Abstract
The interaction between cocoa clones and post-harvest practices, such as fermentation and non-fermentation of beans, influences cocoa quality in the Amazon. However, these effects remain poorly understood. This study evaluated the effects of fermentation and non-fermentation on the biochemical and nutritional attributes of beans from different clones and on plant productivity in the southwestern Brazilian Amazon. Nine cocoa clones cultivated in Rondônia were analyzed under two post-harvest conditions (fermented and non-fermented). The following parameters were assessed: N, P, K, S, Ca, Mg, B, Cu, Fe, Mn, Zn, total phenolic compounds, tannins, anthocyanins, and the metabolites proline, glycine betaine (GB), malondialdehyde (MDA), and superoxide dismutase (SOD) activity. In general, fermented beans showed higher levels of amino acids, SOD, K, Mg, GB, Zn, and Fe, while non-fermented beans exhibited higher contents of sugars, phenolic compounds, tannins, proteins, MDA, and anthocyanins. Clone CCN 51 showed a balanced biochemical profile under both post-harvest conditions, while EEOP 63 combined high productivity with strong functional performance. These findings suggest that selecting appropriate clones for fermented and non-fermented systems can improve cocoa bean quality in the Amazon, aligning productivity, health-related traits, and innovation in the fine chocolate market.
Similar content being viewed by others
Introduction
Cocoa (Theobroma cacao L.) is one of the most socioeconomically important tropical crops worldwide, primarily driven by the food industry for chocolate and its derivatives1.
Although Brazil represents a modest share of global cocoa production, it plays a strategic role as the only major producer cultivating cocoa at scale within the Amazon biome2. This confers unique importance to Brazilian cocoa, given the genetic diversity conserved in the region and its integration into agroforestry and sustainable production systems. Within the Amazon, Rondônia stands out as the third largest cocoa producer in the country. Production reached five thousand tons in 2023, representing a 29.7% increase compared with the previous year3. This growth is expected to persist in the coming years, consolidating Rondônia as an emerging cocoa-producing area in the southwestern Amazon, where cocoa increasingly shares space with coffee, the state’s other major perennial crop.
The quality of cocoa beans is influenced by a combination of genetic, environmental, and processing factors, among which post-harvest fermentation plays a central role in shaping the characteristic sensory profile of the cocoa beans4. During fermentation, yeasts, lactic acid bacteria, and acetic acid bacteria act sequentially and synergistically as the main microorganisms responsible for mucilage degradation, production of ethanol, lactic acid, and acetic acid, and for driving the principal physical and biochemical transformations in cocoa beans1,5.
These microbial and enzymatic activities promote the formation of aroma, flavor, and color precursors6, while also reducing compounds responsible for astringency and bitterness7. In addition to its sensory and functional contributions, fermentation imposes moderate metabolic stress on the beans, triggering biochemical responses associated with the accumulation of osmoprotective and antioxidant compounds, such as proline, glycine betaine (GB), and the enzyme superoxide dismutase (SOD)8,9.
Although fermentation enhances key sensory attributes for chocolate production, it also leads to the degradation of sugars and proteins, as well as the oxidation of phenolic compounds, resulting in a significant loss of natural antioxidants7,10. In this context, alternative approaches involving the use of non-fermented or minimally processed beans have gained interest, aiming to preserve sensitive bioactive compounds, such as phenolics and proteins, and to enhance the functional value of cocoa7,11,12, while promoting the development of health-beneficial specialty products7,12.
Reports suggest that combining beans at different fermentation levels may offer new possibilities for flavor and functionality, similarly, to blending practices11. Most studies evaluating the effects of post-harvest processing, with or without fermentation, have focused on sensory quality, including aroma, flavor, acidity, bitterness, and astringency13, with few assessing the contents of mineral elements, bioactives, and oxidative metabolites. This is relevant because fermentation processes, depending on the genotype, can influence nutrient mobilization, degradation of phenolic compounds, and activation of oxidative defense mechanisms14,15,16. It is possible that the fermentation process, depending on the levels of specific mineral elements, contributes to biochemical pathways related to oxidative metabolism, including proline, GB, SOD, and malondialdehyde (MDA), which may reflect in the functional quality of the beans.
Among cellular damage indicators, MDA is widely used to determine lipid peroxidation. While its application is well established in biomedical studies on cocoa consumption17,18, MDA quantification during post-harvest processing of beans remains poorly explored and may provide important insights into grain physiological integrity and biochemical mechanisms activated in response to oxidative stress.
We hypothesize that certain clones may be better adapted to fermented or non-fermented post-harvest systems, resulting in improved bean quality and higher productivity. Therefore, this study aimed to evaluate the effects of fermentation and non-fermentation on the biochemical, nutritional, and antioxidant attributes of cocoa beans from different clones, as well as on plant productivity in the Amazon. If confirmed, this hypothesis will enable the identification of the most suitable clone for cultivation in Rondônia under different post-harvest systems, providing tailored solutions for the food industry. Selecting the optimal clone can enhance raw material quality without additional costs and strengthen the cocoa production chain in the Amazon region.
Material and methods
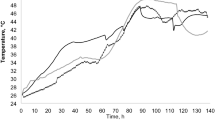
This study was conducted using different genetic materials of cocoa cultivated in Acrisols at the Frederico Afonso Experimental Station of the Executive Commission for the Cocoa Crop Plan (CEPLAC) (10°42′30″S, 62°13′30″W), located in Ouro Preto do Oeste, Brazil. The regional climate is classified as Am according to Köppen, with an average annual temperature of 25.6 °C, average relative humidity of 89%, and annual rainfall exceeding 2200 mm19. Meteorological data during the cultivation and post-harvest evaluation period were obtained from the National Institute of Meteorology20 (Fig. 1).
Daily variation in air temperature, relative humidity, and rainfall in eastern Rondônia, Brazil.
During the harvest and processing of fruits in the experimental area, the average daily temperature was 26.6 ± 4.3 °C, with maximums of 35.0 ± 4.8 °C and minimums of 19.6 ± 3.7 °C, and average relative humidity was 53.4 ± 5.5%20 (Fig. 1). From July 2 to August 10, 2024, no rainfall was recorded. The year 2024 was atypical, with an extended dry season exceeding the historical average.
Study site and experimental design
The study was conducted in an eight-year-old cocoa plantation with 3 m × 3 m spacing, resulting in a planting density of 1111 trees per hectare. Each experimental unit consisted of nine cocoa trees. Treatments included nine cocoa clones combined with two post-harvest processes (fermented and non-fermented) arranged in a randomized block design with three replicates.
The study was conducted using nine cocoa clones representing four genetic groups: Bahia Norte (BN 34), Castro Naranjal Collection (CCN 51), Cocoa Research Center (CEPEC 2005), and Ouro Preto Experimental Station (EEOP 33, EEOP 63, EEOP 65, EEOP 69, EEOP 80, EEOP 96). For consistency, CEPEC 2005 is referred to as CEPEC 5 throughout the text and figures. All clones are listed in the International Cocoa Germplasm Database – ICGD21, except for BN 34, which was not found in the database.
