Abstract
The etiological heterogeneity of spontaneous preterm birth (sPTB) complicates PTB risk prediction and management. Accurate risk stratification at clinical suspicion is critical to administer life-saving interventions for fetuses at genuine risk, while avoiding unnecessary treatments in pregnancies progressing to term to mitigate long-term complications. Current tools lack predictive accuracy, with 40% of suspected PTB delivering at term. This pilot study investigated signaling lipids as potential biomarkers for sPTB. Prospectively collected midstream urine of 30 women with imminent PTB were retrospectively classified into preterm without chorioamnionitis, preterm with chorioamnionitis, and term, and analyzed using liquid-chromatography mass-spectrometry. Overall-preterm vs. term analysis revealed reduced lipoxygenase- and cytochrome P450-derived oxylipins in the preterm group, suggesting impaired inflammation resolution. Pairwise comparisons showed that these differences were primarily driven by the non-infectious preterm group. Notably, 8,9-DiHETrE and 9-HODE emerged as promising etiology-independent biomarkers for sPTB. Furthermore, oxidative stress dominated non-infectious sPTB cases, while inflammatory cascades characterized the infectious cases. A predictive model incorporating 9-HODE and vaginal discharge demonstrated the ability (AUC 84.2%, sensitivity 73.7%, specificity 88.9%) to differentiate between women delivering preterm and at term. These findings highlight the potential of urinary metabolomics as a non-invasive tool to unravel sPTB pathophysiology and enhance risk prediction.
Similar content being viewed by others
Introduction
Preterm birth (PTB), defined as delivery before 37 + 0 weeks of gestation, is the leading cause of perinatal mortality and morbidity globally, affecting approximately 10% of all live births annually1. PTB has a multifactorial origin, involving a complex interplay of maternal, fetal, and environmental factors2. PTB can be categorized into (i) spontaneous PTB (sPTB), which encompasses spontaneous preterm labor with intact membranes or preterm premature rupture of membranes (PPROM), irrespective of eventual mode of delivery, and (ii) iatrogenic PTB (iPTB), wherein delivery is medically indicated due to maternal or fetal complications, through labor induction or pre-labor cesarean section3. Developments in obstetric care, including improved monitoring and management of high-risk pregnancies for both maternal and fetal risk, have contributed to an increase in iPTB and a relative decline in sPTB4,5. sPTB is often associated with inflammation, maternal infections such as chorioamnionitis and urinary tract infection, uterine overdistension, or cervical insufficiency, while iPTB are frequently driven by maternal conditions such as hypertensive and placental disorders of pregnancy, fetal conditions such as fetal growth restriction, or a combination of both3,6. Often, causes of iPTB are identifiable in advance, enabling clinicians to make informed decisions regarding the timing of delivery, balancing the risks of prematurity against potential complications of continuing the pregnancy. However, the mechanisms leading to sPTB remain poorly understood, posing a substantial challenge in their accurate prediction, effective prevention, and timely initiation of treatment2.
Early identification and accurate risk stratification of women at risk of sPTB is crucial for facilitating timely interventions to prolong pregnancy, providing sufficient time for fetal interventions to optimize their preparation for preterm delivery and reduce the risks of adverse neonatal outcomes. Depending on the timing during pregnancy, key interventions include targeted therapies such as cervical cerclage and progesterone for women presenting with a short cervix or a history of PTB, antibiotics to manage maternal bacterial vaginosis or infection, maternal administration of antenatal corticosteroids to accelerate fetal lung maturity, magnesium sulphate for fetal neuroprotection, and additional tocolytics to allow administration of fetal therapy7. Currently, women at high risk of PTB are identified through an assessment of predisposing factors, including maternal and obstetric history, demographic determinants, and behavioral and environmental influences, as well as clinical evaluation of imminent PTB using diagnostic tools such as cervical length screening, fetal fibronectin testing, and biomarkers of inflammation, such as C-Reactive Protein (CRP)8,9. However, these approaches are constrained by their moderate predictive accuracy and limited universal applicability10. Data from randomized clinical trials and population-based studies have shown that nearly 40% of women diagnosed with imminent PTB and exposed to early antenatal corticosteroids, ultimately deliver at term, highlighting the inadequacy of current tools in accurately predicting sPTB11. The timing of corticosteroid administration is critical to maximize efficacy, with optimal outcomes and reduced fetal side effects achieved when administered 2–7 days prior to delivery12. However, unnecessary fetal exposure to corticosteroids in pregnancies that will ultimately end at term is associated with long-term cognitive and behavioral disorders, emphasizing the need for cautious use13,14,15,16. Furthermore, unnecessary hospital admissions contribute to increased healthcare expenditures and impose a burden on hospital resources. Therefore, methods to accurately predict sPTB are urgently needed to enable timely interventions in women at genuine risk while minimizing unnecessary maternal admission and fetal treatment for those ultimately delivering at term.
The vaginal microbiome has emerged as a promising source of potential predictive biomarkers of PTBs, as it has been shown to differ between pregnancies resulting in term and PTBs, with distinct microbial profiles identifiable as early as the first trimester17. Consequently, volatile organic compounds (VOCs), which are the metabolic end-products of biochemical processes such as microbial activity, inflammation, and oxidative stress, are emerging as a promising tool for the prediction of sPTB18,19,20. VOC analysis provides a non-invasive approach to capture microbiome composition, host responses, and their dynamic interactions, offering valuable insights into the metabolic disturbances associated with PTB20,21. In a prospective observational cohort study by Lacey et al., VOCs derived from vaginal swabs collected in the mid-trimester demonstrated moderate predictive capability (AUC 0.79) for sPTB, with predictive accuracy increasing in samples taken closer to delivery (AUC 0.84)21. Similarly, in our previous study, urinary VOC analysis revealed distinct profiles between women admitted for imminent preterm birth who delivered preterm (28 + 0 to 36 + 6) and those who progressed to term (AUC 0.70). In addition, the urinary VOC profiling proved effective in distinguishing sPTB cases with chorioamnionitis (AUC 0.72) or urinary tract infections (AUC 0.97) from those without these complications19. While VOC analysis has demonstrated utility in sPTB prediction and identification of associated infections by providing snapshots of metabolic disturbances, it is inherently limited in its ability to elucidate the molecular mechanisms driving these disturbances.
Signaling lipids, key regulators of inflammation, immune response, and vascular function, offer a powerful framework for deciphering the mechanistic pathways driving sPTB, as a substantial proportion of sPTB cases are associated with inflammation, whether sterile or caused by bacterial infections3,22,23,24,25. These bioactive molecules encompass a diverse array of compound classes, including oxylipins, free fatty acids (FAs), lysophospholipids, endocannabinoids, and bile acids. Oxylipins, derived from the oxygenation of polyunsaturated FAs either enzymatically through cytochrome P450 (CYP450), lipoxygenase (LOX), or cyclooxygenase (COX) pathways, or non-enzymatically via free radical peroxidation, play critical roles in regulating maternal-fetal immune tolerance, uterine quiescence, placental vascularization, and parturition26,27,28,29. Disruptions in oxylipin homeostasis have been implicated in pregnancy complications, including preeclampsia, fetal growth restriction, and PTB, underscoring their significance in maternal-fetal health28. Free FAs and lysophospholipids can be derived from the enzymatic hydrolysis of membrane phospholipids, catalyzed by phospholipase A230. FAs, in addition to serving as precursors for oxylipins, support placental function by promoting angiogenesis, cell signaling, and nutrient transport to the fetus29,31. Among these, omega-3 FAs, such as docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA), are particularly notable for their anti-inflammatory properties, with higher levels associated with a reduced risk of PTB32,33. Lysophospholipids further enhance maternal-fetal adaptation and maintain uterine homeostasis by regulating cellular migration, immune activation, and vascular remodeling34,35,36. Endocannabinoids, also derived from polyunsaturated FAs, contribute to labor onset by stimulating the production of prostaglandins PGE2 and PGF2α, which govern uterine contractions, cervical ripening, and membrane rupture37. Lastly, bile acids, traditionally recognized for their role in lipid metabolism, also function as immunomodulatory signaling molecules38. Elevated levels have been implicated in intrahepatic cholestasis of pregnancy, a condition associated with adverse pregnancy outcomes, including sPTB39,40. The analysis of these signaling lipid molecules in urine provides a powerful approach to investigating their association with sPTB, as urinary metabolites originate from vaginal microbiota-host interactions as well as broader systemic physiological processes41.
