Abstract
Malaria, a vector-borne disease transmitted by female Anopheles mosquitoes, a lethal insect instigator, is typically found in tropical and subtropical locations. In this paper, we developed the new malaria disease fractional order model under the Mittag-Leffler kernel to understand the dynamics and preventive control of malaria transmission in Northern Cyprus. The model is formulated as a deterministic eight-compartmental with subpopulations of humans and mosquitos for this study. The qualitative study of the suggested model is thoroughly presented, including an outline of its essential properties and a review of theorems. Linear growth and Lipschitz criteria are used to verify the existence and uniqueness of solutions. Along with determining the equilibrium points and reproductive number \((R_0)\) for sensitivity analysis of the system with sensitivity index parameters. The malaria fractional order model was analyzed as locally asymptotically stable, and the Lyapunov function first derivative test for global stability. The Newton Polynomial method is used to generate the algorithm for numerical simulations at different fractal order values and fractal dimensions to understand the impact of infection dynamics in the region that support theoretical results. The asymptotically stable worldwide endemic equilibrium revealed by numerical simulations highlights the necessity of major efforts to reduce malaria. The findings indicate a considerable rise in susceptible human populations and a decline in infectious mosquito numbers. It could be worthwhile to consider making a few significant adjustments to the current methodology in future studies.
Similar content being viewed by others
Introduction
Malaria is a severe infectious disease caused by Plasmodium parasites transferred to humans by the bite of infected female Anopheles mosquitos1. Malaria in human beings is caused by five different species of Plasmodium (single-celled protozoa): Plasmodium falciparum, Plasmodium vivax, Plasmodium malariae, Plasmodium ovale, and Plasmodium knowlesi. Plasmodium falciparum has the highest death and morbidity rates. However, Plasmodium vivax is the most widely dispersed species in the world2,3. These two species represent the greatest threat and contribute the most to global malaria prevalence. Plasmodium malariae is the third most prevalent species, with a prevalence range of 15–40%. According to the World Health Organization (WHO) report, globally, 249 million malaria cases were detected in 85 countries in 2022, with an estimated 608,000 malaria-related deaths. In 2022, the African region was home to 94% of malaria cases (233 million) and 95% of malaria deaths (580,000)4. Over the last two decades, global malaria cases have been on a downward trend, dropping from 80 to 57 cases per 1,000 people at risk between 2000 and 2019. Similarly, malaria-related deaths have also declined from 25 to 10 deaths per 100,000 people at risk. Despite malaria elimination initiatives in Sub-Saharan Africa, the disease load remains stable, especially among endemic territories. Despite a decline in death from 444,600 in 2020 to 427,854 in 2021, malaria cases jumped from 165 million to 168 million. Plasmodium falciparum malaria decreased, although Plasmodium malariae and Plasmodium ovale infections increased two to six times5. The COVID-19 pandemic has had a substantial impact on the epidemiology of imported malaria, with travel restrictions reducing the number of travel-related infections. However, statistics indicate that those returning from endemic areas are more likely to contract severe malaria6. Malaria cases documented between 2020 and 2021 were lower than in pre-pandemic years; however, initial data was inconclusive. The number of imported malaria cases increased in 2021 as COVID-related travel restrictions were removed, implying that severe malaria cases may rise.
The European Center for Disease Prevention and Control (ECDC) reported 2432 confirmed malaria cases in 2020 and 4780 in 2021, down from a peak of 8462 in 2019. France, Germany, Spain, and Belgium had the highest instances in 2020 and 20216,7. During the COVID-19 pandemic, the number of imported malaria cases increased as people traveled more. According to research by the Spanish national cooperation network8, the number of severe malaria cases increased significantly between 2020 and 2021. Another study9 found that relaxing COVID-19 limitations led to increased rates of parasitemia, hyperparasitemia, and severe malaria cases. The COVID-19 pandemic presents considerable difficulty in controlling malaria, as both diseases exhibit similar symptoms. Its outgrowths have intensified the incidence of malaria in Africa, threatening millions of lives10. Cyprus is the third-largest island (9.251 km\(^2\)) in the Mediterranean, after Sicily and Sardinia11. Like all countries in the Mediterranean basin, Cyprus has been severely affected by malaria for centuries. In the first half of the 1900s, malaria was endemic in Cyprus. However, with the “Malaria Eradication Project”carried out on the island of Cyprus between April 1946 and July 1949, malaria infection was successfully eradicated from the island12,13. Today, there are no endemic malaria cases in Cyprus, and all cases are imported. All imported malaria cases detected in Cyprus come to the island from endemic countries, mainly to study at universities14. Malaria is among the notifiable diseases in Cyprus. According to Turkish Republic of Northern Cyprus (TRNC) Ministry of Health data, a total of 56 imported malaria cases were detected in Northern Cyprus in the last eight years15. In the southern part of Cyprus, 17 imported malaria patients were reported between 2016 and 202016.
