Abstract
Childhood vaccination is a cornerstone of public health, significantly reducing morbidity and mortality from vaccine-preventable diseases. However, immunization coverage remains suboptimal in many low-resource settings, including Ethiopia. Mobile health (mHealth) interventions, such as SMS reminders, have shown promise in improving healthcare service utilization, yet their effectiveness in enhancing childhood vaccination uptake remains underexplored in Ethiopia. This study assesses the impact of an mHealth intervention on childhood vaccination coverage and associated factors among mothers in the Gamo and Gofa zones of South Ethiopia. A cluster randomized controlled trial (cRCT) was conducted among 680 pregnant mothers recruited from 20 randomly selected health institutions in the Gamo and Gofa zones. Participants were assigned to either the intervention group (n = 340), which received SMS reminders and health education messages, or the control group (n = 340), which received standard care. Eligible participants were pregnant women (≥ 18 years old) with a gestational age of 16–28 weeks, literate, and able to use mobile phones. The primary outcome was childhood vaccination completion, while secondary outcomes included timely Bacillus Calmette-Guérin (BCG) vaccination at birth. Data were collected through structured interviews and analyzed using multilevel logistic regression and generalized linear mixed models. The intervention group demonstrated a significantly higher childhood vaccination completion rate (AOR = 4.80, 95% CI: 1.75–13.01, p < 0.01). Timely BCG vaccination at birth was also significantly improved in the intervention group (Log Odds = 2.19, 95% CI: 0.69–3.70, p < 0.01). Sociodemographic factors, such as maternal age, colostrum feeding, provider, and family planning, were significantly associated with vaccination uptake. The study suggests that mHealth interventions, such as SMS health information and reminders, might significantly enhance childhood vaccination coverage. Implementing mobile-based strategies could help bridge immunization gaps and support national vaccination programs. Future studies should explore the scalability of mHealth interventions in diverse settings.
Clinical Trial Registration number: PACTR202211547107725; Date 22/11/2022.
Similar content being viewed by others
Introduction
Childhood vaccination is one of the most cost-effective public health interventions, significantly reducing morbidity and mortality from vaccine-preventable diseases (VPDs) such as measles, diphtheria, pertussis, tuberculosis, tetanus, hepatitis B, Haemophilus influenzae b, pneumococcal diseases, polio, and rotavirus1,2,3. Ethiopia’s national immunization program follows the Expanded Program on Immunization (EPI) schedule, which has evolved over time, where childhood vaccines are administered. The EPI aims to ensure universal vaccine coverage; however, suboptimal immunization rates continue to pose a challenge in many low- and middle-income countries (LMICs)4. Despite ongoing efforts to boost immunization coverage in Ethiopia, the childhood vaccination rate remains below the WHO target of 90%5,6. Several barriers, including limited maternal awareness, logistical challenges, healthcare accessibility, and socioeconomic disparities, are associated with missed or delayed vaccinations7,8.
Challenges in childhood vaccination coverage
In Ethiopia, childhood vaccination coverage varies significantly by region, ranging from 98% in Addis Ababa to as low as 50% in some rural areas5. Several factors that hinder optimal immunization coverage include:
Limited maternal education and awareness result in lower health literacy and less comprehensive compilation of the immunization schedule9,10.
Low vaccine uptake is linked to long travel distances, inadequate infrastructure, and low attendance at antenatal care11.
Misinformation, distrust, and sociocultural beliefs that significantly influence vaccination and decrease immunization adherence12,13.
The role of mHealth in immunization programs
Mobile health (mHealth) interventions have emerged as a promising strategy to bridge gaps in immunization coverage, particularly in resource-limited settings14. mHealth involves mobile phone-based technologies to deliver health-related information and reminders15. Studies have demonstrated that SMS reminders and mobile-based health education improve vaccine uptake by enhancing maternal knowledge, reinforcing immunization schedules, and reducing forgetfulness16,17,18. In Ethiopia, where mobile phone penetration has recently increased, leveraging mHealth interventions could provide a scalable solution to address vaccination gaps19,20.
According to the Africa Telecom 50 Report 2024 from Revenue Magazine, Ethiopia was the largest telecom market in East Africa in 2024, with approximately 87 million mobile subscriptions, accounting for roughly 28% of the region’s total21. This suggests broad mobile phone penetration within the country. While mobile coverage has expanded substantially, network strength remains uneven across urban, peri-urban, and rural areas, with noticeable gaps in 2G/3G and limited 4G/5G availability in many locations. Some rural regions rely on 2G or edge connectivity, and several communities depend on satellite or microwave backhaul to access the network22. Overall, among an estimated 120 million Ethiopians, nearly 40 million remain unconnected, primarily residing in the country’s peripheral areas22. Mobile Telephone Network (MTN) Group began operations in Ethiopia in 2021 and Safaricom of Kenya in 2022, after regulatory changes allowed increased competition in the mobile sector previously dominated by Ethio Telecom. This entry contributed to expanding mobile network access and introduced competitive dynamics in a market previously characterized by a state-backed monopoly, with implications for pricing, innovation, and service quality23,24.
Rationale for the study
Despite growing evidence supporting mHealth interventions in immunization programs, few studies have rigorously evaluated their effectiveness through cluster randomized controlled trials (cRCTs) in Ethiopia. Previous studies have shown that mobile-based reminders significantly improve ANC visits, skilled birth attendance, and postpartum follow-ups25, but their impact on childhood vaccination outcomes remains underexplored. Furthermore, the study area faces unique challenges related to healthcare accessibility, cultural influences, and maternal health-seeking behaviors, necessitating localized and evidence-based interventions26. Overall, we evaluate the effect of an mHealth intervention on childhood vaccination coverage and its associated factors among mothers in South Ethiopia. The findings will provide policy-relevant evidence on the effectiveness of mobile-based interventions in improving immunization uptake, contributing to national efforts to achieve universal vaccine coverage.
