Abstract
Despite success in preclinical animal models, most cancer prevention drugs fail to show efficacy in clinical trials. One potential reason could be that study end points in animal models are not sufficiently robust. Preclinical efficacy studies often use tumor development, as measured by palpation, as an endpoint. A comprehensive study design that includes more rigorous measures by histologic assessment of all the mammary glands is less common. We hypothesized that the efficacy of drug treatment may vary based on the approach to tumor assessment. SV40C3TAg mice that spontaneously develop TNBC-like tumors were used to assess the efficacy of drugs for breast cancer prevention. Histological grading was performed using a 5-point scale. Animals were treated with fluvastatin and/or aspirin for 16 weeks and the experiment was concluded at 22 weeks, and all the mammary glands removed for histologic assessment. Grossly palpable mammary glands (> 3 mm) and /or histologic grade 4–5 was considered tumor bearing and treatment non-responder. 112 mammary glands from 40 mice were assessed. A high concordance (> 90%) was noted between palpably enlarged glands and the finding of grade 4–5 lesions. Conversely, among 74 non-palpably enlarged mammary glands, more than 66% were histologically high-grade and would have been wrongly classified as tumor-free based on gross tumor measurement. In drug efficacy studies, significant differences were noted in the rate of treatment response based on the method of tumor assessment, with a 22% response rate noted using the histological grading system and a 65% response rate using gross palpation. These analyses suggest that there is poor concordance between the gross tumor measurements and histological tumor assessment in a mouse model of breast cancer. Thus, gross tumor measurement alone is insufficient for determining chemopreventive efficacy in preclinical animal models.
Similar content being viewed by others
Introduction
A large number of breast cancer prevention agents have shown efficacy in preclinical animal models such as metformin, tamoxifen, retinoids receptor inhibitor, lapatinib, rexinoid, LG100268, selective estrogen receptor modulator (arzoxifene or acolbifene), 25-Hydroxy Vitamin D, and anti-progestins (such as mifepristone)1,2,3,4,5,6,7,8,9,10,11,12,13,14. But only a handful of these chemo preventive agents (such as tamoxifen, arzoxifene and raloxifene) have proven to be clinically effective. While this disconnect between preclinical success and clinical efficacy may be attributed to several complex and interrelated factors such as inherent differences between mouse models and humans, the impact of endpoint selection in the preclinical setting and its potential to bias interpretation of efficacy has not been studied. The overwhelming majority of the animal studies with the aforementioned agents used tumor mass palpation method as a measure of response which we hypothesize could have overestimated their potential efficacy1,2,3,4,5,6,7,8,9,10,11,12,13,14.
Several factors determine the choice of study end points for assessing cancer drug efficacy in mouse models. One such determinant is the intended use of an agent -cancer treatment/cancer prevention drug. For cancer treatment studies, drug efficacy is generally assessed through survival studies or regression in tumor size. However, in the context of breast cancer prevention, the lengthy wait time required for tumor development and thus to conduct overall survival (OS) studies often limits the use of OS as an end point. Consequently, many breast cancer-prevention preclinical studies have relied on tumor development as the outcome, measured by presence of enlarged palpable mass.
These tumor palpability-based measures include gross tumor onset, gross tumor incidence, tumor volume and tumor burden/multiplicity, which are frequently used as surrogate endpoints by preclinical animal studies to demonstrate efficacy for prevention of breast cancer. However, gross tumor palpability only provides a crude measure of tumor development, has high inter-observer variability and can miss microscopic breast cancer lesions. An alternative measure of tumor assessment relies on histological evaluation with direct microscopic examination of breast tissue15,16. However, this system is underutilized in preclinical animal-based breast cancer prevention studies, despite its utility to offer valuable insights into the morphological characteristics of breast tumors, disease progression and their clinical behavior17. While tumor size shrinkage can offer a convenient and quick overview of drug efficacy, an endpoint that can confirm histological absence of breast cancer will be more precise and thus desirable for successful development of chemopreventive drugs for breast cancer.