To understand the global dynamics of different diseases, the study of dynamic systems necessitates the development of mathematical models17. Understanding community-based infectious diseases requires an understanding of epidemiological research. Model construction, parameter estimation, parameters’ sensitivity testing, and numerical simulation computation are all aided by mathematical modeling. This aids in comprehending the relationship between infection and disease transmission within the community18. Numerous scholars have effectively showcased mathematical representations of an array of viral diseases19,20,21,22,23. These models help to formulate appropriate policies to control the disease by conducting prospective studies. Although researchers have adopted mathematical models to explain malaria dynamics and management, these efforts have not effectively lowered the disease’s spread24. More model-based research on malaria dynamics and awareness campaigns is required (e.g.,25,26,27).
The order of differential equations limits classical models involving derivatives. To get around these constraints, researchers are turning to a relatively new branch of mathematics called fractional calculus. Mathematical modeling is valuable and widely used across numerous scientific areas because fractional-order models accurately predict real-world phenomena (e.g.,28,29,30). To improve classical calculus for fractional-order modeling, several mathematical strategies have been used (see,31,32,33,34,35). In their study36, Yunus and Olayiwola modeled malaria using fractional-order differential equations and the Caputo fractional derivative, taking into account long-term memory effects and control measures like therapy and enlightenment. Numerical solutions were found using the Laplace-Adomian Decomposition Method, which enabled simulation of epidemic trajectory and assessment of the efficacy of actions in lowering infection levels. According to certain research cited in37,38, vaccination awareness is crucial for preventing infectious diseases like measles and the Ebola virus. The research supports attempts to combat endemic problems by incorporating behavioral elements into a framework of fractional calculus. Two orders, a fractional order and a fractal dimension, have been proposed by Atangana39 for a novel fractional order that combines operators from differential calculus and integral calculus. Researchers have made extensive use of the fractal-fractional model, a unique method for researching infectious diseases40,41,42,43. This will serve as our inspiration as we develop and examine a fractal-fractional model to comprehend the dynamics of malaria transmission.
The goal of this study is to examine the evolution of malaria in Northern Cyprus using a typical SEIR framework and fractal-fractional derivative, emphasizing the region’s distinct demographics and long incubation period. Using a modified model, this study focuses on basic features of the dynamics of malaria. This is how the remainder of the paper is structured. In Section “Model formulation”, the deterministic malaria model is developed. Section “Key features of the proposed model” explains the salient features of the proposed malaria mathematical model, which include positivity, boundedness, and epidemiologically feasible region. Section “Global derivative’s effect” provides information on how the global derivative affects the existence and uniqueness of the malaria model. Section “Qualitative analysis” determines the equilibrium points, the reproductive number \((R_0)\), and its sensitivity analysis. Section “Stability analysis” examines the model’s local and global stability. Section “Numerical solutions” employs the numerical scheme to derive the solution. In Section “Simulation results”, the model’s numerical simulations are run and discussed. The conclusion is found in Section “Conclusion”.
Model formulation
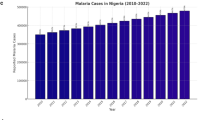
This work is based on the malaria cases real data in North Cyprus between 2014 and 2022. All datasets that were used in this study were taken from some scientific papers and hospitals in North Cyprus, which are publicly available on the website of the Ministry of Health of North Cyprus. Detailed information of cases was taken from the laboratories with ethical approval. The scientific papers that were used are cited in the paper. All of the cases obtained were in the age range of 18–28. 63% of the cases were men, while 37% of them were women. 97% of the cases were imported malaria cases. Moreover, these cases were from countries that have malaria as an endemic disease. The rest of the cases (3%) were local cases. In Fig. 1a,b, the distribution of cases according to years is shown both as numbers and percentages, respectively. The distribution of cases according to months is visualized in Fig. 2a,b as numbers and percentages, respectively. Figure 3a,b are illustrated to indicate the distribution of cases in seasons. These distributions are given both as numbers and percentages, respectively.
The distribution of cases in numbers and percentages according to years.
The distribution of cases in numbers and percentages according to months.
The distribution of cases in numbers and percentages according to seasons.
Atangana39 introduced a new class of fractional notions by combining power-law, exponential law, and modified Mittag-Leffler law with fractal derivatives. This study uses the fractal-fractional operator with the Mittag-Leffler kernel, which is defined below.
Definition 2.1
39,40,43 Let \(u: (a,b) \rightarrow [0, \infty )\) be a continuous, fractal differentiable function of dimension \(\rho\). The fractal-fractional derivative of u using a generalized Mittag-Leffler kernel of order \(\sigma\) is as follows:
where the fractal derivative is given by
\(\sigma ,\rho \in (0,1]\), and \(\textbf{E}_{\sigma }\) presents Mittag-Leffler function. The corresponding integral is as follows:
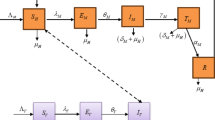
Since Northern Cyprus has distinct social dynamics and longer incubation periods, the evolution of malaria there is represented mathematically using a deterministic compartmental model. This enables us to comprehend the dynamics of malaria transmission more thoroughly. The model considers the populations of human hosts and mosquito vectors at various stages in the following ways:
-
\(S_h\): Susceptible human population;
-
\(E_h\): Exposed human population;
-
\(I_e\): Infected human population (whose countries have an endemic malaria disease);
-
\(I_{ne}\): Infected human population (whose countries have not an endemic malaria disease);
-
R: Recovered human population;
-
\(S_m\): Susceptible mosquito population;
-
\(E_m\): Exposed mosquito population;
-
\(I_m\): Infected mosquito population.
