Abstract
Hyperuricemia (HUA) is a widespread public health problem worldwide, the etiology of HUA is complex, and there is still a lack of accurate indicators to assess HUA when conducting mass epidemiologic screening. Recent studies have shown that insulin resistance (IR) contributes to the development of HUA, and the estimated glucose disposal rate (eGDR) has been recognized as a reliable surrogate for IR. The aim of this study was to investigate the association between eGDR and incident HUA. A total of 9091 participants without HUA at baseline were enrolled in the China Health and Retirement Longitudinal Study. Multivariate logistic regression models were used to analyze the association between eGDR and incident HUA. Restricted cubic spline (RCS) regression modeling was used to examine the dose–response relationship between eGDR and new-onset HUA, and ROC curve analysis was used to test the predictive power of eGDR. Among 9091 participants, 306 new cases of HUA were documented during a median follow-up of 79.4 months. RCS curves showed a significant linear relationship between eGDR and HUA (nonlinear P > 0.05). After full adjustment for covariates (model 3), higher eGDR values were significantly associated with a lower risk of HUA (P < 0.05). The ORs (95% CI) for eGDR Q2-4 were 0.83 (95% CI 0.57–1.19), 0.65 (95% CI 0.45–0.92), and 0.62 (95% CI 0.43–0.89), respectively, for a significantly lower risk of HUA compared with participants with eGDR Q1. When assessed as a continuous variable, each additional standard deviation of eGDR was associated with an 8% reduction in the risk of new-onset HUA (OR 0.92; 95% confidence interval 0.86–0.98; P = 0.006). Higher eGDR was found to be associated with a reduced risk of HUA. This suggests that eGDR may represent one of the promising predictive and intervention targets for HUA.
Similar content being viewed by others
Introduction
Hyperuricemia (HUA) is usually caused by increased synthesis or decreased urinary excretion of uric acid in the body and is characterized by elevated serum uric acid (SUA) levels1. UA is considered the fourth most important risk factor for cardiovascular disease after hyperlipidemia, hypertension, and hyperglycemia, and has become the second most common metabolic disease after diabetes2,3. Recent trends suggest that HUA affects approximately 20% of the world population and has become a major global health problem4,5. Therefore, the study of clinical markers for the early identification of individuals at risk for HUA is essential for public health.
Studies have shown that high serum uric acid levels are associated with the onset and progression of a number of diseases, particularly diabetes, dyslipidemia, chronic kidney disease, and cardiovascular disease3. Research indicates that the prevalence of diabetes in China is estimated at 12.4%. Among individuals with Type 2 diabetes, the prevalence of hyperuricemia reaches 45%, significantly higher than that in the general population. This condition is closely associated with blood glucose control and obesity6,7. For every 60 μmol/L (1 mg/dl) increase in serum uric acid, there is a 20% increase in the risk of developing type 2 diabetes and a 15% increase in the risk of developing type 2 diabetes in offspring8. Previous studies have shown that HUA is strongly associated with abnormal glucose metabolism and insulin resistance (IR), and that reducing IR can reduce serum uric acid levels9. In addition, a prospective study from Japan showed that elevated HOMA-IR independently predicted the subsequent development of HUA and that IR itself or compensated hyperinsulinemia may contribute to the development of HUA10. Another study from the United States found that hyperuricemia in the twenties was an independent marker for predicting diabetes and IR in young adults over the next 15 years11. Another Mendelian randomization study showed that obesity leads to hyperuricemia via IR12, and Ma et al. showed that serum uric acid is the only diagnostic marker for HUA and plays a role in obesity-related insulin resistance13.
Insulin resistance (IR) is a pathophysiological state characterized by decreased insulin sensitivity and responsiveness of insulin target organs or tissues, resulting in impaired glucose uptake and utilization, and is recognized as an important contributor to HUA14,15. Although the hyperinsulinemic-euglycemic clamp is currently the gold standard test for assessing IR, it is difficult to use in large-scale epidemiological studies because it is cumbersome, time-consuming, and costly16,17. Therefore, a simpler indicator, eGDR, has become a reliable alternative to IR. Previous studies have shown that TyG, another surrogate for insulin resistance, is associated with an increased risk of HUA in the general US population18, but to the best of our knowledge, the association between eGDR and new-onset HUA has not been fully evaluated.
To address these knowledge gaps, we recruited CHARLS participants to prospectively assess the relationship between eGDR and HUA incidence. This was done with the aim of providing more valuable insights into the early prevention and management of HUA.
