Abstract
By treating cis-1-amino-2-indanol with different functionalized isocyanates and isothiocyanates, a group of 15 indanol derivatives were synthesized. The synthesis was effectively performed employing a previously proven method and the resulting products were characterized through multiple spectroscopic techniques including NMR spectroscopy and the α-glucosidase inhibitory assay was used to evaluate their α-glucosidase inhibitory capacity. The results indicated a varied level of inhibitory activity against α-glucosidase and 2h showed the highest potency with IC50 value of 9.64 ± 0.24 µM, while compounds 2g, 2c, and 3i also exhibited significant inhibition. The kinetic analysis of the most active inhibitor 2h showed its competitive mode of inhibition, with a Ki value of 7.39 ± 0.088 µM. The binding modes of compounds were predicted by molecular docking analysis within the active site of α-glucosidase enzyme, where our molecules showed significant binding and docking scores. Docking analysis revealed that the thiourea and urea moieties play a critical role in facilitating the interaction of the molecules with one of the catalytic triad residues. The current study provides a basis for the development of a possible lead compound that can inhibit α-glucosidase.
Similar content being viewed by others
1. Introduction
Aminoindanol is an essential scaffold in organic chemistry and medicine. A major focus of organic chemistry encompasses the synthesis of these molecules and the development of innovative methodologies for their acquisition1,2,3,4. These scaffolds have gained the attention of academic and industrial researchers to generate their derivatives with novel applications through multiple synthetic routes. A sequence of enantioselective enzymatic processes has been reported where lipases and transaminases were employed to produce trans-(1R, 2R) and cis-(1 S, 2R)-1-amino-2-indanols, while the (R)-2-hydroxy indanone, 2-acetoxyindanone was hydrolyzed enantio-selectively by Lipase. In water-saturated ethyl acetate, (R)-2-hydroxy indanone was used as an amino donor for the synthesis of trans-1-amino-2-indanol (de >98%), and reductive amination was used to obtain the cis-isomer from the same starting material. The kinetic resolution was carried out using enantioselective ω-transaminase (ω-TA), whose enantioselectivity rose to 22.1 when 5% γ-cyclodextrin was present5,6. These include rigidity, the orientation of the two stereogenic centers, along with the ability of amino and hydroxy groups to coordinate with certain metals or act as hydrogen bond donors or acceptors, and diverse activities exhibited by these entities and their potential for further derivatization7. According to reports, the cis-1-amino-2-indanol’s rigid cyclic structure makes it an essential ligand or chiral auxiliary in chemical synthesis. Furthermore, cis-1-amino-2-indanol is also employed as a chiral auxiliary in several asymmetric transformations, including diastereoselective reduction and diastereoselective enolate alkylation (Fig. 1)8. The substructure of indanol is very interesting for the design of drugs. It is also in KNI-10,006, which is used to treat malaria, or in Indinavir sulfate (Crixivan®), an HIV protease inhibitor created by Merck (Rahway, NJ, USA) to treat AIDS, etc9,10,11,12.
The World Health Organization (WHO) estimates that around 90% of people with diabetes globally have type 2 diabetes mellitus (T2DM), a situation projected to impact 439 million people by the year 2030. Between 2012 and 2014, almost 1.5 million people lost their lives due to complications associated with diabetes13,14. T2DM is an extremely complicated metabolic disease, and despite the availability of FDA-approved anti-diabetic medications, the patients still have hyperglycemia and severe consequences.
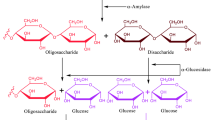
α-Glucosidase (EC 3.2.1.20), a crucial metabolic enzyme that catalyzes the digestion of massive, non-absorbable carbohydrates into smaller glucose molecules to be absorbed by the small intestine. Consequently, the enzyme has become an excellent drug target for the treatment of T2DM15. The rise of blood sugar after consumption of a carbohydrate-rich diet is suppressed when the α-glucosidase enzyme activity is prevented16. Clinically prescribed α-glucosidase inhibitors (AGIs), including acarbose, voglibose, and miglitol, effectively lower postprandial glucose levels in individuals with T2DM17. Nevertheless, these AGIs have inhibitory activity against the α-glucosidase enzyme; however, they are linked to several issues such as diarrhea, flatulence, and gastrointestinal pain18. The adverse effects of these medications demand new small-molecule inhibitors of this enzyme with a high efficacy and safety profile for the management of T2DM. T2DM.
Derivatives of Cis indanol.
The biological activity of indanes, indanols, and iso(thio)cyanates, as well as the demand for other new bioactive compounds and structural diversity, prompted us to combine the above moieties into a single pharmacophoric substance. Indanol serves as an important structural precursor for the synthesis of numerous pharmacologically active compounds. Owing to its unique bicyclic framework and versatile functionalization potential, indanol derivatives have demonstrated significant α-glucosidase inhibitory activity, making them valuable scaffolds in the design and development of novel antidiabetic agents4,10,19,20,21,22. The designed target molecules are similar in structure, differing by the presence of oxygen (in isocyanate) and sulfur (in isothiocyanate) and the alkyl groups present at the side chains (Fig. 1). These tiny variations allow us to figure out how this change would affect interactions with biological targets using α-glucosidase and cytotoxic analyses. Furthermore, the in-silico studies including molecular docking were also performed for better interpretation of results.
Experimental section
General methods
For all reactions, the commercially available chemicals were purchased from Sigma-Aldrich. Chemical reactions were carried out in commercially available solvents, which were dried prior to the chemical reactions. The precoated silica gel plates (Merck, G60 F-254, UV-254) were used for Thin-Layer Chromatography (TLC) experiments. The visualization of the TLC plates was done by using a 254 nm and 366 nm UV light. The IR spectra was recorded on Bruker ATR-Tensor 37 spectrophotometer (νmax in cm− 1). Optical rotations were measured on KRUSS P3000 polarimeter (A. Kruss Optronic, Germany). 1H NMR spectra were recorded on Bruker Avance 600 (150 MHz for13C) spectrometer; chemical shift values (δ) were recorded in ppm, with residual peak of CDCl3 (δ (H) 7.26, δ (C) 77.0) used for referencing.
Chemical synthesis
All the compounds (2a to 4o) were synthesized at room temperature. The equimolar amounts of (1R,2 S)-cis-1-Amino-2-indanol were used to react with the substituted iso(thio)cyanates in toluene for 1–2 h. The precipitates were then filtered and dried. The products obtained were in good yields.
