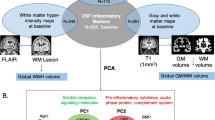
Abstract
Alzheimer’s disease (AD) is characterized by amyloid-beta (Aβ) and tau pathologies that drive neuroinflammation and neurodegeneration. Free water (FW), a diffusion tensor imaging (DTI) metric reflecting extracellular fluid changes, has emerged as a sensitive marker of neuroinflammation. This study examined the role of FW in AD and its associations with plasma biomarkers, brain structural measures, and cognitive decline. We analyzed 968 participants across the AD continuum from Samsung Medical Center. Plasma biomarkers were measured using Single Molecule Array technology. DTI metrics (FW, fractional anisotropy (FA), and mean diffusivity (MD)) and structural MRI-derived hippocampal volume and cortical thickness were assessed. Plasma phosphorylated tau 217 (pTau217) significantly correlated with FW in both white matter (WM) and gray matter (GM), but not with FA or MD. All DTI metrics were associated with reduced hippocampal volume and lower cortical thickness. Mediation analyses revealed that FW in WM and GM had both direct and indirect associations with cognitive decline, while FA and MD were indirectly linked to cognitive outcomes through structural measures. These findings support FW as a sensitive marker of neuroinflammation, linking microstructural changes to macrostructural changes and cognitive outcomes. Integrating plasma biomarkers, DTI, and MRI may improve understanding of AD pathophysiology and support the development of targeted diagnostic and therapeutic approaches.
Similar content being viewed by others
Introduction
Alzheimer’s disease (AD) is characterized by a cascade of pathological events, starting with amyloid-beta (Aβ) and tau protein aggregation, which precede neurodegeneration and ultimately lead to cognitive decline1. In vivo detection of these pathological changes has been revolutionized by positron emission tomography (PET) imaging for Aβ and tau, allowing precise staging of disease progression. More recently, plasma biomarkers have emerged as a promising alternative for detecting AD pathology non-invasively2,3. Plasma phosphorylated tau 217 (pTau217), in particular, is closely associated with core AD pathology, Aβ and tau, providing a direct link to the primary pathological hallmarks of the disease4. In contrast, glial fibrillary acidic protein (GFAP) and neurofilament light chain (NfL) are associated with non-specific processes related to AD, with GFAP reflecting neuroinflammation and NfL indicating neurodegeneration5,6,7,8,9. These plasma biomarkers offer complementary insights into both the core pathologies and the downstream processes of AD, highlighting their potential to bridge gaps in understanding disease progression.
Diffusion tensor imaging (DTI) is a widely used MRI technique that measures the microstructural integrity of brain tissues in both white matter (WM) and gray matter (GM)10,11,12,13,14. Common DTI metrics, such as fractional anisotropy (FA) and mean diffusivity (MD), assess axonal integrity and tissue diffusivity15 while free water (FW) quantifies extracellular fluid and serves as a marker of neuroinflammation in AD15,16,17,18,19. Neuroinflammation plays a critical role in AD pathophysiology20,21,22 contributing to both microstructural disruptions and downstream macrostructural changes. Studies have demonstrated FW’s utility in capturing extracellular changes related to neuroinflammation, but its relationship with other DTI metrics like FA and MD, which may influence FW, has been less explored. This highlights the need for a comprehensive analysis of DTI metrics to better understand their combined role in AD pathology across WM and GM.
DTI metrics, which reflect microstructural changes, and structural MRI measures, such as hippocampal volume and cortical thickness, which capture macrostructural changes, provide complementary insights into AD progression. Microstructural disruptions, such as neuroinflammation and axonal degeneration, contribute to macrostructural alterations like hippocampal atrophy and cortical thinning, which in turn drives cognitive decline. Understanding the interplay between microstructural and macrostructural changes is crucial for elucidating the pathological mechanisms underlying AD and improving the prediction of cognitive outcomes.
In the present study, we aimed to investigate the role of FW on DTI in understanding the pathophysiology of AD, particularly its relationship with core AD pathologies, brain structural measures, and cognitive decline. Specifically, we examined the associations between plasma biomarkers and DTI metrics, the links between DTI metrics and brain structural measures, and how these structural measures mediate the relationship between DTI metrics and cognitive decline. This integrated approach provides a comprehensive understanding of the connections between molecular markers, micro- and macro-structural abnormalities, and clinical outcomes in AD.
Materials and methods
Study population
Participants were recruited from Samsung medical center between 2016 and 2023. We enrolled 968 participants aged ≥ 45 years who underwent Aβ PET, brain MRI including diffusion tensor image (DTI) and plasma AD biomarkers (217 with cognitively unimpaired (CU), 436 with mild cognitive impairment (MCI), and 315 with dementia of the Alzheimer’s type (DAT)23). All participants with CU status satisfied the following criteria: (1) no medical history that was likely to affect cognitive function based on Christensen’s health screening criteria20; (2) no objective cognitive impairment in any cognitive domain on a comprehensive neuropsychological test battery (above at least − 1.0 SD of age-adjusted norms on any cognitive test); and (3) independence in activities of daily living. All participants with amnestic MCI (aMCI) satisfied the criteria for aMCI with the following modifications24,25: (1) subjective cognitive complaints by the participants or caregivers; (2) objective memory impairment below − 1.0 SD on verbal or visual memory tests, (3) no significant impairment in activities of daily living, and (4) non-demented status. All participants with DAT satisfied the diagnostic criteria of the National Institute on Aging and Alzheimer’s Association (NIA-AA).
We excluded participants with structural lesions including territorial infarction, lobar hemorrhage, brain tumors, WMH due to demyelinating disorders, vasculitis, leukodystrophy, radiation injury, and hydrocephalus on MRI. Additionally, participants with abnormal laboratory results for complete blood count, electrolyte, vitamin B12, folate levels, syphilis serology, and liver/kidney/thyroid function tests were excluded.
The Institutional Review Board of Samsung Medical Center approved this study (No. 2021-02-135). All participants provided their informed consent to participate in the study and the data were collected in accordance with to the Declaration of Helsinki.