To complement the findings of our previous VOC analysis, we conducted a pilot study, where 30 women presenting with imminent PTB at gestational age range 28 + 0–32 + 2 weeks were sampled upon admission, to investigate the potential role of signaling lipids in the pathophysiology and accurate risk stratification of sPTB. Participants were stratified into three groups based on subsequent delivery outcomes: spontaneous preterm delivery without histological evidence of chorioamnionitis, spontaneous preterm delivery with histologically confirmed chorioamnionitis, and term delivery. Specifically, the study aimed to (i) explore metabolic differences between pregnancies leading to spontaneous preterm and term deliveries, with a focus on delineating biochemical signatures associated with non-infectious and infection-driven etiologies of sPTB, and (ii) evaluate the potential of urinary metabolites as predictive non-invasive bed-side biomarkers to distinguish, at the time of suspicion, pregnant women who will actually deliver preterm from those who will eventually deliver at term. These investigations will aid in advancing our understanding of the diverse and complex mechanisms contributing to sPTB and could allow the development of more precise diagnostics tools for accurate prediction of sPTB.
Materials and methods
Cohort
The study cohort presented here is a subset of a larger prospective cohort of pregnant women admitted to the maternity ward of the Department of Obstetrics at Erasmus MC Sophia Hospital in Rotterdam, the Netherlands19. Briefly, all participants presented with imminent PTB and were sampled upon admission, prior to any medical treatments. Women between 23 + 5 and 33 + 6 weeks of gestation received antenatal corticosteroids and tocolytics in accordance with local clinical guidelines. For all cases of PTB, placental tissue was subjected to histopathological analysis for the confirmation of chorioamnionitis. Baseline maternal and pregnancy characteristics, as well as delivery details, were systematically retrieved from electronic patient records. The inclusion and exclusion criteria, as well as the admission protocols, have been described extensively in our previous study19.
Sample collection and storage
From the original prospective cohort, a subset of 30 women with gestational ages between 28 + 0 and 34 + 0 was retrospectively selected for inclusion in this pilot study (Fig. 1). This retrospective cohort comprised 10 women who delivered spontaneously preterm without histological evidence of chorioamnionitis, 10 women who delivered spontaneously preterm with histologically confirmed chorioamnionitis, and 10 women who delivered at term, representing the three distinct clinical groups under investigation. Hereafter, preterm birth cases without histologically confirmed chorioamnionitis are also referred to as non-infectious preterm births, and those with chorioamnionitis as infectious preterm births. At admission and prior to any drug treatments, 25 mL of midstream or catheterized urine was collected from each participant, which were stored at 4 °C within 30 min in a 50 mL sterile falcon tube, and subsequently transported for long-term storage at −20 °C. Selected samples were later retrieved and transferred on dry ice to the analytical chemistry laboratory, where they were stored at −20 °C until aliquoting for metabolomic analysis. A 4 mL aliquot of urine was sent to the Diagnostic Core Laboratory at Erasmus MC for osmolality measurement, which was used for sample normalization in downstream analysis.
Overview of study design and sample classification. The figure illustrates the study design, which included 30 women presenting with suspected imminent preterm birth based on the presence of painful uterine contractions, and/or cervical length < 25 mm, and/or cervical effacement and dilation. Participants were categorized into three groups based on their subsequent delivery outcomes: (i) non-infectious preterm group (n = 10); women who delivered preterm spontaneously without chorioamnionitis, (ii) infectious preterm group (n = 10); women who delivered preterm spontaneously with chorioamnionitis, and (iii) term group (n = 10); women who delivered at term.
Metabolomic data acquisition
For a comprehensive analysis of signaling lipids, the clinical urine samples were analyzed using two distinct preparation protocols adapted from Yang et al. and Fu et al.: (i) a standard protocol to extract free lipids, and (ii) an enzymatic deconjugation protocol to hydrolyze glucuronidated lipids, which are conjugated with glucuronic acid to enhance their water solubility for urinary excretion42,43.
Standard sample preparation
In the standard preparation, 400 µL of urine sample was spiked with 10 µL of antioxidant solution (0.2 mg mL− 1 BHT and 0.2 mg mL− 1 EDTA), 20 µL of internal standard (ISTD) solution composed of deuterated targets, and 200 µL of 0.2 M citric acid and 0.4 M disodium hydrogen phosphate buffer (pH 4.5). Lipids were extracted using liquid-liquid extraction using 1 mL of BuOH: MTBE (1:1 v/v). The samples were subjected to a vortexing of 1 min, followed by centrifugation at 15,800 rcf for 10 min at 4 °C. A volume of 800 µL of the upper organic phase was separated, dried under vacuum using a speedVac, and reconstituted in 50 µL of 70:30 (v/v) mixture of MeOH: ACN. The samples were vortexed and centrifuged for 10 min at 4 °C and at 15,800 rcf, following which they were transferred into a vial for subsequent analysis via LC-MS/MS. The detailed methodology, including chemicals and reagents, preparation of ISTD solution, and overall protocol is described in Yang et al.43 While the standard protocol of Yang et al. remains unchanged, the volumes and matrix type were adapted based on the approach outlined by Fu et al.42
Enzymatic deconjugation sample preparation
To hydrolyze conjugated lipids, an additional enzymatic deconjugation step was incorporated prior to lipid extraction based on the protocol of Fu et al.42 In this preparation, 400 µL of urine sample was spiked with 10 µL of antioxidant solution (0.2 mg mL− 1 BHT and 0.2 mg mL− 1 EDTA), 20 µL of ISTD solution, and 200 µL of enzyme solution. The enzyme solution was prepared using β-glucuronidase derived from bovine liver, dissolved in 200 mM acetate buffer at 4.5 pH, with a final enzyme concentration of 1000 units per sample. The samples were incubated at 37 °C for 2 h to facilitate enzymatic hydrolysis of the conjugated lipids. Following incubation, 200 µL of 0.2 M citric acid and 0.4 M disodium hydrogen phosphate buffer (pH 4.5) was added, and lipids were extracted and reconstituted as described in the standard preparation protocol.
Lipid analysis by LC-MS/MS
To ensure a comprehensive coverage of a wide range of compound classes, two chromatographic methods were employed, following the protocol established by Yang et al.43 Briefly, a low pH chromatography was utilized for various oxylipins (such as prostaglandins, isoprostanes (iPs), hydroxyeicosatetraenoic acids (HETEs), dihydroxyeicosatetraenoic acids (DiHETEs), hydroxyoctadecadienoic acids (HODEs), epoxyeicosatrienoic acids (EpETrEs), dihydroxyeicosatrienoic acids (DiHETrEs), hydroxyeicosapentaenoic acids (HEPEs), and hydroxydocosahexaenoic acids (HDoHEs)), endocannabinoids, and bile acids, and a high pH chromatography for FAs and lysophospholipids (such as lysophosphatidic acids (LPAs), cyclic lysophosphatidic acids (cLPAs), lysophosphatidylethanolamines (LPEs), lysophosphatidylinositol (LPIs), and lysophosphatidylglycerol (LPGs)) ranging from C14 to C22 chain length species. Chromatographic separation was performed by a Shimadzu Nexera X2 LC formed by three high pressure pumps (LC-30AD), communication module (CBM-20Alite), autosampler (SIL-30AC), and an oven (CTO-30 A) from Shimadzu Benelux, employing a Waters BEH C18 column (2.1 × 50 mm, 1.7 μm) for the low pH chromatography and a Kinetex EVO C18 column (2.1 × 50 mm, 1.7 μm) for the high pH chromatography. ESI-MS was conducted using a Sciex QTRAP 6500 MS for both chromatography, allowing for polarity switching and dynamic MRM. Peak integration of the acquired MRM data was performed using the vendor software Sciex OS (v2.1.6.59781).