1-(4-chlorophenyl)-3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl) thiourea (2a)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 4-chlorophenylisothiocyanate (85 mg, 0.5 mmol) in dry toluene (5 mL) stirred at room temperature for 4 h. After the reaction was complete, the precipitates were filtered and dried. The resulting off-white solid weighed 151 mg, yielding 95%, m.p: 168 °C; IR: 3392.8, 2929.8, 2316.5, 1693.5, 1519.9 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.93 (d, 1H, J = 14.6, CH2) and 3.25 (dd, 1H, J = 5.1, CH2), 4.80 (m, 1H, J = 5.1, CH), 5.98 (s, 1H, OH), 6.8 (t, 1H, J = 6.6 Hz, CH), 7.2 (m, 1H, NH), 7.23–7.29 (m, 6 H, ArH), 7.35–7.38 (m, 2 H, ArH), 8.21 (d, 1H, NH, J = 25.9), 13C NMR ppm; 39.8, 72.7, 73.6, 124.7, 125.5, 126.1,127.4, 128.2, 128.6, 129.1, 130.2, 132.7, 134.7, 139.8, and 180.9; (ESI) m/z: calculated for protonated C16H15ClN2OS [M + H]+ 319.8190, found 319.8193.
1-(4-fluorophenyl)-3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl)thiourea (2b)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 4-Fluorophenylisothiocyanate (76.6 mg, 0.5 mmol) in dry toluene (5 mL) was stirred at room temperature for 4 h. After the reaction was complete, the precipitates were filtered and dried, yielding 138 mg of an off-white solid with a 92% yield, m.p: 144 °C; IR: 3394.8, 2926, 2312.6, 1697.3, 1523.7 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.93 (d, 1H, J = 16.6, CH2) and 3.25 (dd, 1H, J = 5.7, CH2), 4.79 (s, 1H, CH), 5.97 (s, 1H, OH), 6.66 (s, 1H, CH), 7.18 (t, 2 H, J = 8.4 Hz, ArH), 7.2–7.29 (m, 5 H, ArH, NH), 7.26 (d, 2 H, J = 7.02 Hz, ArH), 8.19 (m, 1H, NH), 13C NMR ppm; 39.7, 63.1, 73.5, 117.0, 117.2, 124.7, 125.5, 127.4, 127.4, 128.6, 132.0, 139.8, 162.2, 181.2; (ESI) m/z: calculated for protonated C16H15FN2OS [M + H]+ 303.3674, found 303.3677.
1-(2-hydroxy-2,3-dihydro-1H-inden-1-yl)-3-(m-tolyl) thiourea (2c)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 3-Methylphenylisothiocyanate (63 µl, 0.5 mmol) was dissolved in dry toluene (5 mL) and stirred at room temperature for 4 h. Upon completion of the reaction, the resulting precipitate was filtered and dried. This process yielded 130 mg of an off-white solid with a 90% yield. m.p: 137 °C; IR: 3307.9, 2912.5, 2024.3, 1591.3, 1519 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.34 (s, 3 H, CH3), 2.94 (d, 1H, J = 16.62, CH2) and 3.25 (d, 1H, J1 = 12.84, CH2,), 4.79 (s, 1H, CH), 5.99 (s, 1H, OH), 6.79 (d, 1H, J = 7.92 Hz, CH), 7.04–7.11 (m, 4 H, ArH), 7.25–7.28 (m, 4 H, ArH), 7.38 (d, 1H, NH, J = 6.72 Hz) 8.11 (d, 1H, NH, J = 13.8 Hz). 13C NMR ppm; 21.3, 39.7, 63.3, 73.6, 121.8, 122.7, 124.7, 125.5, 126.3, 127.3, 128.2, 128.5, 129.3, 130.0, 135.9, 139.9, 140.4, 180.8; (ESI) m/z: calculated for protonated C17H18N2OS [M + H]+ 299.4040, found 299.4043.
1-ethyl-3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl) thiourea (2d)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and ethyl isothiocyanate (43.7 µl, 0.5 mmol) in 5 mL of dry toluene was stirred at room temperature for 4 h. After the reaction was complete, the precipitates were filtered and dried, resulting in 108 mg of an off-white solid with a 92% yield., m.p: 118 °C; IR: 3306, 3034, 2318.4, 1670.3, 1523.7 cm− 1; 1H NMR (CDCl3-d) δ ppm; 1.24 (t, 3 H, CH3, J = 7.2 Hz), 2.91 (d, 1H, CH2, J = 10.6 Hz), 3.25 (dd, 1H, CH2, J = 4.7 Hz), 3.40 (s, 2 H, CH), 4.59 (s, 1H, CH), 5.78 (s, 1H, OH), 6.48 (s, 2 H, ArH), 7.22–7.28 (m, 4 H, 3 ArH & 1NH), 7.34 (d, 1H, NH, J = 7.1 Hz), 13C NMR ppm; 14.1, 39.3, 62.6, 73.6, 124.7, 125.2, 127.2, 128.5, 140.1, 180.9; (ESI) m/z: calculated for protonated C12H16N2OS [M + H]+ 237.3330, found 237.3334.
1-(2-hydroxy-2,3-dihydro-1H-inden-1-yl)-3-(naphthalen-1-yl) thiourea (2e)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 1-Naphthylisothiocyanate (92.62 mg, 0.5 mmol) in dry toluene (5 mL) was stirred at room temperature for 4 h. Then after completion of the reaction, the precipitates were filtered with the help of a filter paper and dried. The white solid obtained (148 mg, yield 90%), m.p: 169 °C; IR: 3255.8, 2976.1, 1975.1, 1537.2 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.82 (d, 1H, CH2, J = 16.6 Hz), 3.15 (dd, 1H, J = 3.54 Hz, CH2), 4.73 (s, 1H, CH), 5.98 (m, 1H, OH), 6.48 (d, 1H, J = 6.4 Hz, CH), 7.13 (t, 1H, J = 7.1 Hz, ArH), 7.16–7.19 (s, 3 H, 2 ArH & 1NH), 7.23 (m, 1H, ArH), 7.48 (t, 1H, J = 7.7 Hz, ArH), 7.52 (d, 1H, J = 7.1 Hz, ArH), 7.48 (t, 1H, J = 7.4 Hz, ArH), 7.64 (t, 1H, J = 7.5 Hz, ArH), 7.85 (d, 1H, J = 8.1 Hz, ArH), 7.91(d, 1H, J = 8.1 Hz, ArH), 8.13 (d, 1H, J = 8.34 Hz, NH), 8.38 (d, 1H, J = 24.5 Hz); 13C NMR ppm; 39.5, 63.2, 73.4, 122.4, 124.5,125.1, 125.8, 127.1, 127.2, 127.5, 128.3, 128.6, 129.1, 129.7, 131.5, 134.6, 139.7, 139.8, 181.9; (ESI) m/z: calculated for protonated C20H18N2OS [M + H]+ 335.4370, found 335.4373.