The CCN 51 was developed by Homero Castro in Ecuador through the cross (ICS 95 × IMC 67) × CCN 122. CEPEC 2005 originated from a Catongo × VB 276 cross, developed by CEPEC/CEPLAC (Bahia, Brazil)23. BN 34 is a hybrid genotype selected in southern Bahia24. EEOP clones were developed by CEPLAC/RO as part of a regional breeding initiative in the southwestern Amazon. EEOP 33 resulted from a cross between POUND 7 and BE 1025. The remaining clones (EEOP 63, 65, 69, 80, and 96) were selected from improved seed material from Amazonian plantations, as described26,27. No voucher specimens of the plant material were found in publicly available herbaria. However, all clones used in this study are conserved ex situ in CEPLAC’s living germplasm collections, both at their respective institutions of origin and at the experimental station where the present study was conducted26.
Compliance with Brazilian biodiversity legislation. The cocoa clones investigated in this study (BN 34, CCN 51, CEPEC 5, and the EEOP series) are established improved genotypes maintained in ex situ CEPLAC germplasm collections and/or registered in the International Cocoa Germplasm Database (ICGD). No in situ collection of wild/native material was carried out. Accordingly, this work does not constitute “access to Brazilian genetic heritage” as defined by Law 13.123/2015 and Decree 8.772/2016, and therefore SISGEN registration is not applicable.
Sample collection and processing
Harvests were carried out on July 2 and July 30, 2024. Healthy, damage-free fruits were individually analyzed to estimate seed mass with mucilage per hectare (kg ha⁻1). Fresh seed samples were divided: one half underwent only pre-drying and was classified as non-fermented (NF), while the other underwent fermentation and was classified as fermented (F).
Fermentation and drying
The fermentation was performed in wooden boxes with drainage and aeration28. The mass was covered with banana leaves to prevent dehydration. Fermentation lasted 5 to 7 days, with mixing for homogenization: the first after 24 h, the second after 48 h, and two additional mixings every 24 h. After fermentation, beans were pre-dried in a closed-circulation air oven at ambient temperature (19.6 ± 3.7 °C). Non-fermented beans underwent the same pre-drying. All samples were then dried in a forced-air oven at 60 °C until its constant weight. This drying temperature was maintained to preserve sensory quality and prevent excessive acid retention and bitterness in the beans29.
After drying, cocoa beans were processed differently depending on the type of analysis. For the determination of macro e micronutrients, sugars, amino acids, lipids, total phenolics, and tannins, the shells were not removed and whole beans (nibs plus shells) were ground together. For the analyses of protein, malondialdehyde, superoxide dismutase, proline, glycine betaine, and anthocyanins, the shells were removed in both fermented and non-fermented samples, and only the nibs (inner part of the beans) were used.
Biochemical and nutritional analysis
The contents of N, P, K, Ca, Mg, B, Cu, Fe, Mn, and Zn were determined30. Total sugars31, amino acids32, and tannins, using gallic acid as standard33, were also quantified. Total phenolics were measured following established protocols34, with absorbance readings performed as described in35. Lipid content was determined according to official procedures36. The levels of proline37, malondialdehyde (MDA)38, glycine betaine (GB)39, anthocyanins40, and superoxide dismutase (SOD) activity41 were evaluated. Soluble proteins used to calculate SOD specific activity were quantified using the Bradford method42.
Statistical analysis
Statistical analysis included ANOVA, and means were compared using Tukey’s test at a 5% significance level, using Python. Principal Component Analysis (PCA) was conducted to identify variability patterns among cocoa clones based on biochemical and nutritional composition. A general PCA was first applied to all samples to assess separation between fermented (F) and non-fermented (NF) groups. Then, separate PCAs were conducted for each group using the mean of replicates per clone to evaluate variable associations in each processing context. The loading matrix was used to identify the variables contributing most to the principal components. All analyses were performed in Python using scikit-learn, matplotlib, pandas, and seaborn libraries.
Pearson’s correlation analysis was performed between biochemical, nutritional, and productivity variables, separately for F and NF groups. Associations were considered relevant when the correlation coefficient (r) was ≥ 0.50 or ≤ –0.50. Analyses were conducted in Python using pandas and scipy.stats libraries.
This manuscript benefited from English language refinement using a large language model (ChatGPT, OpenAI), under the supervision of the authors. The model was used to improve clarity and grammar, without altering the scientific content.
This research was not conducted with endangered species and all methods in this study were carried out in compliance/accordance with relevant institutional, national, and international guidelines and legislation.
Results
The clone EEOP 63 showed the highest seed mass and differed significantly only from EEOP 69 (Fig. 2).
Effects of nine cocoa clones on seed mass per hectare. Different letters indicate significant differences according to Tukey’s test (P < 0.05).
The contents of N and Zn in cocoa beans were not affected by the evaluated factors. For N, there was no significant effect of clone (F = 0.57; p > 0.05), fermentation process (F = 0.10; p > 0.05), or their interaction (F = 1.53; p > 0.05). A similar pattern was observed for Zn, with no significant effect of clone (F = 1.03; p > 0.05), fermentation (F = 0.20; p > 0.05), or interaction (F = 0.94; p > 0.05).
The fermentation process significantly affected the nutrient contents in cocoa beans, except for S, Cu, and Fe (Fig. 3). The effect of clone was also significant, influencing all evaluated nutrients except for N and Zn. The interaction between fermentation and clone was significant only for P and S, indicating that the effect of fermentation on these nutrients varied among genotypes.
Effects of nine cocoa clones (C) and the fermentation process (PF), fermented (F) and non-fermented (NF), on the mean contents of phosphorus (a), potassium (b), calcium (c), magnesium (d), sulfur (e), boron (f), copper (g), manganese (h), and iron (i) in cocoa beans. Uppercase letters compare fermentation levels (F vs. NF) within each clone; lowercase letters compare clones within each fermentation level, according to Tukey’s test (p < 0.05). * = significant, ns = not significant according to the F test (p < 0.05).
Phosphorus content in cocoa beans was influenced by both main factors and their interaction. Higher P levels were observed in non-fermented beans of clone EEOP 63, which did not differ from the other clones, except CCN 51, CEPEC 5, EEOP 33, and EEOP 96. Among fermented beans, higher P content was observed in clone EEOP 69, which differed only from CCN 51, EEOP 65, and EEOP 96. In general, non-fermented beans showed higher P contents than fermented beans, particularly in clones BN 34, EEOP 63, EEOP 65, and EEOP 80.
Potassium content in cocoa beans was affected by fermentation and clone, without significant interaction between these factors (Fig. 3b). Among non-fermented beans, K contents were similar across clones. Among fermented beans, the highest K content was observed in clone CEPEC 5, which differed only from EEOP 33. Fermented beans exhibited higher K levels compared to non-fermented ones, with significant differences in clones CCN 51, CEPEC 5, EEOP 65, and EEOP 96.
