Abstract
In this paper, we present the design, investigation, and optimization of a surface plasmon resonance (SPR) biosensor using a D-shaped photonic crystal fiber (PCF) with layers of gold and titanium oxide (TiO₂). The TiO₂ layer is applied on top of the gold layer to enhance sensitivity. This biosensor aims to simplify the manufacturing process while achieving optimal performance through effective coupling. We improved the design by thoroughly analyzing and optimizing various structural parameters to enhance performance. Our biosensor demonstrated exceptional diagnostic accuracy when tested on multiple samples, including cancer cells (Basal, MDA-MB-231, Jurkat, PC-12, and HeLa). The biosensor achieved a maximum wavelength sensitivity (WS) of 42,000 nm/RIU, a maximum amplitude sensitivity (AS) of – 1862.72 RIU, and a maximum figure of merit (FOM) of 1393.128 RIU⁻¹ across the wavelength range from visible to near-infrared and within a refractive index (RI) range of 1.3–1.4. The results indicate that this optimized biosensor exhibits reliable and accurate detection capabilities, capable of measuring minute changes in RI with high precision.
Similar content being viewed by others
Introduction
Cancer poses a significant biological challenge worldwide, affecting millions of lives each year. Timely and accurate identification of this disease can have a profound impact on individuals’ lives. In this regard, all-optical sensors have attracted significant interest from researchers and industry professionals, primarily because of their remarkable immunity to external electromagnetic fields. This immunity makes them particularly valuable in environments where electromagnetic interference can affect the performance of traditional electronic sensors. By utilizing light for sensing, these sensors offer enhanced reliability and stability, paving the way for advancements in various applications, such as telecommunications, medical diagnostics, and environmental monitoring. Surface plasmon resonance (SPR) biosensors that utilize photonic crystal fiber (PCF) are essential for detecting cancer as optical sensors. These biosensors have demonstrated outstanding promise across various applications, including biomolecule detection, medical diagnostics, blood type analysis, virus detection, food quality and safety inspection, environmental monitoring, and more1,2,3,4,5.
In PCF sensors, selecting suitable materials for the background and plasmonic substance is crucial. Silica (SiO2) is commonly used in PCF-SPR sensors6. The materials used as plasmonic substances include gold, silver, copper, aluminum, Palladium, indium, and sodium, which are capable of generating surface plasmons7,8. Gold and silver are the most well-known among these elements9,10. Sensors using SPR with silver substrates exhibit higher sensitivity compared to those using gold substrates. However, the low chemical stability of silver in SPR sensors restricts its broader application, and it is prone to surface tarnishing and corrosion in air and aqueous environments, and thus typically requires protective passivation layers that complicate fabrication and can degrade the near-field enhancement11. Alternatives like Pd and Pd-based nanostructures are attractive for active gas sensing (hydrogen) because of hydride formation, but their chemical reactivity and associated optical/structural changes make them less suitable as passive, long-term stable RI-biosensor coatings12,13. In contrast, gold has high chemical stability and resistance to oxidation. It also supports reliable, reproducible thin-film deposition and bioconjugation chemistry under ambient and biological conditions, making it the more commonly utilized material in SPR-PCF sensors14,15,16. Furthermore, the integration of various materials with plasmonic metals has become a significant area of investigation in SPR-based optical sensors. The use of materials such as oxides like TiO₂, Transition Metal Dichalcogenides (TMDCs), and other nanostructures like carbon nanotubes (CNTs), graphene, and MXene has been explored to either enhance sensitivity or improve the chemical stability of the plasmonic layer17,18,19,20,21,22. But depositing a metal layer onto PCF presents significant challenges in practice. To address issues such as coating small holes in the internal metal layers of PCF sensors and achieving a uniform thickness without roughness on circular surfaces, D-shaped sensors have been proposed. By polishing the surface of the PCF to create a flat area, it becomes possible to achieve a homogeneous metal coating with minimal roughness. This polished fiber surface positions the metal layer closer to the fiber core, providing an effective solution to the coupling issue between the core mode and the surface plasmon polariton (SPP) mode23.
Recently, there has been a growing focus on D-shaped PCF-based SPR sensors. One of the challenges in implementing and simulating these sensors is achieving optimal detection performance. The sensitivity to changes in the refractive index (RI) and the overall detection quality are indicated by factors such as wavelength sensitivity (WS), amplitude sensitivity (AS), figure of merit (FOM), and resolution, all of which are crucial for enhancing sensor performance. To improve these aspects, the primary challenge lies in finding the ideal combination of materials and structures that can boost sensitivity and detection quality. Many traditional sensors have primarily relied on metals like gold and silver; however, these materials require further optimization to attain high sensitivity and improved chemical stability. To address these challenges, researchers have explored various proposed structures and employed different plasmonic materials to enhance detection capabilities for a wide range of applications. Wu et al. conducted a numerical investigation of a D-shaped PCF sensor coated with gold. They analyzed the effects of different structural parameters on sensor performance and achieved a WS of 21,700 nm/RIU in the RI range of 1.33 to 1.34, with experimental results confirming their numerical analysis24. Ayanar et al. introduced a D-shaped PCF sensor utilizing the phase change material Ge2Sb2Te5 (GST) within the near-infrared (IR) wavelength range and an RI range of 1.35 to 1.40. This sensor demonstrated wavelength sensitivities of 17,600 nm/RIU for the crystalline phase and 8000 nm/RIU for the amorphous phase25. Abdelghafar et al. developed a biosensor with a V-shaped surface coated with ZrN as a plasmonic material for cancer detection. The maximum WS achieved for this structure is 6214.28 nm/RIU, specifically for breast cancer detection26. Pakarzadeh et al. developed a D-shaped PCF sensor that combined gold coating with ten layers of graphene to detect RIs ranging from 1.35 to 1.42 across the visible to