Methods
Study setting
This study is a cluster-randomized controlled trial (cRCT) conducted in the Gamo and Gofa zones. These zones are located in the South Ethiopia Region and are known for their diverse populations, rural-urban disparities, and varying levels of healthcare access. The study considered health institutions, such as primary hospitals, health centers, and health posts, which provide essential maternal and child health services, antenatal care (ANC), and childhood vaccination. To avoid auto-plagiarism, we do not replicate the same methodology across all articles from this trial; we provide an overview summary here.
Study design
We employed a cluster-randomized controlled trial (cRCT) to evaluate the effect of the mHealth intervention on childhood vaccination rates. Clusters were defined at the health institution level, ensuring that all pregnant mothers receiving ANC services within a selected institution were randomly assigned to either the intervention or control group.
Participants
The study enrolled pregnant women registered at the selected health facilities, identifying eligible participants from ANC registers at health centers and from family folders at health posts during routine ANC visits. Twenty health facilities were randomized into two arms, intervention and control, with 10 facilities in each arm. Two primary hospitals were included, one assigned to the control group and one to the intervention group. Health posts were not sampled separately because they are organized under health centers; when a health center was included, all its associated health posts automatically participated.
Inclusion criteria and exclusion criteria
Pregnant women were eligible for inclusion if they were at least 18 years old, had a gestational age between 16 and 28 weeks at recruitment, could read and write in Amharic, and could use a mobile phone independently. At the beginning of the intervention, the gestational age was 24 to 28 weeks. These criteria ensured that participants could fully engage with the mHealth intervention and comprehend the SMS content. Limiting participation to literate mothers helped ensure that observed outcomes were attributable to the intervention rather than differences in message interpretation. Requiring literacy also mitigated privacy and confidentiality concerns related to personal health information, especially on sensitive topics such as reproductive health and family planning. If illiterate mothers relied on others to read messages, this could introduce variability and contamination in message delivery, differences in tone, timing, or accuracy of interpretation. At the intervention’s outset, there was an opportunity to provide mobile phones to women who lacked them, which can make the program accessible to a broad group of participants. In a country with rapidly increasing mobile subscriptions, the intervention has the potential to benefit a broad segment of pregnant women21. Additionally, expanding delivery modes to include video and voice could benefit less-literate mothers in the future.
In Ethiopia (as in many countries), individuals under 18 are legally considered minors and cannot provide full informed consent on their own. Including them would have required parental or guardian consent, which is often complex and may compromise autonomy in sensitive topics like reproductive health and family planning.
Participants were excluded from the study if they reported mobile network or internet interruptions in their residential area during the pre-intervention interview, as consistent mobile access was essential for the intervention. Additionally, women who planned to relocate from the study area before the end of the follow-up period, or those with high-risk pregnancies requiring specialized medical care, were excluded to ensure safety and maintain consistent follow-up throughout the study duration. This is done because a woman might move to an area where the network can be interrupted.
Sample size determination
The sample size was determined using estimates from previous studies on childhood vaccination rates. Considering the evidence that shows a 70.9% vaccination rate among controls and 82.6%27 among the intervention group, we further considered 12% clinically significant differences, 0.011 ICC28, 80% power, 5% significance level, 95% CI, 1.37 design effect, and 20% attrition rate to calculate sample size. The final sample size was 680 women.
Randomization and allocation
A two-stage randomization process was used:
Health institution selection: Twenty health facilities were randomly selected from the 102 available. These were randomly assigned into equal intervention (n = 10) and control (n = 10) groups drawn from an opaque envelope. The opaque envelope contains pieces of paper with sequential numbers that contain the names of the health facilities. One person picks control, and the second person picks intervention from the same opaque envelope.
Individual participant selection: Eligible pregnant mothers were randomly selected within their respective clusters based on a computer-generated random sequence of numbers. The family number of the family folder and ANC registration numbers at health facilities were used as the frame for selection. That mean the recruitment team randomly selected women from the control and intervention health facilities. The recruitment team collected baseline data from all eligible women who signed a consent form. Randomization was conducted in health facilities because a randomization frame is difficult to find outside, and recording of pregnancy and gestational ages was also necessary for inclusion (Fig. 1).
Recruitment
In a two-stage cluster randomized technique, we first selected HFs from the sequentially numbered 102 HFs in an opaque envelope. Health facilities include district hospitals, health centers, and health posts. Each health center may encompass more than one Satellite health post. After HF’s selection, ANC registration and family folder numbers are used as a sampling frame to select mothers. For this purpose, a computer-generated sequence number is applied to those frames to get the desired HFs. The health centers oversee their associated health posts (community clinics) within the same catchment area, since health posts are part of the health centers. These health posts support women who do not visit the health center’s (main site) but remain in the community. We stopped recruitment after achieving an average of 34 women per facility and 340 participants per arm.
Intervention
Mothers under the mHealth intervention received tailored, scheduled SMS messages and reminders. The intervention started at the end of the second trimester of pregnancy and continued until six months postnatally. Mothers received messages on ANC services (ANC visits, danger signs, birth preparedness, nutritional advice, complications, institutional delivery (child and maternal), etc.) and PNC services (postnatal visits, partner and family support, family planning, vaccination, and child feeding). The vaccination-based SMS is sent along with all other messages. The mothers’ schedule led to the type of message they should receive during the week. In this document, we are focusing on vaccination-related information.
The intervention consisted of two types of message systems:
Mothers receive SMS reminders on their cellphones regarding their children’s immunization schedules. They also received health education messages on the importance of immunization and vaccines.