Calcium content was influenced by fermentation and clone, also without significant interaction (Fig. 3c). Among fermented beans, higher Ca levels were found in EEOP 69, CEPEC 5, and EEOP 65 compared to CCN 51. In non-fermented beans, EEOP 69 showed the highest Ca content, differing only from CCN 51. Non-fermented beans had higher Ca contents than fermented beans, especially BN 34 and EEOP 33.
Magnesium content was significantly affected by fermentation and clone, with no interaction (Fig. 3d). In non-fermented beans, Mg levels were similar among clones. In fermented beans, the highest content was found in CEPEC 5, differing only from EEOP 96. Fermented beans had higher Mg content than non-fermented ones, with significant differences in BN 34, CCN 51, CEPEC 5, and EEOP 69.
Sulfur content was influenced by clone and the interaction between factors, but not by fermentation alone (Fig. 3e). Among non-fermented beans, S levels were similar across clones. In fermented beans, the highest S content was found in EEOP 80, differing only from CCN 51 and EEOP 96. Non-fermented beans showed higher S content than fermented ones, especially in CCN 51 and EEOP 96.
Boron content was influenced by both fermentation and clone, without significant interaction (Fig. 3f). In non-fermented beans, the highest B content was observed in CEPEC 5, which differed only from EEOP 63 and EEOP 80. Among fermented beans, B content was similar across clones. Non-fermented beans showed higher B levels than fermented ones, especially in BN 34, CCN 51, CEPEC 5, and EEOP 96.
Copper content was affected only by clone, with no effect from fermentation or the interaction (Fig. 3g). In non-fermented beans, the highest Cu levels were found in EEOP 80, CEPEC 5, and EEOP 63, differing only from EEOP 65, BN 34, and EEOP 96. In fermented beans, CEPEC 5 showed the highest Cu content, differing only from EEOP 96, EEOP 65, and EEOP 33.
Manganese content was affected by fermentation and clone, without interaction (Fig. 3h). Among non-fermented beans, EEOP 69 showed the highest Mn content, differing only from EEOP 96, EEOP 65, EEOP 33, and CCN 51. Among fermented beans, the highest Mn contents were observed in EEOP 69 and CEPEC 5, followed by EEOP 80, with the two former differing from CCN 51 and EEOP 33. Non-fermented beans had higher Mn content than fermented ones, with significant differences in BN 34, CCN 51, EEOP 33, EEOP 63, and EEOP 65.
Iron content was influenced only by clone, with no effect of fermentation or interaction (Fig. 3i). Among non-fermented beans, Fe content was similar across clones. In fermented beans, the highest Fe content was observed in CEPEC 5, differing only from CCN 51 and EEOP 33.
Sugar content in cocoa beans was influenced by fermentation and the interaction between factors (Fig. 4a). In non-fermented beans, sugar content was similar across clones. Among fermented beans, the highest sugar contents were observed in EEOP 65 and EEOP 96, differing only from BN 34 and EEOP 63. Non-fermented beans showed higher sugar content than fermented ones, with significant differences in all evaluated clones.
Effects of nine cocoa clones (C) and the fermentation process (PF)—fermented (F) and non-fermented (NF)—on the mean contents of sugars (a), amino acids (b), lipids (c), total phenolics (d), tannins (e), and anthocyanins (f) in cocoa beans. Uppercase letters compare fermentation levels (F vs. NF) within each clone; lowercase letters compare clones within each fermentation level, according to Tukey’s test (p < 0.05). * = significant; ns = not significant (F test, p < 0.05).
Amino acid content in cocoa beans was influenced by fermentation, clone, and their interaction (Fig. 4b). Among non-fermented beans, the highest amino acid content was observed in clone CCN 51, which differed from EEOP 63, EEOP 65, EEOP 69, and EEOP 80. In fermented beans, CCN 51 also showed the highest amino acid content, while the lowest levels were found in EEOP 69, CEPEC 5, EEOP 33, BN 34, and EEOP 63. Non-fermented beans exhibited higher amino acid levels than fermented beans, particularly in CCN 51, EEOP 65, EEOP 69, EEOP 80, and EEOP 96.
Lipid content in cocoa beans was influenced only by clone, with no effect of fermentation or interaction between the factors (Fig. 4c). In both fermented and non-fermented beans, CEPEC 5 showed the highest lipid content, differing from all other genotypes. Lipid levels remained stable between the two post-harvest processes.
Total phenolic content in cocoa beans was influenced by fermentation, clone, and their interaction (Fig. 4d). In non-fermented beans, the highest phenolic content was observed in EEOP 96, which differed only from BN 34. Among fermented beans, CCN 51 showed the highest phenolic content, while EEOP 65 and EEOP 96 had the lowest values. Non-fermented beans exhibited higher phenolic content than fermented beans, with significant differences across all clones.
Tannin content in cocoa beans was influenced by fermentation and the interaction between factors, with no effect of clone (Fig. 4e). Tannin levels were similar among clones within both fermentation groups. Non-fermented beans had higher tannin content than fermented beans, with significant differences in all clones except EEOP 33, EEOP 63, and EEOP 65.
Anthocyanin content in cocoa beans was influenced by fermentation, clone, and their interaction (Fig. 4g). Among non-fermented beans, the highest content was observed in EEOP 65, which differed from CCN 51 and CEPEC 5. In fermented beans, the highest anthocyanin content was found in EEOP 96, which differed from BN 34. Non-fermented beans had higher anthocyanin levels than fermented ones, except in EEOP 33 and EEOP 65, which showed no significant difference between treatments.
Protein content in cocoa beans was influenced by fermentation, clone, and their interaction (Fig. 5a). Among non-fermented beans, the highest protein content was observed in EEOP 33, which differed only from EEOP 63. Non-fermented beans had higher protein content than fermented ones, with significant differences across all clones. In fermented beans, EEOP 80 showed the highest protein content, differing only from EEOP 65.
Effects of nine cocoa clones (C) and the fermentation process (PF)—fermented (F) and non-fermented (NF)—on the mean contents of protein (a), SOD (b), MDA (c), proline (d), and glycine betaine (e) in cocoa beans. Uppercase letters compare fermentation levels (F vs. NF) within each clone; lowercase letters compare clones within each fermentation level, according to Tukey’s test (p < 0.05). * = significant; ns = not significant (F test, p < 0.05).
SOD enzyme activity in cocoa beans was affected by fermentation, clone, and their interaction (Fig. 5b). In non-fermented beans, SOD activity was similar among clones. In fermented beans, the highest SOD activity was observed in EEOP 65, which differed only from EEOP 80. Fermented beans showed higher SOD activity compared to non-fermented ones, except for clones EEOP 69, EEOP 80, and EEOP 96, where no significant differences were found between treatments.