Aβ PET acquisition and quantification using Centiloid values
All the participants underwent Aβ PET18 F-florbetaben (FBB) PET or18 F-flutemetamol (FMM) PET using a Discovery STe PET/CT scanner (GE Medical Systems, Milwaukee, WI, USA). For FBB PET or FMM PET, a 20 min emission PET scan in dynamic mode (consisting of 4 × 5 min frames) was performed 90 min after injection of a mean dose of 311.5 MBq FBB or 197.7 MBq18F-flutemetamol, respectively. Three-dimensional PET images were reconstructed in a 128 × 128 × 48 matrix with 2 mm × 2 mm × 3·27 mm voxel size using the ordered-subsets expectation maximization algorithm (FBB, iteration = 4 and subset = 2018; F-flutemetamol, iteration = 4 and subset = 20).
We used regional direct comparison centiloid (rdcCL) method, to harmonize the quantitative analysis of Aβ PET across different ligands26,27. Data processing was conducted utilizing Statistical Parametric Mapping version 8 (SPM 8). The acquired PET images were co-registered with the individual MRI scans. Subsequently, The PET and MRI images were spatially normalized using the transformation parameters of the SPM 8 unified segmentation method for T1-weighted MRIs. The whole cerebellum was used as a reference region to calculate the standardized uptake value ratio (SUVR) using regional masks obtained from the GAAIN website (http://www.GAAIN.org). The common cortical target region was obtained by comparing the scans of 25 Aβ PET-positive Alzheimer’s disease dementia patients and 18 Aβ PET-negative controls among patients who underwent both FBB and FMM-PET with ligand-to-head. The MRI-based regional volumes of interest (VOIs) were defined by applying the common cortical target VOI (FMM-FBB global CTX VOI) based on the automated anatomical labeling (AAL) atlas. Finally, individual rdcSUVR values in the FMM-FBB global CTX VOI were calculated for all PET images and rdcSUVR values of the FBB-FMM CTX VOI were converted to rdcCL units using a conversion equation28. A more detailed process for obtaining global rdcCL is discussed elsewhere27. For clarity and consistency, global rdcCL will be referred to as centiloid in the following sections.
MRI acquisition and processing
We acquired three-dimensional T1 Turbo Field Echo MRI scans for all participants using a 3.0 T MRI scanner (Philips 3.0T Achieva) with the following imaging parameters: sagittal slice thickness, 1.0 mm with 50% overlap; no gap; repetition time of 9.9 ms; echo time of 4.6 ms; flip angle of 8°; and matrix size of 240 × 240 pixels reconstructed to 480 × 480 over a field view of 240 mm. All T1 scans were processed by CIVET pipeline (version 2.1.0)29 developed at the Montreal Neurological Institute for fully automated structural image analysis. In brief, T1 scans were transformed to MNI-152 template30,31 and N3 algorithms31 was used for correction of intensity non-uniformity. Next, tissue classification32 was performed, and WM and GM surfaces were constructed using the Constrained Laplacian-based Automated Segmentation with Proximities (CLASP)33 algorithm. The cortical thickness was defined of the Euclidean distance between the linked vertices of the WM and GM surfaces. A more detailed process is described in elsewhere34.
To measure hippocampal volume (HV), we employed an automated hippocampus segmentation method using a graph cut algorithm combined with an atlas-based segmentation and morphological opening as described in an earlier study35.
DTI acquisition
Axial diffusion-weighted images were acquired using a single-shot echo-planar imaging (EPI) sequence with b-values of 0 and 600 s/mm2. We applied 45 directions of motion probing gradients, and other imaging parameters for the diffusion MRI were as follows: repetition time (TR), 9300 ms; echo time (TE), 91 ms; section thickness, 2 mm; flip angle, 90°; field-of-view, 220 × 220 mm2; acquisition matrix, 128 × 128; scan time, 7 min and 26 s. All axial sections were acquired parallel to the anterior commissure-posterior commissure line.
Calculation of DTI metrics
All diffusion MRI data were processed using the functional magnetic resonance imaging of the brain (FMRIB) software library tool (FSL) (www.fmrib.ox.ac.uk/fsl)36. Briefly, correction for head movements and eddy current distortions were performed and brain masks were defined for each subject using the Brain Extraction Toolbox (BET) for the single B0 images. Single-tensor fractional anisotropy (FA) and mean diffusivity (MD) maps were generated using the DTIFIT tool implemented in FMRIB Software. We used FW imaging to estimate fractional volume of freely diffusing extracellular water molecules. The FW and FW-corrected maps (FAt and MDt) were then calculated using the open-source software package Diffusion Imaging in Python (DIPY, https://dipy.org/)37. A more detailed description of the bi-tensor model is extensively discussed elsewhere16.
Plasma collection and processing
We obtained 8 mL of blood from each participant and placed in a 0.5 M EDTA-containing tube and mixed it for 5 min. Plasma was extracted from the blood sample after a 10-min centrifuge (1300g) and dispensed into 5 or 10 vials at a volume of 0.3 mL each. All plasma samples were kept frozen at -75 °C until analysis. This process complied with the manual for human resource collection and registration of the National Biobank of the Republic of Korea. The median interval between plasma collection and Aβ PET scans was 4 days (interquartile range [IQR], 0–69 days).
Plasma samples were shipped in a frozen state at − 70 degrees to the Department of Psychiatry and Neurochemistry, University of Gothenburg for analysis. Plasma samples were thawed on wet ice, centrifuged at 500×g for 5 min at 4 °C. Plasma pTau 217 concentration was meaused using the commercial ALZpath pTau217 immunoassay on a Single molecule array HD-X instrument (Quanterix, Billerica, MA), a paramagnetic microbead-based sandwich enzyme-linked immunosorbent assay system. Plasma GFAP and NfL were quantified using the commercial Neurology 4-Plex E kit on the same instrument (Quanterix).
Clinical and vascular assessments
Of the 968 participants, 592 underwent follow-up Clinical Dementia Rating–Sum of Boxes (CDR-SOB) assessments. The number of follow-up visits varied by individual (mean ± SD, 1.79 ± 0.96 visits), with a mean interval of 3.06 ± 1.59 years between the baseline MRI and the final follow-up. Annual CDR-SOB change rates were then calculated using linear regression. Diabetes mellitus (DM) was defined as self-reported diagnosis by specialist or current use of diabetic medications or insulin, and hypertension (HTN) was defined as self-reported diagnosis or current use of antihypertensive medications.
