Abstract
Sustainable biomaterials are essential for advancing tissue engineering. This study investigates the in vivo biocompatibility and regenerative potential of seaweed cellulose (SC) scaffolds derived from Ulva sp. and Cladophora sp. as connective support matrices. SC scaffolds were fabricated using an optimized decellularization process that preserved their distinct porous (Ulva) and fibrous (Cladophora) architectures. Subcutaneous implantation in Sprague–Dawley rats demonstrated minimal foreign body response and successful integration over an eight-week period. Histological analysis revealed architecture-driven healing dynamics: Ulva sp. scaffolds promoted compartmentalized healing, characterized by distributed vascularized connective tissue, while Cladophora sp. scaffolds supported stratified tissue organization with aligned collagen deposition. Both scaffolds exhibited progressive vascularization and reduced foreign body response, with no adverse inflammatory reactions observed. These findings highlight the potential of SC scaffolds for regenerative applications that require tailored tissue responses, while their renewable, marine- origin underscores their potential as sustainable biomaterials in advanced healthcare solutions.
Similar content being viewed by others
Introduction
The development of biomaterials for tissue engineering faces ongoing challenges, particularly in creating scaffolds that closely mimic native extracellular matrix (ECM) to support cellular growth and tissue regeneration. The ECM is a dynamic and complex structure that provides both mechanical and biochemical cues essential for cell adhesion, proliferation, and differentiation. Replicating these properties in synthetic or natural biomaterial scaffolds has been a central focus in the field, as such scaffolds are critical for advancing regenerative medicine and related applications1,2,3,4.
Among the materials investigated for tissue engineering, cellulose-based scaffolds have emerged as promising candidates due to their biocompatibility, structural integrity, and mechanical robustness5. Cellulose, the most abundant biopolymer in nature, is composed of glucose monomers organized into a highly crystalline structure, imparting it with tensile strength and stability over extended periods6,7. These properties make cellulose suitable for applications requiring long-term structural support, such as wound healing and tissue regeneration8,9,10.
Marine green macroalgae, commonly known as seaweed, provide an alternative, not explored and sustainable source of cellulose for biomaterials11,12,13. Unlike terrestrial plant cellulose, seaweed biomass is lignin-free, making the extraction of seaweed cellulose (SC) less energy and chemicals-intensive and more cost-effective14. Additionally, macroalgae grow rapidly in aquatic environments, requiring no arable land, freshwater, or fertilizers15,16. These characteristics make SC a renewable and environmentally friendly material forbiomedical applications17,18,19,20. Moreover, the structural diversity of SC, ranging from porous (Ulva sp.) to fibrous (Cladophora sp.) architectures, offers a unique opportunity to tailor scaffolds for specific tissue engineering applications21. The structural differences between Ulva sp. and Cladophora sp. cellulose scaffolds are particularly relevant to their function as connective support scaffolds. Ulva sp. features a comb-like porous structure that facilitates nutrient diffusion and multidirectional cell growth, while Cladophora sp. exhibits a dense fibrous matrix that supports unidirectional cell alignment and proliferation21. These distinct morphologies are hypothesized by us to influence not only in vitro cell behavior but also in vivo tissue responses, including vascularization and healing dynamics.
To be clinically viable, biomaterials must exhibit biocompatibility in vivo, with minimal foreign body reaction (FBR) and effective tissue integration22,23,24. FBR is an immune-mediated response characterized by protein adsorption, acute and chronic inflammation, and eventual fibrosis25,26. Materials that reduce FBR while promoting tissue regeneration and angiogenesis are ideal candidates for regenerative medicine27,28,29,30. SC scaffolds, by virtue of their natural composition and structural properties, hold significant potential to modulate FBR and enhance tissue integration in vivo.
Following our previous in vitro biocompatibility study21, which confirmed that both seaweed cellulose (SC) scaffolds are non-toxic, the present study focuses on evaluating the in vivo biocompatibility of Ulva sp. and Cladophora sp. cellulose scaffolds. Using a rat subcutaneous implantation model31,32, we examine scaffold-mediated effects on tissue integration, immune response, and angiogenesis over an eight-week period. This investigation emphasizes how scaffold architecture shapes the spatial and temporal dynamics of healing, aiming to clarify the role of porous versus fibrous structures in guiding tissue responses.
By integrating renewable marine-derived materials into tissue engineering, this research highlights the dual benefits of SC scaffolds: clinical efficacy and environmental sustainability. The use of macroalgae-derived cellulose aligns with global efforts to reduce the ecological footprint of biomedical materials while addressing the demand for scalable and cost-effective solutions. Future directions include enhancing the degradability of SC scaffolds and developing hybrid biomaterials to expand their applicability in regenerative medicine.
Results
Structural characterization of seaweed cellulose scaffolds
In our previous study21, we developed cellulose scaffolds from green macroalgae species Ulva sp. and Cladophora sp. as potential connective support scaffolds alternatives. Decellularization effectively removed cellular content while preserving structural integrity, as confirmed by SEM, DNA analysis, histology imaging (Fig. 1A-G), and fluorescence imaging, verified cellulose as the primary component. Scaffolds structural analysis revealed distinct morphologies (Fig. 1B,E): Ulva sp. displayed a porous structure with 20.2 ± 4 µm pore size, while Cladophora sp. exhibited a fibrous structure with fibers ranging from 5 µm to over 80 µm, overlaid with microfibrils measuring 55–400 nm (Fig. 1H).
Seaweed derived from green marine macroalgae species (A-C) Ulva sp. and (D-F) Cladophora sp. Imaging of (A, D) fresh seaweed thallus before decellularization. Macro view of decellularized SC scaffolds reveal (C) Ulva sp. with highly organized porous architecture and (D) Cladophora sp. with highly fibrous architecture. (E, F). Hematoxylin and eosin (H&E) staining cross-sections of seaweed after decellularization reveal eosin stain of the matrix and no hematoxylin (cell nucleus). (G) DNA quantification analysis using a NanoDrop spectrophotometer, reveal DNA concentration of fresh (f) and decellularized (d), Ulva sp. (U) and Cladophora sp. (C) seaweed samples, confirming both scaffolds as acellular post decellularization. Additionally, closeup SEM imaging of (H) decellularized porous Ulva sp. and fibrous Cladophora sp. and (I) fibroblast, 4 weeks post recellularization, reveal Cell–Matrix interactions, and the impact of SC scaffold structural properties on cell morphologies and spreading. Ulva sp. facilitate stretched morphologies and 2D spreading opportunities in all direction, while the Cladophora sp. facilitate spindle morphologies and 1D spreading opportunities along the fiber elongated axis. (J) Representative plots of fibroblast cell size post recellularization. (K) summarized information Table for each SC scaffold and cells features. Correlation between the SC matrices and the fibroblast behavior, with significant difference between the two scaffolds, demonstrating fibroblast cell size larger than the Ulva sp. pore size, while smaller than the Cladophora sp. fiber width impact cell behavioral tendencies. Scale Bar: *Scale Bars: (A) = 5 cm, (B) = 0.5 cm, (C, D) = 50 µm, (E, F) = 100 um, (H) = 5 µm, (I) = 10, 20 µm, (Bar-Shai, et al. 2021).
Cell growth analysis demonstrated high viability for up to 40 days in culture, highlighting the distinct impact of each scaffold’s structure on cell behavior and proliferation rates. The porous Ulva sp. scaffold supported rapid, multidirectional fibroblast proliferation, reaching saturation by week 3, whereas Cladophora sp. fibrous scaffold promoted elongated cell growth along fiber axes with consistent, linear proliferation over time (Fig. 1I). In vitro Biocompatibility assessments using alamarBlue assays confirmed scaffold non-toxicity, demonstrating their suitability for long-term cell culture.
These findings emphasize the direct correlation between scaffold structural properties and cell behavior, survival, and proliferation, underscoring the potential of macroalgae-derived cellulose scaffolds for biomedical applications such as tissue engineering and wound healing.
Implantation of seaweed cellulose scaffolds and tissue collection
Following decellularization21, sterilized seaweed cellulose implants (Ø = 8 mm) from porous Ulva sp. (Fig. 2A) and filamentous Cladophora sp. (Fig. 2B) were evaluated for in vivo biocompatibility as alternative implants in a wound-healing model using male Sprague–Dawley rats (250 g, n = 24, 8 weeks old). Animals were divided into test groups (with implants) and controls (without implants) over an 8-week period. Subcutaneous implantations of Ulva sp. and Cladophora sp. SC scaffolds were performed in two upper dorsal incisions (1 cm each) (Fig. 2D,E), with biocompatibility assessed at weeks 1, 4, and 8 post implantation, for tissue with and without the subcutaneous implants (Fig. 2F). Dissected tissue sites (1–2 cm2) (Fig. 2H,I) were collected at each time point for histopathological analysis, revealing minimal inflammation, structural stability, and intact shapes with minimal alteration throughout the study.