When developing a mathematical model, we began by making the following fundamental assumptions.
-
For biological considerations, it is assumed that all variables are non-negative.
-
The total human population at time t, denoted by \(N_H(t)\), is given by
$$\begin{aligned} N_h(t)=S_h(t)+E_h(t)+I_e(t)+I_{ne}(t)+R(t), \end{aligned}$$(4)and the total mosquito population at time t, denoted by \(N_m(t)\), is given by
$$\begin{aligned} N_m(t)=S_m(t)+E_m(t)+I_m(t). \end{aligned}$$(5) -
Due to comparable birth and death rates, the overall population of both human beings and mosquitoes remains constant.
-
Those who recover from malaria develop a temporary immunity to reinfection.
-
The susceptible human population is influenced by recruitment rates \(\Lambda\) and \(\Phi\), as well as reinfection or fading immunity of recovered populations at the rate of q.
-
The force of infection, \(\frac{(\beta _{h_1}+\beta _{h_2}) {S}_h {I}_m}{{N}_h}\), shifts the susceptible human population to the exposed class. The exposed class develop infection at rates of \(\alpha _1\) and \(\alpha _2\), and hence relocate to the infectious compartments.
-
Infected people recover at rates of \(\gamma _1\) and \(\gamma _2\).
-
The logistic equation \(\Theta N_m (1-\delta )\left( 1-\frac{N_m}{p}\right)\) describes mosquito recruitment rates in susceptible mosquito population.
-
The force of infection in mosquitoes, \(\frac{(\beta _{m_1}+\beta _{m_2})({I}_{e}+{I}_{ne}) {S}_m}{{N}_m}\), shifts susceptible mosquitoes to the exposed compartment. The exposed mosquitoes develop infection at a rate of \(\alpha _3\) and hence relocate to the infectious mosquito compartment.
-
The natural deaths of humans and mosquitoes are provided by \(\mu _h\) and \(\mu _m\), respectively.
This is consistent with the notion that fractional models have implications and the potential to be useful. The purpose of this work is to create a mathematical model that uses simulation to compare significant changes in the malaria disease analysis. Fractional-order derivatives will demonstrate how well the malaria system captures memory effects and non-local behaviors. The model incorporates a new fractal-fractional operator that improves accuracy, provides deeper insights into disease dynamics, and proposes new intervention techniques. Based on the aforementioned assumptions, below is the newly constructed malaria model with fractal-fractional derivative order:
Initial conditions are as follows:
The dynamical system is illustrated in Fig. 4.
The Schematic diagram illustrating the dynamical system.
The study of a malaria model with distinct infectious classes contributes to a better understanding of the impact of population migrations and local control efforts on global and regional malaria spread, allowing for the assessment of travel’s impact on disease persistence and evaluation of focused treatments. The values of all parameters were derived using data taken from the TRNC Ministry of Health’s website. In addition, some parameter values were determined using the formulations and calculations provided in45,46,47. All parameter values are given in Table 1. The flowchart of the proposed method in the model is given in Fig. 4.
Key features of the proposed model
Positivity
The fundamental analysis demonstrates the superiority of the proposed solutions in addressing real-world problems with constructive values, while this subsection explores the conditions that guarantee the model’s advantageousness.
Theorem 3.1
The proposed system’s solutions will remain positive in \(\mathbb {R}_+^8\) for \(t>0\) if the initial conditions satisfy the following.
Proof
We establish the norm:
then, we have
In case of fractal-fractional derivative44, we have
\(\square\)
Biological feasibility
Theorem 3.2
The model (6) is unique and limited in \(\mathbb {R}^8_{+}\), together with the initial conditions (7).
Proof
We get
Since \((S_{h_0},E_{h_0},I_{e_0},I_{{ne}_0},R_0,S_{m_0},E_{m_0},I_{m_0})\in \mathbb {R}_+^8\), (17) states that the solution is not allowed to leave the hyperplane. This proves that the domain of positive invariance is the set \(\mathbb {R}^8_+\). \(\square\)
Theorem 3.3
All solutions of the system (6) are bounded when the initial conditions (7) are positive.