Messages were tailored to align with the child’s age-appropriate vaccination schedule and were sent in local languages. Vaccination schedules occur at the sixth week, tenth week, fourteenth week, and ninth month after childbirth3. A reminder message was dispatched to mothers one day before the scheduled time. All messaging processes are conducted through the computerized system connected to the Ethiopian telecommunications network. The reminder message schedules were adjusted during the second data collection in the first month after childbirth and were regularly monitored. The messages sent to mothers are kept concise to avoid being tedious to read. All mothers can read and understand the Amharic language. Messages were prepared in a way that considers the appropriate cultural orientation of the area. The national Reproductive, Maternal, Child Health, Family Planning, behavioural change communication strategies, and WHO guidelines were used to develop messages29. The prepared educational messages are delivered to mothers every other week. There are reminder messages interspersed among the educational messages.
Overall, 6,760 messages were sent to intervention groups during the postpartum period, and 98.5% were successfully delivered and read/seen by participants. We used a secure VPN provided by the Ethiopian Telecommunication Corporation, which was installed at Arba Minch University’s Health Informatics Department. A research technical assistant and manager with a computer background controlled sending, scheduling, and the status of messages. For scaling up to a wider area, we think about a centralized system instead of each health facility managing VPNs. A national or regional SMS gateway, managed by a government body, can send messages automatically, alleviating the burden on local staff. The use of simpler mobile-based dashboards (rather than VPNs) that can be introduced to frontline health workers can be a cost-effective method. This can enable mHealth in health extension programs in rural areas. In Ethiopia, health facilities have internet access (except health posts) and health facility-level health information technicians who can bridge the skills gap.
FrontlineSMS software was customized to fit the nature of the intervention. The trial manager was responsible for the software and following the message status. If the mother is not reading or opening messages, we make a voice call to check her situation. If the mother is not receiving messages and cannot be contacted through the registered addresses, she is recorded as lost to follow up.
Blinding
Due to the nature of the intervention, participants could not be blinded. However, healthcare providers and data collectors were blinded to minimize bias during data collection and follow-up assessments.
Control group
Mothers in the control group received standard maternal and child healthcare services without additional mHealth reminders or educational messages. All mothers in the control group continued to receive the established standard of antenatal, delivery, postnatal, and vaccination services (the usual care). The intervention (SMS messages and reminders) served as an additional supportive strategy, rather than a substitute for essential care. At the time of the study design, there was scientific uncertainty about the effectiveness of SMS-based mHealth health education and reminders in enhancing vaccination uptake in these settings. Therefore, conducting the trial was justified to generate evidence.
Variables
Sociodemographic characteristics
Maternal age, maternal and partner education, maternal and partner occupational status, household income, and residence.
Reproductive health factors
ANC visits, parity, mode of delivery, birth complications, and postnatal care (PNC) attendance.
Family planning factors
Contraceptive use, birth spacing, and fertility intentions.
Vaccination information
BCG, pentavalent 1, pentavalent 2, pentavalent 3, oral polio vacine 1, oral polio vaccine 2, oral polio vaccine 3, measles, vitamin A.
Health information and media exposure
Sources of health information, access to media (phones(static), TV, and radio), frequency of exposure to health messages, and birth preparedness information.
Primary outcome
Full childhood vaccination coverage is defined as the proportion of children receiving all recommended vaccines within the expected timeframe.
Secondary outcome
BCG vaccination at birth was assessed by maternal recall and, when available, by the presence of a vaccination scar on the child’s right upper arm. These are the common methods of checking child vaccination (BCG), including in the Ethiopian Demographic Health Survey30. The use of maternal recall as evidence for vaccination could potentially influence the magnitude of observed effects; nonetheless, recall remains a widely used and pragmatic approach in low-resource settings31,32.
Women and Mothers: In this document, the terms “mothers” and “women” are used interchangeably; however, first-time pregnant women (expectant mothers) are also included.
Data collection and quality control
The questionnaire was adapted from validated tools used in previous studies on maternal and child health33,34,35,36, ensuring contextual relevance and content validity. The tool underwent thorough pre-testing in a comparable population to assess clarity, flow, and cultural appropriateness. Based on the pretest results, necessary modifications were made to enhance the consistency and quality of the data collection process. Field supervision included spot checks, daily reviews of completed questionnaires, and feedback for data collectors. Data were gathered by trained professionals with prior research experience from Arba Minch University and Arba Minch College of Health Sciences, who received comprehensive training on the study objectives, interview techniques, confidentiality, and ethical conduct. Their knowledge of the local context and language significantly improved the effectiveness and reliability of the data collection. All completed interviews were reviewed daily, and any inconsistencies or errors were promptly addressed to ensure data quality. Both phone and in-person interviews adhered to standardized procedures to minimize interviewer bias and enhance data comparability.
Statistical analysis
Descriptive statistics were employed to summarize baseline characteristics. Chi-square tests and t-tests compared categorical and continuous variables between groups. Multilevel logistic regression and generalized linear mixed models (GLMM) assessed the effect of the mHealth intervention on vaccination outcomes, adjusting for clustering effects at the health institution level. Generalized Linear Mixed Models are powerful statistical methods used to analyze situations where the outcome variable is not normally distributed, whether binary, count, or categorical data. The data have a hierarchical or clustered structure (participants are nested within health facilities, or individuals within HF/catchment areas). We utilized multilevel logistic regression since the data were obtained from clusters; traditional logistic regression is not an appropriate method of analysis. Multilevel logistic regression (hierarchical logistic regression or mixed-effects logistic regression) is used because we have a binary dependent variable (vaccinated/not vaccinated: yes/no). This method allows for simultaneous analysis of individual-level and group-level effects and accounts for intra-cluster correlation that can bias standard logistic regression. This approach avoids underestimating standard errors, which can lead to incorrect conclusions and models contextual effects (e.g., how health facility characteristics influence outcomes). Level 1 variables are individual-level measurements (e.g., maternal age, education, parity, media exposure, etc.), while group-level variables measure (e.g., treatment, residence, aggregated women’s education, etc.).