IR wavelength spectrum. By adjusting the chemical potential of the graphene, they achieved tunable RI sensitivity, reaching a maximum WS of 7500 nm/RIU at a chemical potential of 1.2 eV27. Mu et al. presented a D-shaped PCF sensor using a dual layer of Au/Ti3C2Tx-MXene. This sensor operates within a wavelength range of 800–2020 nm and an RI range of 1.39 to 1.415, achieving a remarkable WS of 64,600 nm/RIU28. Li et al. have demonstrated that sensors enhanced with molybdenum disulfide (MoS2) can outperform silver-based sensors. Their experimental results indicate that within the RI range of 1.415 to 1.420, this sensor achieves a maximum WS of 16,000 nm/RIU. This enhancement has led to a 167.35% increase in performance compared to silver-based sensors29. Liu et al. introduced a D-shaped PCF sensor using silver and TiO2, which exhibited a maximum WS of 30,000 nm/RIU in an RI range of 1.22 to 1.3630. Elmahdi et al. developed an SPR PCF-based biosensor that utilizes a rhodium plasmonic material with a grating structure. This biosensor is designed for the detection of basal, breast, and cervical cancers, featuring WS of 19,750 nm/RIU, 20,428 nm/RIU, and 20,041 nm/RIU for these respective cell types31. Additionally, Chen et al. analyzed a D-shaped PCF sensor that utilized a dual layer of Au/MgF2 with a rectangular microchannel. This sensor operates within an RI range of 1.27 to 1.43 and a temperature range of 45 to 100 degrees Celsius, achieving a maximum WS of 31,800 nm/RIU32. Sardar et al. developed a sensor based on dual polished photonic crystal fibers for cancer detection. This sensor showed a maximum WS of 7143 nm/RIU, an AS of – 270 \(\:{\text{R}\text{I}\text{U}}^{-1}\), and an FOM of 900 \(\:{\text{R}\text{I}\text{U}}^{-1}\)33. Lastly, Dong et al. introduced and numerically analyzed a D-shaped sensor that incorporates a MoO2 nanofilm as the plasmonic layer. This sensor operates within an RI range of 1.38–1.43 and demonstrates a WS of 24,000 nm/RIU34. Despite the advancements in the development of SPR PCF sensors and the incorporation of various structural elements, challenges still exist in achieving reliable and stable detection performance across all diagnostic parameters. The introduced sensors have struggled to meet the criteria for excellent performance in every aspect. Specifically, certain parameters such as AS, FOM, and resolution are either not adequately addressed or demonstrate clear shortcomings in their performance.
The D-shaped PCF-based SPR biosensor presented in this study demonstrates remarkable sensitivity for precise cancer detection. The novelty of this work is threefold: first, a comprehensive optimization process yields a design with simultaneously high and balanced performance across all key metrics (WS, AS, and FOM), enhancing its diagnostic reliability. Second, we propose a practical sensing approach using cell cytoplasm, which links detection to protein concentration changes and simplifies experimental procedures. Third, the synergistic combination of optimized Gold/TiO₂ layers is shown to be critical for achieving the sensor’s exceptional sensitivity. This innovative design enables the biosensor to operate efficiently within the visible to near-infrared spectrum, responsive to RI changes between 1.3 and 1.4 RIU. Its performance is exemplified by a maximum WS of 42,000 nm/RIU and a FOM reaching 1393.128 RIU⁻¹, offering superior resolution and accuracy. Furthermore, its broad applicability is demonstrated through the capability to identify multiple cancer cell lines, such as Basal, HeLa, Jurkat, PC-12, and MDA-MB-231. With its high sensitivity and specificity, this biosensor holds significant promise for applications in early cancer detection and other biomedical diagnostics.
Proposed cancer detection system
The experimental setup for evaluating the performance of this biosensor is illustrated in Fig. 1a. Firstly, an electromagnetic wave is generated by a tunable optical laser source and transmitted through a single-mode fiber to a polarizer and then to the biosensor. The polarizer converts unpolarized light into polarized light. The analyte is introduced to and removed from the biosensor via a programmable pump, controlled through tubes and valves at an appropriate pressure. The output light from the biosensor is then sent through a single-mode fiber to an optical spectrum analyzer. The collected data is then transmitted to a computer for analysis35. In this detection configuration, the analyte under investigation will be cancer cells, but it’s challenging to completely inject the cell into the biosensor because of its micrometer-scale dimensions (Fig. 1b). Standard centrifugation methods can be used to separate different parts of a cell, such as the cytoplasmic liquid, which can then be injected into the biosensor for measurement (Fig. 1(c)). The change in the RI of the analyte leads to a change in the resonance wavelength. Cancer cells have a higher RI than normal cells due to the larger amount of proteins in the cytoplasm. Therefore, by measuring the changes in resonance wavelength, it is possible to measure the change in the RI of the analyte and detect cancer cells (Fig. 1(d, e))36.
Although the cytoplasmic fluid contains water, its overall refractive index is primarily determined by biomolecules such as proteins and lipids. Water exhibits an absorption peak in the mid-infrared region; however, in the near-infrared region relevant to the sensor, water absorption is minimal and does not significantly impact the measurement process. This characteristic allows changes in the cellular refractive index to influence shifts in the resonance wavelength, independent of the presence of water. Another consideration in biosensing is the scattering of light caused by cellular components. However, the sensor operates based on the shift of the resonance wavelength rather than the intensity of light, which minimizes the effect of scattering. Additionally, using cytoplasmic fluid instead of whole cells significantly reduces light scattering, as the fluid does not contain the large cellular organelles that typically cause strong scattering of light37,38,39,40.
(a) Experimental setup illustration of the proposed biosensor. (b) Cell and cytoplasm. (c) Schematic diagram of the biosensor. (d) Normal cell image. (e) Cancerous cell image.
Design and numerical characteristics of the proposed biosensor
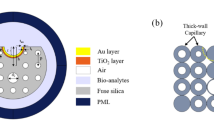
The proposed biosensor is a D-shaped PCF-based SPR sensor featuring 12 air holes. These holes are strategically arranged to achieve a specific geometry that controls the core-guided mode and its interaction with the plasmonic surface. The biosensor design features strategically placed holes to create an asymmetric structure, which induces birefringence and supports the Y-polarized mode.