Proof
Since, we have
So, we find
The comparison theorem gives us the following:
If \(N_h(0)<\frac{\Lambda +\Phi }{\mu _h}\), then
Consequently,
Additionally, the total mosquitoes population is
then
Therefore,
Hence, the system (6)’s solutions are limited to the biological feasible region \(\Upsilon\):
\(\square\)
Global derivative’s effect
Widely recognized in the literature, among the most frequently recurring integral is the Riemann-Stieltjes integral, of which the standard integral is a specific example. For
the Riemann-Stieltjes integral is
The global derivative of Z(t) in terms of y(t) is provided by
To examine if it has an impact on the malaria model (6), we are going to substitute out the global derivative with the classical derivative.
Here, we assume that y is differentiable. As a result, we have
Let
Examine the following system:
where \(\omega =(S_h,E_h,I_e,I_{ne},R,S_m,E_m,I_m)\). We will see from this example that the proposed system has a single solution.
To ensure the existence and uniqueness of our system, it is crucial to ensure the following:
-
1.
\(\big | \chi _i(t,\omega (t))\big |^2 ~ \le ~ K_i(1+ |\omega |^2)\), i=1,2,3,...,8.
-
2.
\(\forall\) \({\omega },\tilde{\omega }\), \({\parallel \chi _i(t,\omega (t))- \chi _i(t,\tilde{\omega }(t)) \parallel }_{\infty }^2 \ \le \ \overline{K}_i{\parallel \omega -\tilde{\omega } \parallel }_{\infty }^2\).
We have
under the condition \(\frac{2\big (\frac{(\beta _{h_1}+\beta _{h_2 } )^2|I_m|^2}{|N_h|^2}+\mu _h^2\big )}{\Lambda ^2+\Phi ^2+q^2 |R|^2}<1\), where \(K_1=2|y^\prime |^2(\Lambda ^2+\Phi ^2+q^2 |R|^2)\). Similarly we have a results for others compartments and this proves that function satisfies the linear growth condition. Next, we verify that the Lipschitz requirement is satisfied. Now let \(\xi _1=(t,S_{h_1},E_h,I_e,I_{ne},R,S_m,E_m,I_m)\) and \(\xi _2=(t,S_{h_2},E_h,I_e,I_{ne},R,S_m,E_m,I_m)\). We have
where \(\overline{K}_1=\Vert y^{\prime }\Vert ^2_{\infty }\big \{\frac{2(\beta _{h_1}+\beta _{h_2 })^2 \Vert I_m\Vert ^2_{\infty }}{\Vert N_h\Vert ^2_{\infty }}+2\mu _h^2\big \}\).
The system (6) has a unique solution under the following conditions for all compartments in general:
Qualitative analysis
Equilibrium points
In this portion of the article, we give a comprehensive review of equilibrium points. These equilibrium locations can only be obtained by setting the left side of Equation (6) to zero, as the following illustration indicates. According to the suggested model, the disease-free equilibrium point is
as well as the endemic equilibrium point is \(P_{2}(S_h^{*},E_h^{*},I_e^{*},I_{ne}^*, R^{*}, S_m^{*}, E_m^{*}, I_m^{*})\), where
Reproductive number and sensitivity of parameters
Understanding stability conditions is facilitated by the reproduction number \((R_0)\), which is an essential tool in epidemiological modeling. It is dictated by the vectors F and V, which stand for the origin of novel diseases and the dissemination of preexisting infections. We analyze these vectors at the disease-free equilibrium point \(D_1\). Consider the system
Therefore, we have
At \(P_{1}(S_h^0,~E_h^0,~I_e^0,~I_{ne}^0,~R^0,~S_m^0,~E_m^0,~I_m^0)\), we find the Jacobian matrices \((\textsf{F},\textsf{V})\) of F and V. And after that, we have
Given that the reproduction number \((R_0)\) of matrix \(\textsf{F} {\textsf{V}}^{-1}\) is the dominating eigenvalue, thus, we have
Sensitivity analysis is a technique used to assess a model’s resilience by looking at how variations in input parameters impact the model’s results. It works especially well with uncertain models and ambiguous data. It measures how inputs and outputs relate to one another, highlighting important factors that have a big influence on the model’s output. This aids researchers in identifying model limitations, honing important elements, and reaching well-informed conclusions. By computing the partial derivatives with respect to the pertinent parameters, we can examine the sensitivity of \(R_{0}\), it is evident that the value of \(R_{0}\) is quite sensitive when we change the settings. The parameters \(\alpha _3, \beta _{h_1}, \beta _{h_2}\) in our analysis show growth, whereas the parameters \(\alpha _1,\alpha _2,\mu _h,\mu _m\) show contraction. As a result, for efficient infection management, prevention should come before treatment. Figure 5 represents a graphic representation of the link between \({R}_0\) and the given parameters.
\({R}_{0}\)’s sensitivity to various parameters.
Stability analysis
Local stability
Theorem 6.1
When \(R_0\) is less than 1, the disease-free equilibrium point of the proposed malaria model is found to be locally asymptotically stable; however, when \(R_0\) is greater than 1, it becomes unstable.