Methods
Study design and population
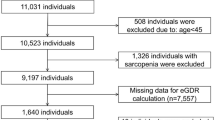
The data for this study were obtained from the China Health and Retirement Longitudinal Study (CHARLS), an ongoing longitudinal survey of China’s aging population. Initiated in 2011, the CHARLS employs a probability proportional to size methodology to conduct surveys on a nationwide sample. All survey respondents are invited to participate in follow-up surveys at two- to three-year intervals. To date, the CHARLS has conducted five surveys: in 2011, 2013, 2015, 2018, and 2020. In this study, data from the 2011 to 2020 waves of the CHARLS survey were employed for analysis. The baseline survey constituted a nationally representative sample of 17,708 individuals selected from 10,257 households between June 2011 and March 2012 for the initial cohort. Individuals with missing data on hyperuricemia (HUA), uric acid, age, education, residence, alcohol consumption, eGDR, HDL, LDL, TG, TC, and BUN were excluded from the study (N = 8617). In conclusion, a total of 9091 participants were included in the analysis and subsequently classified into four subgroups based on eGDR quartiles (Q) (Fig. 1).
Overview of participants screening.
Definitions of eGDR and eGDR’
-
eGDR(mg/kg/min) = 21.158 − (0.09 × WC) − (3.407 × hypertension) − (0.551 × HbA1c) [WC (cm), hypertension (yes = 1/no = 0), and HbA1c (%)].
-
eGDR’ = WC + HbA1c [WC (cm), HbA1c (%)].
Assessment of HUA and other covariates
In accordance with the findings of preceding studies, HUA was defined as serum uric acid (SUA) levels exceeding 7.0 mg/dL in males and 6.0 mg/dL in females19,20.
Any single SUA measurement that exceeds the threshold is recorded as an event. The event date is the date of the first follow-up blood draw. If there is no SUA data from 2013 or 2015, the most recent available value is used instead. Participants with two missing measurements are considered lost to follow-up. The continuous variable of SUA was transformed into a dichotomous variable (HUA/Non-HUA) for the purposes of analysis. The study considered a number of factors (covariates) that may potentially influence the relationship between eGDR and HUA. These covariates included a range of demographic characteristics, such as age, gender, place of residence, marital status, educational level, and smoking and drinking habits.
Statistical analysis
RStudio 4.3.2 software was employed for all statistical analyses. A two-tailed P value of less than 0.05 was considered to be statistically significant. When applicable, continuous variables were presented as mean ± standard deviation (SD) or median (interquartile range). The baseline data were compared using either an analysis of variance (ANOVA) or a Kruskal–Wallis H-test, depending on whether the data were normally distributed or skewed. Categorical variables were expressed as counts and percentages, and differences were determined by chi-square tests. To investigate the relationship between eGDR and HUA, a series of multivariate logistic regression models were constructed. Model 1 examined the relationship between eGDR and HUA without controlling for potential confounding variables. In contrast, Model 2 was adjusted for age, gender, education, residence, and marital status, as these factors were identified as potential confounders. Model 3 was further adjusted for the following variables: smoking, alcohol consumption, blood urea nitrogen (BUN), total cholesterol (TC), triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), and C-reactive protein (CRP). To investigate the dose–response relationship between eGDR and HUA incidence, restricted cubic splines (RCS) were employed in a logistic regression model, with the eGDR value at OR 1 used as a reference. Receiver operating characteristic (ROC) curves were constructed for the purpose of evaluating the predictive capacity of eGDR for HUA incidence. Moreover, subgroup analyses were performed to assess the impact of eGDR on the prevalence of HUA across diverse demographic groups, including age (< 60, ≥ 60 years), sex (male, female), educational attainment (primary school or below, middle school, high school or above), place of residence (rural, urban), marital status (married, unmarried), smoking status (yes, no), and alcohol consumption (yes, no). Subsequently, a Bayesian approach was employed to analyze the mediating effect, examining the indirect effect, direct effect, and total effect. Additionally, we excluded patients with hypertension and conducted sensitivity analyses using multivariate logistic regression models to assess the robustness of the results (adjusted covariates were consistent with the primary analysis).