1-(2-hydroxy-2,3-dihydro-1H-inden-1-yl)-3-(2-morpholinoethyl) thiourea (2f)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 2-Morpholinoethylisothiocyanate (121.12 µl, 0.5 mmol) in dry toluene (5 mL) was converted into compound 7, obtained (140 mg, yield 88%), m.p: 206 °C; IR: 3265.4, 2983.8, 1978.9, 1539.2, 1506.4 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.42 (s, 2 H, CH2), 2.59 (s, 2 H, CH2), 2.69 (t, 1H, J = 6.7 Hz, CH2), 2.60 (d, 1H, J = 6.6 Hz, NH), 2.98 (dd, 2 H, J = 2.5 Hz, CH2), 3.27 (s, 1H, J = 5.5 Hz, CH2), 3.62 (t, 1H, J = 6.1 Hz, CH2), 3.74 (s, 2 H, CH2), 4.81(d, 1H, CH), 5.81 (s, 1H, OH), 6.74 (s, 1H, CH), 7.25 (d, 1H, CH), 7.26 (d, 1H, NH), 7.30 (m, 2 H, CH), 7.37 (d, 1H, NH, J = 7.3 Hz), 8.86 (1H, NH); 13C NMR ppm; 39.4, 41.4, 42.9o, 53.32, 53.6, 57.6, 66.2, 66.9, 73.5, 125.1, 125.5,
(ESI) m/z: calculated for protonated C16H23N3O2S [M + H]+ 321.4550, found 321.4553.
1-(3-chlorophenyl)-3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl) thiourea (2g)
Compound 2 g was synthesized by the reaction of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 3-Chlorophenylisothiocyanate (89.4 mg, 0.5 mmol) in dry toluene (5 mL), at room temperature. After completion of the reaction, the precipitates were filtered and dried. White solid (140 mg, yield 87%), m.p: 128 °C; IR: 3251.9, 2983.8, 1541.1, 1508.3 cm− 1;1H NMR (CDCl3-d) δ ppm; 2.94 (d, 1H, J = 16.6 Hz, CH2), 3.23 (dd, 1H, J = 5.1 Hz, CH2), 4.78 (t, 1H, J = 4.2 Hz, CH), 5.97 (s, 1H, OH), 6.95 (d, 1H, J = 7.7 Hz, CH), 7.17–7.22 (m, 3 H, ArH), 7.25 (m, 1H, CH), 7.27–7.33 (m, 3 H, ArH,1H, NH), 7.39 (d, 1H, J = 7.1 Hz, ArH), 8.38 (s, 1H, NH); 13C NMR ppm; 39.8, 63.1, 73.5, 122.5, 124.6, 125.3, 125.4 126.9, 127.3, 128.2, 128.6, 129.0, 130.9, 135.5, 137.6, 139.7, 139.8, 180.6; (ESI) m/z: calculated for protonated C16H15ClN2OS [M + H]+ 319.8190, found 319.8193.
1-(2-hydroxy-2,3-dihydro-1 H-inden-1-yl)-3-(4-methoxyphenyl) thiourea (2h)
(1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 2-Morpholinoehylisothiocyanate (69 µl, 0.5 mmol) were reacted in dry toluene (5 mL), at 25 °C for 4 h with stirring. Filtered the reaction mixture and dried. The white solid obtained (135 mg, yield 86%), m.p: 124 °C; IR: 3255.8, 2985, 1535.3, 1510.2 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.83 (dd, 1H, J = 1.9 Hz, CH2), 3.14 (dd, 1H, J = 5.3 Hz, CH2), 4.72 (ddd, 1H, J = 2.2 Hz, CH), 5.8 (dd, 1H, J = 5.2 Hz, CH), 6.41 (d, 1H, J = 8.1 Hz, OH), 6.82 (d, 1H, J = 2.1 Hz, ArH), 7.13 (d, 2 H, J = 8.9 Hz, ArH), 7.14–7.17 (m, 3 H, ArH), 7.25 (d, 1H, NH), 7.75 (s, 1H, NH); 13C NMR ppm; 39.6, 55.5, 63.2, 73.5, 115.4, 124.6, 125.4, 127.3, 127.4,128.2, 128.5, 139.9, 140.1, 181.5; (ESI) m/z: calculated for protonated C17H18N2O2S [M + H]+ 315.4030, found 315.4033.
1-(4-chlorophenyl)-3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl) urea (3i)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 4-Chlorophenyleisocyanate (63.9 µl, 0.5 mmol) reacted in dry toluene (5 mL) with stirring for 4 h at room temperature. Filtered the precipitates and dried. The white solid obtained (139 mg, 93% yield), m.p: 244 °C; IR: 3261.6, 2982, 1539.1, 1508.3 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.86 (dd, 1H, J = 16.3 Hz, CH2), 3.05 (dd, 1H, J = 4.6 Hz, CH2), 4.49 (d, 1H, J = 4.4 Hz, CH), 4.78 (d, 1H, J = 4.5 Hz, OH), 5.16 (dd, 1H J = 5.1 Hz, OH), 6.46 (d, 1H, J = 8.6 Hz, NH), 6.82 (d, 1H, J = 7.9 Hz, Ar-H), 7.08–7.17 (m, 2 H, ArH), 7.25 (d, 1H, ArH), 7.65 (d, 2 H, NH), 8.68 (s, 1H, NH); 13C NMR ppm;, 39.9, 57.8, 73.8, 116.1, 117.8, 121.1, 125.1,126.5, 127.5, 129.8, 134.0, 140.4, 141.9, 142.6, 155.8; (ESI) m/z: calculated for protonated C16H15ClN2O2 [M + H]+ 303.7580, found 303.7582.