Proof
To assess the stability of the model (6) at the disease-free equilibrium, assume the Jacobian matrix at \(P_{1}(S_h^0,~E_h^0,~I_e^0,~I_{ne}^0,~R^0,~S_m^0,~E_m^0,~I_m^0)\) is as follows:
where
Thus, the characteristics equation is
We can write
Upon solving the determinant matrix mentioned above, we obtain the following eigenvalues \((\lambda )\):
Since all of the matrix’s eigenvalues have negative real components, the system is locally asymptotically stable, which means that its solutions will eventually converge to malaria-free equilibrium point. \(\square\)
Global Stability Using Lyapunov for Endemic Equilibrium
Lemma 6.1
Let \(H \in \mathbb {R}^{+}\) be a continuous function such that for any \(t \ge t_0\)
Theorem 6.2
If the \(R_0>1\), the endemic equilibrium \(P_2\) of system (6) is globally asymptotically stable; otherwise, they are unstable.
Proof
Initially, we establish the Lyapunov function:
We have
We get
Now, using \(S_h=S_h-S_h^*\), \(E_h=E_h-E_h^*\), \(I_e=I_e-I_e^*\), \(I_{ne}=I_{ne}-I_{ne}^*\), \(R=R-R^*\), \(S_m=S_m-S_m^*\), \(E_m=E_m-E_m^*\), and \(I_m=I_m-I_m^*\). After rearranging and simplification the equality above can be written as
where
and
It is evident that in the case where \({\eta _1<\eta _2}\),
But, if \(S_h=S_h^*\), \(E_h=E_h^*\), \(I_e=I_e^*\), \(I_{ne}=I_{ne}^*\), \(R=R^*\), \(S_m=S_m^*\), \(E_m=E_m^*\), and \(I_m=I_m^*\), then
It is our understanding that the largest compact invariant set for the proposed model (6) in
is the endemic equilibrium \(D_2(S^*,E^*,I^*,Q^*,R^*)\) of the system (6). In the event that \({\eta _1<\eta _2}\), we can conclude that \(P_2(S_h^{*},E_h^{*},I_e^{*},I_{ne}^*, R^{*}, S_m^{*}, E_m^{*}, I_m^{*})\) is globally asymptotically stable in \(\Upsilon\). \(\square\)
Numerical solutions
The work proposes a numerical scheme for the suggested model (6) that used the fractal-fractional operator with the Mittag-Leffler kernel and procedure for evolution is shown in Fig. 6.
where \(\omega =(S_h,E_h,I_e,I_{ne},R,S_m,E_m,I_m)\). Let the abstract description of (49) be
where \(\chi =(\chi _1,\chi _2,\chi _3,\chi _4,\chi _5,\chi _6,\chi _7,\chi _8)\).
Following from integral (3), we have the Voltera formula:
Since, Newton polynomial can be written as:
After some calculations, we obtain
The flowchart of proposed method.
Simulation results
From the Figs. 1 and 2, it can be concluded that the number of malaria cases are in rise since 2020. Moreover, the vast majority of the obtained cases are imported malaria cases which came from the countries that has malaria as an endemic disease. A statistical study was conducted in China, in 2020, to indicate the importance of imported malaria cases and to emphasize the recent increase of cases in China. From the Figs. 2 and 3, it is obvious that most of the cases obtained in autumn months, especially in October. Human and mosquito population density, resource availability, infrastructure, and growth potential all have a major impact on the dynamics of malaria transmission in human society. The work uses numerical simulations under various fractional orders and fractal dimensions to show the malaria model’s accuracy and viability. The two-order fractal-fractional form operator is more helpful for data and practical difficulties, but the fractal-fractional malaria model is essential for understanding epidemiological model. Given the following initial conditions and parameter values listed in Table 1, the fractal-fractional malaria model is computed by numerical simulations.
To understand how parameters affect the results of state variables associated with malaria, we ran several simulations.
-
Figures 7, 8, 9, 10, 11, 12, 13 and 14 display our model’s graphical solution for all compartments at different fractional orders \((\sigma )\) and fractal dimensions \((\rho )\). The dynamics of human populations and mosquito vectors against different fractional orders are comprehensively illustrated in the paper.
-
The fractal dimension of 1.0 is used to simulate Figs. 7a, 8, 9, 10, 11, 12, 13 and 14a, whereas Figs. 7b, 8, 9, 10, 11, 12, 13 and 14b are simulated against fractal dimension of 0.9.
-
Figure 7 shows the mathematical simulation for susceptible human populations versus different fractional orders. while the rate of infection declines, the susceptible human population first rises and then falls at higher values of \(\sigma\), suggesting that while the rate of infection declines, the number of susceptible individuals naturally rises.
-
Figures 8, 9 and 10 illustrate how the infection against greater fractional orders is spreading concurrently because susceptible persons are spreading the infection to exposed and affected classes.
-
Figure 11 illustrates the fast expanding recovered class in comparison to larger fractional orders that indicate the infected individuals’ recovery rate.