Results
Participants characteristics
As shown in Baseline Table 1, this study included a total of 9091 participants with an average age of 58.0 years (45.1% male, 54.9% female). The mean age, proportion of participants who were married, proportion of participants who resided in urban areas, proportion of participants who did not drink alcohol, TC, TG, LDL, CRP, UA, HbA1c, and WC all decreased with increasing eGDR (all P < 0.001). However, individuals with higher levels of eGDR were more likely to reside in rural areas and to be married. To more accurately assess the relationship between HUA and the dependent variable, we divided the data into two groups based on baseline hypertension status. As shown in Table 2, the study included 9091 participants: 6758 without hypertension, and 2333 with hypertension. The median age was 68.0 years for all participants and 61.0 years for hypertensive individuals (42.6% male, 57.4% female). The hypertensive group exhibited significantly higher median age, lower educational attainment, and elevated levels of TC, TG, HDL, LDL, CRP, and URA compared to the non-hypertensive group (P < 0.05). Conversely, the hypertensive group demonstrated significantly lower proportions of males, married individuals, urban residents, alcohol consumers, smokers, and eGDR levels compared to the non-hypertensive group (P < 0.05).
Association between eGDR and emerging incidence of HUA
Logistic regression analysis was performed with the occurrence of HUA as the dependent variable and the eGDR index as the independent variable. Multifactorial regression analysis revealed a significant correlation between eGDR and the prevalence of HUA. When HUA was assessed as a continuous variable, the risk of incident HUA decreased by 8% per 1-SD increase in eGDR (model-adjusted). In addition, when HUA was assessed as a categorical variable, the ORs (95% CI) for eGDR Q2–4 were 0.83 (95% CI 0.57–1.19), 0.65 (95% CI 0.45–0.92), and 0.62 (95% CI 0.43–0.89), respectively, with a significant reduction in the risk of HUA compared to participants with eGDR Q1. These results suggest that eGDR is an independent and significant predictor of the incidence of HUA. The association between eGDR and HUA remained strong even after adjustment for a wide range of demographic, clinical, and biochemical variables (Table 3).
RCS curves to evaluate the risk of eGDR in patients with HUA
In this study, RCS curves were used to visualize the relationship between eGDR index and HUA risk. The results showed that after adjusting for age, sex, marital status, education, residence, smoking, alcohol consumption, blood urea nitrogen (BUN), total cholesterol (TC), triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), C-reactive protein (CRP), and serum uric acid (SUA), there was a significant linear relationship (overall P < 0. 005, nonlinear P > 0.05), with a higher eGDR index being associated with a lower risk of developing HUA (Fig. 2).
Restricted cubic spline curves for HUA according to the eGDR. (A) Unadjusted models. (B) Adjusted models. The adjusted models adjusted Age, Gender, Education, Living place, Marital status, Drinking, Smoking, BUN, TC, TG, HDL, LDL, CRP, SUA. Restricted cubic spline regression model was conducted with 4 knots at the 5th, 35th, 65th, and 95th percentiles of eGDR. The shaded area represents the 95% confidence interval for the spline model (reference is 10.44 mg kg−1 min−1).
Subgroup analyses and interaction tests
Subgroup analyses were used to assess whether the association between eGDR and HUA events varied by prespecified subgroup. Subgroup analysis is exploratory in nature. Since interaction p values have not undergone multiple comparisons correction, they should be interpreted with caution and are only meant to suggest potential directions of effect modification. After adjusting for confounders and treating eGDR as a continuous variable, a significant association between eGDR and the incidence of HUA was demonstrated in most subgroups (P < 0.05). Higher eGDR indices were associated with a lower risk of developing HUA. No significant interaction was observed for the association between eGDR and HUA incidence in these subgroups (Fig. 3).
Subgroup and interaction analyses of HUA across different subgroups.
Mediation analysis
To ensure model stability and interpretability, this study employed the variance inflation factor (VIF) method to detect and eliminate multicollinearity. First, VIF calculations were performed for all independent variables. Values exceeding 10 indicate severe multicollinearity. Results showed VIF values of 15.20 and 12.37 for the TC and LDL variables, respectively, indicating high multicollinearity between them. Based on theoretical logic and variable importance, TC was retained, while LDL was excluded. After adjustment, all variables in the model exhibited VIF values below 5, effectively resolving multicollinearity. Final results confirmed no excessive multicollinearity issues (Table 4). Mediation effects were analyzed using Bayesian methods to examine indirect, direct, and total effects. As shown in Table 5, the total effect (β_total) of eGDR on the HUA outcome was -0.09 (95% CI [− 0.15, − 0.03]), the direct effect (β_direct) was − 0.09 (95% CI [-0.15, -0.03]), and the mediation effect (β_indirect) was − 0.00 (95% CI [− 0.03, 0.00]), with a mediation proportion of 0% (1 − β_direct/β_total = 0%). Although significant associations existed between eGDR and SUA, and between SUA and HUA, the effect of eGDR on HUA outcomes was entirely mediated through a direct effect, with no significant mediating effect observed. This suggests that eGDR may directly reduce HUA risk, rather than acting through modulating SUA levels. The 0% mediation proportion indicates that eGDR’s influence on HUA outcomes is scarcely affected by the mediating role of SUA levels.
