Abstract
Parkinson’s disease (PD) is characterized by the loss of dopaminergic neurons in the substantia nigra (SN), accumulation of alpha-synuclein (a-Syn)-rich aggregates, and the development of both motor and non-motor symptoms. The absence of a cure for PD underscores the critical need for animal models that recapitulate both its molecular hallmarks and the spectrum of behavioral symptoms to elucidate pathogenic mechanisms and advance therapeutic development. While the virus-mediated a-Syn overexpression rat model recapitulates progressive dopaminergic neurodegeneration and motor deficits, its non-motor phenotypic profile remains poorly characterized. In this study, we investigated the behavioral consequences of targeted adeno-associated virus (AAV)-mediated overexpression of aggregate-prone mutant A53T a-Syn within the rat SN bilaterally. We confirmed dopaminergic neurodegeneration within 6 weeks, along with progressive motor impairments evident as reduced locomotion in the open field test and increased foot slips in the grid walking test as early as 3 weeks post-AAV injection. The dopaminergic origin of the motor deficit in the grid walking was confirmed at the 6-week timepoint by its attenuation with L-DOPA treatment. Crucially, animals overexpressing A53T a-Syn exhibited reduced responsiveness to palatable stimulation in the sucrose preference test in the context of advanced neurodegeneration. No changes in anxiety-like behaviors, olfactory function, or recognition memory were observed when compared with age-matched controls. These findings suggest that, in addition to motor impairments, bilateral A53T a-Syn overexpression induces depression-like behavior, providing a valuable tool for studying the pathogenesis and treatment of non-motor affective symptoms in PD.
Similar content being viewed by others
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative movement disorder characterized by bradykinesia, rigidity, and tremor. While its exact cause remains unknown, the loss of dopaminergic neurons in the substantia nigra pars compacta (SN), due to the abnormal accumulation of alpha-synuclein (a-Syn), is widely considered to be the pathological substrate of PD-related motor dysfunction1,2,3,4. Beyond its well-known motor impairments, PD is also marked by non-motor symptoms—including depression5, anxiety6, cognitive impairment7, and olfactory dysfunction8—that are increasingly recognized as critical facets of the disease9. These non-motor symptoms can precede motor deficits by years or even decades, significantly impacting patients’ quality of life and posing unique challenges for diagnosis and treatment10,11,12. Depression and anxiety may be present in up to 50% of PD patients and are associated with faster disease progression and increased disability5,13. Olfactory dysfunction is one of the earliest non-motor symptoms observed and affects upwards of 90% of PD patients8,14,15. Cognitive impairment, ranging from mild deficits to dementia, may also affect a substantial proportion of patients as the disease progresses7,16,17,18. Emerging evidence suggests that a-Syn accumulation is linked to both motor and non-motor symptoms of PD19, but it remains unclear whether the degeneration of SN dopaminergic neurons is the primary driver of these clinical features, underscoring the need for animal models that recapitulate a-Syn-related pathology and its behavioral consequences.
Animal models play an essential role in advancing our understanding of PD pathogenesis and in developing therapeutic strategies. Traditional neurotoxin-based models, such as 6-hydroxydopamine (6-OHDA) and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), provide valuable insights into the relationship between nigrostriatal dopamine depletion and the development of motor symptoms4,20,21,22,23. However, these toxin models have limitations, as they fail to capture the progressive nature of the disease and do not develop a-Syn aggregates, which are a hallmark of PD and implicated in the dysregulation of critical processes such as synaptic vesicle recycling and neurotransmitter release4,24,25. Genetic models, including transgenic mice that overexpress wild-type or mutant a-Syn, are used to replicate the proteinopathic features of PD, but robust dopaminergic cell loss in the SN is often absent26, and it can take an extended period for motor impairments to manifest27. Viral vector-mediated a-Syn overexpression models, particularly those using adeno-associated virus (AAV), offer a compelling alternative. These models recapitulate key features of PD, such as progressive dopaminergic neuron loss, a-Syn aggregation, and significant motor deficits within weeks4,28,29,30,31,32,33. Despite these advantages, the characterization of non-motor behaviors in AAV-based a-Syn overexpression models remains limited and marked by inconsistent findings across studies33,34,35.
To address this gap, we employed mutant A53T a-Syn, which exhibits accelerated aggregation kinetics and induces more pronounced nigrostriatal degeneration than the wild-type protein36. While motor and molecular changes in the AAV-mediated A53T a-Syn overexpression rat model are well established by our group37,38 and others29,30,31,32,39, its non-motor behavioral profile has not been systematically defined. For this behavioral profiling, we focused on female Sprague–Dawley rats to maintain continuity with the original description and subsequent characterization of this model, which have been conducted predominantly in females29,30,32,37, although male rats have recently been shown to also exhibit dose-dependent dopaminergic neurodegeneration and motor impairment following A53T a-Syn overexpression40. While men may have a higher incidence of PD in Western populations41, women seem to experience a more rapid progression in some studies and report distinct burdens of non-motor symptoms, such as depression42. The exclusive use of female rats in the present study reduced a major source of biological variability allowing for a more precise and internally consistent analysis of core behavioral sequelae of targeted A53T a-Syn overexpression, while establishing a clear baseline for future sex-comparative studies.
Here, we conducted a longitudinal study following AAV-mediated A53T a-Syn overexpression in the SN of female rats and demonstrated that bilateral nigrostriatal neurotoxicity drives gradual motor impairment and the late emergence of an anhedonia-like phenotype characterized by reduced responsiveness to palatable stimulation. These findings establish this model as a suitable platform for studying motor symptom progression and depression-like behavior in the context of a-Syn-mediated dopaminergic neurodegeneration.
Materials and methods
This study was reported in accordance with the ARRIVE guidelines. All methods were performed in accordance with the relevant guidelines and regulations. Materials and Methods are available in the manuscript.
Animals
The University Health Network Animal Care Committee approved all animal procedures in accordance with the guidelines and regulations set by the Canadian Council on Animal Care (AUP #3818). The number of animals per group was determined by a priori power analysis based on primary motor outcomes from pilot unilateral studies and adjusted to account for the increased technical variability associated with bilateral SN targeting (see Supplementary Note 1), in accordance with the principle of the 3Rs. Thirty-nine adult female Sprague–Dawley rats were used in the study. Rats were purchased from a commercial producer (Envigo, IN, USA) and group-housed in the animal care facility at the Toronto Western Hospital under standard conditions (ad libitum access to food and water unless stated otherwise, 21 \(^\circ \text{C}\), 12-h light/dark cycle).
