid, 5-Deoxykievitone hydrate, Daphniphylline, Gingerol and Cinnamyl phenylacetate. In the comparison between the ACN and POP groups, the top 5 upregulated metabolites were Triethanolamine, 3-Oxo-7-hydroxychol-4-enoic acid, (all-E)-1,7,9-Heptadecatriene-11, 13, 15-triyne, 3-Buten-2-one 1-(2,3,6-trimethylphenyl) and 2-Aminoacrylic acid. The top 5 downregulated metabolites were Oleanolic acid, 28-O-Beta-D-glucopyranoside, Glycyrrhetic acid, 3-O-Beta-D-glucuronide and Glomeratose A.
Based on the OPLS-DA model (VIP > 1 and p < 0.05), 2-Aminoacrylic acid was identified as a significant differential metabolite, exhibiting a significant decrease in the model group (p < 0.05), but a significant increase following POP intervention (Fig. 5E, F). Studies identified binary metabolites 2-Aminoacrylic acid involved in important metabolic pathways in the control, model, and POP groups by correlation analysis. Relevant studies have shown that 2-Aminoacrylic acid levels are significantly decreased in diabetic patients, suggesting that 2-Aminoacrylic acid has an effect on the development of diabetes23. As we have observed in the present study, 2-Aminoacrylic acid abnormalities affect glucose and lipid metabolism and insulin sensitivity from KEGG24 Enrichment data Display (Fig. 6A–D), After the ACN intervention, 2-Oxocarboxylic acid metabolism, Biosynthesis of amino acids, Nucleotide metabolism, Biosynthesis of unsaturated fatty acids, Purine metabolism, Glycine, Serine and threonine metabolism Abnormalities affected the steroid metabolism pathway, the metabolic pathways of cysteine and methionine, Thus causing a series of reproductive hormone secretion disorders, oxidative stress, Adherence responses such as chronic inflammation and gut dysbiosis.
(A) KEGG Enrichment for ACN group vs control group and (B) KEGG Enrichment for POP group vs ACN group. (C)Pathway analysis for ACN group vs control group and (D) Pathway analysis for POP group vs ACN group.
Discussion
Chronic occupational exposure to environmental pollutants, particularly acrylonitrile (ACN), has been well documented to disrupt endocrine homeostasis and adversely affect male reproductive health25. As a volatile petrochemical intermediate extensively used in acrylic fiber production, food packaging, and medical materials, ACN poses significant health risks through prolonged exposure. Clinical studies of ACN-exposed male workers have revealed multiple reproductive impairments, including marked decreases in serum testosterone (T) with concurrent elevation of estradiol (E2) levels26,27, reduced sperm density and total count, increased sex chromosome aneuploidy (XX, YY, XY diploids), and significant increases in sperm DNA fragmentation as evidenced by comet assay parameters (increased tail length and percentage of comet sperm)28. Beyond its established immunotoxic28, neurotoxic29, and pulmonary effects30, ACN’s reproductive toxicity has been increasingly recognized31, with emerging research highlighting a novel mechanistic pathway through gut microbiota-testis axis disruption. This suggests that environmental toxins like ACN may compromise male fertility via this important biological crosstalk. This biological crosstalk has been substantiated by several key findings: First, experimental studies have verified that environmental toxin exposure can significantly damage the male reproductive system in offspring while disrupting gut microbiota composition32,33,34. Second, microbiota transplantation studies demonstrate that transferring gut microbiota from adult male mice to immature females elevates testosterone levels and induces metabolic changes35. Third, beyond classical renal excretion pathways, gut microbiota have been recognized as a major site of androgen metabolism36,37.
Our study demonstrated that acrylonitrile (ACN) exposure induced significant reproductive toxicity in male mice, manifesting as disrupted testicular architecture, impaired spermatogenesis, elevated sperm malformation rates, and steroid hormone dysregulation. Comprehensive microbiome analysis revealed three key dimensions of gut microbial alterations induced by ACN exposure. First, at the community level, β-diversity metrics (PCoA/NMDS) showed significant compositional divergence between ACN-exposed and control/POP groups, particularly evidenced by an elevated Firmicutes/Bacteroidetes ratio (1.58 in model vs 1.07 in control and 1.12 in POP groups), indicating substantial microbiota imbalance38. Second, these structural changes translated to functional consequences, where increased Firmicutes abundance potentially enhanced SCFA production (e.g., 2-aminoacrylic acid) that may systemically influence testicular function, while reduced Bacteroidetes levels compromised immune regulation. Notably, Firmicutes overgrowth could induce gut inflammation that may affect testicular immunity via the gut-testis axis38. Third, genus-specific alterations included decreased beneficial Lactobacillus (impairing gut barrier function39), increased pro-inflammatory HT002 strain, and modified abundance of Prevotella and Bacteroides (family Rikenellaceae) known to influence immune-metabolic pathways34,40,41. Collectively, these results establish a robust association between ACN-induced microbial dysbiosis and spermatogenic dysfunction, reinforcing the gut microbiota’s pivotal role in maintaining reproductive health42.