Voxel-wise whole brain analysis
A tract-based spatial statistics (TBSS)38 and GM-based spatial statistic (GBSS)39 were used to elevate the relationship between plasma and DTI metrics for WM and GM, respectively.
Analysis of WM microstructural changes were performed using the skeleton projection of TBSS with the following procedure. First, FA maps of all subjects were aligned to the Montreal Neurological Institute (MNI 152) standard space using the FMRIB non-linear registration tool36. Next, a mean FA image was generated and thinned to create the mean FA skeleton, which represented the centers of all tracts common to the groups. FA > 0.20 was used as the threshold of the mean FA skeleton to include major WM pathways and exclude peripheral tracts and GM. Finally, the aligned FA map of each subject was projected onto the FA skeleton and other maps (FAt, MDt, and FW) were projected on the mean FA skeleton.
Analysis of GM microstructural changes were performed using GBSS, a GM along of TBSS, using the following steps. First, the Brain Extraction Tool was used to remove non-brain tissues from 3D T1-weighted images. Next, each skull-stripped 3D T1-weighted images was linear and non-linearly aligned to MNI152 1 mm standard space using the FMRI registration tool. Next, filed bias was corrected, and skull-stripped images were segmented into GM, WM, and CSF using the FMRIB automated segmentation tool40. Next, GM images were used to create median GM skeleton with a threshold of 0.2 to minimize the contribution of voxels from WM and CSF. After then, B0 maps of each subject were aligned to 3D T1-weighted images. All DTI maps were linearly and non-linearly aligned into MNI152 1 mm standard space and aligned maps of each subject were projected on the median GM skeleton.
Statistical analyses
Clinical characteristics of the study participants are presented as mean ± standard deviation for continuous variables and as frequency (%) for categorical variables. The level of pTau 217, GFAP, NfL, and centiloid were transformed to log-transformed to satisfy the assumption of linearity.
For TBSS and GBSS analyses, a general linear model framework, with age, sex, BMI status (only for plasma analyses), and APOE4 genotype as covariates, was used. The randomize tool (FSL) with 5000 permutations was applied to test the relationships between DTI metrics and plasma biomarkers. Results were then corrected for multiple comparisons by controlling the family-wise error (FWE) at a 0.01 significance level and applying threshold-free cluster enhancement (TFCE).
To examine the relationship between DTI metrics and brain structural measures, mean cortical thickness and hippocampal volume, linear regression was performed, with age, sex, education, APOE4 genotype, and intracranial volume (ICV) as covariates. The regression analysis was conducted using standardized variables, with DTI metrics (FAt, MDt,, and FW) calculated as the mean within the skeleton mask.
To investigate whether the brain structural measure mediates the effect of DTI metrics on longitudinal cognitive decline, we conducted mediation with DTI metrics and FW as predictor, brain structural measures (mean cortical thickness or hippocampal volume) as a mediator, and CDR-SOB score as an outcome after controlling age, sex, education, APOE4 genotype, and ICV. Bootstrapping was used to verify the significance of the direct and indirect effects.
Results
Characteristics of participants
Table 1 shows the characteristics of the 968 participants. The age (mean ± standard deviation [SD]) was 70.6 ± 8.6 years, and 600 out of 968 (62.0%) participants were females. The frequencies of diagnosis were 217 (22.4%) for CU, 436 (45.0%) for MCI, and 315 (32.5%) for ADD. The frequency of BMI status was 34 (3.5%) for underweight, and 263 (27.2%) for obese. The frequency of the APOE4 genotype was 429 (44.3%) for ε4 carriers.
Relationships between AD biomarkers and DTI metrics
Figure 1 show the results of the TBSS analyses for FAt (Fig. 1A), MDt, (Fig. 1B), and FW (Fig. 1C). The voxel-wise linear regression indicated that lower FAt in widespread brain WM regions were associated with increased levels of NfL and GFAP, while lower MDt were linked to increased NfL, GFAP, and centiloid. The results are TFCE-enhanced and reported at p < 0.01, with FWE correction. There were no regions showing an association between FAt and pTau217 or centiloid, nor between MDt and pTau217. Furthermore, higher FW in widespread WM regions was found to be associated with higher levels of NfL, GFAP, and centiloid, while increased FW in specific brain regions, including the corpus callosum, corona radiata, and posterior thalamic radiation, showed a significant correlation with elevated pTau217 levels. However, several projection fibers, including the posterior limb of internal capsule and the anterior limb of internal capsule, did not show significant associations.
Association between plasma biomarkers, centiloid, and WM integrity. TBSS showing the correlations between FA (A), MD (B), and FW (C) and plasma biomarkers (NLF, GFAP, pTau217, and centiloid). Results are TFCE enhanced, reported at p < 0.01, FWE corrected. The green voxels indicate the white matter skeleton used for statistical testing, and voxels with significant correlations (corrected p < 0.01) are shown in red to yellow. TBSS, tract-based spatial statistics; WM, white matter; FA, fractional anisotropy; MD, mean diffusivity; FW, free water, NfL, neurofilament light chain; GFAP, glial fibrillary acidic protein.
Figure 2 shows the results of the GBSS analyses. The results indicated a widespread association between these DTI metrics and GFAP across various brain areas, with lower FAt (Fig. 2A) and MDt values associated with higher GFAP levels (Fig. 2B). FAt showed a strong association with NfL in certain brain regions, including the cerebellum, the left and right inferior temporal gyri, the left and right fusiform gyri, the left parahippocampal gyrus, the left middle temporal gyrus, and the left lingual gyrus, whereas no significant associations were observed in others. There was no region showing an association between FAt and pTau217 or centiloid, nor between MDt and NfL, pTau217, or centiloid. FW showed significant associations with plasma biomarkers and centiloid in most cortical regions, with higher FW associated with elevated plasma and centiloid levels (Fig. 2C).
Association between plasma biomarkers, centiloid, and GM integrity. GBSS showing the correlations between FA (A), MD (B), and FW (C) and plasma biomarkers (NLF, GFAP, pTau217, and centiloid). Results are TFCE enhanced, reported at p < 0.01, FWE corrected. The green voxels indicate the gray matter skeleton used for statistical testing, and voxels with significant correlations (corrected p < 0.01) are shown in red to yellow. GBSS, gray matter based spatial statistics; GM, gray matter; FA, fractional anisotropy; MD, mean diffusivity; FW, free water, NfL, neurofilament light chain; GFAP, glial fibrillary acidic protein.