Neurosurgical procedures
The stereotaxic surgical procedure was adapted from our previous work with the unilateral AAV A53T a-Syn rat model38,43. Animals were randomly assigned to receive A53T a-Syn (n = 20) or empty vector (EV) control (n = 19). The surgical procedure was performed under isoflurane/oxygen anesthesia (2.5–3%) and ketoprofen (5 mg/kg) analgesia. Adult female rats with an average weight of 270 g were bilaterally injected in the SN with either 2.0 \(\upmu\)L of empty AAV1/2 vector (EV) or 2.0 \(\upmu\)L of AAV1/2 expressing human mutant A53T a-Syn (A53T) at a concentration of 3.4 × 1012 genomic particles (gp)/mL using coordinates from bregma: − 5.2 mm anteroposterior (AP); either − 2.0 mm (right) or + 2.0 mm (left) mediolateral (ML); − 7.5 mm dorsoventral (DV) according to a rat brain atlas44. The rat’s body temperature was stabilized at 37 °C–38 °C throughout the surgery with a heating pad. Daily monitoring was performed for one week after surgery.
Behavioral tests
Behavioral tests were conducted at weeks 3 and 6 post-AAV injection, corresponding to mid-stage and late-stage neurodegeneration, respectively32. Rats underwent 5 min of daily habituation for 5 consecutive days prior to the initiation of any experimental procedures. During this period, animals were habituated to the experimenter through gentle handling, which involved removal from the home cage and allowing rats to rest on the experimenter’s hands and arms. Tests were distributed throughout the week to minimize interactions and carry-over effects (Fig. 1). For each assay, the testing apparatus was cleaned with 70% ethanol between trials to remove urine and fecal residues and limit potential odors from conspecifics.
Schematic representation of the experimental design for testing the bilateral AAV1/2 A53T a-Syn overexpression rat model of Parkinson’s disease (PD). Behavioral experiments were conducted at 3- and 6-weeks post-injection, following a standardized schedule throughout the week: Monday, sucrose preference test; Tuesday, novelty-suppressed feeding test; Wednesday, habituation-dishabituation test; Thursday, open field and novel object recognition tests; Friday, grid walking test. L-DOPA or saline (control) was administered to a subset of animals at 6 weeks post-injection, and the grid walking test was repeated to confirm the dopaminergic origin of motor impairment. Animals were sacrificed following the completion of all experiments at 6 weeks post-AAV injection. The schematic was created using BioRender (http://www.biorender.com/).
Sucrose preference test
The sucrose preference test was used to evaluate depression-like behavior according to a modified protocol45. Before testing, rats underwent 3 days of habituation during which they were water deprived for 1 h and then placed in a standard cage with two bottles, one containing water and the other containing a 20% sucrose solution for another 1 h. This procedure was repeated on the test day and sucrose preference was calculated by dividing the amount of sucrose solution consumed by the total fluid intake from both bottles during the 1 h test.
Novelty-suppressed feeding test
The modified novelty-suppressed feeding protocol was applied to evaluate anxiety-like behavior46. The test was performed in a testing cage (26 cm W × 48 cm D × 20 cm H) with an illuminated dish containing 20 crisps. Rats were subjected to 12 h of food deprivation before the test to enhance food-seeking behavior. Consistent with the standard protocol for this test47,48,49, no habituation to the testing arena was performed in order to preserve the essential “novelty” component required to elicit the approach-avoidance conflict that measures anxiety-like behavior. The latency to feeding, defined as the time needed to start eating the first crisp, was manually recorded using a stopwatch. Additionally, the number of crisps consumed within 10 min was measured as an indicator of responsiveness to palatable stimulation.
Habituation-dishabituation test
The habituation-dishabituation test was conducted following a modified protocol to evaluate anosmia and odor discrimination capacity50. Rats were individually placed in a testing cage (26 cm W × 48 cm D × 20 cm H) and exposed to the habituation scent (orange) using a cotton pad for 2-min trials separated by 1-min intertrial intervals. Following the habituation phase (3 trials), the dishabituation phase began with the introduction of a novel scent (cherry). Fresh cotton pads soaked in the cherry scent were presented in 2-min trials with 1-min intertrial intervals. The active sniffing duration, defined as the animal’s nose orientation towards the cotton pad within a < 2 cm distance, was visually scored and recorded with a stopwatch51. The cross-habituation index (CHI), defined as the difference in normalized sniffing duration between the first and the fourth trials, was used to evaluate odor discrimination abilities52.
Open field test
The open field test, used to assess locomotor abilities and anxiety-like behavior, was conducted in a rectangular arena with 60 cm W × 40 cm D dimensions. Each rat was placed in the center of the arena and tracked for 10 min using the Panlab Smart 3.0 video tracking tool (Panlab, Barcelona, Spain) for automated assessments of speed, total distance traveled, and resting time. Total time spent in the center of the arena (10 cm from each wall) and in the periphery (within 10 cm from the nearest wall) was also calculated.
Novel object recognition test
The novel object recognition test was used to assess recognition memory over three distinct phases53. During the habituation phase, animals were permitted to freely explore an open field arena (60 cm W × 40 cm D dimensions) for 10 min. During the training phase, animals were allowed to explore two identical objects placed in opposite corners of the arena for 10 min. One hour after the end of the habituation phase, rats were re-introduced to the arena containing one object from the training phase and one novel object. Rats were allowed to explore the objects for 10 min during this testing phase. Object positions were randomized, and the setup was cleaned between trials to avoid confounding effects. Exploration time, defined as the rat’s head within 2 cm from the object and facing towards the object, was recorded with the Panlab Smart 3.0 video tracking tool (Panlab, Barcelona, Spain). The recognition index—calculated as the ratio of time spent exploring the novel object to the total object exploration time—was used to compare recognition memory53.
Grid walking test
Gait and motor coordination were assessed using the grid walking test54. An elevated horizontal grid (70 cm W × 90 cm H) with evenly spaced bars was used for this test. During the testing sessions, the rat was placed at the center of the grid and allowed to freely explore the arena for 5 min. Two parameters were manually recorded: the number of foot slips, indicating instances where the rats’ paws slipped through or between the bars, and the total number of steps. The slip percentage was calculated as the ratio of foot slips to the total number of steps.
L-DOPA administration
A single intraperitoneal (i.p.) injection of L-3,4-dihydroxyphenylalanine methyl ester (L-DOPA, D1507, Sigma-Aldrich, USA) or saline control was performed on a subset of rats (n = 7 for L-DOPA, n = 4 for saline) 6 weeks post administration of A53T a-Syn. L-DOPA at a concentration of 6 mg/kg and benserazide hydrochloride (B7283, Sigma-Aldrich, Canada) at a concentration of 15 mg/kg were dissolved in saline (1 mL/kg) for i.p. injection. The grid walking test was carried out 1 h after i.p. injection to assess the effectiveness of dopaminergic treatment. Week 6 grid walking test results, taken a few days before L-DOPA treatment, were used for comparison.
