Abstract
To compare the safety of intracameral levofloxacin and intracameral cefazolin in patients undergoing cataract surgery. In this prospective, double-blind, randomized study, patients with senile cataracts who were undergoing phacoemulsification were assigned to receive intracameral levofloxacin (0.1 ml/0.5 mg; Cravit® 0.5%; Santen Pharmaceutical Ltd.) or intracameral cefazolin (0.1 ml/1 mg; generic). The endpoints were the occurrence of endophthalmitis during the 3-month follow-up period, best-corrected visual acuity (BCVA), the presence of anterior chamber (AC) inflammation with grading of AC cells and presence of flare, along with intraocular pressure (IOP), central corneal thickness (CCT), central foveal thickness (CFT), and cell density. A total of 50 patients (50 eyes) were enrolled, randomized, and completed the study. No cases of endophthalmitis were reported in either treatment group. There were no significant differences in BCVA, AC inflammation, IOP, CCT, CFT, or cell density between the groups at any timepoint. Importantly, no dose errors or serious adverse events were reported in either group. This study provides proof of concept that prophylactic intracameral levofloxacin (0.1 ml/0.5 mg) may have a comparable safety profile to intracameral cefazolin (0.1 ml/1 mg) in patients undergoing cataract surgery. Intracameral levofloxacin (0.1 ml/0.5 mg) may offer a viable alternative to cefazolin (0.1 ml/1 mg), particularly for patients with penicillin allergies, while also reducing the risk of dilution errors by eliminating the need for reconstitution. Larger, multicenter studies are warranted to confirm the efficacy of levofloxacin (0.1 ml/0.5 mg) in preventing endophthalmitis.
NCT06710977, 02/12/2024, retrospectively registered.
Similar content being viewed by others
Introduction
With the global prevalence of cataract surgery increasing due to an aging population and greater accessibility in developing regions, addressing postoperative infections remains crucial1,2. Despite optimal aseptic techniques, endophthalmitis can occur when microbial-laden ocular fluid enters the anterior chamber (AC) through the corneal incision that is made to remove the cataractous lens. In the United States of America and Europe, bacteria are nearly always the cause of postoperative endophthalmitis, but in tropical regions, such as India, 10–20% of cases are caused by fungi3. While the exact incidence of postoperative endophthalmitis is unknown and likely underreported, published estimates range from 0.001 to 0.3%4. Visual complications can progress from decreased visual acuity (< 20/200) and severe eye pain to complete vision loss5. Patients also report decreased quality of life, increased financial costs, and prolonged hospitalization6,7.
To prevent endophthalmitis, prophylactic treatment with cephalosporins, aminoglycosides, fluoroquinolones, and/or chloramphenicol are recommended as part of infection prevention protocols. Intracameral cefuroxime, a second-generation cephalosporin, inhibits the formation of the peptidoglycan layer of the bacterial cell wall, which results in cell death8. The pivotal European Society of Cataract & Refractive Surgeons (ESCRS) study showed that intracameral cefuroxime significantly reduced the rates of postoperative endophthalmitis9,10. Intracameral cefuroxime boasts broad-spectrum effects, a wide safety margin, and has an acceptable tolerability profile, with most studies reporting few associated adverse events. Based on these findings, cephalosporins are widely used in clinical practice, including at Tan Tock Seng Hospital in Singapore, where cefazolin (0.1 ml/1 mg), a first-generation cephalosporin, is the standard of care for endophthalmitis prevention in cataract surgery. However, anaphylactic reactions (urticaria, bronchoconstriction, or life-threatening circulatory reactions) have been reported following treatment with cephalosporins in patients with penicillin allergies5,11. Additionally, the typical need for manual reconstitution of cephalosporins increases the risk of dilution errors, which can compromise the safety and efficacy of the treatment. These factors underscore the need for alternative options that can minimize these risks while maintaining effective prophylaxis.
The recently published Asian study on Perioperative Antibiotic prophylaXis (APAX) highlights the current variations in antimicrobial prophylaxis practice while also emphasizing the need for research and evidence-based guidelines, particularly in light of increasing concerns over the development of antimicrobial resistance12. A study from Tan Tock Seng Hospital demonstrated that gram-negative organisms make up a larger proportion of microbial isolates (25%) than reported in Western studies13. Levofloxacin (0.1 ml/0.5 mg), a third-generation fluoroquinolone antibacterial agent with broad-spectrum activity against gram-positive and gram-negative organisms, may have potential as a broad-spectrum candidate for postoperative ophthalmic prophylaxis, especially in Asia where gram-negative organisms are more prevalent than in Europe and the United States of America13. Animal and human studies suggest that levofloxacin is nontoxic to the eye14 and effective against Streptococcus pneumoniae (S. pneumoniae), a common cause of endophthalmitis15. Furthermore, it is suitable for patients with penicillin allergies. Two separate intracameral levofloxacin studies from the Philippines indicate an acceptable safety profile16,17 and Malaysian surgeons have also reported the successful use of intracameral levofloxacin18.