(a) Cross-sectional view of the proposed biosensor. (b) 3D schematic view of the proposed biosensor. (c) FEM-based meshing.
Figure 2a illustrates the cross-section, while Fig. 2b shows the 3D view of the proposed biosensor, highlighting its structural parameters, and Fig. 2c shows the finite element method (FEM)-based meshing in the structure. The numerical solution for this biosensor uses the FEM. The sensor simulation is based on a two-dimensional cross-sectional area, as the structure is uniform along the propagation direction. The complete mesh consists of 17,326 domain elements and 16,726 boundary elements. The optimization of this biosensor follows a manual step-by-step approach, focusing on achieving the desired outputs for optimization. In the design of this biosensor, the holes labeled d1 and d2 primarily serve to confine the core-guided mode. The hole labeled dc is strategically positioned to lower the effective RI of the core and to create phase-matching conditions between the core-guided mode and the SPP mode. Meanwhile, the hole labeled d3 regulates the leakage of the core mode field to the plasmonic surface. The placement and dimensions of these holes are carefully engineered to provide adequate space for the core mode field to reach the plasmonic metal surface, thus maximizing sensitivity. The top surface of the biosensor is polished, and a layer of gold is deposited on this polished surface. Subsequently, a layer of TiO2 is coated over the gold layer. The analyte flows around the optical fiber and over the polished surface. To minimize unwanted reflections at the boundary regions, a perfectly matched layer (PML) is incorporated as the final layer.
In PCF-based sensors, silica is commonly used as the background material due to its favorable optical properties. The RI of silica is a critical parameter that influences the sensor’s performance and is described by the Sellmeier equation, as expressed in Eq. (1)41.
In this relationship, the wavelength of incident light is on the micrometer scale. B1, B2, and B3 are the Sellmeier coefficients and are equal to 0.69616300, 0.407942600, and 0.897479400 respectively. C1, C2, and C3 are the wavelength-independent parameters related to the material’s electronic band structure and are equal to 0.00467914826 µm2, 0.0135120631 µm2, and 97.9340025 µm2, respectively.
As mentioned previously, a dual layer of Au/TiO2 is utilized as the plasmonic layer in this biosensor. The detection performance of the biosensor is highly dependent on the characteristics of this layer. These characteristics include the permittivity of gold, obtained using the Drude-Lorentz approximation, as described in Eq. (2)42, and the RI of TiO2, which is given by the relation in Eq. (3)23. The relevant values for these relations are provided in Table 1.
Where \(\:{\uplambda\:}\) is the incident wavelength in µm and \(\:\text{Im}\left({\text{n}}_{\text{e}\text{f}\text{f}}\right)\:\)represents the imaginary part of the effective RI of the fundamental guided mode.
The length of the biosensor is inversely related to the maximum CL at the resonant wavelength. The larger this number, the longer the core and plasmon modes need to interact effectively, as described by Eq. (5)44:
One of the key parameters in assessing the performance of an SPR-PCF sensor is its sensitivity. Changes in the RI of the analyte lead to corresponding changes in the resonance wavelength of the sensor. The extent of the shift in resonance wavelength due to changes in RI is a key factor in assessing the performance of this biosensor, known as WS. To determine WS, a wavelength interrogation method is employed, which is based on Eq. (6)45:
In this equation, \(\:{{\Delta\:}{\uplambda\:}}_{\text{p}\text{e}\text{a}\text{k}}\:\)denotes the changes in the resonant wavelength in nm, and \(\:{\Delta\:}{\text{n}}_{\text{a}}\) represents the changes in the RI of the analyte.
Another type of sensitivity analysis in SPR-PCF sensors is AS, which is performed using the amplitude interrogation method. This sensitivity can be expressed by Eq. (7)46:
in this equation \(\:{\upalpha\:}({\uplambda\:},{\text{n}}_{\text{a}})\) shows the CL value at resonance wavelength, and \(\:\varDelta\:{\upalpha\:}({\uplambda\:},{\text{n}}_{\text{a}})\) is the change in the CL value between two resonance wavelengths.
Another aspect that can be mentioned for evaluating the performance of a biosensor is its ability to detect the smallest changes in RI, referred to as sensor resolution, which is determined by Eq. (8)47:
In this equation, \(\:{\Delta\:}{\text{n}}_{\text{a}}\) is the changes in the RI, \(\:{\Delta\:}{{\uplambda\:}}_{\text{min\:}}\) represents the smallest value of the spectral resolution, and \(\:{\Delta\:}{{\uplambda\:}}_{\text{peak}\text{}}\)is the shift of resonance wavelength in nm scale.
An important component in the plot of CL of the biosensor is the full spectral width of CL at half-maximum, which significantly influences the biosensor’s performance. Smaller values of this component enhance metrics such as FOM and detection accuracy (DA). The overall performance of a biosensor can be compared and evaluated by FOM derived from Eq. (9)45:
Additionally, achieving smaller values of FWHM means the resonance peak is more precisely located within a narrow range of wavelengths. This leads to greater accuracy in detecting changes in the RI. Consequently, a sensor with a smaller FWHM and a larger DA will be able to detect minor changes with increased precision. the DA of a biosensor is determined by Eq. (10)48:
In this biosensor, gold is used as the plasmonic material. To optimize the biosensor’s performance and select an appropriate thickness of gold, the performance was evaluated for three different thicknesses. The CL spectrum and AS graphs for various gold layer thicknesses are illustrated in Fig. 3a and b, respectively. As shown in this figure, increasing the thickness of the gold layer reduces the evanescent wave’s ability to penetrate this layer due to the inherent damping properties of gold, resulting in a decrease in CL and a blue shift in resonance wavelength. It is also evident that the Tg = 30 nm layer has the highest AS. Other necessary information is provided in Table 2. As shown in Table 2, the highest WS, AS, and FOM are achieved with the Tg = 30 nm layer. Therefore, the optimal thickness for this part is considered to be Tg = 30 nm.
