Abstract
Psoriasis is a common chronic inflammatory skin disease that significantly affects patients’quality of life. There is no cure for psoriasis, and available treatments are not completely effective. We have previously found that hydroxytyrosol (HT) has anti-psoriatic effects in vitro. In the present study, we aimed to investigate the therapeutic effects of HT on psoriasis in vivo and to explore the underlying mechanisms. We explored the effects and molecular mechanisms of HT on imiquimod (IMQ)-induced psoriasis-like dermatitis in mice and an M5-induced in vitro cell model using real-time PCR, western blotting, hematoxylin-eosin staining, immunohistochemistry, and enzyme-linked immunosorbent assay. HT (10 mg/kg/d or 50 mg/kg/d, by gavage) ameliorated IMQ-induced clinical manifestations in mice. Moreover, HT ameliorated the histopathological changes and decreased the spleen index and levels of pro-inflammatory cytokines, such as interleukin (IL)-17 A, IL-23, and IL-22, in mouse serum or skin. Mechanistically, HT application inhibited the activation of ERK and NF-кB signaling in the skin samples. Consistently, in vitro analysis showed that HT significantly inhibited inflammation via ERK and NF-κB signaling in a cellular model of psoriasis. Our results indicate that HT alleviates IMQ-induced psoriasis-like dermatitis by inhibiting the ERK and NF-κB signaling pathways, suggesting that HT has a promising therapeutic application in psoriasis treatment.
Similar content being viewed by others
Introduction
Psoriasis, a prevalent chronic immune-mediated dermatosis, manifests as scaly red plaques1. It is characterized by epidermal hyperplasia and intense inflammation2. Patients with psoriasis may suffer from several comorbidities, such as psoriatic arthritis, cardiovascular disorders, and metabolic syndrome, and experience a reduction in their quality of life with substantial economic and psychological burden3,4,5,6. However, there is no cure for psoriasis, and current treatments are not effective for all patients2,7.
Hydroxytyrosol (HT) is a natural phenolic compound with several remarkable biological properties, including anti-proliferation and anti-inflammation8,9. It is extremely safe even at high doses10,11,12. Owing to these activities, HT has been suggested to treat neurodegenerative diseases, metabolic syndrome, cardiovascular diseases, cancer and so on12,13,14,15,16. Studies have hypothesized that HT could be a potential drug for psoriasis17,18. Unfortunately, studies that provide direct evidence of the anti-psoriasis effects of HT are rare.
We previously elucidated that HT exerts anti-psoriasis effects in vitro by suppressing aberrant keratinocyte proliferation and inflammation17. This study further investigated the effects of HT on the imiquimod (IMQ)-induced psoriasis-like dermatitis and explored the underlying mechanisms.
Materials and methods
Cell culture and treatment
HaCaT cells (an immortalized human keratinocyte cell line; ATCC®) were cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin at 37 °C. HaCaT cells were treated with TNF-α, IL-1α, oncostatin-M, IL-17 A, and IL-22 (Peprotech) (M5 cytokine cocktail, each cytokine at 10 ng/ml) to establish an in vitro model of psoriasis. For HT treatment (C8H10O3, APP-Chem Bio), HaCaT cells were pre-incubated with the indicated HT for 2 h, followed by exposure to the M5 cytokine cocktail.