While topical levofloxacin is available in Singapore for ophthalmic conditions19, the intracameral effects have not been widely examined under clinical study conditions. Therefore, this study aimed to compare the safety profile of intracameral levofloxacin (0.1 ml/0.5 mg) with intracameral cefazolin (0.1 ml/1 mg) in patients from Singapore undergoing cataract surgery.
Methodology
Study design and participants
This was a prospective, single-center, double-blind, interventional, randomized controlled pilot study that was conducted in Singapore at Ang Mo Kio Specialist Centre, a Ministry of Health-accredited satellite medical center that is part of Tan Tock Seng Hospital (NCT06710977, retrospective registration date 02/12/2024). The trial took place between June 2022 and February 2024.
Study investigators recruited patients who were listed for cataract surgery in clinics, as well as those who were already scheduled and awaiting preoperative counselling at Tan Tock Seng Hospital. Patients were eligible to enroll if they were ≥ 50 to < 80 years of age and undergoing cataract surgery under the care of one of three study investigators, all of whom were consultant-level cataract surgeons. Only those with senile cataracts, which were assessed using the Lens Opacities Classification System III (LOCS III), and without features of complex cataracts, were included. Other exclusion criteria included prior ocular trauma; previous history of refractive, corneal, or intraocular surgery; corneal endothelial cell count of < 1500/mm2; concomitant ocular disease; allergy to penicillin, fluoroquinolone or cephalosporin; and abnormal biometric measurements. Full eligibility criteria are detailed in Supplementary Table 1.
Given the low incidence of endophthalmitis, this proof-of-concept study was designed to test the feasibility of the methodology and to ensure comparable safety before scaling up to a larger study. Therefore, no formal power analysis was conducted. Instead, a target sample size of 50 was determined based on feasibility considerations. Estimating a screen failure rate of 10%, up to five patients who failed screening were replaced.
Ethical approval was obtained from the National Healthcare Group Domain Specific Review Board. The study was conducted in accordance with Good Clinical Practice Guidelines and the ethical principles of the Declaration of Helsinki. All patients provided written informed consent prior to enrollment.
Procedures
Patients were randomly assigned to receive intracameral levofloxacin (0.1 ml/0.5 mg, Cravit® 0.5%; Santen Pharmaceutical Ltd.) or intracameral cefazolin (0.1 ml/1 mg, AFT Pharmaceutical Ltd) in a 1:1 ratio using a computerized system. Only the local study coordinator and a solo dedicated scrub nurse were aware of the treatment allocation; clinical staff were blinded throughout. The same scrub nurse prepared all study treatments. A total amount of 0.2 ml of levofloxacin (0.1 ml/0.5 mg) was drawn up into an unmarked 1.0 mL syringe from the ampule supplied by the manufacturer. Cefazolin was supplied in powder form and was first reconstituted with 10 mL of 0.9% normal saline. From this solution, 1 mL was withdrawn and further diluted with 9 mL of normal saline. A final volume of 0.2 mL was drawn into an unmarked 1.0 mL syringe. For both medications, immediately prior to instillation, 0.1 mL was expelled from the syringe to leave a final volume of 0.1 mL, which was then instilled into the eye. The appearance of the solutions and syringes was identical, ensuring the surgeon remained blinded to the treatment allocation.
In the surgical procedure, standard phacoemulsification was performed using the Stellaris Elite phacoemulsification system with standard viscoelastic (HEALON GV PRO, Johnson & Johnson, USA) and intraocular lens implantation (Sensar IOL, Johnson & Johnson, USA).