-
The graphs in Figs. 12, 13 and 14 show how mosquitoes grow into three different classes, each of which shows a positive relationship with fractional order. At higher values of \(\sigma\), the population of mosquito vectors increases quickly in all classes.
The fractal-fractional order model offers a wide range of calibrations for varying levels of infectivity in populations of mosquitoes and humans. The study discovered that an increase in oscillations causes the memory effects of infected vectors and humans to rise as the derivative order \((\sigma )\) approaches 1. Furthermore, an apparent change in the dynamics of different compartments occurs with a decrease in the fractal dimension \((\rho )\). The figures demonstrate how, within the constrained domain, the solution approaches a steady state as the value of \(\sigma\) declines. To get to this point, each graph travels a non-linear path, emphasizing the significance of fractional order. Greater \(\sigma\) values correspond to a faster rate of convergence. The fractal dimension \(\rho\)’s self-similar pattern and declining amplitudes suggest that the malaria system’s paths are moving in the direction of stability. Researchers can find trends and patterns that can direct population management and sustainable development strategies by analyzing the impact of different parametric values on population density.
Dynamics and forecasting \(S_h(t)\) at higher quartile coefficient values index.
Dynamics and forecasting \(E_h(t)\) at higher quartile coefficient values index.
Dynamics and forecasting \(I_e(t)\) at higher quartile coefficient values index.
Dynamics and forecasting \(I_{ne}(t)\) at higher quartile coefficient values index.
Dynamics and forecasting R(t) at higher quartile coefficient values index.
Dynamics and forecasting \(S_m(t)\) at higher quartile coefficient values index.
Dynamics and forecasting \(E_m(t)\) at higher quartile coefficient values index.
Dynamics and forecasting \(I_m(t)\) at higher quartile coefficient values index.
Conclusion
To better understand malaria transmission dynamics in Northern Cyprus, researchers used a deterministic eight compartmental mathematical model. The model reveals distinct social dynamics and an extended incubation period, with reproductive number serving as the primary metric. To better understand the model’s behavior under constrained conditions, sensitivity and stability analyses were performed. Fractal geometry and fractional calculus were employed to improve forecasting accuracy and comprehension of population dynamics. The study employed a Newton polynomial numerical approach to obtain solutions and runs simulations to examine the global impact of parameters on malaria virus symptomatic and asymptomatic rates. Fractal geometry and fractional calculus were employed to improve forecasting accuracy and comprehension of population dynamics. Human and mosquito growth rates are proportional to the fractional order \((\sigma )\), with the memory effect increasing as the derivative order decreases from 1. This research is critical for comprehending viral transmission dynamics and developing effective control measures. The study underscores the significance of control strategies for imported malaria cases during entry into a country, suggesting that policymakers should implement awareness-raising measures, test applications during entry, and vaccination programs to combat the endemic of malaria. The uncertainty of Cyprus’ epidemiological status, particularly in the north, makes it difficult to establish a baseline and reliable historical data for a malaria model. Most cases are imported malaria cases from endemic countries, with diagnosis dates typically falling in October, coinciding with university openings. The model’s accuracy is based on precise, region-specific data on human, vector, and disease prevalence, which may be scarce in Northern Cyprus. Complex models are required to account for unique factors such as tourism-induced vector introduction and diverse immune status, but overcomplicated models can be overly parameterized and difficult to analyze. The model must account for local conditions such as climate, water bodies, and human behavior, which can be difficult to generalize from models developed for other regions. Limited or noisy data can make it difficult to identify specific parameter values in models, affecting both analysis and prediction. To overcome over-parameterization and improve parameter estimation, model reduction techniques may be required, which simplify complex models while preserving essential transmission dynamics. To improve this model, localized data such as climate, mosquito density, and treatment-seeking behaviors should be included. Complex epidemiological factors such as spatial heterogeneity, treatment efficacy, and traditional medicine should be addressed. Future research should employ numerical optimization approaches and algorithms, such as non-linear least squares curve fitting or machine learning methods, to reduce the discrepancy between anticipated epidemic curves and real-world data while also estimating unknown parameters and fractional orders. Experimenting with other fractional derivatives and numerical methods can help identify transmission drivers and assess intervention strategies.
Data availability
The data for this study are available from the corresponding author upon a reasonable request. All data generated or analysed during this study are included in this article.
References
Collins, O. C. & Duffy, K. J. A mathematical model for the dynamics and control of malaria in Nigeria. Infect. Dis. Modell. 7(4), 728–741 (2022).
Lazrek, Y. et al. Molecular detection of human Plasmodium species using a multiplex real time PCR. Sci. Rep. 13(1), 11388 (2023).
Güler, Emrah, Hürdoganoglu, Ulas & Süer, Kaya. An imported malaria case associated with pregnancy in northern cyprus. Türk Mikrobiyoloji Cemiyeti Dergisi 53(2), 138–142 (2023).
World Health Organization. Malaria. World Health Organization. https://www.who.int/news-room/fact-sheets/detail/malaria (2024).