MDA content in cocoa beans was influenced by fermentation, clone, and their interaction (Fig. 5c). Among non-fermented beans, the highest MDA content was observed in EEOP 63, which differed only from CEPEC 5. In fermented beans, MDA levels were similar across clones. Fermented beans showed lower MDA levels than non-fermented ones, with significant differences across all clones.
Proline content in cocoa beans was influenced by the interaction between factors, with no isolated effect of fermentation (Fig. 5d). In non-fermented beans, proline levels were similar across clones. In fermented beans, the highest proline content was observed in CCN 51, differing only from EEOP 96. Fermented beans had higher proline levels than non-fermented ones in clones CEPEC 5, EEOP 63, and EEOP 69. On the other hand, non-fermented beans showed higher proline content only in EEOP 65. No differences between fermentation treatments were observed for the remaining clones.
Glycine betaine content in cocoa beans was influenced by fermentation, clone, and their interaction (Fig. 5e). In non-fermented beans, the highest GB content was found in CCN 51, differing only from EEOP 80, EEOP 63, EEOP 69, and EEOP 65. In fermented beans, the highest GB contents were observed in CCN 51 and CEPEC 5, with a notable distinction for CCN 51, which differed from all other clones. Fermented beans had higher GB content in CCN 51, CEPEC 5, and EEOP 33 compared to the remaining clones.
Principal Component Analysis (PCA) revealed the multivariate variation pattern among cocoa clones in fermented (F) and non-fermented (NF) samples. The first two components explained 57.0% of the total variance, with 40.1% attributed to PC1 and 16.9% to PC2 (Fig. 6). The biplot showed clear separation between treatments along the PC1 axis, with fermented clones predominantly located in the right quadrant and non-fermented clones on the left, indicating biochemical differentiation between groups. Traits such as SOD, amino acids, proline, glycine betaine (GB), Zn, and seed mass were positively associated with fermented clones, whereas protein, tannins, reducing sugars, and MDA were more related to non-fermented clones.
Biplot of overall PCA (a) and heatmap of correlations with PC1 and PC2 (b), based on biochemical and nutritional variables from fermented (F) and non-fermented (NF) samples of cocoa clones cultivated in the Amazon.
The distribution of clones across the four quadrants of the PCA biplot further emphasizes the biochemical distinctions between treatments (Fig. 6a). In the upper right quadrant (PC1 + and PC2 +), CEPEC-F, EEOP69-F, and EEOP80-F stood out, associated with variables such as lipids, Fe, K, and Mg. The upper left quadrant (PC1– and PC2 +) included CEPEC-NF, EEOP80-NF, and EEOP69-NF, which showed stronger associations with B, Mn, and Ca. The lower left quadrant (PC1– and PC2–) grouped clones such as EEOP33-NF, BN34-NF, EEOP63-NF, EEOP65-NF, EEOP96-NF, and CCN51-NF, associated with sugars, protein, anthocyanins, tannins, and MDA. In the lower right quadrant (PC1 + and PC2–), CCN51-F, EEOP33-F, EEOP63-F, EEOP65-F, EEOP96-F, and BN34-F were prominent, located near vectors such as SOD, proline, GB, and amino acids, indicating a biochemical profile related to adaptive stress response.
The correlation of biochemical and nutritional variables with the principal components (Fig. 6b) showed that PC1 was positively associated with K, Mg, Fe, SOD, GB, amino acids, and seed mass, and negatively associated with P, phenolics, tannins, protein, and MDA, highlighting the contrast between functional compounds and stress markers. Meanwhile, PC2 was more strongly influenced by Mn, Ca, lipids, Fe, Zn, and Mg, variations in mineral composition and macronutrient distribution. These patterns align with the distribution of clones in Fig. 6 and help explain the separation between fermented and non-fermented groups along the main axes.
Separate PCA analyses for the fermented (Fig. 7a) and non-fermented (Fig. 7b) groups revealed that seed mass clustered with variables such as SOD, GB, proline, and amino acids in both contexts, especially in the fermented group (Fig. 7a). This pattern suggests that higher productivity was associated with a biochemical profile linked to adaptive stress response, particularly in clone EEOP 63. In contrast, clones with lower productivity, such as EEOP 69, were positioned in regions of the biplots associated with stress and degradation-related compounds such as MDA, tannins, and reducing sugars.
PCA biplots with biochemical and nutritional variable vectors, showing the distribution of fermented (a) and non-fermented (b) samples derived from cocoa clones cultivated in the Amazon.
Finally, the correlation analysis revealed relevant associations among biochemical and nutritional variables in both fermented and non-fermented groups. In the fermented group, a strong correlation was observed between K and Mg (r = 0.81), in addition to moderate associations between Ca and P (r = 0.56) and between Mn and MDA (r = 0.59). In the non-fermented group, the most prominent findings were very strong negative correlations between protein and SOD (r = –0.93), protein and phenolics (r = –0.73), and protein and amino acids (r = –0.74), indicating that higher levels of these compounds were associated with lower total protein content. Strong correlations were also identified between P and Mn (r = 0.71), Ca and Mn (r = 0.78), as well as between SOD and amino acids (r = 0.64), protein and B (r = –0.66), and MDA and Cu (r = –0.61).
Discussion
Unraveling the impact of cocoa clone and fermentation on the reduction of sugars and the antinutrient tannin in nutrient availability in cocoa beans cultivated in southwestern Amazonia
Fermentation is a key step in cocoa processing, driving biochemical changes essential for the development of the chocolate’s characteristic sensory profile. During this process, mucilage degradation, polyphenol oxidation, and the activation of enzymatic pathways result in the formation of aroma, flavor, and color precursors, in addition to reducing the astringency and bitterness of the beans4,6. An initial relevant aspect of fermentation is the microbial consumption of sugars, followed by tannin degradation, which is considered an antinutrient. In the present study, the reduction in sugar content in fermented beans was significant and consistent across all evaluated clones.
Sugar levels decreased by more than 95% in all clones, indicating intense and consistent degradation of these compounds. This reduction is largely attributed to microbial activity during the initial stages of fermentation, when yeasts and lactic acid bacteria metabolize sugars to ethanol, acetic acid, and lactic acid43,44. In addition, diffusion of metabolites into the beans and possible residual pulp adhering to the testa (seed coat), which was included in the ground samples, may also contribute to the observed decline. These findings agree with previous reports describing drastic sugar depletion in cocoa fermentation43,44.
As fermentation progresses, tannin levels also decrease. Tannins are phenolic compounds classified as antinutrients due to their ability to form insoluble complexes with proteins and minerals such as iron, zinc, and calcium, impairing their absorption in the gastrointestinal tract45,46. This reduction is associated with the polymerization and complexation of catechins, leading to the formation of higher molecular weight tannins with lower reactivity47. In this study, an average reduction of 49% in tannin content was observed, with significant interclonal variation. Clones such as EEOP 96 showed losses greater than 70%, whereas CCN 51, EEOP 63, and EEOP 33 maintained more stable levels (around 40%).