The multilevel equation is mathematically represented as follows:
\(\:\text{log}\left(\frac{{{\uppi\:}}_{\text{i}\text{j}}}{1-{{\uppi\:}}_{\text{i}\text{j}}}\right)={\beta\:}_{0}+{\beta\:}_{0}{\text{X}}_{\text{i}\text{j}}\)+\(\:{\beta\:}_{2}{\text{Z}}_{\text{i}\text{j}}+\dots\:uij\); Where i and j are the level 1 (individual) and level 2 (community) units, respectively; X and Z refer to individual and community-level variables, respectively. πij is the probability of the childhood vaccination for the ith mother in the jth community.
To know cluster-level correlation strength, we calculated the Intra-Cluster Correlation as ICC=\(\:\frac{{{{\upsigma\:}}^{2}}_{a}}{{{{\upsigma\:}}^{2}}_{a}+{{{\upsigma\:}}^{2}}_{b}}\); where, \(\:{{{\upsigma\:}}^{2}}_{a}\) is the community-level variance and \(\:{{{\upsigma\:}}^{2}}_{b}\) is individual-level variance. The individual-level variance is constant (\(\:{\:{\uppi\:}}^{2}/3\)). Likelihood Ratio (LR) test for model comparison and deviance (−2LL) for goodness of fit were calculated, while37. The generalized linear mixed model.
g(µij)= β0 + β1 × 1 + β2 × 2+.+uj.
Where:
g(⋅): Link function (logit).
µij: Expected value of the outcome for individual iii in cluster j.
β0,β1,β2: Fixed-effect coefficients.
uj: Random effect for cluster j.
Adjusted odds ratios (AORs) with 95% confidence intervals (CIs) were reported to quantify associations. Statistical significance was set at p < 0.05. Analyses were conducted using Stata software. We recognize that risk ratios (RRs) can offer a more intuitive measure of intervention effects in public health research, especially when the outcome is common, as RRs directly convey the likelihood of an event occurring in one group compared to another. ORs, in contrast, may overestimate the strength of association when the outcome prevalence is high38,39. The models were assessed using various criteria such as the intraclass correlation coefficient, Akaike Information Criterion, Bayesian Information Criterion, Log-likelihood, Model variances, and Deviance.
Research process
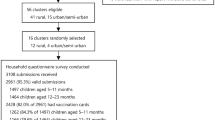
The flow diagram below indicates that the study enrolled 680 women, with 340 in the intervention group and 340 in the control group. Five participants were lost to follow-up, leaving 675 women assessed for vaccination completion from birth to six months (337 in the intervention group and 338 in the control group) (Fig. 1).
The research process diagram.
Results
For this specific objective, 675 women were assessed for vaccination completion from birth to six months. The four articles from the data share similar study characteristics, including sociodemographic, socioeconomic, and reproductive health factors. To avoid overlapping the same basic characteristics, we presented them in another article, and only vaccine variables are included here. The overall dropout and lost follow-up rate was limited to < 1%. 95% of mothers in the mHealth intervention group vaccinated their children compared to 87% of mothers in the control group. Both groups had low coverage of BCG, at 60% for the intervention and 42% for the controls, respectively. The overall child vaccination rate among the study participants was 91% (Table 1).
Table 2 shows the effect of different factors, including the mHealth intervention, on childhood vaccination using a multilevel model. The mHealth intervention significantly increased childhood vaccination rates (more than double the odds compared to controls) (AOR = 4.80, 95% CI: 1.75–13.01, p < 0.01). Children who received colostrum for three days had 3.41 times higher odds of vaccination than those who did not (AOR = 2.82, 95% CI: 1.03–7.68, p < 0.05). Mothers who used postpartum family planning were 7.2 times more likely to vaccinate their children (AOR = 6.18, 95% CI: 3.20–11.91, p < 0.001). When the service provider is health extension workers (HEWs), childhood vaccination reduced by 65% (AOR = 0.35, 95%CI: 0.12–0.99, p < 0.05) (Table 2).
The final model (Model 3) provides the best fit. Adding individual-level predictors, such as colostrum feeding, postpartum family planning (PPFP), mHealth, initiation time for breastfeeding, and other factors listed in Table 2, significantly improves the prediction of vaccination uptake. As more predictors are added to the model, all the group-level variances are reduced (0.47 to 0.00), and the inter-cluster correlation is reduced (ICC: 0.12 to 0.00), indicating that these variables explain most of the previously observed clustering. The log-likelihood ratio (LLR) improves markedly (from − 201 to −135), reflecting a better overall fit. Correspondingly, the AIC and BIC values decrease, further confirming the enhanced model performance (Table 3).
Secondary outcomes
Table 4 examines BCG at birth, its effects on next childhood vaccinations, and associated factors. The intervention significantly increased the likelihood of childhood vaccination, reinforcing previous findings (Log Odds = 2.19, 95% CI: 0.69–3.70, p < 0.01). Children whose mothers received iron supplementation had lower odds of vaccination (Log Odds = 0.69, 95% CI: 0.06–1.23, p < 0.05). Age of the mother (Log Odds = 0.08, 95% CI: 0.04–14, p < 0.01). Positive effect, suggesting that older mothers are more likely to vaccinate their children (Table 4).
Although the model performance improved from the random effect model to the final models, we cannot perfectly avoid variance at the cluster and individual levels.
Discussion
The findings of this study provide valuable insights into how mHealth interventions, maternal health behaviors, and socio-demographic factors affect childhood vaccination rates. The results underscore the importance of digital health solutions, early infant feeding practices, and integrated healthcare services for increasing immunization coverage, with standard healthcare and health education provided to both groups and the intervention delivered exclusively to the intervention group.