1-(4-fluorophenyl)-3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl) urea (3j)
Compound 3j was obtained by the mixing of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 4-Flourophenylisocyanate (56.8 µl, 0.5 mmol) in dry toluene (5 mL) and the precipitates were obtained after 4 h stirring of the reaction mixture at 25 °C. The white solid was 120 mg, 87% yield; m.p: 248 °C; IR: 3261.8, 2985.8, 1537.2, 1508.3 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.81 (d, 1H, J = 16.3 Hz, CH2), 3.15 (dd, 1H, J = 4.6 Hz, CH2), 4.48 (t, 1H, J = 4.4 Hz, CH), 4.77 (t, 1H, J = 4.3 Hz, OH), 5.16 (dd, 1H, J = 5.0 Hz, CH), 6.46 (d, 1H, J = 8.6 Hz, ArH), 6.82 (d, 2 H, J = 7.9 Hz, ArH), 7.69 (d, 1H, J = 6.5 Hz, ArH), 7.34–7.38 (m, 2 H, ArH), 7.66 (d, 1H, J = 5.22 Hz, NH), 8.53 (d, 1H, NH); 13C NMR ppm;, 40.1, 57.9, 72.8, 115.1, 115.1, 119.5, 119.5, 124.4, 125.1,126.5, 127.4, 136.7, 140.5, 142.8, 156.2; (ESI) m/z: calculated for protonated C16H15FN2O2 [M + H]+ 287.3064, found 287.3067.
1-(2-hydroxy-2,3-dihydro-1 H-inden-1-yl)-3-phenylthiourea (2k)
From the reaction of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) with Phenyl Isothiocyanate (60 µl, 0.5 mmol), compound 2k was obtained in the form of white solid (123 mg, 90% yield), in dry toluene (5 mL) after 4 h stirring at 25 °C. The m.p: 127 °C; IR: 3296.3, 2983.8, 2324.2, 1975.1, 1595.1, 1545 cm− 1; 1H NMR (DMSO-d6) δ ppm; 2.83 (d, 1H, J = 16.2 Hz, CH2), 3.12 (dd, 1H, J = 4.6 Hz, CH2), 4.54 (q, 1H, CH), 5.42 (d, 1H, CH), 5.87 (t, 1H, OH), 7.13 (t, 1H, ArH), 7.22 (p, 2 H, J = 7.5 Hz, ArH), 7.26 (d, 1H, J = 7.0 Hz, ArH), 7.31–7.35 (m, 3 H, 3CH, ArH), 7.57 (d, 2 H, J = 7.7 Hz, ArH), 7.81 (d, 1H, NH), 9.99 (s, 1H, NH); 13C NMR ppm;, 39.8, 61.8, 72.4, 123.3, 124.6, 124.7, 125.5., 126.8, 127.8, 129.1, 139.8, 140.9, 142.5, 180.9; (ESI) m/z: calculated for protonated C16H16N2OS [M + H]+ 285.3770, found 285.3773.
1-benzyl-3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl) thiourea (2L)
According to the above procedure compound 2L was obtained (142 mg, 86% yield), by the reaction of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and Benzyl Isothiocyanate (89.5 mg, 0.5 mmol). m.p: 121 °C; IR: 3342.6, 2991.6, 2320.3, 2029.1, 1977, 1579.7 cm− 1; 1H NMR (CDCl3-d) δ ppm; 2.81 (dd, 1H, J = 16.6 Hz, CH2), 3.09 (dd, 1H, J = 4.4 Hz, CH2), 4.49 (q, 1H, J = 4.4 Hz, CH), 4.77 (s, 1H, OH), 5.3 (s, 1H, CH), 5.82 (s, 1H, ArH), 7.15–7.20 (p, 2 H, J = 7.4 Hz, ArH), 7.24 (d, 2 H, J = 6.9 Hz, ArH), 7.29 (t, 1H, J = 6.5 Hz, ArH), 7.38 (q, 4 H, J = 7.3 Hz, ArH), 7.69 (d, 1H, J = 1.86 Hz, NH), 8.3 (s, 1H, NH); 13C NMR ppm;, 39.8, 72.5, 124.5, 125.4, 126.6, 127.4, 127.9, 128.8, 140.5, 173.3; (ESI) m/z: calculated for protonated C17H18N2OS [M + H]+ 299.4040, found 299.4043.
1-(2-hydroxy-2,3-dihydro-1 H-inden-1-yl)-3-(m-tolyl) Urea (3m)
Compound 3 m was synthesized by the reaction of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and m-Tolyl Isocyanate (64.44 µl, 0.5 mmol) into product in dry toluene (5 mL) at room temperature for 4 h. Precipitates were obtained and filtered/dried (115 mg, 90% yield), m.p: 236 °C; IR: 3346.5, 2983.8, 2318.4, 1977.1, 1589.3 cm− 1; 1H NMR (DMSO-d6) δ ppm; 2.26 (s, 3 H, CH3), 2.81 (d, 1H, J = 16.1 Hz, CH2), 3.08 (dd, 1H, J = 4.7 Hz, CH2), 4.45 (q, 1H, J = 8.3 Hz, CH), 5.1 (q, 1H, J = 4.9 Hz, CH), 5.10 (dd, 1H, J = 4.3 Hz, OH), 5.27 (d, 1H, J = 4.3 Hz, CH), 6.44 (d, 1H, J = 8.6 Hz, ArH), 6.74 (d, 1H, J = 7.4 Hz, ArH), 7.13 (t, 1H, J = 7.8 Hz, ArH), 7.18–7.24 (m, 4 H, ArH), 7.23 (m, 1H, NH), 7.28 (s, 1H, ArH), 8.7 (s, 1H, NH); 13C NMR ppm;, 39.8, 57.7, 72.5, 115.4, 118.4, 122.2, 124.4, 125.3, 126.7, 127.3, 140.9, 143.6, 155.7; (ESI) m/z: calculated for protonated C17H18N2O2 [M + H]+ 283.3430, found 283.3432.
1-(2-hydroxy-2,3-dihydro-1H-inden-1-yl)-3-(4-nitrophenyl) Urea (3n)
(1R,2 S)- cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 4-Nitrophenylisocyanate (82 mg, 0.5 mmol) were reacted to give compound 3n in dry toluene (5 mL) at room temperature by 4 h stirring. The yellowish solid was obtained (144 mg, yield 94%), m.p: 243 °C; IR: 3311.7, 2991.6, 2322.3, 1979, 1558.5 cm− 1; 1H NMR (DMSO-d6) δ ppm; 2.83 (d, 1H, J = 16.2 Hz, CH2), 3.11 (dd, 1H, J = 16.2 Hz, CH2), 4.47 (s, 1H, CH), 5.12 (s, 1H, OH), 5.35 (s, 1H, CH), 6.72 (s, 1H, J = 7.45, NH), 7.20–7.24 (t, 4 H, J = 7.8 Hz, ArH), 7.68 (d, 2 H, ArH), 8.19 (t, 2 H, CH), 9.64 (s, 1H, NH); 13C NMR ppm; 40.1, 57.6, 72.4, 117.2, 124.4, 125.4, 125.7, 126.8, 127.7, 128.6, 140.9, 143.1, 147.6,155.7; (ESI) m/z: calculated for protonated C16H15N3O4 [M + H]+ 314.3130, found 314.3132.