The magnitude of this response reinforces the importance of selecting genotypes with higher susceptibility to tannin degradation, particularly in productive contexts of the Amazon region, where studies on this aspect remain scarce. The reduction of these compounds may enhance the bioavailability of nutrients in cocoa beans, representing a relevant factor for improving the nutritional quality of the fermented product45,46.
Similar patterns of tannin reduction have also been reported in cocoa by-products. Djali et al.48 observed decreases of 68% and 93% in cocoa bean shells after fermentation and drying, respectively, reinforcing the role of processing in reducing these compounds, although the magnitude differs from that observed in the kernels.
During cocoa fermentation, increases in K content were observed, particularly in clones CCN 51, CEPEC 5, EEOP 65, and EEOP 96, and increases in Mg content were noted in clones BN 34, CCN 51, and CEPEC 5. Although some studies have reported reductions in these elements throughout fermentation, these discrepancies may be related to differences in fermentation systems, turning frequency, mucilage composition, leaching losses, and the genetic characteristics of the clones49. In the present study, it is likely that intracellular mineral release triggered by cell collapse outweighed leaching losses, resulting in higher residual levels in the fermented beans.
Although studies have evaluated mineral dynamics in cocoa pulp, their results indicate that fermentation promotes substantial increases in K, Mg, and Zn contents, while Fe levels tend to decrease43. K and Mg are not only associated with improved functional profiles of cocoa49,50 but also play key roles in human health: K contributes to blood pressure regulation and neuromuscular function, whereas Mg acts as a cofactor in numerous enzymatic reactions, being linked to cardiovascular health and energy metabolism51,52. Their presence in cocoa beans contributes to the nutritional relevance of the product.
Conversely, this study found that nutrients such as P, Ca, S, B, and Mn showed lower concentrations in fermented cocoa beans compared to non-fermented ones, suggesting potential losses due to leaching, particularly during mass turning, or microbial uptake. These results indicate that while fermentation promotes beneficial sensory and functional transformations, it may also lead to selective nutritional losses. Similar findings were reported by49, who observed significant reductions in Ca, Fe, Mg, Mn, Zn, and other minerals during fermentation, confirming the susceptibility of certain elements to the biochemical and physical dynamics of the process.
When compared with literature data, the mineral concentrations observed in this study were within or above the range reported for cocoa beans. Potassium averaged 14.2 g/kg, higher than the values reported by Millena et al.49, which decreased from 12.7 to 6.8 g/kg during fermentation. Calcium levels (~ 0.9 g/kg) were consistent with the range described by the same authors (1.25–1.19 g/kg). Magnesium averaged 3.4 g/kg, in line with their findings (4.6–2.3 g/kg), while iron levels (~ 41 mg/kg) were slightly higher than the reported values (34–26 mg/kg). These comparisons reinforce that the concentrations found here are nutritionally relevant and consistent with those described in the literature.
Thus, a dual effect of fermentation on the nutritional and functional quality of cocoa beans becomes evident. On the one hand, fermentation can favor the retention or even accumulation of certain nutrients such as K and Mg, which are often associated with improved cocoa functionality and human health benefits15,43,50,53. On the other hand, it tends to reduce the levels of elements such as P, Ca, S, B, and Mn, highlighting the delicate balance between functional gains and nutritional losses inherent to the process49. Therefore, the decision to ferment or not should consider not only the desired sensory profile but also the nutritional and technological objectives of the final product, especially in regions like the Amazon, where tailored strategies may add value to cocoa beans.
In this context, non-fermented beans, by preserving higher levels of certain minerals, hold potential for differentiated formulations aimed at health promotion. Their functional value thus lies in the ability to retain essential minerals with recognized benefits to human health, such as blood pressure regulation, energy metabolism support, and prevention of chronic disorders associated with micronutrient deficiencies18,52,53. Accordingly, the choice between fermentation and non-fermentation should reflect both sensory goals and nutritional and technological priorities.
Based on the data obtained, it was also possible to identify clones with superior nutritional performance under different fermentation conditions. From a nutritional perspective, clone EEOP 63 stood out for its high productivity and stability, maintaining satisfactory nutrient levels even after fermentation. In the fermented group, CEPEC 5 showed the best performance, with increases in K and Mg contents and consistently high levels of other nutrients. In the non-fermented group, clone EEOP 65 was the most productive, combining high concentrations of most evaluated nutrients, despite moderate levels of Mn, B, and Cu.
Unveiling the impact of cocoa clone and fermentation on antioxidant traits in cocoa beans cultivated in the southwestern Amazon
Fermentation plays an important role in the formation of new volatile compounds, the development of complex cocoa aromas, and the generation of bioactive compounds, induced by microbial activity and biochemical reactions. During this process, enzymatic and oxidative reactions lead to profound changes in the profiles of phenolic compounds, amino acids, and other functional metabolites, directly impacting the nutritional quality of the final product.
One of the most evident effects observed in this study was the significant reduction in total phenolic content and anthocyanins in fermented cocoa beans, with mean reductions of 88% and 73%, respectively, across the studied clones. The degradation of anthocyanins can reach up to 90% of their initial levels prior to fermentation11,54,55. Despite these losses, the residual levels of these compounds in the present study were higher than those reported in previous studies16,56. Nevertheless, the mechanisms underlying the sharp decline in these compounds during fermentation remain poorly understood. One possible explanation is oxidative stress, which may trigger the degradation of phenolics and anthocyanins through the action of enzymes such as polyphenol oxidase and peroxidase57.
However, this hypothesis appears to be weak in the present study, as low MDA concentrations and high SOD activity were observed, indicating an efficient enzymatic antioxidant system across all evaluated clones. Furthermore, the accumulation of osmoprotective compounds with antioxidant function was detected, including proline, known for stabilizing proteins and membranes58, and glycine betaine, a precursor of glutathione and modulator of the redox system59, identified for the first time in cocoa beans. These findings suggest that no significant oxidative stress occurred during bean fermentation.
Therefore, it is more plausible that the degradation of phenolic compounds and anthocyanins is associated with polymerization processes and complexation with proteins, which reduces their solubility and contributes to decreased astringency of the product47, although it may also compromise the antioxidant quality of the beans7,29. Additionally, in the specific case of anthocyanins, their degradation may involve the direct action of glycosidases, which hydrolyze them into anthocyanidins, followed by oxidation into quinones, a process linked to the brown coloration characteristic of fermented beans60.
Most previous studies on the effects of fermentation on cocoa’s functional quality were conducted using clones that are no longer widely cultivated, and there is a lack of research on agronomically relevant genotypes. In general, all clones evaluated in this study showed adequate antioxidant capacity, modulated by high SOD activity and low MDA levels after fermentation.
Among the evaluated genotypes, the CCN 51 clone stood out for exhibiting not only enzymatic antioxidant activity but also high levels of phenolic compounds, glycine betaine, and proline, compounds with well-established antioxidant, osmoregulatory, and anti-inflammatory properties7,9,58,59. The positive correlation between glycine betaine and proline reinforces the coordinated action of these compounds, highlighting the non-enzymatic antioxidant potential of CCN 51. This characteristic may provide greater oxidative stability in subsequent processing steps, such as roasting, which involves intense thermal degradation, although this hypothesis still needs to be confirmed by further studies.