It remains to be determined if there is a consensus between gross presence of enlarged mass in the mouse mammary gland and histologic finding of tumor and thus whether palpation of a mass alone is appropriate surrogate end point for breast cancer prevention drug efficacy studies. We hypothesize that there is poor concordance between grossly palpable mass in the mammary gland and histological presence of tumor in mouse models of cancer and thus gross palpability alone may be an inadequate surrogate end point for preclinical studies testing of novel agents for breast cancer prevention.
Results
Choice of study end point in evaluating cancer progression in a mouse model
To address if gross tumor measurement is an adequate end point for tracking cancer development in a spontaneous mouse model of TNBC, we measured the concordance between gross tumor palpability and high histologic grade representative of in situ or invasive carcinoma. We first evaluated the distribution of histological grades within the palpable and non-palpable mammary glands from all 40 adult SV40C3TAg mice (representing112 distinct mammary glands) in the study, regardless of treatment group (Fig. 1). Out of the 38 grossly palpable mammary glands, approximately 89.4% were found to be grade 5 (invasive cancer) and 5.3% of tumors were classified as grade 4 (in situ cancer) histologically. Another 2 glands (5.3%) showed the highest-grade lesion of atypia (grade 3) (Fig. 2a). Sections representative of these histologic grades are shown in Fig. 2d. The positive predictive value for a grade 4 or 5 tumor of a palpable nodule in the mammary gland was greater than 94%. This highlights a strong concordance between gross morphology and histological findings for palpably enlarged mammary gland nodules in SV40 C3TAg mice.
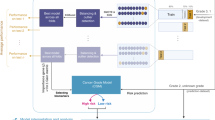
Study schematic. SV40C3TAg mice were treated starting at 6 weeks of age to 22 weeks of age. Glands were collected either at 22 weeks or prior if tumor size surpassed the maximal allowable tumor burden (< 20 mm). For gross measurements, glands were classified as tumors if the gland measured greater than 3 mm in any single dimension. Histological grades 4 and 5 were considered Tumor bearing (cancer), while grades 0–3 were considered Tumor-free (benign).
Concordance between histologic grade and gross assessment of tumor. (A) Bar Graph of the distribution of histologic grading across all palpable and non-palpable glands. Each mammary gland was considered a separate data point. (B) Analysis of concordance between histologic grade and gross tumor assessment by gland. Each mammary gland was considered a separate data point. (C) Analysis of concordance between histologic grade and gross assessment by mouse. Mice were considered to be tumor bearing if at least one gland was palpable and/or carried histologic grade 4 or 5 lesion. (D) H/E-stained images of various histological grades observed with in the palpable and non-palpable mammary glands.
Conversely, among the 74 mammary glands categorized as non-palpable, 44 glands (59.4%) consisted of grade 5 lesions and 5 glands (6.76%) were classified as grade 4. The grade distribution in the remaining, histologically tumor free glands was grade 3 in 16.2%, grade 2 in 5.4%, grade 1 in 6.78% and grade 0 in 5.4% (Fig. 2a). Sections representative of these histologic grades are shown in Fig. 2d. Importantly, 2/3rds of non-palpable mammary glands were identified as tumor bearing (DCIS or invasive) upon microscopic examination. While there was a significantly higher likelihood that a non-palpable mammary gland would harbor non-malignant low grade (0–3) lesions, as compared to a palpable mammary gland (33.8% vs. 5%, p = 0.008, Fig. 2b), overall, the negative predictive value for tumor (grade 4–5 lesion) of a non-palpable gland was very low at 33.8%, and can erroneously suggest absence of cancer in animal studies.