1,1′-(1,4-phenylene)bis(3-(2-hydroxy-2,3-dihydro-1H-inden-1-yl)urea) (4o)
A mixture of (1R,2 S)-cis-1-amino-2-indanol (74.5 mg, 0.5 mmol) and 1,4-Phenylenediisocyanate (80.1 mg, 0.5 mmol), in dry toluene (5 mL), at room temperature, was reacted to afford compound 4o (white solid precipitates; 144 mg, yield 95%), m.p: 267 °C; m.p: 267 °C; IR: 3338.7, 3280.9, 2941.4, 1637.5, 1564.2 cm− 1; 1H NMR (DMSO-d6) δ ppm; 2.81 (dd, 2 H, J = 16.1 Hz, CH2), 3.08 (dd, 2 H, J = 3.9 Hz, CH2), 4.43 (d, 2 H, J = 3.4 Hz, CH2), 5.09 (q, 2 H, J = 12.8 Hz, OH), 5.24 (d, 2 H, J = 3.1 Hz, CH), 6.35 (d, 2 H, J = 8.6 Hz, NH), 7.19–7.23 (m, 8 H, J = 11.7 Hz, ArH), 7.32 (s, 4 H, ArH), 8.69 (s, 2 H, NH); 13C NMR ppm; 40.2, 57.7, 72.6, 118.8, 124.4, 125.3, 126.7, 127.5, 134.9, 140.9, 143.7, 155.9; (ESI) m/z: calculated for protonated C26H26N4O4 [M + H]+ 459.5180, found 459.5183.
α-Glucosidase inhibition of synthetic derivatives
The α-glucosidase inhibitory activity of the synthesized compounds was measured in 50 mM phosphate buffer, pH 6.8. The enzyme (from Saccharomyces cerevisiae) was diluted to 1 U/2 mL in the phosphate buffer, and 20 L of enzyme was added per well. Each sample was incubated in a reaction buffer of 135 L of phosphate buffer per well with 20 L of each sample dissolved in 0.5 mM DMSO and transferred to 96-well plate. The plates were then incubated at normal temperature of 37 °C for 15 min. After incubation, 0.7 mM of the substrate “p-nitrophenyl-D-glucopyranoside” was added to the plates and the absorbance was measured at 400 nm wavelength. Acarbose and 7% DMSO were used as positive controls23.
Molecular docking
The 3D coordinate (PDB ID: 3A4A) of α-glucosidase (Saccharomyces cerevisiae) complexed with the inhibitor (maltose) was retrieved from the Protein Data Bank (RCSB) at a resolution of 1.60 Å11. The Loop modeler tool within the Molecular Operating Environment (MOE) version 2024.06 was utilized to reconstruct the absent residues in the protein structure to complete overall structure12,24. Amber14:EHT forcefield was applied which integrates the ff14SB Amber forcefield with Extended Huckel Theory (EHT) for molecular modeling, enabling precise predictions and optimizations of the geometry, energy, and interactions of biomolecules, especially proteins and nucleic acids. The C- and N- terminals of α-glucosidase were charged electrically to indicate the appropriate beginning and ending positions25.
Ligands were prepared in ChemDraw and imported into MOE for 3D-minimization of structures. AM1-BCC charges were added to each structure, and their structures were minimized with the MMFF-94x forcefield with an RMS gradient of 0.1 Kcal/mol/Å2. Afterwards, docking was carried out using the Triangle Matcher algorithm with the London dG scoring function with a total of 30 conformations for each ligand26. The appropriate binding mode of each molecule was further selected by conformational sampling. Protein-ligand interactions between our docked derivative and α-glucosidase were quantitatively evaluated using Protein-Ligand Interaction Fingerprints (PLIF) in MOE27 which measures several types of interactions like hydrogen bonds, water-ligand-residue bridges, surface contact, metal coordination and arene interactions28.
Results and discussion
Aminoindanol scaffolds are recognized as versatile building blocks in various chemical and biological applications due to their unique structural, chemical, and biological properties. These properties include their inherent rigidity, the spatial arrangement of two stereogenic centers, and the ability of hydroxy and amino groups to coordinate with metals as well as hydrogen-bond donors/acceptors. Additionally, their distinct catalytic activities and potential for derivatization further enhance their utility. Aminoindanol derivatives have been widely utilized in asymmetric catalysis and as ligands in organometallic catalytic complexes. Notable examples include their application in transfer-hydrogenation reactions catalyzed by bifunctional chiral ruthenium complexes, which are employed in synthesizing peptide mimics containing the intriguing trifluoroethylamine moiety29,30,31,32,33. The aminoindanol core is also a recurrent motif in several organocatalytic species, gaining prominence in asymmetric organo-catalysis34,35.
Key examples of cis-aminoindanol applications include the enantioselective reduction of ketones via the in situ formation of catalytically active oxazaborolidines36,37 and the synthesis of cooperative thiourea-urea-based organo-catalysts38. Similarly, the use of trans-aminoindanol as an efficient organo-catalyst has been reported in the enantioselective synthesis of natural products, such as the TMC-954 core39,40. These and other examples41 show the high catalytic potential of this versatile motif and prompted us towards the synthesis of its derivatives in the present study (Scheme 1).
Synthesis of compounds 2a-4o.
Chemistry
The urea and thiourea derivatives were prepared based on the versatile applications of the aminoindanols and the iso(thio)cyanate in organic and medicinal chemistry. The chemical reactions were carried out at room temperature employing dry toluene as the solvent. The equimolar amounts of the aminoindanols and variously substituted iso(thio)cyanate were reacted for approximately 4 h to afford the desired products in good to excellent yields. The molecular structure of compounds 2c, 2f, and 3n were confirmed by single crystal X-ray diffraction studies. Single crystals of compound 2c, 2f, and 3n were grown via slow evaporation from ethyl hexane-acetate mixture (1:9) solvent (Fig. 2 and Table S1).