Scaffolds implantation, resection and collection: seaweed cellulose (SC) scaffold derived from (A) porous Ulva sp. and (B) fibrous Cladophora sp., cut into (C) uniformed disks (diameter: Ø = 8 mm) and used as implants in (D) male Sprague–Dawley rat model (250 g, n = 20, 8 weeks old). The surgical procedure consisted of two symmetrical and parallel full-thickness skin incision (1 cm each), on the right and left sides, spaced 2.5 cm apart, along the upper dorsal area of each rat. (E) Each scaffold was implanted into a surgically created subcutaneous pouch on the right (R) and left (L) sides. (F) Scheme of experiment timeline, describe collection timepoint of implants and surgical tissue sites at week 1, 4 and 8 post implantations, including three animals (n = 3) for each time point, for animal groups with implants and control groups without implants. Representative photographs of animal’s dorsal surgical sites (n = 2, left and right): (G) prior to dissection, (H) after implant resection (1–2 cm2 ), and (I) excised tissue collection with the subcutaneous Ulva sp. (R) and Cladophora sp. (L) implants.
Both Ulva sp. and Cladophora sp. implants retained consistent dimensions and their original shape from the initial implantation (diameter: Ø = 8 mm; thickness: 2 ± 0.06 mm and 1.7 ± 0.05 mm, respectively) through week 8 of the experiment (Fig. 7D,E). Throughout the experiment, including surgical procedures and handling, the animals exhibited no signs of discomfort or behavioral changes, maintained consistent weight gain, and tolerated the implants well. Although three animals died early from causes unrelated to the test items, as confirmed by the study pathologist, their exclusion, along with the removal of one additional animal, left 20 rats for statistical analysis, establishing the 8-week experiment timeline.
In vivo biocompatibility and foreign body response
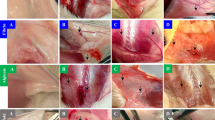
Excision sites with and without SC implants were processed for histopathological evaluations. Assessments were based on adversity criteria, as well as semiquantitative scoring conducted by a board-certified pathologist, for Ulva sp. and Cladophora sp. scaffolds. Global view imaging (Figs. 3, 4, top row) and representative view imaging of the implants’ center (Figs. 3, 4, bottom row), highlight their performance as biomaterial implants. Evaluations include inflammation reactions (Hematoxylin & Eosin staining) Fig. 3, collagen depositions (Masson’s Trichome staining), Fig. 4, and vascularization developments (anti-CD31 antibody staining) Fig. 5, concluded with cell and tissue semiquantitative scoring Fig. 6.
Biocompatibility and cell infiltration: Hematoxylin and eosin (H&E) staining of tissue cross-sections with porous (A) Ulva sp. and fibrous (B) Cladophora sp. Seaweed Cellulose implants and sections of (C) control group with no implants, for the evaluation of biocompatibility, cell infiltration and foreign body reaction (FBR), including cell type and cell response, at week 1, 4 and 8. Top row reveal FBR global view, and bottom row reveal representative view of implant center ROI. *Scale Bars: Top row = 1 mm, Bottom row = 100 μm.
Connective tissue deposition: tissue cross sections of seaweed cellulose (SC) porous (A) Ulva sp. implant, fibrous (B) Cladophora sp. implant and sections of (C) control group with no implant, stained with Masson’s Trichrome, for the evaluation of extracellular matrix deposition, including fibroplasia, fibrosis, and neovascularization at week 1, 4 and 8. Top row reveal global view with fibrosis capsule of both SC implants, and bottom row reveal representative view of implant center ROI, with fibrosis and angiogenesis. Collagen is shown in blue, cellular cytoplasm in pink/red, and blood vessels in yellow/orange. (a) *Scale Bars: Top row = 1 mm, Bottom row = 100 μm.
Vascularization and angiogenesis: tissue cross sections of seaweed cellulose (SC) A. Ulva sp. porous implant, B. Cladophora sp. fibrous implant and sections of C. control group with no implant, stained with Anti-CD31/PECAM-1 immunohistochemistry staining, for the evaluation of vascularization and angiogenesis, at week 8. The top row reveals global view, the middle (20x) and bottom (40x) rows reveal representative view of center ROI. Imaging of both SC implant sites (A, B) show the presence of endothelial cells, confirming angiogenesis process with multiple blood vessels within the implant sites. *Scale Bars: Top row = 1 mm, Middle row = 100 μm and Bottom row = 50 μm.
Representative plots of Semi-quantitative scoring of histopathology parameters for SC implants Ulva sp. and Cladophora sp. and control group of animal tissue without implant, at week 1, 4 and 8 (t = 1, 4, 8), post implantation. (A) the overall FBR reaction, including MC (yellow) and FBGC (orange), (B) inflammatory reaction PMNN (purple) and necrosis (green), (C) overall granulation tissue formation, including Fibrosis / Fibroplasia (blue), and Neovascularization (grey). The plots present scoring for the Ulva sp. implant (dashedline), Cladophora sp. implant (solid line) and control tissues without implants (dotted line), at weeks 1, 4 and 8. Scoring for cell and tissue response and the total scoring were calculated according to criteria of ISO 10993–6:2016, Table E.3 by a board-certified pathologist. The individual categories were ranked: no change (0), minimal (1), mild (2), moderate (3) or severe (4); the columns represent the mean values, n = 3.
Additionally, morphological characteristics and the distinct impact of the SC scaffolds structural characteristic on the healing process were studied Fig. 7. Significant differences were observed in the healing processes for both Ulva sp. and Cladophora sp. SC implants. The histological responses of the two SC implants are shown in Figs. 3, 4, 5 and 6, indicating an ongoing healing process during this study. Notably, some SC implant samples exhibited a bent shape due to handling during the formalin fixation, causing deformation before histological staining.
Implantation summary: vascularization and angiogenesis representative images of tissue cross sections (A) without implants and of Seaweed Cellulose (SC). (B) Ulva sp. porous implant and (C) Cladophora sp. fibrous implant, stained with Hematoxylin–Eosin (H&E), Masson’s Trichrome (MT), and Anti-CD31/PECAM-1 (CD31), close-up view (X40), for the evaluation of vascularization and angiogenesis, representative view of implants’ center ROI, after 8 weeks of implantation. Imaging of both SC implant sites (B,C) show the presence of blood cells (H&E, red; MT, yellow) and endothelial cells (CD31, brown; MT, dark pink), confirming angiogenesis process with multiple blood vessels developments within both implant sites. Additionally, (D) scaffolds before implantation (t = 0) and (E) implants with tissue resections at week 8 post implantation (t = 8), reveal scaffolds structural stability obtaining original diameter (Ø = 8 mm) with no deformation or degradability post implantation, as well as animal positive response. The implanted SC scaffolds are pink with no discoloration and show no visual sign of inflammation or edemas. The implants were soft and appears to be well integrated with the host’s tissues, confirming a healthy tissue. *Scale Bars: (HE, MT) = 100 μm, (CD31) = 50 μm.
Inflammation response
Hematoxylin and eosin (H&E) staining (Fig. 3) revealed distinct patterns of inflammation and cell infiltration at the implantation sites of porous Ulva sp. and fibrous Cladophora sp. scaffolds, compared to control tissues without implants. Both scaffolds elicited inflammatory reactions at week 1, which diminished or resolved entirely by week 8, indicative of an active healing process. However, their responses differed significantly. Ulva sp. implant (Fig. 3A) showed a milder initial inflammatory response at week 1, characterized by polymorphonuclear-hemorrhage interspersed throughout the expanded necrotic implant site. The reduction of polymorphonuclear cells (PMNNs), notably neutrophils, was gradual over the study period, with smaller necrotic cavities persisting among the implant fragments until week 8, suggesting a slower inflammation resolution. In contrast, Cladophora sp. implant (Fig. 3B) exhibited a more pronounced initial inflammatory reaction at week 1, characterized by PMNN clusters at the implant’ expanded necrotic center. However, by week 4, the necrotic center diminished, PMNNs had markedly reduced, and none were observed by week 8, indicating faster inflammation resolution and a more accelerated healing process. Control tissues without implants (Fig. 3C) displayed minimal inflammatory reactions at week 1, followed by complete healing in subsequent weeks, highlighting the differences in healing dynamics between the two implants and compared to tissues without implants.
Foreign body reaction (FBR)
Additionally, foreign body reactions (FBR), notably macrophages (MCs) and foreign body giant cells (FBGCs), were observed at both implant sites at week 1, albeit with distinct differences. The Ulva sp. implant exhibited a gradual increase in FBGC presence by week 4, predominantly scattered throughout necrotic cavities surrounding implant fragments, followed by steady reduction over the 8-week study period. Conversely, the Cladophora sp. implant elicited an earlier resolution of FBR, with transformation of MCs into FBGCs predominantly at the implant periphery. By week 4, FBR at the implant’ center significantly diminished, leaving moderate FBGCs activity persisting at the periphery, which further decreased by week 8. This suggests faster integration and recovery process in the Cladophora sp. implant compared with the more prolonged and scattered FBR response in the Ulva sp. implant. Control tissues without implants showed minimal FBR at week 1 and 4, and none by week 8, confirming an earlier and complete healing response in the absence of implants. These observations align with Masson’s Trichrome staining (Fig. 4), which demonstrated the gradual replacement of inflammation and FBR with connective tissue in both scaffolds.
