Abstract
Lysyl oxidase-like 2 (LOXL2) is a key enzyme involved in extracellular matrix remodeling, and its high expression is associated with cancer progression. This study aims to assess the relationship between LOXL2 expression and clinical prognosis in cancer patients through meta-analysis and bioinformatics. A comprehensive literature search was conducted in PubMed, Cochrane Library, Web of Science, and Embase databases until December 2023. Studies reporting the relationship between LOXL2 expression and overall survival (OS) or disease-free survival (DFS) were included. Hazard ratios (HR) and odds ratios (OR) with 95% confidence intervals (CIs) were calculated. Subgroup analyses and meta-regression were performed to evaluate sources of heterogeneity. A total of 30 studies with 5021 patients were included. High LOXL2 expression was significantly associated with poor OS (HR = 1.92, 95% CI: 1.65–2.23, P < 0.001) and DFS (HR = 1.81, 95% CI: 1.39–2.36, P < 0.001). LOXL2 expression correlated with several clinicopathological features, including tumor size (OR = 1.53, P = 0.006), lymph node metastasis (OR = 1.71, P = 0.016), and distant metastasis (OR = 2.63, P < 0.001). Sensitivity analysis confirmed the robustness of these results. High LOXL2 expression is a reliable prognostic biomarker for various cancers, correlating with poor survival outcomes and advanced clinical features. LOXL2 may serve as a therapeutic target to improve cancer prognosis and treatment.
Similar content being viewed by others
Introduction
Cancer is one of the major causes responsible for mortality worldwide, causing a serious economic burden on countries around the world1. The incidence rate of cancer among people under 50 years old has been rising2. It is estimated that by 2040, the number of cancer cases in the world will reach 28.4 million3. The current reported cancer treatment methods mainly include surgery, radiation therapy and systemic therapy, including chemotherapy, targeted therapy, hormone therapy and immunotherapy4. However, with the increase of incidence rate and mortality, cancer remains a major public health problem.
Lysyl oxidase-like 2 (LOXL2) is a key extracellular matrix remodelling enzyme involved in the cross-linking of collagen and elastin, thereby influencing the formation and maintenance of the tumour microenvironment5,6. The LOX family consists of five members: LOX and lysine oxidase like proteins 1–4 (LOXL1, LOXL2, LOXL3, and LOXL4). The expression of LOX family members and their impact on enzyme activity are important factors in cancer progression7,8,9. In recent years, the importance of LOXL2 in cancer research has been gradually highlighted. Studies have shown that LOXL2 promotes tumour progression through multiple mechanisms, including promotion of epithelial-mesenchymal transition (EMT), enhancement of tumour cell invasion and metastasis, and regulation of tumour angiogenesis10,11. In breast cancer, high expression of LOXL2 was closely associated with enhanced invasiveness and metastasis of tumour cells12; in hepatocellular carcinoma, LOXL2 significantly affected tumour growth and metastasis by promoting angiogenesis and stromal remodelling13. In addition, there are significant differences in the expression levels and functions of LOXL2 in different cancer types, and in gastric and colorectal cancers, high LOXL2 expression is usually associated with poor prognosis14,15.
In recent years, an increasing number of literatures have shown that LOXL2 is highly expressed in various malignant tumors, and is correlated with clinical pathological features and prognosis, indicating its potential as a biomarker for prognosis and treatment16,17,18. Moreover, in anti-extracellular matrix therapy, LOXL2 may be a favorable target for preventing tumor metastasis19. Although a large number of studies have explored the role of LOXL2 in cancer, its specific mechanisms in different cancer types and its relationship with prognosis still need to be further clarified. Therefore, this study systematically assessed the relationship between LOXL2 expression and cancer prognosis through meta-analysis, aiming to provide more comprehensive evidence to support LOXL2 as a potential therapeutic target.
Materials and methods
Literature search
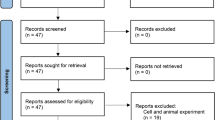
This meta-analysis was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The completed PRISMA checklist is provided as Supplementary Material 120. From its establishment until December 10, 2023, a comprehensive literature search was conducted on the Cochrane Library, Medline, Web of Science, Embase, and PubMed databases. The literature search terms are used in the following different combinations: (“tutor” or “cancer” or “neoplasm” or “cancer”) and (“LOXL2 protein” or “lysyl oxidation like 2 protein” or “LOXL2”) (Fig. 1). The detailed search strategies for all databases are provided in Supplementary Material 2 (Search Strategies). The literature is limited to English publications and human studies only.
The flow diagram of the eligible literature retrieval and selection processes.
Inclusion and exclusion criteria
Two researchers independently checked articles that met the criteria. When there are different opinions, reach a consensus through discussion with the third author.
The inclusion criteria are as follows: (1) All patients have been diagnosed with cancer through histopathological examination; (2) Grouping patients based on the expression level of LOXL2; (3) Detecting the relationship between LOXL2 expression and clinical pathological features and prognosis; (4) Complete data on overall survival (OS), odds ratio (OR), or hazard ratio (HR) can be obtained directly or indirectly with a 95% confidence interval (95% CI).
The exclusion criteria are as follows: (1) Non main research articles, such as case reports, conferences, abstracts, animal experiments, reviews, or editorials; (2) No LOXL2 grouping was performed; (3) There is no full text research; (4) Lack of prognostic results or repeated published studies.
Data extraction and quality evaluation
In this study, the main data was independently analyzed and extracted by two reviewers, including author name, publication year, cancer type, number of patients, sample size, clinical pathological features, and overall survival (OS) and Disease-free-survival (DFS); The availability of HR; HR (95% CI) and follow-up time. The survival HR (95% CI) is directly obtained from the study, and if there is a Kaplan Meier curve, it is calculated using the Engage Digitizer 11.1 software.
The quality of the included literature was evaluated using the Newcastle Ottawa Scale (NOS) score21 by two independent reviewers. The NOS assesses studies across three dimensions: (1) Selection (adequacy of case definition, representativeness of the cases, selection and definition of controls), (2) Comparability (control for the most important factor and any additional factor), and (3) Outcome (ascertainment of exposure, same method of ascertainment for all subjects, and non-response rate). Any discrepancies in the quality scores between the two reviewers were resolved through discussion until a consensus was reached, or by consultation with a third reviewer if necessary. The total NOS score ranges from 0 to 9 points, with a higher score indicating higher methodological quality. Consistent with common practice in meta-analyses, we pre-specified a score of ≥ 6 points as the threshold for indicating a study of satisfactory quality. All studies meeting the inclusion criteria also met this quality threshold and were therefore included in the final analysis.