The effect of this discordance between histology and gross assessment of the mammary gland was then analyzed at the organism level, with mice classified as tumor bearing if a palpable tumor and/or histologically confirmed tumor was noted in at least 1 of the 4 mammary glands. Out of the 25 mice with one or more palpably enlarged glands, 96% showed histology consistent with tumor. However, for the 13 mice without any palpably enlarged glands and thus grossly deemed to be tumor free, 85% were noted to have tumors when assessed histologically (Fig. 2c). Therefore, the absence of a palpable mass in the mammary gland is not a reliable indicator of the absence of tumor histologically. In fact, the overall incidence of histologically confirmed grade 4 or 5 lesions between grossly tumor bearing (palpable) and grossly tumor free (non-palpable) animals were not significantly different (Fig. 2c, p = 0.27) and further supports the use of histological evaluation for correct assessment. These findings suggest that a study design that is solely based on gross evaluation of the mammary gland for the presence of tumor does not capture true incidence of disease.
Choice of study end point in assessment of preclinical drug efficacy
Previously, we used the gross palpability method to assess the efficacy of fluvastatin, aspirin and the combination to prevent the development of mammary tumors in SV40 C3TAg animals18. We have extended these analyses further to now compare the impact of the tumor assessment approach on estimations of drug efficacy in animal studies. SV40C3TAg mice treated with fluvastatin alone, aspirin alone or combination were used for these analyses and all drug treated animals were combined into a single group for comparison with vehicle treated group. Among the 81 mammary glands examined, 53 (65%) were deemed tumor-free based on gross assessments and thus considered responsive to treatment, however only 18 of these (22%) glands were characterized as histologically negative for tumor (Fig. 3A). Consequently, there was a significant difference in drug efficacy based on the tumor assessment method chosen (65% vs. 22% p < 0.01).
Comparison between gross assessment and histological grading- two different methods of drug efficacy measurement. Analysis limited to animals in drug treatment groups. (A) Analysis of drug efficacy per gland irrespective of the treatment. Response to treatment was significantly greater when tumor assessments were performed by palpation method vs. histologic grading method. (B) Analysis of drug efficacy per mouse. Mice were considered to be non-responders if at least one gland was palpable and/or carried histologic grade 4 or 5 lesion.
We also assessed the proportion of responder animals based on gross vs. histologic determination of treatment response. For this analysis, a mouse with a palpable tumor and/or high-grade lesion in any one of its four mammary glands examined was categorized as a tumor-bearing (non-responder) mouse. Out of the 28 mice in the treatment groups, 10 mice (36%) were classified as tumor-free based on gross assessments, but only 2 mice (7%) were determined to be tumor-free histologically, again resulting in a significant discrepancy in the determination of drug efficacy (36% response rate grossly vs. 7% response rate histologically, P = 0.03) (Fig. 3B).
We further evaluated the effect of method of tumor assessment (gross palpability vs. histological grading) on drug efficacy by analyzing each of the 4 treatment groups separately. These treatment groups included fluvastatin alone, aspirin alone, fluvastatin + aspirin and the vehicle treatment. These analyses revealed that tumor incidence (proportion of animals with one or more tumors) driven from histological grading was higher in all treatment groups compared to the assessment based on gross tumor palpability (Fig. 4, statistically not significant). While aspirin was found to be ineffective in preventing breast cancer in SV40C3TAg based on either gross palpitation or histologic determination, the efficacy of fluvastatin varied remarkably based on the assessment method. When assessed using gross palpation method, fluvastatin appeared to be effective with tumor incidence of 30% as compared to an incidence of 70% in control animals (p = 0.057); however, when evaluated histologically 100% of the animals harbored high grade tumors (grade 4 or 5) thus showing no real efficacy of fluvastatin as compared to vehicle treated animals. Despite the histological presence of tumors in all of the fluvastatin treated mice, the overall disease burden appears to be lesser as indicated by the low proportion of mice with grossly evident disease (Fig. 4) and the previously reported delayed average onset of gross tumors by > 3 weeks18. Whether this lower gross burden of disease translates to better overall survival outcome requires additional investigation. These findings suggest that a more holistic/comprehensive end point, such as overall survival, may provide a better measure of drug efficacy.