The results of the TBSS analyses with additional adjustment for HTN and DM are presented in Supplementary Fig S1, and those of the GBSS analyses with the same covariates are shown in Supplementary Fig. S2. These sensitivity analyses revealed comparable spatial patterns and statistical significance, supporting the robustness of our findings regardless of vascular risk factor adjustment.
Relationships between DTI metrics and brain structural measures on MRI
The FAt was positively associated with cortical thickness (β = 0.046, p < 0.0001) (Fig. 3A) and hippocampal volume (β = 159.6, p < 0.0001) in WM (Fig. 3B), and similarly, in GM, it showed significant associations with cortical thickness (β = 0.08, p < 0.0001) (Fig. 3A) and hippocampal volume (β = 251.5, p < 0.0001) (Fig. 3B) after controlling for age, sex, APOE4 genotype, education, and ICV. The MDt showed a positive association with cortical thickness (β = 0.032, p < 0.0001) and hippocampal volume (β = 109.2, p < 0. 0001) in WM, while in GM, it was positively correlated with cortical thickness (β = 0.099, p < 0.0001) and hippocampal volume (β = 300.8, p < 0.0001). Contrary to the findings for FA and MD, the FW showed a negative association with cortical thickness (β = − 0.043, p < 0.0001) and hippocampal volume (β = − 103.7, p < 0.0001) in WM, and similarly, in GM, it was positively correlated with cortical thickness (β = − 0.097, p < 0.0001) and hippocampal volume (β = − 292.1, p < 0.0001), after controlling for the same covariates. Forest plots of the beta coefficients from the full models with all covariates are shown in Supplementary Figs. S3 and S4, and those additionally adjusted for HTN and DM are presented in Supplementary Figs. S5 and S6.
Forest plot of beta coefficients from linear regression of brain atrophy and DTI metrics. The forest plots display the standardized beta coefficients from linear regression analyses with 95% confidence intervals, adjusted for age, sex, BMI, APOE4 genotype, and ICV. The forest plots show each regressor’s standardized beta linear regression coefficients with 95% confidence interval. Plots displaying the beta coefficients from linear regression analyses of cortical thickness with FA, MD, and FW (A), as well as hippocampal volume with FA, MD, and FW (B). FA, fractional anisotropy; MD, mean diffusivity; FW, free water.
Relationships between DTI metrics brain structural measures, and clinical progression
Figures 4 and 5 illustrate the relationships between DTI metrics in WM and GM, brain structural measures, and cognitive decline.
Diagram of mediation analyses among DTI metrics in WM, brain atrophy measures, and CDR-SOB. Mediation analyses were performed using DTI metrics as a predictor, brain atrophy measures as a mediator, and CDR-SOB as an outcome. Significance levels are follows: *p < 0.05, **p < 0.01, ***p < 0.001. FA, fractional anisotropy; MD, mean diffusivity; CDR-SOB, clinical dementia rating sum of boxes; WM, white matter.
Diagram of mediation analyses among DTI metrics in GM, brain atrophy measures, and CDR-SOB. Mediation analyses were performed using DTI metrics as a predictor, brain atrophy measures as a mediator, and CDR-SOB as an outcome. Significance levels are follows: *p < 0.05, **p < 0.01, ***p < 0.001. FA, fractional anisotropy; MD, mean diffusivity; CDR-SOB, clinical dementia rating sum of boxes; GM, gray matter.
For the DTI metrics in WM (Fig. 4), the relationship between the FAt and MDt metrics and annual CDR-SOB change was fully mediated by cortical thickness (FAt: p for indirect effect < 0.001, p for direct effect = 0.38; MDt: p for indirect effect < 0.001, p for direct effect = 0.43) and hippocampal volume (FAt: p for indirect effect < 0.001, p for direct effect = 0.39; MDt: p for indirect effect < 0.001, p for direct effect = 0.93). In contrast, the relationship between the FW and annual CDR-SOB change was partially mediated by brain structural measures, including cortical thickness (p for indirect effect < 0.001, p for direct effect < 0.01) and hippocampal volume (p for indirect effect < 0.001, p for direct effect < 0.01).
For the DTI metrics in GM (Fig. 5), the relationship between the FAt and annual CDR-SOB change was fully mediated by cortical thickness (p for indirect effect < 0.001, p for direct effect = 0.15) and partially mediated by hippocampal volume (p for indirect effect < 0.001, p for direct effect = 0.02). The relationships between the MDt and FW metrics and annual CDR-SOB change were partially mediated by cortical thickness (MDt: p for indirect effect = 0.01, p for direct effect < 0.001; FW: p for indirect effect = 0.01, p for direct effect < 0.001) and hippocampal volume (MDt: p for indirect effect < 0.001, p for direct effect < 0.001; FW: p for indirect effect < 0.001, p for direct effect < 0.001). Supplementary Figs. S7 and S8 present the results of additional mediation analyses that included HTN and DM as covariates. The findings were largely consistent with those from the original models, indicating that the inclusion of HTN and DM did not substantially alter the observed mediation effects.
Discussion
In the present study, we investigated the role of FW on DTI in understanding the pathophysiology of AD, with a particular focus on its relationships with plasma biomarkers, structural changes on MRI, and cognitive decline. Our major findings were as follows. First, core biomarkers, including Aβ and pTau217, showed significant associations with FW, but not with FA or MD, in both WM and GM. In contrast, plasma biomarkers of non-specific AD-related processes, such as GFAP and NfL, were associated with all DTI metrics, including FW, FA, and MD, in both WM and GM. Second, in both WM and GM, all DTI metrics’ abnormalities, representing unspecific microstructural changes, were associated with macrostructural changes such as reduced hippocampal volume or cortical thinning. Finally, these macrostructural changes, in turn, predict cognitive decline, highlighting their critical role in linking microstructural integrity with clinical outcomes. Taken together, our findings suggest that FW on DTI reflects a continuum of pathological processes in AD, linking plasma biomarkers and microstructural changes to macrostructural alterations and subsequent cognitive decline. These findings underscore the utility of FW as a sensitive biomarker for detecting early pathological changes, predicting cognitive decline, and guiding targeted interventions through integration with structural MRI and plasma biomarkers.