Polysaccharides are a class of high molecular weight compounds formed by the combination of ten or more monosaccharides through glycosidic bonds. Their constituent monosaccharides can be identical or different, resulting in a complex and large molecular structure. Polysaccharides are widely found in nature, with abundant resources, high safety, and minimal side effects, possessing a variety of pharmacological activities such as hypoglycemic, antioxidant, anti-inflammatory, antitumor, and immunomodulatory effects43,44. Plant polysaccharides are considered natural bioactive macromolecules found in medicinal and edible plants, known for their multiple bioactivities and health benefits45.
Recent studies have increasingly demonstrated the protective role of plant-derived polysaccharides in mitigating reproductive toxicity and restoring reproductive function. For instance, Dendrobium polysaccharides significantly ameliorate cyclophosphamide-induced oxidative stress in the male murine reproductive system43, while Ganoderma polysaccharide peptides alleviate cyclophosphamide-induced male reproductive damage by reducing oxidative stress and apoptosis46. Multiple studies have also shown that plant polysaccharides can participate in the development and progression of diseases by regulating gut microbiota. For instance, Yahui Jia et al. studied the effects of Rehmannia glutinosa polysaccharides on the immune system and gut microbiota in an immunosuppressed mouse model, finding that the polysaccharides alleviated cyclophosphamide-induced gut microbiota damage by increasing the populations of Muribaculaceae and Erysipelotrichaceae, while inhibiting the growth of Lactobacillaceae45 Guo et al. investigated the effects of Grifola frondosa polysaccharides on glucose and lipid metabolism as well as gut microbiota in type 2 diabetic mice47. POP, a naturally active polysaccharide, has been widely concerned for its unique advantage of improving the microecology in the gut as probiotics. Investigators have verifed that the interaction mechanism between melatonin and intestinal microbiota constitutes a complex and multifaceted network. POP has been shown to be able to lower intestinal pH and increase SCFA utilization in aged rats, stimulating probiotic growth (e.g., lactic acid bacteria) and inhibiting pathogenic bacteria (e.g., Shigella)48. In addition, POP can reduce the abundance of some obesity-related bacteria in aged rats, such as reducing the ratio of Firmicutes to Bacteroidetes and decreasing the abundance of mucin-degrading bacteria Akkermansia, which is associated with healthy mucosal19. These microbiota-mediated effects likely contribute to POP’s broad pharmacological profile, which encompasses immunomodulatory, metabolic (hypolipidemic/hypoglycemic), antimicrobial, antitumor, and antioxidant/anti-aging activities, with particular efficacy in promoting Bifidobacterium proliferation48,49.
Nonetheless, there is currently no published research on how purslane polysaccharides regulate gut flora to alleviate reproductive impairment. This study utilized the microecological modulation effects of Portulaca oleracea to explore the potential secondary impacts of dysbiosis caused by ACN-induced reproductive damage. The results indicated that POP can promote the growth of normal gut microbiota, regulate dysbiosis, and serve as an effective microecological modulator. POP significantly decreases the F/B ratio and regulates the imbalance of the microbial flora. The POP intervention improved the damage of spermatogenic tubules, increased sperm count, repaired testicular structure, and significantly increased the number of offspring born after mating with female mice, as well as oxidative metabolism indicators and hormone levels. These findings strongly suggest that POP can alleviate inflammatory responses and oxidative stress, leading to the improvement of lipid peroxidation. Based on the 16S rDNA sequencing results, certain microbial communities may be associated with ACN-induced testicular damage. At the phylum level, the relative abundance of Bacteroidetes decreased in the POP group compared to the model group, which has been suggested to be positively correlated with testicular function38. At the genus level, the relative abundance of Prevotella was significantly downregulated in the POP group compared to the ACN group (79.87%), and it has been reported to be associated with testicular function. Ding et al. found that the combined relative abundance of Prevotella and Bacteroides was strongly negatively correlated with sperm motility15. In addition, the combined abundance of Prevotella and Bacteroides was significantly decreased in the POP group compared to the ACN groups, which was closely associated with higher sperm motility. Moreover, we identified several unexplained plant-related genera, such as Turicibacter, that may have potential impacts on the male reproductive system. Future efforts may include exploring the effects of these molecular-level changes on sperm production, motility, and morphology, as well as their potential involvement in inflammatory processes within the male reproductive system.