A trend of better drug efficacy with gross palpability as compared to histological assessment of mammary glands in SV40C3TAg mouse model. A mouse was considered tumor bearing if it had a palpable or high grade (grade 4 or 5) lesion in one or more of its mammary glands. p values are indicated.
Discussion
The optimal approach to diagnosis of breast tumors in preclinical mouse models is not well established, with most studies using gross palpation, rather than extirpation and histologic evaluation of the main mammary glands as the basis for classification of disease. Whether gross palpation of enlarged tumor mass is an appropriate and sufficient surrogate for histologic findings is unclear, and our main objective in this study was to evaluate the concordance between these two diagnostic strategies. Inclusion of a surrogate measure in a preclinical animal study design that accurately captures the extent of cancer progression and predicts treatment response is important to optimize selection of agents for translation from the laboratory to clinical trial. In the current study, we report that 2/3rd of grossly normal appearing mammary glands in 22-week-old SV40 C3TAg mice were in fact tumor bearing microscopically. This finding suggests that relying on tumor detection through palpation is an insufficient endpoint marker for tracking the presence of cancer or evaluating efficacy of a drug. We found that reliance on gross palpation of enlarged mass for assessing efficacy of chemo-preventive agents resulted in a 5-fold overestimation of the drug’s true efficacy.
There are several advantages of tracking tumor burden by Vernier calipers or imaging such as non-invasiveness, ease of measurement, feasibility to periodically track tumor development, and quantitative response that can be used as a first pass to exclude ineffective agents in preclinical drug treatment studies. However, only about 1/3rd of animal trials found tumor burden to have an association with cancer progression. In a recent meta- analysis that included 1830 patient derived xenograft-based mouse trials with 61 different cancer treatment drugs, only 30% of studies found tumor burden to be a true surrogate that associated with disease progression19. Thus, gross evaluation of disease appears to have only modest utility in the context of assessing the efficacy of drugs in treatment studies and as noted in our study, gross evaluation of the mammary glands also had very limited utility when assessing the efficacy of drugs for prevention of breast cancer.
While histological grading of the mammary gland necessitates removal of the gland and thus eliminates the ability to track tumor over time, this approach provides better estimation of the presence or absence of disease, a key metric when agents are being tested for prevention of breast cancer. While our data makes clear that histologic grading of the mammary glands is superior to gross palpation in determining the chemo-preventive potential of drugs, whether this histologic endpoint is more likely to be associated with improvements in survival, the ultimate goal of preventive treatment, is unknown. Well-designed animal studies incorporating multiple surrogate end points (such as gross examination and histological grading) along with overall survival are needed to understand the true utility of these surrogate endpoints in the design of preclinical studies.
Our study has a number of limitations. It should be noted that standard histologic examination does not fully exclude the presence of focal areas of pre-invasive or invasive disease, as small foci may be missed when processing in 5-micron thick histologic sections. Additionally, we chose to examine only 2 levels (1st cut and 5th cut) from each gland, which again may have resulted in underestimation of the true extent of microscopic disease. However, our approach approximates standard histopathologic processes, and this approach still constitutes a reasonable comparative estimate across groups. Additional limitations of our study include the use of only one mouse model of breast cancer and two chemoprevention drugs. It remains to be further explored if our findings are generalizable across other mouse models and drugs.
In conclusion, our results underscore the limitations of using palpation of enlarged mammary gland for determination of tumor and efficacy of chemo-preventive agents for breast cancer. Preclinical study designs should include either histologic evaluation or an additional measure of drug responsiveness such as survival in order to improve the study reproducibility and enhance their potential for translation in the clinic.