Our first major finding was that core biomarkers such as Aβ and pTau217 were significantly associated with FW in both WM and GM, but not with FA or MD. This suggests that these biomarkers primarily reflect extracellular changes41,42,43,44 including neuroinflammation and tissue atrophy, rather than axonal or microstructural integrity, indicating their close link to extracellular fluid dynamics and neuroinflammatory processes. These findings align with previous studies, such as Ji et al.45 and Nakaya et al.46 which reported significant associations between FW, neuroinflammation, and tau pathology in AD45,46,47. Our study extends this understanding by demonstrating the specificity of these associations to FW, highlighting its sensitivity to extracellular alterations like neuroinflammation and synaptic dysfunction. While FW is sensitive to extracellular fluid changes associated with neuroinflammation, it may also capture neurodegenerative processes such as Wallerian degeneration and myelin breakdown16,48. Future work using multimodal imaging and region-specific analyses is needed to disentangle the contributions of inflammation versus degeneration to FW alterations. In contrast, FA and MD, which reflect axonal density and fiber organization, may remain relatively unaffected during early or extracellular-dominant stages of pathology. These results emphasize FW’s utility as a biomarker for detecting extracellular changes and clarifying the role of core biomarkers in AD.
In contrast, plasma biomarkers of non-specific processes involved in AD pathophysiology, such as GFAP and NfL, were associated with all DTI metrics, reflecting their relationship with microstructural changes, in both WM and GM. Their associations with all DTI metrics suggest that these biomarkers capture a broader spectrum of microstructural changes, encompassing both extracellular alterations measured by FW and intracellular disruptions reflected by FA and MD. Our findings align with previous studies, such as Ji et al.45 which demonstrated the sensitivity of FW to neuroinflammation mediated by GFAP, and Nakaya et al.46 which reported the association of NfL with axonal damage. Together, these results could be explained by the concept that GFAP and NfL represent secondary and unspecific processes following core biomarker-mediated pathology, with GFAP reflecting neuroinflammation and NfL indicating neurodegeneration and axonal damage. While consistent with existing findings, our study extends the current understanding by demonstrating that GFAP and NfL are associated with both extracellular changes, captured by FW, and intracellular disruptions, reflected by FA and MD, across WM and GM. This broader role of non-specific biomarkers highlights their ability to capture the full spectrum of microstructural changes, offering a more comprehensive view of AD pathophysiology beyond core biomarker-driven processes.
Our second major finding was that in both WM and GM, all DTI metrics’ abnormalities, representing microstructural changes49 are associated with macrostructural changes such as hippocampal volume loss or cortical thinning50,51 bridging the gap between cellular-level changes and large-scale structural damage. These findings suggest that microstructural disruptions, such as neuroinflammation and axonal degeneration, precede and potentially drive macrostructural changes in AD52. In WM, processes such as Wallerian degeneration (anterograde axonal degeneration following upstream injury)10 or the dying-back phenomenon (distal axonal loss preceding cell body loss)53 may propagate damage to connected GM regions. FW, reflecting neuroinflammation, indicates extracellular matrix disruptions, while FA and MD abnormalities suggest axonal degeneration and demyelination, which impair structural connectivity and contribute to secondary GM atrophy. In GM, FW highlights extracellular changes linked to neuroinflammation54 while FA and MD reflect intracellular disruptions49 such as neuronal shrinkage or synaptic loss, directly driving cortical thinning and hippocampal volume loss. Together, these findings underscore distinct but interconnected pathways: in WM, axonal degeneration impacts connected GM regions, while in GM, localized microstructural changes directly lead to volume loss. This highlights the importance of assessing both WM and GM to understand the cascading effects of AD pathology.
Our final major finding was that the relationship between DTI metrics and cognitive decline was mediated by brain structural measures, with distinct patterns in WM and GM. Especially, FW in both WM and GM exhibited both indirect and direct effects on cognitive decline, suggesting that its influence extends beyond its impact on structural changes. This direct effect of FW highlights the potential role of neuroinflammation in driving cognitive impairment, independent of macrostructural degeneration. This finding aligns with evidence that neuroinflammation disrupts neural function independently of structural degeneration by impairing synaptic integrity, altering extracellular fluid homeostasis, and inducing chronic gliosis55,56,57,58,59. In contrast, FA and MD primarily contributed to cognitive decline through their effects on brain structural measures. In WM, FA and MD were fully mediated by cortical thickness and hippocampal volume, indicating that axonal integrity and diffusivity influence cognition indirectly via macrostructural changes. In GM, FA was fully mediated by cortical thickness and partially by hippocampal volume, while MD was partially mediated by both cortical thickness and hippocampal volume. These findings suggest that while axonal and cellular integrity are key correlates of structural brain changes, FW uniquely captures neuroinflammatory processes that may be more proximally associated with cognitive decline. Microstructural alterations—captured by changes in FW, FA, and MD—reflect underlying processes such as neuroinflammation, axonal degeneration, and demyelination. These processes compromise extracellular matrix integrity and axonal transport, leading to neuronal loss and synaptic pruning. Over time, such cumulative microstructural damage manifests as measurable reductions in cortical thickness and hippocampal volume. Our mediation analyses thus link these cellular- and tissue-level pathologies to large-scale structural degeneration in AD.