Post-mortem analyses
Rats were euthanized 6 weeks post injection via transcardial perfusion under deep isoflurane anesthesia. Animals were first placed in an induction chamber and deeply anesthetized with 5% isoflurane in oxygen until the cessation of breathing and loss of pedal reflex was confirmed37. They were then immediately perfused transcardially with 100 mL heparinized saline solution, followed by immersion fixation in 4% PFA in 0.1 M PBS for 2 days and cryoprotection in 15% sucrose in 0.1 M PBS followed by 30% sucrose in 0.1 M PBS solution as previously described in detail37. Coronal sections were cut at a thickness of 40 μm on a sliding microtome (Leica Microsystems Inc., Richmond Hill, ON), and serial sections were stored in cryoprotectant (30% glycerol, 30% ethoxyethanol, 40% PBS). Free-floating sections were washed with PBS-T, incubated in a blocking solution and incubated overnight in primary antibodies for tyrosine hydroxylase (TH) (1:500, Abcam, ab76442) and human a-Syn (1:500, Invitrogen, 32–8100). Following washes with PBS-T, sections were incubated in secondary antibodies (1:500, Alexa Fluor 555, Invitrogen, A-21437; 1:500, Alexa Fluor 647, Invitrogen, A-21236) for 1 h and then counterstained with DAPI. Stained sections were mounted on slides and imaged with a Zeiss AxioObserver Widefield fluorescence microscope at 10× magnification. The number of puncta was assessed using ImageJ software (Version 1.54 g, NIH, USA)55. TH + cells per mm2 were quantified by counting TH-immunofluorescent-labeled cells within the ROI and dividing by the total analyzed area37,38. a-Syn puncta per mm2 were detected with an antibody specific for human a-Syn (Syn 211) and quantified using a thresholding method in ImageJ following a previously described protocol56. Briefly, a threshold value was applied to segment puncta with a circularity 0.3–1.0, detected points within the ROI were counted and values were divided by the total analyzed area.
Statistical analyses
Data are expressed as the mean ± standard error of the mean (SEM) and were analyzed with GraphPad Prism 8 software (GraphPad, MA, USA). Normality of the data was tested using the Shapiro–Wilk test. Two-way analysis of variance (ANOVA) was used for behavior analyses, with Group (EV and A53T) as a between-group factor and Time (week 3 and week 6) as a within-group factor. For L-DOPA treatment effects, Treatment (saline and L-DOPA) was the between-group factor, and Injection (baseline and post-injection) was the within-group factor. Bonferroni post hoc tests were employed to compare behavioral outcomes. The unpaired two-tailed t-test was used to compare TH + counts between groups. Statistical significance\(\propto \hspace{0.17em}\)considered significant when p < 0.05. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
Results
Severe dopaminergic cell loss with bilateral SN overexpression of A53T a-Syn
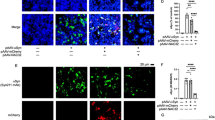
We applied the commonly used method of stereotactic injection of AAV-A53T a-Syn to achieve targeted overexpression of A53T a-Syn in the rat SN30,32,33,37,38,57. Unilateral injections are associated with the development of hemiparkinsonism-like motor impairments that parallel progressive dopaminergic neurodegeneration over the course of several weeks30,31,32,33,37,38. While PD motor symptoms are typically asymmetric, the disease process is not restricted to a unilateral SN. Therefore, we opted for a bilateral injection approach to more closely mimic PD in humans58. To achieve this, adult female rats received bilateral injections of either AAV1/2 expressing human mutant A53T a-Syn (A53T) or empty AAV1/2 vector (EV) control in the SN. To enable longitudinal assessment of behavioral progression, the same cohort of animals was tested at both the 3- and 6-week timepoints. Post-mortem assessment of the midbrain was performed at the study endpoint, 6 weeks post-infection, using immunofluorescent staining for a-Syn and tyrosine hydroxylase (TH), a marker of dopaminergic neurons (Fig. 2A). We observed 274 \(\pm\) 97 a-Syn puncta per mm2 detected with an antibody specific for human a-Syn in the SN, with no such puncta detected in the EV group (Fig. 2B). Widefield microscopy images revealed comparable numbers of TH + cells in both hemispheres within each group, so TH + cell counts from both sides were pooled for the assessment of neurodegeneration. We found a ~ 60% reduction in TH + cells in the SN of A53T a-Syn overexpressing animals compared to EV controls (Fig. 2C). These results suggest that bilateral accumulation of mutant a-Syn in the rat SN over 6 weeks recapitulates dopaminergic cell loss seen in PD and thus represents a useful model to study the progression of neurobehavioral manifestations secondary to a-Syn-induced nigrostriatal toxicity.
Dopaminergic cell loss due to bilateral A53T a-Syn overexpression in the rat substantia nigra (SN). (A) Representative images of immunostaining with anti-tyrosine hydroxylase (TH) and human a-Syn antibodies in the SN of rats bilaterally injected with mutant A53T a-Syn or empty vector (EV) control (scale bar = 200 μm applicable for all images). (B) Quantification of a-Syn puncta per mm2 in the SN (no a-Syn puncta were detected in the EV group). (C) Quantification of TH + cell counts per mm2 in the SN (t (76) = 30.18, p < 0.0001, unpaired two-tailed t-test). Sample sizes: EV, n = 38 data points; A53T, n = 40 data points). Bars represent means ± SEM ****p < 0.0001.
Motor impairment due to bilateral SN overexpression of A53T a-Syn is rescued by L-DOPA
Motor impairments are critical features that support face validity of animal models of PD. We assessed motor function at 3- and 6-weeks post-AAV injection, representing mid-stage and late-stage neurodegeneration in the A53T a-Syn group. We first employed the open field, a classical test of exploratory locomotor behavior in which rats were permitted to spontaneously explore a spacious environment for a duration of 10 min. Impairment in locomotion was evident in A53T a-Syn overexpressing animals in the open field with significant differences seen between groups in total distance (Fig. 3A, Table 1) and speed (Fig. 3B, Table 1) 6 weeks post injection. Since no differences in resting time were observed between the groups at either timepoint (Fig. 3C), the reduced distance traveled by A53T a-Syn overexpressing animals at week 6 was likely due to decreased velocity rather than increased immobility.