Methods
Animals
SV40C3 TAg hemizygous female mice that spontaneously develop triple negative breast cancer were used for the study. These mice were generated by crossing hemizygous transgene (SV40 T antigen) carrying male mice with wild-type female mice of the FVB/N strain. The SV40 T antigen (TAg) oncogene driven by the C3 (1) fragment of rat prostate steroid binding protein promoter, starts expressing in the mammary cells of female mice when the mice reach about 4 weeks of age18,20,21. This leads to p53 mutations and inactivation in RB pathway triggering breast tumorigeneis, starting with atypia around 8 weeks of age, followed by DCIS at approximately 12 weeks, and eventually progressing to invasive breast cancer, which can occur as early as 16 weeks (Fig. 1). By 22 weeks of age, at least 80% of animals are reported to be tumor bearing18. All animal experiments were conducted following the guidelines of UTMD Anderson Cancer Center’s Institution animal care and use committee (IACUC) and is in compliance with the ARRIVE guidelines (https://arriveguidelines.org).
Drug treatments and tumor measurements
Female mice of approximately 6 weeks age, were treated with fluvastatin (10 mg/kg b.wt), aspirin (20 mg/kg b.wt in 0.01% ethanol), fluvastatin and aspirin combination (10 mg/kg b.wt and 20 mg/kg b.wt respectively) or vehicle control (0.01% ethanol) via drinking water. The treatments were replenished in the fresh water every other day. Water consumption was recorded twice a week and drug doses were adjusted to maintain their specified dose. The general health check of mice including body weight was conducted weekly and no adverse effect was noted. At the end of the 16-week treatment period (6 weeks of age to age 22 weeks), all mice were humanely euthanized, and the thoracic and inguinal mammary glands were harvested, regardless of gross appearance.
Onset of tumors grossly was tracked by biweekly physical exam to monitor any palpable tumors, starting at the age of 10 week until the age of 21 weeks. A palpable mass of 3 mm or larger in any single dimension as measured by Vernier Calipers was considered a tumor. This threshold was used because it was challenging to reliably detect or identify masses smaller than 3 mm in the mammary gland. Tumor width and length were measured by Vernier calipers. Mice tumors were not allowed to grow more than 1.5 cm size in any linear measurement. If multiple tumors were present in a mouse, the combination of two longest diameters was not allowed to exceed more than 2 cm as per UTMD Anderson Cancer center’s IACUC guidelines. Animals were humanely euthanized if the tumor size approached these limits. Tumor incidence was defined as the number of animals with palpable lesions or grade 4–5 lesions at 22 weeks just prior to necropsy.
Tissue processing and histology grading
At the time of necropsy, thoracic and inguinal mammary glands from each mouse were harvested, regardless of whether they were grossly tumor bearing, weighed and each gland processed separately. Mammary glands were fixed in formalin followed by embedding in paraffin (FFPE). FFPE blocks were cut into 5-µm-thick sections and first and fifth level cut were stained with hematoxylin and eosin (H/E).
A histological grading system ranging from grade 0–5, described by Dawson et al. and Visscher et al., was used as the basis for histological grading in this study12,22. Briefly, this grading system ranged from simple tubules with 1–2 cell layers (grade 0); simple tubules with > 2 cell layers but no architectural complexity (grade 1); complex hyperplasia (grade 2); atypical hyperplasia (grade 3); ductal carcinoma in situ (grade 4), and invasive carcinoma (grade 5) with following modifications. A grade was classified as recommended if majority of features described in the histological grading criteria (by Dawson et al.) were present even in the absence of one or two defining features22. This approach approximates clinical practice where the pathologist will frequently consider the preponderance of features for classification of disease. Grading in this study was overseen by a breast pathologist (CTA). For purposes of our analysis, grades 0–3 were considered low grade or tumor free, while grades 4–5 were considered high grade or tumor bearing. If a spectrum of grades was noted within a gland, highest grade was recorded and used in the analysis.
Statistical analysis
Fisher’s exact test was used to determine drug efficacy among various groups and to assess the level of concordance between histologic grade and gross evaluation of tumor (palpable vs. non-palpable glands). McNemar’s test was utilized to determine the agreement between the two assessment approaches used (gross vs. histologic) in measuring drug efficacy. P < 0.05 was considered statistically significant difference.