Fibroplasia/fibrosis response
Masson’s Trichrome staining (Fig. 4) revealed collagen deposition and fibrosis within and around the implant sites, supporting new tissue formation. Both Ulva sp. and Cladophora sp. scaffolds exhibited fibrotic reactions starting at week 4, gradually replacing inflammation and FBR, with scaffold-specific fibrosis-fibroplasia patterns. Ulva sp. implant (Fig. 4A) demonstrated non-homogeneous fibrosis scattered across the implant site, steadily replacing necrotic-FBR areas with mature connective tissue septa among implant fragments. Collagen infiltration and replacement was slower but gradual and more pronounced. However, some necrotic-FBR collections persisted through week 8, indicating ongoing healing. In contrast, Cladophora sp. implant (Fig. 4B) elicited denser and more homogeneous fibrosis, replacing the necrotic center by week 4 and progressively expanding towards the periphery by week 8, with collagen deposition engulfing individual fibers, suggesting more advanced tissue integration. Fibroplasia capsules were observed surrounding both SC implants throughout the 8-week study, reflecting FBR responses and healing progression. Control tissues without implants (Fig. 4C) displayed early fibrosis by week 1 and fully mature connective tissue regeneration by weeks 4 and 8, highlighting faster and more complete healing in the absence of implants. These observations underscore the structural influence of the two SC scaffolds on fibrosis development and healing dynamics.
Angiogenesis and tissue integration
Anti-CD31/PECAM-1 immunohistochemistry (Fig. 5) revealed endothelial cells (stained brown) within Ulva sp. and Cladophora sp. implant sites at week 8, confirming vascularization and angiogenesis. This was supported by Masson’s Trichrome staining imaging (Fig. 4), which showed neovascularization alongside fibroplasia and fibrosis, progressively replacing inflammation and FBR through the study. At week 1, early-stage neovascularization was observed primarily at the periphery of both implant sites. By week 4, blood vessels began forming in the fibrosis implants’ center, with distinctdifferences between scaffolds. Ulva sp. implants exhibited displayed a continuous decrease in vascularization at the periphery while extending into the connective tissue septa among necrotic-FBR collections, reflecting slower and ongoing healing. In contrast, Cladophora sp. implants showed neovascularization at both the FBR periphery and fibrosis center by week 4, with vascularization shifting predominantly to the fibrosis center by week 8, forming between the implant’ fibers, indicating advanced healing and tissue integration. Control tissues without implants demonstrated early vascularization throughout the scar tissue at week 1, which decreased by week 4 as connective tissue matured. By week 8, vascularization was absent, indicating correlation with the inflammation decrees and the complete healing process in the absence of implants. Overall, Cladophora sp. implants demonstrated significantly greater neovascularization than Ulva sp. at week 8, highlighting their superior capacity to support angiogenesis and integration during healing.
Semi-quantitative histopathological scoring
The histopathological semi-quantitative scoring was conducted according to ISO 10993–6:2016 standards, following the reference criteria outlined in Tables E.1 - E.3 of the guideline33, to assess inflammation, foreign body reaction (FBR), fibroplasia/fibrosis, and neovascularization at Ulva sp. and Cladophora sp. implantation sites (Table 1, Fig. 6). Tissue cross-sections were evaluated for cell types and tissue responses, using a five-point grading scale (0–4) based on histological changes observed via Hematoxylin & Eosin (H&E), Masson’s Trichrome, and anti-CD31 staining over the 8-week study period. Scoring was performed by a board-certified pathologist (Table 1), with representative plots illustrating trends for MCs (yellow) and FBGCs (orange) (Fig. 6B), PMNNs (purple) and necrosis (green) (Fig. 6C), and collagen deposition (blue) and blood cells/vessels (gray) (Fig. 6D). The Ulva sp. implant was represented by a dashed line, Cladophora sp. by a solid line, and control tissues without implants by a dotted line.
Inflammation response
Both scaffolds triggered inflammatory responses at week 1, composed primarily of polymorphonuclear neutrophils (PMNN). The Ulva sp. implants exhibited a mild (2) inflammatory response that steadily declined to minimal (1) by week 4 and resolved entirely (0) by week 8. In contrast, Cladophora sp. scaffolds elicited a more pronounced moderate (3) inflammation at week 1, with a steeper resolution, reducing to none (0) by week 4. Control tissues displayed minimal (1) inflammation at week 1, resolving completely by week 4.
Foreign body reaction (FBR)
Ulva sp. scaffolds exhibited upward trend of MCs and FBGC’s by week 4, followed by a downward trend by week 8. FBR-associated MCs increased from minimal (1) FBR at week 1 to mild (2) FBR by week 4, then decreased to minimal (1) FBR by week 8. This transition coincided with FBGC formation, with minimal (1) FBR/FBGCs present at weeks 4 and 8. Whereas Cladophora sp. scaffolds showed a decreasing trend in MCs and an increasing trend in FBGCs. FBR was moderate (3) at week 1, with MCs concentrated at the implant core, decreasing to mild (2) FBR/MCs at weeks 4 and 8, confined to the periphery. FBGC formation progressed outward, increasing from minimal (1) FBR/FBGCs at week 1 to mild (2) at week 4, and reaching moderate (3) FBR/FBGCs by week 8, only at the periphery. Control tissues demonstrated minimal (1) FBR at week 1 and 4, with no FBR (0) by week 8.
Fibroplasia/fibrosis
Collagen deposition and connective tissue formation varied between scaffolds. The Ulva sp. scaffolds showed a gradual increase in fibrosis, starting as mild (2) at weeks 1 and 4, progressing to moderate (3) by week 8. Fibrosis was scattered among implant fragments, indicating steady but slower tissue remodeling. Cladophora sp. scaffolds exhibited earlier and more homogenous fibrosis, scoring mild (2) at week 1 and moderate (3) by week 4, with complete replacement of necrotic centers and denser fibrosis by week 8. Control tissues exhibited advanced fibrosis early, scoring moderate (3) at week 1 and severe (4) by weeks 4 and 8.
Neovascularization
Neovascularization followed different trends for each scaffold. The Ulva sp. implants demonstrated an initial increase in vascularization at weeks 1 and 4 (mild, 2), followed by a decline to minimal (1) by week 8, reflecting slower angiogenesis. In contrast, Cladophora sp. scaffolds maintained steady vascularization, scoring mild (2) throughout the study, with enhanced vessel formation in fibrotic regions. Control tissues exhibited declining vascularization, scoring mild (2) at week 1 and minimal (1) by week 4, with no vascularization (0) by week 8.
Foreign debris
FBR associated with the presence of foreign debris remnants, including hair shafts and cotton fibers from surgical residues, was observed at both SC implant sites and surrounding tissues. as well as at the animal tissue without implant. Ulva sp. scaffolds showed mild (2) response at weeks 1 and 4, decreased to minimal (1) by week 8. Whereas Cladophora sp. scaffolds exhibited moderate (3) foreign debris-associated FBR at week 1, decreasing to mild (2) and week 4 following an increase at week 8. Control tissue without implant showed minor (1) FBR linked to surgical debris, which resolved by week 8. Local scant FBR was assessed based on MCs and FBGCs presence surrounding residual material, which did not undergo degradation, scattered at the implant sites.
Total scoring and healing trends
Overall, both scaffolds demonstrated progressive healing, with inflammation and FBR transitioning to fibrosis and vascularization by week 8. Cladophora sp. scaffolds exhibited faster integration and more advanced tissue remodeling, while Ulva sp. scaffolds supported gradual and steady healing. Control tissues without implants showed earlier and complete healing by week 8, with minimal tissue reactions. Both scaffolds were biocompatible, non-toxic, and effective in promoting tissue regeneration, highlighting their potential as connective support scaffolds alternatives.
Discussion
Our study evaluates the in vivo biocompatibility and healing dynamics of two seaweed-derived cellulose (SC) scaffolds, specifically Ulva sp. (porous) and Cladophora sp. (fibrous), in a rat subcutaneous implantation model over an 8-week period. The selection of Ulva sp. and Cladophora sp. scaffolds was guided by their distinctive structural architectures, sustainable sourcing, and promising stability, all of which are critical for long-term tissue integration. The assessment, conducted by a board-certified toxicological pathologist, complemented our prior in vitro findings21, provided critical insights into scaffold integration, modulation of immune response, and long-term physiological function, as well as the impact of scaffold morphology on host immune response, tissue integration, spatial and temporal progression of foreign body response (FBR), fibrosis and angiogenesis. The assessment, conducted by a board-certified toxicological pathologist, complemented our prior in vitro findings21, provided critical insights into scaffold integration, modulation of immune response, and long-term physiological function, as well as the impact of scaffold morphology on host immune response, tissue integration, spatial and temporal progression of foreign body response (FBR), fibrosis and angiogenesis.
Subcutaneous implantation proved effective for monitoring scaffold integration, host tissue interactions, and ease of retrieval while minimizing animal discomfort. No animals exhibited implant rejection symptoms such as swelling or discharge, suggesting successful scaffold integration. However, three of the initial 24 animals succumbed to surgical complications unrelated to the implant material, as confirmed by pathological analysis. To maintain a balanced cohort, one additional subject was excluded, yielding a final study population of 20 animals with adjusted tissue collection at 1-, 4-, and 8-weeks post-implantation.
Differences healing dynamics and spatial FBR distribution between the two SC scaffold may result fromvariations in biomaterial source, biodegradability, structure, permeability, swelling behavior, and overall scaffold morphology. These factors collectively impact the host response and influence biocompatibility, tissue regeneration, and implant success. Understanding these interactions is essential for optimizing scaffold design for specific clinical applications2,24,28,29,30.