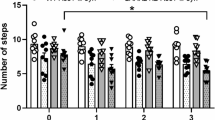
Progressive sensorimotor impairments in A53T a-Syn overexpressing rats are amenable to L-DOPA administration. (A) Total distance traveled in the open field test was significantly reduced in A53T a-Syn overexpressing animals compared to EV controls at week 6 (16,918 ± 337 cm [EV], 16,025 ± 344 cm [A53T], p = 0.0347), but not at week 3 (17,067 ± 361 cm [EV], 16,338 ± 254 cm [A53T], p = 0.2239) (Group: F (1, 74) = 7.956, p = 0.0061) timepoint. (B) Mean speed in the open field was significantly lower in the A53T group at week 6 (26.84 ± 0.55 cm/s [EV], 25.31 ± 0.53 cm/s [A53T], p = 0.0186), with no significant difference at week 3 (26.85 ± 0.58 cm/s [EV], 25.84 ± 0.49 cm/s [A53T], p = 0.3786) (Group: F (1, 74) = 7.982, p = 0.0061). (C) Resting time was comparable between groups at both timepoints in the open field test (week 3: 6.78 ± 1.07 s [EV], 6.71 ± 0.96 s [A53T], p > 0.9999; week 6: 7.37 ± 0.96 s [EV], 8.76 ± 1.30 s [A53T], p = 0.7337) (Group: F (1, 74) = 0.3748, p = 0.5423). (D) In the grid walking test, A53T a-Syn overexpressing rats exhibited a significantly higher foot slip ratio compared to EV controls at both timepoints (week 3: p = 0.0244; week 6: p = 0.0002) (Group: F (1, 74) = 22.28, p < 0.0001). (E) No difference in the number of steps was seen between groups at both timepoints (week 3: p = 0.4319; week 6: p = 0.1905) (Group: F (1, 74) = 4.316, p = 0.0412). (F) L-DOPA administration significantly reduced the slip ratio compared to week 6 baseline performance (p = 0.0425) with no difference seen between baseline and saline injection (p = 0.7889) (Treatment: F (1, 18) = 4.919, p = 0.0397). Sample sizes: n = 20 for A53T, n = 19 for EV, n = 4 for saline, n = 7 for L-DOPA. Data are shown as mean ± SEM. *p \(<\hspace{0.17em}\)0.05, ***p \(<\hspace{0.17em}\)0.001 for the post hoc Bonferroni comparison following two-way ANOVA.
Next, we employed the grid walking test to evaluate more subtle impairments in sensorimotor coordination and function. Rats were placed on an elevated grid with evenly spaced bars and permitted to move freely for assessment of foot slips over a five-minute period. Whereas deficits were initially observed at 6 weeks in the open field, A53T a-Syn overexpressing animals exhibited a higher foot slip percentage—calculated as the ratio between foot slips and total number of steps—compared to EV controls at the earlier 3-week timepoint (Fig. 3D). This difference persisted when animals were re-tested 6 weeks post-injection (Fig. 3D, Table 1). The total number of steps taken by both A53T a-Syn overexpressing animals and EV controls was comparable at both timepoints (Fig. 3E). These results indicate progressive motor impairment following bilateral accumulation of mutant A53T a-Syn in the rat SN, with deficits in sensorimotor coordination emerging as early as 3 weeks post-injection.
To determine whether sensorimotor coordination deficits were due to nigrostriatal dopamine deficiency, we administered L-DOPA to a subset of A53T a-Syn overexpressing animals at week 6 and repeated the grid walking test. L-DOPA administration caused a reduction in the foot slip ratio in these animals compared to their baseline performance (Fig. 3F, Table 1). A similar reduction in foot slip ratio was not observed in saline-treated animals, demonstrating that sensorimotor impairments in the bilateral A53T a-Syn model are responsive to dopaminergic therapy.
Blunted sucrose sensitivity with bilateral SN overexpression of A53T a-Syn
Following characterization of motor deficits in this model, we next examined for behavioral abnormalities that may represent neuropsychiatric features. We first performed the two-bottle sucrose preference test59, a widely used paradigm to assess hedonic responsiveness to a sweetened solution in rodents including rats. At 3 weeks post-AAV injection, both A53T a-Syn overexpressing and EV control animals demonstrated similar sucrose preference, calculated as the amount of sucrose solution consumed relative to the total fluid intake from both bottles (Fig. 4A, Table 1). However, by week 6, A53T a-Syn overexpressing animals displayed a lower sucrose preference compared to EV controls. Specifically, there was a significant increase in sucrose preference from week 3 to week 6 in the EV group, but no change in the A53T group over the same period. Total fluid consumption did not differ between groups or timepoints, indicating that the observed changes in preference were not due to alterations in the ability to drink fluids (Fig. 4B). These results suggest that severe dopaminergic neurodegeneration in the A53T a-Syn model may be associated with the late-stage emergence of depression-like behavior.
Depression-like behavior, anxiety-like behavior, and cognitive functioning in EV and A53T a-Syn overexpressing animals. (A) A53T a-Syn overexpressing animals showed reduced sucrose preference compared to EV controls at week 6 (0.850 ± 0.014 [A53T], 0.895 ± 0.012 [EV]; p = 0.0370) with no difference at week 3 (0.839 ± 0.015 [A53T], 0.830 ± 0.012 [EV]; p > 0.999), indicating elevated depression-like behavior in the A53T group at the late timepoint (Group x Time: F (1, 74) = 4.169, p = 0.0447). (B) No difference in total fluid consumption during the sucrose preference test was found between groups at both timepoints (week 3: 13.84 ± 0.57 g [EV], 14.25 ± 0.44 g [A53T], p > 0.9999; week 6: 14.11 ± 0.69 g [EV], 13.65 ± 0.67 g [A53T], p > 0.9999) (Group: F (1, 74) = 0.001548, p = 0.9687). (C) No anxiety-like behavior was detected in the novelty-suppressed feeding test, as latency to eat did not differ between groups at any timepoint (week 3: 51.2 ± 11.6 s [EV], 36.4 ± 6.5 s [A53T], p = 0.3591; week 6: 28.8 ± 6.6 s [EV], 38.2 ± 4.8 s [A53T], p = 0.7878) (Group: F (1, 74) = 0.1237, p = 0.7260). (D) A non-significant trend towards reduced palatable food consumption was observed in the A53T group in the novelty-suppressed feeding test (week 3: 8.8 ± 0.8 [EV], 8.0 ± 0.5 [A53T], p = 0.3591; week 6: 9.3 ± 1.1 [EV], 7.7 ± 0.5 [A53T], p = 0.2460) (Group × Time: F (1, 74) = 0.3173, p = 0.5749). (E) Comparable time was spent in the periphery in the open field test by both groups at both timepoints (week 3: 17.4 ± 3.5 s [EV], 20.5 ± 3.4 s [A53T], p = 0.8981; week 6: 9.8 ± 1.6 s [EV], 11.1 ± 1.8 s [A53T], p > 0.9999) (Group: F (1, 74) = 0.5779, p = 0.4498). (F) Recognition memory, assessed by the novel object recognition test, was intact in both groups at all timepoints (p > 0.9999) (Group: F (1, 74) = 0.04472, p = 0.8331). Sample sizes: n = 20 for A53T, n = 19 for EV. Data are shown as mean ± SEM. *p \(<\hspace{0.17em}\)0.05, **p \(<\hspace{0.17em}\)0.01 for the post hoc Bonferroni comparison following two-way ANOVA.