All the methods used in the manuscript were carried out in accordance with UTMD Anderson Cancer Center’s regulations and committees such as Environment health and safety (EH&S) and Institutional committee for animal care and use (IACUC).
Data availability
All the data supporting the results reported in this article are presented in the form of figures in here.
Abbreviations
- SV40C3(1)TAg:
-
Simian Virus 40 C3(1) large T-antigen
- DCIS:
-
Ductal carcinoma in situ
- H/E:
-
Hematoxylin and eosin
- TNBC:
-
Triple negative breast cancer
- FFPE:
-
Formalin fixed paraffin embedded
- OS:
-
Overall survival
References
Anisimov, V. N. et al. Effect of Metformin on life span and on the development of spontaneous mammary tumors in HER-2/neu Transgenic mice. Exp. Gerontol. 40, 685–693. https://doi.org/10.1016/j.exger.2005.07.007 (2005).
Anisimov, V. N. et al. Metformin extends life span of HER-2/neu Transgenic mice and in combination with melatonin inhibits growth of transplantable tumors in vivo. Cell. Cycle. 9, 188–197. https://doi.org/10.4161/cc.9.1.10407 (2010).
Bojkova, B. et al. Metformin in chemically-induced mammary carcinogenesis in rats. Neoplasma 56, 269–274. https://doi.org/10.4149/neo_2009_03_269 (2009).
Liby, K. et al. The combination of the rexinoid, LG100268, and a selective Estrogen receptor modulator, either arzoxifene or acolbifene, synergizes in the prevention and treatment of mammary tumors in an Estrogen receptor-negative model of breast cancer. Clin. Cancer Res. 12, 5902–5909. https://doi.org/10.1158/1078-0432.CCR-06-1119 (2006).
Liby, K. et al. Prevention and treatment of experimental Estrogen receptor-negative mammary carcinogenesis by the synthetic triterpenoid CDDO-methyl ester and the rexinoid LG100268. Clin. Cancer Res. 14, 4556–4563. https://doi.org/10.1158/1078-0432.CCR-08-0040 (2008).
Moon, R. C. et al. N-(4-Hydroxyphenyl)retinamide, a new retinoid for prevention of breast cancer in the rat. Cancer Res. 39, 1339–1346 (1979).
Poole, A. J. et al. Prevention of Brca1-mediated mammary tumorigenesis in mice by a progesterone antagonist. Science 314, 1467–1470. https://doi.org/10.1126/science.1130471 (2006).
Rossdeutscher, L. et al. Chemoprevention activity of 25-hydroxyvitamin D in the MMTV-PyMT mouse model of breast cancer. Cancer Prev. Res. (Phila). 8, 120–128. https://doi.org/10.1158/1940-6207.CAPR-14-0110 (2015).
Strecker, T. E. et al. Effect of lapatinib on the development of Estrogen receptor-negative mammary tumors in mice. J. Natl. Cancer Inst. 101, 107–113. https://doi.org/10.1093/jnci/djn436 (2009).
Suh, N. et al. Prevention and treatment of experimental breast cancer with the combination of a new selective Estrogen receptor modulator, arzoxifene, and a new rexinoid, LG 100268. Clin. Cancer Res. 8, 3270–3275 (2002).
Unni, E., Kittrell, F. S., Singh, U. & Sinha, R. Osteopontin is a potential target gene in mouse mammary cancer chemoprevention by Se-methylselenocysteine. Breast Cancer Res. 6, R586–592. https://doi.org/10.1186/bcr914 (2004).
Visscher, D. W., Nanjia-Makker, P., Heppner, G. & Shekhar, P. V. Tamoxifen suppresses histologic progression to atypia and DCIS in MCFIOAT xenografts, a model of early human breast cancer. Breast Cancer Res. Treat. 65, 41–47. https://doi.org/10.1023/a:1006490000659 (2001).