In vivo biocompatibility assessment
Biocompatibility was evaluated through histopathological analyses, including hematoxylin and eosin (H&E), Masson’s Trichrome, and anti-CD31 staining, coupled with semi-quantitative scoring, aligned with ISO 10993–633 and Schafer et al. 201834. Biocompatibility depends greatly on the foreign body response (FBR), a critical factor for evaluating scaffold performance. Effective FBR resolution promotes tissue remodeling and integration27, whereas prolonged or an unbalanced response may cause persistent inflammation, fibrotic encapsulation, or chronic rejection25,35. Therefore, assessing FBR mechanisms is critical for optimizing biomaterial performance in tissue engineering.
Our in vivo biocompatibility tests enabled a comprehensive evaluation of both implants’ behavior within a living organism over the course of the study. Both SC scaffolds exhibited promising biocompatibility throughout the 8-week study, showing mild to moderate differences in FBR between the two implants (Fig. 7), which progressively subsiding over time, consistent with biomaterial-associated FBR22,28,29, Cladophora sp. scaffolds demonstrated more rapid resolution of polymorphonuclear neutrophils (PMNNs), macrophages (MCs) and a necrotic core (~ 60% of the implant site), which resolved into fibrotic tissue by week 4, indicating an active tissue remodeling at its core, accompanied by FBR-periphery (~ 40%) associated with foreign body giant cells (FBGCs) that decreased to ~ 20% by week 8. Whereas Ulva sp. scaffolds showed a delayed transition characterized by lingering interspersed FBR-inflammation response and necrosis clusters among implant fragments, which gradually decreased and transitioned into more progressive septal fibrosis structures and neovascularization, supporting tissue integration across the implant, yet dispersed inflammatory and persistent FBGCs were still observed by week 8. indicating ongoing healing process.
Consistent with previous studies on natural36 and synthetic32 biomaterials, persistent FBGCs and fibrotic encapsulation in non-degradable cellulose scaffolds suggest delayed wound resolution29. Nevertheless, despite the prolonged FBR, both SC scaffolds supported pro-fibrotic and pro-angiogenic responses that progressively transitioned into organized connective tissue, with no adverse outcomes such as rejection or infection, thereby confirming their biocompatibility.
Biodegradability impact on foreign body response
Biodegradability plays a key role in shaping the severity and persistence of the FBR25,29,30 to implanted biomaterials (Table 2). Biodegradable scaffolds, whether natural (e.g., collagen, elastin and fibrin29) or synthetic (e.g., PLGA37, PDO38, and PLA39,), typically elicit a transient inflammatory response, followed by scaffold resorption and tissue replacement, that closely mimic native ECM27,39. These materials often promote biocompatibility and integration30,40, although in synthetic materials, degradation rates must be carefully controlled to avoid harmful byproducts and leachables27,39. whereas in natural materials mechanical, economical, ethical and ecological present concerns. Emerging plant-based biomaterials, including soy41, alginates28, and algae-derived biomaterials17, show promise as biodegradable alternatives29, inducing mild FBR and supporting tissue integration. However, structural modifications are often required to better mimic the complexity of ECM and optimize their performance for biomedical applications41.
In contrast, non-degradable biomaterials, such as plant cellulose10,36,48, bacterial nanocellulose (BNC)44,45, composites31,49,50, and synthetic polymers like silicone32,51, tend to provoke prolonged FBR, including persistent macrophages (MCs) and FBGCs, and fibrotic encapsulation25,28,32,39. These could induce frustrated phagocytosis of indigestible materials25,39, potentially delaying full tissue integration26,27. However, such materials generally avoid acute inflammation and offer stable, long-term biocompatibility29,30,52,53. See Table 2 for comparative overview of SC scaffolds with other reported biodegradable and non- biodegradable biomaterials in subcutaneous implant models.
Our Ulva sp. and Cladophora sp. SC scaffolds, maintained their structural integrity throughout the 8-week study (Fig. 7D,E), demonstrating high stability, similar to other cellulose-based implants5,52. This supported sustained cellular infiltration, tissue ingrowth and neovascularization. While the SC scaffolds exhibited low inflammation, they did induce prolonged FBR marked by persistent FBGCs and encapsulation, consistent with other non-degradable cellulose-based materials26,27,29. Nonetheless, they did not trigger harmful immune responses or generate toxic degradation products24,39.
The SC scaffolds used in this study were processed using our previously described decellularization method21, which preserve their native architecture but involves organic solvents, acid/base solutions, and heat. While effective, this approach presents scalability and sustainability trade-offs. To address these, several studies have proposed cost-effective and ecologically viable methods for cultivating and processing marine algae14,15,16, supporting seaweed cellulose as a renewable, low-impact material source.
Although we did not conduct formal shelf-life testing, preliminary observations in our lab indicate that dry-stored or water-immersed samples remained structurally stable for 12, 18, and even 48 months. Future studies should validate these findings under clinically relevant storage conditions. Nevertheless, the absence of degradable byproducts, coupled with the scaffold’s durability, highlights its promise for applications requiring long-term structural support, such as wound healing, bone regeneration, or drug delivery29.
Scaffolds structure and healing dynamics
Ulva sp. (porous) and Cladophora sp. (fibrous) SC scaffolds provided a comparative model for examining host immune responses. Our study revealed clear differences in spatiotemporal FBR distribution and healing dynamics, driven by scaffold topology, composition, density, and form, which collectively shaped tissue response and regeneration (Fig. 8, Table 3).
Tissue-matrix interactions, summary—implantation-sites scheme: Histological H&E imaging and schematic representation associated with each healing phase ROI morphology and coverage during the 8-week study, at both (A) porous Ulva sp. and (B) fibrous Cladophora sp. SC implant sites, in comparison to (C) animal tissue without implant, for cell and tissue response at the implantation’s sites. In vivo biocompatibility evaluations including global view (top row), as well as center and periphery ROI (20x), of each implant site, at week 1, 4 and 8. The imaging global view and the overlay schematic illustration display the distinct trend of the healing process as influenced by the SC scaffolds structural properties during the 8-weeks healing recovery (necrosis, yellow; neutrophils, purple; FBR MCs and FBGCs, orange; collagen, light blue; blood cells and vessels, gray). *Scale Bars: (× 1.5) = 1 mm, (× 20) = 100 μm, 250 μm.
Healing dynamics in porous and fibrous implant
Scaffold composition, including pore size and fiber diameter, strongly influences FBR dynamics by regulating oxygen/nutrient diffusion, protein adsorption, cell infiltration, collagen deposition, angiogenesis, and fibrotic capsule formation28.
Porous scaffolds Pore size regulates infiltration, tissue ingrowth, and vascularization. Optimal micropores (10–40 µm) enhance cell attachment54, promote capillary formation (≈34 µm)25,28,29,54,55, limit fibrosis and reduce FBR.56 Whereas macropores (50–300 µm) support larger vessels25,28,29,55 and collagen deposition (≈160 µm)57, reduce inflammation28,58 and promote long-term integration, though they often form thicker fibrotic encapsulation57. Very large pores (≈4000 µm) elicited more favorable immune responses than smaller pores (≈460 µm), underscoring pore-size-dependent reactions56. In contrast, nonporous/dense implants hinder infiltration and angiogenesis, increase inflammatory adherence on the implant surface leading to dense encapsulation37,57. The Ulva sp. scaffold in our study displayed intermediate interconnected pores (Fig. 1H, K) and cavities among implant fragments (Fig. 8B), creating a micro- and macro-pore structure that mimicked macroporous implants57, This intermixed composition supported infiltration, vascularization and remodeling despite persistent MCs and FBGCs. Although healing was incomplete at week 8, cavity reduction and tissue ingrowth confirmed favorable biocompatibility and a pro-healing response.
Fibrous scaffolds Fiber diameter, alignment, and inter-fiber spacing influence inflammation25,28,29, cell attachment and morphology59, fibrosis, vascularization and integration38,47,56. Nanofibers (~ 1–100 nm) mimic natural ECM, enhancing attachment and angiogenesis while reducing fibrosis47,59. Microfibers (1–100 µm) provide structural support but induce moderate inflammation and fibrosis, while macrofibers (> 100 µm) offer mechanical durability for high-load applications, they often limit cell interaction, sustain inflammation and form denser encapsulation, limiting vascularization and slowing integration. Thus, nanofibers, with higher surface-to-volume ratio, enhance attachment and angiogenesis, while microfibers provide superior structural support. Orientation and surface topography also modulates healing responses: nanoscale grooves (400–500 nm) promote protein adsorption and fibroblast ingrowth, accelerating FBR resolution, compared to smooth fibers59,60,61, whereas aligned fibers guide anisotropic growth (useful for muscle/nerve), and random fibers promote isotropic infiltration, advantageous for wound healing28. The Cladophora sp. scaffold in our study exhibited diverse fiber diameters (Fig. 1H, K) and dense inter-fiber spacing (Fig. 8C), resembling microfiber structures, showing induced inflammatory-FBR followed by remodeling at its core by week 4, and persistent FBGC’s at the periphery. Compared to Ulva sp., the tightly packed, dense fibrous structure led to a stratified healing pattern with earlier core resolution but slower peripheral integration. The overlay nanoscale microfibrils (Fig. 1H) likely enhanced contact guidance and surface area-to-volume ratio, promoting protein adsorption, collagen deposition and advanced FBR resolution. Additionally, random fiber orientation, mimicking natural ECM, likely enhanced homogeneous cellular interactions and modulated FBR distribution.