Animal experiments
Eight-week-old female BALB/c mice were purchased from the Animal Experiment Center of Xi’an Jiaotong University and they were kept under a 12 h light/dark cycle with specific pathogen-free conditions. After one week of acclimatization, 24 mice were randomly divided into four groups (n = 6): control, model (IMQ), low-dose HT (IMQ + 10 mg/kg/d HT), and high-dose HT (IMQ + 50 mg/kg/d HT). Mice in the low-dose HT and high-dose HT groups were administered HT solution (10 mg/kg/d or 50 mg/kg/d) by gavage for nine consecutive days, and other mice were administered normal saline. From the 4th day of the experiment, mice except the control group received daily 62.5 mg IMQ cream (Mingxin, Sichuan, China) treatment on the shaved back for 6 days. Mice in the control group received the same dose of the vehicle cream. On the 10th day, mice were euthanized by CO₂ asphyxiation at 35% chamber replacement rate for 5 min. Blood was collected via retro-orbital bleeding, and dorsal skin biopsies and spleens were harvested for further analysis. This study was approved by the Ethics Committee of Fujian Provincial Hospital and all experimental procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Evaluation severity of lesions
The severity of the mouse skin lesions was evaluated on days 1, 3, 5, and 7 using the psoriasis area severity index (PASI) after treatment with IMQ. Erythema, scaling, and skin thickness were scored independently on a scale of 0–4, with 0 indicating no severity and 4 indicating the highest severity. The cumulative PASI score was calculated as the sum of the above three scores.
Measurement of spleen weight index
On day 10th, we took the photos of the spleen and body and spleen weights were taken. Spleen weight index was calculated by normalizing it to body weight.
Histology and immunohistochemistry
The skin tissues were fixed in 10% formalin and embedded in paraffin. Section (5 μm thick) were stained with hematoxylin and eosin. For immunohistochemical staining, sections were incubated with specific primary antibodies against PCNA (YM3031), CD3 (YT0761), Ly6G (Ab238132), and integrin αx (YT5924).
Real-time PCR
Total RNA was extracted from mouse skin tissues or cells using the TRIzol reagent (Invitrogen). The mRNA expression levels of TNF-α, IL-1β, IL-6, IL-17, IL-22, involucrin (IVL) or filaggrin (FLG) were determined, and the primer sequences are shown in Table 1.
Western blot
Proteins were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and electrotransferred onto polyvinylidene fluoride (PVDF) membranes. The blots were incubated with primary antibodies anti-ERK (YT1625), anti-p-ERK (YP0101), anti-p-p65 (YP0191), and anti-p65 (YM3111). The bands were detected using the ECL method.
Measurement of cytokine levels with ELISA
Levels of IL-23, IL-17 A, and IL-22 in mouse serum and IL-1β, IL-6 and IL-23 in cell culture supernatants were measured using ELISA kits from Elabscience according to the manufacturer’s protocols.
Statistical analysis
Experimental data are presented as the mean ± SD. Statistical analyses were performed using Graphpad Prism 5.0. Dunnett’s test was used to compare the differences among groups. p < 0.05 was considered statistically significant.
Results
HT ameliorated psoriasis-like dermatitis induced by IMQ in mice
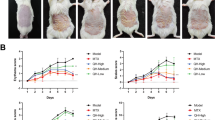
As showed in Figure. 1, mice were given HT by gavage for 9 days to investigate its effect on IMQ-induced psoriasis-like dermatitis. As expected, IMQ treatment induced psoriasis-like lesions and increased PASI scores compared to the control. HT at 10 mg/kg and 50 mg/kg efficiently alleviated the skin manifestations with reduced PASI scores in a dose-dependent manner (Fig. 2a and e).
A schematic diagram of the animal experimental schedule. HT: hydroxytyrosol; IMQ: imiquimod.
HT improved IMQ-induced psoriasis-like dermatitis in mice. (a) Representative clinical photos of mice back skins on days 1, 3, 5 and 7 after treatment with IMQ. (b) Erythema, (c) Scaling, (d) Skin thickness and (e) Cumulative score was scored on days 1, 3, 5 and 7 after treatment with IMQ based on the PASI. Data were presented as mean ± SD. n = 6 per experiment. * p < 0.05, ** p < 0.01, *** p < 0.001. Ctr: control; HT: hydroxytyrosol.
HT alleviated the histopathological morphologies of IMQ-induced psoriasis
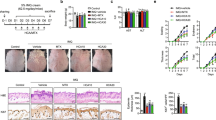
Histopathological analysis revealed that IMQ caused hyperkeratosis, epidermal hyperplasia, acanthosis, and inflammatory cell infiltration (Fig. 3a). HT treatment partially alleviated these histopathological changes (Fig. 3a and b). IHC staining confirmed that IMQ induced epidermal hyperproliferation, as evidenced by the high expression of the proliferative marker PCNA, and HT decreased PCNA expression in the lesion (Fig. 3c and d).