Data preprocessing
Quality control was performed utilizing mzQuality software based on pooled study samples, method blanks, and ISTDs44. To account for variability in sample processing and instrument performance, peak area ratios were calculated using the closest eluting ISTD for each metabolite. The metabolite dataset was curated by selecting each metabolite from either the standard or the enzymatic deconjugation analysis, based on the method with higher median peak area and superior peak shape and quality. Metabolites demonstrating a relative standard deviation (RSD) below 30% in pooled quality control samples and a background signal of less than 40% in method blanks were retained for downstream analysis. Fisher’s exact test was applied to evaluate potential associations between missing data and experimental groups, and metabolites with over 20% missingness were excluded from subsequent analyses. Additionally, biologically pertinent metabolite ratios of product to precursor (e.g. 20-HETE to AA ratio) and sums of metabolites within the same subclass (e.g. sum of all HETEs), were computed to provide insights into enzyme activities and shifts in metabolic pathways. All metabolic data was normalized to osmolality, log2 transformed, and missing values were imputed using the Quantile Regression Imputation of Left-Censored data method45. The data was further auto-scaled for multivariable analysis.
Statistics
All descriptive statistics, statistical tests, and visualization were performed in RStudio (v4.3.1). Clinical cohort characteristics were summarized using median and range for continuous variables and number with percentage for categorical variables. To determine whether these variables differed significantly between the overall preterm and term groups, Welch’s t-test was applied to continuous variables, and logistic regression for categorical variables. Univariate differential analysis was performed using linear regression, with metabolite as the response and a dichotomous variable representing the comparison groups as the predictor. Effect sizes of the linear regression models are provided by the model’s β coefficients. Models were corrected for the study-specific statistical confounders for preterm outcome identified from the baseline clinical characteristics. The primary analysis compared the combined preterm groups to the term group, to provide an overarching assessment of preterm-associated metabolic alterations, independent of etiology. Subsequently, pairwise comparisons were conducted to examine group-specific differences: (i) preterm deliveries without chorioamnionitis versus term, (ii) preterm deliveries with chorioamnionitis versus term, and (iii) preterm deliveries with chorioamnionitis versus preterm deliveries without chorioamnionitis. Univariate and multivariable logistic regression analysis was performed using potential biomarkers identified from the univariate linear regression analysis to distinguish women who delivered preterm from those who delivered at term. Given the limited sample sizes of individual subgroups (non-infectious and infectious preterm), the overall preterm cohort was analyzed as a single group to ensure model stability and prevent overfitting. Additionally, vaginal discharge, as self-reported by patients, was evaluated independently as well as in combination with the metabolic models to assess their predictive performance, as it reflects changes in the vaginal microenvironment, which has been associated with preterm birth46. Leave-one-out-cross-validation was used to evaluate model performances. A significance threshold of 0.05 was applied to p-values across all statistical analyses. To account for multiple testing, p-values were adjusted using Benjamini-Hochberg method implemented by the p.adjust function. These adjusted p-values, termed q-values, were subjected to a significance threshold of 0.1. These corrections accounted for the total number of metabolic features in univariate tests (n = 133).
Ethics
The study was granted an exemption from formal ethical approval by the local institutional Medical Ethics Committee in accordance with the Dutch Medical Research involving Human Subjects Act (MEC-2018-1302). Written informed consent was obtained from all participants prior to their inclusion in the study. The research was supported by the Erasmus MC Sophia, Departments of Obstetrics and Gynecology and Neonatology, and conducted in strict adherence to the ethical principles outlined in the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Results
Clinical cohort characteristics
A summary of key demographic and clinical characteristics is provided in Table 1. The gestational age at birth ranged from 29 + 0 to 32 + 5 in the preterm groups and 37 + 1 to 41 + 1 in the term group, while gestational age at the time of inclusion and collection of the urinary samples was comparable across groups, ranging from 28 + 0 to 32 + 2, thus reflecting uniformity in the stage of pregnancy during participant enrollment. Apart from chorioamnionitis, PTBs in this cohort were not associated with other clinical conditions commonly linked to PTB, such as urinary tract infections and pre-eclampsia. The median CRP level of the preterm group with chorioamnionitis was higher compared to the other groups, although mean differences were not statistically significant. Based on the t-test and logistic regression analysis comparing overall preterm and term groups, differences in number of prior births (p = 0.01), a history of miscarriages (p = 0.03), and presence of vaginal discharge (p = 0.006) were found to be significantly associated with preterm birth outcomes.
Metabolic cohort characteristics
The quality control review excluded one outlier in the term group, which was identified based on visual anomalies during chromatographic peak integration and further validated through principal component analysis. Among the 182 metabolites targeted using the two chromatographic methods, including 79 oxylipins, 64 lysophospholipids, 16 fatty acids, 8 bile acids, 15 endocannabinoids, and 1 steroid hormone, 102 passed the predefined acceptance thresholds. Based on the median peak area and peak quality, data of oxylipins and FAs were obtained from the enzymatic deconjugation analysis while lysophospholipids, endocannabinoids, and bile acids were derived from the standard analysis. A total of 15 metabolites exhibited varying degrees of missing data, of which three metabolites exceeding 20% missingness, were excluded from further analysis. A total of 34 derived metabolic features were computed, including 23 ratios and 11 sums, to enhance the characterization of metabolic pathways. A list of all included metabolic features and abbreviations is provided in Table S1, and the complete dataset used for statistical analyses is available in Table S2. The three statistical confounders identified in the clinical cohort characteristics were accounted for in the linear regression models: number of prior births, miscarriages (yes/no), and vaginal discharge (yes/no).
Differential metabolic regulation in preterm and term deliveries
Overall preterm birth versus term birth
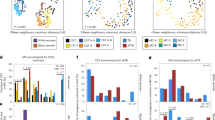
The univariate analysis of differential metabolic abundances between overall preterm and term deliveries revealed metabolic associations (p < 0.05) with sPTB, identifying 13 metabolites with lower levels in the preterm group compared to the term group (Fig. 2a). Among the 13 metabolites, 11 were oxylipins predominantly generated via the LOX and CYP450 pathways, utilizing arachidonic acid (AA), linoleic acid (LA), DHA, or EPA as precursors, while the remaining 2 metabolites were lysophospholipids. The LOX-derived metabolites included 15-HETE, HDoHEs (8-HDoHE, 10-HDoHE), and HODEs (9-HODE, sum HODEs), with β coefficients ranging from −1.42 to −0.85. Oxylipins generated via the CYP450 pathway comprised DiHETrEs (8,9-DiHETrE, 11,12-DiHETrE, 14,15-DiHETrE, sum DiHETrEs), 20-HETE, and 17,18-DiHETE (β = −1.49 to −1.04). The two lysophopholipids identified were LPA(18:2) (β = −2.04) and LPG(14:0) (β = −2.80). Detailed results of the analysis, including effect estimates and p-values for all metabolites, are provided in Table S3.
Differentially regulated metabolites across term and preterm births. The figure presents volcano plots and boxplots illustrating metabolite differences across study groups. Panels (a–b) display volcano plots based on univariate linear regression analysis for the following comparisons: (a) term birth vs. overall preterm birth, (b) term birth vs. non-infectious preterm birth. Panel (c) shows the boxplot of 9-HODE, highlighting its distribution across study groups. Panels (d–e) present volcano plots for (d) term birth vs. infectious preterm birth and (e) non-infectious preterm birth vs. infectious preterm birth. Panel (f) shows the boxplot of cLPA(18:1) which was statistically significant in the non-infectious vs. infectious preterm birth comparison. In all volcano plots, the x-axis represents the regression estimate and the y-axis depicts statistical significance (-log10 p-value). Metabolites are color-coded by compound classes, including bile acids, fatty acids, lysophospholipids, and oxylipins. Data points are shaped to indicate significance levels: significant after FDR correction, significant before FDR correction, and not significant. The vertical dashed line indicates an estimate of zero effect, and horizontal dashed lines denote significance thresholds. In each boxplot, the central line represents the median, with box edges denoting the interquartile range. Individual data points represent osmolality-normalized, log₂-transformed metabolite area ratio in each sample, and the black diamond represents the mean. The letters denote significance between groups from the linear-mixed effect models: shared letters indicate no significant difference (p > 0.05), while unique letters indicate significant difference (p < 0.05). FDR = False Discovery Rate; IP = infectious preterm; NIP = non-infectious preterm; T = term.