Spatiotemporal FBR distribution driven by porous and fibrous implants
Scaffold architecture dictated distinct spatiotemporal FBR patterns and healing dynamics. While both scaffolds supported progressive healing, Ulva sp. scaffold facilitated dispersed, compartmentalized healing dynamics, characterized by inflammatory clusters and tissue growth septa distributed among implant fragments, while Cladophora sp. scaffold exhibited concentric stratification FBR pattern, guiding layered center-to-periphery healing (Fig. 8). These architectural differences modulated inflammatory distribution, matrix remodeling, and integration outcomes.
Across timepoints, both scaffolds exhibited early expansion due to FBR-associated inflammation with PMNN infiltration and hemorrhagic necrosis (Fig. 8, yellow). Cladophora sp. developed a large necrotic core (~ 60%), surrounded by a MC-rich periphery (~ 40%), whereas Ulva sp. displayed widespread necrosis interspersed with inflammatory clusters. By week 4, necrosis resolved in Cladophora sp., replaced by fibrotic connective tissue; FBR-inflammatory (Fig. 8, orange) became peripheral and decreased to ~ 20% of implant site by week 8, dominated by FBGCs. Conversely, Ulva sp. necrotic cavities evolved into compartmentalized inflammatory clusters, distributed among scaffold fragments, that persisted with FBGCs at week 8. Thus, Cladophora sp. facilitated earlier necrosis FBR- inflammation resolution and tissue remodeling whereas, Ulva sp. exhibited slower inflammatory resolution. In humans, comparable necrotic responses may take longer to resolve, potentially prolonging inflammation or fibrosis, underscoring the need for porosity optimization or surface functionalization to improve integration. As implants’ inter-spacing contracted, FBR-inflammation shifted toward connective tissue formation (Fig. 8, blue). In Cladophora sp., fibrosis emerged at the implant core by week 4, expanding outward to ~ 80% by week 8, with neovascularization infiltrating the fibrotic center (Fig. 8, gray). Ulva sp. exhibited connective tissue septa among scaffold fragments and necrotic cavities, increasing from ~ 20% at week 4 to ~ 40% by week 8 (Fig. 8, blue). However, persistent FBR clusters indicated delayed resolution. Neovascularization followed scaffold-dependent patterns: Cladophora sp. promoted branching angiogenesis within its dense fibrous matrix, while Ulva sp.’s spacious cavities facilitated luminal-like vascular formation (Fig. 7B-C).
Healing dynamics driven by porosity and density
Scaffold porosity and density directly impact inflammatory persistence, oxygen/nutrient influx, waste removal, and overall FBR distribution47,56. Porous structures promote cell infiltration, connective tissue development, and angiogenesis, whereas dense, non-porous scaffolds can hinder infiltration and diffusion, limit tissue integration38,56,57,62,63. Optimal porosity is critical, as excessively large or small pores impair integration and may lead to scaffold failure56. Porous matrices are generally more biocompatible compared to dense, non-textured surfaces or much looser structures, which sometimes delay complete resolution. Swellability further alters pore size and shape over time, affecting cell-scaffold interactions47,56. For example, dense drug-releasing implants erode into randomly shaped pores (≈2 μm) post-drug release, increasing internal pore volume56.
In our study, both SC scaffolds preserved structural integrity over 8-weeks (Fig. 7D-E). The inter-fiber spacing within the Cladophora sp. and the Ulva sp. macro-cavity spacing, can be described as scaffold porosity. Ulva sp. cavities and Cladophora sp. inter-fiber voids gradually contracted, driving progressive FBR resolution and connective tissue replacement (Figs. 3A,B, 4A,B, 8B,C). These dynamics underscore their potential in drug-release applications where porosity evolution modulates tissue interactions56.
Limitation and translational considerations
While SC scaffolds showed promising healing outcomes, several limitations may impact clinical translation. Long-term degradation, mechanical stability, and host responses require further investigation to fully understand scaffold performance over extended periods.
Cladophora sp. scaffolds facilitated earlier necrosis resolution, whereas the porous Ulva sp. promoted sustained regeneration. However, healing in rats is inherently faster than in humans, where necrosis may resolve more slowly, risking prolonged inflammation or fibrosis, which could impact scaffold integration. To enhance biocompatibility and tissue remodeling, further studies should evaluate long-term performance and stability to host response. Scaffold optimization may include adjusting form, porosity, interconnectivity, or hybrid designs combining both fibrous and porous structures to mitigate FBR and better align with human healing dynamics.
The fibrous Cladophora sp. scaffold may be advantageous for applications requiring rapid stabilization, while the porous Ulva sp. could be beneficial for sustained regeneration. Looking ahead, the principles and methods developed in this study could be extended to other bio-based materials, where refined architecture may enhance healing efficiency and minimize inflammatory responses, and hybrid design strategies, could balance structural stability withintegration to open new opportunities for optimizing scaffold performance across diverse biomedical applications.
Conclusion
This study evaluated seaweed cellulose (SC) scaffolds from Ulva sp. and Cladophora sp. as sustainable biomaterials for tissue engineering using an eight-week rat subcutaneous implantation model. Both porous (Ulva sp.) and fibrous (Cladophora sp.) scaffolds demonstrated excellent biocompatibility, minimal foreign body reaction, and successful tissue integration. The scaffolds structural diversity shaped distinct healing dynamics: Ulva sp. facilitated compartmentalized tissue ingrowth and vascularization, while Cladophora sp. promoted stratified tissue organization with collagen alignment and angiogenesis.
The renewable origin and ecological advantages of SC scaffolds, coupled with their capacity to modulate scaffold-dependent tissue responses, positions them as versatile platforms with dual benefit in addressing both clinical and sustainability goals. These scaffolds are promising for applications requiring durable, non-degradable, non-leachable, non-conductive materials, either as standalone implants or as reinforcement in composites.
Future research should aim to enhance long-term strategies, tailor mechanical and biological properties, and explore hybrid designs to expand their versatility. Overall, SC scaffolds bridge innovative biomaterials with sustainable practices, offering a strong platform for next-generation healthcare solutions and beyond.
Experimental section
Animal model
All surgical procedure and animal handling were performed at Tel Aviv University Animal Facility. The Study was approved by the Tel Aviv University Animal Care Committee (approval number TAU-R-1012196) and were conducted according to the highest ethical standards and in compliance with the ARRIVE guidelines64, the Institutional Animal Care and Use Committee (IACUC), and the Operational Guidelines for Ethics Committees for Biomedical Research.
A total of 24 male Sprague–Dawley rats65,66 (approximately 250 g, 8 weeks old) were obtained from Envigo laboratories (Israel). The animals were housed in groups of three per cage under controlled temperature and humidity conditions, with a 12-h light/dark cycle and ad libitum access to food and water. All measures were taken to minimize the number of animals used and to prevent pain and discomfort during the experiments.
The study design included two experimental groups. A test group implanted with Ulva sp. and Cladophora sp. scaffolds (on the left and right dorsal sites, respectively, see Fig. 2), and a control group without implants. Each group included three animals per time point (1, 4, and 8 weeks). However, three animals died early due to causes unrelated to the test items, as confirmed by the study pathologist, and one additional animal was excluded due to unrelated technical issues. Consequently, 20 animals were included in the final statistical analysis.
Seaweed selection and preparation
Green marine macroalgae species Ulva sp. and Cladophora sp. specimens were obtained in this work as a model for their structural composition variation: a porous and a fibrous matrix structure, respectively. The two species, which are found in the intertidal and shallow waters of the Israeli Mediterranean seashores, are known for their fast growth rates16, and are considered as potential feedstock for biorefineries15,67. Seaweeds were cultivated under controlled conditions using cylindrical, sleeve-like macroalgae photo-bioreactors (MPBR, Polytiv, Israel), with sleeve dimensions of 100 cm length, 200 μm thickness, 40 cm width, and total circulation volume of 3400 l seawater (salinity 3.9%, pH 8.2)15. All samples were cleaned and sorted manually to achieve clean monocultures and documented for their morphology and histology evaluations.
Seaweed decellularization: cellulose extraction and purification
A whole organ and tissue decellularization approach68 was used to isolate and extract the support matrix of seaweed tissue from its inhabiting cells, leaving a “ghost” ECM-like scaffold of the original tissue. Following an efficient decellularization treatment69,70 and its optimization for a whole tissue culture, cellular content was extracted from the two macroalgae species Ulva sp. and Cladophora sp. Fresh Ulva sp. and Cladophora sp. seaweed biomass (100 g wet weight ) were obtained, cleaned and sorted. Next, the samples were boiled in acetone bath (20% w/v) at 60 °C for 60 min, repeatedly 4 times, in order to remove pigments (chlorophyll) and proteins. Residual biomass was boiled in acetate buffer bath, containing 1.17 g Sodium Chlorite (NaClO2) (20% w/v), at 60 °C for 6–8 h, spurring bleaching and remove polysaccharides of simpler structure than cellulose. The bleached seaweed residues were then pH neutralized by washing with distilled water. Next, the samples were alkylated in 0.5 M Sodium Hydroxide (NaOH) bath (20% w/v), at 60 °C for 8–10 h, to remove all excessive lipids and hemicellulose within the cell wall. Following the alkali treatment, the seaweed residues were once again pH neutralized by washing with distilled water and then acidified in a hydrochloric acid (HCI) (5% v/v), at 100 °C for 10 min (20% w/v), or until boiling started. The samples were left in the acid treatment to rest overnight at room temperature to remove all excessive polysaccharides, such as starch, that might remain close to the cell wall. Finally, the samples were carefully rinsed repeatedly in DW, until reaching a neutral pH (SevenExcellence pH Meter).