Study visits were conducted at baseline and at Week 1, Month 1, and Month 3 after surgery (Fig. 1). In addition to standard surgical measures, study assessments included best-corrected visual acuity (BCVA), which was measured using the Snellen Chart, as well as specular microscopy to assess both endothelial cell count (ECC) and central cornea thickness (CCT). ECC was assessed to evaluate corneal endothelial health while CCT was measured to quantify corneal edema. Spectral domain optical coherence tomography (SD-OCT) was also employed to monitor macular thickness and detect cystoid macular edema. A flare meter was used to quantify the amount of flare in the anterior segment of the undilated eye, with a minimum of three readings and a target of five readings per assessment. Clinical grading of the AC, for both AC cells and AC flare, was performed using the Standardization of Uveitis Nomenclature (SUN) grading scheme. Intraocular pressure (IOP) was measured in mmHg using Goldmann applanation tonometry. At Week 1 post procedure, an anterior segment examination was performed. A full manifest refraction was conducted at Month 1 and Month 3, along with a comprehensive slit-lamp biomicroscopy examination of the anterior and posterior segments of the operated eye.
Study schedule of ophthalmologic measures.
Outcomes
Assessing the safety profile of levofloxacin (0.1 ml/0.5 mg) was the overall aim of the study. Endpoints included the occurrence of endophthalmitis (yes/no) during the 3-month follow-up period, BCVA; the presence of AC inflammation, including grading of AC cells and the presence of flare (yes/no); IOP; CCT; central foveal thickness (CFT); and cell density. The incidence of adverse events, including dose and dilution errors, was monitored throughout the study.
Statistical analyses
Patient demographic characteristics were summarized using descriptive statistics. Occurrence of endophthalmitis was compared between the two treatment groups using Fisher’s exact test. The same method was used to compare the occurrence of AC flare between groups. The Mann–Whitney U test was used to compare AC cell grades between groups, accounting for the category orders. For the remaining outcomes, which were continuous in nature, treatment groups were compared using analysis of covariance. The outcome variable was the postoperative measurement (either Week 1, Month 1, or Month 3), with the equivalent outcome at baseline included as a covariate in the analysis. Variables that were found to follow an approximate normal distribution were analyzed on the original scale of measurement. To meet the assumptions of the statistical test, variables that followed a positively skewed distribution (BCVA and CFT) were analyzed on a log scale. For BCVA, a small constant was added to all values before log transformation to account for the zero values.
Results
A total of 129 patients were assessed for eligibility (Fig. 2). Of these, 50 patients were enrolled, with 25 randomly assigned to receive levofloxacin (0.1 ml/0.5 mg) and 25 to cefazolin (0.1 ml/1 mg) (Fig. 2). All participants completed the trial.
CONSORT study design showing patient flow.
Patient demographics were similar between treatment groups (Table 1). The mean age ± SD in the levofloxacin (0.1 ml/0.5 mg) group was 69.2 ± 4.6 years and 68.4 ± 6.9 years in the cefazolin (0.1 ml/1 mg) group. Across both groups, there were more male than female patients, and most patients were of Chinese ethnicity. There was a slight majority of right study eyes in both groups.
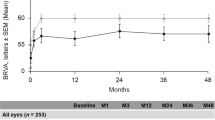
No cases of endophthalmitis were detected in either the levofloxacin (0.1 ml/0.5 mg) group or the cefazolin (0.1 ml/1 mg) group throughout the follow-up period (0% [95% CI: –13, 13]). BCVA outcomes were comparable between the groups (Table 2). Similarly, no differences were observed between the treatment groups in the occurrence of AC flare over the follow-up period, or in AC cell grade at any timepoint (Table 2). No differences were observed between treatment groups in CFT, IOP, CCT, or cell density at any timepoint (Table 2).
Most patients in both groups did not experience AC flare at Week 1. Only 1 (4%) patient in each group recorded a faint flare (SUN score = 1), with no scores higher than 1. Most patients recorded an AC cell grade of 0.5 at Week 1 post procedure, including 20 (80%) patients in the levofloxacin (0.1 ml/0.5 mg) group and 22 (88%) in the cefazolin (0.1 ml/1 mg) group. At Week 1, three (12%) patients in each group reported an AC cell grade of 1. By Month 1 over 90% of patients, in both groups, reported an AC grade of 0, indicating no inflammation. At Month 3, only one patient in each group reported an AC cell grade of 1 (Table 3).
Discussion
Our pilot trial is the first double-blind, randomized controlled study to compare intracameral levofloxacin (0.1 ml/0.5 mg) to intracameral cefazolin (0.1 ml/1 mg). Treatment with levofloxacin (0.1 ml/0.5 mg) was tolerated and no new safety signals emerged.