Statistical analysis
All data extracted from the article were statistically analyzed using STATA 17.0 software. A P-value < 0.05 indicates that the difference is statistically significant. Analyze the relationship between LOXL2 expression and survival data OS/DFS using HR and corresponding 95% CI and describe the association between LOXL2 expression and clinical pathological features using OR and its 95% CI. The heterogeneity statistic was used to assess the variability of the results between studies; Cochran’s Q statistic were used to test the significance of heterogeneity, with P < 0.05 indicating significance, while I²statistic indicated the degree of heterogeneity, with I²>50% indicating high heterogeneity. When heterogeneity was significant, a random effects model was used to adjust the analysis. Use Begg’s and Egger’s tests to evaluate potential publication bias and use sensitivity analysis to evaluate the reliability of the results.
Sensitivity analysis for publication bias
Using STATA 17.0 software, Egger’s test was used to evaluate publication bias in the literature. Verify the stability of the results through sensitivity analysis. P < 0.05 indicates a statistically significant difference.
Enrichment analysis drug sensitivity prediction
Gene Ontology (GO) and Kyoto Encyclopedia of Genomes (KEGG) pathway enrichment analyses were performed using the R package “clusterProfiler” (v4.0.5), which screens for significantly enriched entries by the Hypergeometric Distribution Test (FDR-corrected P < 0.05), and which has the advantage of supporting the integration of multiorganomics data and interactive visualisation (parameter settings: minGSSize = 10, maxGSSize = 500). For drug sensitivity prediction, the CallMiner™ (v2.3.1) platform was selected to process the NCI-60 compound activity data in this study, which can quantitatively assess drug sensitivity association characteristics in the pan-cancer range by building a Spearman rank correlation model (|ρ|>0.3, P < 0.01) between the expression level of LOXL2 and the IC50 value of drugs. A central consideration in the selection of CallMiner™ was its integration of a database of 2,862 FDA-approved drugs and 1,974 clinical trial compounds, validated by the TCGA Pharmacogenomics Benchmark22. The “ggplot2” (visualisation), “impute” (KNN algorithm for missing values) and “ggpubr” (statistical plotting) software packages to assist in the analysis were installed via Bioconductor.
Results
Characteristics of studies
The detailed process of literature selection is shown in Fig. 1. The initial search result was 2651 articles, of which 1058 duplicate studies were excluded. In addition, 1364 unrelated articles were excluded after filtering the titles and abstracts. Among the remaining 229 articles, 189 reports were excluded due to non-primary research, failure to group patients based on LOXL2 expression levels, or to investigate the association between expression and prognosis, while 10 reports were excluded due to a lack of research data on prognosis or clinical pathological features. Finally, 30 articles were included in the current meta-analysis.
As shown in Table 1, a total of 30 articles and 5021 patients were included, with cancer types including colorectal cancer15,23,24,25,26, glioma27, lung cancer28,29, hepatocellular carcinoma30,31,32,33, cholangiocarcinoma34,35,36, pancreatic cancer37,38,39,40, cervical cancer41, head and neck squamous cell carcinoma42, esophageal cancer43,44,45,46,47, gastric cancer48,49, breast cancer12, oral cancer50. The publication period is from 2009 to 2023. The number of cases ranges from 37 to 548. Among the 30 studies included, one study investigated two groups of cases from different sources; 26 studies provided overall survival (OS), of which 17 directly provided HR values, 12 studies provided disease-free survival (DFS), of which 7 directly provided HR values, and other studies only provided K-M survival curves. One study used qPCR to detect the expression of LOXL2, while the remaining studies used immunohistochemistry (IHC) for detection and used immunostaining scores to determine cutoff values. Seven studies were unable to report specific cutoff values. Four studies scored 9 points, nine studies scored 8 points, eight studies scored 7 points, and the remaining nine studies scored 6 points. All studies clearly defined case and control groups and controlled for major confounders in their design and analyses. Most studies used independent and blinded assessment of outcomes and had a sufficiently long follow-up period with good completeness of follow-up.
The quality assessment using the NOS revealed that the overall methodological quality of the 30 included studies was high, with scores ranging from 6 to 9 points. The distribution of scores was as follows: four studies (13.3%) scored 9 points, nine studies (30.0%) scored 8 points, eight studies (26.7%) scored 7 points, and the remaining nine studies (30.0%) scored 6 points. All studies demonstrated adequate selection of cohorts and outcome assessment. The comparability of cohorts, particularly regarding the control for key prognostic factors like tumor stage, was well addressed in studies scoring 7 points or higher. The detailed scores for each domain for all included studies are presented in Supplementary Material 2 (Supplementary Table 1).
Association between LOXL2 and prognostic indicators
Association between LOXL2 expression and OS
26 articles involving 4657 patients reported the relationship between LOXL2 expression levels and the prognosis of cancer patients. The heterogeneity test showed an I²value of 52.3% (P = 0.001), indicating significant heterogeneity between studies. Therefore, random effects model was used for combined analysis. The results showed that high expression of LOXL2 predicted poor cancer survival (pooled HR = 1.92 95% CI 1.65–2.23, P < 0.001) (Fig. 2A). Subgroup analysis and meta regression showed a strong correlation between sample size and study area and OS, suggesting that sample size and study area may be sources of heterogeneity (Table 2). The results of subgroup analysis showed that high expression of LOXL2 was associated with poor OS in tumor patients.
Meta analysis forest plots of OS and DFS. (A) Correlation between LOXL2 expression and overall survival (OS). (B) The correlation expression between LOXL2 and disease-free survival (DFS).
Association between LOXL2 expression and DFS
12 studies have reported DFS data, which can be used to evaluate the prognostic value of LOXL2. The heterogeneity test showed an I²value of 65.1% (P = 0.001), indicating significant heterogeneity between studies. Therefore, random effects model was used for combined analysis. Our analysis showed that high expression of LOXL2 was associated with poor DFS (pooled HR = 1.81, 95% CI 1.39–2.36, P < 0.001) (Fig. 2B).