Seaweed cellulose scaffold fabrication
Obtaining a clear clean biomass, the decellularized seaweed were then filtered and dried at 40 °C in an oven for 24 to 48 h, or at room temperature (RT °C), on a flat surface for a period of 4 to 7 days, achieving a final whole algal support matrix, making them a ready-to-be-used acellular seaweed cellulose (SC) scaffolds. Using a digital caliper (Holex), Ulva sp. and Cladophora sp. scaffolds were measured for their thickness, 0.1 mm and 0.15 mm, respectively, and for their area dimensions for each of the study evaluation tests. Decellularized samples with area dimensions that range between 1 and 2 mm2 were used for observation imaging analysis. Scaffolds for in vitro biocompatibility analysis were fabricated with specific dimension area for the viability direct test (uniformed Ø = 2 mm diameter disks, ) and cytotoxicity indirect test (6 cm2 per 1 ml). Scaffolds for in vivo biocompatibility analysis were fabricated with specific dimension area for implantation with animal model (uniformed Ø = 8 mm diameter disks), as described below.
Cellulose determination
Fluorescence staining solution (1:1) consisting of Calcofluor White reagent (Ref. 18909; Sigma-Aldrich), which binds to cellulose in the plant cell wall, and 10% potassium hydroxide (KOH) (Ref. P5958; Sigma-Aldrich) was used to determine the presence of cellulose in the seaweed-scaffolds post-decellularization treatment. The Calcofluor White fluorescent dye solution was deposited directly onto the seaweed decellularized samples, which were placed onto glass slides. Fluorescence Microscopy was used to observe the samples. The Evans blue present in the stain, emits fluorescence at a wavelength of 395–415 nm and permits a rapid visualization of cellulose presence in the decellularized seaweed cell wall.
Seaweed histological analysis
The determination of the seaweed decellularization treatment, achieving acellular cellulose scaffolds, was carried out using histological analysis of samples from fresh seaweed and decellularized scaffold samples. The samples were embedded in paraffin and sectioned into 4 μm thick slices perpendicular to the surface. The sections were mounted on glass slides (4 sections per slides), stained with hematoxylin and eosin (H&E) reagent (Patholab, IL) and visualized under an optical microscope (Nikon Eclipse TS2, Japan). All image processing was performed with ImageJ software (ImageJ v. 1.51, NIH).
DNA contamination quantification
The evaluation of acellular scaffold, emptied from its cellular organelles post decellularization, were further determined using plant genomic DNAconcentration and purification analysis (Thermo Scientific GeneJET # K0791). The concentration was measured with a NanoDrop spectrophotometer (ND-2000, Thermo Scientific), used for a quick and simple wavelength absorbance analysis. Fresh and decellularized Ulva sp. and Cladophora sp. samples were examined (n = 3 for each sample). Wavelength absorbance of all samples (1 μl solvent) were compared with blank sample and purified DNA sample with a nucleic acid to protein (A260/280) indicator and ratio between 1.7 and 1.9. Furthermore, gel electrophoresis (Invitrogen, E-Gel, 1.2%) was used to determine the concentration results for both fresh and decellularized samples. Purified DNA samples (20 μl solvent) of fresh seaweed and decellularized scaffolds were analyzed and documented (ENDURO GDS, Labnet; Omega Fluor, software).
Scanning electron microscopy analysis
Decellularized SC scaffolds were evaluated and analyzed using scanning electron microscopy (SEM) (JCM-6000, JEOL, Life Sciences, Tel Aviv University). Samples were visualized and recorded at × 50, 130, 400, 650, 1000, 1700, 4000 and × 7000 magnification to analyze the decellularization treatment and evaluate the isolation of the algal inhabiting cells from its extracellular matrix cell-wall. Further, structural characterization of the two-seaweed species was carried to study the varied cellulose-based matrices morphology and surface area. Pore size, cell wall width, and fiber diameter were observed and determined using image analysis software ImageJ (ImageJ v. 1.51, NIH). To evaluate the Ulva sp. pore size, 50 regions of interest (ROI) were identified in a given SEM image of the decellularized sample, and 10 ROI were identified to determine the Ulva sp. cell wall thickness. Further, 55 ROI were identified to determine Cladophora sp. fiber width, and 50 ROI were identified to determine Cladophora sp. overlay microfibrils width. The mean dimensions and standard deviation were reported.
Sterilization
Decellularized cellulose from Ulva sp. and Cladophora sp. species were sterilized and pretreated prior to the in vitro biocompatibility tests. Single seaweed cellulose samples were placed in individual wells and soaked in 70% Ethanol (1 ml/well) overnight at RT°C, in a tissue culture flow hood. Samples were then washed in ultrapure water (UPW) (UltraPure DNsese/RNase-Free, Biolab-Chemicals), three times, then soaked overnight (2 ml/well) at RT°C, in a tissue culture flow hood. Next, the samples were treated in PBS (Dulbecco’s Phosphate Buffered Saline ( −) Calcium ( −), Magnesium, Biological Industries) (1 ml/well) and incubated overnight (37 °C, %5CO2). Finally, the samples were treated in GM consisting of DMEM-HG (1 ml/well), and incubated overnight (37 °C, %5CO2). Successively, the media was discarded, and the samples were dried in a tissue culture flow hood, before recellularization seeding took place.
Implants preparation
In vivo biocompatibility evaluation took place following implantation of Ulva sp. and Cladophora sp. cellulose scaffolds. The SC’s biomaterial were cut into uniformed circle disks, using a hole puncher device (diameter: Ø = 8 mm; weight: 0.0042 ± 0.0008 g and 0.006 ± 0.0007 g; thickness: 2 ± 0.06 mm and 1.7 ± 0.05 mm, respectively, see (Fig. 2A–C)). The scaffolds were then sterilized using 70% ethanol, as previously described, following additional sterilization with a steam autoclave (121 ˚C, 30 min), using sterilization pouches (YIPAK) ready to be used as scaffolds, suitable for in vivo implantation.
Surgical procedure: seaweed cellulose (SC) scaffolds implantation
All animals, including control groups without implants, and experimental test groups with SC scaffold implantation, underwent a standardized subcutaneous dorsal surgical procedure to evaluate the in vivo biocompatibility of the seaweed cellulose (SC) scaffolds. Experimental animals received SC scaffold implantation with Ulva sp. on the right side, and Cladophora sp. on the left side (Fig. 2D,E). Control animals underwent the same surgical procedure without scaffold implantation.
Before the implantation procedure, all animals were co-administered with Ketamine/Xylazine (Clorketam/Sedaxylan, Phibro) anesthesia combination and confirmed complete anesthesia by eliciting no response to a tail/paw pinch-induced nociception stimulation. An ophthalmic ointment was applied to prevent corneal irritation and drying. The dorsal area of each rat was shaved using an electric clipper. The exposed shaved skin area was disinfected with povidone iodine (VITAMED). To maintain sedation during surgery, isoflurane anesthetic inhalation (USP 100%; Terrell, Piramal) was administrated, and physiological parameters including, hydration, body temperature, and respiration, were monitored throughout the procedure. Following, two symmetrical and parallel, full-thickness 1 cm skin incisions were made using a sterile scalpel, on the upper dorsal area of each rat (left and right sides). Subcutaneous pouches were made with blunt dissection, and one SC scaffold of each type, Ulva sp. on the right (R) and Cladophora sp. on the left (L) were implanted in the respective pouches. Incisions were then sutured using 4/0 monofilament nylon polyamide (Atlas Medical) and disinfected. All animal received postoperative analgesia (Rheumocam solution) and were monitored during recovery.
Explantation procedure: scaffold and tissue resections
Scaffold and tissue explantation was performed at 1-, 4-, and 8-weeks post-implantations. Rats were euthanized using carbon dioxide (CO2) overdose. Skin Sects. (1–2 cm2) from the dorsal surgical sites (left and right) were carefully dissected and collected (Fig. 2G-I). , including both scaffold and surrounding tissue (in test groups) or tissue only (in controls). Samples were documented and immediately fixed in 4% paraformaldehyde (PFA, Biological Chemicals) for histological analysis.
Histological analysis
Collected tissue samples, with and without scaffold implants, were embedded in paraffin blocks, sectioned at 5 μm thickness, and mounted on glass slides (approximately 2–3 tissue sections from each block). All samples were cut through the center of the wound along a line perpendicular to the head-to-tail axis of the animal. Samples with implants were cut perpendicular to the scaffold. Tissue sections were processed and stained with Hematoxylin–Eosin (H&E) and Masson’s trichrome (MT) staining by Patho-Lab Diagnostics Ltd. (Rehovot, Israel) for the evaluation of cell infiltration and extracellular matrix deposition. Color images of each entire tissue section were acquired using a NanoZoomer digital pathology slide scanner with a 40 × objective and NDP.view2 image viewing software (Hamamatsu, Hamamatsu City, Japan). Additional tissue sections from week 8 were processed and stained by Patho-Logica (Rehovot, Israel) with anti-CD31/PECAM-1 immunohistochemistry staining for the evaluation of vascularization (angiogenesis). Color images of each tissue section were acquired using a KF-FL-400 digital pathology slide scanner with a 40 × objective and K-Viewer software (KFBIO, China).