The study demonstrated that mHealth interventions might significantly improve childhood vaccination uptake (AOR = 4.80, p < 0.01). This finding aligns with other studies showing that mobile health technologies enhance maternal engagement with healthcare services by providing timely reminders, improving access to health information, and reinforcing positive health behaviors15,40. Studies in low-resource settings reported that mobile messaging and telehealth interventions increased immunization rates by addressing common barriers such as forgetfulness, misinformation, and logistical challenges27,41,42. Based on this linkage between mHealth exposure and vaccination, there is a need to scale up digital health strategies, particularly in rural and underserved areas, where traditional healthcare outreach is limited43. Future programs might explore multi-platform approaches (SMS, voice messages, social media) to enhance engagement and sustain behavioral change among caregivers.
The findings suggest that colostrum feeding (AOR = 2.82, p < 0.05) strongly predicts childhood vaccination. This is consistent with studies showing that early breastfeeding behaviors reflect overall maternal health-seeking tendencies, where mothers who engage in recommended breastfeeding practices are more likely to adhere to immunization schedules44,45,46. This indicates that considering the entire service could be more beneficial than focusing solely on individual components. Since exclusive breastfeeding reduces childhood morbidity and enhances immune function, promoting breastfeeding-friendly policies and strengthening infant feeding education during antenatal and postnatal care may also indirectly improve vaccination adherence47. This ensures that maternal and child health services can coexist effectively. Women who utilize child services, such as childhood vaccination, are also likely to seek maternal services, including postpartum family planning (AOR = 6.18, p < 0.001). This aligns with evidence suggesting that women who access reproductive health services are more likely to seek preventive care for their children48,49. Integrated healthcare models that combine immunization with family planning services have been successfully implemented in Malawi, Benin, Kenya, Uganda, and Liberia, resulting in higher vaccine uptake and better maternal health outcomes50,51,52. These findings suggest that strengthening postpartum care by integrating family planning and immunization could be an effective dual-intervention approach to improve maternal and child health. While health extension workers (HEWs) conduct most vaccination activities, their overall influence on childhood vaccination rates appears limited. Evidence also shows that strained relationships between community health workers and the formal health system weaken the connections between CHWs and the communities they serve, potentially undermining vaccination efforts53.
Health extension workers in the Ethiopian health system are equivalent to community health workers in other countries. They typically hold diploma-level educational qualifications and serve as front-line health workers in villages. The relationship they maintain with the community and the information they provide may require evaluation due to their comparatively low influence relative to other professions. An environment that motivates health workers could be crucial in enhancing overall performance.
Children of mothers who received SMS had 2.19 times higher Logodds (95% CI: 0.69–3.70, p < 0.05) of receiving the BCG vaccination at birth. This reinforces the idea that structured interventions, especially those involving health education and behavioral modifications, can significantly improve maternal and child health outcomes over time. Similar findings have been reported in other studies, where targeted interventions increased health service utilization, breastfeeding rates, and vaccination coverage15,54. These results underscore the importance of scaling up evidence-based interventions and ensuring wider program coverage to maximize public health impact. Iron supplementation was associated with a higher uptake of BCG at birth (Logodds = 0.69, p < 0.05). The trend suggests that iron supplementation is linked to BCG uptake at birth, influencing early childhood development and maternal motivation to stay in services27,55,56. Age had a positive association (Logodds = 0.08, p < 0.01) with receiving the BCG at birth, suggesting that the likelihood of this desired outcome also increases. This finding aligns with studies showing that older mothers are more experienced, have better health literacy, and are more likely to engage in positive health-seeking behaviors49. As anemia remains a major challenge in low-resource settings, expanding iron and micronutrient supplementation programs during antenatal care services and community-based initiatives may enhance maternal well-being and improve child health indicators.
Strengths of the study
The study employed multilevel models to enhance the robustness of estimates and the reliability of inferences by accounting for clustering and hierarchical data. It included a range of predictors such as maternal age, intervention effects, transportation methods, partner support, and iron supplementation. By adjusting for potential confounders, the analysis enhances internal validity and supports more generalizable conclusions. The study assessed the potential impact of the intervention on maternal and child health outcomes, with significant findings showing its scalability and informing policymakers about effective maternal health strategies. Model comparison, using AIC, BIC, and ICC values, adds statistical rigor to the study’s conclusions.
Limitations
Participants may exhibit slight variations in health-seeking behaviors, and residual confounding may persist despite statistical adjustments. The study’s short-term impact assessment complicates the evaluation of intervention sustainability over time. In instances where data collectors were unable to locate mothers, mobile interviews were conducted, which could hinder the verification of vaccine evidence. The findings are derived from a specific population and geographical context, which may not apply to other regions with differing healthcare infrastructures, cultural norms, or economic conditions. But this may not affect the application in Ethiopia and similar regions’ scale-up. Although approximately 73% of the population in Ethiopia has mobile subscriptions, we believe that including only women who can read and write presents certain limitations. Additionally, missing values complicate some models and removed data with many missing values. This may affect the representativeness of the report; however, considering low instances with missing, statistical power remain intact. The limited mobile network coverage in the country’s peripheral regions could hinder large-scale implementation.
Conclusion and recommendations
Despite these limitations, the study provides robust evidence on the effectiveness of maternal health interventions and the role of socio-environmental factors. The study reveals that mHealth interventions improved childhood vaccination rates and underscores the crucial role of maternal health behaviors in adherence. Integrating healthcare strategies that combine maternal education, family planning, breastfeeding promotion, and immunization services to optimize child health outcomes. By implementing these evidence-based strategies, policymakers and health practitioners can increase childhood vaccination rates and improve maternal-child health outcomes. Mobile health initiatives should be implemented to deliver personalized vaccine reminders and educational messages. The study indicates that the motivation of front-line health professionals is essential for improving childhood vaccination. Future studies might explore alternative mHealth modalities (such as voice calls, interactive voice response [IVR] systems, or audio messages in local languages) that can include illiterate mothers.