Sensitivity analysis
To further validate the robustness of the model results, we conducted a sensitivity analysis to examine the relationship between the redefined eGDR’ (excluding hypertension status) and hyperuricemia (HUA) after excluding hypertension status. The analysis revealed that eGDR’ was significantly associated with HUA in both the hypertension and non-hypertension groups (P < 0.001). Within the hypertensive cohort, the association between eGDR’ and HUA remained significant even after excluding hypertension status. In Model 1, the OR for eGDR’ associated with HUA was 0.70 (95% CI 0.64, 0.76), with a p value < 0.001. In Model 2, after adjusting for baseline factors, including age, sex, education, residence, and marital status, the OR for eGDR’ associated with HUA was 0.70 (95% CI 0.64, 0.76), P < 0.001. This further confirmed the significant association between eGDR’ and HUA, which persisted after adjusting for baseline factors. In Model 3 (fully adjusted): After further adjusting for confounding factors, including alcohol consumption, smoking, BUN, TC, TG, HDL, LDL, CRP, and SUA, the OR for eGDR’ associated with HUA was 0.71 (95% CI 0.64, 0.77), P < 0.001. This indicates that, even after full adjustment, the association between eGDR’ and HUA remains significant, with individuals having higher eGDR’ exhibiting a lower risk of HUA. The relationship between eGDR’ and HUA was also significant in the non-hypertensive group. In Model 1, the OR for eGDR’ associated with HUA was 0.69 (95% CI 0.65, 0.72), P < 0.001. This indicates a significant association between eGDR’ and HUA, even without adjusting for other confounding factors. In Model 2, which adjusted for baseline factors including age, sex, education, residence, and marital status, the OR for eGDR’ associated with HUA was 0.69 (95% CI 0.65, 0.72), P < 0.001. This further validated the significant association between eGDR’ and HUA, which persisted after adjusting for baseline factors. In Model 3 (fully adjusted), additional confounding factors, including alcohol consumption, smoking, BUN, TC, TG, HDL, LDL, CRP, and SUA, were further adjusted. The OR for eGDR’ and HUA was 0.68 (95% CI 0.65, 0.72), P < 0.001. This indicates that the association between eGDR’ and HUA remained significant even after full adjustment, with individuals having higher eGDR’ exhibiting a lower risk of HUA, as shown in Table 6.
Predictive performance in the incident HUA
The basic model (including age, sex, marital status, education, residence, smoking, drinking status, BUN, TC, TG, HDL, CRP, and eGDR). The baseline model’s area under the curve (AUC) value was 0.696 (95% confidence interval [CI]: 0.670–0.609). Its coefficient estimates, standard errors, and 95% confidence intervals are detailed in Supplementary Table S1 of the Supplementary Materials. The eGDR model’s AUC value was 0.703 (95% CI 0.745–0.553) (see Fig. 4). Coefficient estimates, standard errors, and 95% confidence intervals for the eGDR model are detailed in Supplementary Table S2. The area under the ROC curve (AUC), calibration slope, and calibration intercept for both models are detailed in Supplementary Table S3. The p value for the DeLong test was 0.078, indicating no statistically significant difference in AUC values between the two models. This suggests that incorporating baseline serum uric acid levels did not substantially enhance the model’s predictive capability for hyperuricemia outcomes, and did not obscure the incremental value of eGDR. The model is well calibrated (Fig. 5). The p values obtained from the Hosmer–Lemeshow test were 0.73 and 0.28, respectively, both greater than 0.05. This indicates no statistically significant deviation between predicted and observed values. Notably, the DCA curve indicates that the eGDR model achieves optimal clinical net benefit within the 15–75% threshold range in the test cohort (Fig. 6).
