Abstract
Lumbar puncture (LP) to extract cerebral spinal fluid (CSF) to quantify analytes relevant to brain health can lead to post-dural puncture headache (PDPH). Incidences vary across studies. This prospective memory clinic study assessed the incidence and intensity of PDPH after protocolised extraction and post-procedural rest. Patient and procedural risk factors for PDPH were explored. In 724 patients or healthy volunteers we performed 1147 procedures with intravenous infusion of 500 ml of 0.9% saline, LP with 22-gauge non-cutting pencil point needles, CSF collection (13.5–15 ml) without negative pressure, and observed bed and chair rest for 45 min. The overall incidence of PDPH was 9.6% at 24 h and 4.7% at seven days. All PDPHs were mild to moderate and self-limited (no blood patches or hospitalisations required). Patients managed symptoms with low doses of acetaminophen or non-steroidal anti-inflammatory drugs, along with oral caffeine intake. At 24 h PDPH was significantly associated with a low (> 18.5–24.9) or very high (> 40) body mass index (P = 0.021), a history of chronic pain (P = 0.007), or a lower puncture site (P = 0.002). We conclude that protocolised extraction of large volumes of CSF with a medium-gauge non-traumatic needle, controlled fluid loading and short bed rest can lower the incidence of PDPH, favouring safe CSF research.
Similar content being viewed by others
Introduction
Lumbar puncture (LP) is widely used for administering regional anaesthetics and treating diseases. It also serves for extracting cerebrospinal fluid (CSF) to diagnose and study neurodegenerative conditions. When performed correctly by a specialist, LP is safe and rarely causes complications other than post-dural puncture headache (PDPH)1,2,3.
The current International Classification of Headache Disorders defines PDPH as a headache resulting from decreased CSF pressure, typically manifesting within 5 days of LP4. PDPH worsens when patients stand and improves when they lie down. Pain is usually in the frontal or occipital regions and associated symptoms may include neck stiffness, nausea, photophobia, tinnitus and hearing disturbances5. CSF leakage at the puncture site is thought to lead to decreased intracranial pressure5. Conservative management includes supine positioning, hydration, caffeine, and oral analgesics; although PDPH generally resolves spontaneously within two weeks, severe cases may require treatment with an autologous epidural blood patch to seal the CSF leak4. A patch is recommended when patients do not respond to conservative management or require hospitalisation3,6. Some patients may also experience non-specific headache that does not meet the clinical criteria for a diagnosis of PDPH7,8.
Patient factors that have been associated with PDPH include younger age, female biological sex, pregnancy, lower body mass index and headache history9,10,11. Factors related to technique include type of bevel (traumatic cutting bevels, for example, increase risk) and larger needle gauge9,10,11,12. For spinal anaesthesia, small-gauge needles (25-gauge or smaller) are often used to avoid PDPH, but to obtain high-volume samples, such as in memory clinic studies, larger needles (22- or 20-gauge) are preferred. Studies of PDPH risk after diagnostic LP to extract large volumes13,14,15,16,17,18,19,20,21 have not given specifics regarding whether or not protocols were followed for procedures or post-procedure rest periods.
The first objective of this study was to evaluate the incidence and severity of PDPH at 24 h and seven days in a prospectively created case registry of healthy adults scheduled to undergo LP according to a standardised memory clinic protocol with 22-gauge non-cutting, pencil point needles. We hypothesised that by systematically applying a standardised protocol for large-volume CSF extraction and rest, we could achieve a low incidence of severe PDPH. Secondarily, we aimed to explore possible risk factors (such as age, sex, body mass index, and history of chronic pain and/or migraine) that might be associated with the development of PDPH.
Methods
Study design, setting and participants
This was an observational study of data from a prospectively gathered case registry of LPs performed at Barcelonaβeta Brain Research Centre (BBRC)22,23. The registry was created for diagnostic and other clinical and counselling purposes (follow-up) as well as for research (hence, all cases followed the same protocol for procedures and rest throughout the recruitment period). Cases were patients with memory loss scheduled for diagnostic LPs or follow-up or healthy volunteers whose parents were diagnosed with Alzheimer’s disease.