Data availability
The data generated during this study are fully available on proper request from the corresponding author.
Abbreviations
- PPFP:
-
postpartum Family Planning
- BCG:
-
Bacillus Calmette-Guérin
- HFs:
-
Health Facilities
- UNICEF:
-
United Nations Children Emergency Fund
- ANC:
-
Antenatal Care
- PNC:
-
Postnatal Care
- ICC:
-
Intra-Cluster Correlation
- HEWs:
-
Health Extension Workers
- AIC:
-
Akaike Information Criterion
- BIC:
-
Bayesian Information Criterion
- GA:
-
Gestational Age
- Penta:
-
pentavalent
- OPV:
-
oral polio vaccine
- AOR:
-
Adjusted Odds Ratio
- CI:
-
Confidence Interval
References
WHO. Global Vaccine Action Plan. Vaccine ;31:B5–B31. DOI: https://doi.org/10.1016/j.vaccine.2013.02.015. (2013).
Strategic Advisory Group of Experts on Immunization, REVIEW. The Global Vaccine Action Plan 2011–2020. Review and lessons learned [Internet]. Geneva: World Health Organization. (2019). Available from: https://www.who.int/publications/i/item/global-vaccine-action-plan-and-decade-of-vaccines-review-and-lessons-learned-reports
Ministry of Health-Ethiopia. National Implementation Guideline for Expanded Program on Immunization. ;(June):1–33. (2021). Available from: https://e-library.moh.gov.et/library/wp-content/uploads/2022/07/National-Implementation-Guidline-for-Expanded-Program-on-Immunization.pdf
UNICEF. Every child survives and thrives[anual report]. 2019;1-219. Available at: https://www.unicef.org/report
MoH (Ethiopian Ministry of Health). Ethiopia National Expanded Program on Immunization. July 2021. 1-122. Available at: http://dataverse.nipn.ephi.gov.et/bitstream/handle/123456789/1657/Ethiopia-National-Expanded-Program-on-Immunization.pdf?sequence=1
Biset, G., Woday, A., Mihret, S. & Tsihay, M. Full immunization coverage and associated factors among children age 12–23 months in Ethiopia: systematic review and meta-analysis of observational studies. Hum Vaccines Immunother [Internet] 17(7), 2326–2335. https://doi.org/10.1080/21645515.2020.1870392 (2021).
Mebrat, A. et al. Determinants of Incomplete childhood vaccination among children aged 12–23 months in Gambela Region, Southwest Ethiopia: a case control study. Ethiop J Heal Sci. 31(1), 63–72. https://doi.org/10.4314/ejhs.v31i1.8 (2021) (PMID: 34158753).
Desalew, A., Semahegn, A., Birhanu, S. & Tesfaye, G. Incomplete vaccination and its predictors among children in ethiopia: A systematic review and Meta-Analysis. Glob Pediatr. Heal. 7, 1–20. https://doi.org/10.1177/2 (2020). 7/2333794X20968681.
Tesema, G. A., Tessema, Z. T., Tamirat, K. S. & Teshale, A. B. Complete basic childhood vaccination and associated factors among children aged 12–23 months in East africa: a multilevel analysis of recent demographic and health surveys. BMC Public. Health. 20 (1), 1–14 (2020).
Girmay, A. & Dadi, A. F. Full immunization coverage and associated factors among children aged 12–23 months in a Hard-to-Reach areas of Ethiopia. Hindawi 2019, 8 (2019).
Eshete, A., Shewasinad, S. & Hailemeskel, S. Immunization coverage and its determinant factors among children aged 12–23 months in ethiopia: a systematic review, and Meta- analysis of cross- sectional studies. BMC Pediatr. 20, 1–13. https://doi.org/10.1186/s12887-020-02163-0 (2020).
Dubé, E., Gagnon, D., MacDonald, N. & SAGE Working Group on Vaccine Hesitancy. Strategies intended to address vaccine hesitancy: review of published reviews. Vaccine 33(34):4191–4203. https://doi.org/10.1016/j.vaccine.2015.04.041. Epub.
Chernet, A. G., Dumga, K. T. & Cherie, K. T. Home delivery practices and associated factors in Ethiopia. J. Reprod. Infertil. 20 (2), 102–108 (2019).
Hurt, K., Walker, R. J., Campbell, J. A. & Egede, L. E. mHealth Interventions in Low and Middle-Income Countries: A Systematic Review. Glob J Health Sci. 8(9), 54429. https://doi.org/10.5539/gjhs.v8n9p183 (2016) (PMID: 27157176).
Free, C. et al. The effectiveness of Mobile-Health technologies to improve health care service delivery processes: A systematic review and Meta-Analysis. PLoS Med. 10 (1), 1–26. https://doi.org/10.1371/journal.pmed.1001363 (2013).
Haji, A. et al. Reducing routine vaccination dropout rates: evaluating two interventions in three Kenyan districts, 2014. BMC Public Health [Internet]. 1–8. (2016). Available from: https://doi.org/10.1186/s12889-016-2823-5
Jaca, A., Mathebula, L., Iweze, A., Pienaar, E. & Wiysonge, C. S. A systematic review of strategies for reducing missed opportunities for vaccination. Vaccine [Internet]. 36(21):2921–2927. (2018). https://doi.org/10.1016/j.vaccine.2018.04.028. Available from: https://doi.org/10.1016/j.vaccine.2018.04.028.
Bangure, D. et al. Effectiveness of short message services reminder on childhood immunization programme in Kadoma, Zimbabwe - A randomized controlled trial, 2013. BMC Public. Health. 15 (1), 1–8. https://doi.org/10.1186/s12889-015-1470-6 (2015).
Mekonnen, Z. A., Gelaye, K. A. & Were, M. C. Mothers intention and preference to use mobile phone text message reminders for child vaccination in Northwest Ethiopia. BMJ Heal Care Inf. 28 (1), 1–9. https://doi.org/10.1136/bmjhci-2020-100193 (2021).