The modified novelty-suppressed feeding protocol was applied to assess the development of anxiety-like behaviors in A53T a-Syn overexpressing animals. This test evaluates the delay in initiating feeding when rats are placed in an unfamiliar environment after a period of food deprivation46. Latencies to eat remained comparable between the A53T and EV groups at both the 3- and 6-week timepoints (Fig. 4C, Table 1). Similarly, no significant differences were noted in the amount of food consumed by either group in the novelty-suppressed feeding test (Fig. 4D). In the open field test described above, the time spent in the periphery—a measure of anxiety—did not differ between groups at either timepoint (Fig. 4E, Table 1). Together, these findings suggest that bilateral SN accumulation of mutant A53T a-Syn and subsequent degeneration of dopaminergic neurons is associated with sensorimotor impairments and blunted sucrose sensitivity but not with anxiety-like behavior.
Intact olfactory function and novel object recognition with bilateral SN overexpression of A53T a-Syn
Olfactory dysfunction is a common early symptom of PD and has been proposed as a clinical biomarker for early diagnosis and prognosis15. To assess olfactory function following bilateral SN overexpression of A53T a-Syn, we performed the habituation-dishabituation test using orange and cherry aromas50. A53T a-Syn overexpressing and EV control animals exhibited similar habituation trajectories, as indicated by a steady decline in sniffing time upon repeated exposure to the same odor (trials 1–3 and 4–6) (Fig. 5A,B). Additionally, both groups displayed dishabituation, with increased sniffing time upon introduction of a novel odor (transition from trial 3 to trial 4). The cross-habituation index (CHI), which measures the ability to discriminate between odors, did not differ between the A53T and EV groups at either timepoint (Fig. 5C, Table 1). These results suggest that bilateral SN degeneration due to A53T a-Syn overexpression does not impair olfactory function or the ability to differentiate between scents.
Olfactory function in EV and A53T a-Syn overexpressing animals assessed by the habituation-dishabituation test. Both groups exhibited olfactory habituation, as shown by reduced sniffing time during repeated exposure to the same odor (trials 1–3 and trials 4–6) at week 3 (A) and week 6 (B) post-injection. Sniffing time for each trial was normalized to the first trial in (A) and (B). (C) Odor discrimination, measured by the cross-habituation index (CHI), did not differ between groups at either timepoint (week 3: p = 0.2887; week 6: p = 0.9256), indicating intact olfactory function in A53T a-Syn overexpressing animals (Group: F (1, 74) = 2.450, p = 0.1218). Sample sizes: n = 20 for A53T, n = 19 for EV. Data are shown as mean ± SEM. The post hoc Bonferroni comparison following two-way ANOVA was used in the analysis.
Dementia is a common late-stage feature of PD17. We assessed whether bilateral overexpression of mutant A53T a-Syn in the SN impairs recognition memory using the novel object recognition test. Both A53T a-Syn overexpressing and EV control animals were able to discriminate between the familiar and novel objects, spending significantly more time exploring the novel object during the testing phase. Indeed, no differences in the recognition index were observed between the A53T a-Syn and EV groups at either timepoint (Fig. 4F, Table 1). These results suggest that A53T a-Syn overexpression in the SN does not impair recognition memory within the timeframe and conditions of this study.
Discussion
In this study, we characterized the neurobehavioral consequences of A53T a-Syn overexpression in the bilateral SN of rats, providing a comprehensive evaluation of motor and non-motor phenotypes at early and late timepoints. Our findings demonstrate that this model recapitulates some, but not all, key features of PD. Specifically, we observed dopaminergic neurodegeneration, progressive motor impairments, and depression-like behavior. However, we did not observe any evidence supporting the development of an anxiety-like phenotype, olfactory deficits, or cognitive impairment following bilateral overexpression of A53T a-Syn in the SN. These results highlight the potential utility of the bilateral A53T a-Syn model to study PD-related motor and depressive features, while also underscoring its limitations in replicating the full spectrum of PD-related symptomatology.
In this bilateral AAV-A53T a-Syn model, we observed approximately 63% TH + cell loss in the SN 6 weeks post-injection, which represents greater degeneration than the 53% loss previously reported in the unilateral A53T model using a similar viral titer31. The bilateral A53T a-Syn model was also associated with approximately 20% greater neurodegeneration compared to bilateral SN overexpression of wild-type a-Syn, even when the latter used a 30-fold higher viral titer and a longer post-injection timepoint35. Our findings align with evidence that the A53T mutation enhances a-Syn toxicity, leading to more rapid and severe neurodegeneration36.
Motor deficits in PD are closely linked to dopaminergic neuron loss in the SN60, and our findings support this well-established relationship. The grid walking test, which assesses sensorimotor coordination, detected subtle motor impairments even at the early timepoint, while the open field test revealed more pronounced deficits as neurodegeneration progressed. The responsiveness of the sensorimotor impairments to L-DOPA provides face validity for this model. Interestingly, the model exhibited fine motor skill deficits in the grid walking before gross motor impairments were observed in the open field, which is representative of the progression of motor symptoms in humans.
The absence of olfactory and cognitive deficits in our model likely reflects the localized overexpression of A53T a-Syn in the SN4,61. In humans, olfactory dysfunction and cognitive impairment are strongly associated with pathology in the olfactory bulb14 and cortex62, respectively. Indeed, a-Syn overexpression in the olfactory bulb14 and cerebral cortex63 recapitulates these symptoms.
Depression-like behavior in our model was observable at 6 weeks post-injection, coinciding with severe dopaminergic neuron loss. This is not unexpected, as the basal ganglia are functionally integrated with limbic circuits64, and disruption of these pathways is known to contribute to motivational and affective disturbances64,65. Consistent with this, toxin-based PD models with severe nigrostriatal degeneration caused by intranigral 6-OHDA or MPTP injections also display depression-like phenotypes66,67. By impairing synaptic vesicle dynamics and neurotransmitter release4,25, a-Syn overexpression itself may alter signaling pathways that contribute to affective dysfunction. The inconsistent observation of depression-like behaviors among a-Syn overexpression studies34,35 may reflect differences in the severity of dopaminergic degeneration. Our findings are consistent with this threshold effect, as depression-like behavior was absent at 3 weeks post-injection but emerged once cell loss exceeded ~ 60%. Taken together, these results suggest that while a-Syn pathology may impair neurotransmitter systems, the emergence of a depression-like phenotype is dependent on the extent of nigrostriatal neurodegeneration.