Histopathological and biocompatibility qualitative assessment
Slides were evaluated by a board-certified toxicological pathologist, according to the international ISO-10993 standard 6 [ISO 10993–6:2016, Table E.1, E.2,33 used for the biological evaluation of medical devices in animal testing and clinical trials, Table E.1 for cell type/ response and Table E.2 for Tissue response. Samples were examined in a “blinded” manner, i.e., without prior knowledge of the treatment groups. Histopathological evaluation was based on a grading system given to tissue tolerance parameters (e.g., inflammation, necrosis, foreign body response (FBR), fibrosis and neovascularization), to document the lesions severity which reflected the extent of each reaction, its distribution, and the actual degree of severity, according to the Harmonization of nomenclature and diagnostic criteria in toxicologic pathology (IN HAND) standards.71 Histopathological changes were described and scored by the study pathologist using semi-quantitative grading system of a five-point grading scale (0–4), taking into consideration the severity of the changes (0 = No Change, 1 = Minimal Change, 2 = Mild Change, 3 = Moderate Change, 4 = Severe Change), based on the criteria published by Schafer et al. 201834. The scoring reflected the predominant degree of the specific lesion seen in the entire field of the histology section. The grading system was presented to each lesion parameter and to the overall total scoring, according to ISO 10993–6:2016, Table E.3, and specified to study test samples.
Histopathologic severity grades is a semiquantitativeassessment method which is used to describe and compare between the study groups. The grading scale is often not linear and based on variable criteria concluded by the study pathologist. Thus, statistical analysis is not recommended in this method34,71.
Criteria for adversity
The study pathologist included in the assessment an appropriate judgment and conclusive statement concerning potential adversity or not adversity for each type of treatment-related lesion. The criteria for adversity are based on the position papers published by the Society of Toxicologic Pathology (STP) and the European ESTP72,73,74,75. This statement is referring to the animal species used in the experimental conditions specific to this study and will help the study director determine the No Observed Adverse Effect (NOAEL) level (and Pass/Fail, in case of need). Parameters that may be taken into consideration for the determination of adversity, including the presence of ulceration, necrosis, mineralization, and thrombosis, and the potential recovery of this phase, are included in the study design76. In particular, the severity grade and extension of such a potential adverse lesion will be considered34. In the event that such a listed lesion is focal and of minor grade (up to grade 2 of 4), the lesion will potentially be considered not adverse. However, if such a lesion is extensive and of a higher grade than 2, it may be considered adverse. In any case, the determination of adversity should always be analyzed and considered case-by-case, and the rationale for the suggested adversity should be justified with appropriate references.
Statistical analysis
In vivo experiments were all standardized; animals of the same age were used. At least three wounds from different animals for each treatment option were used. At least nine wounds (n = 2 sites) were used as untreated, no implants, control groups. All experiments were carried out with at least three replicates. Values are presented as the mean ± standard deviation (SD), paired with a two-sample T-test coupled with Fischer’s Combined Probability test. A value of p < 0.05 was considered statistically significant.
Data availability
All data generated or analyzed during this study are available from the corresponding author (Nurit Bar-Shai) on reasonable request.
References
Lanza, R., Langer, R., Vacanti, J. & Atala, A. Principles of Tissue Engineering. (Academic Press, 2020).
Wagner, W. R., Sakiyama-Elbert, S. E., Zhang, G. & Yaszemski, M. J. Biomaterials Science: An Introduction to Materials in Medicine. (Elsevier, 2020).
O’Brien, F. J. Biomaterials & scaffolds for tissue engineering. Mater. Today 14, 88–95 (2011).
Langer, R. Perspectives and challenges in tissue engineering and regenerative medicine. Adv. Mater. 21, 3235–3236 (2009).
Hickey, R. J. & Pelling, A. E. Cellulose biomaterials for tissue engineering. Front. Bioeng. Biotechnol. 7, (2019).
Klemm, D., Heublein, B., Fink, H.-P. & Bohn, A. Cellulose: Fascinating biopolymer and sustainable raw material. Angew. Chem. Int. Ed. 44, 3358–3393 (2005).
Gibson, L. J. The hierarchical structure and mechanics of plant materials. J. R. Soc. Interface 9, 2749–2766 (2012).
Bilirgen, A. C. et al. Plant-based scaffolds in tissue engineering. ACS Biomater. Sci. Eng. 7, 926–938 (2021).
Fu, L., Zhang, J. & Yang, G. Present status and applications of bacterial cellulose-based materials for skin tissue repair. Carbohydr. Polym. 92, 1432–1442 (2013).
Shang, L., Wang, S. & Mao, Y. Recent advances in plant-derived polysaccharide scaffolds in tissue engineering: A review. Int. J. Biol. Macromol. 277, 133830(2024).
Wang, Z. et al. Current application and modification strategy of marine polysaccharides in tissue regeneration: A review. Biomater. Adv. 154, 213580 (2023).
Mihranyan, A. Cellulose from cladophorales green algae: From environmental problem to high-tech composite materials. J. Appl. Polym. Sci. 119, 2449–2460 (2011).
Lahaye, M. & Robic, A. Structure and functional properties of ulvan, a polysaccharide from green seaweeds. Biomacromol 8, 1765–1774 (2007).
Baghel, R. S., Reddy, C. R. K. & Singh, R. P. Seaweed-based cellulose: applications and future perspectives. Carbohydr. Polym. 118241 (2021).
Chemodanov, A., Robin, A. & Golberg, A. Design of marine macroalgae photobioreactor integrated into building to support seagriculture for biorefinery and bioeconomy. Bioresour. Technol. 241, 1084–1093 (2017).
Fernand, F. et al. Offshore macroalgae biomass for bioenergy production: Environmental aspects, technological achievements and challenges. Renew. Sustain. Energy Rev. 75, 35–45 (2017).
Iravani, S. & Jamalipour Soufi, G. Algae-derived materials for tissue engineering and regenerative medicine applications: current trends and future perspectives. Emergent Mater. 5, 631–652 (2022).
Kumar, S., Marrero-Berrios, I., Kabat, M. & Berthiaume, F. Recent advances in the use of algal polysaccharides for skin wound healing. Curr. Pharm. Des. 25, 1236–1248 (2019).
Zhou, S., Nyholm, L., Strømme, M. & Wang, Z. Cladophora cellulose: Unique biopolymer nanofibrils for emerging energy, environmental, and life science applications. Acc. Chem. Res. 52, 2232–2243 (2019).
Madub, K. et al. Green seaweeds ulvan-cellulose scaffolds enhance in vitro cell growth and in vivo angiogenesis for skin tissue engineering. Carbohydr. Polym. 251, (2021).
Bar-Shai, N., Sharabani-Yosef, O., Zollmann, M., Lesman, A. & Golberg, A. Seaweed cellulose scaffolds derived from green macroalgae for tissue engineering. Sci. Rep. 11, 11843 (2021).
Ratner, B. D. The biocompatibility of implant materials. In Host Response to Biomaterials: The Impact of Host Response on Biomaterial Selection 1–25 (Elsevier, 2015).
Marin, E., Boschetto, F. & Pezzotti, G. Biomaterials and biocompatibility: An historical overview. J. Biomed. Mater. Res. A 108, 1617–1633 (2020).
Ratner, B. D. & Schoen, F. J. Characterization of biomaterials. In Biomaterials Science (eds. Wagner, W. R., Sakiyama-Elbert, S. E., Zhang, G. & Yaszemski, M. J.) 843–849 (Elsevier, 2020).
Anderson, J. M., Rodriguez, A. & Chang, D. T. Foreign body reaction to biomaterials. Semin. Immunol. 20, 86–100 (2008).
Noskovicova, N., Hinz, B. & Pakshir, P. Implant fibrosis and the underappreciated role of myofibroblasts in the foreign body reaction. Cells 10, (2021).
Ibrahim, R., Nyska, A. & Ramot, Y. Biocompatibility of polymers. In Biomaterials and Biopolymers (eds. Domb, A., Mizrahi, B. & Farah, S.) 235–253 (Springer, 2023).
Zhang, D. et al. Dealing with the foreign-body response to implanted biomaterials: Strategies and applications of new materials. Adv. Funct. Mater. 31, 1–22 (2021).
Litowczenko, J. et al. Milestones and current achievements in development of multifunctional bioscaffolds for medical application. Bioact. Mater. 6, 2412–2438 (2021).
Jurak, M., Wiącek, A. E., Ładniak, A., Przykaza, K. & Szafran, K. What affects the biocompatibility of polymers? Adv. Colloid Interface Sci. 294, (2021).
Peltokallio, N. M. M. et al. Comparative in vivo biocompatibility of cellulose-derived and synthetic meshes in subcutaneous transplantation models. Biomacromolecules (2024).
Yamakawa, T. et al. Modified foreign body reaction to silicone imbedded in subcutaneous tissues by different mouse systemic immune conditions. J. Biomed. Mater. Res. A 110, 1921–1931 (2022).
International Organization for Standardization (ISO). ISO 10993–6: Biological evaluation of medical devices — Part 6: Tests for local effects after implantation. (2016).
Schafer, K. A. et al. Use of severity grades to characterize histopathologic changes. Toxicol. Pathol. 46, 256–265 (2018).