Eba, K. et al. Mobile health service as an alternative modality for hard-to-reach pastoralist communities of Afar and Somali regions in Ethiopia. Pastoralism [Internet]. 13(1):1–12. (2023). Available from: https://doi.org/10.1186/s13570-023-00281-9
Africa_Business_Community. Ethiopia tops East Africa’s mobile market with 86.6m subscriptions [Internet]. pp. 1–2. (2025). Available from: https://africabusinesscommunities.com/tech-24/ethiopia-tops-east-africa’s-mobile-market-with-86.6m-subscriptions/
Nigusie, M. Ethiopia Struggles to close digital divide as 40 million remain offline [Internet]. pp. 1–2. (2025). Available from: https://birrmetrics.com/ethiopia-struggles-to-close-digital-divide-as-40-million-remain-offline/#:~:text=While 3G coverage reaches nearly the entire population%2 C,many urban and almost all rural areas underserved.
AerolLeads Top Mobile Companies In Ethiopia [Internet]. p. 1. (2025). Available from: https://aeroleads.com/list/top-mobile-companies-in-ethiopia#:~:text=Mobile companies in Ethiopia are few in number%2 C,company and has a monopoly over the industry.
GSMArena. Network coverage in Ethiopia [Internet]. pp. 1–2. (2025). Available from: https://www.gsmarena.com/network-bands.php3?sCountry=ETHIOPIA
Gibson, D. G. et al. Mobile phone-delivered reminders and incentives to improve childhood immunisation coverage and timeliness in Kenya (M-SIMU): a cluster randomised controlled trial. Lancet Glob Health. 5(4), e428–e438. https://doi.org/10.1016/S2214-109X(17)30072-4 (2017).
Darebo, T. D., Oshe, B. B. & Diro, C. W. Full vaccination coverage and associated factors among children aged 12 to 23 months in remote rural area of Demba Gofa District, Southern Ethiopia. PeerJ 10 (March), 1–17. https://doi.org/10.7717/peerj.13081 (2022).
Mekonnen, Z. A., Gelaye, K. A., Were, M. & Tilahun, B. Effect of mobile phone text message reminders on the completion and timely receipt of routine childhood vaccinations: Superiority randomized controlled trial in Northwest Ethiopia. JMIR mHealth uHealth 9(6), 1–31. https://doi.org/10.2196/27603 (2021) (10.2196/27603 PMCID:).
Taljaard, M. et al. Intracluster correlation coefficients from the 2005 WHO global survey on maternal and perinatal health: implications for implementation research. Paediatr. Perinat. Epidemiol. 22 (2), 117–125 (2008).
Wagle, K. & Septmeber 23. pp. 1–8. Available from: https://publichealthnotes.com Behavior Change Communication (BCC): Importance and Strategies. (2019).
Central Statistical Agency (CSA) [Ethiopia] and ICF. Ethiopia Demographic and Health Survey. Addis Ababa, Ethiopia, and Rockville, Maryland, USA: CSA and ICF. [Internet]. (2016). Available from: https://www.dhsprogram.com/pubs/pdf/FR328/FR328.pdf
Miles, M., Ryman, T. K., Dietz, V., Zell, E. & Luman, E. T. Validity of vaccination cards and parental recall to estimate vaccination coverage: a systematic review of the literature. Vaccine 31 (12), 1560–1568. https://doi.org/10.1016/j.vaccine.2012.10.089 (2013).
Langsten, R. & Hill, K. The accuracy of mothers’ reports of child vaccination: evidence from rural Egypt. Soc. Sci. Med. 46 (9), 1205–1212. https://doi.org/10.1016/S0277-9536(97)10049-1 (1998).
Megersa, N. D., Tariku, E. Z., Yesera, G. E. & Gutema, B. T. Prevalence of prelacteal feeding and its associated factors among mothers of under-24-month-old children at Arba minch Zuria district, ethiopia: A cross-sectional study. SAJCH South. Afr. J. Child. Heal. 15 (2), 112–117. https://doi.org/10.7196/sajch.2021.v15i2.01698 (2021).
Downs, S. M. et al. The impact and implementation of an mHealth intervention to improve infant and young child feeding in senegal: IIMAANJE protocol for a cluster randomized control trial. Front. Public. Heal. 11, 1–8. https://doi.org/10.3389/fpubh.2023.1258963 (2023).
Gilano, G., Dekker, A. & Fijten, R. Effect of mobile phone messaging on uptake of maternal and child health service in southern Ethiopia: Protocol for cluster randomized controlled trial. Clin Nutr Open Sci [Internet]. ;54:89–99. (2024). https://doi.org/10.1016/j.nutos.2024.02.002. Available from: https://doi.org/10.1016/j.nutos.2024.02.002.
Adam, M. et al. The philani MOVIE study: A cluster-randomized controlled trial of a mobile video entertainment-education intervention to promote exclusive breastfeeding in South Africa. BMC Health Serv. Res. 19 (1), 1–14. https://doi.org/10.1186/s12913-019-4000-x (2019).
Belachew, A. B., Kahsay, A. B. & Abebe, Y. G. Individual and community-level factors associated with introduction of prelacteal feeding in Ethiopia. Arch Public Heal [Internet] 74(1), 1–11. https://doi.org/10.1186/s13690-016-0117-0 (2016).
Zou, G. A modified Poisson regression approach to prospective studies with binary data. Am. J. Epidemiol. 159 (7), 702–706. https://doi.org/10.1093/aje/kwh090 (2004).
Zhang, J. & Yu, K. F. What’s the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA 280 (19), 1690–1691 (1998).