Recruitment and data collection took place between November 2016 and October 2023. The BBRC registry was initially created for three projects with protocols registered at ClinicalTrials.gov: ALFA-FPM-0311 (NCT02485730), EPAD-UoE-001 (NCT02804789), and β-AARC_BBRC2021 (NCT04935372). The registry has since been made available for further research. The present analysis of the registered data was approved by the human research ethics committee of Hospital de Mar Research Institute (CEIC-PSMAR) in Barcelona (file number 2022/10251) and was carried out in accordance with the ethical principles stipulated in the Helsinki Declaration (2013 revision from Fortaleza, Brazil).
Patients and healthy individuals participated in the study voluntarily, gave their informed consent and did not receive compensation. Exclusion criteria included coagulation disorders, anticoagulant or antiaggregatory therapies, and lumbar spine conditions evaluated by BBRC physicians that could impair the performance of an intradural LP.
Standardised protocols for LP and follow-up
During the procedure, continuous blood pressure, oxygen saturation and 3-lead electrocardiogram monitoring was in place. A peripheral venous catheter was inserted to infuse 500 ml of 0.9% saline. If the volunteer exhibited anxiety, 0.25 mg of oral alprazolam was offered.
All LPs were performed after a 6-h fasting period with participants positioned either in a left lateral decubitus or seated position. The lateral decubitus position was primarily chosen as the initial position, while the seated position was chosen if a difficult LP was expected based on inspection of the back anatomy or a history of previous difficult LP. If LP in lateral decubitus proved challenging, the volunteer was repositioned to the seated position for subsequent attempts.
The puncture sites utilised were L2-L3, L3-L4, L4-L5, or L5-S1. An initial subcutaneous infiltration of 2% lidocaine was administered to the skin area corresponding to the interspinous space chosen. After 2 min, another intramuscular injection with 2% lidocaine was given at the same puncture site. One minute later, the LP was performed with a 22-gauge non-cutting (Whitacre) pencil point spinal needle (Temena GmbH, Felsberg, Germany) to extract 13.5–15 ml of CSF. CSF was collected by free flow: no negative pressure was applied during the extraction. All LPs were conducted by senior anaesthesiologists, each with over 15 years of experience.
Once extraction was completed, the volunteer remained in supine position for 30 min with monitors connected. After this rest period, if the volunteer was stable and had no headache or dizziness, the head of the bed was raised 45 degrees, and he or she remained in this position 15 min longer. Monitors were then disconnected and the volunteer was helped to stand up and move to a chair for further rest; a light breakfast was provided. After 30 min, if oral tolerance was satisfactory, a final blood pressure reading was recorded, recommendations were made (moderate physical activity during the first week, abundant hydration favouring beverages containing caffeine, and no flights for 1 week). A telephone number was offered so that certain symptoms (headache, backache, or other discomfort or incidents) could be reported if they appeared or help with complications was needed. Further strict bed rest was not recommended.
At 24 h and seven days after extraction, a nurse specialised in LP follow-up contacted each volunteer to check for complications and offer help if needed.
Variables
On enrolment, a medical history, including such factors as chronic non-migraine headache of any type, migraine and chronic pain (any pain lasting longer than three months) was taken. Age, sex, and body mass index (BMI) were also on record. Procedure variables included positioning for LP and puncture site, the number of attempts required, and the extraction time.
Follow-up variables assessed at 24 h and seven days of LP were the presence and severity of PDPH and/or non-specific headache, back pain, and any analgesic treatments taken. The follow-up information was collected by specialised BBRC staff nurses. Mild complications like nausea, vomiting, pain radiating to the trapezius, dizziness, flashing/flickering lights (photopsia), tinnitus and tearing were also recorded.
PDPH was defined as a headache that worsens within 15 min of standing and improves or resolves within 30 min of lying down. It was classified based on its severity as follows: mild (when there was no alteration of the volunteer’s normal activities), moderate (when there was deterioration in the volunteer’s activities), and severe (when hospitalisation was required). Non-specific headache was defined as any headache without the abovementioned characteristics that define PDPH.
Statistical analysis
SPSS 29.0 for Windows (IBM, Armonk, NY, US) was used for statistical analysis. Continuous variables were described with the mean and standard deviation (SD) and categorical variables with the number and percentage of volunteers in each response category. Statistical techniques were applied to verify the assumed data distributions before the appropriate parametric tests were used to compare means (t tests) or categorical variables (Fisher exact tests). Values of P < 0.05 were considered significant in all tests. We explored candidate risk factors with multiple logistic regression analysis and calculated odds ratios (ORs) and 95% CIs.