(a) CL spectrum for different thicknesses of gold layer for RI = 1.38, RI = 1.39. (b) AS for different thicknesses of gold layer for RI = 1.38.
The use of TiO2 in SPR-PCF sensors is aimed at enhancing SPR and increasing sensitivity. In this biosensor, a layer of TiO2 is used on top of the gold layer to evaluate the performance of this biosensor using the TiO2 layer. Figure 4a shows the CL spectrum and Fig. 4b shows the AS graph for different TiO₂ thicknesses. As shown in Fig. 4a, increasing the thickness of this layer, which acts as a sensitivity-enhancing dielectric layer, results in increased CL, and the resonance peak shifts towards longer wavelengths. while the TiO₂ layer enhances the field, there is an optimal thickness (Tti = 10 nm) that maximizes the WS. A layer that is too thin provides insufficient enhancement, while a layer that is too thick can begin to shield the evanescent field from the analyte, reducing the overall sensitivity. As shown in Table 2, the highest values for WS, AS, FOM, and DA are achieved with the Tti = 10 nm layer. Therefore, a thickness of Tti = 10 nm is considered optimal.
(a) CL spectrum for different thicknesses of TiO2 layer for RI = 1.38, RI = 1.39, and Tg = 30 nm. (b) AS for different thicknesses of TiO2 layer for RI = 1.38 and Tg = 30 nm.
It is critical to optimize and place the air holes correctly when designing an efficient biosensor. The location and size of these holes have a significant impact on factors such as mode field distribution, coupling between core and SPP modes, birefringence, and ultimately the biosensor’s performance.
(a) CL spectrum for different diameters of d1 hole for RI = 1.38, RI = 1.39, Tg = 30 nm, and Tti = 10 nm. (b) AS for different diameters of d1 hole for RI = 1.38, Tg = 30 nm, and Tti = 10 nm.
Figure 5a shows the CL spectrum, and Fig. 5b shows the AS graph for different d1 diameters. With an increase in the diameter of this hole for an RI of 1.38, CL increases, while for RI = 1.39, the CL first increases and then decreases, resulting in a shift of the CL peak towards longer wavelengths. This behavior occurs because, for smaller diameters, the confinement of the core-guided mode and its coupling towards the plasmonic surface is less intense. As indicated in Table 2, the highest values of AS, WS, and FOM correspond to a diameter of d1 = 2 μm. Therefore, the diameter of d1 = 2 μm is considered the optimal value for this component.
(a) CL spectrum for different diameters of d2 hole for RI = 1.38, RI = 1.39, Tg = 30 nm, Tti = 10 nm, and d1 = 2 μm. (b) AS for different diameters of d2 hole for RI = 1.38, Tg = 30 nm, Tti = 10 nm, and d1 = 2 μm.
Figure 6a shows the CL spectrum, and Fig. 6b shows the AS graph for different d2 diameters. The purpose of this hole is to engineer and confine the core-guided mode; therefore, increasing the diameter of this hole beyond d2 = 1 μm prevents the core mode from accessing the plasmonic surface, resulting in a decrease in CL. However, changing the diameter of this hole does not alter the resonance wavelength. According to the results in Table 2, WS remains the same for all three values. However, for the other parameters, d2 = 1 μm shows the highest value and is considered the optimal component.
(a) CL spectrum for different diameters of dc hole for RI = 1.38, RI = 1.39, Tg = 30 nm, Tti = 10 nm, d1 = 2 μm, and d2 = 1 μm. (b) AS for different diameters of dc hole for RI = 1.38, Tg = 30 nm, Tti = 10 nm, d1 = 2 μm, and d2 = 1 μm.
Figure 7a shows the CL spectrum and Fig. 7b shows the AS graph for different dc diameters. As shown in this figure, with an increase in the diameter of the dc hole, there is an increase in CL and a shift of the resonance peak towards longer wavelengths. This is because, with an increase in the diameter of this hole, the distribution of the core-guided mode field increases, leading to a more evanescent field penetrating the plasmonic surface, which results in higher CL. According to Table 2, the highest WS corresponds to the 0.6 μm diameter, and the highest values of AS, DA, and FOM correspond to dc = 0.5 μm. Considering the trade-off between the available parameters, a value of dc = 0.5 μm can be regarded as optimal.
(a) CL spectrum for different diameters of d3 hole for RI = 1.38, RI = 1.39, Tg = 30 nm, Tti = 10 nm, d1 = 2 μm, d2 = 1 μm, and dc = 0.5 μm. (b) AS for different diameters of d3 hole for RI = 1.38, Tg = 30 nm, Tti = 10 nm, d1 = 2 μm, d2 = 1 μm, and dc = 0.5 μm.
Figure 8a shows the CL spectrum and Fig. 8b shows the AS graph for different d3 diameters. The d3 hole is positioned to manage and control the leakage of the core mode field to the plasmonic surface. With an increase in the diameter of this hole, naturally, a smaller amount of the field of the core-guided mode will reach the plasmonic surface. Therefore, it can be expected that CL will decrease with an increase in the diameter of d3. According to the results in Table 2, it can be inferred that WS is highest at d3 = 0.5 μm and d3 = 0.4 μm. AS and FOM are higher at d3 = 0.4 μm compared to the others, and DA is highest at d3 = 0.3 μm. Considering the trade-off between the parameters, d3 = 0.4 μm is deemed the optimal value.