Association between LOXL2 expression and clinicopathological features
The relationship between LOXL2 expression and clinical pathological features is shown in Table 3. The results show that LOXL2 expression was significantly correlated with patient age (OR = 1.33, 95% CI 1.06–1.66, P = 0.014), tumor size (OR = 1.53, 95% CI 1.13–2.08, P = 0.006), lymphovascular involvement (OR = 2.62, 95% CI 1.11–6.19, P = 0.028), T stage (OR = 2.02, 95% CI 1.33–3.07, P = 0.001), lymph node metastasis (OR = 1.71, 95% CI 1.11–2.64, P = 0.016), distant metastasis (OR = 2.63, 95% CI 1.98–3.46, P = 0.016), TNM staging (OR = 1.83, 95% CI 1.28–2.62, P = 0.001) and venous invasion (OR = 3.19, 95% CI 1.60–6.36, P = 0.001). However, high expression of LOXL2 was not significantly correlated with gender (OR = 1.07, 95% CI 0.92–1.24, P = 0.4), histological grade (OR = 1.38, 95% CI 0.88–2.16, P = 0.16), differentiation (OR = 1.33, 95% CI 0.99–1.79, P = 0.061) and perineural invasion (OR = 1.24, 95% CI 0.70–2.19, P = 0.46) (Fig. 3).
Forest plots of the correlation between LOXL2 expression and clinical pathological parameters. (A) Age. (B) Gender. (C) T stage. (D) Lymph node metastasis. (E) TNM stage. (F) Differentiation. (G) Tumor size. (H) Distant metastasis. (I) Pathological grade. (J) Lymphovascular involvement. (K) Portal vein invasion. (L) Perineural invasion.
Sensitivity analysis and publication bias
To assess the stability of the meta-analysis results, Leave-one-out Analysis was used to assess the effect of individual articles on the combined overall survival (OS) results. The results of the sensitivity analyses showed that the range of variation in the pooled effect sizes after excluding any of the studies was small (HR = 1.85–1.95), and the 95% confidence intervals for all outcomes did not cross the null line (HR = 1). This suggests that the results of the sensitivity analyses were stable and none of the literature significantly altered the OS results, further validating the robustness of the overall results. Egger’s test was performed to assess the effect of publication bias on the results. The results showed that there was a significant publication bias in OS (P > |t| = 0.002). To assess the effect of publication bias on outcomes, this study used the Trim and Fill method to generate nine imputed studies. These studies were generated by Monte Carlo simulation, based on the effect size distribution parameters of real data, and were used to correct for potential publication bias. To assess the effect of publication bias on the combined results, this study used the “trim and fill method”. A total of nine imputed studies were generated by iteratively pruning the right extreme value studies and filling in the left missing data. The results showed a random-effects model with Q = 102.187, P < 0.001, and a composite effect indicator of HR = 1.597 (95% CI: 1.347–1.892, P < 0.001). Compared with the pre-correction results (HR = 1.92, 95% CI: 1.65–2.23), the corrected pooled effect sizes decreased slightly but remained consistent with the original results, indicating that the combined results were less sensitive to publication bias and that the overall results had high stability. In addition, the funnel plot showed a roughly symmetrical distribution, further supporting the plausibility of using a random-effects model to estimate HR across studies (Fig. 4). The results of these analyses indicate that the results of this meta-analysis have a high degree of stability and reliability, despite the presence of some publication bias.
Sensitivity analysis and publication bias test. (A) Sensitivity analysis for OS; (B) Sensitivity analysis for DFS; (C) The star plot test of OS Heterogeneity. (D) The Egger’s test and linear regression plot for the publication bias for OS. (E) The funnel plot of OS. (F) The trim-and-fill test of OS.
Prediction of LOXL2 function and drug sensitivity in cancer
The potential molecular mechanism of LOXL2 was determined through GO and KEGG enrichment analysis, as shown in Fig. 5. It is expected that LOXL2 will mainly participate in biological processes (BPs), such as extracellar matrix organization, extracellar structure organization, and external encapsulation structure organization; Cellular components (CCs), such as collagen containing extracellular matrix and endoplasic reticulum lumen; And molecular functions (MFs), extracellar matrix structural constitution, conferring tension strength growth factor binding (Fig. 5A and B). The KEGG pathway analysis results show that the interacting genes mainly participate in ECM receptor interaction, focal adhesion, PI3K Akt signaling pathway, and protein digestion and absorption (Fig. 5C and D). This indicates that LOXL2 mainly regulates the occurrence and development of tumors by influencing the remodeling of tumor extracellular matrix, which is consistent with previous reports51. In addition, using CellMiner ™, we further investigated the correlation between LOXL2 expression levels and drug sensitivity. The research results showed that the expression of LOXL2 was positively correlated with the drug sensitivity of P-529, BLU-667, Staurosporine, IDH-C227, spebrutinib, Telatinib, Lenvatinib, Momelotinib, dimethylfasudil, JNJ-3,887,618, IDH-Comp 301, and Dasatinib (Fig. 6A, B, C, E, F, G, H, I, K, L, O, P). The expression of LOXL2 is negatively correlated with Tamoxifen, CT-GSK183, AMG-900, By Product of CUDC-305 (Fig. 6D, J, M, N). The above-mentioned drugs, such as P-529, Staurosporine and spebrutinib, are mostly enzyme inhibitors, indicating that LOXL2’s involvement in chemotherapy resistance may be related to enzyme metabolism and transcriptional dysregulation. These data suggest that LOXL2 may be involved in the mechanism of chemotherapy resistance.
KEGG and GO term enrichment for LOXL2. (A) Barplots of GO enrichment. (B) Bubble charts of GO enrichment; (C) Barplots of KEGG molecular mechanisms. (D) Bubble charts of KEGG molecular mechanisms.
Drug sensitivity analysis of LOXL2. The correlation between the expression of LOXL2 and (A) P-529, (B) BLU-667, (C) Staurosporine, (D) Tamoxifen, (E) IDH-C227, (F) spebrutinib, (G) Telatinib, (H) Lenvatinib, (I) Momelotinib, (J)CT-GSK183, (K) dimethylfasudil, (L) JNJ-3,887,618, (M)AMG-900, (N) By-Product of CUDC-305, (O) IDH1-Comp 301, (P) Dasatinib.
Discussion
This meta-analysis, encompassing 30 studies with 5,021 patients, provides robust evidence that elevated LOXL2 expression serves as a significant prognostic indicator across multiple cancer types. Our pooled results demonstrate that high LOXL2 expression is substantially associated with poorer overall survival (HR = 1.92, 95% CI: 1.65–2.23) and disease-free survival (HR = 1.81, 95% CI: 1.39–2.36). Furthermore, LOXL2 expression significantly correlates with aggressive clinicopathological features including advanced T stage, lymph node metastasis, distant metastasis, and advanced TNM stage. These clinical observations are mechanistically supported by our bioinformatics analyses, which reveal LOXL2’s predominant involvement in extracellular matrix organization and PI3K-Akt signaling pathways - both established mediators of tumor progression and metastasis.