HT alleviated the histopathological morphologies of IMQ-induced psoriasis. (a) H&E staning of mice dorsal skin tissues in each group. Bar = 100 μm. (b) The epidermal thickness was counted (magnification, ×400). (c) Immunohistochemical staining for PCNA (Bar = 100 μm) and (d) quantification of PCNA positive cells in mice dorsal skins. Data were presented as mean ± SD. n = 3 per experiment. * p < 0.05, ** p < 0.01, *** p < 0.001. Ctr: control; HT: hydroxytyrosol; IMQ: imiquimod.
HT improved the immune micro-environment in IMQ-treated mice
qRT-PCR was used to detect the mRNA levels of TNF-α, IL-1β, IL-6, IL-17 A, and IL-22 in the dorsal skin of mice. The data showed that IMQ significantly increased the mRNA levels of TNF-α, IL-1β, IL-6, IL-17 A, and IL-22, and HT treatment at 10 mg/kg and 50 mg/kg significantly decreased the levels of these cytokines in IMQ-treated mice (Fig. 4a). IHC staining showed that infiltration of T cells (CD3+, Fig. 4b), neutrophils (Ly6G, Fig. 4b), and dendritic cells (integrin αx, Fig. 4b) was abundant in the IMQ group. HT treatment effectively reduced inflammatory cell infiltration.
HT improved the immune micro-environment in IMQ-treated mice. (a) The mRNA levels of TNF-α, IL-1β, IL-6, IL-17 A and IL-22 in mice skins were detected by RT-qPCR. (b) Immunohistochemical photographs of CD3, Integrin αx and Ly6G in mice skins. Bar = 100 μm. Data were presented as mean ± SD. n = 3 per experiment. ** p < 0.01, *** p < 0.001. Ctr: control; HT: hydroxytyrosol; IMQ: imiquimod; TNF-α: tumor necrosis factor-α; IL: interleukin; CD3: cluster of differentiation 3; Ly6G: Lymphocyte antigen 6G.
HT inhibited systemic inflammation in IMQ-treated mice
To further explore whether HT can affect IMQ-induced systemic inflammation, we measured spleen weight and detected the expression of IL-17 A, IL-22 and IL23 in mice serum. IMQ significantly induced enlarged spleens and increased the spleen index compared with the control group. However, HT apparently decreased these indices (Fig. 5a and c). ELISA results revealed that in the eyeball blood, the expression of IL-17 A, IL-22 and IL23 in the IMQ group was elevated compared to that in the control group, whereas HT treatment decreased the expression of IL-17 A, IL-22 and IL23, at high-dose HT (Fig. 5d).
HT inhibited systemic inflammation in IMQ-treated mice. (a) Representative images of spleen tissues of each group. (b) Spleen weight. (c) Spleen index. (d) The levels of IL-17 A, IL-22 and IL-23 in the mice serum were measured by elisa. Data were presented as mean ± SD. n = 3 per experiment. * p < 0.05, ** p < 0.01, *** p < 0.001. Ctr: control; HT: hydroxytyrosol; IMQ: imiquimod; IL: interleukin.
HT suppressed the activation of ERK and NF-κB pathways in psoriasis-like dermatitis
Western blot analysis showed that IMQ application significantly increased the p-p65 and p-ERK protein levels (Fig. 6a and c). HT treatment decreased the phosphorylation of p65 and ERK (Fig. 6a and c).
Effect of HT on the activation of ERK and NF-κB pathways in mice. (a) Western blot analysis was performed to measure the protein levels of p65, p-p65, ERK and p-ERK. Images were cropped, and full-length blots were presented in Supplementary Fig. 6a. (b) Semi-quantification analysis of p-p65 normalized to p65 and (c) p-ERK normalized to ERK. Data were presented as mean ± SD. n = 3 per experiment. * p < 0.05, ** p < 0.01. Ctr: control; HT: hydroxytyrosol; IMQ: imiquimod.