ROC curves for the prediction of HUA. (A) Basic model. (B) basic model + SUA. basic model (including age, sex, marital status, education, residence, smoking, drinking, BUN, TC, TG, HDL, CRP, and eGDR).
Accuracy of the predictive model. (A) Basic model. (B) Basic model + SUA. basic model (including age, sex, marital status, education, residence, smoking, drinking, BUN, TC, TG, HDL, CRP, and eGDR).
Clinical benefit of the predictive model. (A) Basic model. (B) Basic model + SUA. basic model (including age, sex, marital status, education, residence, smoking, drinking, BUN, TC, TG, HDL, CRP, and eGDR).
Discussion
In this study, we investigated for the first time the predictive value of eGDR for new-onset HUA in non-HUA patients. We used a logistic regression model to adjust for potential confounding factors and to further explore the independent impact of eGDR on the risk of new-onset hyperuricemia (HUA) in patients without HUA. Additionally, RCS analysis revealed a linear association between eGDR and HUA. This indicates that higher eGDR scores are associated with a lower risk of HUA. Subgroup analyses revealed a significant association between eGDR and HUA incidence in most subgroups (P < 0.05). To ensure model stability and interpretability, we employed variance inflation factor (VIF) methods to detect and eliminate multicollinearity. After adjustment, all variables in the model exhibited VIF values below 5, effectively resolving multicollinearity. The final results confirmed that there were no excessive multicollinearity issues. Subsequently, a Bayesian approach was employed to analyze mediating effects, examining indirect, direct, and total effects. Significant associations were identified between eGDR and SUA, as well as between SUA and HUA. However, the influence of eGDR on HUA outcomes was entirely mediated through direct effects, with no significant mediating effects observed. The DeLong test further supported these findings: although baseline serum uric acid levels correlated with hyperuricemia outcomes, incorporating them into predictive models did not dominate the prediction.
Additionally, hypertension itself may influence uric acid metabolism through multiple mechanisms, such as affecting renal hemodynamics and glomerular filtration rate, thereby impacting uric acid excretion. Therefore, including hypertension status in the eGDR may introduce an inherent association with hyperuricemia, potentially affecting our accurate assessment of the relationship between eGDR and hyperuricemia21. To address this, we conducted a stratified analysis based on baseline hypertension status. The median age of participants was 68.0 years, while the median age of hypertensive individuals was 61.0 years (42.6% male, 57.4% female). The hypertension group exhibited significantly higher median age, lower educational attainment, and elevated levels of TC, TG, HDL, LDL, CRP, and SUA compared to the non-hypertension group (P < 0.05). Conversely, the hypertension group showed significantly lower proportions of males, married individuals, urban residents, alcohol consumers, smokers, and eGDR levels compared to the non-hypertension group (P < 0.05). Sensitivity analysis further excluded the influence of hypertension status on eGDR, thereby validating the robustness of the model results. Detailed statistical analyses revealed a significant statistical association between eGDR and HUA. This discovery not only provides a novel biomarker for early HUA diagnosis, but also offers potential guidance for clinical decision-making. To translate this statistical association (eGDR–HUA) into a clinically actionable decision framework22, we further evaluated the performance of eGDR testing in terms of discriminatory ability, calibration, and clinical utility. We validated the predictive performance and clinical applicability of the model using receiver operating characteristic curves (ROC), calibration curves, and decision curves23.
Prior research has demonstrated a robust correlation between hyperinsulinemic-euglycemic clamp and IR. The hyperinsulinemic-euglycemic clamp is currently the gold standard for evaluating IR. However, its intricate, time-consuming, and costly nature presents challenges for its use in large-scale epidemiological surveys9. In a study conducted by the National Health and Nutrition Examination Survey (NHANES) and the China Longitudinal Study of Health and Aging (CHARLS), another surrogate, the triglyceride-glucose (TyG) index, was employed as a biomarker for IR. The findings indicated that the TyG index was associated with an elevated risk of hyperuricemia24. However, the relationship between the estimated glucose disposal rate (eGDR), an indicator of insulin resistance, and the risk of new-onset HUA remains underexplored.
In light of these findings, it is proposed that IR may serve as a more straightforward, practical, and reliable alternative marker for predicting HUA events.