A major strength of our study is the comprehensive integration of plasma biomarkers, DTI metrics, and structural MRI measures to elucidate the differential contributions of microstructural and macrostructural abnormalities to cognitive decline in AD. However, several limitations should be acknowledged. First, one limitation of this study is the use of a single non-zero b-value (600 s/mm2), which may reduce sensitivity to free-water components. Although multi-shell or higher b-value protocols are optimal16,60 our single-shell approach with spatial regularization produced plausible FW maps and significant group effects. Future studies should employ multi-shell acquisitions for improved free-water modeling. Second, we relied on global DTI metrics for association and mediation analyses, which, while robust, may obscure regional microstructural variations. Moreover, FW is a non-specific indicator of extracellular alterations, and future studies should apply voxel-wise or ROI-based approaches to capture anatomical specificity and disentangle distinct pathological processes. Third, although cognitive trajectories were modeled longitudinally, all plasma biomarkers and imaging measures—including DTI metrics—were obtained cross-sectionally. As a result, causal inferences linking neuroinflammation, structural brain changes, and cognitive decline remain limited. Fourth, we included participants aged 45 years and older to capture early midlife microstructural and inflammatory changes that may precede clinical onset of AD. While this broader age range increases the study’s generalizability to preclinical stages, it may also introduce heterogeneity compared to cohorts restricted to typical late-onset AD. Future studies could stratify analyses by age groups to confirm the reproducibility of our findings in older samples. Fifth, while FW is a widely used marker of neuroinflammation15,16,17,18,19 it is not specific to neuroinflammation alone and may also reflect other extracellular alterations such as edema or vascular dysfunction. In our sensitivity analyses additionally adjusting for vascular risk factors including hypertension and diabetes mellitus, the main results remained unchanged, suggesting that FW alterations are not solely attributable to vascular dysfunction. Nevertheless, the potential for residual confounding by unmeasured vascular or other systemic factors cannot be entirely excluded, and the non-specificity of FW should be considered when interpreting these findings. Finally, despite our efforts to control for confounders, unmeasured variables such as comorbid cerebrovascular disease may have influenced the observed associations. Nevertheless, our findings provide novel insights into the role of neuroinflammation in AD progression, highlighting FW as a potential biomarker that bridges microstructural disruption, structural measures, and cognitive decline, and underscoring its relevance for both disease monitoring and therapeutic targeting.
Conclusions
Our integrated analysis combining plasma biomarkers, microstructural DTI metrics, and macrostructural MRI measures provides a comprehensive framework for understanding the continuum of pathological changes in AD. Notably, our findings reveal that extracellular alterations—reflected by increased FW—are closely linked to core AD pathologies and cognitive decline, with brain structural measures mediating these relationships. These insights underscore the promise of multi-modal biomarker integration for advancing early diagnosis and targeted therapeutic strategies in AD.
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Abbreviations
- AD:
-
Alzheimer’s disease
- Aβ:
-
Amyloid-beta
- BMI:
-
Body mass index
- CDR-SOB:
-
Clinical dementia rating scale sum of box
- CU:
-
Cognitively unimpaired
- DAT:
-
Dementia of the Alzheimer’s type
- DM:
-
Diabetes mellitus
- DTI:
-
Diffusion tensor image
- FA:
-
Fractional anisotropy
- FBB:
-
18F-florbetaben
- FMM:
-
18F-flutemetamol
- FW:
-
Free water
- FWE:
-
Family-wise error
- GBSS:
-
Gray matter-based spatial statistics
- GFAP:
-
Glial fibrillary acidic protein
- HTN:
-
Hypertension
- ICV:
-
Intracranial volume
- MCI:
-
Mild cognitive impairment
- MD:
-
Mean diffusivity
- NfL:
-
Neurofilament light
- pTau217:
-
Phosphorylated tau 217
- rdcCL:
-
Regional direct comparison centiloid
- SUVR:
-
Standardized uptake value ratio
- TBSS:
-
Tract-based spatial statistics
- TFCE:
-
Threshold-free cluster enhancement
References
Scheltens, P. et al. Alzheimer’s disease. The Lancet vol. 397 Preprint at (2021). https://doi.org/10.1016/S0140-6736(20)32205-4
Verberk, I. M. W. et al. Combination of plasma amyloid beta(1–42/1–40)and glial fibrillary acidic protein strongly associates with cerebral amyloid pathology. Alzheimers Res. Ther 12, (2020).
Palmqvist, S. et al. Discriminative accuracy of plasma Phospho-tau217 for Alzheimer disease vs other neurodegenerative disorders. JAMA J. Am. Med. Assoc. 324, (2020).
Mattsson-Carlgren, N. et al. Soluble P‐tau217 reflects amyloid and Tau pathology and mediates the association of amyloid with Tau. EMBO Mol. Med 13, (2021).
Mielke, M. M. et al. Plasma and CSF neurofilament light: relation to longitudinal neuroimaging and cognitive measures. Neurology 93, (2019).
Mattsson, N. et al. Association of plasma neurofilament light with neurodegeneration in patients with Alzheimer disease. JAMA Neurol 74, (2017).
Cicognola, C. et al. Plasma glial fibrillary acidic protein detects Alzheimer pathology and predicts future conversion to Alzheimer dementia in patients with mild cognitive impairment. Alzheimers Res. Ther 13, (2021).
Chatterjee, P. et al. Plasma glial fibrillary acidic protein is elevated in cognitively normal older adults at risk of Alzheimer’s disease. Transl Psychiatry 11, (2021).
Benedet, A. L. et al. Differences between plasma and cerebrospinal fluid glial fibrillary acidic protein levels across the Alzheimer disease continuum. JAMA Neurol 78, (2021).
Coleman, M. P. & Freeman, M. R. Wallerian degeneration, WldS, and Nmnat. Annual Review of Neuroscience vol. 33 Preprint at (2010). https://doi.org/10.1146/annurev-neuro-060909-153248
Bozzali, M., Cercignani, M., Sormani, M. P., Comi, G. & Filippi, M. Quantification of brain Gray matter damage in different MS phenotypes by use of diffusion tensor MR imaging. Am. J. Neuroradiol. 23, (2002).
Pierpaoli, C. & Basser, P. J. Toward a quantitative assessment of diffusion anisotropy. Magn Reson. Med 36, (1996).
Basser, P. J., Mattiello, J. & LeBihan, D. MR diffusion tensor spectroscopy and imaging. Biophys J 66, (1994).
Raghavan, S. et al. Diffusion models reveal white matter microstructural changes with ageing, pathology and cognition. Brain Commun 3, (2021).
Assaf, Y. & Pasternak, O. Diffusion tensor imaging (DTI)-based white matter mapping in brain research: A review. J. Mole. Neurosci. vol. 34 Preprint at (2008). https://doi.org/10.1007/s12031-007-0029-0
Pasternak, O., Sochen, N., Gur, Y., Intrator, N. & Assaf, Y. Free water elimination and mapping from diffusion MRI. Magn Reson. Med 62, (2009).
Ofori, E. et al. Free-water imaging of the hippocampus is a sensitive marker of Alzheimer’s disease. Neuroimage Clin 24, (2019).
Pasternak, O., Shenton, M. E. & Westin, C. F. Estimation of extracellular volume from regularized multi-shell diffusion MRI. in Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics) vol. 7511 LNCS (2012).