Results
From November 2016 to October 2023, 1162 LPs were performed in 724 patients or healthy volunteers: 378 underwent a single LP, 299 underwent two LPs, 58 underwent three LPs, and three underwent four LPs (Fig. 1). Fifteen LPs were excluded because the procedure failed, or the volunteers were lost to follow-up at 24 h or seven days. Ultimately, data for 1147 LPs were analysed for the first objective (PDPH incidence and severity).
Recruitment and analysis flowchart. LP, lumbar puncture; PDPH, post-dural puncture headache.
For the exploration of risk PDPH risk factors, the number of procedures for which variables were recorded varied slightly, attributable to changes in data collection logistics while the registry was being created or to the nature of the variable. For example, data collection began on paper and evolved to a digital platform. In the tables below, we report the numbers on which the percentages and ORs were based. All analyses were based on more than 1000 cases.
Demographic data
The characteristics of the study population are summarised in Table 1. The mean (SD) age was 63.9 (5.6) years and 58.1% of the participants were women. The mean BMI was 27.5 (4.4) kg/m2. A history of chronic non-migraine headache was reported by 16.8% of the participants, migraine was reported by 15.7%, and 25.7% had a history of chronic pain.
LP procedure data
LP characteristics are summarised in Table 2. In more than 80% of the cases, the LP was performed with the volunteer in lateral decubitus position; 18.3% of the participants underwent LP in seated position. The procedure was performed in a single attempt in over 80% of the cases.
Post-LP complications
Table 3 summarises the post-procedural complications at 24 h and seven days.
At the 24-h follow-up call the incidence of PDPH was 9.6% and 14.7% reported non-specific headaches. Most participants with PDPH reported mild pain (95.5%). No participants reported severe PDPH. Analgesics were used by 87.3% of participants to treat PDPH, and 36.4% increased their caffeine consumption. Back pain was reported by 42% (mild for the majority of those affected). No severe complications that required a blood patch or hospitalisation were reported.
At seven days’ follow-up, the incidence of PDPH was 4.7% and 8.9% reported non-specific headache. Most of the participants with PDPH (87%) had mild pain; none reported severe pain. Analgesics were used by 92.6% of participants with PDPH and 22.2% increased their caffeine consumption. Back pain was reported by over 11%; pain was mild for 92.5% and moderate for 7.5%. As at the earlier follow-up, no severe complications that required a blood patch or hospitalisation occurred.
Risk factors for PDPH at 24 h and seven days
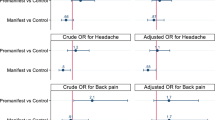
OR analysis (Table 4) showed that the incidence of PDPH at 24 h was higher in those with a history of chronic pain (P = 0.007), either a low or very high BMI (P = 0.021), and when the LP was performed at lower levels (L4-L5 and L5-S1) (P = 0.002). At seven days, no significant associations were found between the incidence of PDPH and any of the analysed factors.
Mean (SD) age did not differ between individuals with or without PDPH at 24 h (63.3 [5.9] vs. 63.0 [6.0] years; P = 0.226) or seven days. Likewise, extraction times did not differ (9.8 [2.9] vs. 9.9 [2.9] min; P = 0.704).
Discussion
We report PDPH incidences of 9.6% at 24 h and 4.7% at seven days achieved under the standardised protocol we applied in this study. These figures are markedly lower than those reported in the literature on high-volume CSF extractions in general and in memory-clinic settings; they are also lower than figures reported in the three studies most similar to ours (Table 5)16,17,19. Most of our participants reported that pain was both mild and self-limited (in over 95% of PDPH cases at 24 h and 87% at seven days). Pain that was not self-limited was managed with simple analgesics (acetaminophen or non-steroidal anti-inflammatory drugs) or caffeine intake in all cases, so no extra measures or hospitalisations were needed.
Reported incidences of PDPH at 24 h after large-volume CSF extraction have ranged widely, from as low as 2.6%12 and 9%13 to as high as 17.5%14 and 20.3%15. This variability can be explained by methodological differences in such variables as diagnostic criteria, data collection, staff experience, needle gauge, patient positioning, and clinical setting to name but a few possibilities besides patient characteristics. Therefore, for a closer comparison with our study, we narrowed our literature selection to studies that classified outcomes by severity, involved large-volume CSF extractions in individuals with similar demographic characteristics, and with LPs performed by experienced physicians. We identified three such studies (Table 5)16,17,19, thus excluding any performed by residents or non-specialised staff19,24. Whereas these three studies did see cases of severe PDHP, as defined by need for hospitalisation or blood patches, none of our participants required extraordinary treatment after use of a standard protocol for LP, hydration and rest. The higher incidence of severity reported in the earlier studies might possibly be attributable to the lack of such a protocol.