In clinical practice, given the invasive and consumptive nature of traditional methods of assessing IR status, the eGDR has been developed and demonstrated to have a high degree of IR accuracy, and the calculation of the eGDR is based only on the participant’s WC, HbA1c, and presence of hypertension, and the parameters required to calculate the eGDR are readily available, making it more suitable for large-scale clinical application10,25,26. Furthermore, the eGDR has a similar accuracy to the hyperinsulinemic-euglycemic clamp in assessing IR status27,28. The eGDR is closely associated with IR, which is an important risk factor for hyperuricemia. This suggests that eGDR may be a reliable alternative marker for IR in epidemiological studies evaluating IR, which is important for early detection and intervention of HUA disease in the population and has strong practicality, validity and applicability. At present, the relationship between eGDR and HUA is still poorly understood. However, our study showed that higher eGDR was significantly associated with a lower risk of developing HUA.
Although our results suggest that eGDR is linearly associated with the incidence of HUA and that higher eGDR is significantly associated with a lower risk of HUA, the exact biological mechanisms remain unclear. However, it can be elucidated through an understanding of the underlying pathophysiological processes, as follows: Firstly, in a state of insulin resistance, glycolytic intermediates are diverted to ribulose 5-phosphate and ribulose phosphate pyrophosphate, resulting in increased UA production29. This, in turn, results in the deposition of substantial quantities of UA in pancreatic islet cells, impairing the insulin secretion function of pancreatic islet B-cells and necessitating compensatory insulin secretion. Concurrently, HUA causes endothelial cell damage and a diminished capacity to consume glucose, producing an effect analogous to that of IR30. Secondly, IR results in a sustained hyperglycemic state, which impairs renal function and affects UA excretion. Furthermore, the hyperinsulinemia induced by IR increases renal reabsorption of UA, leading to elevated UA levels31,32. Thirdly, hyperglycemia and hyperlipidemia have been observed to reduce glyceraldehyde 3-phosphate dehydrogenase activity, which in turn has been demonstrated to promote uric acid synthesis33. Additionally, IR can indirectly enhance NADPH production by stimulating the lipolysis pathway, which is a principal source of serum uric acid and ultimately results in hyperuricemia34,35. In conclusion, IR can indirectly elevate NADPH production by stimulating the lipolysis pathway, which represents a significant source of serum uric acid and ultimately contributes to hyperuricemia.
The strength of this study lies in its population-based design, which employed a representative sample of the general Chinese adult population. This approach reduces estimation bias and improves the study’s internal validity. It should be noted, however, that the present study is not without certain limitations. Firstly, As CHARLS only collected blood samples in the 2011–2015 waves, 2018/2020 SUA data is unavailable, which could lead to an underestimation of the true incidence density. Consequently, further investigation into the impact of eGDR trajectories over time on HUA is recommended. Second, existing research suggests that dietary factors may influence uric acid levels. Although the original CHALRS study design incorporated multiple covariates, unmeasured confounders such as dairy, vegetable, and meat intake may introduce bias into the analysis due to their exclusion from this specific study population14. We applied the E-value method, yielding a result of 1.4. This value indicates that our study’s conclusions are robust in the face of unmeasured confounding factors. Even if unidentified confounding variables exist, fundamentally reversing the study results would require these confounders to exhibit strong associations with both the exposure and outcome variables—a scenario with a low probability in practical research. Third, although HbA1c measurements are typically conducted using standard methods, any condition that alters the lifespan of red blood cells (e.g., renal anemia resulting from erythropoietin use, hemolytic anemia, and severe iron-deficiency anemia) or recent transfusions can impact the accuracy of HbA1c measurements36. Fourth, the study population had an average age of 58.2 years, with 95% being Han Chinese adults. Due to differences in metabolic characteristics, vascular aging rates, and genetic backgrounds, caution should be exercised when extrapolating conclusions to younger populations or non-East Asian ethnicities. Future validation should be conducted in multi-ethnic, prospective cohorts. Fifth, since the on-site questionnaire did not list specific drug components, we could not adjust directly for potential confounding factors, such as thiazide, thiazide-loop diuretics, and urate-lowering therapy. Previous studies indicate that thiazide users have elevated serum uric acid (SUA) levels by an average of 30–40 μmol/L. If the usage rates and patterns of this cohort align with those in the literature, the association between eGDR and hyperuricemia may be slightly overestimated. Subsequent subgroup analyses by medication are required to quantify the effect of drug interference. Finally, we excluded samples containing missing values, which could introduce potential estimation bias.
Conclusion
Our study showed that a higher eGDR, an indicator of insulin resistance, was associated with a lower risk of developing HUA. eGDR is a reliable surrogate for assessing the prevalence of HUA and a strong predictor of HUA.