Kamagata, K. et al. Advanced diffusion magnetic resonance imaging in patients with Alzheimer’s and Parkinson’s diseases. Neural Regener. Res. 15 Preprint at (2020). https://doi.org/10.4103/1673-5374.276326
Calsolaro, V. & Edison, P. Neuroinflammation in Alzheimer’s disease: Current evidence and future directions. Alzheimer’s and Dementia vol. 12 Preprint at (2016). https://doi.org/10.1016/j.jalz.2016.02.010
Leng, F. & Edison, P. Neuroinflammation and microglial activation in Alzheimer disease: where do we go from here? Nat. Rev. Neurol. vol. 17 Preprint at (2021). https://doi.org/10.1038/s41582-020-00435-y
Onyango, I. G., Jauregui, G. V., Čarná, M., Bennett, J. P. & Stokin, G. B. Neuroinflammation in Alzheimer’s disease. Biomedicines vol. 9 Preprint at (2021). https://doi.org/10.3390/biomedicines9050524
Jang, H. et al. Korea-Registries to Overcome Dementia and Accelerate Dementia Research (K-ROAD): A Cohort for Dementia Research and Ethnic-Specific Insights. Dement Neurocogn Disord. 23(4), 212–223 https://doi.org/10.12779/dnd.2024.23.4.212
Petersen, R. C. Clinical practice. Mild cognitive impairment. - supplementary data. N Engl. J. Med 364, (2011).
Jeong, H. J. et al. [18F]THK5351 PET imaging in patients with mild cognitive impairment. Journal Clin. Neurol. (Korea) 16, (2020).
Klunk, W. E. et al. The centiloid project: standardizing quantitative amyloid plaque Estimation by PET. Alzheimer’s Dement. 11, 1–15e4 (2015).
Kim, S. J. et al. Development and clinical validation of CT-based regional modified centiloid method for amyloid PET. Alzheimers Res. Ther 14, (2022).
Cho, S. H. et al. A new centiloid method for 18F-florbetaben and 18F-flutemetamol PET without conversion to PiB. Eur J. Nucl. Med. Mol. Imaging 47, (2020).
Ivanov, O. et al. The CIVET image-processing environment: A fully automated comprehensive pipeline for anatomical neuroimaging research yasser references & credits. Proceedings of the 12th annual meeting of the organization for human brain mappin 17, (2005).
Collins, D. L., Neelin, P., Peters, T. M. & Evans, A. C. Automatic 3d intersubject registration of Mr volumetric data in standardized Talairach space. J. Comput. Assist. Tomogr. 18, 192–205 (1994).
Sled, J. G. & Zijdenbos, P. Evans, a C. A nonparametric method for automatic correction of intensity nonuniformity in MRI data. IEEE Trans. Med. Imaging. 17, 87–97 (1998).
Zijdenbos, A., Forghani, R. & Evans, A. Automatic quantification of MS lesions in 3D mri brain data sets: Validation of INSECT. in Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics) vol. 1496 (1998).
June, S. K. et al. Automated 3-D extraction and evaluation of the inner and outer cortical surfaces using a laplacian map and partial volume effect classification. Neuroimage 27, 210–221 (2005).
Kim, J. et al. Prediction of Tau accumulation in prodromal Alzheimer’s disease using an ensemble machine learning approach. Sci Rep 11, (2021).
Kwak, K. et al. Fully-automated approach to hippocampus segmentation using a graph-cuts algorithm combined with atlas-based segmentation and morphological opening. Magn. Reson. Imaging. https://doi.org/10.1016/j.mri.2013.04.008 (2013).
Jenkinson, M., Beckmann, C. F., Behrens, T. E. J., Woolrich, M. W. & Smith, S. M. Fsl Neuroimage 62, 782–790 (2012).
Garyfallidis, E. et al. Dipy, a library for the analysis of diffusion MRI data. Front Neuroinform 8, (2014).
Smith, S. M. et al. Tract-based Spatial statistics: Voxelwise analysis of multi-subject diffusion data. Neuroimage 31, (2006).
Ball, G. et al. Development of cortical microstructure in the preterm human brain. Proc Natl. Acad. Sci. U S A 110, (2013).
Zhang, Y., Brady, M. & Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imaging 20, (2001).
Ashton, N. J. et al. Differential roles of Aβ42/40, p-tau231 and p-tau217 for Alzheimer’s trial selection and disease monitoring. Nat Med 28, (2022).
Wang, S. et al. Biofluid biomarkers for Alzheimer’s disease. Front. Aging Neurosci. 16, 1380237 (2024).
Sattarov, R. et al. Phosphorylated Tau in cerebrospinal fluid-derived extracellular vesicles in Alzheimer’s disease: A pilot study. Sci. Rep. 14, 25419 (2024).
Heneka, M. T. et al. Neuroinflammatory fluid biomarkers in patients with Alzheimer’s disease: A systematic literature review. Mol Psychiatry 1–16 (2025).
Ji, F. et al. Associations of blood cardiovascular biomarkers with brain free water and its relationship to cognitive decline: A Diffusion-MRI study. Neurology 101, (2023).
Nakaya, M. et al. Free water derived by multi-shell diffusion MRI reflects tau/neuroinflammatory pathology in Alzheimer’s disease. Alzheimer’s Dementia: Translational Res. Clin. Interventions 8, (2022).
Dumont, M. et al. Free water in white matter differentiates MCI and AD from control subjects. Front Aging Neurosci 11, (2019).
Hoy, A. R. et al. Microstructural white matter alterations in preclinical Alzheimer’s disease detected using free water elimination diffusion tensor imaging. PLoS One 12, (2017).
Alexander, A. L., Lee, J. E., Lazar, M. & Field, A. S. Diffus. Tensor Imaging Brain Neurotherapeutics 4, (2007).
Spotorno, N. et al. Diffusion MRI tracks cortical microstructural changes during the early stages of Alzheimer’s disease. Brain 147, (2024).
Zhang, B., Xu, Y., Zhu, B. & Kantarci, K. The role of diffusion tensor imaging in detecting microstructural changes in prodromal Alzheimer’s disease. CNS Neuroscience and Therapeutics vol. 20 Preprint at (2014). https://doi.org/10.1111/cns.12166
van Uden, I. W. M. et al. Diffusion tensor imaging of the hippocampus predicts the risk of dementia, the RUN DMC study. Hum Brain Mapp 37, (2016).