Preterm birth without chorioamnionitis versus term birth
The pairwise comparison between non-infectious sPTB and term birth revealed metabolic differences (p < 0.05) that were similar with those identified in the overall preterm vs. term analysis (Fig. 2b). AA-derived metabolites, including HETEs (20-HETE, sum HETEs), and DiHETrEs (5,6-DiHETrE, 8,9-DiHETrE, 11,12-DiHETrE, 14,15-DiHETrE, sum DiHETrEs) exhibited lower levels in the non-infectious preterm group (β = −2.23 to −1.30). Similar trends were observed for LA-derived HODEs (9-HODE, sum HODEs), DHA-derived 8-HDoHE, EPA-derived DiHETEs (14,15-DiHETE, 17,18-DiHETE, sum DiHETEs) and 9-HEPE (β = −1.65 to −1.23), as well as lysophospholipid LPG(14:0) (β = −3.57). Detailed results of the analysis, including effect estimates and p-values for all metabolites, are provided in Table S4.
Preterm birth with chorioamnionitis versus term birth
In the comparison between preterm deliveries with infection and term deliveries, only three metabolites were found to be lower in the infectious preterm group compared to the term group with p < 0.05: 9-HODE (Fig. 2c), sum HODEs, and 8,9-DiHETrE (β = −1.58 to −1.15) (Fig. 2d). Detailed results of the analysis, including effect estimates and p-values for all metabolites, are provided in Table S5.
Preterm birth without chorioamnionitis versus preterm birth with chorioamnionitis
The comparison between non-infectious and infectious preterm deliveries revealed distinct metabolic profiles, with 13 metabolites higher and 3 metabolites lower with p < 0.05 in the infectious group compared to the non-infectious group (Fig. 2e). Among the metabolites with higher levels, cLPAs featured prominently, including cLPA(18:0), cLPA(18:1), cLPA(18:2) (β = 1.16 to 1.57), with cLPA(18:1) (Fig. 2f) demonstrating a statistically significant difference (q < 0.1). Additional lysophospholipids with greater abundances included several LPIs (LPI(16:0), LPI(18:1), LPI(20:4)), LPEs (LPE(16:0), LPE(18:1), LPE(18:2)), and LPG(18:0) (β = 1.01 to 1.75). Additionally, elevated levels of AA and bile acids, glycodeoxycholic acid (GDCA) and glycochenodeoxycholic acid (GCDCA), were also observed in the infectious group (β = 0.86 to 1.07). In contrast, lower levels were observed in ratios related to oxidative stress and lipid peroxidation: 10-HDoHE/DHA, 5-iPF2α-VI/AA, and 8,12-iPF2α-VI/AA (β = −1.03 to −0.85). Detailed results of the analysis, including effect estimates and p-values for all metabolites, are provided in Table S6.
Predictive role of urinary signaling lipids in preterm birth
Univariate and multivariable analyses using logistic regression were performed to stratify women, at the time of clinical suspicion of imminent PTB, into those who would deliver preterm and those who would deliver at term. Predictive metabolomic models were constructed using 8,9-DiHETrE and 9-HODE, which were identified through the univariate linear regression analyses. These metabolites were selected based on their consistent identification in both non-infectious preterm vs. term and infectious preterm vs. term comparisons, suggesting their potential relevance in sPTB regardless of the underlying etiology. The predictive performance of individual models for 8,9-DiHETrE, 9-HODE, and vaginal discharge, yielded AUCs of 0.65, 0.54, and 0.58 respectively. A multivariable model with the two-metabolite panel (8,9-DiHETrE + 9-HODE), achieved an AUC of 0.61. The inclusion of vaginal discharge as an additional predictive feature in the metabolomic models enhanced performance, with AUCs increasing to 0.77 for 8,9-DiHETrE, 0.84 for 9-HODE, and 0.82 for the combined metabolite model. The most optimal model was 9-HODE + vaginal discharge, achieving an AUC of 84.2%, with a sensitivity of 73.7% and a specificity of 88.9%. Receiver operating characteristic curves and model parameters are provided in Fig. 3.
Receiver operating characteristic curves for predicting term versus preterm delivery at the moment of suspected imminent birth. The figure illustrates the diagnostic performance of predictive models in distinguishing between women who would deliver at term and those who would deliver preterm, when both are under the suspicion of imminent preterm birth. Receiver operating characteristic curves derived from leave-one-out cross-validation are shown for (a) individual models (8,9-DiHETrE, 9-HODE, vaginal discharge) and (b) multivariable models (8,9-DiHETrE + 9-HODE, 8,9-DiHETrE + vaginal discharge, 9-HODE + vaginal discharge, 8,9-DiHETrE + 9-HODE + vaginal discharge). The table presents the model parameters, including area under the curve, sensitivity, specificity, and optimal threshold. The optimal threshold refers to the probability cutoff used to classify individuals as preterm or term, determined to maximize the balance between sensitivity and specificity. DiHETrE = dihydroxyeicosatrienoic acid; HODE = hydroxyoctadecadienoic acid.
Discussion
Our study uniquely investigates the potential of maternal urinary signaling lipids at the moment of clinical suspicion of imminent PTB at a gestional age range 28 + 0–32 + 2 weeks, to elucidate underlying sPTB pathophysiology and differentiate between women who will deliver preterm and those who will deliver term. Our approach captures the critical time point when interventions should be initiated, addressing a critical gap in existing predictive tools, which often fail to accurately predict pregnancy outcomes at suspicion. This inaccuracy leads to an overcautious clinical approach where some women suspected for imminent PTB are exposed to unnecessary interventions despite delivering at term, leading to unintended long-term neonatal complications. Moreover, our study stratifies sPTB into non-infectious and infectious subgroups, providing deeper insights into the distinct metabolic pathways underlying these etiologies. This explorative study leveraging urinary metabolomics captures a complementary biochemical landscape against the traditional plasma or serum metabolome, exploring its bed-side diagnostic potential to enhance accurate risk stratification and targeted interventions.
Our study highlights disruptions in oxylipin metabolism at the critical moment of clinical suspicion of imminent PTB in women who delivered preterm, providing crucial insights into potential metabolic perturbations associated with sPTB (Fig. 4). The reduced urinary levels of several LOX- and CYP450-derived oxylipins indicate a dysregulation in pathways integral to inflammation and vascular regulation. Evidence from our study suggests that an impaired resolution of inflammation, reflected by diminished levels of anti-inflammatory mediators, may be associated with sPTB. Specifically, the lower levels of HDoHEs, derived from omega-3 FA DHA known for its anti-inflammatory properties, suggest an impaired anti-inflammatory response, potentially tilting the balance toward a pro-inflammatory state, a hallmark of sPTB pathophysiology and initiation of preterm labor47,48. This finding aligns with the clinical evidence demonstrating that supplementation of DHA reduces the risk of PTB, likely mediated through the enhanced production of protective oxylipins33,49. Furthermore, low plasma concentrations of EPA and DHA during pregnancy have been identified as a strong risk factor for early preterm birth in a Danish cohort, underscoring their critical role in mitigating sPTB risk50.
Network illustrating metabolic pathways, enzymes, and differential regulation of oxylipins across term and preterm births. The figure depicts the metabolic pathway of the differentially regulated oxylipins identified in this study, showing the relationships between precursors, products, and the enzymes mediating their conversion. The network illustrates the metabolism of (a) AA, (b) EPA, (c) DHA, and (d) LA. Arrows represent the enzymatic reactions mediated by the LOX, CYP450, or sEH enzymes, indicating the directionality of metabolite conversions. The network highlights the differential regulation of oxylipins across the following analyses: (i) overall preterm vs. term, (ii) non-infectious preterm vs. term, and (iii) infectious preterm vs. term. Each node represents a metabolite, color-coded to reflect its direction of regulation (upregulated or downregulated) in the preterm group compared to the term and its significance, based on univariate linear regression analysis. AA = arachidonic acid; EPA = eicosapentaenoic acid; DHA = docosahexaenoic acid; LA = linoleic acid; LOX = lipoxygenase; CYP450 = cytochrome P450; sEH = soluble epoxide hydrolase; DiHETrE = dihydroxyeicosatrienoic acid; EpETrE = epoxyeicosatrienoic acid; HETE = hydroxyeicosatetraenoic acid; DiHETE = dihydroxyeicosatetraenoic acid; EpETE = epoxyeicosatetraenoic acid; HEPE = hydroxyeicosapentaenoic acid; HDoHE = hydroxydocosahexaenoic acid; HODE = hydroxyoctadecadienoic acid.