Safety outcomes – as measured by CFT, IOP, CCT, and cell density – were acceptable, and there were no differences between the study groups. Inflammation, as assessed via AC cell grades, decreased rapidly, such that after Month 1, over 90% of patients reported AC cell grades of 0. Only one patient in each group reported AC flare. There were no reported adverse drug reactions in the levofloxacin (0.1 ml/0.5 mg) or cefazolin (0.1 ml/1 mg) groups. Additionally, there were no cases of endophthalmitis in either treatment group. These safety results support earlier levofloxacin studies16,17. In a prospective, single-arm, interventional study, Espiritu and colleagues (2007) evaluated the safety of intracameral injection of levofloxacin 0.5% in 50 patients who were undergoing cataract extraction and intraocular lens implantation. Similar to our study, there were no cases of endophthalmitis and no safety concerns relating to BCVA, CFT, CCT, and AC inflammation. A 2022 study of 114 eyes that compared intracameral levofloxacin 0.5% and moxifloxacin 0.5% also reported no cases of endophthalmitis and no safety concerns relating to CFT, CCT, and cell density in both treatment groups16.
The findings of this study are crucial in demonstrating the safety of intracameral levofloxacin (0.1 ml/0.5 mg), given the known variation of infection prophylaxis practice patterns in cataract surgery across Asia12. The APAX study surveyed clinicians from the Asia region, including Singapore, and reported that the use of intracameral antibiotics was 60.0% in low-risk cases and 66.7% in high-risk cases12. A commonly cited barrier to greater usage of intracameral antibiotics is the lack of a commercial preparation approved for use in the country, resulting in off-label use of other agents12,20. Worryingly, not all institutions reported having established protocols for antimicrobial use in cataract surgery, nor did they have monitoring programs for emerging antimicrobial resistance. While preoperative topical antibiotic use is declining in Europe and Australia, it remains prevalent in Asian countries such as China, India, and Japan12. Postoperative use also remains high; more than half of the respondents in the APAX survey reported prescribing topical antibiotics for 2–4 weeks post procedure. Postoperative antibiotic use can contribute to antimicrobial resistance due to patients not always adhering to prescribed dosing and timing regimens. Additionally, patients may not consistently follow best practices such as washing hands, avoiding contamination of the bottle tip, correctly instilling drops, and using the correct number of drops. This issue is further compounded by the large discrepancy between patients’ self-reported perspectives and their observed techniques21. In the current healthcare landscape, implementing evidence-based policies is vital in preventing antimicrobial resistance, and ensuring that antimicrobials are used where they are most effective.
Accumulating evidence suggests that intracameral delivery, such as that used in this study, is optimal to prevent endophthalmitis, with one meta-analysis study showing that the occurrence of endophthalmitis was reduced from 1 in 2855 procedures to 1 in 485 (p < 0.00001), representing a relative risk reduction of 0.12 (95% CI: 0.08, 0.18)20. A more recent meta-analysis of over 6.8 million eyes showed that intracameral injection of vancomycin, cefazolin, cefuroxime, or moxifloxacin prevented postoperative endophthalmitis2. Due to the rare occurrence of postoperative endophthalmitis, there are few randomized studies in this area. The pivotal ESCRS study recruited and randomized over 16,000 patients into four groups to receive intracameral cefuroxime, intracameral cefuroxime with topical levofloxacin, no intracameral injection and placebo vehicle drops, or no intracameral injection and topical levofloxacin. The absence of intracameral cefuroxime was associated with a 4.92-fold increase (95% CI: 1.87, 12.9) in the risk of postoperative endophthalmitis10. It was on the basis of these results that intracameral cefazolin, a first-generation cephalosporin, was used as the control therapeutic in this study (and is currently used at Tan Tock Sen Hospital as standard of care). Cefazolin and cefuroxime are used widely, and continue to show efficacy in preventing endophthalmitis2; however, they require reconstitution and dilution, which can lead to administration errors and subsequent toxicities22,23,24. Erroneous use of cefuroxime has been associated with corneal edema, raised IOP, pigment precipitates in the AC, loss of corneal endothelial cells, pigmentary changes on the retina, tritanopia-type color-vision defects, impairments in dark adaptation, lowered threshold in the visual field, changes in electroretinography values, reduced rod photoreceptor cell function, development of retinal infarction, noninfectious panuveitis, macular detachment, and permanent loss of vision23,24,25,26. Toxic anterior segment syndrome – a postoperative inflammatory reaction with permanent sequelae, such as corneal decompensation, intractable glaucoma, and cystoid macular edema27 – has also been associated with inadvertent overdoses of cefuroxime28,29. Importantly, in this study, there were no drug administration errors. Levofloxacin (0.1 ml/0.5 mg) is packaged in a ready-to-use formulation and does not require reconstitution or dilution, greatly reducing the potential for administrative errors and toxicities.