Our study was conducted exclusively in female rats, a design that requires consideration within the broader context of sexual dimorphism in PD. It is often reported that the male-to-female prevalence ratio of PD is approximately 1.5:168, although more recent studies suggest that this sex difference may be less pronounced69. Clinically, women often exhibit a later onset, a higher prevalence of tremor-dominant subtypes70, and report a greater burden of specific non-motor symptoms (e.g., depression, anxiety) and levodopa-induced dyskinesias71. These disparities are likely due to a complex interplay of genetics, neuroanatomy, neuroimmune function, and the neuroprotective effects of estrogen72,73,74,75. Preclinical studies in toxin-based models often report greater nigrostriatal vulnerability in males, whereas emerging data from genetic a-Syn-based models suggest females may develop more pronounced a-Syn pathology76. Crucially, sex differences in the AAV-mediated A53T a-Syn model remain unexplored. Therefore, our characterization establishes an essential behavioral and pathological baseline in females. The specific depression-like phenotype we observed aligns with the clinical burden of affective symptoms in women with PD71. Future studies directly comparing sexes in this model will be critical to determine if anhedonia represents a core feature of A53T a-Syn-mediated neurodegeneration or exhibits sex-dependent expression, thereby refining the model’s translational utility for studying PD-related non-motor symptoms.
While this study provides a detailed characterization of motor and non-motor phenotypes in the bilateral AAV-A53T a-Syn model, several limitations should be acknowledged. Despite performing assays for cognition, olfaction, and anxiety, we did not observe deficits in these domains, suggesting that overexpression restricted to the SN may be insufficient to recapitulate the broader spectrum of non-motor symptoms. Future studies could address this by targeting additional PD-relevant regions, such as the olfactory bulb or cortical areas, either individually or in combination, to better model the multisystem nature of PD. Mechanistic investigations are also warranted to clarify how a-Syn pathology contributes to neurotransmitter dysfunction, for example by measuring monoamine levels across the striatum and probing a-Syn effects on synaptic plasticity and neurotrophic factor signaling. Finally, while the behavioral assays employed here are validated and widely used, complementary tasks could further strengthen the evaluation of non-motor domains, provided care is taken to minimize motor confounds and carry-over effects. Together, these directions represent important opportunities to refine a-Syn overexpression models and improve their translational relevance for both motor and non-motor aspects of PD.
Conclusion
Our findings demonstrate that the rat model with bilateral SN overexpression of A53T a-Syn recapitulates progressive motor impairments and depression-like behavior. The emergence of depression-like behavior at the late timepoint, coinciding with severe dopaminergic neuron loss, highlights the complex interplay between a-Syn pathology, neurodegeneration, and neuropsychiatric symptoms. This model represents a promising tool for studying PD-related motor and depressive symptoms. However, its localized pathology limits its ability to replicate the full spectrum of PD symptoms, highlighting the need for future studies to explore extranigral a-Syn overexpression and its effects on other non-motor symptoms.
Data availability
All data generated or analysed during this study are included in this published article or available from the corresponding author on reasonable request.
Abbreviations
- 6-OHDA:
-
6-Hydroxydopamine
- AAV:
-
Adeno-associated virus
- ANOVA:
-
Analysis of variance
- AP:
-
Anteroposterior
- a-Syn:
-
Alpha-synuclein
- CHI:
-
Cross-habituation index
- DV:
-
Dorsoventral
- EV:
-
Empty vector
- i.p.:
-
Intraperitoneal
- L-DOPA:
-
L-3,4-dihydroxyphenylalanine methyl ester
- ML:
-
Mediolateral
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- PD:
-
Parkinson’s disease
- SEM:
-
Standard error of the mean
- SN:
-
Substantia nigra pars compacta
- TH:
-
Tyrosine hydroxylase
References
Kalia, L. V., Kalia, S. K., McLean, P. J., Lozano, A. M. & Lang, A. E. α-Synuclein oligomers and clinical implications for Parkinson disease. Ann. Neurol. 73, 155–169 (2013).
Meade, R. M., Fairlie, D. P. & Mason, J. M. Alpha-synuclein structure and Parkinson’s disease – lessons and emerging principles. Mol. Neurodegener. 14, 29 (2019).
Calabresi, P. et al. Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction. Cell Death Dis. 14, 176 (2023).
Koprich, J. B., Kalia, L. V. & Brotchie, J. M. Animal models of α-synucleinopathy for Parkinson disease drug development. Nat. Rev. Neurosci. 18, 515–529 (2017).
Laux, G. Parkinson and depression: Review and outlook. J. Neural. Transm. 129, 601–608 (2022).
Broen, M. P. G., Narayen, N. E., Kuijf, M. L., Dissanayaka, N. N. W. & Leentjens, A. F. G. Prevalence of anxiety in Parkinson’s disease: A systematic review and meta-analysis. Mov. Disord. 31, 1125–1133 (2016).
Kehagia, A. A., Barker, R. A. & Robbins, T. W. Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson’s disease. Lancet Neurol. 9, 1200–1213 (2010).
Doty, R. L. Olfactory dysfunction in Parkinson disease. Nat. Rev. Neurol. 8, 329–339 (2012).
McGregor, M. M. & Nelson, A. B. Circuit mechanisms of Parkinson’s disease. Neuron 101, 1042–1056 (2019).
Chaudhuri, K. R., Healy, D. G. & Schapira, A. H. Non-motor symptoms of Parkinson’s disease: Diagnosis and management. Lancet Neurol. 5, 235–245 (2006).
Church, F. C. Treatment options for motor and non-motor symptoms of Parkinson’s disease. Biomolecules 11, 612 (2021).
Blesa, J., Foffani, G., Dehay, B., Bezard, E. & Obeso, J. A. Motor and non-motor circuit disturbances in early Parkinson disease: Which happens first?. Nat. Rev. Neurosci. 23, 115–128 (2022).
Brown, R. G. et al. Depression and anxiety related subtypes in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 82, 803–809 (2011).
Chen, F. et al. α-Synuclein aggregation in the olfactory bulb induces olfactory deficits by perturbing granule cells and granular–mitral synaptic transmission. NPJ Parkinsons Dis. 7, 114 (2021).
Fullard, M. E., Morley, J. F. & Duda, J. E. Olfactory dysfunction as an early biomarker in Parkinson’s disease. Neurosci. Bull. 33, 515–525 (2017).