The extracellular matrix (ECM) is one of the main components of tumors, and the synthesis and modification of ECM proteins play multiple key roles in the development of cancer, including mechanical support and regulation of the microenvironment52. The LOX family is a key enzyme in ECM remodeling, which can promote covalent crosslinking and stabilization of ECM structural components and participate in various pathological processes related to connective tissue. The LOX family leads to tumor occurrence and metastasis by reshaping the tumor microenvironment53,54. LOXL2 is a copper dependent amine oxidase that can initiate cross-linking of collagen and elastin and induce connective tissue proliferation matrix and tumor progression49,55,56. Many studies have reported that LOXL2 promotes the progression, migration, and invasion of various tumors and is associated with poor prognosis57,58,59,60. Due to recent research finding that LOXL2 is highly correlated with various human tumors and significantly correlated with patient prognosis. Previous studies have found that high expression of plasma LOX after tumor surgery is more likely to promote tumor metastasis, and LOXL2 levels in serum sEV (Small extrasellar vessels) of HNSCC patients are significantly increased42,61, Therefore, LOXL2 may serve as a new tumor serum marker to predict the prognosis of various tumors.
In this study, we used the Egger’s test to detect publication bias in the included articles and found that there was significant publication bias (Pr > |t| = 0.002). However, it is important to note that if too many studies are included in the meta-analysis or if there is too much heterogeneity among the original studies, this may also lead to test failure and thus inaccurate conclusions. To further assess the impact of publication bias, we conducted additional analyses of all studies using the “trim and fill” method62. The results showed that after filling in the data from nine dummy studies, the random-effects model was used to test for heterogeneity, with Q = 102.187, P < 0.001, and the combined effect indicator was HR = 1.597 (95% CI: 1.347–1.892, P < 0.001). The results before and after correction did not change significantly (P < 0.001), and the combined results remained stable, suggesting that there may not be significant publication bias in the included articles. In addition, the funnel plot showed a roughly symmetrical distribution, further supporting the reliability of the results. After meta-analysis of the 26 eligible studies, the results showed that high expression of LOXL2 was significantly associated with poor patient prognosis, including overall survival (OS: HR = 1.92, 95% CI: 1.65–2.23, P < 0.001) and disease-free survival (DFS: HR = 1.81, 95% CI: 1.39–2.36, P < 0.001). However, in the OS analysis, we observed significant heterogeneity (I² = 52.3%, P = 0.001). To assess the stability of the results, we performed sensitivity analyses and recalculated the pooled effect sizes by excluding each study in turn using Leave-one-out Analysis. The results showed that the range of variation in the pooled effect sizes was small (HR = 1.85–1.95) and the 95% confidence intervals of all the results did not cross the null line (HR = 1) after the exclusion of any of the studies, suggesting that the results were less dependent on individual studies and that the overall results had a high degree of stability. The funnel plot showed that although some studies fell outside the confidence intervals, suggesting possible heterogeneity, the majority of studies were concentrated in the narrow area at the top of the funnel plot, indicating a large sample size of included studies and high reliability of the results. In conclusion, despite the publication bias and heterogeneity, the robustness of the meta-analysis results was verified by the trim-and-fill method and sensitivity analysis, and the significant association between high LOXL2 expression and poor prognosis of tumour patients has high scientific credibility. However, various tumours differ in their origins, molecular pathways and microenvironmental compositions, and LOXL-2 is involved in multiple signalling pathways such as stromal remodelling, epithelial-mesenchymal transition (EMT) and immune regulation, which may play different roles in different tumours. For example, in breast and lung cancers, LOXL-2 up-regulation is more likely to promote tumour cell invasion and metastasis, leading to a significantly poorer prognosis12; whereas in some other tumour types, LOXL-2 may be more involved in local fibrosis and microenvironmental regulation, and its impact on overall survival is relatively limited13,14,15. This heterogeneity in prognostic value suggests the complexity of LOXL-2 in clinical applications and provides an important reference for more targeted research and individualised treatment strategies in the future. Future studies should further adopt standardised protocols and homogeneous populations to validate the clinical value of LOXL2 as a prognostic marker for tumours.
Despite significant heterogeneity observed in this meta-analysis (I² = 75.6%, P < 0.001), meta-regression analysis revealed that sample size (adjusted R² = 41.41%, P = 0.03) and geographic region (adjusted R² = 24.43%, P = 0.015) were major contributors, while tumor type and HR estimation methods contributed minimally to heterogeneity. Subgroup analysis further demonstrated that, despite residual heterogeneity in some subgroups, the association between LOXL2 overexpression and poor prognosis remained consistent across all subgroups. This robustness enhances the reliability of the study conclusions. Heterogeneity likely stemmed from variations in LOXL2 detection methods, positivity criteria, threshold settings, and patient baseline characteristics. Notably, given the predominance of Asian populations in the included studies, these findings hold greater applicability within this demographic. Future studies should enhance comparability by standardizing detection protocols, defining positive criteria, and establishing consistent thresholds. Despite observed heterogeneity, the consistent findings across subgroups support LOXL2’s potential value as a prognostic biomarker.
In addition, we also analyzed the relationship between LOXL2 expression levels and clinical pathological features. The combined OR results showed that high expression of LOXL2 was significantly correlated with age, tumour size, vascular infiltration, T stage, lymph node metastasis, distant metastasis, TNM stage and venous invasion, suggesting that LOXL2 may play an important role in tumour progression and metastasis. LOXL2 enhances tumour cell invasion and metastatic ability, while its role in angiogenesis may explain its significant correlation with vascular infiltration and venous invasion10,11. In addition, our study also found that high expression of LOXL2 was significantly correlated with patient age, a result that may be related to age-related changes in the tumour microenvironment. With age, the functions of stromal cells and immune cells in the tumour microenvironment may change, which may affect LOXL2 expression and function. Also, age-related genomic instability may lead to upregulation of LOXL2 expression, suggesting that LOXL2 may serve as a potentially reliable tumour prognostic indicator, especially in elderly patients63,64. However, LOXL2 expression did not significantly correlate with gender, histological grade and perineural invasion, which may reflect differences in the function of LOXL2 in different clinicopathological features. The function of LOXL2 is mainly associated with tumour invasion and metastasis, and these processes may not be significantly different in male and female patients. For histological grading, LOXL2 expression levels may be more consistent across tumours with different degrees of differentiation, and its function is reflected more in the processes of tumour invasion and metastasis than in the degree of differentiation. For perineural invasion, LOXL2 may not play a major role in the neural invasion of tumours, and its function is more in extracellular matrix remodelling and distant metastasis. In addition, data bias and other factors (e.g., differences in sample sizes, assay methods, and adjudication criteria) may also affect the significance of these results. Future studies should use larger sample sizes and standardised protocols to reduce data bias and further validate these results.