Risk for PDPH at 24 h in our study was associated with low BMI (≤ 25) or very high BMI (> 40), chronic pain, and LP performed at a lower interspinous level (L4-L5 and L5-S1). Although some groups have reported higher risk for high-BMI patients18,25,26, others have found no significant association27,28. History of chronic pain has also previously been related to higher risk for PDPH at 24 h10,15. However, ours is the only study to report a statistically significant association between puncture site level and PDPH at 24 h. It is possible that the lower interspinous sites are prone to greater CSF leakage due to gravity, resulting in lower intracranial pressure and favouring the development of PDPH. However, this warrants further research. We did not observe significant associations for other independent variables that have been described as predictors of PDPH, such as the number of puncture attempts needed26,29,30 or use of the seated position for LP26,31. This difference may be attributable to either physician experience or an appropriate rest protocol. Likewise, we found no relationship between the incidence of PDPH and biological sex, even though previous studies have found that women are at a significantly higher risk of developing PDPH18,21,29,30,31. In addition, patients with PDPH were not older than those who did not develop this complication, despite age being identified as a risk factor in other studies16,17,18,25. However, the narrow, older age range in our cohort, in which many volunteers had parents with Alzheimer’s disease, would explain this difference. Similarly, extraction times were similar for patients with and without PDPH.
A Cochrane review on interventions to prevent PDPH32 concluded that neither prolonged bed rest nor additional fluid supplementation provided clear benefits in preventing this complication. In fact, it was concluded that prolonged bed rest, even for as long as 24 h, could possibly slightly increase risk for PDPH compared to early mobilisation. Additionally, increased fluid intake, whether oral or intravenous, did not significantly reduce PDPH incidence according to the Cochrane reviewers. The approach we used to protect against PDPH combined protocolised postural management, controlled hydration, and personalised rest. Our volunteers were then encouraged to resume normal daily activities, while avoiding excessive physical exertion. Furthermore, we administered 500 ml of intravenous saline to optimise hydration. We think that this approach, although differing from Cochrane’s general recommendations, contributed to the lower incidence of PDPH observed in our study.
A limitation of this study is the lack of a control group because a historical control cohort was not available. Although a randomised control group would have allowed clearer evaluation of the protocol, ethical concerns made this approach unfeasible. As a result, and because we found that no guidelines existed for performing LP in memory clinic settings, we opted for a protocol providing protective strategies based on our clinical consensus and reading of the Cochrane review32. The protocol included measures that would ensure the safety of participants while avoiding the exclusion of any potentially beneficial intervention.
We conclude that the standardised protocol used in this study for extracting large volumes of CSF can potentially reduce the incidence of PDPH, especially severe PDPH, a goal which is crucial for ensuring the safety and feasibility of research in this setting. Systematic use of non-cutting pencil point needles, controlled fluid overload and a standardised, supervised early rest protocol should prove to be a promising strategy for preventing severe complications of LP. We believe it merits testing in memory clinics and similar settings, possibly with comparison to retrospective cohorts.
Data availability
The patient registry and other datasets are not publicly posted but can be obtained from the corresponding author on reasonable request.
Abbreviations
- LP:
-
Lumbar puncture
- CSF:
-
Cerebrospinal fluid
- PDPH:
-
Post-dural puncture headache
- BMI:
-
Body mass index
- BBRC:
-
Barcelonaβeta brain research centre
- OR:
-
Odds ratio
- SD:
-
Standard deviation
- NS:
-
Not specified
References
Buddeberg, B. S., Bandschapp, O. & Girard, T. Post-dural puncture headache. Minerva Anestesiol. 85, 543–553 (2019).
Doherty, C. M. & Forbes, R. B. Diagnostic lumbar puncture. Ulster Med J. 83, 93–102 (2014).
Geisbush, T. R., Matys, T., Massoud, T. F. & Hacein-Bey, L. Dural puncture complications. Neuroimaging Clin N Am. 35, 53–76 (2025).
Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition. Cephalalgia. 2018;38:1–211.
Vallejo, M. C. & Zakowski, M. I. Post-dural puncture headache diagnosis and management. Best Pract. Res. Clin. Anaesthesiol. 36, 179–189 (2022).