Janvin, C., Aarsland, D., Larsen, J. P. & Hugdahl, K. Neuropsychological profile of patients with Parkinson’s disease without dementia. Dement. Geriatr. Cogn. Disord. 15, 126–131 (2003).
Levy, G. et al. Memory and executive function impairment predict dementia in Parkinson’s disease. Mov. Disord. 17, 1221–1226 (2002).
Khan, H. F. et al. Site-specific seeding of Lewy pathology induces distinct pre-motor cellular and dendritic vulnerabilities in the cortex. Nat. Commun. 15, 10775 (2024).
Mazzotta, G. M. & Conte, C. Alpha synuclein toxicity and non-motor Parkinson’s. Cells 13, 1265 (2024).
Konnova, E. A. & Swanberg, M. Animal models of Parkinson’s disease. in Parkinson’s Disease: Pathogenesis and Clinical Aspects 83–106 (Codon Publications, 2018).
Blesa, J. & Przedborski, S. Parkinson’s disease: Animal models and dopaminergic cell vulnerability. Front. Neuroanat. 8, 155 (2014).
Cenci, M. A. & Björklund, A. Animal models for preclinical Parkinson’s research: An update and critical appraisal. 252, 27–59 (2020).
Schober, A. Classic toxin-induced animal models of Parkinson’s disease: 6-OHDA and MPTP. Cell Tissue Res 318, 215–224 (2004).
Kanaan, N. M. & Manfredsson, F. P. Loss of functional alpha-synuclein: A toxic event in Parkinson’s disease?. J. Parkinsons Dis. 2, 249–267 (2012).
Cheng, F., Vivacqua, G. & Yu, S. The role of alpha-synuclein in neurotransmission and synaptic plasticity. J. Chem. Neuroanat. 42, 242–248 (2011).
Dawson, T. M., Ko, H. S. & Dawson, V. L. Genetic animal models of Parkinson’s disease. Neuron 66, 646–661 (2010).
Fernagut, P.-O. & Chesselet, M.-F. Alpha-synuclein and transgenic mouse models. Neurobiol. Dis. 17, 123–130 (2004).
Ip, C. W. et al. AAV1/2-induced overexpression of A53T-α-synuclein in the substantia nigra results in degeneration of the nigrostriatal system with Lewy-like pathology and motor impairment: a new mouse model for Parkinson’s disease. Acta Neuropathol. Commun. 5, 11 (2017).
Chung, C. Y., Koprich, J. B., Siddiqi, H. & Isacson, O. Dynamic changes in presynaptic and axonal transport proteins combined with striatal neuroinflammation precede dopaminergic neuronal loss in a rat model of AAV α-synucleinopathy. J. Neurosci. 29, 3365–3373 (2009).
Koprich, J. B. et al. Progressive neurodegeneration or endogenous compensation in an animal model of Parkinson’s disease produced by decreasing doses of alpha-synuclein. PLoS ONE 6, e17698 (2011).
Musacchio, T. et al. Temporal, spatial and molecular pattern of dopaminergic neurodegeneration in the AAV-A53T α-synuclein rat model of Parkinson’s disease. Behav. Brain Res. 432, 113968 (2022).
Koprich, J. B., Johnston, T. H., Reyes, M. G., Sun, X. & Brotchie, J. M. Expression of human A53T alpha-synuclein in the rat substantia nigra using a novel AAV1/2 vector produces a rapidly evolving pathology with protein aggregation, dystrophic neurite architecture and nigrostriatal degeneration with potential to model the pathology of Parkinson’s disease. Mol. Neurodegener. 5, 43 (2010).
Musacchio, T., Koprich, J. B. & Ip, C. W. Modeling with the A53T α-synuclein model of Parkinson’s disease. in Genetics, Neurology, Behavior, and Diet in Parkinson’s Disease 677–687 (Elsevier, 2020).
Campos, F. L. et al. Rodent models of Parkinson’s disease: Beyond the motor symptomatology. Front. Behav. Neurosci. 7, 175 (2013).
Caudal, D., Alvarsson, A., Björklund, A. & Svenningsson, P. Depressive-like phenotype induced by AAV-mediated overexpression of human α-synuclein in midbrain dopaminergic neurons. Exp. Neurol. 273, 243–252 (2015).
Lu, J., Sun, F., Ma, H., Qing, H. & Deng, Y. Comparison between α-synuclein wild-type and A53T mutation in a progressive Parkinson’s disease model. Biochem. Biophys. Res. Commun. 464, 988–993 (2015).
Hui, S. et al. Mitophagy upregulation occurs early in the neurodegenerative process mediated by α-synuclein. Mol. Neurobiol. 61, 9032–9042 (2024).
Nim, S. et al. Disrupting the α-synuclein-ESCRT interaction with a peptide inhibitor mitigates neurodegeneration in preclinical models of Parkinson’s disease. Nat. Commun. 14, 2150 (2023).
Musacchio, T. et al. Subthalamic nucleus deep brain stimulation is neuroprotective in the A53T α-synuclein Parkinson’s disease rat model. Ann. Neurol. 81, 825–836 (2017).
Hofman, K. et al. Low β predicts motor output and cell degeneration in the A53T Parkinson’s disease rat model. Brain 148, 4058-4071 (2025).
Van Den Eeden, S. K. Incidence of Parkinson’s disease: Variation by age, gender, and race/ethnicity. Am. J. Epidemiol. 157, 1015–1022 (2003).
Martinez-Martin, P. et al. Gender-related differences in the burden of non-motor symptoms in Parkinson’s disease. J. Neurol. 259, 1639–1647 (2012).
Chen, K. S. et al. Small molecule inhibitors of α-synuclein oligomers identified by targeting early dopamine-mediated motor impairment in C. elegans. Mol. Neurodegener. 16, 77 (2021).
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Coordinates. (2013).
López-Alonso, V. E. et al. The central blockade of the dopamine DR4 receptor decreases sucrose consumption by modifying the microstructure of drinking behavior in male rats. IBRO Neurosci. Rep. 14, 195–201 (2023).
Blasco-Serra, A., González-Soler, E. M., Cervera-Ferri, A., Teruel-Martí, V. & Valverde-Navarro, A. A. A standardization of the novelty-suppressed feeding test protocol in rats. Neurosci. Lett. 658, 73–78 (2017).
Dulawa, S. C. & Hen, R. Recent advances in animal models of chronic antidepressant effects: The novelty-induced hypophagia test. Neurosci. Biobehav. Rev. 29, 771–783 (2005).
Bodnoff, S. R., Suranyi-Cadotte, B. E., Quirion, R. & Meaney, M. J. Role of the central benzodiazepine receptor system in behavioral habituation to novelty. Behav. Neurosci. 103, 209–212 (1989).