LOXL2 is involved in multiple carcinogenic pathways, and previous studies using data mining and bioinformatics analysis have shown differential expression and prognostic value of LOXL2 in pan cancer65. Previous studies have shown that members of the LOX family have a promoting effect on endothelial cell activity and angiogenesis in tumors66,67. And LOXL2 induces epithelial mesenchymal transition by affecting the expression of SNAIL, regulating the FAK/Src pathway to promote tumor metastasis27,48. HIF-1 α inducing epithelial mesenchymal transition and tumor cell migration and invasion by regulating LOXL232. On the other hand, LOXL2 upregulates HIF-1α/VEGF signaling pathway through the Snail/FBP1 axis to promote the progression of hepatocellular carcinoma68. LOXL2 also has the effect of promoting tumor cell migration and invasion by activating EGFR through IFIT1 and IFIT350. LOXL2 not only promotes the invasion and metastasis of ESCC through phosphorylation of ezrin, but also increases tumor proliferation, invasion, and metastasis by inhibiting the expression of E-cadherin43,69,70. Other studies have also shown that Wnt9a and Wnt7 can induce LOXl2 expression, thereby affecting tumor invasiveness11. Moreover, LOXL2 mediated ECM remodeling can lead to the activation of ROCK, thereby enhancing tumor invasion71. In addition, LOXL2 promotes the progression of phosphorylation of AKT by regulating the PI3K/AKT signaling pathway47,72. In addition, the interaction between LOXL2 and GATA6 can promote angiogenesis, thereby enhancing the expression of VEGFA and ultimately leading to tumor progression36. However, direct experimental evidence supporting the molecular mechanisms of LOXL2 in carcinogenesis remains limited. Future studies are warranted to validate these mechanisms through robust experimental approaches, which would further elucidate the role of LOXL2 and its potential as a therapeutic target.
The results of the present study showed that LOXL2 expression was positively correlated with drug sensitivity to various enzyme inhibitors (e.g., P-529, staurosporine, spebrutinib). Although studies on the relationship between LOXL2 and chemotherapeutic agents (e.g. P-529, Staurosporine, etc.) are relatively limited, the potential role of LOXL2 in the regulation of the tumour microenvironment suggests that it may indirectly affect the permeability and effect of chemotherapeutic agents by modulating extracellular matrix components or influencing the activity of tumour-associated fibroblasts73. In addition, LOXL2 may influence the metabolism and efficacy of chemotherapeutic drugs by modulating the activity or expression levels of certain enzymes related to chemotherapeutic drug metabolism (e.g., cytochrome P450, glutathione S-transferase, etc.) and thereby affecting the metabolism and efficacy of chemotherapeutic drugs. However, these mechanisms have not been directly experimentally validated, and further studies are needed to clarify the specific pathways of LOXL2’s role in chemotherapy sensitivity. Future studies could focus on the interactions between LOXL2, and enzymes related to chemotherapeutic drug metabolism, as well as the potential clinical applications of LOXL2 inhibitors in combination with chemotherapeutic drugs.
Overall, our study suggests that high expression of LOXL2 is significantly associated with poor prognosis and advanced clinical features in various human cancers, and it may serve as a new prognostic biomarker. At the same time, the characteristics of LOXL2 in expression patterns, survival prognosis, signaling pathways, drug sensitivity, and other aspects were revealed. However, this study also has some limitations: 1) Some research directly provided HRs and 95% confidence intervals, while others only provided survival curves, which may not be accurate enough, resulting in some reporting bias. 2) The inconsistency in the thresholds for LOXL2 expression levels and the varying methods used to assess LOXL2 expression across the included studies may have introduced a potential bias in the final results. It is recommended that future studies adopt a standardised study design protocol to ensure the reliability of the findings. Such standardisation will enhance the comparability of results from different studies and improve the overall reliability of conclusions. 3) This study primarily analyzed data from a limited number of geographic regions and predominantly Asian populations, which may restrict the generalizability of the findings. Therefore, it is recommended that a multicentre, large-sample prospective study be conducted to validate the prognostic value of LOXL2 in different cancer types and to assess its generalizability to different populations. 4) The literature included in this study is a retrospective study, and the quality level of the literature may be relatively low. To ensure quality and high representativeness, appropriate statistical software and methods might be improved to collect as much relevant data as possible. 5) The presence of publication bias in this study may have constrained the comprehensiveness of the findings. Nevertheless, the trim-and-fill method indicated that publication bias had a minimal impact on the results, which remained consistent. The existence of publication bias may be attributed to the fact that negative results are not readily published. Indeed, negative results are as valuable as positive results, and it is recommended that a robust research project registration system and seamless access to published and unpublished data be established and enhanced in the future. It is proposed that the establishment and improvement of a robust research project registration system and seamless access to information on published and unpublished literature will significantly reduce the occurrence of publication bias. 6) The criteria for defining ‘high’ versus ‘low’ LOXL2 expression, particularly the cut-off values, were not uniformly reported across all included studies. While the methodological quality assessment (NOS) confirmed that all studies employed a clear and consistent method for grouping patients within their own cohorts, the variation in unreported cut-offs may contribute to heterogeneity and complicates direct comparisons. Future studies would benefit from pre-specifying and transparently reporting the exact cut-off values used for IHC scoring to enhance reproducibility and comparability.
Conclusion
The high expression of LOXL2 is closely associated with poor patient prognosis, tumour progression and chemotherapy resistance, suggesting that LOXL2 not only has an important application in tumour diagnosis and prognostic assessment, but may also be a potential target for reversing drug resistance and developing targeted therapies. However, current studies on LOXL2 have certain limitations. Future studies should further explore the specific molecular mechanisms of LOXL2 in tumour progression and drug resistance to provide a more solid theoretical basis for its clinical application. It should also develop LOXL2-based prognostic models and targeted therapeutic strategies to evaluate its safety and efficacy in clinical practice. By addressing the limitations of existing studies and conducting standardised studies, LOXL2 is expected to become a reliable prognostic marker and therapeutic target, providing a new strategy for precision treatment of tumour patients.