A distinct behavior was observed for the EEOP 69 clone, which showed high MDA levels in both fermented and non-fermented beans, while still maintaining relatively high phenolic content, especially after fermentation. This preservation suggests the action of alternative antioxidant protection mechanisms, such as the presence of phenolic compounds less susceptible to oxidation, reduced polymerization degree, and subcellular compartmentalization, factors that limit the exposure of phenolics to oxidative enzymes61,62,63.
This profile reinforces that the antioxidant response can vary among clones even under the same fermentation conditions and highlights the importance of considering the functional stability of bioactive compounds in genotype selection.
Despite the benefits associated with fermentation, the loss of antioxidant quality has led to the development of blending strategies, involving the combination of fermented and non-fermented beans11, or even limited fermentation, aiming to maximize the bioactive potential of cocoa7. Therefore, the retention of sensitive bioactive compounds non-fermented cocoa as a valuable raw material for emerging markets seeking to combine distinct aromas and even blend with fermented beans to produce novel differentiated products.
Overall, the results also revealed that non-fermented beans showed high levels of phenolics, anthocyanins, and MDA due to an inefficient antioxidant system, characterized by low SOD activity and reduced levels of protective compounds such as GB and proline, although these effects varied across clones. The higher availability of bioactives in non-fermented beans is likely more related to limited polymerization with proteins and an oxidative stress environment that, although present, was not sufficient to trigger extensive degradation63.
The enzymatic antioxidant system mediated by SOD in non-fermented cocoa beans is relatively limited, although most clones exhibited similar antioxidant capacity, with some variations. In contrast, under fermentation, only one clone clearly stood out. It is also relevant to note that not all clones under postharvest systems without fermentation exhibit high levels of bioactive compounds. For instance, the BN 34 clone showed the lowest levels of total phenolics and anthocyanins, making it less suitable for strategies aimed at maximizing the nutritional value of cocoa. Conversely, the EEOP 96 clone stood out in the same system by maintaining high levels of phenolics and anthocyanins, even under conditions of high MDA levels and low SOD activity. These results suggest that non-enzymatic or structural mechanisms, such as compartmentalization or greater molecular stability, may have contributed to the functional preservation of the material, making it a promising option for applications involving non-fermented beans.
Finally, the results obtained in this study reveal that the clones’ response to fermentation is highly variable and depends not only on the postharvest system but also on the intrinsic biochemical and functional composition of each genotype. This diversity underscores the importance of selecting genetic materials that combine agronomic performance, antioxidant stability, and nutritional potential, regardless of the postharvest system.
Clones such as CCN 51, EEOP 63, EEOP 65, CEPEC 5, EEOP 69, and EEOP 96 exhibited distinct strategies for functional preservation and emerged as promising alternatives for the development of differentiated products. In this context, blending strategies involving fermented and non-fermented beans may be further explored to optimize both sensory and functional attributes.
These findings are consistent with recent work conducted in the Brazilian Amazon by Lima et al.64, who also reported strong genotype-dependent responses to fermentation. Using a targeted metabolomics approach, they observed pronounced reductions in polyphenols such as catechin and epicatechin, while highlighting CCN 51 as one of the top four clones with the highest concentrations of bioactive compounds. Together, these complementary results strengthen the evidence that genetic diversity within Amazonian cocoa plays a decisive role in shaping both the functional stability and the nutritional potential of cocoa beans.
Overall, this study indicates the potential to enhance the quality of cocoa beans produced in the Amazon through the selection of differentiated clones under both fermented and non-fermented postharvest systems, generating valuable insights for the food industry and with potential implications for human health.
Conclusion
This study demonstrated that fermentation significantly alters the biochemical, nutritional, and antioxidant profiles of cocoa beans from different Amazonian clones. Reductions of more than 95% in sugar content and nearly 50% in tannins were observed, accompanied by marked losses of phenolics and anthocyanins. These changes were associated with the activation of enzymatic and non-enzymatic antioxidant systems, particularly the accumulation of osmoprotective compounds such as proline and glycine betaine, which were identified for the first time in cocoa beans. Fermentation also promoted increases in key minerals such as potassium and magnesium, while other elements including calcium, phosphorus, and manganese showed selective losses, highlighting the dual nutritional effects of this process.
At the clonal level, distinct adaptive responses were identified. CCN 51 combined high antioxidant activity with elevated levels of phenolics, proline, and glycine betaine, while EEOP 96 preserved high phenolic and anthocyanin levels under non-fermented conditions. These findings reinforce the importance of genotype selection to optimize the nutritional and functional quality of cocoa beans and provide tailored solutions for the food industry.
From a practical perspective, the results emphasize the potential of non-fermented beans as raw material for differentiated formulations aimed at health-oriented products, while fermentation remains essential for developing desirable sensory attributes. The contrasting profiles revealed here suggest opportunities for blending strategies, combining fermented and non-fermented beans to balance flavor and functionality.
Future research should explore the biochemical mechanisms underlying clonal differences, the optimization of fermentation protocols adapted to Amazonian conditions, and the development of specialty cocoa products that valorize regional diversity. Such efforts may consolidate the Amazon, particularly Rondônia, as a strategic frontier for sustainable cocoa production with high scientific and commercial relevance.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Campos, S. D. M. et al. The role of microbial dynamics, sensorial compounds, and producing regions in cocoa fermentation. Microbiol. Res. 16(4), 75 (2025).
ICCO - International Cocoa Organization. ICCO quarterly bulletin of cocoa statistics 2023. (Accessed 04 Mar 2025). https://www.icco.org/wp-content/uploads/Production_QBCS_XLIX_No.-2.pdf. (2023).
IBGE - Instituto Brasileiro de Geografia e Estatística. Produção agrícola municipal: culturas temporárias e permanentes (IBGE, 2023).
Švarc-Gajić, J. et al. ESG approach in the valorization of cocoa (Theobroma cacao) by-products by subcritical water: Application in the cosmetic industry. Sustain. Chem. Pharm. 31, 100908. https://doi.org/10.1016/j.scp.2022.100908 (2023).
De Vuyst, L. & Leroy, F. Functional role of yeasts, lactic acid bacteria and acetic acid bacteria in cocoa fermentation processes. FEMS Microbiol. Rev. 44, 432–453. https://doi.org/10.1093/femsre/fuaa019 (2020).
Kadow, D. The biochemistry of cocoa flavor – A holistic analysis of its development along the processing chain. J. Appl. Bot. Food Qual. 93, 300–312. https://doi.org/10.5073/JABFQ.2020.093.037 (2020).