Numerical performance of the proposed biosensor
The CL curve for the Y-polarized mode, the real part of the effective RI of the SPP mode, and the core mode and their mode field distribution for RI = 1.38 are shown in Fig. 9. According to the optimizations and results obtained in previous sections, the structural parameters of the proposed biosensor have the following values: Tg = 30 nm, Tti = 10 nm, d1 = 2 μm, d2 = 1 μm, d3 = 0.4 μm, and dc = 0.5 μm. According to Fig. 9a, when the real part of the effective RI of the core mode intersects with that of the SPP mode, a maximum CL is achieved at that point. This phenomenon indicates a coupling between the two modes, known as the phase-matching condition. This maximum CL occurs at the resonance wavelength λSPR. Figure 9b and c present the 2-D and 3-D mode field distributions of the core guided mode, respectively. Similarly, the 2-D and 3-D field profiles of the SPP mode are illustrated in Fig. 9d and e, while those of the SPR mode are shown in Fig. 9f and g. CL diagram for RI of 1.3 to 1.4 and AS of this biosensor for RI from 1.3 to 1.39 in 0.01 increments are illustrated in Fig. 10a and b, respectively. The resonance wavelength values and corresponding WSs for each RI from 1.3 to 1.4 and AS values for different RIs are listed in Table 3. As shown in this table, the highest WS of this biosensor is associated with an RI of 1.39 with a value of 42,000 nm/RIU, the average value is 7400 nm/RIU, and the highest AS is associated with an RI of 1.39 and is equal to -1862.72 RIU. The average AS for this biosensor is -479.63 RIU. The analysis of the CL diagram in Fig. 10a and the sensor response data in Table 3 reveals that sensor performance and CL improve at RIs of 1.38 and higher. This enhancement can be attributed to the increased coupling between the core guided mode and the plasmonic mode. As the RI of the analyte rises, the effective RI of the plasmonic mode also increases, bringing it closer to the effective RI of the core mode. This alignment strengthens the phase-matching condition. As a result of this improved phase matching, there is greater energy transfer between the two modes, leading to a stronger resonance. Additionally, as the RI of the analyte increases, the penetration depth of the evanescent field also rises, allowing for more interaction between light and the surrounding environment. This ultimately results in increased sensitivity, a reduced FWHM, and a higher FOM for the sensor49,50.
(a) CL curve for y-pol core mode and the phase matching condition among the SPP mode and core mode. The 2-D mode field distribution of (b) core guided mode (d) SPP mode, (f) SPR mode. The 3-D mode field distribution of (c) core guided mode, (e) SPP mode, (g) SPR mode.
The biosensor’s performance is also represented by other factors such as FWHM, DA, and FOM. Figure 11(a) displays the graph of FWHM and FOM values, and Fig. 11(b) shows the graph of FWHM and DA values. The exact values of FWHM, FOM, and DA for RI ranging from 1.3 to 1.4 in increments of 0.01 are provided in Table 3. These parameters provide a comprehensive understanding of the biosensor’s detection capabilities and its overall efficiency. The maximum FOM of this biosensor is 1393.128 RIU− 1 associated with an RI of 1.39 and its average value is 201.377 RIU− 1.
(a) CL spectrum of the biosensor for RIs from 1.3 to 1.4. (b) AS of the biosensor for RI of 1.3 to 1.39.
(a) Graph of FWHM and FOM values in the RI range of 1.3–1.39 in increments of 0.01. (b) Graph of FWHM and DA values in the RI range of 1.3–1.4 in increments of 0.01.
Moreover, this biosensor’s shortest and longest lengths correspond to RIs of 1.4 and 1.3, with values of 1.485 × 10− 5 m and 7.1551 × 10− 4 m, respectively. According to Fig. 12, as the CL increases, the biosensor length decreases.
Sensor length and CL variation with RI from 1.3 to 1.4.
The smallest change in the RI that the biosensor can detect is known as the sensor resolution. The data for RIs ranging from 1.3 to 1.4 is shown in Table 3. As illustrated in the table, the smallest detectable change by the biosensor is 2.38 × 10− 6 RIU, with an average value of 4.43 × 10− 5 RIU.
One of the key factors in evaluating SPR-PCF sensors is the relationship between the resonance wavelength and the RI of the analyte, as illustrated in Fig. 13. This relationship enables the prediction of the resonance wavelength based on variations in the RI. The polynomial fitting equation governing this biosensor is expressed as: y = 61,771.56 × 4 − 330,881.90 × 3 + 664,555.36 × 2 − 593,127.16x + 198,489.12. In this equation, y represents the resonance wavelength in micrometers (µm), while x denotes the RI of the analyte. The high R² value of 0.99 indicates that the polynomial fitting equation accurately reflects the relationship between the resonance wavelength and the RI of the analyte. This information is crucial for the design and optimization of the biosensor for various applications.
Resonance wavelength variation with RI from 1.3 to 1.4.
Cancer detection by the proposed biosensor
Changes in the RI of biological cells are significant components of biological processes, influenced by alterations in biochemical compounds and proteins present in the cytoplasm. It is important to note that the RI of biological tissues varies with wavelength. Generally, they exhibit normal dispersion behavior, meaning that, according to the Cauchy and Sellmeier equations, the RI slightly decreases as the wavelength increases. However, in the operational wavelength range of our sensor, this variation remains minimal, and researches conducted in the near-infrared range indicate that although the absolute value of the RI changes with wavelength, the difference in RI between cancer cells and normal cells remains significant. This distinction makes the method based on RI changes a valid approach for cancer diagnosis37,51,52.
CL spectrum of normal and affected cells (a) Basal cell, (b) HeLa cell, (c) Jurkat cell, (d) PC-12 cell, (e) MDA-MB-231 cell. (f) AS graph for Basal cell, HeLa cell, Jurkat cell, PC-12 cell, MDA-MB-231 cell.