Data availability
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
References
Vineis, P. & Wild, C. P. Global cancer patterns: causes and prevention. Lancet 383 (9916), 549–557. 10.1016/s0140-6736(13)62224-2 (2014).
Ugai, T. et al. Is early-onset cancer an emerging global epidemic? Current evidence and future implications. Nat. Rev. Clin. Oncol. 19 (10), 656–673. https://doi.org/10.1038/s41571-022-00672-8 (2022).
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71 (3), 209–249. https://doi.org/10.3322/caac.21660 (2021).
Miller, K. D. et al. Cancer treatment and survivorship statistics, 2022. CA Cancer J. Clin. 72 (5), 409–436. https://doi.org/10.3322/caac.21731 (2022).
Chitty, J. L., Setargew, Y. F. I. & Cox, T. R. Targeting the Lysyl oxidases in tumour desmoplasia. Biochem. Soc. Trans. 47 (6), 1661–1678. https://doi.org/10.1042/bst20190098 (2019).
Molnar, J. et al. Structural and functional diversity of Lysyl oxidase and the LOX-like proteins. Biochim. Biophys. Acta. 1647 (1–2), 220–224. https://doi.org/10.1016/s1570-9639(03)00053-0 (2003).
Csiszar, K. Lysyl oxidases: a novel multifunctional amine oxidase family. Prog Nucleic Acid Res. Mol. Biol. 70, 1–32. https://doi.org/10.1016/s0079-6603(01)70012-8 (2001).
Setargew, Y. F. I., Wyllie, K., Grant, R. D., Chitty, J. L. & Cox, T. R. Targeting Lysyl oxidase family meditated matrix Cross-Linking as an Anti-Stromal therapy in solid tumours. Cancers (Basel). 13 (3). https://doi.org/10.3390/cancers13030491 (2021).
Vallet, S. D., Berthollier, C., Salza, R., Muller, L. & Ricard-Blum, S. The interactome of Cancer-Related Lysyl oxidase and Lysyl oxidase-Like proteins. Cancers (Basel). 13 (1). https://doi.org/10.3390/cancers13010071 (2020).
Lin, Z. Y., Chuang, Y. H. & Chuang, W. L. Cancer-associated fibroblasts up-regulate CCL2, CCL26, IL6 and LOXL2 genes related to promotion of cancer progression in hepatocellular carcinoma cells. Biomed. Pharmacother. 66 (7), 525–529. https://doi.org/10.1016/j.biopha.2012.02.001 (2012).
Matsuoka, K. et al. Wnt signaling and Loxl2 promote aggressive osteosarcoma. Cell. Res. 30 (10), 885–901. https://doi.org/10.1038/s41422-020-0370-1 (2020).
Ahn, S. G. et al. LOXL2 expression is associated with invasiveness and negatively influences survival in breast cancer patients. Breast Cancer Res. Treat. 141 (1), 89–99. https://doi.org/10.1007/s10549-013-2662-3 (2013).
Radić, J. et al. Multiple roles of LOXL2 in the progression of hepatocellular carcinoma and its potential for therapeutic targeting. Int. J. Mol. Sci. 24 (14). https://doi.org/10.3390/ijms241411745 (2023).
Wang, L., Cao, S., Zhai, R., Zhao, Y. & Song, G. Systematic analysis of expression and prognostic values of Lysyl oxidase family in gastric cancer. Front. Genet. 12, 760534. https://doi.org/10.3389/fgene.2021.760534 (2021).
Torres, S. et al. LOXL2 is highly expressed in cancer-Associated fibroblasts and associates to poor colon cancer survival. Clin. Cancer Res. 21 (21), 4892–4902. https://doi.org/10.1158/1078-0432.Ccr-14-3096 (2015).
Salvador, F. et al. Lysyl Oxidase-like protein LOXL2 promotes lung metastasis of breast cancer. Cancer Res. 77 (21), 5846–5859. https://doi.org/10.1158/0008-5472.Can-16-3152 (2017).
Zou, H. Y. et al. A truncated splice variant of human Lysyl oxidase-like 2 promotes migration and invasion in esophageal squamous cell carcinoma. Int. J. Biochem. Cell. Biol. 75, 85–98. https://doi.org/10.1016/j.biocel.2016.04.003 (2016).
Weidenfeld, K. et al. Dormant tumor cells expressing LOXL2 acquire a stem-like phenotype mediating their transition to proliferative growth. Oncotarget 7 (44), 71362–71377. https://doi.org/10.18632/oncotarget.12109 (2016).
Ferreira, S., Saraiva, N., Rijo, P. & Fernandes, A. S. LOXL2 inhibitors and breast cancer progression. Antioxid. (Basel). 10 (2). https://doi.org/10.3390/antiox10020312 (2021).
Page, M. et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. Bmj 372, 160. https://doi.org/10.1136/bmj.n160 (2021).
Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 25 (9), 603–605. https://doi.org/10.1007/s10654-010-9491-z (2010).
Reinhold, W. C. et al. CellMiner: a web-based suite of genomic and Pharmacologic tools to explore transcript and drug patterns in the NCI-60 cell line set. Cancer Res. 72 (14), 3499–3511. https://doi.org/10.1158/0008-5472.Can-12-1370 (2012).
Wang, F. et al. ZEB1 promotes colorectal cancer cell invasion and disease progression by enhanced LOXL2 transcription. Int. J. Clin. Exp. Pathol. 14 (1), 9–23 (2021).
Park, P. G. et al. Role of LOXL2 in the epithelial-mesenchymal transition and colorectal cancer metastasis. Oncotarget 8 (46), 80325–80335. https://doi.org/10.18632/oncotarget.18170 (2017).
Liang, Y. et al. Prognostic significance of abnormal matrix collagen remodeling in colorectal cancer based on histologic and bioinformatics analysis. Oncol. Rep. 44 (4), 1671–1685. https://doi.org/10.3892/or.2020.7729 (2020).
Cui, X. et al. Lysyl oxidase-like 2 is highly expressed in colorectal cancer cells and promotes the development of colorectal cancer. Oncol. Rep. 40 (2), 932–942. https://doi.org/10.3892/or.2018.6452 (2018).