The key strengths of this study include the prospective study design, robust randomization and blinding protocol, and 100% retention rate at 3 months. The outcome measures were well established, clinically meaningful, and precisely calibrated. Timepoints were extended beyond the perioperative period to ensure that all related adverse events were captured. A further notable strength is the involvement of a small number of surgeons, only three, which minimized variation in patient care and surgical techniques.
While the population for this pilot study was small and not designed to detect the extremely rare occurrence of endophthalmitis, it aligns with the populations studied in other intracameral ophthalmology studies16,17. The exploratory nature of this study means that it may have been underpowered to show differences in safety outcomes and any findings require confirmation in a prospective, powered study. A limitation of this study is that it was conducted at a single center, which may limit the generalizability of the results to other regions. However, these findings will help to inform the design of a larger, multicenter study with subgroup analyses.
In conclusion, this pilot study provides proof of concept that intracameral levofloxacin (0.1 ml/0.5 mg) and intracameral cefazolin (0.1 ml/1 mg) may have a comparable safety profile in patients who are undergoing routine cataract surgery. Intracameral levofloxacin may offer a viable alternative to cefazolin, particularly for patients with penicillin allergies, while also reducing the risk of dilution errors by eliminating the need for reconstitution. Larger, multicenter, randomized controlled studies are warranted to confirm these findings and explore their broader applicability in preventing postoperative endophthalmitis.
Data availability
Data is provided within the manuscript or supplementary information files. The full dataset that supports the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. Data are located in controlled access data storage at Tan Tock Seng Hospital.
References
Grzybowski, A. Recent developments in cataract surgery. Ann Transl Med. 8, 1540. https://doi.org/10.21037/atm-2020-rcs-16 (2020).
Kato, A. et al. Prophylactic antibiotics for postcataract surgery endophthalmitis: a systematic review and network meta-analysis of 6.8 million eyes. Sci Rep. 12, 17416. https://doi.org/10.1038/s41598-022-21423-w (2022).
Gupta, A. et al. Spectrum and clinical profile of post cataract surgery endophthalmitis in north India. Indian J. Ophthalmol. 51, 139–145 (2003).
Levison, A. L., Mendes, T. S. & Bhisitkul, R. Postprocedural endophthalmitis: A review. Expert Rev. Ophthalmol. 8, 45–62 (2013).
Althiabi S., Aljbreen A. J., Alshutily A., Althwiny, F. A. (2022) Postoperative endophthalmitis after cataract surgery: an update. Cureus 14
Clark, A. et al. Quality of life after postoperative endophthalmitis. Clin. Exp. Ophthalmol. 36, 526–531 (2008).
Schmier, J. K., Hulme-Lowe, C. K., Covert, D. W. & Lau, E. C. An updated estimate of costs of endophthalmitis following cataract surgery among Medicare patients: 2010–2014. Clin. Ophthalmol. 10, 2121–2127 (2016).
Scott, L. J., Ormrod, D. & Goa, K. L. Cefuroxime axetil: An updated review of its use in the management of bacterial infections. Drugs 61, 1455–1500 (2001).
Barry, P. et al. ESCRS study of prophylaxis of postoperative endophthalmitis after cataract surgery: Preliminary report of principal results from a European multicenter study. J. Cataract Refract. Surg. 32, 407–410 (2006).
Endophthalmitis Study Group. Prophylaxis of postoperative endophthalmitis following cataract surgery: Results of the ESCRS multicenter study and identification of risk factors. J. Cataract Refract. Surg. 33, 978–988 (2007).
Moisseiev, E. & Levinger, E. Anaphylactic reaction following intracameral cefuroxime injection during cataract surgery. J. Cataract Refract. Surg. 39, 1432–1434 (2013).
Garg, P., Khor, W.-B., Roy, A. & Tan, D.T.-H. A survey of Asian eye Institutions on perioperative antibiotic prophylaxis in cataract surgery. Int. Ophthalmol. 43, 4151–4162 (2023).
Tan, C. S. H., Wong, H. K. & Yang, F. P. Epidemiology of postoperative endophthalmitis in an Asian population: 11-year incidence and effect of intracameral antibiotic agents. J. Cataract Refract. Surg. 38, 425–430 (2012).