Raff, M. C., Whitmore, A. V., Finn, J. T. & Neuroscience Axonal self-destruction and neurodegeneration. Science vol. 296 Preprint at (2002). https://doi.org/10.1126/science.1068613
Pasternak, O., Westin, C. F., Dahlben, B., Bouix, S. & Kubicki, M. The extent of diffusion MRI markers of neuroinflammation and white matter deterioration in chronic schizophrenia. Schizophr Res 161, (2015).
Bronzuoli, M. R., Iacomino, A., Steardo, L. & Scuderi, C. Targeting neuroinflammation in Alzheimer’s disease. J. Inflamm. Res. vol. 9 Preprint at (2016). https://doi.org/10.2147/JIR.S86958
Adamu, A., Li, S., Gao, F. & Xue, G. The role of neuroinflammation in neurodegenerative diseases: current understanding and future therapeutic targets. Front. Aging Neurosci. vol. 16 Preprint at (2024). https://doi.org/10.3389/fnagi.2024.1347987
Jung, Y. J., Tweedie, D., Scerba, M. T. & Greig, N. H. Neuroinflammation as a Factor of Neurodegenerative Disease: Thalidomide Analogs as Treatments. Frontiers in Cell and Developmental Biology vol. 7 Preprint at (2019). https://doi.org/10.3389/fcell.2019.00313
Kurki, S. N. et al. Acute neuroinflammation leads to disruption of neuronal chloride regulation and consequent hyperexcitability in the dentate gyrus. Cell Rep 42, (2023).
Zhang, W., Xiao, D., Mao, Q. & Xia, H. Role of neuroinflammation in neurodegeneration development. Signal Transduct.Targeted Therapy vol. 8 Preprint at (2023). https://doi.org/10.1038/s41392-023-01486-5
Hoy, A. R., Koay, C. G., Kecskemeti, S. R. & Alexander, A. L. Optimization of a free water elimination two-compartment model for diffusion tensor imaging. Neuroimage 103, (2014).
Acknowledgements
This study utilized BeauBrain Amylo’s image processing technology to quantify amyloid uptakes using PET-CT.
Funding
This work was partly supported by Institute of Information & communications Technology Planning & Evaluation (IITP) grant funded by the Korea government(MSIT) (No.RS-2021-II212068, Artificial Intelligence Innovation Hub), a grant of the Korea Dementia Research Project through the Korea Dementia Research Center(KDRC), funded by the Ministry of Health & Welfare and Ministry of Science and ICT, Republic of Korea (grant number: RS-2020-KH106434), the National Research Foundation of Korea(NRF) grant funded by the Korea government(MSIT) (RS-2019-NR040057), Future Medicine 20*30 Project of the Samsung Medical Center [#SMX1250081], and the “Korea National Institute of Health” research project(2024-ER1003-01), the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number : RS-2025-02223212), and is also supported by Henrik Zetterberg, who is a Wallenberg Scholar and a Distinguished Professor at the Swedish Research Council, supported by grants from the Swedish Research Council (#2023-00356, #2022-01018 and #2019-02397), the European Union’s Horizon Europe research and innovation programme under grant agreement No 101053962, Swedish State Support for Clinical Research (#ALFGBG-71320), the Alzheimer Drug Discovery Foundation (ADDF), USA (#201809–2016862), the AD Strategic Fund and the AlzheimeAlzheimer’s Association (#ADSF-21-831376-C, #ADSF-21-831381-C, #ADSF-21-831377-C, and #ADSF-24-1284328-C), the European Partnership on Metrology, co-financed from the European Union’s Horizon Europe Research and Innovation Programme and by the Participating States (NEuroBioStand, #22HLT07), the Bluefield Project, Cure Alzheimer’s Fund, the Olav Thon Foundation, the Erling-Persson Family Foundation, Familjen Rönströms Stiftelse, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden (#FO2022-0270), the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 860197 (MIRIADE), the European Union Joint Programme—Neurodegenerative Disease Research (JPND2021-00694), the National Institute for Health and Care Research University College London Hospitals Biomedical Research Centre, the UK Dementia Research Institute at UCL (UKDRI-1003), and an anonymous donor.
Author information
Authors and Affiliations
Contributions
Bo-Hyun Kim: conceptualization, software, formal analysis, visualization, writing—original draft, writing—review and editing. Daeun Shin: data curation. Sung Hoon Kang: data curation. Hyemin Jang: data curation. Jihwan Yun: data curation. YuHyun Park: formal analysis. Henrik Zetterberg: formal analysis, writing—review and editing. Kaj Blennow: formal analysis, writing—review and editing. Fernando Gonzalez-Ortiz: formal analysis, writing—review and editing. Nicholas J. Ashton: formal analysis, writing—review and editing. Sung Tae Kim: data curation. Hee Jin Kim: data curation. Duk L. Na: data curation. Jun Pyo Kim: conceptualization, supervision, data curation, writing—review and editing. Sang Won Seo: conceptualization, supervision, data curation, writing—review and editing. The authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The Institutional Review Board of Samsung Medical Center approved this study (No. 2021-02-135). All participants provided their informed consent to participate in the study, and all methods were in accordance with the Declaration of Helsinki.
Consent for publication
Not applicable.
Competing interests
Henrik Zetterberg has served at scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZPath, Amylyx, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Merry Life, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave, has delivered lectures in symposia sponsored by Alzecure, Biogen, Cellectricon, Fujirebio, Lilly, Novo Nordisk, and Roche, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work).Kaj Blennow has served as a consultant and on advisory boards for Abbvie, AC Immune, ALZPath, AriBio, BioArctic, Biogen, Eisai, Lilly, Moleac Pte. Ltd, Novartis, Ono Pharma, Prothena, Roche Diagnostics, and Siemens Healthineers; has served on data monitoring committees for Julius Clinical and Novartis; has delivered lectures, produced educational materials, and participated in educational programs for AC Immune, Biogen, Celdara Medical, Eisai and Roche Diagnostics; and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program, outside the work presented in this paper. All other authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, BH., Shin, D., Kang, S.H. et al. Mapping Alzheimer’s disease pathology using free water through integrated analysis of plasma biomarkers, microstructural DTI metrics, and macrostructural MRI measures. Sci Rep 15, 30702 (2025). https://doi.org/10.1038/s41598-025-14200-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14200-y