Data availability
The data supporting the findings of this study are available the CHARLS website (http://charls.pku.edu.cn/en). The code used and/or analyzed in this study will be made available by the corresponding author upon reasonable request.
References
Maiuolo, J., Oppedisano, F., Gratteri, S., Muscoli, C. & Mollace, V. Regulation of uric acid metabolism and excretion. Int. J. Cardiol. 213, 8–14 (2016).
Wu, J. et al. Hyperuricemia and clustering of cardiovascular risk factors in the Chinese adult population. Sci. Rep. 7(1), 5456 (2017).
Multidisciplinary expert task force on hyperuricemia and related diseases. Chinese multidisciplinary expert consensus on the diagnosis and treatment of hyperuricemia and related diseases. Chin. Med. J. (Engl). 130(20), 2473–2488 (2017).
Chen-Xu, M., Yokose, C., Rai, S. K., Pillinger, M. H. & Choi, H. K. Contemporary prevalence of gout and hyperuricemia in the United States and Decadal Trends: The National Health and Nutrition Examination Survey, 2007–2016. Arthritis Rheumatol. 71(6), 991–999 (2019).
Kumar, A. U. A. et al. Temporal trends in hyperuricaemia in the Irish health system from 2006–2014: A cohort study. PLoS ONE 13(5), e0198197 (2018).
Wang, L. et al. Prevalence and treatment of diabetes in China, 2013–2018. JAMA 326(24), 2498–2506 (2021).
Chen, Q. et al. Evaluating the risk of developing hyperuricemia in patients with type 2 diabetes mellitus using least absolute shrinkage and selection operator regression and machine learning algorithm. Digit Health 10, 20552076241241380 (2024).
Bhole, V., Choi, J. W., Kim, S. W., de Vera, M. & Choi, H. Serum uric acid levels and the risk of type 2 diabetes: A prospective study. Am. J. Med. 123(10), 957–961 (2010).
Liu, C. et al. RBP4 is associated with insulin resistance in hyperuricemia-induced rats and patients with hyperuricemia. Front. Endocrinol. (Lausanne) 12, 653819 (2021).
Nakamura, K. et al. HOMA-IR and the risk of hyperuricemia: A prospective study in non-diabetic Japanese men. Diabetes Res. Clin. Pract. 106(1), 154–160 (2014).
Krishnan, E., Pandya, B. J., Chung, L., Hariri, A. & Dabbous, O. Hyperuricemia in young adults and risk of insulin resistance, prediabetes, and diabetes: A 15-year follow-up study. Am. J. Epidemiol. 176(2), 108–116 (2012).
Panlu, K. et al. Associations between obesity and hyperuricemia combing mendelian randomization with network pharmacology. Heliyon 10(6), e27074 (2024).
Ma, L., Wang, J., Ma, L. & Wang, X. M. The link between hyperuricemia and diabetes: Insights from a quantitative analysis of scientific literature. Front. Endocrinol. (Lausanne). 15, 1441503 (2025).
Petersen, M. C. & Shulman, G. I. Mechanisms of insulin action and insulin resistance. Physiol. Rev. 98(4), 2133–2223 (2018).
Kahaer, M. et al. Triglyceride glucose index is more closely related to hyperuricemia than obesity indices in the medical checkup population in Xinjiang, China. Front. Endocrinol. (Lausanne). 13, 861760 (2022).
Bonora, E. et al. Homeostasis model assessment closely mirrors the glucose clamp technique in the assessment of insulin sensitivity: studies in subjects with various degrees of glucose tolerance and insulin sensitivity. Diabetes Care 23(1), 57–63 (2000).
Nevárez-Sida, A. & Guerrero-Romero, F. The triglycerides and glucose index: A cost-effectiveness analysis compared with the homeostatic model assessment for insulin resistance. Value Health Reg. Issues 37, 49–52 (2023).
Wang, L. et al. A national study exploring the association between triglyceride-glucose index and risk of hyperuricemia events in adults with hypertension. Prev. Med. Rep. 43, 102763 (2024).
Huang, X. et al. Visceral adipose accumulation increased the risk of hyperuricemia among middle-aged and elderly adults: A population-based study. J. Transl. Med. 17(1), 341 (2019).
Zhang, Y. et al. Association of hypertension and hypertriglyceridemia on incident hyperuricemia: An 8-year prospective cohort study. J. Transl. Med. 18(1), 409 (2020).