As we know, this is the first multi-omics analysis that combines metabolome and microbiome data to reveal the intrinsic mechanisms underlying ACN-induced reproductive toxicity and the protective effects of POPs on reproduction. However, several limitations need to be addressed. Firstly, the study is constrained by a small sample size; further research with a larger scale is necessary to confirm these findings. Secondly, deeper investigations are required to further verify the causal relationship between ACN-induced changes and POP intervention on specific microbiota and differential metabolites, such as 2-Aminopropionic acid, in relation to male reproductive decline. Additionally, the mechanism should be confirmed using either fecal microbiota transfer (FMT) operations or in vitro models. Thirdly, this study did not design and establish independent groups for VE and POP, neglecting the contribution of individual components to reproductive protection. This oversight has impacted the persuasiveness of the experimental results. Future steps of the study will enhance the experimental group settings to improve the scientific value of the outcomes. Fourthly, the limitations of animal model studies must be acknowledged, as the results of animal experiments may not be entirely applicable to human reproductive health conclusions. The next phase will involve conducting more population-based, long-term follow-up studies to more comprehensively assess the potential risks of ACN to human reproductive health.
This study confirms that gut microbiota dysbiosis can adversely affect male reproductive function, a finding consistent with previous research. Through multi-omics analysis, we demonstrate that acrylonitrile (ACN) exposure significantly alters both the gut microbial structure and metabolic profiles in male mice. These alterations primarily mediate ACN-induced fertility decline through dysregulation of lipid metabolism pathways. Notably, the natural polysaccharide POP exhibits significant protective effects against ACN-induced reproductive damage, although its precise molecular mechanisms require further elucidation. The animal model established in this study successfully recapitulates key pathological features, providing valuable preclinical data. These findings not only reveal the substantial reproductive risks posed by environmental toxins but also highlight the therapeutic potential of natural product interventions. Our results advance the understanding of gut-testis axis interactions and establish a theoretical foundation for developing POP-based functional foods or pharmaceuticals to prevent and treat environmental toxin-mediated reproductive disorders.
Data availability
The date that supports the findings of this study are available in [Non-target metabolomics of mouse faecal LCMS raw data] at https://ngdc.cncb.ac.cn/omix/select-edit/OMIX008428. The gut microbial data have been uploaded to the NCBI database and reviewer link is https://dataview.ncbi.nlm.nih.gov/object/PRJNA1203095.
References
Wang, Y. et al. Exploring the role of gut microbiome in male reproduction. Andrology 10(3), 441–450 (2022).
Infertility prevalence estimates, 1990–2021. Geneva: World Health Organization.WHO. 2023.
Agarwal, A. et al. Male infertility: Causes, diagnosis, and treatment strategies. Nat. Rev. Urol. 20(3), 123–145 (2023).
Zhang, T. et al. Gut microbiota may contribute to the postnatal male reproductive abnormalities induced by prenatal dibutyl phthalate exposure. Chemosphere 287(Pt 1), 132046 (2022).
Nordkap, L. et al. Regional differences and temporal trends in male reproductive health disorders: Semen quality may be a sensitive marker of environmental exposures. Mol. Cell. Endocrinol. 355(2), 221–230 (2012).
Tian, X. et al. Lactobacillus plantarum TW1-1 alleviates diethylhexylphthalate-induced testicular damage in mice by modulating gut microbiota and decreasing inflammation. Front. Cell Infect. Microbiol. 9, 221 (2019).
Ning, K. et al. Portulaca oleracea L. polysaccharide alleviates dextran sulfate sodium-induced ulcerative colitis by regulating intestinal homeostasis. Int J Biol Macromol 256, 128375 (2024).
Neal, B. H. et al. Weight-of-the-evidence review of acrylonitrile reproductive and developmental toxicity studies. Crit. Rev. Toxicol. 39(7), 589–612 (2009).