Du, X. G. & Zhu, M. J. Clinical relevance of Lysyl oxidase-like 2 and functional mechanisms in glioma. Onco Targets Ther. 11, 2699–2708. https://doi.org/10.2147/ott.S164056 (2018).
Zhan, P., Lv, X. J., Ji, Y. N., Xie, H. & Yu, L. K. Increased Lysyl oxidase-like 2 associates with a poor prognosis in non-small cell lung cancer. Clin. Respir J. 12 (2), 712–720. https://doi.org/10.1111/crj.12584 (2018).
Zhan, P. et al. Down-regulation of Lysyl oxidase-like 2 (LOXL2) is associated with disease progression in lung adenocarcinomas. Med. Oncol. 29 (2), 648–655. https://doi.org/10.1007/s12032-011-9959-z (2012).
Shao, B. et al. LOXL2 promotes vasculogenic mimicry and tumour aggressiveness in hepatocellular carcinoma. J. Cell. Mol. Med. 23 (2), 1363–1374. https://doi.org/10.1111/jcmm.14039 (2019).
Choi, J. et al. Increased expression of the Matrix-Modifying enzyme Lysyl Oxidase-Like 2 in aggressive hepatocellular carcinoma with poor prognosis. Gut Liver. 13 (1), 83–92. https://doi.org/10.5009/gnl17569 (2019).
Wang, M. et al. HIF-1α promoted vasculogenic mimicry formation in hepatocellular carcinoma through LOXL2 up-regulation in hypoxic tumor microenvironment. J. Exp. Clin. Cancer Res. 36 (1), 60. https://doi.org/10.1186/s13046-017-0533-1 (2017).
Ninomiya, G. et al. Significance of Lysyl oxidase–like 2 gene expression on the epithelial–mesenchymal status of hepatocellular carcinoma. Oncol. Rep. 39 (6), 2664–2672. https://doi.org/10.3892/or.2018.6349 (2018).
Xu, J. et al. 67 laminin receptor promotes the malignant potential of tumour cells up-regulating Lysyl oxidase-like 2 expression in cholangiocarcinoma. Dig. Liver Dis. 46 (8), 750–757. https://doi.org/10.1016/j.dld.2014.03.017 (2014).
Bergeat, D. et al. Impact of stroma LOXL2 overexpression on the prognosis of intrahepatic cholangiocarcinoma. J. Surg. Res. 203 (2), 441–450. https://doi.org/10.1016/j.jss.2016.03.044 (2016).
Peng, T. et al. The interaction of LOXL2 with GATA6 induces VEGFA expression and angiogenesis in cholangiocarcinoma. Int. J. Oncol. 55 (3), 657–670. https://doi.org/10.3892/ijo.2019.4837 (2019).
Tanaka, N. et al. Clinical implications of Lysyl Oxidase-Like protein 2 expression in pancreatic cancer. Sci. Rep. 8 (1), 9846. https://doi.org/10.1038/s41598-018-28253-9 (2018).
Park, J. S. et al. Emerging role of LOXL2 in the promotion of pancreas cancer metastasis. Oncotarget 7 (27), 42539–42552. https://doi.org/10.18632/oncotarget.9918 (2016).
Kim, I. K. et al. Specific protein 1(SP1) regulates the epithelial-mesenchymal transition via Lysyl oxidase-like 2(LOXL2) in pancreatic ductal adenocarcinoma. Sci. Rep. 9 (1), 5933. https://doi.org/10.1038/s41598-019-42501-6 (2019).
Li, R. et al. Reciprocal regulation of LOXL2 and HIF1α drives the Warburg effect to support pancreatic cancer aggressiveness. Cell. Death Dis. 12 (12), 1106. https://doi.org/10.1038/s41419-021-04391-3 (2021).
Elsayed, W. H. et al. NR2F6, LOXL2 and DMBT1 expression in cervical cancer tissues, prognostic and clinicopathological implications. talian J Gynæcology & Obstetrics, 33(3), https://doi.org/10.36129/jog.33.03.05 (2021).
Zhu, G. et al. LOXL2-enriched small extracellular vesicles mediate hypoxia-induced premetastatic niche and indicates poor outcome of head and neck cancer. Theranostics 11 (19), 9198–9216. https://doi.org/10.7150/thno.62455 (2021).
Zhan, X. H. et al. LOXL2 upregulates phosphorylation of Ezrin to promote cytoskeletal reorganization and tumor cell invasion. Cancer Res. 79 (19), 4951–4964. https://doi.org/10.1158/0008-5472.Can-19-0860 (2019).
Liu, Y. et al. LOXL2 promotes tumor proliferation and metastasis by FAK/Src signaling in esophageal squamous cell carcinoma. Electron. J. Biotechnol. 63, 18–28. https://doi.org/10.1016/j.ejbt.2023.01.002 (2023). https://doi.org/https://doi.
Li, T. Y. et al. Reduced nuclear and ectopic cytoplasmic expression of Lysyl oxidase-like 2 is associated with lymph node metastasis and poor prognosis in esophageal squamous cell carcinoma. Hum. Pathol. 43 (7), 1068–1076. https://doi.org/10.1016/j.humpath.2011.07.027 (2012).
Jiao, J. W. et al. LOXL2-dependent deacetylation of aldolase A induces metabolic reprogramming and tumor progression. Redox Biol. 57, 102496. https://doi.org/10.1016/j.redox.2022.102496 (2022).
Fan, Z. et al. Phosphorylation of AKT by Lysyl oxidase-like 2 activates the PI3K/AKT signaling pathway to promote proliferation, invasion and metastasis in esophageal squamous carcinoma. Clin. Transl Oncol. 25 (8), 2487–2498. https://doi.org/10.1007/s12094-023-03133-5 (2023).
Peng, L. et al. Secreted LOXL2 is a novel therapeutic target that promotes gastric cancer metastasis via the Src/FAK pathway. Carcinogenesis 30 (10), 1660–1669. https://doi.org/10.1093/carcin/bgp178 (2009).
Kasashima, H. et al. Lysyl oxidase-like 2 (LOXL2) from stromal fibroblasts stimulates the progression of gastric cancer. Cancer Lett. 354 (2), 438–446. https://doi.org/10.1016/j.canlet.2014.08.014 (2014).