Zhu, P. et al. Metformin selectively targets tumor-initiating cells in ErbB2-overexpressing breast cancer models. Cancer Prev. Res. (Phila). 7, 199–210. https://doi.org/10.1158/1940-6207.CAPR-13-0181 (2014).
Zhu, Z., Jiang, W., Thompson, M. D., McGinley, J. N. & Thompson, H. J. Metformin as an energy restriction mimetic agent for breast cancer prevention. J. Carcinog. 10, 17. https://doi.org/10.4103/1477-3163.83043 (2011).
Ehinger, A. et al. Histological grade provides significant prognostic information in addition to breast cancer subtypes defined according to St Gallen 2013. Acta Oncol. 56, 68–74. https://doi.org/10.1080/0284186X.2016.1237778 (2017).
Schwartz, A. M., Henson, D. E., Chen, D. & Rajamarthandan, S. Histologic grade remains a prognostic factor for breast cancer regardless of the number of positive lymph nodes and tumor size: a study of 161 708 cases of breast cancer from the SEER program. Arch. Pathol. Lab. Med. 138, 1048–1052. https://doi.org/10.5858/arpa.2013-0435-OA (2014).
Rakha, E. A. et al. Breast cancer prognostic classification in the molecular era: the role of histological grade. Breast Cancer Res. 12, 207. https://doi.org/10.1186/bcr2607 (2010).
Bhardwaj, A., Embury, M. D., Rojo, R. D., Albarracin, C. & Bedrosian, I. Efficacy of Fluvastatin and aspirin for prevention of hormonally insensitive breast cancer. Breast Cancer Res. Treat. 187, 363–374. https://doi.org/10.1007/s10549-021-06229-0 (2021).
Hitesh, B. M. The search for surrogacy in patient derived xenograft mouse trials: glass is less than half full. bioRxiv. https://doi.org/10.1101/2020.12.03.409730 (2020).
Bhardwaj, A., Embury, M. D., Ju, Z., Wang, J. & Bedrosian, I. Gene signature associated with resistance to Fluvastatin chemoprevention for breast cancer. BMC Cancer. 22, 282. https://doi.org/10.1186/s12885-022-09353-2 (2022).
Green, J. E. et al. The C3(1)/SV40 T-antigen Transgenic mouse model of mammary cancer: ductal epithelial cell targeting with multistage progression to carcinoma. Oncogene 19, 1020–1027. https://doi.org/10.1038/sj.onc.1203280 (2000).
Dawson, P. J., Wolman, S. R., Tait, L., Heppner, G. H. & Miller, F. R. MCF10AT: a model for the evolution of cancer from proliferative breast disease. Am. J. Pathol. 148, 313–319 (1996).
Acknowledgements
The study was partially funded through Institutional funding (Duncan Family Institute/DFI) provided to the PI (IB).
Author information
Authors and Affiliations
Contributions
AB, AK, RDR, and MDE performed experiments; AB, AK, JS, CTA and IB did data analysis; AK, AB and IB wrote the manuscript. All authors have read the manuscript and agree to publish in the current form.
Corresponding authors
Ethics declarations
Competing interests
Authors have no conflict of interests to disclose.
Ethical approval
All the animal experiments and other procedures were conducted as per the guidelines of as per UTMD Anderson Cancer center’s institutional animal care and use committee (IACUC) and other relevant committees such as Environment health and safety. Animals were humanely euthanized by pressure regulated CO2 inhalation followed by opening of the thoracic cavity as a secondary method to ensure death as per IACUC endorsed procedure. The study was carried out and is reported in compliance with the ARRIVE guidelines (https://arriveguidelines.org).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bhardwaj, A., Koh, A., Embury, M.D. et al. Gross tumor assessment is not a reliable measure of the efficacy of chemo-preventive agents for breast cancer in preclinical mouse models. Sci Rep 15, 39402 (2025). https://doi.org/10.1038/s41598-025-19423-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-19423-7