Abe, Y. et al. Survey of volatile substances in kitchen utensils made from acrylonitrile–butadiene–styrene and acrylonitrile–styrene resin in Japan. Food Sci. Nutr. 2(3), 236–243 (2014).
Souter, I. et al. Urinary concentrations of phthalate metabolite mixtures in relation to serum biomarkers of thyroid function and autoimmunity among women from a fertility center. Environ. Health Perspect. 128(6), 67007 (2020).
Niu, W. et al. Efects of molecular weight of chitosan on anti-inflammatory activity and modulation of intestinal microflora in an ulcerative colitis model. Int. J. Biol. Macromol. 193, 1927–1936 (2021).
Jin, Y. et al. Oral exposure of mice to carbendazim induces hepatic lipid metabolism disorder and gut microbiota dysbiosis. Toxicol. Sci. 147(1), 116–126 (2015).
Zhang, L. et al. Persistent organic pollutants modify gut microbiota-host metabolic homeostasis in mice through aryl hydrocarbon receptor activation. Environ. Health Perspect. 123(7), 679–688 (2015).
Ding, N. et al. Impairment of spermatogenesis and spern motility by the high-fat diet-induced dysbiosis of gut microbes. Gut Microbiota 69(9), 1608–1619 (2020).
Haro, C. et al. Intestinal Microbiota Is Influenced by Gender and Body Mass Index. PLoS ONE 11(5), e0154090 (2016).
Shen, H. et al. Purification and characterization of an antitumor polysaccharide from Portulaca oleracea L.. Carbohyd. Polym. 93(2), 395–400 (2013).
He, Y. et al. Antibacterial diarrhea effect and action mechanism of Portulaca oleracea L. water extract based on the regulation of gut microbiota and fecal metabolism. J. Sci. Food Agric. 103(14), 7260–7272 (2023).
Paul Claman, M. D. et al. Men at disk: Occupation and male infertility. Fertil. Steril. 81(2), 19–26 (2004).
Zhuang, S. et al. Portulaca oleracea L. polysaccharide ameliorates lipopolysaccharide-induced inflammatory responses and barrier dysfunction in porcine intestinal epithelial monolayers. J. Funct. Foods 91, 104997 (2022).
Wang, Z. et al. Protective effects of polysaccharide extracted from portulacae oleracea L. on colitis induced by dextran sulfate sidium. J. Med. Foods 23(2), 125–131 (2020).
Fu, Q. et al. Portulaca oleracea polysaccharides reduce serum lipid levels in aging rats by modulating intestinal microbiota and metabolites. Front. Nutr. 9, 965693 (2022).
Naghdi, M. et al. Effects of common fig (Ficus carica) leaf extracts on sperm parameters and testis of mice intoxicated with formaldehyde. Evid.-Based Complement. Altern. Med. 2, 1–9 (2016).
Xu, W.-Y. et al. 2-Aminoadipic acid protects against obesity and diabetes. J. Endocrinol. 243(2), 111–123 (2019).
Kanehisa, M. et al. KEGG: Biological systems database as a model of the real world. Nucleic Acids Res 53(D1), D672–D677 (2025).
Giribabu, N., Sainath, S. B. & Sreenivasula Reddy, P. Prenatal di-n-butyl phthalate exposure alters reproductive functions at adulthood in male rats. Environ. Toxicol. 29(5), 534–544 (2014).
Farombi, E. O. et al. Neuroprotective role of 6-Gingerol-rich fraction of Zingiber oficinale (Ginger) against acrylonitrile-induced neurotoxicity in male Wistar rats. J. Basic Clin. Physiol. Pharmacol. https://doi.org/10.1515/jbcpp-2018-0114 (2019).
El-shafey, R. S. et al. The ameliorative impacts of wheat germ oil against ethanol-induced gastric ulcers: involvement of anti-inflammatory, antiapoptotic, and antioxidant activities. Toxicol. Res. 11(2), 325–338 (2022).
Barbara, H. et al. Weight-of-the-evidence review of acrylonitrile reproductive and developmental toxicity studies. Crit. Rev. Toxicol. 39(7), 589–612 (2009).
Jin, T., et al. Effect of acrvlonitrile on male reproductive health among occupationally exposed workers. Environ. Sci. (2004).
Walker, V. E. et al. Analysis of biomarkers of DNA damage and mutagenicity in mice exposed to acrylonitrile. Chem. Res. Toxicol. 33(7), 1623–1632 (2020).