X-ray crystal structures of 2c (A), 2f (B), and 3n (C).
In vitro α-glucosidase Inhibition of synthetic derivatives
The α-glucosidase inhibitory potential of compounds was assessed, with acarbose being used as a standard inhibitor. Except for 2d, all the compounds showed inhibitory potential for α-glucosidase better than that of the reference inhibitor. Compound 2h exhibited the highest inhibitory potency with IC50 of 9.64 ± 0.24 µM, followed by 2 g, 2c, 3i and 2a that exhibited IC50 in range of 11.60 ± 0.47 to 13.20 ± 0.28 µM. Later compounds 3n, 3j, 3 m and 2k also displayed IC50 values from 15 to 17.73 µM, indicating their significant inhibitory capability. While five compounds (2L, 2b, 2f, 2e and 4o) exhibited the IC50 values from 23 to > 35 µM. The % inhibition and IC50 values of compounds are given in Table 1.
Kinetic analysis
To elucidate the inhibition mechanism, the most potent compound 2h was subjected to detailed in vitro kinetic analysis. The compound exhibited a concentration-dependent inhibitory effect, consistent with a competitive mode of inhibition, with a Ki value of 7.39 ± 0.088 µM. In this inhibition type, the inhibitor competes with the substrate for binding at the enzyme’s active site, leading to an increase in Km while the Vmax remains unchanged (Fig. 3). These findings confirm that compound 2h directly interacts with the catalytic site residues, thereby preventing substrate access without altering the enzyme’s maximal catalytic efficiency.
The inhibition of α-glucosidase by compounds 2h (A) Lineweaver-Burk plot presented 1/V (velocities) on their Y-axis, while that of reciprocal of substrate (4-Nitrophenyl α-D glucopyranoside ) on their X-axis. Order of the concentrations of inhibitor include in the absence of compound (■), and in the presence of 20.00 µM (○), 10.00 µM (●), and 5.00 µM (□) of compounds 2h. (B) Secondary replot of Lineweaver-Burk was plotted between the slopes (Km/Vmax) on Y-axis and 1/Inhibitor on X-axis, and (C) Dixon plot presented 1/V on Y-axis, while 1/Inhibitor on X-axis.
Molecular docking results and predictive structure-activity relationship
All the compounds (except 2d) displayed good inhibitory potential for α-glucosidase; hence, their mode of binding was predicted by molecular docking within the active site of α-glucosidase. The residues ASP352, ASP69, ARG213, HIS351, ARG442, TYR72, PHE178, and HIS112 of α-glucosidase interact with the inhibitor (maltose). To ensure the effectiveness of our docking method, we conducted a thorough evaluation by re-docking the co-crystallized ligands in the active pocket. The RMSD value between the co-crystalized and re-docked ligands calculated is 0.28 Å (Fig. 4), indicating accuracy of applied docking method as the docking method accurately predicts the experimentally determined orientations of co-crystalized ligand.
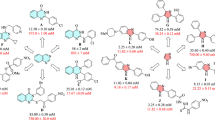
Our new compounds exhibited good binding interaction with the active pocket of α-glucosidase with good docking scores. Compound 2h shows the highest activity with the IC50 value of 9.64 ± 0.24 µM. When tested in the kinetic analysis, this molecule showed competitive mode of inhibition. Interestingly when docked with α-glucosidase, this molecule fitted well in the active site and mediated excellent interaction with GLU277 which is a part of catalytic tried (ASP215, ASP352 and GLU277). The thiourea moiety of 2h which links the indanol group with R group, participates in the binding of molecule with the surrounding residues. In compound 2h, one of the amino groups donate hydrogen bond to the side chain of GLU277 while thiol group accepted a hydrogen bond from the side chain of GLN353. Moreover, the indanol –OH group also mediates a hydrogen bond with the side chain of GLU279. The methoxy phenyl (R group) is nicely fitted between several hydrophobic residues including TYR72, HIS112, TYR158, PHY159 and PHY178. Due to these excellent interactions and well fitted orientation of compound, the ligand 2h produces highly negative docking score of − 6.52 Kcal/mol, which is the highest docking score among all the docked molecules, further confirming the robustness of the docking protocol. Compound 2g shows the second highest activity in in-vitro findings. We observed similar binding pattern of 2g and 2h with minor conformational variation. Both the amino moieties of 2 g mediated hydrogen bonding with two residues of catalytic triad (GLU277 and ASP352). While thiol and the indanol groups did not show any polar interactions with the surrounding residues. The chlorophenyl ring of 2 g is fitted well among the hydrophobic residues. This molecule produced the docking score of -6.41 Kcal/mol. The docking score and binding interaction clearly reflect differences in the inhibitory potency of ligands 2 h and 2 g.
Later, compound 2c, 2a, 2k, 2b, 2f, 2e and 2L showed IC50 values in range of (12 to > 23 µM). We observed the conformational changes to predict the structure activity relationship (SAR) among these compounds. In compound 2c (docking score= − 6.37 kcal/mol), we observed the slight variation in its orientation as compared to 2g. The indanol linked amino group formed a hydrogen bond with ASP352, while the hydroxyl group formed a hydrogen bond with the side chain of GLU411. The thiol group lost interaction with GLU277 or GLU279, which may be the reason for slightly lesser activity of this inhibitor than the 2g and 2h. In compound 2a (docking score= − 6.25 Kcal/mol), 2k (− 6.12 kcal/mol) 2 L (− 6.09 kcal/mol) and 2b (− 6.06 kcal/mol), the indanol moiety slipped towards the entrance of active site. Due to this movement, the indanol –OH, the amino and the thiol moieties did not interact with the surrounding residues, however the other amino group formed a hydrogen bond with the side chain of ASP352 in 2a, Glu277 in (2k and 2b) and ASP352 in 2L. In these compounds, we observed that the R moiety has moved out of the hydrophobic groove. In compound 2f, the propyl morpholine is added as a R group. The addition of morpholine group has made the molecule adopt a more bent orientation, thus, the thiourea and morpholine moieties did not interact with residues while indanol –OH group retained hydrogen bond with the side chain of GLU114 with a docking score of − 6.06 kcal/mol. The addition of naphthalene moiety in 2e made the molecule tilted conformation to be fitted inside the active site. The R group (i.e. naphthalene) instead of fitting in the core of active site, faces the entrance of active site while indanol group is oriented towards the core of active site. Due to this drastic conformational change, this molecule has lost all the polar interactions, while only HIS351 and PHE303 provided hydrophobic interactions to this molecule. The docking score of 2e (− 5.81 kcal/mol) reflects its lesser activity as observed in in-vitro.