HT inhibited inflammatory cytokines expression and activation of ERK and NF-κB pathways in vitro
To further investigate the underlying mechanism of HT in psoriasis-like inflammation, M5-stimulated HaCaT cells were used as an in vitro model in present study. The ELISA data showed that HT pretreatment significantly suppressed the upregulation of IL-1β, IL-6 and IL-23 levels in M5-treated HaCaT cells (Fig. 7a). While HT could not reverse changes in mRNA levels of the differentiation markers IVL and FLG that were induced by M5 treatment in HaCaT, indicating that HT maybe not involved in the regulation of psoriatic keratinocyte differentiation. The western blot assay results confirmed that HT pretreatment markedly decreased the levels of p-p65 and p-ERK induced by M5 (Fig. 7b and d). Overall, these results strongly suggest that HT effectively alleviates the M5-induced inflammatory response via the ERK and NF-κB signaling pathways in HaCaT cells.
Effect of HT on inflammatory cytokines expression and the activation of ERK and NF-κB signaling pathways in M5-stimulated HaCaT cells. (a) Elisa assay was performed to measure the amount of TNF-α, IL-1β, IL-6, IL-17 A, IL-22 and IL-23 release and PCR was performed to measure the mRNA levels of IVL and FLG. (b) Western blot analysis was used to determine the protein levels of p65, p-p65, ERK and p-ERK. Images were cropped, and full-length blots were presented in Supplementary Fig. 7b. (c) Semi-quantification analysis of p-p65 normalized to p65 and (d) p-ERK normalized to ERK. Data were presented as mean ± SD. n = 3 per experiment. * p < 0.05, ** p < 0.01, *** p < 0.001. Ctr: control; HT: hydroxytyrosol; TNF-α: tumor necrosis factor-α; IL: interleukin.
Discussion
We previously found that HT possesses anti-psoriasis potential in vitro17. In the study, the protective effect of HT on psoriasis-like dermatitis was evaluated using IMQ-induced mouse model and M5-induced in vitro cell model. HT alleviated psoriasis-like dermatitis and systemic inflammation induced by IMQ through inactivation of the NF-κB and ERK pathways. These results suggest that HT is a potential candidate for psoriasis treatment.
Several lines of evidence have shown that IMQ treatment of the mouse skin can induce psoriasis-like lesions, and this model has been extensively used to investigate drug candidates19,20. In this study, we used this model to detect the effects of HT on psoriasis-like dermatitis in vivo. These data show that HT application significantly ameliorated the severity of psoriasis-like dermatitis. We first investigated the antihyperproliferative activity of HT in mice, and histopathological analysis showed that HT alleviated IMQ-induced epidermal hyperplasia, as evidenced by a noteworthy reduction in skin thickness and PCNA levels.
Another hallmark of psoriasis is intense inflammation, and it is well known that the IL-17/IL-23 axis is critically involved in the pathogenesis of psoriasis21. IL-17 A, IL23, IL-22, IL-1, IL-6, and TNF-α play key roles in mediating the immune response and skin recruitment of T cells, neutrophils and dendritic cells21,22,23,24. Many in vitro and in vivo studies have observed that HT can modulate several cytokines25. An HT-based formulation could lower IL-1α and IL-8 secretion in reconstructed human epidermis26. Furthermore, HT suppressed the expression of IL-1β, TNF-α, COX-2, and iNOS in macrophages treated with LPS27. We previously found that HT decreased the mRNA levels of IL-6, IL-8, TNF-α, h-BD2, S100A7, S100A8, and S100A9 in psoriatic keratinocytes17. In the current study, HT treatment lowered the expression of TNF-α, IL-1β, IL-6, IL-17 A, and IL-22 induced by IMQ in the dorsal skin of mice. In vitro studies have shown that HT significantly suppresses upregulation of IL-1β, IL-6 and IL-23 protein levels. Additionally, IHC staining showed that HT effectively reduced the infiltration of T cells, neutrophils, and dendritic cells into the psoriatic skin lesions. Taken together, these results demonstrate that HT ameliorates IMQ-induced psoriasis-like dermatitis partly by improving the localized inflammatory microenvironment.