The potential impairment in inflammation resolution is further supported by the reduced levels of DiHETrEs, which are produced through the hydrolysis of their CYP450-derived precursors, EpETrEs, by the soluble epoxide hydrolase (sEH) enzyme51. While EpETrEs are potent anti-inflammatory mediators, their metabolites, DiHETrEs, exhibit significantly diminished anti-inflammatory activity. Elevated levels of DiHETrEs in plasma have been reported in previous studies, which likely reflect the loss of EpETrEs’ protective effects, contributing to an unresolved pro-inflammatory state driving sPTB52,53. In the LIFECODES birth cohort, 8,9-DiHETrE and 11,12-DiHETrE in plasma were significantly associated with increased odds of PTB53. Similarly, in the study by Svenvik et al., higher plasma levels of 11,12-DiHETrE were found to be associated with an increased risk of delivery before 34 weeks of gestation in women with preterm labor52. However, the reduced urinary levels of DiHETrEs observed in our study highlight a discrepancy between plasma and urinary profiles, potentially revealing a distinct spatial compartmentalization in oxylipin metabolism. Supporting this hypothesis, an animal study has shown that urinary DiHETrEs are primarily derived from the renal synthesis rather than systemic activity, as demonstrated by the negligible excretion of radio-labelled 14,15-DiHETrE in urine following plasma administration of its precursor 14,15-EpETrE54. This compartmentalization suggests that urinary oxylipin levels may not always be representative of systemic activity, emphasizing the need for cautious interpretation of urinary oxylipin profiles which may be influenced by heightened systemic utilization, reduced renal synthesis, or altered excretion dynamics. Similarly, research in rats has demonstrated a significant increase in the synthesis of 20-HETE in renal tubules during the late third trimester, accompanied by a corresponding rise in its urinary excretion55,56. This mechanism functions to facilitate the excretion of sodium, aiding in blood pressure reduction to maintain a healthy pregnancy. The reduced urinary levels of 20-HETE observed in our study may therefore indicate an impairment in its natriuretic effect, potentially leading to increased vascular resistance and compromised placental perfusion. While placental insufficiency is well-recognized in iPTB, emerging evidence suggests it may also play a role in sPTB57.
The pairwise comparisons revealed that the metabolic differences observed in the overall preterm vs. term analysis were predominantly driven by the non-infectious preterm group, highlighting the unique pathophysiological mechanisms underlying non-infectious sPTB (Fig. 4). This finding suggests that metabolomic profiling of signaling lipids could be a particularly effective tool for identifying non-infectious cases of sPTB. In contrast, the metabolic profiles of infectious preterm and term deliveries, at the moment of suspicion, exhibited less substantial differences, suggesting an activation of shared inflammatory pathways. This similarity likely stems from the overlap of mechanisms, such as production of inflammatory mediators, hormonal changes, cervical remodeling, and uterine activation, in both infection-driven preterm labor and the physiological preparation for term labor47,58. In term pregnancies, these processes are typically gradually initiated several weeks before birth as part of a tightly controlled physiological preparation59,60. However, in some women, hormonal fluctuations, transient inflammatory responses, or heightened stress, may lead to earlier cervical and uterine changes, prompting clinical suspicion of sPTB61. In such cases, the body may effectively compensate and resolve these triggers, allowing pregnancy to successfully progress to term. Conversely, in infection-driven sPTB, pathogen-associated molecular patterns may trigger a more rapid and intense activation of these pathways. The exaggerated inflammatory response can amplify the labor cascade beyond a critical threshold, ultimately resulting in sPTB. Therefore, alternate techniques like VOC analysis, which has shown promise in detecting infection-related sPTB, may be better suited than metabolic profiling of signaling lipids for identifying infectious sPTB at clinical suspicion. Despite this overlap, the consistent reduction in 8,9-DiHETrE and 9-HODE (Fig. 2c) across both preterm subgroups highlights their potential as universal markers of metabolic dysregulation in sPTB, regardless of etiology. The LA-derived oxylipin, 9-HODE, serves as a natural ligand for the activation of the peroxisome proliferator-activated receptor gamma (PPARγ), a nuclear receptor known to mediate anti-inflammatory responses62. By modulating gene expression, PPARγ plays a crucial role in maintaining immune homeostasis and uterine quiescence via the negative regulation of NF-kB during pregnancy63,64. The observed reduction in 9-HODE levels in preterm deliveries may indicate a dysregulated or impaired activation of these protective pathways, potentially contributing to the pathogenesis of sPTB.
The comparative analysis between non-infectious and infectious sPTB revealed critical insights into the potential mechanistic heterogeneity underlying sPTB. In the context of infectious sPTB, chorioamnionitis triggers a robust inflammatory and immune response, driven in part by the enzymatic activity of phospholipase A265. The elevated levels of cLPAs (Fig. 2f), LPIs, LPEs, and LPG observed in the infectious PTB group aligns with PLA2’s role in mobilizing these lipids as part of the immune defense against pathogens. Notably, the elevated levels of cLPA, a specific and high-affinity antagonist of PPARγ, suggest an enhanced suppression of PPARγ’s anti-inflammatory activity, potentially exacerbating the pro-inflammatory milieu66,67,68. Similarly, increased levels of LPI may reflect its role in orchestrating immune responses to pathogenic insults, including the promotion of chemotaxis to direct immune cells to sites of inflammation and the stimulation of cytokine production69. Concurrently, elevated AA levels highlight its pivotal role as a precursor for pro-inflammatory lipid mediators such as prostaglandins and leukotrienes, which are integral to amplifying the inflammatory cascade and mediating immune defense mechanisms70. Furthermore, prostaglandins PGE2 and PGF2α, have been well documented to mediate uterine contractions and cervical ripening, processes integral to the initiation of parturition71. Consistent with our findings, increased levels of AA have been previously reported in blood of women who delivered preterm, which were associated with an increased sPTB risk72. Additionally, elevated bile acid levels observed in the infectious sPTB group suggest their involvement in PTB through enhanced oxytocin-mediated uterine contractility as bile acids have been shown to increase the expression and responsiveness of the human myometrial oxytocin receptor73. Supporting this observation, previous studies have demonstrated a strong correlation between serum total bile acid concentrations and increased rates of PTB, irrespective of underlying hepatic conditions74. In contrast, the non-infectious sPTB group exhibited heightened oxidative stress, as evidenced by elevated lipid peroxidation ratios. This finding suggests that in infectious sPTB, AA metabolism may be preferentially directed toward prostaglandin synthesis, reducing its availability for isoprostane formation. While prostaglandin-mediated pathways appear more pronounced in infectious sPTB, oxidative stress may contribute more substantially to the non-infectious phenotype, suggesting potential mechanistic differences between these etiologies. Numerous studies have recognized F2 isoprostanes as biomarkers of oxidative stress which have been consistently associated with an increased likelihood of PTB28,75,76. Oxidative stress-induced damage to intrauterine tissues, particularly the fetal membranes, play a pivotal role in labor initiation by promoting fetal cell senescence via the p38 MAPK pathway77. While this process is physiologically timed at term, premature senescence triggered by a redox imbalance is more pronounced in PTB, especially in cases involving PPROM, underscoring oxidative stress as a key mechanistic factor in the pathogenesis of sPTB78.