Shi, Y. et al. Protection of apigenin against acrylonitrile-induced sperm and testis injury in rats: involvement of activation ofASK1-JNK/p38 signaling pathway. Toxicol Res (Camb) 10(2), 159–168 (2021).
Al-Asmakh, M. et al. The gut microbiota and developmental programming of the testis in mice. PLoS ONE 9(8), e103809 (2014).
Jiang, D. et al. Gut microbiota combined with metabolome dissects Fluorene-9-bisphenol exposure-induced male reproductive toxicity. Environ. Pollut. 364, 125339 (2025).
Liu, J. et al. Glyphosate-induced gut microbiota dysbiosis facilitates male reproductive toxicity in rats. Sci. Total Environ. 805, 150368 (2022).
Shi, Y. et al. Protection of apigenin against acrylonitrile-induced sperm and testis injury in rats: involvement of activation of ASK1-JNK/p38 signaling pathway. Toxicol. Res. 10, 299–311 (2021).
Schiffer, L., Arlt, W. & Storbeck, K. H. Intracrine androgen biosynthesis, metabolism and action revisited. Mol. Cell Endocrinol. 465, 4–26 (2018).
Colldén, H. et al. The gut microbiota is a major regulator of androgen metabolism in intestinal contents. Am. J. Physiol. Endocrinol. Metabol. 317(6), E1182–E1192 (2019).
Xu, P. et al. Melatonin prevents obesity through modulation of gut microbiota in mice. J. Pineal Res. 62(4), e12399 (2017).
Dickerson, R. et al. Research progress on the potential mechanism and application of lactobacillus in the prevention and treatment of acute myocardial infarction. Tianjin Med. J. 52(1), 107–112 (2024).
Liu, Y. et al. Harnessing human microbiomes for disease prediction. Trends Microbiol. 23, 00339–00346 (2024).
Zhang, T. et al. Disrupted spermatogenesis in a metabolic syndrome model: The role of vitamin A metabolism in the gut–testis axis. Gut 71(1), 78–87 (2022).
Krautkramer, K. A. et al. Gut microbial metabolites as multikingdom intermediates. Nat. Rev. Microbiol. 19(2), 77–94 (2021).
Mu, Y. et al. Dendrobium oficinale polysaccharides improved reproductive oxidative stress injury in male mice treated with cyclophosphamide. Environ. Sci. Pollut. Res. Int. 30(48), 106431–106441 (2023).
Zhang, H. et al. Ganoderma lucidum polysaccharide peptide alleviates cyclophosphamide-induced male reproductive injury by reducing oxidative stress and apoptosis. Biomedicines 12(8), 1632 (2024).
Jia, Y. et al. Effects ofpolysaccharide from hot-compressed steamed Rehmannia glutinosa on the immune system and gut microbiota in an immunosuppressed mice model. Int. Immunopharmacol. 142(Pt B), 113202 (2024).
Borchers, A. & Pieler, T. Programming pluripotent precursor cells derived from Xenopus embryos to generate specific tissues and organs. Genes 1(3), 413–426 (2010).
Guo, W. L. et al. Hypoglycemic and hypolipidemic activities of Grifola frondosa polysaccharides and their relationships with the modulation of intestinal microflora in diabetic mice induced by high-fat diet and streptozotocin. Int. J. Biol. Macromol. 153, 1231–1240 (2020).
Fu, Q. et al. Portulaca oleracea polysaccharides modulate intestinal microflora in aged rats in vitro. Front. Microbiol. 13, 841397 (2022).
Wang, Z. et al. Protective effects of polysaccharide extracted from Portulacae oleracea L. on colitis induced by dextran sulfate sodium. J. Med. Food. 23(2), 125–131 (2020).
Acknowledgements
This work was supported by grants from Natural Science Foundation of Ningxia (2022AAC03202).
Author information
Authors and Affiliations
Contributions
W.L. designed the study. H.L., J.W., Y.L. perfoemed the experiment. W.L., C.J. and Y.L. analyzed the date. W.L. wrote original draft preparation. W.L. and Y.L. wrote—review and editing. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, W., Li, H., Wu, J. et al. Purslane oleracea polysaccharides alleviate male reproductive damage caused by acrylonitrile by improving gut microbiota dysbiosis and metabolic disorder. Sci Rep 15, 27450 (2025). https://doi.org/10.1038/s41598-025-12090-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-12090-8