Several studies have demonstrated that NF-κB and ERK signaling pathway might trigger inflammation, promote epidermal hyper-proliferation and exacerbate psoriatic pathogenesis28,29,30. NF-κB is a central orchestrator of inflammation, controlling expression of a myriad of pro-inflammatory genes28. ERK phosphorylation could activate Th17 cells in psoriasis and inhibition of ERK could be a potential drug target29. It was demonstrated that HT could exhibit anti-inflammatory activity via the ERK and NF-κB pathway31,32,33. In current study, we found that HT reversed the activation of ERK and NF-κB signaling pathways, that were induced by IMQ. Consistently, HT reduced p65 and ERK phosphorylation in the M5-stimulated HaCaT cells. Thus, our data showed HT could alleviate psoriatic inflammation response and abnormal proliferation of keratinocyte via ERK and NF-κB pathways.
As we all know, psoriasis is a chronic, systemic inflammatory disease. Thus, the amelioration of psoriasis and its comorbidities may be an ideal treatment strategy. We found that HT decreased the IMQ-induced spleen weight and spleen index in mice. Furthermore, HT decreased IMQ-induced IL-17 A, IL-22, and IL-23 expression in mouse eyeball blood, indicating that HT might improve systemic inflammation status in psoriasis. Considering hydroxytyrosol’s antioxidant properties and the role of oxidative stress in psoriasis, future studies should assess oxidative stress markers to clarify its therapeutic mechanism in psoriasis.
Conclusions
To the best of our knowledge, this is the first study to explore the in vivo effects of HT on psoriasis. Our findings demonstrate that HT relieves psoriasis-like dermatitis and systemic inflammation induced by IMQ via the ERK and NF-κB signaling pathways, making it a potential therapeutic agent for psoriasis and psoriasis-associated comorbidities. However, there are some limitations in our study and the main limitations were as follows: (1) we just focused on the anti-inflammation and anti-proliferation effects of HT on psoriasis; (2) we do not investigate the antioxidant effect of HT on psoriasis; (3) Further basic and clinical research are warranted to fully explore the anti-psoriasis effects of HT and the molecular pathological mechanisms.
Data availability
All data presented in this study are included in the article, and further inquiries can be directed to the corresponding author.
References
Griffiths, C. E. M., Armstrong, A. W., Gudjonsson, J. E. & Barker, J. N. W. N. Psoriasis. Lancet 397, 1301–1315. https://doi.org/10.1016/S0140-6736(20)32549-6 (2021).
Armstrong, A. W., Read, C. & Pathophysiology Clinical presentation, and treatment of psoriasis: A review. JAMA 323, 1945–1960. https://doi.org/10.1001/jama.2020.4006 (2020).
Yamazaki, F. & Psoriasis Comorbidities. J. Dermatol. 48, 732–740. https://doi.org/10.1111/1346-8138.15840 (2021).
Korman, N. J. Management of psoriasis as a systemic disease: what is the evidence? Br. J. Dermatol. 182, 840–848. https://doi.org/10.1111/bjd.18245 (2020).
Armstrong, A. W. et al. Patient perspectives on psoriatic disease burden: results from the global psoriasis and beyond survey. Dermatology 239, 621–634. https://doi.org/10.1159/000528945 (2023).
Min, S., Wang, D., Xia, J., Lin, X. & Jiang, G. The economic burden and quality of life of patients with psoriasis treated with biologics in China. J. Dermatolog Treat. 34, 2247106. https://doi.org/10.1080/09546634.2023.2247106 (2023).
Bakshi, H., Nagpal, M., Singh, M., Dhingra, G. A. & Aggarwal, G. Treatment of psoriasis: A comprehensive review of entire therapies. Curr. Drug Saf. 15 https://doi.org/10.2174/1574886315666200128095958 (2020).