The individual predictive performance of potential metabolic biomarkers of sPTB (8,9-DiHETrE and 9-HODE), as well as vaginal discharge, demonstrated limited discriminatory power (AUCs ranging from 0.54 to 0.65) in stratifying women at the suspicion of imminent PTB based on their likelihood to deliver preterm or term. However, including vaginal discharge as an additional predictive feature with the metabolomic models significantly improved models’ performances, highlighting the synergistic effect of integrating clinical observations with metabolomic data to improve risk stratification. Notably, 9-HODE + vaginal discharge emerged as the optimal model, with a high predictive performance of AUC 0.84, sensitivity 0.74, and specificity 0.89. The high specificity is particularly valuable in clinical context, as it reduces the likelihood of unnecessary interventions in women who will ultimately deliver at term. 9-HODE reflects impaired anti-inflammatory processes driving preterm labor, while vaginal discharge likely captures localized physiological changes, such as cervical ripening, or underlying pathological conditions like infection or inflammation, underscoring its utility in complementing metabolic data79,80. This integrated model enables a more comprehensive assessment that accounts for both physiological and pathological processes driving preterm labor.
The design and scope of this study present several strengths and limitations that warrant consideration. A key strength lies in the homogeneity of the cohort, with participants recruited from a single center, all when admitted and treated for suspicion of PTB, ensuring consistency in clinical protocols and sample handling. The inclusion of well-characterized preterm subgroups, one with histologically confirmed chorioamnionitis and another without chorioamnionitis, facilitated the investigation of mechanistic differences underlying non-infectious and infection-driven sPTB. These findings reinforce and extend our previous VOC-based research, underscoring the multifactorial nature of sPTB and need for complementary analytical approaches to capture its diverse etiologies. The urinary metabolome offers a non-invasive tool to study lipid signaling dynamics underlying sPTB, which may reflect both local and systemic physiological processes. As samples were collected prior to any clinical intervention, the observed metabolic profiles represent unbiased snapshots at clinical presentation of imminent preterm birth. Importantly, this design enables differentiation within a symptomatic, high-risk population, identifying which women presenting with signs of imminent preterm birth are likely to deliver preterm, thereby supporting clinical decision-making to avoid unnecessary treatments in those that would eventually deliver at term. The small sample size of our pilot study limits the statistical power to detect subtle metabolic differences and reduces the generalizability of the results. However, it still provides valuable preliminary insights into the mechanistic and predictive potential of urinary metabolomics in sPTB, as demonstrated by the identification of relevant biological pathways supported by existing literature. The selection of a subset from the larger cohort introduces potential selection bias, limiting the ability to detect and account for all associations with established risk factors such as prior preterm births and lifestyle variables. Additionally, vaginal discharge was self-reported by patients, which may introduce bias due to its subjective assessment and variability in individual perception. Furthermore, since urinary metabolites represent a cumulative output over time and are subject to modifications during renal filtration, reabsorption, or secretion, their systemic origin or functional role may be distorted, complicating their interpretation in the context of pathophysiological processes. Such findings underscore the necessity of integrating simultaneous plasma and urinary analyses in future studies to disentangle renal and systemic metabolic contributions, thereby strengthening mechanistic insights. To address these limitations and advance the clinical impact of this work, future research should incorporate larger, multicenter cohorts to validate the results and enhance the robustness and reliability of findings, facilitating the development of improved complementary diagnostic tools for sPTB. Furthermore, while this study specifically focuses on symptomatic women at presentation, future research may also explore earlier gestational time points to determine whether these metabolic patterns hold predictive value prior to symptom onset.
Conclusion
sPTB remains a significant challenge in maternal and neonatal healthcare due to the interplay of multifactorial mechanisms that hinder its timely and accurate prediction. This diagnostic uncertainty often results in the unwarranted administration of interventions at the moment of suspected imminent PTB, posing risks of adverse maternal and neonatal outcomes. Our study offers valuable insights into potential metabolic perturbations associated with different etiologies of sPTB and highlights the potential of urinary signaling lipids as non-invasive biomarkers for identifying women at genuine risk at the critical moment of clinical suspicion. A pivotal insight from the study is the potential role of impaired resolution of inflammation in sPTB, as evidenced by reduced levels of anti-inflammatory mediators in urinary profiles. Moreover, our findings delineate the metabolic heterogeneity between non-infectious and infectious sPTB, with oxidative stress predominating in the former and pro-inflammatory cascades in the latter. These observations point to phenotype-specific biological processes that could inform future diagnostic strategies. Notably, urinary metabolomic profiling exhibited greater potential for distinguishing non-infectious sPTB from term deliveries, whereas infection-driven cases showed less distinct metabolic signatures likely due to activation of overlapping pathways, suggesting the potential of alternative techniques, such as VOC analysis, to enhance diagnostic precision. Furthermore, discrepancies between our urinary metabolite findings and plasma trends reported in the literature suggest a compartmentalized metabolic response. This underscores the need for cautious interpretation of urinary metabolomic data, while also suggesting that urine may capture distinct metabolic alterations that could serve as non-invasive predictive biomarkers. This emphasizes the importance of adopting a holistic approach that accounts for contributions from different systems to comprehensively elucidate their mechanistic roles. In conclusion, this pilot study sheds light on the intricate pathophysiology of sPTB, highlighting the potential of urinary signaling lipids as biomarkers for risk stratification. This research establishes a strong foundation for future metabolomic investigations aimed at refining clinical and biochemical characteristics into diagnostic tools and developing targeted interventions, ultimately striving to enhance outcomes for women and neonates at risk of sPTB.
Data availability
All data generated and analyzed in this study are available in the Supplementary Information Files.
Abbreviations
- AA:
-
Arachidonic Acid
- ACN:
-
Acetonitrile
- AUC:
-
Area Under the Curve
- BHT:
-
Butylated Hydroxytoluene
- BuOH:
-
Butanol
- cLPA:
-
Cyclic Lysophosphatidic Acid
- COX:
-
Cyclooxygenase
- CRP:
-
C-Reactive Protein
- CYP450:
-
Cytochrome P450
- DHA:
-
Docosahexaenoic Acid
- DiHETE:
-
Dihydroxyeicosatetraenoic Acid
- DiHETrE:
-
Dihydroxyeicosatrienoic Acid
- EDTA:
-
Ethylenediaminetetraacetic Acid
- EPA:
-
Eicosapentaenoic Acid
- EpETrE:
-
Epoxyeicosatrienoic Acid
- FA:
-
Fatty Acid
- GCDCA:
-
Glycochenodeoxycholic Acid
- GDCA:
-
Glycodeoxycholic Acid
- HDoHE:
-
Hydroxydocosahexaenoic Acid
- HEPE:
-
Hydroxyeicosapentaenoic Acid
- HETE:
-
Hydroxyeicosatetraenoic Acid
- HODE:
-
Hydroxyoctadecadienoic Acid
- iP:
-
Isoprostane
- iPTB:
-
Iatrogenic Preterm Birth
- ISTD:
-
Internal Standard
- LA:
-
Linoleic Acid
- LASSO:
-
Least Absolute Shrinkage and Selection Operator
- LC-MS/MS:
-
Liquid Chromatography-Tandem Mass Spectrometry
- LOX:
-
Lipoxygenase
- LPA:
-
Lysophosphatidic Acid
- LPE:
-
Lysophosphatidylethanolamine
- LPG:
-
Lysophosphatidylglycerol
- LPI:
-
Lysophosphatidylinositol
- MeOH:
-
Methanol
- MTBE:
-
Methyl Tert-Butyl Ether
- PTB:
-
Preterm Birth
- PPROM:
-
Preterm Premature Rupture of Membranes
- sEH:
-
Soluble Epoxide Hydrolase
- sPTB:
-
Spontaneous Preterm Birth
- VOC:
-
Volatile Organic Compound
References
Ohuma, E. O. et al. National, regional, and global estimates of preterm birth in 2020, with trends from 2010: a systematic analysis. Lancet 402, 1261–1271 (2023).
Rubens, C. E. et al. Prevention of preterm birth: Harnessing science to address the global epidemic. Sci Transl Med 6, 262sr5 (2014).
Goldenberg, R. L., Culhane, J. F., Iams, J. D. & Romero, R. Epidemiology and causes of preterm birth. Lancet 371, 75–84 (2008).
Mensah, N. A. et al. Examining recent trends in spontaneous and iatrogenic preterm birth across race and ethnicity in a large managed care population. Am. J. Obstet. Gynecol. 228, 736.e1-736.e15 (2023).