The docking results of compounds 3i, 3n, 3j and 3 m were compared, these molecules exhibited IC50 in range of 12 to > 17 µM. Structurally, compound 3i is similar to 2a with the replacement of thiourea moiety in 2a with urea moiety in 3i. Interestingly, both 2a and 3i showed exactly similar conformation and docking score ( 3i= − 6.35 kcal/mol) where one of the amino group mediates hydrogen bonding with ASP352 in 3i. Interestingly, the benzoic moiety (R-group) of 3n formed a hydrogen bond with the side chain of HIS112 while its amino group interacted with GLU277. Compound 3n exhibited a docking score of − 6.52 kcal/mol which is slightly higher than that of 2a. Compound 3j is similar to 3b with the exception of thiourea moiety replaced with urea. In 2b, the thiourea moiety interacts with GLU277 while in 3j, the urea moiety formed bi-dentate interaction with the side chain of ASP352 which is making 3j more active than 3b, this is also reflected by the docking score of 3j (− 6.23 kcal/mol). In compound 3 m, the amino group interact with the side chain of GLU411 with the docking score of − 6.21 kcal/mol. At last, compound 4o showed lesser activity than the rest of the compounds and compound 2d was found inactive in this study. As the molecular size increased in 4o, the molecule moved more towards the entrance of active site. One of the indanol moieties remained surface exposed, while the other indanol moiety and a thiol group mediated hydrogen bonds with the side chain of GLU411 and GLN279, respectively. Furthermore, the benzamide group was stabilized by a hydrophobic interaction provided by HIS280. It is noted that the urea and thiourea moieties play vital role in binding with the one of residue of catalytic triad while indanol moiety (-OH group) further stabilize the molecule at active site entrance loop. We obtained a clear correlation between the experimental IC50 value and the docking score of compounds. The docking results are tabulated in Table 2 and the binding mode of the most active compound (2h) is depicted in Fig. 4.


(A) Re-docked conformation of co-crystalized ligand (blue) with RMSD of 0.28 Å is shown at its X-ray conformation (red) (B) The binding mode of compound 2h is shown in the active site of α-glucosidase enzyme.
Conclusion
The convenient synthesis of iso(thio)cyanates derivatives of aminoindanols have been reported in the present study. The NMR spectroscopy and other spectral techniques were used for the structure elucidation of the synthesized products. The α-glucosidase inhibition studies suggest 2h, 2 g, 2c and 3i as promising anti-diabetic molecules. While the rest of the compounds (except 2d) also showed good to moderate to low inhibition of the α-glucosidase enzyme. The kinetic analysis of the most potent inhibitor (2h) reflects its competitive mode of inhibition, suggesting that these molecules directly bind at the active site of enzyme which is also confirmed by molecular docking analysis. Docking results highlight the crucial role of the thiourea and urea moieties in mediating interactions with key residues, identifying them as the primary pharmacophoric features of these scaffolds. The current study, thus, provided an insight into the biological potential of the synthesized molecules, and provided a basis for designing new and improved α-glucosidase inhibitors.
Data availability
No datasets were generated or analyzed during the current study.
References
Saddique, F. A. et al. Recent trends in ring opening of epoxides by amines as nucleophiles. Synth. Commun. 46, 831–868 (2016).
Weng, C., Zhang, H., Xiong, X., Lu, X. & Zhou, Y. Evolution of epoxides to synthesize β-amino alcohols. ChemInform 46 (2015).
Karjalainen, O. K. & Koskinen, A. M. Diastereoselective synthesis of vicinal amino alcohols. Org. Biomol. Chem. 10, 4311–4326 (2012).
Gallou, I. & Senanayake, C. H. cis-1-Amino-2-indanol in drug design and applications to asymmetric processes. Chem. Rev. 106, 2843–2874 (2006).
Ghosh, A. K., Fidanze, S. & Senanayake, C. H. cis-1-Aminoindan-2-ol in asymmetric syntheses. Synthesis 1998, 937–961 (1998).
Yun, H., Kim, J., Kinnera, K. & Kim, B. G. Synthesis of enantiomerically pure trans-(1R, 2R)‐and cis‐(1S, 2R)‐1‐amino‐2‐indanol by lipase and ω‐transaminase. Biotechnol. Bioeng. 93, 391–395 (2006).
Dorsey, B. D. et al. L-735,524: the design of a potent and orally bioavailable HIV protease inhibitor. J. Med. Chem. 37, 3443–3451 (1994).
Reider, P. J. Advances in AIDS chemotherapy: the asymmetric synthesis of CRIXIVAN®. Chimia 51, 306–306 (1997).
Abdel-Rahman, H. M. et al. Design of inhibitors against HIV, HTLV-I, and Plasmodium falciparum aspartic proteases (2004).
Mendas, I., Gastaldi, S. & Suppo, J. S. Strategies for accessing cis-1-Amino-2-Indanol. Molecules 29, 2442 (2024).
Selvaraju, K., Raguraman, V., Yadav, H. N., Hariprasad, P. & Malik, A. Spectral characterization and binding dynamics of bioactive compounds from chlorella minutissima against α-glucosidase: an in vitro and in Silico approach. Algal Res. 75, 103281 (2023).
Edmonds, M. A. Virtual Screening of Hamigeromycin B Natural Product Derivatives in JNK3, EGFR, and Hsp90 (Tennessee Technological University, 2023).
Organization, W. H. Global health estimates: deaths by cause, age, sex and country, 2000–2012. Geneva, WHO 9 (2014).
Kidane, Y. et al. In Vitro Inhibition of α-Amylase and α‐Glucosidase by Extracts from Psiadia punctulata and Meriandra bengalensis. Evid. Based Complement. Alter. Med. 2018, 2164345 (2018).
Rehman, N. U. et al. New α-glucosidase inhibitors from the resins of Boswellia species with structure–glucosidase activity and molecular Docking studies. Bioorg. Chem. 79, 27–33 (2018).
Kim, K., Nam, K., Kurihara, H. & Kim, S. Potent α-glucosidase inhibitors purified from the red Alga Grateloupia elliptica. Phytochemistry 69, 2820–2825 (2008).
Martin, A. E., Montgomery, P. A. & Acarbose An α-glucosidase inhibitor. Am. J. health-system Pharm. 53, 2277–2290 (1996).