Edwar, L., Bisara, B. B., Setiabudi, R., Susanto, E. & Badruddin, G. H. Safety of intracameral injection of levofloxacin 0.5% eye drops single dose 0.6 ml preservative free on rabbit eye. F1000Res 12, 772. https://doi.org/10.12688/f1000research.133293.1 (2023).
Miller, J. J. et al. Endophthalmitis caused by Streptococcus pneumoniae. Am. J. Ophthalmol. 138, 231–236 (2004).
Cruz, F. M., Uy, H. S., Rubio, C. J. & Chan, P. S. Postoperative safety outcomes in patients undergoing routine phacoemulsification cataract surgery with intraoperative intracameral injection of preservative-free moxifloxacin versus levofloxacin. Philipp. J. Ophthalmol. 47, 15–22 (2022).
Espiritu, C. R. G. & Bolinao, J. G. Prophylactic intracameral levofloxacin in cataract surgery–an evaluation of safety. Clin. Ophthalmol. 11, 2199–2204 (2017).
Wai, Y. Z., Chong, Y. Y., Lim, L. T., Chan, J. B. & Chandrasekaran, S. Preferred cataract surgery practices in Malaysia: A survey based study. BMC Res. Notes. 16, 116. https://doi.org/10.1186/s13104-023-06391-2 (2023).
Cravit 1.5% Product Information Sheet. Santen. (2020).
Kessel, L. et al. Antibiotic prevention of postcataract endophthalmitis: A systematic review and meta-analysis. Acta Ophthalmol. 93, 303–317 (2015).
An, J. A., Kasner, O., Samek, D. A. & Lévesque, V. Evaluation of eyedrop administration by inexperienced patients after cataract surgery. J. Cataract Refract. Surg. 40, 1857–1861 (2014).
Sakarya, Y. & Sakarya, R. Cefuroxime dilution error. Eur. J. of Ophthalmol. 20, 460–461 (2010).
Delyfer, M. N. et al. Ocular toxicity after intracameral injection of very high doses of cefuroxime during cataract surgery. J. Cataract Refract. Surg. 37, 271–278 (2011).
Olavi, P. Ocular toxicity in cataract surgery because of inaccurate preparation and erroneous use of 50mg/ml intracameral cefuroxime. Acta Ophthalmol. 90, e153-154 (2012).
Sül, S. & Karalezli, A. Development of retinal infarct due to intracameral cefuroxime injection following complicated cataract surgery. Turk. J. Ophthalmol. 48, 317 (2018).
Kamal-Salah, R., Osoba, O. & Doyle, E. Ocular toxicity after inadvertent intracameral injection of high dose of cefuroxime during cataract surgery: A case series. Retin. Cases Brief Rep. 13, 269–272 (2019).
Verma, L., Malik, A., Maharana, P. K., Dada, T. & Sharma, N. Toxic anterior segment syndrome (TASS): A review and update. Indian J. Ophthalmol. 72, 11–18 (2024).
Çakır, B. et al. Toxic anterior segment syndrome after uncomplicated cataract surgery possibly associated with intracamaral use of cefuroxime. Clin. Ophthalmol. 9, 493–497 (2015).
Sengillo, J. D. et al. Postoperative endophthalmitis and toxic anterior segment syndrome Prophylaxis: 2020 Update. Ann. Transl. Med. 8, 1548. https://doi.org/10.21037/atm-2019-rcs-02 (2020).
Acknowledgements
Medical writing assistance was provided by Jessica Reid, PhD, and Hanne Stotesbury PhD, from Synergy Vision Ltd in accordance with Good Publication Practice (GPP 2022) guidelines, and statistical assistance was provided by Paul Bassett from Statsconsultancy Ltd, with funding from Santen Pharmaceutical Asia Pte. Ltd.
Funding
This research study has received financial support from Santen Pharmaceutical Asia Pte. Ltd.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection and analyses were performed by Yan Tong Koh and Zhi Hong Toh. Supervision was provided by Don Chern Kuok Pek, Tun Kuan Yeo, John Xin Hao Wong, Wei Kiong Ngo and Han Bor Fam. Development of the first draft of the manuscript was performed by Yan Tong Koh, and all authors commented on all versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
Ethical approval was obtained from the National Healthcare Group Domain Specific Review Board.
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Koh, Y., Yeo, T., Toh, Z.H. et al. A double-blind, randomized controlled pilot study comparing the safety of intracameral levofloxacin and intracameral cefazolin in patients undergoing cataract surgery. Sci Rep 15, 34444 (2025). https://doi.org/10.1038/s41598-025-17553-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17553-6