Marcolino, M. S. et al. The impact of mHealth interventions: systematic review of systematic reviews. JMIR mHealth uHealth. 6 (1), 1–12. https://doi.org/10.2196/mhealth.8873 (2018).
Knop, M. R. et al. Impact of mHealth interventions on maternal, newborn, and child health from conception to 24 months postpartum in low- and middle-income countries: a systematic review. BMC Med [Internet]. 22(1):1–19. (2024). Available from: https://doi.org/10.1186/s12916-024-03417-9
Atnafu, A., Otto, K. & Herbst, C. H. The role of mHealth intervention on maternal and child health service delivery: findings from a randomized controlled field trial in rural Ethiopia. Mhealth 3, 39. https://doi.org/10.21037/mhealth.2017.08.04 (2017).
WHO. Global strategy on digital health 2020–2025. Geneva: World Health Organization; Licence: CC BY-NC-SA 3.0 IGO. [Internet]. 18. August. (2021). Available from: https://www.who.int/publications/i/item/9789240020924
Edmond, K. et al. Timing of initiation, patterns of breastfeeding, and infant survival: Prospective analysis of pooled data from three randomised trials. Lancet Glob Heal [Internet]. 4(4):1–10. (2016). Available from: https://doi.org/10.1016/S2214-109X(16)00040-1
Smith, E. R. et al. Delayed breastfeeding initiation and infant survival: A systematic review and meta-analysis. PLoS One. 12 (7), 1–16. https://doi.org/10.1371/journal.pone.0180722 (2017).
Mericq, V. et al. Long-term metabolic risk among children born premature or small for gestational age. Nat. Rev. Endocrinol. 13 (1), 50–62. https://doi.org/10.1038/nrendo.2016.127 (2017).
Victora, C. et al. Breastfeeding series Group. Breastfeeding in the 21st century: epidemiology, mechanisms, and lifelong effect. Lancet 387 (100017), 475–490. https://doi.org/10.1016/S0140-6736(15)01024-7 (2016).
Ahmed, S., Li, Q., Liu, L. & Tsui, A. Maternal deaths averted by contraceptive use: an analysis of 172 countries. Lancet 380(9837):111–125. https://doi.org/10.1016/S0140-6736(12)60478-4. Epub 2.
Cleland, J., Harbison, S. & Shah, I. H. Unmet need for contraception: issues and challenges. Stud. Fam Plann. 45 (2), 105–122. https://doi.org/10.1111/j.1728-4465.2014.00380.x (2014).
Cooper, C. M. et al. Successful proof of concept of family planning and immunization integration in Liberia. Glob Heal Sci. Pract. 3 (1), 71–84. https://doi.org/10.9745/GHSP-D-14-00156 (2015).
Hamon, J. K. et al. Integrated delivery of family planning and childhood immunisation services: A causal loop analysis of service responsiveness in Malawi. SSM - Qual. Res. Heal. 2 (June), 885–894. https://doi.org/10.1016/j.ssmqr.2022.100159 (2022).
Hamon, J. K. et al. Integrated delivery of family planning and childhood immunisation services: a mixed methods assessment of service responsiveness. BMC Health Serv Res [Internet]. 22(1):1–11. (2022). Available from: https://doi.org/10.1186/s12913-022-07983-7
Kok, M. C. et al. Optimising the benefits of community health workers’ unique position between communities and the health sector: A comparative analysis of factors shaping relationships in four countries. Glob Public Health [Internet]. 12(11):1404–32. (2017). Available from: https://doi.org/10.1080/17441692.2016.1174722
Sara, S. & Kim, Manish Patel, A. H. Use of m-Health in polio eradication and other immunization activities in developing countries. Vaccine Elsevier. 35 (10), 1373–1379. https://doi.org/10.1016/j.vaccine.2017.01.058 (2017).
Kabongo, E. M., Mukumbang, F. C., Delobelle, P. & Nicol, E. Explaining the impact of mHealth on maternal and child health care in low- and middle-income countries: a realist synthesis. BMC Pregnancy Childbirth. 21 (1), 1–17. https://doi.org/10.1186/s12884-021-03684-x (2021).
Gilano, G., Sako, S., Molla, B., Dekker, A. & Fijten, R. The effect of mHealth on childhood vaccination in Africa: A systematic review and meta-analysis. PLoS One [Internet]. 19(2 February):1–21. Available from: http://dx.doi.org/10.1371/journal.pone.02944. (2024). Available from: https://doi.org/10.1371/journal.pone.0294442
Acknowledgements
The authors thank Arba Minch and Maastricht University for their invaluable support and assistance in facilitating this study.
Funding
We received financial support from the Arba Minch University Research Directorate Office. The grant number for the project is GOV/AMU/PhDMast/TH3/CMHS/HI/RCO/01/15.
Author information
Authors and Affiliations
Contributions
GG developed the proposal, wrote the methods and results, and drafted the manuscript. AD is the first supervisor of the study and development of the hypothesis, introduction, methods section, revising, and editing of the final manuscript. RF is the second supervisor and is involved in all sections as AD.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Informed consent was obtained from all participants before their inclusion in the study. Trial number: PACTR202211547107725. Date of approval: 22/11/2022. Trial status: registered in Pan African Clinical Trial Registry. Trial website: www.pactri.org. We obtained Ethical approval from the Arba Minch University Institutional Review Board (AMU-IRB) committee, Ref. No 1326/2022. Confidentiality and anonymity were maintained throughout the study, and participants were assured of their right to withdraw at any time. The trial’s purpose and procedures were communicated to all study participants. The study adheres to Good Clinical Practice guidelines and the Declaration of Helsinki’s ethical principles for research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gilano, G., Dekker, A. & Fijten, R. The effect of mHealth on childhood vaccination and its associated factors among South Ethiopian mothers: a cluster randomized controlled trial. Sci Rep 15, 41668 (2025). https://doi.org/10.1038/s41598-025-25568-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-25568-2