Chougale, A. D., Ghadyale, V. A., Panaskar, S. N. & Arvindekar, A. U. Alpha glucosidase Inhibition by stem extract of Tinospora cordifolia. J. Enzyme Inhib. Med. Chem. 24, 998–1001 (2009).
Santoso, M. et al. Synthesis, α-glucosidase inhibition, α-amylase inhibition, and molecular Docking studies of 3, 3-di (indolyl) indolin-2-ones. Heliyon 8 (2022).
Li, M. et al. Anti-α-glucosidase, SAR analysis, and mechanism investigation of Indolo [1, 2-b] isoquinoline derivatives. Molecules 28, 5282 (2023).
Sayahi, M. H. et al. Design, synthesis, in vitro, and in Silico anti-α-glucosidase assays of N-phenylacetamide-1, 2, 3-triazole-indole-2-carboxamide derivatives as new anti-diabetic agents. Sci. Rep. 14, 15791 (2024).
Kamimura, H., Ogata, H. & Takahara, H. Alpha-glucoside formation of xenobiotics by rat liver alpha-glucosidases. Drug Metab. Dispos. 20, 309–315 (1992).
Lazar, A., Sharma, P., Singh, A. & Chiral VIVO-Sal-Indanol complex over modified SBA-15: an efficient, reusable enantioselective catalyst for asymmetric sulfoxidation reaction. Microporous Mesoporous Mater. 170, 331–339 (2013).
Vu, T. Y. et al. Estimation of the binding affinities of glycogen phosphorylase inhibitors by molecular Docking to support the treatment of type 2 diabetes. Phys. Chem. Res. 12, 821–835 (2024).
Ullah, A. et al. Novel natural inhibitors for glioblastoma by targeting epidermal growth factor receptor and phosphoinositide 3-kinase. Curr. Med. Chem. (2024).
Corbeil, C. R., Williams, C. I. & Labute, P. Variability in Docking success rates due to dataset Preparation. J. Comput. Aided Mol. Des. 26, 775–786 (2012).
El-Hassab, M. A. E. M., El-Bastawissy, E. E. & El-Moselhy, T. F. Identification of potential inhibitors for HCV NS5b of genotype 4a by combining dynamic simulation, protein–ligand interaction fingerprint, 3D pharmacophore, Docking and 3D QSAR. J. Biomol. Struct. Dynamics. 38, 4521–4535 (2020).
Ye, W. L. et al. Improving Docking-based virtual screening ability by integrating multiple energy auxiliary terms from molecular Docking scoring. J. Chem. Inf. Model. 60, 4216–4230 (2020).
Dai, X. & Cahard, D. Enantioselective synthesis of α-Trifluoromethyl arylmethylamines by Ruthenium‐Catalyzed transfer hydrogenation reaction. Adv. Synth. Catal. 356, 1317–1328 (2014).
Sani, M., Volonterio, A. & Zanda, M. The trifluoroethylamine function as peptide bond replacement. ChemMedChem: Chem. Enabling Drug Discovery. 2, 1693–1700 (2007).
Piras, M., Fleming, I. N., Harrison, W. T. & Zanda, M. Linear trifluoroethylamine RGD peptidomimetics: stereoselective synthesis and integrin αvβ3 affinity. Synlett 23, 2899–2902 (2012).
Dalko, P. I. Comprehensive Enantioselective Organocatalysis: Catalysts, Reactions, and Applications, 3 Volume Set (Wiley, 2013).
Gaunt, M. J., Johansson, C. C., McNally, A. & Vo, N. T. Enantioselective organocatalysis. Drug Discovery Today. 12, 8–27 (2007).
Berkessel, A. & Gröger, H. Asymmetric organocatalysis: From biomimetic concepts to applications in asymmetric synthesis (2005).
Jørgensen, D. K. B. Asymmetric Organocatalysis–Synthesis of Centrally and Axially Chiral Compounds (2020).
Sibi, M. P., Cook, G. R. & Liu, P. Enantioselective reduction of ketones. Examination of bifunctional ligands. Tetrahedron Lett. 40, 2477–2480 (1999).
Turgut, Y., Azizoglu, M., Erdogan, A., Arslan, N. & Hosgoren, H. β-Hydroxyamide derivatives of Salicylic acid as organocatalysts for enantioselective reductions of prochiral ketones. Tetrahedron: Asymmetry. 24, 853–859 (2013).
Probst, N. et al. Cooperative assistance in bifunctional organocatalysis: enantioselective Mannich reactions with aliphatic and aromatic Imines. Angew. Chem. Int. Ed. 51, 8495–8499 (2012).
Kohno, J. et al. Structures of TMC-95A – D: novel proteasome inhibitors from Apiospora m ontagnei Sacc. TC 1093. J. Org. Chem. 65, 990–995 (2000).
Coste, A., Couty, F. & Evano, G. TMC-95A–D and analogues: chemistry and biology. Comptes Rendus Chim. 11, 1544–1573 (2008).
Coste, A., Bayle, A., Marrot, J. & Evano, G. A convergent synthesis of the fully elaborated macrocyclic core of TMC-95A. Org. Lett. 16, 1306–1309 (2014).
Acknowledgements
The authors are thankful to The Oman Research Council (TRC) through the Research Grant Program (BFP/RGP/CBS/18/096). The authors also extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through large Research project grant number RGP2/191/46.
Funding
The authors are thankful to The Oman Research Council (TRC) through the Research Grant Program (BFP/RGP/CBS/18/096). The authors also extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through large Research project grant number RGP2/191/46.
Author information
Authors and Affiliations
Contributions
S.K.: Methodology, Writing—original draft, F.S.: Conceptualization, Supervision, Project administration, Writing—review & editing. L.A.: Methodology, Formal analysis, A.K.: Software, Validation, Writing—original draft. M.U.A.: Methodology, Formal analysis, Data curation. A.U.: Methodology, Validation, Software. S.A.H.: Software, Formal analysis and Writing—review & editing. F.K.: Methodology, Data curation. S.U.: Methodology, Data curation. J.U.: Funding acquisition, Resources, Formal analysis, A.A.-H.: writing—review & editing, Supervision & Project administration. J.H.: writing—review & editing, Supervision & Project administration.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Khan, S., Shaheen, F., Ali, L. et al. Synthesis and pharmacological profiling of cis-1-amino-2-indanol derivatives as α-glucosidase inhibitors. Sci Rep 16, 271 (2026). https://doi.org/10.1038/s41598-025-29712-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-29712-w