Bertelli, M. et al. A natural compound with promising Pharmacological activities. J. Biotechnol. 309, 29–33. https://doi.org/10.1016/j.jbiotec.2019.12.016 (2020). Hydroxytyrosol.
Velotti, F. & Bernini, R. Hydroxytyrosol interference with inflammaging via modulation of inflammation and autophagy. Nutrients 15 https://doi.org/10.3390/nu15071774 (2023).
Auñon-Calles, D., Canut, L. & Visioli, F. Toxicological evaluation of pure Hydroxytyrosol. Food Chem. Toxicol. 55, 498–504. https://doi.org/10.1016/j.fct.2013.01.030 (2013).
Auñon-Calles, D., Giordano, E., Bohnenberger, S. & Visioli, F. Hydroxytyrosol is not genotoxic in vitro. Pharmacol. Res. 74, 87–93. https://doi.org/10.1016/j.phrs.2013.06.002 (2013).
Robles-Almazan, M. et al. Hydroxytyrosol: bioavailability, toxicity, and clinical applications. Food Res. Int. 105, 654–667. https://doi.org/10.1016/j.foodres.2017.11.053 (2018).
D’Angelo, C., Franceschelli, S., Quiles, J. L. & Speranza, L. Wide biological role of hydroxytyrosol: possible therapeutic and preventive properties in cardiovascular diseases. Cells 9 https://doi.org/10.3390/cells9091932 (2020).
de Las Hazas, M. C. L., Rubio, L., Macia, A. & Motilva, M. J. Hydroxytyrosol: emerging trends in potential therapeutic applications. Curr. Pharm. Des. 24, 2157–2179. https://doi.org/10.2174/1381612824666180522110314 (2018).
Micheli, L. et al. Role of Hydroxytyrosol and Oleuropein in the prevention of aging and related disorders: focus on neurodegeneration, skeletal muscle dysfunction and gut microbiota. Nutrients 15 https://doi.org/10.3390/nu15071767 (2023).
Peyrol, J., Riva, C. & Amiot, M. J. Hydroxytyrosol in the prevention of the metabolic syndrome and related disorders. Nutrients 9 https://doi.org/10.3390/nu9030306 (2017).
Chen, C. et al. Anti-psoriasis activities of Hydroxytyrosol on HaCaT cells under psoriatic inflammation in vitro. Immunopharmacol. Immunotoxicol. 45, 328–333. https://doi.org/10.1080/08923973.2022.2143373 (2023).
Santangelo, C. et al. Anti-inflammatory activity of extra Virgin Olive oil polyphenols: which role in the prevention and treatment of Immune-Mediated inflammatory diseases?? Endocr. Metab. Immune Disord Drug Targets. 18, 36–50. https://doi.org/10.2174/1871530317666171114114321 (2018).
Singh, T. P., Zhang, H. H., Hwang, S. T. & Farber, J. M. IL-23- and Imiquimod-Induced models of experimental psoriasis in mice. Curr. Protoc. Immunol. 125, e71. https://doi.org/10.1002/cpim.71 (2019).
Gangwar, R. S., Gudjonsson, J. E. & Ward, N. L. Mouse models of psoriasis: A comprehensive review. J. Invest. Dermatol. 142, 884–897. https://doi.org/10.1016/j.jid.2021.06.019 (2022).
Grän, F., Kerstan, A., Serfling, E., Goebeler, M. & Muhammad, K. Current developments in the immunology of psoriasis. Yale J. Biol. Med. 93, 97–110(2020).
Moos, S., Mohebiany, A. N., Waisman, A. & Kurschus, F. C. Imiquimod-Induced psoriasis in mice depends on the IL-17 signaling of keratinocytes. J. Invest. Dermatol. 139, 1110–1117. https://doi.org/10.1016/j.jid.2019.01.006 (2019).
Tokuyama, M. & Mabuchi, T. New treatment addressing the pathogenesis of psoriasis. Int. J. Mol. Sci. 21 https://doi.org/10.3390/ijms21207488 (2020).