Dao, F. et al. The prevalence of human Plasmodium species during peak transmission seasons from 2016 to 2021 in the rural commune of Ntjiba. Mali. Trop. Med. Infect. Dis. 8(9), 438 (2023).
González-Sanz, M., Berzosa, P. & Norman, F. F. Updates on malaria epidemiology and prevention strategies. Curr. Infect. Dis. Rep. 25(7), 131–139 (2023).
Surveillance Atlas of Infectious Diseases. Europa.eu. https://atlas.ecdc.europa.eu/public/indexaspx?Dataset=27 (2025).
Norman, F., et al. (2022). Trends in imported malaria during the COVID-19 pandemic, Spain (+Redivi Collaborative Network). J. Travel Med. 29(6).
Choy, B., Bristowe, H., Khozoee, B. & Lampejo, T. Increased imported severe Plasmodium falciparum malaria involving hyperparasitaemia \((> 10\%)\) in a UK hospital following relaxation of COVID-19 restrictions compared to the pre-pandemic period. J. Travel Med. 29(8), taac116 (2022).
Hussien, H. H. COVID-19 and malaria: A comparative study of epidemiology, burden, and challenges. Adv. Infect. Dis. 12(4), 758–775 (2022).
Senol, C. The Turkish Republic of Northern Cyprus hydrographic structure, water problem and solution suggestions. J. Cyprus Stud. 21(45), 77–98 (2020).
An A. Valuable Persons Raised in Cyprus (1782-1899), 1st ed. 17.07.2002; s.416-420, Akçaǧ publications. Ankara.
An, A. et al. (eds) The Cyprus Turkish Medical Association Publication (Nicosia, Cyprus, April 2014).
Güler, E., Güvenir, M. & Süer, K. The amazing eradication story and current situation of malaria in Cyprus. Parasitol. United J. 15(1), 1–4 (2022).
Statistical Information, Notifiable Diseases. Gov.ct.tr. https://www.saglik.gov.ct.tr/online-hizmetler/istatistikibilgiler/ihbari-zorunlu-hastaliklar-istatistikleri (2025).
Malaria Annual Epidemiological Report for 2020 Key facts. (n.d.). https://www.ecdc.europa.eu/sites/default/files/documents/AER-malaria-2020.pdf.
Ahmad, A. et al. Flip bifurcation analysis and mathematical modeling of cholera disease by taking control measures. Sci. Rep. 14(1), 10927 (2024).
Farman, M., Akg, I. A., Ahmad, A. & Baleanu, D. Dynamical transmission of coronavirus model with analysis and simulation. Comput. Model. Eng. Sci. 127(2), 753–769 (2021).
Tabassum, M. F., Saeed, M., Akgül, A., Farman, M. & Chaudhry, N. A. Treatment of HIV/AIDS epidemic model with vertical transmission by using evolutionary Pad -approximation. Chaos, Solitons Fractals 134, 109686 (2020).
James Peter, O., Ojo, M. M., Viriyapong, R. & Abiodun Oguntolu, F. Mathematical model of measles transmission dynamics using real data from Nigeria. J. Differ. Equ. Appl. 28(6), 753–770 (2022).
Batool, M., Farman, M., Ahmad, A. & Nisar, K. S. Mathematical study of polycystic ovarian syndrome disease including medication treatment mechanism for infertility in women. AIMS Public Health 11(1), 19 (2024).
Gweryina, R. I., Madubueze, C. E., Bajiya, V. P. & Esla, F. E. Modeling and analysis of tuberculosis and pneumonia co-infection dynamics with cost-effective strategies. Results Control Optim. 10, 100210 (2023).
Kulachi, M. O., Ahmad, A., Hincal, E., Ali, A. H., Farman, M., & Taimoor, M. Control of conjunctivitis virus with and without treatment measures: A bifurcation analysis. J. King Saud Univ.-Sci. 103273 (2024).
Al Basir, F. & Abraha, T. Mathematical modelling and optimal control of malaria using awareness-based interventions. Mathematics 11(7), 1687 (2023).
Ndii, M. Z. & Adi, Y. A. Understanding the effects of individual awareness and vector controls on malaria transmission dynamics using multiple optimal control. Chaos, Solitons Fractals 153, 111476 (2021).
Basir, F. A., Banerjee, A. & Ray, S. Exploring the effects of awareness and time delay in controlling malaria disease propagation. Int. J. Nonlinear Sci. Numer. Simul. 22(6), 665–683 (2021).
Ibrahim, M. M., Kamran, M. A., Naeem Mannan, M. M., Kim, S. & Jung, I. H. Impact of awareness to control malaria disease: A mathematical modeling approach. Complexity 2020(1), 8657410 (2020).
Nisar, K. S., Farman, M., Abdel-Aty, M. & Ravichandran, C. A review of fractional order epidemic models for life sciences problems: Past, present and future. Alex. Eng. J. 95, 283–305 (2024).