Lanaspa, M. A., Andres-Hernando, A. & Kuwabara, M. Uric acid and hypertension. Hypertens. Res. 43(8), 832–834 (2020).
Okechukwu, C. C. et al. Enhanced therapeutic efficacy of the nanoscale fluoropyrimidine polymer CF10 in a rat colorectal cancer liver metastasis model. Cancers (Basel) 16(7), 1360 (2024).
Huddleston, C. et al. Evaluating efficacy of cervical HPV-HR DNA testing as alternative to PET/CT imaging for posttreatment cancer surveillance: retrospective proof-of-concept study. Cancer Epidemiol. Biomarkers Prev. 34(8), 1264–1268 (2025).
Gou, R. et al. Association between triglyceride glucose index and hyperuricemia: A new evidence from China and the United States. Front. Endocrinol. (Lausanne) 15, 1403858 (2024).
Helliwell, R. et al. Body mass index, estimated glucose disposal rate and vascular complications in type 1 diabetes: Beyond glycated haemoglobin. Diabet Med. 38(5), e14529 (2021).
Girgis, C. M., Scalley, B. D. & Park, K. E. Utility of the estimated glucose disposal rate as a marker of microvascular complications in young adults with type 1 diabetes. Diabetes Res. Clin. Pract. 96(3), e70–e72 (2012).
Zabala, A. et al. Estimated glucose disposal rate and risk of stroke and mortality in type 2 diabetes: a nationwide cohort study. Cardiovasc. Diabetol. 20(1), 202 (2021).
Liu, C. et al. Predictive worth of estimated glucose disposal rate: evaluation in patients with non-ST-segment elevation acute coronary syndrome and non-diabetic patients after percutaneous coronary intervention. Diabetol. Metab. Syndr. 14(1), 145 (2022).
Leyva, F., Wingrove, C. S., Godsland, I. F. & Stevenson, J. C. The glycolytic pathway to coronary heart disease: A hypothesis. Metabolism 47(6), 657–662 (1998).
Borghi, C. et al. Expert consensus for the diagnosis and treatment of patient with hyperuricemia and high cardiovascular risk. Cardiol. J. 25(5), 545–563 (2018).
Rathmann, W., Funkhouser, E., Dyer, A. R. & Roseman, J. M. Relations of hyperuricemia with the various components of the insulin resistance syndrome in young black and white adults: the CARDIA study. Coronary Artery Risk Development in Young Adults. Ann Epidemiol. 8(4), 250–261 (1998).
Facchini, F., Chen, Y. D., Hollenbeck, C. B. & Reaven, G. M. Relationship between resistance to insulin-mediated glucose uptake, urinary uric acid clearance, and plasma uric acid concentration. JAMA 266(21), 3008–3011 (1991).
Gao, Z. et al. Renal impairment markers in type 2 diabetes patients with different types of hyperuricemia. J. Diabetes Investig. 10(1), 118–123 (2019).
Clausen, J. O., Borch-Johnsen, K., Ibsen, H. & Pedersen, O. Analysis of the relationship between fasting serum uric acid and the insulin sensitivity index in a population-based sample of 380 young healthy Caucasians. Eur. J. Endocrinol. 138(1), 63–69 (1998).
Vuorinen-Markkola, H. & Yki-Järvinen, H. Hyperuricemia and insulin resistance. J Clin Endocrinol Metab. 78(1), 25–29 (1994).
American Diabetes Association. 6. Glycemic Targets: Standards of Medical Care in Diabetes-2021. Diabetes Care 44(Suppl 1), S73-S84 (2021).
Funding
This study was supported by National Natural Sciences Foundation of China (Grant No. 81860524).
Author information
Authors and Affiliations
Contributions
Y. J. L. and N.Z. designed this study. Y. J. L. and M.S.Z. performed the statistical analysis. Y. J. L. wrote the first draft of the manuscript. Y.P.Z., T.X., J.J.T. and L.M.F. wrote some sections of the manuscript. Y. J. L. and N.Z. revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
CHARLS was approved by the Institutional Review Board of Peking University (approval number: IRB00001052-11015 for household survey and IRB00001052-11014 for blood sample), and all participants provided written consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liao, Y., Zuo, M., Xu, T. et al. Estimated glucose disposal rate and risk of incident hyperuricemia: insights from CHARLS. Sci Rep 15, 39439 (2025). https://doi.org/10.1038/s41598-025-22952-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-22952-w