In this section, we examine the performance of the proposed biosensor in various applications, including the detection of different types of cancer cells such as Basal, MDA-MB-231, Jurkat, PC-12, and HeLa33,36. The biosensor’s ability to detect and identify cancer cells, along with numerical values for detection parameters for each type of cancer, will be discussed. Figure 14 presents the CL spectra of both normal and affected cells for five different cell types: Basal (Fig. 14a), HeLa (Fig. 14b), Jurkat (Fig. 14c), PC-12 (Fig. 14d), and MDA-MB-231 (Fig. 14e). The AS graphs for each cell type are shown in Fig. 14f. Information related to this figure and the numerical performance of the biosensor is provided in Table 4. According to the results presented in the table, the proposed biosensor demonstrates a range of WS of (4000–32142.85), AS range of (569–1826.77), SR range of (2.5 × 10–5–3.11 × 10− 6), and FOM range of (74.83–683.32). By conducting investigations on the biosensor and observing its performance against various analytes, the excellent performance of this biosensor was demonstrated across all detection metrics, which makes this biosensor a suitable option for various applications, such as biosensing.
After a comprehensive analysis of the biosensor’s performance and the numerical results, it is evident that the biosensor has great potential to be used in medical diagnostics. Its high WS, FOM, and SR make it suitable for detection using the wavelength interrogation method. Additionally, its high AS and excellent performance in detecting cancer samples make it a strong candidate for medical diagnostics through the amplitude interrogation method. A performance comparison with other state-of-the-art sensors is presented in Table 5. The data clearly indicates that the proposed biosensor exhibits superior or highly competitive performance across all key diagnostic metrics, highlighting its significant advantages over existing designs. Although several reported sensors exhibit higher WSs, this improvement often comes at the expense of broader resonance linewidths (larger FWHM), which reduces the FOM and limits DA. Other designs achieve sharp resonances but suffer from weaker AS or narrower operational ranges. By contrast, the proposed sensor offers a balanced trade-off. The proposed SPR-PCF biosensor maintains high WS and AS while achieving a narrow FWHM, resulting in a superior overall FOM. This balance between sensitivity and spectral sharpness ensures reliable performance for practical biosensing applications.
Feasibility of fabricating the proposed biosensor
Although this study is based on numerical simulations, the proposed sensor is highly feasible for practical implementation by addressing its fabrication challenges. The primary challenge is ensuring structural uniformity. Achieving the precise dimensions and correct arrangement of air holes along the fiber’s structure is critical for the sensor’s consistent performance. Various fabrication methods can be employed for this biosensor, such as sol-gel, stack and draw, 3D printing, extrusion, and others57,58,59,60,61. The automated stack-and-draw method, utilizing high-precision capillaries, can guarantee the preservation of the sensor’s geometry during the drawing process62.
A second challenge is obtaining a smooth, polished surface at the appropriate depth without damaging the fiber’s core structure. This can be accomplished using the wheel polishing method (WPM), which allows for high-precision surface polishing. Furthermore, real-time monitoring of the polishing depth during the process, for instance with an optical microscope, is essential to prevent over-polishing63.
The final challenge involves the thin-film deposition of gold and TiO₂ to achieve a smooth, uniform surface. High-vacuum deposition methods are recommended. While Chemical Vapor Deposition (CVD) can be utilized for the gold layer, the TiO₂ layer requires a high-precision technique such as Atomic Layer Deposition (ALD). This method produces highly uniform, conformal, and pinhole-free films with angstrom-level precision, which is crucial for achieving the sensor’s ideal performance64,65.
Conclusion
This paper introduces a highly effective biosensor utilizing SPR PCF. We meticulously examined, analyzed, and optimized the structural parameters of the biosensor. To tackle manufacturing challenges, we implemented a specialized coating on a polished surface, effectively addressing issues such as achieving uniform metal surfaces on circular geometries and significantly reducing both costs and complexity in internal metal coating for sensors. This D-shaped design represents a key step towards practical and scalable fabrication. We selected gold and TiO₂ for their exceptional stability against oxidation. Through the optimization of structural parameters, we achieved remarkable enhancements in detection capabilities, which we compared favorably with findings from other studies. The biosensor demonstrated outstanding performance in detecting various cancer types, including Basal, MDA-MB-231, Jurkat, PC12, and HeLa, underscoring its potential in advancing medical diagnostics. Looking forward, future research can enhance sensor sensitivity and stability by using advanced plasmonic materials like MXene and graphene-metal. Optimizing deposition techniques such as atomic layer deposition (ALD) will improve coating uniformity and efficiency. While these advanced approaches promise significant performance gains, future work must also address their practical feasibility, including higher fabrication costs and complexity, to ensure a viable path toward clinical adoption. Expanding the refractive index detection range through innovative designs and employing artificial intelligence for spectral analysis will increase accuracy. This integration of AI will focus on both developing machine learning models for automated cell classification and utilizing signal processing algorithms to enhance the accuracy of experimental data. Ultimately, the path to clinical translation will require these advancements to be integrated into a robust prototype. Additionally, in vitro validation with real biological samples and the development of portable microfluidic systems can support clinical applications and aid early cancer detection.
Data availability
The data that support this study are available from the corresponding author upon reasonable request.
References
Goodrich, T. T., Lee, H. J. & Corn, R. M. Direct detection of genomic DNA by enzymatically amplified SPR imaging measurements of RNA microarrays. J. Am. Chem. Soc. 126 (13), 4086–4087 (2004).
Wu, Q. et al. A 2D transition metal carbide MXene-based SPR biosensor for ultrasensitive carcinoembryonic antigen detection. Biosens. Bioelectron. 144, 111697 (2019).
Fallahi, V., Kordrostami, Z. & Hosseini, M. Sensitivity and quality factor improvement of photonic crystal sensors by geometrical optimization of waveguides and micro-ring resonators combination. Sci. Reports. 14(1), 2001 (2024).
Khodatars Dashtmian, M. R. et al. Optimized SPR-PCF sensor for sucrose detection inspired by vertical pupil geometry. Sci. Rep. 15 (1), 29076 (2025).
Singh, S. et al. Gold immobilized SPR-Enhanced PCF biosensor for detection of cancer cells: a numerical simulation. Plasmonics 20 (6), 3535–3544 (2025).
Pinto, A. M. & Lopez-Amo, M. Photonic crystal fibers for sensing applications. J. Sens. 2012 (1), 598178 (2012).