Yunus, A. O., Olayiwola, M. O. & Ajileye, A. M. A fractional mathematical model for controlling and understanding transmission dynamics in computer virus management systems. Jambura J. Biomath. (JJBM) 5(2), 116–131 (2024).
Yunus, A. O., & Olayiwola, M. O. A mathematical model for assessing vaccination’s efficacy as a preventative strategy against newly emerging COVID-19 variants. Vacunas 500392 (2025).
Zheng, Z. et al. A new fractional-order model for time-dependent damage of rock under true triaxial stresses. Int. J. Damage Mech. 32(1), 50–72 (2023).
Farman, M. et al. Generalized Ulam-Hyers-Rassias stability and novel sustainable techniques for dynamical analysis of global warming impact on ecosystem. Sci. Rep. 13(1), 22441 (2023).
Grabowski, D., Jakubowska-Ciszek, A. & Klimas, M. Fractional-order model of electric arc furnace. IEEE Trans. Power Deliv. 38(6), 3761–3770 (2023).
Naik, P. A. et al. Forecasting and dynamical modeling of reversible enzymatic reactions with a hybrid proportional fractional derivative. Front. Phys. 11, 1307307 (2024).
Olayiwola, M. O., Yunus, A. O., Ismaila, A., Alaje, & Adedeji, J. A. Modeling within-host Chikungunya virus dynamics with the immune system using semi-analytical approaches. BMC Res. Notes 18(1), 201 (2025).
Yunus, A. O. & Olayiwola, M. O. Mathematical modeling of malaria epidemic dynamics with enlightenment and therapy intervention using the Laplace-Adomian decomposition method and Caputo fractional order. Frank. Open 8, 100147 (2024).
Yunus, A. O. & Olayiwola, M. O. Dynamics of Ebola virus transmission with vaccination control using Caputo-Fabrizio Fractional-order derivative analysis. Model. Earth Syst. Environ. 11(3), 1–18 (2025).
Yunus, A. O. & Olayiwola, M. O. Simulation of a novel approach in measles disease dynamics models to predict the impact of vaccinations on eradication and control. Vacunas (English Edition) 26(2), 100385 (2025).
Atangana, A. Fractal-fractional differentiation and integration: connecting fractal calculus and fractional calculus to predict complex system. Chaos, Solitons Fractals 102, 396–406 (2017).
Naik, P. A., Farman, M., Zehra, A., Nisar, K. S. & Hincal, E. Analysis and modeling with fractal-fractional operator for an epidemic model with reference to COVID-19 modeling. Part. Differ. Equ. Appl. Math. 10, 100663 (2024).
Partohaghighi, M., Mortezaee, M., Akgül, A., Hassan, A. M. & Sakar, N. Numerical analysis of the fractal-fractional diffusion model of ignition in the combustion process. Alex. Eng. J. 86, 1–8 (2024).
Akgül, A. & Conejero, J. A. Fractal Fractional Derivative Models for Simulating Chemical Degradation in a Bioreactor. Axioms 13(3), 151 (2024).
Farman, M., Shehzad, A., Nisar, K. S., Hincal, E. & Akgül, A. A mathematical fractal-fractional model to control tuberculosis prevalence with sensitivity, stability, and simulation under feasible circumstances. Comput. Biol. Med. 178, 108756 (2024).
Atangana, A. Mathematical model of survival of fractional calculus, critics and their impact: How singular is our world? Adv. Differ. Equ. 2021(1), 403 (2021).
Wu, H. & Hu, Z. Malaria Transmission Model with Transmission-Blocking Drugs and a Time Delay. Math. Probl. Eng. 2021, 1–17 (2021).
Collins, O. C. & Duffy, K. J. A mathematical model for the dynamics and control of malaria in Nigeria. Infect. Dis. Modell. 7(4), 728–741 (2022).
Herdicho, F. F., Chukwu, W. & Tasman, H. An optimal control of malaria transmission model with mosquito seasonal factor. Results Phys. 25, 104238 (2021).
Funding
The authors received no financial support for the research and publication of this article.
Author information
Authors and Affiliations
Contributions
The contributions of each author to this research are as follows: M.F., N.G: Conceptualization, Methodology, Formal Analysis, Writing-Original Draft, Writing-Review and Editing, Visualization and Numerical Analysis. U.H.: Conceptualization, Methodology, Investigation, Writing-Original Draft, Writing-Review and Editing, Visualization. N.H: Conceptualization, Investigation, Writing-Original Draft, Writing-Review and Editing, Visualization. E.G, A.S.: Conceptualization, Investigation, Writing-Review and Editing, Visualization. E.H., A.S.: Methodology, Formal Analysis, Investigation, Writing-Review and Editing, Visualization. K.S.: Methodology, Investigation, Writing-Review and Editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Farman, M., Hurdoganoglu, U., Hincal, N. et al. Fractal fractional approach to investigate the forecasting and dynamics of malaria disease for early control precaution. Sci Rep 15, 42879 (2025). https://doi.org/10.1038/s41598-025-27152-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27152-0