Aughey, H. et al. Iatrogenic and spontaneous preterm birth in england: A population-based cohort study. BJOG Int. J. Obstet. Gynaecol. 130, 33–41 (2023).
Behrman, R. E., Butler, A. S. & Outcomes, I. Of M. (US) C. on U. P. B. and A. H. Causes of preterm birth. in Preterm Birth: Causes, Consequences, and Prevention (National Academies Press (US), (2007).
World Health Organization. WHO Recommendations on Interventions To Improve Preterm Birth Outcomes (World Health Organization, 2015).
Zeng, X., Jiang, W., He, X. & Zhang, J. Preterm birth: Epidemiology, risk Factors, Pathogenesis, and prevention. Oxf. Res. Encyclopedia Global Public. Health. https://doi.org/10.1093/acrefore/9780190632366.013.515 (2024).
Hezelgrave, N. L., Shennan, A. H. & David, A. L. Tests to predict imminent delivery in threatened preterm labour. BMJ 350, h2183–h2183 (2015).
Gravett, M. G. et al. Assessment of current biomarkers and interventions to identify and treat women at risk of preterm birth. Front Med 11, 1414428 (2024).
Ninan, K. et al. The proportions of term or late preterm births after exposure to early antenatal corticosteroids, and outcomes: systematic review and meta-analysis of 1.6 million infants. BMJ 382, e076035 (2023).
Hamm, R. F., Combs, C. A., Aghajanian, P. & Friedman, A. M. Society for Maternal-Fetal medicine special statement: quality metrics for optimal timing of antenatal corticosteroid administration. Am. J. Obstet. Gynecol. 226, B2–B10 (2022).
Fee, E. L., Stock, S. J. & Kemp, M. W. Antenatal steroids: benefits, risks, and new insights. J Endocrinol 258, e220306 (2023).
Ninan, K., Liyanage, S. K., Murphy, K. E., Asztalos, E. V. & McDonald, S. D. Evaluation of Long-term outcomes associated with preterm exposure to antenatal corticosteroids: A systematic review and Meta-analysis. JAMA Pediatr. 176, e220483 (2022).
Melamed, N. et al. Neurodevelopmental disorders among term infants exposed to antenatal corticosteroids during pregnancy: a population-based study. BMJ Open. 9, e031197 (2019).
Räikkönen, K., Gissler, M. & Kajantie, E. Associations between maternal antenatal corticosteroid treatment and mental and behavioral disorders in children. JAMA 323, 1–10 (2020).
Haque, M. M., Merchant, M., Kumar, P. N., Dutta, A. & Mande, S. S. First-trimester vaginal Microbiome diversity: A potential indicator of preterm delivery risk. Sci. Rep. 7, 16145 (2017).
Shen, Q., Liu, Y., Li, G. & An, T. A review of disrupted biological response associated with volatile organic compound exposure: insight into identification of biomarkers. Sci. Total Environ. 948, 174924 (2024).
Ronde, E. et al. Detection of spontaneous preterm birth by maternal urinary volatile organic compound analysis: A prospective cohort study. Front. Pediatr. 10, 1063248 (2022).
Ophelders, D. R. M. G. et al. Screening of chorioamnionitis using volatile organic compound detection in exhaled breath: A Pre-clinical proof of concept study. Front Pediatr 9, 617906 (2021).
Lacey, L., Daulton, E., Wicaksono, A., Covington, J. A. & Quenby, S. Volatile organic compound analysis, a new tool in the quest for preterm birth prediction—an observational cohort study. Sci. Rep. 10, 12153 (2020).
Wei, S. Q., Fraser, W. & Luo, Z. C. Inflammatory cytokines and spontaneous preterm birth in asymptomatic women: a systematic review. Obstet. Gynecol. 116, 393–401 (2010).
Chatterjee, P., Chiasson, V. L., Bounds, K. R. & Mitchell, B. M. Regulation of the Anti-Inflammatory cytokines Interleukin-4 and Interleukin-10 during pregnancy. Front. Immunol. 5, 253 (2014).
Dutta, S., Sengupta, P. & Haque, N. Reproductive Immunomodulatory functions of B cells in pregnancy. Int. Rev. Immunol. 39, 53–66 (2020).
Boeldt, D. & Bird, I. Vascular adaptation in pregnancy and endothelial dysfunction in preeclampsia. J. Endocrinol. 232, R27–R44 (2017).
Liu, H. et al. Lipid imbalance and inflammatory Oxylipin cascade at the maternal-fetal interface in recurrent spontaneous abortion. Heliyon 10, e40515 (2024).
Mosaad, E., Peiris, H. N., Holland, O., Morean Garcia, I. & Mitchell, M. D. The Role(s) of eicosanoids and exosomes in human parturition. Front. Physiol. 11, 594313 (2020).
Welch, B. M., McNell, E. E., Edin, M. L. & Ferguson, K. K. Inflammation and oxidative stress as mediators of the impacts of environmental exposures on human pregnancy: evidence from Oxylipins. Pharmacol. Ther. 239, 108181 (2022).
Duttaroy, A. K. & Basak, S. Maternal dietary fatty acids and their roles in human placental development. Prostaglandins Leukot. Essent. Fat. Acids 155, 102080 (2020).
Besenboeck, C., Cvitic, S., Lang, U., Desoye, G. & Wadsack, C. Going into labor and beyond: phospholipase A2 in pregnancy. Reproduction 151, R91–R102 (2016).
Duttaroy, A. K. & Basak, S. Maternal fatty acid metabolism in pregnancy and its consequences in the Feto-Placental development. Front Physiol 12, 787848 (2022).
Fu, J. Y. et al. The role of omega-3 polyunsaturated fatty acids in the prevention of preterm birth. Med J. Aust 220, 502–504 (2024).
Cetin, I. et al. Omega-3 fatty acid supply in pregnancy for risk reduction of preterm and early preterm birth. Am. J. Obstet. Gynecol. MFM. 6, 101251 (2024).
Gräler, M. H. & Goetzl, E. J. Lysophospholipids and their G protein-coupled receptors in inflammation and immunity. Biochim Biophys. Acta BBA - Mol Cell. Biol. Lipids. 1582, 168–174 (2002).
Beltrame, J. S. et al. Lysophosphatidic acid induces the crosstalk between the endovascular human trophoblast and endothelial cells in vitro. J. Cell. Physiol. 234, 6274–6285 (2019).
Nagamatsu, T. et al. Emerging roles for lysophospholipid mediators in pregnancy. Am. J. Reprod. Immunol. N Y N 1989. 72, 182–191 (2014).
Chan, H. W., McKirdy, N. C., Peiris, H. N., Rice, G. E. & Mitchell, M. D. The role of endocannabinoids in pregnancy. Reproduction 146, R101–R109 (2013).
Fiorucci, S., Biagioli, M., Zampella, A. & Distrutti, E. Bile acids activated receptors regulate innate immunity. Front. Immunol. 9, 1853 (2018).
Pataia, V., Dixon, P. H. & Williamson, C. Pregnancy and bile acid disorders. Am. J. Physiol. -Gastrointest Liver Physiol. 313, G1–G6 (2017).
Sarker, M., Zamudio, A. R., DeBolt, C. & Ferrara, L. Beyond stillbirth: association of intrahepatic cholestasis of pregnancy severity and adverse outcomes. Am. J. Obstet. Gynecol. 227, 517.e1-517.e7 (2022).
Bouatra, S. et al. The human urine metabolome. PLoS ONE. 8, e73076 (2013).
Fu, J. et al. Metabolomics profiling of the free and total oxidised lipids in urine by LC-MS/MS: application in patients with rheumatoid arthritis. Anal. Bioanal Chem. 408, 6307–6319 (2016).
Yang, W. et al. A comprehensive UHPLC-MS/MS method for metabolomics profiling of signaling lipids: markers of oxidative stress, immunity and inflammation. Anal. Chim. Acta. 1297, 342348 (2024).
Peet, M. et al. mzQuality: A tool for quality monitoring and reporting of targeted mass spectrometry measurements. 2025.01.22.633547 Preprint at https://doi.org/10.1101/2025.01.22.633547 (2025).