Babensee, J. E., Anderson, J. M., McIntire, L. V. & Mikos, A. G. Host response to tissue engineered devices. Adv. Drug Deliv. Rev. 33, 111–139 (1998).
Modulevsky, D. J., Cuerrier, C. M. & Pelling, A. E. Biocompatibility of subcutaneously implanted plant-derived cellulose biomaterials. PLoS ONE 11, (2016).
Ward, W. K. A review of the foreign-body response to subcutaneously-implanted devices: The role of macrophages and cytokines in biofouling and fibrosis. J. Diabetes Sci. Technol. 2, 768–777 (2008).
Garg, K., Pullen, N. A., Oskeritzian, C. A., Ryan, J. J. & Bowlin, G. L. Macrophage functional polarization (M1/M2) in response to varying fiber and pore dimensions of electrospun scaffolds. Biomaterials 34, 4439–4451 (2013).
Ramot, Y., Haim-Zada, M., Domb, A. J. & Nyska, A. Biocompatibility and safety of PLA and its copolymers. Adv. Drug Deliv. Rev. 107, 153–162 (2016).
Terzopoulou, Z. et al. Biocompatible synthetic polymers for tissue engineering purposes. Biomacromol 23, 1841–1863 (2022).
Las Heras, K. et al. Soy protein and chitin sponge-like scaffolds: From natural by-products to cell delivery systems for biomedical applications. Green Chem. 22, 3445–3460 (2020).
Rahmanian-Schwarz, A. et al. In vivo biocompatibility and biodegradation of a novel thin and mechanically stable collagen scaffold. J. Biomed. Mater. Res. A 102, 1173–1179 (2014).
Chien, K. B., Aguado, B. A., Bryce, P. J. & Shah, R. N. In vivo acute and humoral response to three-dimensional porous soy protein scaffolds. Acta Biomater. 9, 8983–8990 (2013).
Girard, V.-D. et al. In vitro and in vivo biocompatibility of bacterial cellulose. J. Biomed. Mater. Res. B Appl. Biomater. 112, e35488 (2024).
Helenius, G. et al. In vivo biocompatibility of bacterial cellulose. J. Biomed. Mater. Res. A 76, 431–438 (2006).
Maiborodin, I. V. et al. Reaction of rat tissues to implantation of lactic acid-based biodegradable polymer. Bull. Exp. Biol. Med. 156, 874–879 (2014).
Wang, K. et al. Overcoming foreign-body reaction through nanotopography: Biocompatibility and immunoisolation properties of a nanofibrous membrane. Biomaterials 102, 249–256 (2016).
Contessi Negrini, N., Toffoletto, N., Farè, S. & Altomare, L. Plant tissues as 3D natural scaffolds for adipose, bone and tendon tissue regeneration. Front. Bioeng. Biotechnol. 8, (2020).
Mahendiran, B. et al. In vitro and in vivo biocompatibility of decellularized cellulose scaffolds functionalized with chitosan and platelet-rich plasma for tissue engineering applications. Int. J. Biol. Macromol. 217, 522–535 (2022).
Janmohammadi, M. et al. Cellulose-based composite scaffolds for bone tissue engineering and localized drug delivery. Bioact. Mater. 20, 137–163 (2023).
Zare, M., Ghomi, E. R., Venkatraman, P. D. & Ramakrishna, S. Silicone-based biomaterials for biomedical applications: Antimicrobial strategies and 3D printing technologies. J. Appl. Polym. Sci. 138, 50969 (2021).
Wei, C. et al. Biomaterials in skin tissue engineering. Int. J. Polym. Mater. Polym. Biomater. 71, 993–1011 (2022).
Gopinath, V. et al. Multifunctional applications of natural polysaccharide starch and cellulose: An update on recent advances. Biomed. Pharmacother. 146, 112492 (2022).
Ortiz, J. et al. Dietary fiber, amino acid, fatty acid and tocopherol contents of the edible seaweeds Ulva lactuca and Durvillaea antarctica. Food Chem. 99, 98–104 (2006).
Sadtler, K. et al. Design, clinical translation and immunological response of biomaterials in regenerative medicine. Nat. Rev. Mater. 1, (2016).
Hernandez, J. L. & Woodrow, K. A. Medical applications of porous biomaterials: Features of porosity and tissue-specific implications for biocompatibility. Adv. Healthc. Mater. 11, 1–25 (2022).
Sussman, E. M., Halpin, M. C., Muster, J., Moon, R. T. & Ratner, B. D. Porous implants modulate healing and induce shifts in local macrophage polarization in the foreign body reaction. Ann. Biomed. Eng. 42, 1508–1516 (2014).
Almeida, C. R. et al. Impact of 3D-printed PLA- and chitosan-based scaffolds on human monocyte/macrophage responses: Unraveling the effect of 3D structures on inflammation. Acta Biomater. 10, 613–622 (2014).
Luu, T. U., Gott, S. C., Woo, B. W. K., Rao, M. P. & Liu, W. F. Micro- and nanopatterned topographical cues for regulating macrophage cell shape and phenotype. ACS Appl. Mater. Interfaces 7, 28665–28672 (2015).
Rosengren, A., Bjursten, L. M., Danielsen, N., Persson, H. & Kober, M. Tissue reactions to polyethylene implants with different surface topography. J. Mater. Sci. Mater. Med. 10, 75–82 (1999).
Yadav, T. C. & Bachhuka, A. Tuning foreign body response with tailor-engineered nanoscale surface modifications: Fundamentals to clinical applications. J. Mater. Chem. B 11, 7834–7854 (2023).
Sanders, J. E., Stiles, C. E. & Hayes, C. L. Tissue response to single-polymer fibers of varying diameters: Evaluation of fibrous encapsulation and macrophage density. J. Biomed. Mater. Res. 52, 231–237 (2000).
Ward, W. K., Slobodzian, E. P., Tiekotter, K. L. & Wood, M. D. The effect of microgeometry, implant thickness and polyurethane chemistry on the foreign body response to subcutaneous implants. Biomaterials 23, 4185–4192 (2002).
Percie du Sert, N. et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biol. 18, e3000410 (2020).
Yang, G. et al. Assessment of the characteristics and biocompatibility of gelatin sponge scaffolds prepared by various crosslinking methods. Sci. Rep. 8, 1616 (2018).
Golberg, A. Emerging electroporation-based technologies for wound care. In Innovations and Emerging Technologies in Wound Care 155–170 (Academic Press, 2020).
Polikovsky, M. et al. Towards marine biorefineries: Selective proteins extractions from marine macroalgae Ulva with pulsed electric fields. Innov. Food Sci. Emerg. Technol. 37, 194–200 (2016).
Crapo, P. M., Gilbert, T. W. & Badylak, S. F. An overview of tissue and whole organ decellularization processes. Biomaterials 32, 3233–3243 (2011).
Trivedi, N. et al. An integrated process for the extraction of fuel and chemicals from marine macroalgal biomass. Sci. Rep. 6, 1–8 (2016).
Prabhu, M. S., Israel, A., Palatnik, R. R., Zilberman, D. & Golberg, A. Integrated biorefinery process for sustainable fractionation of Ulva ohnoi (Chlorophyta): Process optimization and revenue analysis. Bioresour. Technol. 32, 2271–2282 (2020).
Mann, P. C. et al. International harmonization of toxicologic pathology nomenclature: An overview and review of basic principles. Toxicol. Pathol. 40, 7S-13S (2012).
Kerlin, R. et al. Scientific and regulatory policy committee: Recommended (‘best’) practices for determining, communicating, and using adverse effect data from nonclinical studies. Toxicol. Pathol. 44, 147–162 (2016).
Palazzi, X. et al. Characterizing ‘adversity’ of pathology findings in nonclinical toxicity studies: Results from the 4th ESTP international expert workshop. Toxicol. Pathol. 44, 810–824 (2016).
Wancket, L. M., Bolon, B., Funk, K. A. & Schuh, J. C. L. Toxicologic pathology forum*: Opinion on assessing and communicating adversity for implantable medical devices. Toxicol. Pathol. (2024).
O’Brien, M. T. et al. Scientific and regulatory policy committee points to consider for medical device implant site evaluation in nonclinical studies. Toxicol. Pathol. 50, 512–530 (2022).
Baldrick, P. et al. Toxicology paradise: Sorting out adverse and non-adverse findings in animal toxicity studies. Int. J. Toxicol. 39, 365–378 (2020).
Acknowledgements
The authors thank Planning and Budgeting Committee of the Council for Higher Education of Israel, FUTUREWISE project. The authors also wish to thank Ayelet Lesman, School of Mechanical Engineering, Tel Aviv University, and Libi Hackam, Alpha Program, Tel Aviv University.
Author information
Authors and Affiliations
Contributions
Nurit Bar-Shai– study design, experiments, data analysis, wrote the manuscript Din Mann- performed the experiments, Avshalom Shalom- study design, reviewed the manuscript Abraham Nyska- did pathology analysis, reviewed the manuscript Alexander Golberg- study design, edited the manuscript, supervised the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bar-Shai, N., Mann, D., Shalom, A. et al. Biocompatibility and structural insights of seaweed-derived cellulose scaffolds in a subcutaneous implantation model. Sci Rep 15, 35667 (2025). https://doi.org/10.1038/s41598-025-19632-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-19632-0