Ona, X.B., Osorio, D., Cosp, X.B. Drug therapy for treating post-dural puncture headache. Cochrane Database Syst Rev. 2015;2015(7).
Santanen, U., Rautoma, P., Luurila, H., Erkola, O. & Pere, P. Comparison of 27-gauge (0.41-mm) Whitacre and Quincke spinal needles with respect to post-dural puncture headache and non-dural puncture headache. Acta Anaesthesiol. Scand. 48, 474–479 (2004).
Maranhao, B., Liu, M., Palanisamy, A., Monks, D. T. & Singh, P. M. The association between post-dural puncture headache and needle type during spinal anaesthesia: a systematic review and network meta-analysis. Anaesthesia 76, 1098–1110 (2021).
Pagani-Estévez, G. L. et al. Procedural predictors of epidural blood patch efficacy in spontaneous intracranial hypotension. Reg. Anesth. Pain Med. 44, 212–220 (2019).
Kwak, K. H. Postdural puncture headache. Korean J. Anesthesiol. 70, 136–143 (2017).
Sachs, A. & Smiley, R. Post-dural puncture headache: the worst common complication in obstetric anesthesia. Semin. Perinatol. 38, 386–394 (2014).
Sempere, A. P., Berenguer-Ruiz, L., Lezcano-Rodas, M. & Mira-Berenguer, F. W. M. Lumbar puncture: its indications, contraindications, complications and technique. Rev Neurol. 45, 433–436 (2007).
Van Oosterhout, W. P. J. et al. Postdural puncture headache in migraineurs and nonheadache subjects: a prospective study. Neurology 80, 941–948 (2013).
Lybecker, H., Moller, J. T., May, O. & Nielsen, H. K. Incidence and prediction of postdural puncture headache. A prospective study of 1021 spinal anesthesias. Anesth. Analg. 70, 389–394 (1990).
Monserrate, A. E., Ryman, D. C., Ma, S. & Xiong, C. Factors associated with the onset and persistence of post–lumbar puncture headache. Physiol. Behav. 176, 139–148 (2016).
Duits, F. H. et al. Performance and complications of lumbar puncture in memory clinics: results of the multicenter lumbar puncture feasibility study. Alzheimers Dement. 12, 154–163 (2016).
Alcolea, D. et al. Feasibility of lumbar puncture in the study of cerebrospinal fluid biomarkers for Alzheimer’s disease: a multicenter study in Spain. J. Alzheimers Dis. 39, 719–726 (2014).
Almeida, S. M. et al. Incidence of post-dural puncture headache in research volunteers. Neurology 51, 1503–1510 (2012).
Moulder, K. L. et al. Factors influencing successful lumbar puncture in Alzheimer research. Alzheimer Dis Assoc. Disord. 31, 287–294 (2017).
Barreras, P. et al. A dedicated lumbar puncture clinic: performance and short-term patient outcomes. J. Neurol. 264, 2075–2080 (2017).
Zetterberg, H. et al. Low incidence of post-lumbar puncture headache in 1,089 consecutive memory clinic patients. Eur. Neurol. 63, 326–330 (2010).
Molinuevo, J. L. et al. The ALFA project: a research platform to identify early pathophysiological features of Alzheimer’s disease. Alzheimers Dement. Transl. Res. Clin. Interv. 2, 82–92 (2016).
Solomon, A., Kivipelto, M., Molinuevo, J. L., Tom, B. & Ritchie, C. W. European prevention of Alzheimer’s dementia longitudinal cohort study (EPAD LCS): study protocol. BMJ Open 8, e021017 (2018).
Engedal, T. S., Ørding, H. & Vilholm, O. J. Changing the needle for lumbar punctures: results from a prospective study. Clin. Neurol. Neurosurg. 130, 74–79 (2015).
Al-Hashel, J., Rady, A., Massoud, F. & Ismail, I. I. Post-dural puncture headache: a prospective study on incidence, risk factors, and clinical characterization of 285 consecutive procedures. BMC Neurol. 22, 1–10 (2022).
Uppal, V. et al. Evidence-based clinical practice guidelines on postdural puncture headache: a consensus report from a multisociety international working group. Reg. Anesth. Pain Med. 49, 471–501 (2024).
Souki, F. G. & Zbeidy, R. Efficacy and ease of use of a newly designed pencil-point epidural needle compared to conventional Tuohy epidural needle: a randomized single-blind pilot study. Cureus. 14, e30341 (2022).