Mousavi Monazah, S. M., Salehi, M. R. & Emami, F. Mathematical model for highly sensitive photonic crystal fiber sensor based on hyperbolic black holes. Sci. Rep. 14 (1), 25997 (2024).
Fallahi, V., Hosseini, M. & Kordrostami, Z. Optimization of highly sensitive three-layer photonic crystal fiber sensor based on plasmonic. Phys. Scr. 99 (10), 105577 (2024).
Janjan, B. et al. Ultra-wideband high-speed Mach–Zehnder switch based on hybrid plasmonic waveguides. Appl. Opt. 56 (6), 1717–1723 (2017).
Divya, J. et al. Surface Plasmon Based Plasmonic Sensors: A Review on their past, Present and Future Vol. 11, p. 100175 (X, 2022).
Hang, Y., Wang, A. & Wu, N. Plasmonic silver and gold nanoparticles: shape-and structure-modulated plasmonic functionality for point-of-caring sensing, bio-imaging and medical therapy. Chem. Soc. Rev. 53 (6), 2932–2971 (2024).
Naik, G. V., Shalaev, V. M. & Boltasseva, A. Alternative plasmonic materials: beyond gold and silver. Adv. Mater. 25 (24), 3264–3294 (2013).
Darmadi, I., Nugroho, F. A. A. & Langhammer, C. High-performance nanostructured palladium-based hydrogen sensors—current limitations and strategies for their mitigation. ACS Sens. 5 (11), 3306–3327 (2020).
Szunerits, S. et al. Recent advances in the development of graphene-based surface plasmon resonance (SPR) interfaces. Anal. Bioanal. Chem. 405, p1435–1443 (2013).
Tan, Y. et al. Graphene-deposited photonic crystal fibers for continuous refractive index sensing applications. Opt. Express. 23 (24), 31286–31294 (2015).
Kim, N. H. et al. Sensitivity and stability enhancement of surface plasmon resonance biosensors based on a large-area Ag/MoS2 substrate. Sensors 19 (8), 1894 (2019).
Khodatars Dashtmian, M. R. et al. Highly sensitive dual-side polished SPR PCF sensor for ultra-wide analyte range in the visible to near-IR operating band. Opt. Quant. Electron. 56 (7), 1187 (2024).
Karki, B. et al. Ultra-sensitive early detection of colorectal cancer using surface plasmon resonance sensor: theoretical analysis. Microchim. Acta. 192 (2), 126 (2025).
Karki, B. et al. Platinum diselenide and graphene-based refractive index sensor for cancer detection. Plasmonics 19 (2), 953–962 (2024).
Karki, B. et al. Tuning sensitivity of bimetallic, MXene and graphene-based SPR biosensors for rapid malaria detection: a numerical approach. J. Comput. Electron. 23 (4), 920–929 (2024).
Kumar, R. et al. Ultrasensitive Fano resonance surface plasmon resonance sensor using lithium fluoride and hexagonal Boron nitride. Microchem. J. 213, 113869 (2025).
Mayank et al. Detection of acetone concentration in water using a refractive index-based surface plasmon resonance sensor. Plasmonics, pp. 1–13. (2025). https://doi.org/10.1007/s11468-025-03133-x
Khalaf, M. K. et al. Au-TiO2 coated dielectric micro-channel based plasmonic refractive index sensor. Opt. Quant. Electron. 55 (7), 612 (2023).
Wu, T. et al. Surface plasmon resonance biosensor based on gold-coated side-polished hexagonal structure photonic crystal fiber. Opt. Express. 25 (17), 20313–20322 (2017).
Ayyanar, N. et al. Photonic crystal fiber-based reconfigurable biosensor using phase change material. IEEE Trans. Nanobiosci. 20 (3), 338–344 (2021).
Abdelghaffar, M. et al. Highly sensitive V-shaped SPR PCF biosensor for cancer detection. Opt. Quant. Electron. 55 (5), 472 (2023).
Pakarzadeh, H. et al. Graphene-assisted tunable D-shaped photonic crystal fiber sensor in the visible and IR regions. JOSA B. 39 (6), 1490–1496 (2022).
Mu, R. et al. Design and theoretical analysis of high-sensitive surface plasmon resonance sensor based on Au/Ti 3C2Tx-MXene hybrid layered D-Shaped photonic crystal fiber. IEEE Sens. J. 23 (16), 18160–18167 (2023).
Li, K. et al. Experimental study on high-sensitivity MoS 2 thickness modulation refractive index sensor based on SPR-PCF. IEEE Sens. J. 24 (12), 19007–19012 (2024).
Liu, X. et al. D-shaped photonic crystal fiber sensor based on the surface plasmon resonance effect for refractive index detection. Appl. Opt. 62 (16), E83–E91 (2023).
Elmahdy, N. A. et al. Highly sensitive plasmonic-grating PCF biosensor for cancer cell detection. Opt. Quant. Electron. 56 (4), 688 (2024).
Chen, L. Q. et al. Highly sensitive dual-function sensor for refractive index and temperature using D-shaped microchannel photonic crystal fiber. Opt. Express. 32 (7), 12405–12418 (2024).
Sardar, M. R. & Faisal, M. Dual-core dual-polished pcf-spr sensor for cancer cell detection. IEEE Sens. J. 24 (7), 9843–9854 (2024).
Dong, J. et al. Ultra-high sensitivity D-type photonic crystal fiber sensor deposited with MoO2 nanofilm. Opt. Mater. 152, 115483 (2024).
Khodatars Dashtmian, M. R. et al. Gold nanowire-enhanced SPR-PCF biosensor for high-throughput cancer cell detection in near-infrared. Plasmonics. https://doi.org/10.1007/s11468-025-03252-5 (2025).
Fallahi, V., Kordrostami, Z. & Hosseini, M. Cancer detection by photonic crystal optical biosensors: effect of hexagonal micro ring resonator design. Mater. Sci. Semiconduct. Process. 174, 108188 (2024).