Lu, Y. J. et al. Lysyl oxidase-like 2 promotes stemness and enhances antitumor effects of gefitinib in head and neck cancer via IFIT1 and IFIT3. Cancer Sci. 114 (10), 3957–3971. https://doi.org/10.1111/cas.15912 (2023).
Wen, B., Xu, L. Y. & Li, E. M. LOXL2 in cancer: regulation, downstream effectors and novel roles. Biochim. Biophys. Acta Rev. Cancer. 1874 (2), 188435. https://doi.org/10.1016/j.bbcan.2020.188435 (2020).
Huang, J. et al. Extracellular matrix and its therapeutic potential for cancer treatment. Signal. Transduct. Target. Ther. 6 (1), 153. https://doi.org/10.1038/s41392-021-00544-0 (2021).
Liu, X. et al. Elastic fiber homeostasis requires Lysyl oxidase-like 1 protein. Nat. Genet. 36 (2), 178–182. https://doi.org/10.1038/ng1297 (2004).
Xiao, Q. & Ge, G. Lysyl oxidase, extracellular matrix remodeling and cancer metastasis. Cancer Microenviron. 5 (3), 261–273. https://doi.org/10.1007/s12307-012-0105-z (2012).
Cano, A., Santamaría, P. G. & Moreno-Bueno, G. LOXL2 in epithelial cell plasticity and tumor progression. Future Oncol. 8 (9), 1095–1108. https://doi.org/10.2217/fon.12.105 (2012).
Martin, A. et al. Lysyl oxidase-like 2 represses Notch1 expression in the skin to promote squamous cell carcinoma progression. Embo j. 34 (8), 1090–1109. https://doi.org/10.15252/embj.201489975 (2015).
Akiri, G. et al. Lysyl oxidase-related protein-1 promotes tumor fibrosis and tumor progression in vivo. Cancer Res. 63 (7), 1657–1666 (2003).
Hase, H. et al. LOXL2 status correlates with tumor stage and regulates integrin levels to promote tumor progression in CcRCC. Mol. Cancer Res. 12 (12), 1807–1817. https://doi.org/10.1158/1541-7786.Mcr-14-0233 (2014).
Barker, H. E., Bird, D., Lang, G. & Erler, J. T. Tumor-secreted LOXL2 activates fibroblasts through FAK signaling. Mol. Cancer Res. 11 (11), 1425–1436. https://doi.org/10.1158/1541-7786.Mcr-13-0033-t (2013).
Rückert, F., Joensson, P., Saeger, H. D., Grützmann, R. & Pilarsky, C. Functional analysis of LOXL2 in pancreatic carcinoma. Int. J. Colorectal Dis. 25 (3), 303–311. https://doi.org/10.1007/s00384-009-0853-5 (2010).
Rachman-Tzemah, C. et al. Blocking surgically induced Lysyl oxidase activity reduces the risk of lung metastases. Cell. Rep. 19 (4), 774–784. https://doi.org/10.1016/j.celrep.2017.04.005 (2017).
Duval, S. & Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56 (2), 455–463. https://doi.org/10.1111/j.0006-341x.2000.00455.x (2000).
Wu, Y. et al. Lysyl oxidase-like 2 inhibitor rescues D-galactose-induced skeletal muscle fibrosis. Aging Cell. 21 (7). https://doi.org/10.1111/acel.13659 (2022). e13659.
Steppan, J. et al. Lysyl oxidase-like 2 depletion is protective in age-associated vascular stiffening. Am. J. Physiol. Heart Circ. Physiol. 317 (1), H49–h59. https://doi.org/10.1152/ajpheart.00670.2018 (2019).
Zhang, X., Huang, J., You, F., Li, W. & Zou, Z. Prognostic and clinicopathological significance of LOXL2 in cancers: A systematic review and meta-analysis. J. Cell. Physiol. 234 (11), 21369–21379. https://doi.org/10.1002/jcp.28746 (2019).
Baker, A. M. et al. Lysyl oxidase plays a critical role in endothelial cell stimulation to drive tumor angiogenesis. Cancer Res. 73 (2), 583–594. https://doi.org/10.1158/0008-5472.Can-12-2447 (2013).
Zaffryar-Eilot, S. et al. Lysyl oxidase-like-2 promotes tumour angiogenesis and is a potential therapeutic target in angiogenic tumours. Carcinogenesis 34 (10), 2370–2379. https://doi.org/10.1093/carcin/bgt241 (2013).
Fan, Z. et al. LOXL2 upregulates hypoxia–inducible factor–1α signaling through Snail–FBP1 axis in hepatocellular carcinoma cells. Oncol. Rep. 43 (5), 1641–1649. https://doi.org/10.3892/or.2020.7541 (2020).
Schietke, R. et al. The Lysyl oxidases LOX and LOXL2 are necessary and sufficient to repress E-cadherin in hypoxia: insights into cellular transformation processes mediated by HIF-1. J. Biol. Chem. 285 (9), 6658–6669. https://doi.org/10.1074/jbc.M109.042424 (2010).
Peinado, H. et al. A molecular role for Lysyl oxidase-like 2 enzyme in snail regulation and tumor progression. Embo j. 24 (19), 3446–3458. https://doi.org/10.1038/sj.emboj.7600781 (2005).
Wong, C. C. et al. Lysyl oxidase-like 2 is critical to tumor microenvironment and metastatic niche formation in hepatocellular carcinoma. Hepatology 60 (5), 1645–1658. https://doi.org/10.1002/hep.27320 (2014).
Wu, S. et al. The pathological significance of LOXL2 in pre-metastatic niche formation of HCC and its related molecular mechanism. Eur. J. Cancer. 147, 63–73. https://doi.org/10.1016/j.ejca.2021.01.011 (2021).
Levental, K. R. et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139 (5), 891–906. https://doi.org/10.1016/j.cell.2009.10.027 (2009).
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization: Shenghong Yang, Qin Zhang, Pan Yuan, Yixi Ran; Methodology: Shenghong Yang, Qin Zhang, Pan Yuan, Yixi Ran; Formal analysis and investigation: Shenghong Yang, Qin Zhang; Writing - original draft preparation: Shenghong Yang, Qin Zhang; Writing - review and editing: Pan Yuan, Yixi Ran; Supervision: Pan Yuan, Yixi Ran.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, S., Zhang, Q., Ran, Y. et al. The prediction value of LOXL-2 in the clinical pathology and prognosis of cancers: A meta-analysis and bioinformatics analysis. Sci Rep 16, 1195 (2026). https://doi.org/10.1038/s41598-025-30923-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30923-4