Rawel, H. M., Huschek, G., Tchewonpi Sagu, S. & Homann, T. Cocoa bean proteins-characterization, changes and modifications due to ripening and post-harvest processing. Nutrients 11, 428. https://doi.org/10.3390/nu11020428 (2019).
Ashraf, M. & Foolad, M. R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 59, 206–216 (2007).
Ding, X. et al. BTH treatment delays the senescence of postharvest pitaya fruit in relation to enhancing antioxidant system and phenylpropanoid pathway. Foods 10, 846. https://doi.org/10.3390/foods10040846 (2021).
Calvo, A. M. et al. Dynamics of cocoa fermentation and its effect on quality. Sci. Rep. 11, 16746. https://doi.org/10.1038/s41598-021-95703-2 (2021).
Melo, T. S. et al. Evaluation of the content of bioactive compounds in cocoa beans during the fermentation process. J. Food Sci. Technol. 58, 1947–1957. https://doi.org/10.1007/s13197-020-04706-w (2021).
Cortez, L. C., de Souza, A. C. & Martinez, S. J. Changes in the bioactive compounds during fermentation of cocoa beans harvested in the Amazon region. Curr. Res. Food Sci. 6, 100494. https://doi.org/10.1016/j.crfs.2023.100494 (2023).
Pérez, M. A., Rodríguez, L. M. & Gómez, J. F. Sensory and physical evaluation of cocoas (Theobroma cacao L.) from different origins. Acta Sci. Nutr. Health 5, 47–52. https://doi.org/10.31080/ASNH.2020.05.0847 (2021).
Cruz, J. F. M., Leite, P. B., Soares, S. E. & Bispo, E. D. S. Bioactive compounds in different cocoa (Theobroma cacao L.) cultivars during fermentation. Food Sci. Technol. 35, 279–284. https://doi.org/10.1590/1678-457X.6541 (2015).
Mihai, R. A. et al. Abiotic factors from different Ecuadorian regions and their contribution to antioxidant, metabolomic and organoleptic quality of Theobroma cacao L. beans, variety “Arriba Nacional”. Plants 11, 976. https://doi.org/10.3390/plants11070976 (2022).
Zapata-Álvarez, A. et al. Molecular, biochemical, and sensorial characterization of cocoa (Theobroma cacao L.) beans: A methodological pathway for the identification of new regional materials with outstanding profiles. Heliyon 10, e24544. https://doi.org/10.1016/j.heliyon.2024.e24544 (2024).
Katz, D. L., Doughty, K. & Ali, A. Cocoa and chocolate in human health and disease. Antioxid. Redox Signal. 15, 2779–2811. https://doi.org/10.1089/ars.2010.3697 (2011).
Parsaeyan, N., Mozaffari-Khosravi, H., Absalan, A. & Mozayan, M. R. Beneficial effects of cocoa on lipid peroxidation and inflammatory markers in type 2 diabetic patients and investigation of probable interactions of cocoa active ingredients with prostaglandin synthase-2 (PTGS-2/COX-2) using virtual analysis. J. Diabetes Metab. Disord. 13, 30. https://doi.org/10.1186/2251-6581-13-30 (2014).
Barbosa, R. C. M. & Neves, A. D. S. Levantamento semidetalhado dos solos da Estação Experimental de Ouro Preto de Oeste, RO (CEPLAC/CEPEC, 1983).
INMET - Instituto Nacional de Meteorologia. Dados históricos anuais – Ano 2024 (Ministério da Agricultura e Pecuária, 2024).
Turnbull, C. J. International Cocoa Germplasm Database (ICGD) (CRA Ltd./ICE Futures Europe/University of Reading, 2025).
Boza, E. J. et al. Genetic characterization of the cacao cultivar CCN 51: its impact and significance on global cacao improvement and production. J. Am. Soc. Hortic. Sci. 139, 219–229. https://doi.org/10.21273/JASHS.139.2.219 (2014).
Lopes, U. V. et al. Cacao breeding in Bahia, Brazil: strategies and results. Crop Breed. Appl. Biotechnol. 11, 73–81. https://doi.org/10.1590/S1984-70332011000500011 (2011).
Sodré, G. A. & Nery, I. D. Relações entre massas de frutos e amêndoas para estimar rendimentos em cacauicultura. Agrotropica 35, 15–20. https://doi.org/10.21757/0103-3816.2023v35n1p15-20 (2023).
Almeida, C. D., Pires, J. L., Silva, A. P. & Gomes, L. P. Desempenho agronômico de variedades clonais de cacaueiros em Ouro Preto do Oeste, Rondônia. Agrotropica 28, 221–232. https://doi.org/10.21757/0103-3816.2016v28n3p221-232 (2016).
Almeida, C. M. V. C. et al. Recursos genéticos de cacaueiro em Rondônia: retrospectiva histórica, origem e inventário. Agrotropica 27, 93–124. https://doi.org/10.21757/0103-3816.2015v27n2p93-124 (2015).
de Almeida, C. M. V. C., Barriga, J. P., Machado, P. F. R. & Bartley, B. G. D. Evolução do programa de conservação dos recursos genéticos de cacau na Amazônia Brasileira. Boletim Técnico CEPLAC/DEPEA 5, 108 (1987).
Silva Neto, P. J., Matos, P. G. G., Martins, A. C. S. & Silva, A. P. Sistema de produção de cacau para a Amazônia brasileira (CEPLAC, 2001).
Dzelagha, C. N., Emmanuel, A. O. & Esther, A. I. A review of cocoa drying technologies and the effect on bean quality. Int. J. Food Sci. 2020, 8830127. https://doi.org/10.1155/2020/8830127 (2020).
Bataglia, O. C., Furlani, A. M. C., Teixeira, J. P. F., Furlani, P. R. & Gallo, J. R. Métodos de análise química de plantas (Boletim Técnico 78) (Inst. Agron Campinas, 1983).
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A. & Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 28, 350–356. https://doi.org/10.1021/ac60111a017 (1956).
Yemm, E. W. & Cocking, E. C. The determination of amino acids with ninhydrin. Analyst 80, 209–213. https://doi.org/10.1039/an9558000209 (1955).
Price, M. L. & Butler, L. G. Rapid visual estimation and spectrophotometric determination of tannin content of sorghum grain. J. Agric. Food Chem. 25, 1268–1273. https://doi.org/10.1021/jf60214a034 (1977).
Fantozzi, P. & Montedoro, G. Dosage dês composés phénoliques dans drupes d’olives récoltées à différents stades de maturation. Ind. Aliment. Agric. 95, 1335–1339 (1978).
Gutfinger, T. Polyphenols in olive oils. J. Am. Oil Chem. Soc. 58, 966–968 (1981).
Instituto Adolfo Lutz. Normas analíticas do Instituto Adolfo Lutz: Métodos químicos e físicos para análise de alimentos (IMESP, 2008).
Bates, L. S., Waldren, R. P. & Teare, I. D. Rapid determination of free proline for water-stress studies. Plant Soil 39, 205–207. https://doi.org/10.1007/BF00018060 (1973).