Francis-Oliveira, J. et al. Fluoxetine exposure during pregnancy and lactation: Effects on acute stress response and behavior in the novelty-suppressed feeding are age and gender-dependent in rats. Behav. Brain Res. 252, 195–203 (2013).
Tarland, E. & Brosda, J. Male rats treated with subchronic PCP show intact olfaction and enhanced interest for a social odour in the olfactory habituation/dishabituation test. Behav. Brain Res. 345, 13–20 (2018).
Arbuckle, E. P., Smith, G. D., Gomez, M. C. & Lugo, J. N. Testing for odor discrimination and habituation in mice. J Vis. Exp. 99, 52615 (2015).
Wang, Y., Wu, Z., Bai, Y.-T., Wu, G.-Y. & Chen, G. Gad67 haploinsufficiency reduces amyloid pathology and rescues olfactory memory deficits in a mouse model of Alzheimer’s disease. Mol. Neurodegener. 12, 73 (2017).
Antunes, M. & Biala, G. The novel object recognition memory: neurobiology, test procedure, and its modifications. Cogn. Process 13, 93–110 (2012).
Chao, O. Y., Pum, M. E., Li, J.-S. & Huston, J. P. The grid-walking test: assessment of sensorimotor deficits after moderate or severe dopamine depletion by 6-hydroxydopamine lesions in the dorsal striatum and medial forebrain bundle. Neuroscience 202, 318–325 (2012).
Shihan, M. H., Novo, S. G., Le Marchand, S. J., Wang, Y. & Duncan, M. K. A simple method for quantitating confocal fluorescent images. Biochem. Biophys. Rep. 25, 100916 (2021).
Phan, J.-A. et al. Early synaptic dysfunction induced by α-synuclein in a rat model of Parkinson’s disease. Sci. Rep. 7, 6363 (2017).
Visanji, N. P. et al. α-Synuclein-based animal models of Parkinson’s disease: Challenges and opportunities in a new era. Trends Neurosci. 39, 750–762 (2016).
Deumens, R., Blokland, A. & Prickaerts, J. Modeling Parkinson’s disease in rats: An evaluation of 6-OHDA lesions of the nigrostriatal pathway. Exp. Neurol. 175, 303–317 (2002).
Liu, M.-Y. et al. Sucrose preference test for measurement of stress-induced anhedonia in mice. Nat Protoc 13, 1686–1698 (2018).
Lee, C. S., Sauer, H. & Björklund, A. Dopaminergic neuronal degeneration and motor impairments following axon terminal lesion by intrastriatal 6-hydroxydopamine in the rat. Neuroscience 72, 641–653 (1996).
Gómez-Benito, M. et al. Modeling Parkinson’s disease with the alpha-synuclein protein. Front. Pharmacol. 11, 356 (2020).
Mattila, P. M. et al. Alpha-synuclein-immunoreactive cortical Lewy bodies are associated with cognitive impairment in Parkinson’s disease. Acta. Neuropathol. 100, 285–290 (2000).
Hatami, A. & Chesselet, M.-F. Transgenic rodent models to study alpha-synuclein pathogenesis, with a focus on cognitive deficits. Curr. Top. Behav. Neurosci. 22, 303–330 (2015).
Buot, A. & Yelnik, J. Functional anatomy of the basal ganglia: Limbic aspects. Rev. Neurol. (Paris) 168, 569–575 (2012).
Azizi, S. A. Monoamines: Dopamine, norepinephrine, and serotonin, beyond modulation, “switches” that alter the state of target networks. Neuroscientist 28, 121–143 (2022).
Santiago, R. M. et al. Induction of depressive-like behavior by intranigral 6-OHDA is directly correlated with deficits in striatal dopamine and hippocampal serotonin. Behav. Brain. Res. 259, 70–77 (2014).
Santiago, R. M. et al. Depressive-like behaviors alterations induced by intranigral MPTP, 6-OHDA, LPS and rotenone models of Parkinson’s disease are predominantly associated with serotonin and dopamine. Prog. Neuropsychopharmacol. Biol. Psychiatry 34, 1104–1114 (2010).
Elbaz, A., Carcaillon, L., Kab, S. & Moisan, F. Epidemiology of Parkinson’s disease. Rev. Neurol. (Paris) 172, 14–26 (2016).
Zirra, A. et al. Gender differences in the prevalence of Parkinson’s disease. Mov. Disord. Clin. Pract. 10, 86–93 (2023).
Reekes, T. H. et al. Sex specific cognitive differences in Parkinson disease. NPJ Parkinsons Dis. 6, 7 (2020).
Maas, B. R. et al. Age and gender differences in non-motor symptoms in people with Parkinson’s disease. Front. Neurol. 15, 1339716 (2024).
Sawada, H. & Shimohama, S. Estrogens and Parkinson disease: Novel approach for neuroprotection. Endocrine 21, 77–80 (2003).
Gillies, G. E., Murray, H. E., Dexter, D. & McArthur, S. Sex dimorphisms in the neuroprotective effects of estrogen in an animal model of Parkinson’s disease. Pharmacol. Biochem. Behav. 78, 513–522 (2004).
Murray, H. E. et al. Dose- and sex-dependent effects of the neurotoxin 6-hydroxydopamine on the nigrostriatal dopaminergic pathway of adult rats: differential actions of estrogen in males and females. Neuroscience 116, 213–222 (2003).
Bovenzi, R. et al. Sex-specific immune-biological profiles in Parkinson’s disease. J. Neuroimmunol. 403, 578610 (2025).
Lamontagne-Proulx, J. et al. Sex and age differences in a progressive synucleinopathy mouse model. Biomolecules 13, 977 (2023).
Funding
This project was supported by the RR Tasker Chair in Functional Neurosurgery (SKK), the Wolfond and Krembil Chair in PD Research (LVK) and CRANIA support funds (SKK, TV and LM).
Author information
Authors and Affiliations
Contributions
Conceptualization: LK, MK, LVK, SKK; Methodology: LK, MK, IS; Formal analysis: LK, MK, IS; Investigation: LK; Resources: SKK, LVK, LM, TV; Data curation: LK, CT, RM, SJ, HC, SH, PQ, JG; Writing—original draft: LK, MK; Writing—review and editing: LK, MK, SKK, LVK; Visualization: LK; Supervision: SKK, LVK, TV, LM; Project administration: SKK, LVK; Funding acquisition: SKK, LVK, TV, LM.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kondrataviciute, L., Kapadia, M., Chau, H. et al. Characterization of motor and non-motor features associated with bilateral nigral degeneration due to A53T alpha-synuclein in female rats. Sci Rep 16, 5462 (2026). https://doi.org/10.1038/s41598-025-34884-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-34884-6