Wei, R. et al. Missing value imputation approach for mass Spectrometry-based metabolomics data. Sci. Rep. 8, 663 (2018).
Bayar, E., Bennett, P. R., Chan, D., Sykes, L. & MacIntyre, D. A. The pregnancy Microbiome and preterm birth. Semin Immunopathol. 42, 487–499 (2020).
Romero, R. et al. Inflammation in preterm and term labor and delivery. Semin Fetal Neonatal Med. 11, 317–326 (2006).
Habelrih, T. et al. Inflammatory mechanisms of preterm labor and emerging anti-inflammatory interventions. Cytokine Growth Factor. Rev. 78, 50–63 (2024).
Ramsden, C. E. et al. Plasma Oxylipins and unesterified precursor fatty acids are altered by DHA supplementation in pregnancy: can they help predict risk of preterm birth? Prostaglandins Leukot. Essent. Fat. Acids 153, 102041 (2020).
Olsen, S. F. et al. Plasma Concentrations of Long Chain N-3 Fatty Acids in Early and Mid-Pregnancy and Risk of Early Preterm Birth. EBioMedicine. 35, 325–333 (2018).
Spector, A. A., Fang, X., Snyder, G. D. & Weintraub, N. L. Epoxyeicosatrienoic acids (EETs): metabolism and biochemical function. Prog Lipid Res. 43, 55–90 (2004).
Svenvik, M. et al. Plasma Oxylipin levels associated with preterm birth in preterm labor. Prostaglandins Leukot. Essent. Fat. Acids. 166, 102251 (2021).
Aung, M. T. et al. Prediction and associations of preterm birth and its subtypes with eicosanoid enzymatic pathways and inflammatory markers. Sci. Rep. 9, 17049 (2019).
O’Donnell, V., Milne, G., Nogueira, M., Giera, M. & Schebb, N. Quantitation of Oxylipins in biological Samples, focusing on Plasma, and urine. 317–350 (2023). https://doi.org/10.1002/9783527836512.ch12
Kikut, J., Komorniak, N., Ziętek, M., Palma, J. & Szczuko, M. Inflammation with the participation of arachidonic (AA) and Linoleic acid (LA) derivatives (HETEs and HODEs) is necessary in the course of a normal reproductive cycle and pregnancy. J. Reprod. Immunol. 141, 103177 (2020).
Wang, M. H., Zand, B. A., Nasjletti, A. & Laniado-Schwartzman, M. Renal 20-hydroxyeicosatetraenoic acid synthesis during pregnancy. Am. J. Physiol. -Regul Integr. Comp. Physiol. 282, R383–R389 (2002).
Preston, M., Hall, M., Shennan, A. & Story, L. The role of placental insufficiency in spontaneous preterm birth: A literature review. Eur. J. Obstet. Gynecol. Reprod. Biol. 295, 136–142 (2024).
Gonzalez, J. M., Dong, Z., Romero, R. & Girardi, G. Cervical Remodeling/Ripening at term and preterm delivery: the same mechanism initiated by different mediators and different effector cells. PLoS ONE. 6, e26877 (2011).
Thornton, J. M., Browne, B. & Ramphul, M. Mechanisms and management of normal labour. Obstet. Gynaecol. Reprod. Med. 30, 84–90 (2020).
Behrman, R. E., Butler, A. S. & Outcomes, I. Of M. (US) C. on U. P. B. and A. H. Biological pathways leading to preterm birth. in Preterm Birth: Causes, Consequences, and Prevention (National Academies Press (US), (2007).
Zangeneh, F. Z. & Hantoushzadeh, S. The physiological basis with uterine myometrium contractions from electro-mechanical/hormonal myofibril function to the term and preterm labor. Heliyon 9, e22259 (2023).
Christofides, A., Konstantinidou, E., Jani, C. & Boussiotis, V. A. The role of peroxisome Proliferator-Activated receptors (PPAR) in immune responses. Metabolism 114, 154338 (2021).
Wieser, F., Waite, L., Depoix, C. & Taylor, R. PPAR Action in Human Placental Development and Pregnancy and Its Complications. PPAR Res. 2008, 527048 (2008).
Mahendra, J. et al. Evidence linking the role of placental expressions of PPAR-γ and NF-κB in the pathogenesis of preeclampsia associated with periodontitis. J Periodontol 87, 962–970 (2016).
De Luca, D. et al. Secretory phospholipase A2 expression and activity in preterm clinical chorioamnionitis with fetal involvement. AJP Lung Cell. Mol. Physiol 323, L121–L128 (2022).
Tsuda, S. et al. Cyclic phosphatidic acid is produced by autotaxin in blood. J. Biol. Chem. 281, 26081–26088 (2006).
Tsukahara, T. et al. Phospholipase D2-dependent Inhibition of the nuclear hormone receptor PPARγ by Cyclic phosphatidic acid. Mol. Cell. 39, 421–432 (2010).
Tsukahara, T. PPARγ Networks in Cell Signaling: Update and Impact of Cyclic Phosphatidic Acid. J. Lipids. 2013, 246597 (2013).
Martínez-Aguilar, L. M. et al. Lysophosphatidylinositol promotes chemotaxis and cytokine synthesis in mast cells with differential participation of GPR55 and CB2 receptors. Int. J. Mol. Sci. 24, 6316 (2023).
Dennis, E. A. & Norris, P. C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 15, 511–523 (2015).
Olson, D. M. The role of prostaglandins in the initiation of parturition. Best Pract. Res. Clin. Obstet. Gynaecol. 17, 717–730 (2003).
Reece, M. S., McGregor, J. A., Allen, K. G. & Harris, M. A. Maternal and perinatal long-chain fatty acids: possible roles in preterm birth. Am. J. Obstet. Gynecol. 176, 907–914 (1997).
Germain, A. M. et al. Bile acids increase response and expression of human myometrial Oxytocin receptor. Am. J. Obstet. Gynecol. 189, 577–582 (2003).
You, S. et al. Dysregulation of bile acids increases the risk for preterm birth in pregnant women. Nat. Commun. 11, 2111 (2020).
Eick, S. M. et al. Urinary oxidative stress biomarkers are associated with preterm birth: an ECHO program study. Am. J. Obstet. Gynecol. 228, 576.e1-576.e22 (2023).
Ferguson, K. K., Meeker, J. D., McElrath, T. F., Mukherjee, B. & Cantonwine, D. E. Repeated measures of inflammation and oxidative stress biomarkers in preeclamptic and normotensive pregnancies. Am. J. Obstet. Gynecol. 216, 527e1–527e9 (2017).
Menon, R. & Papaconstantinou, J. p38 mitogen activated protein kinase (MAPK): A new therapeutic target for reducing the risk of adverse pregnancy outcomes. Expert Opin. Ther. Targets. 20, 1397–1412 (2016).
Menon, R. Oxidative stress damage as a detrimental factor in preterm birth pathology. Front. Immunol. 5, 567 (2014).
Khaskheli, M., Baloch, S., Baloch, A. S. & Shah, S. G. S. Vaginal discharge during pregnancy and associated adverse maternal and perinatal outcomes. Pak J. Med. Sci. 37, 1302–1308 (2021).
Becher, N., Waldorf, K. A., Hein, M. & Uldbjerg, N. The cervical mucus plug: structured review of the literature. Acta Obstet. Gynecol. Scand. 88, 502–513 (2009).
Author information
Authors and Affiliations
Contributions
Study conception: IKMR, SS, TH; Sample Collection: ER, SS; Data acquisition and curation: MUT, HLE, LL, AH; Data analysis: MUT; Data interpretation: MUT; Data visualization: MUT; Manuscript drafting: MUT, ER; Critical revision of manuscript: MUT, ER, SS, HLE, LL, AH, YBR, AF, TGJM, AK, IKMR, TH. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Thangavelu, M.U., Ronde, E., Lamont, L. et al. Maternal urinary metabolomic signatures preceding spontaneous preterm birth: A pilot study. Sci Rep 15, 45771 (2025). https://doi.org/10.1038/s41598-025-28436-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-28436-1