Thakur, S. et al. Incidence and risk factors of “postdural puncture headache” in women undergoing cesarean delivery under spinal anesthesia with 26G Quincke spinal needle, experience of medical college in rural settings in India 2019: a prospective cohort study design. J. Pharm. Bioallied. Sci. 14(Suppl 1), S209–S213 (2022).
Khlebtovsky, A. et al. Risk factors for post lumbar puncture headache. Clin. Neurol. Neurosurg. 131, 78–81 (2015).
Evans, R. W. Complications of lumbar puncture. Neurol. Clin. 16, 83–105 (1998).
Stichaller, L. et al. Antiplatelet therapy is not associated with increased risk of complications after lumbar puncture. J. Neurol. 272, 1–7 (2025).
Arevalo-Rodriguez, I., Ciapponi, A., Roqué i Figuls, M., Muñoz, L., Bonfill Cosp, X. Posture and fluids for preventing post-dural puncture headache. Cochrane Database Syst. Rev. 2016;2016(3).
Acknowledgements
We thank the participants in the ALFA, EPAD and β-AARC studies that prospectively collected data for the lumbar puncture registry on which the present study was based. We also thank the members of Barcelonaβeta Brain Research Centre who helped us with this research. We also thank collaborators of the ALFA Study: Federica Anastasi, Annabella Beteta, Anna Brugulat-Serrat, Raffaele Cacciaglia, Marta del Campo, Lidia Canals, Alba Cañas, Irene Cumplido-Mayoral, Carme Deulofeu, Ruth Dominguez, Maria Emilio, Karine Fauria, Ana Fernández-Arcos, Sherezade Fuentes, Marina García, Patricia Genius, Juan Domingo Gispert, Armand González-Escalante, Laura Hernández, Felipe Hernández-Villamizar, Gema Huesa, Jordi Huguet, Laura Iglesias, Esther Jiménez, Ferran Lugo, David López-Martos, Paula Marne, Tania Menchón, Carolina Minguillón, José Luis Molinuevo, Paula Ortiz-Romero, Wiesje Pelkmans, Albina Polo, Sandra Pradas, Blanca Rodríguez-Fernández, Iman Sadeghi, Gemma Salvadó, Mahnaz Shekari, Lluís Solsona, Anna Soteras, Laura Stankeviciute, Marc Suárez-Calvet, Gonzalo Sánchez-Benavides, Núria Tort-Colet, Marc Vilanova, and Natalia Vilor-Tejedor. Mary Ellen Kerans advised on English language expression in some versions of the manuscript.
Funding
The ALFA+study receives ongoing funding from “La Caixa” Foundation (agreement LCF/PR/SC22/68000001), the Pasqual Maragall Foundation for Alzheimer’s disease, the Alzheimer’s Association, and an anonymous international charity through the TriBEKa Imaging Platform (TriBEKa17519007). Additional support has been received from the Catalan government’s Universities and Research Secretariat, Ministry of Business and Knowledge (grant 2021 SGR 00913). The EPAD project received support from the EU/EFPIA Innovative Medicines Initiative Joint Undertaking EPAD (grant 115736) and the Pasqual Maragall Foundation (grant SG-21-818099-EPAD). The β-AARC study receives ongoing funding from the Alzheimer’s Drug Discovery Foundation (grants RDADB-201906-2018897 and 201809-2016862). Additionally, the β-AARC study has been funded by the Health Department of the Catalan Government.
Author information
Authors and Affiliations
Contributions
Antonio Montes-Pérez, Guillem Pérez-Garcia, Lluis Gallart-Gallego, Olga Comps-Vicente, Luis Molto-García and Oriol Grau-Riera wrote the main manuscript. Antonio Montes-Pérez, Guillem Pérez-García, Lluis Gallart-Gallego, Olga Comps-Vicente and Luis Molto-García prepared tables and figure. Antonio Montes-Pérez, Guillem Pérez-García, Lluís Gallart-Gallego, Olga Comps-Vicente, Luis Molto-García, Oriol Grau-Riera, Sandra Pradas and Anna Soteras reviewed the manuscript. Antonio Montes-Pérez and Guillem Pérez-García corresponding authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Pérez-García, G., Gallart-Gallego, L., Comps-Vicente, O. et al. Protocol to avoid post-dural puncture headache after large-volume cerebral spinal fluid extraction in a prospective observational memory clinic study. Sci Rep 16, 1931 (2026). https://doi.org/10.1038/s41598-025-31634-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-31634-6