Heath, R. L. & Packer, L. Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125, 2141–2145. https://doi.org/10.1016/0003-9861(68)90654-1 (1968).
Grieve, C. M. & Grattan, S. R. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 70, 303–307. https://doi.org/10.1007/BF02374789 (1983).
Lichtenthaler, H. K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. In Methods in Enzymology (eds Douce, R. & Packer, L.) (Academic Press, 1987).
Giannopolitis, C. N. & Ries, S. K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 59, 309–314. https://doi.org/10.1104/pp.59.2.309 (1977).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254. https://doi.org/10.1016/0003-2697(76)90527-3 (1976).
Afoakwa, E. O., Quao, J., Takrama, J., Budu, A. S. & Saalia, F. K. Chemical composition and physical quality characteristics of cocoa (Theobroma cacao L.) beans as affected by pulp preconditioning and fermentation. Int. Food Res. J. 20, 1843–1853. https://doi.org/10.1007/s13197-011-0446-5 (2013).
Sari, A. B. T. et al. Chemical composition and sensory profiles of fermented cocoa beans obtained from various regions of Indonesia. Int. J. Food Sci. 2023, 5639081. https://doi.org/10.1155/2023/5639081 (2023).
Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 56, 317–333. https://doi.org/10.1111/j.1753-4887.1998.tb01670.x (1998).
Hagerman, A. E. et al. High molecular weight plant polyphenolics (tannins) as biological antioxidants. J. Agric. Food Chem. 46, 1887–1892. https://doi.org/10.1021/jf970975b (1998).
Serra-Bonvehí, J. & Ventura Coll, F. Evaluation of bitterness and astringency of polyphenolic compounds in cocoa powder. Food Chem. 60, 365–370. https://doi.org/10.1016/S0308-8146(96)00353-6 (1997).
Djali, M., Setiasih, I. S. & Rindiantika, T. S. Chemical characteristics, phytochemicals and cacao shell toxicity changes during the processing of cocoa beans. Asian J. Agric. Biol. 6, 103–114 (2018).
Millena, C. G. et al. Effect of fermentation stages on the nutritional and mineral bioavailability of cacao beans (Theobroma cacao L.). J. Food Compos. Anal. 115, 104886. https://doi.org/10.1016/j.jfca.2022.104886 (2023).
Van Vliet, J. A. & Giller, K. E. Mineral nutrition of cocoa: A review. In Adv. Agron. (ed. Sparks, D. L.) (Elsevier, 2017).
de Prado, R. Mineral Nutrition of Tropical Plants (Springer Nature, 2021).
D’Elia, L., Barba, G., Cappuccio, F. P. & Strazzullo, P. Potassium intake, stroke, and cardiovascular disease: a meta-analysis of prospective studies. J. Am. Coll. Cardiol. 57, 1210–1219. https://doi.org/10.1016/j.jacc.2010.09.070 (2011).
Volpe, S. L. Magnesium in disease prevention and overall health. Adv. Nutr. 4, 378S-383S. https://doi.org/10.3945/an.112.003483 (2013).
Aprotosoaie, A. C., Luca, S. V. & Miron, A. Flavor chemistry of cocoa and cocoa products—An overview. Compr. Rev. Food Sci. Food Saf. 15, 73–91. https://doi.org/10.1111/1541-4337.12180 (2016).
Misnawi, J. Polyphenol changes during fermentation and their influence on astringency and bitterness of cocoa beans. Food Chem. 86, 403–410. https://doi.org/10.1046/j.1365-2621.2003.00674.x (2003).
Freitas, L. S. et al. Elite cacao clonal cultivars with diverse genetic structure, high potential of production, and good organoleptic quality are helping to rebuild the cocoa industry in Brazil. Int. J. Mol. Sci. 26, 3386. https://doi.org/10.3390/ijms26073386 (2025).
Oracz, J. & Nebesny, E. Effect of roasting parameters on the physicochemical characteristics of high-molecular-weight Maillard reaction products isolated from cocoa beans of different Theobroma cacao L. groups. Eur. Food Res. Technol. 245, 111–128. https://doi.org/10.1007/s00217-018-3144-y (2019).
Sánchez-Mundo, M. L., Bautista-Muñoz, C. & Jaramillo-Flores, M. E. Characterization of protease activities in a crude extract of germinated cacao. CyTA J. Food 13, 578–587. https://doi.org/10.1080/19476337.2015.1023359 (2015).
Deus, V. L., Bispo, E. S., Franca, A. S. & Gloria, M. B. A. Understanding amino acids and bioactive amines changes during on-farm cocoa fermentation. J. Food Compos. Anal. 97(103776), 2015. https://doi.org/10.1016/j.jfca.2020.103776 (2021).
Ruiz-Santiago, F. L. et al. Physicochemical and biochemical changes in cocoa during the fermentation step. Fermentation 10, 405. https://doi.org/10.3390/fermentation10080405 (2024).
Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 56, 317–333. https://doi.org/10.1111/j.1753-4887.1998.tb01670.x (2000).
Cerri, M., Reale, L. & Zadra, C. Metabolite storage in Theobroma cacao L. seed: Cyto-histological and phytochemical analyses. Front. Plant Sci. 10, 1599. https://doi.org/10.3389/fpls.2019.01599 (2019).
Agati, G., Azzarello, E., Pollastri, S. & Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 196, 67–76. https://doi.org/10.1016/j.plantsci.2012.07.014 (2012).
Lima, G. V. S. et al. Targeted metabolomics for quantitative assessment of polyphenols and methylxanthines in fermented and unfermented cocoa beans from 18 genotypes of the Brazilian Amazon. Food Res. Int. https://doi.org/10.1016/j.foodres.2025.116394 (2025).
Funding
This research was supported by the Brazilian Commission for Cocoa Cultivation Development (Comissão Executiva do Plano da Lavoura Cacaueira – CEPLAC) and the São Paulo State University (UNESP) and which provided institutional resources for the field experiment and laboratory analyses, respectively. Additionally, the postdoctoral fellowship of Dr. Edilaine Istéfani Franklin Traspadini was funded by the São Paulo Research Foundation (Fundação de Amparo à Pesquisa do Estado de São Paulo – FAPESP; process no. 2024/00775-7, Linked to grant: 22/10358-9). No specific funding was received for the publication of this article.
Author information
Authors and Affiliations
Contributions
E.I.F.T. performed the sample collection, conducted the laboratory analyses, and wrote the manuscript. R.M.P. contributed to the writing and revision of the manuscript. E.G.R. assisted in the laboratory analyses and contributed to the revision. P.L.G., P.G.S.W., S.B.R. and D.M.P.S. contributed to the manuscript revision. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Traspadini, E.I.F., de Mello Prado, R., Reis, E.G. et al. Fermentation and clone selection modulate the biochemical and nutritional profile of cocoa beans grown in the southwestern Amazon. Sci Rep 15, 43999 (2025). https://doi.org/10.1038/s41598-025-27795-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-27795-z