Liang, X. et al. Determination of refractive index for single living cell using integrated biochip. in The 13th International Conference on Solid-State Sensors, Actuators and Microsystems, Digest of Technical Papers. TRANSDUCERS’05. IEEE. 2005. 2: pp. 1712–1715. (2005).
Sharma, P., Sharan, P. & Deshmukh, P. A photonic crystal sensor for analysis and detection of cancer cells. in International conference on pervasive computing (ICPC). 2015. IEEE. 2015. IEEE. (2015).
Ayyanar, N. et al. Photonic crystal fiber-based refractive index sensor for early detection of cancer. IEEE Sens. J. 18 (17), 7093–7099 (2018).
Ahmed, K. et al. Refractive index-based blood components sensing in Terahertz spectrum. IEEE Sens. J. 19 (9), 3368–3375 (2019).
Akowuah, E. K. et al. Numerical analysis of a photonic crystal fiber for biosensing applications. IEE J. Quantum Electron. 48 (11), 1403–1410 (2012).
Monazah, S. M. M. et al. Design and numerical analysis of a highly sensitive nano-layer coated photonic crystal fiber biosensor. Laser Phys. 33 (8), 086201 (2023).
Gao, D. et al. Multi-hole fiber based surface plasmon resonance sensor operated at near-infrared wavelengths. Opt. Commun. 313, 94–98 (2014).
Islam, M. S. et al. Dual-polarized highly sensitive plasmonic sensor in the visible to near-IR spectrum. Opt. Express. 26 (23), 30347–30361 (2018).
Fallahi, V., Hosseini, M. & Kordrostami, Z. Analysis of manufacturing process errors on the functionality of photonic crystal sensors. IEEE Sens. J. 25 (17), 32572–32579 (2025).
Gauvreau, B. et al. Photonic bandgap fiber-based surface plasmon resonance sensors. Opt. Express. 15 (18), 11413–11426 (2007).
Hautakorpi, M., Mattinen, M. & Ludvigsen, H. Surface-plasmon-resonance sensor based on three-hole microstructured optical fiber. Opt. Express. 16 (12), 8427–8432 (2008).
Mollah, M. A. et al. Designing highly sensitive surface plasmon resonance sensor with dual analyte channels. IEEE Access. 9, 139293–139302 (2021).
Knight, J. C. et al. All-silica single-mode optical fiber with photonic crystal cladding. Opt. Lett. 21 (19), 1547–1549 (1996).
Piliarik, M. & Homola, J. Surface plasmon resonance (SPR) sensors: approaching their limits? Opt. Express. 17 (19), 16505–16517 (2009).
Li, J. et al. Refractive indices of liquid crystals for display applications. J. Disp. Technol. 1 (1), 51 (2005).
Palik, E. D. Handbook of Optical Constants of Solids Vol. 3 (Academic, 1998).
Yasli, A. Cancer detection with surface plasmon resonance-based photonic crystal fiber biosensor. Plasmonics 16, 1605–1612 (2021).
Ibrahimi, K. M., Kumar, R. & Pakhira, W. C-grooved dual-core PCF SPR biosensor with graphene/au coating for enhanced early cancer cell detection. Appl. Phys. A. 130 (6), 439 (2024).
Arafin, S., Fatema, T. & Rahman, M. S. Numerical analysis and detection of cancer cells using a dual-core gold coated plasmonic biosensor. Opt. Mater. Express. 14 (6), 1603–1619 (2024).
Ashrafian, M., Olyaee, S. & Seifouri, M. Highly sensitive cancer detection using an open D-channel PCF-based SPR biosensor. Sci. Rep. 15 (1), 10168 (2025).
Li, D. et al. High sensitivity refractive index sensor based on multicoating photonic crystal fiber with surface plasmon resonance at near-infrared wavelength. IEEE Photonics J. 9 (2), 1–8 (2017).
El Hamzaoui, H. et al. Sol-gel derived ionic copper-doped microstructured optical fiber: a potential selective ultraviolet radiation dosimeter. Opt. Express. 20 (28), 29751–29760 (2012).
Vienne, G. et al. Ultra-large bandwidth hollow-core guiding in all-silica Bragg fibers with nano-supports. Opt. Express. 12 (15), 3500–3508 (2004).
Issa, N. A. et al. Fabrication and study of microstructured optical fibers with elliptical holes. Opt. Lett. 29 (12), 1336–1338 (2004).
Daher, M. G. et al. Innovative Sensor Based on Photonic Crystal Fiber for Efficient Determination of Various Explosives. Plasmonics, : pp. 1–10. (2025). https://doi.org/10.1007/s11468-025-03038-9
Pysz, D. et al. Stack and draw fabrication of soft glass microstructured fiber optics. Bull. Pol. Acad. Sci. Tech. Sci. 62 (4), 667–682 (2014).
Xie, Q. et al. Characteristics of D-shaped photonic crystal fiber surface plasmon resonance sensors with different side-polished lengths. Appl. Opt. 56 (5), 1550–1555 (2017).
Sardar, M. R. & Faisal, M. Numerical analysis of highly sensitive twin-core, gold-coated, D-shaped photonic crystal fiber based on surface plasmon resonance sensor. Sensors 23 (11), 5029 (2023).
Fu, P. H. et al. Optimization for gold nanostructure-based surface plasmon biosensors using a microgenetic algorithm. ACS Photonics. 5 (6), 2320–2327 (2018).
Acknowledgements
This work was supported by Shahid Rajaee Teacher Training University under Grant No. 1404/386017.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
M.KH. and V.F. conceived of the presented idea and developed the theory and performed the computations andwrote the main manuscript text. M.S. and S.O. investigate and supervised the findings of this work. All authorsdiscussed the results and contributed to the final manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Khodatars Dashtmian, M., Fallahi, V., Seifouri, M. et al. Advanced SPR biosensor with optimized gold-TiO2 D-shaped photonic crystal fiber for precise multi-cancer detection. Sci Rep 15, 41003 (2025). https://doi.org/10.1038/s41598-025-24697-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24697-y