Zhou, X., Chen, Y., Cui, L., Shi, Y. & Guo, C. Advances in the pathogenesis of psoriasis: from keratinocyte perspective. Cell. Death Dis. 13, 81. https://doi.org/10.1038/s41419-022-04523-3 (2022).
Yu, H. et al. Hydroxytyrosol ameliorates intervertebral disc degeneration and neuropathic pain by reducing oxidative stress and inflammation. Oxid. Med. Cell. Longev. 2022 (2240894). https://doi.org/10.1155/2022/2240894 (2022).
Smeriglio, A. et al. Safety and efficacy of hydroxytyrosol-based formulation on skin inflammation: in vitro evaluation on reconstructed human epidermis model. Daru 27, 283–293. https://doi.org/10.1007/s40199-019-00274-3 (2019).
Yonezawa, Y. et al. Anti-inflammatory effects of olive-derived Hydroxytyrosol on lipopolysaccharide-induced inflammation in RAW264.7 cells. J. Vet. Med. Sci. 80, 1801–1807. https://doi.org/10.1292/jvms.18-0250 (2018).
Goldminz, A. M., Au, S. C., Kim, N., Gottlieb, A. B. & Lizzul, P. F. NF-κB: an essential transcription factor in psoriasis. J. Dermatol. Sci. 69, 89–94. https://doi.org/10.1016/j.jdermsci.2012.11.002 (2013).
Huang, X. et al. ERK inhibitor JSI287 alleviates imiquimod-induced mice skin lesions by ERK/IL-17 signaling pathway. Int. Immunopharmacol. 66, 236–241. https://doi.org/10.1016/j.intimp.2018.11.031 (2019).
Guo, J. et al. Signaling pathways and targeted therapies for psoriasis. Signal. Transduct. Target. Ther. 8, 437. https://doi.org/10.1038/s41392-023-01655-6 (2023).
Yu, Y. B., Zhuang, H. Z., Ji, X. J., Dong, L. & Duan, M. L. Hydroxytyrosol suppresses LPS-induced intrahepatic inflammatory responses via Inhibition of ERK signaling pathway activation in acute liver injury. Eur. Rev. Med. Pharmacol. Sci. 24, 6455–6462. https://doi.org/10.26355/eurrev_202006_21544 (2020).
Zhang, L. et al. Hydroxytyrosol inhibits LPS-Induced neuroinflammatory responses via suppression of TLR-4-Mediated NF-κB P65 activation and ERK signaling pathway. Neuroscience 426, 189–200. https://doi.org/10.1016/j.neuroscience.2019.12.005 (2020).
Aparicio-Soto, M. et al. Olive-Oil-Derived polyphenols effectively attenuate inflammatory responses of human keratinocytes by interfering with the NF-κB pathway. Mol. Nutr. Food Res. 63, e1900019. https://doi.org/10.1002/mnfr.201900019 (2019).
Acknowledgements
This work was supported by joint funds for the innovation of science and technology of Fujian Province (2020Y9022, 2024Y9008), and the National Natural Science Foundation of China (81903220,82404140).
Funding
This work was supported by joint funds for the innovation of science and technology of Fujian Province under Grant [2020Y9022, 2024Y9008], and the National Natural Science Foundation of China under Grant [81903220, 82404140].
Author information
Authors and Affiliations
Contributions
Conceptualization, Meng Liu and Caifeng Chen; methodology and formal analysis, Meng Liu and Caifeng Chen and Li Chen; writing-review and editing, Meng Liu, Ruihan Fan, Caifeng Chen and Li Chen; project administration, Meng Liu; supervision, Caifeng Chen and Danqun Zhang. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Approval for animal experiments
This study was approved by the Ethics Committee of Fujian Provincial Hospital and all experimental procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. Specifically, animal experiments were performed in compliance with the ARRIVE guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, M., Fan, R., Chen, L. et al. Hydroxytyrosol ameliorates imiquimod-induced psoriasis-like dermatitis by modulating ERK and NF-κB signaling pathways in mice. Sci Rep 15, 30125 (2025). https://doi.org/10.1038/s41598-025-16198-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-16198-9