Abstract
In the cultivation and production of sweet cherry, the cost of picking fruit is high due to inconsistency in the maturation period, which has affected the development of the cherry industry. In this study, the effects of exogenous abscisic acid (ABA) on the sweet cherry variety ‘Luying 3’ fruit quality and maturation stage were observed and recorded, and the physiological and molecular mechanisms were explored to systematically analyze the effects of ABA on sweet cherry fruit ripening to promote the development of the cherry industry. Exogenous ABA (400 mg L−1) enhanced the color of ‘Luying 3’ fruit in the developing stage but had no significant effect on the fruit weight, soluble solid content, titratable acid content, and sugar-acid ratio in the mature stage. The application of ABA significantly promoted the secretion of endogenous ABA, gibberellin (GA) and salicylic acid (SA). A total of 766 differentially expressed genes (DEGs) were obtained between the treatment group and the control group at 47 and 54 d after flowering. The DEGs were significantly enriched in plant hormone signal transduction pathway, MAPK plant signal transduction pathway and glycolysis pathway. Six genes related to the synthesis of endogenous hormones were screened, of which five were upregulated and one was downregulated. Four DEGs related to the sweet cherry fruit metabolic rate were upregulated by ABA, which positively regulated fruit ripening. Eight differentially expressed AP2/ERF transcription factors were identified, of which 5 were upregulated and 3 were downregulated. This study provides a theoretical foundation for the application of ABA in promoting the consistency of cherry fruit maturity.
Similar content being viewed by others
Introduction
The European sweet cherry (Prunus avium) is native to Europe and South Asia, including a small isolated area in the western Himalayas, and is commonly referred to as sweet cherry1. Sweet cherry, which belongs to the Rosaceae family, is a Prunus species in the subgroup Cerasus2. Sweet cherry cultivation has the advantages of early fruiting, quick effects, environmental safety, and so on. According to the Food and Agriculture Organization of the United Nations (FAO) database (https://www.fao.org/home/zh), in 2020, sweet cherry was the main product or one of the main products, with a global cultivation area of 451,000 hectares, and the total output reached 2.687 million tons. Given China’s booming economy in recent years, the cultivation area has reached 11,000 hectares. In 2020, with an annual output of 44,000 tons of cherries, China was the world’s largest producer and importer of cherries, with an annual import volume of more than 40,000 tons. Cherries have become an important and lucrative tree species for farmers. However, despite the rapid development of the sweet cherry industry, several factors limit its growth. For example, the maturity of the fruit of a single tree is not consistent during the production of sweet cherry, making it difficult to pick fruit, as the fruits need to be picked individually, resulting in an increase in harvesting costs. Therefore, plant growth regulators are widely used to regulate the ripening of sweet cherry and achieve the greatest economic benefits3,4,5.
Physiological changes occur during fruit ripening, starting with changes in the respiration rate. Changes in the respiration rate play a decisive role in fruit ripening and aging6, and fruits can be classified as climacteric fruits (such as tomatoes, apples and bananas) or non-climacteric fruits (such as grapes, strawberries and cherries). These processes differ in terms of ethylene production and respiration patterns at the beginning of the maturation process7,8,9. Climacteric fruits exhibit a respiratory burst at the onset of ripening, accompanied by a large increase in ethylene production, which regulates chloroplast pigment differentiation and carotenoid accumulation, and fruit ripening can also be initiated by exposure to exogenous ethylene10,11. In non-climacteric fruits, in which increased respiration does not occur, abscisic acid (ABA), but not ethylene, appears to be the key factor affecting the ripening process. Because the activity of biosynthetic enzymes was inhibited, the activity of ABA-degrading enzymes (CYP and UGT) was relatively high, and the level of ABA was low in strawberry during the early stage of development. With continuous ripening, the ABA level gradually increased with the upregulation of the FvNCED1 and FvABA2 genes. It peaked at the “full red” stage of strawberry fruits12,13. During the ripening of these fruits, ABA is directly associated with reduced fruit acidity, changes in peel color (mainly by promoting anthocyanin production), increased vacuolar hexose concentrations, and cell wall modifications that contribute to fruit softening14,15. Exogenous ABA treatment of figs has been shown to regulate the expression of endogenous ABA synthesis genes (FcNCED2, etc.) and ethylene synthesis genes (FcACO2, FcACS4, etc.), thereby affecting the secretion of endogenous ABA and ethylene and promoting the ripening of fig fruit14. In tomato, banana and other fruits, the phenomena associated with fruit ripening, such as deepening of the color, increased softening, and an increased soluble sugar content, were found to occur earlier after ABA application16,17,18. Sweet cherry is also a type of non-climacteric fruit. Studies have shown that the application of ABA can promote the ripening of cherry fruits and make the ripening stage of cherry fruits more consistent, which is conducive to one-time harvesting and reduces harvesting costs19,20,21. However, studies of the effects of exogenous ABA treatment on fruit ripening in sweet cherry have been limited to in vitro shoots or harvested fruits19,22, and further research is needed to investigate the maternal manifestations. Therefore, in this study, a cherry variety (‘Luying 3’) was treated with 400 mg L−1 ABA to observe the changes in the appearance and internal quality of sweet cherry fruit, and the differentially expressed genes (DEGs) after ABA treatment were analyzed to identify the key genes that are regulated by ABA treatment. This study aimed to explore the effects of ABA treatment on the ripening of sweet cherry fruit at the physiological and molecular levels and to provide a theoretical basis for the rational use of ABA in the production and cultivation of sweet cherry fruit.
Materials and methods
Test material
The sweet cherry variety ‘Luying 3’ which was a new variety independently bred by our team was selected as the experimental material, and its father and mother were ‘Tieton’ and ‘Summit’, respectively. It was approved by the Shandong Forest Tree Varieties Examination and Approval Committee in 2018, and the approval number is’ Lu S-SV-CA-016-2018’. The test site is located in our own test base, -the Jinniushan Test Demonstration Base, Daiyue District, Tai ‘an City, Shandong Province, China. Forty days after flowering, the fruit entered the color transition period. Six new trees of the cherry variety ‘Luying 3’ with more orderly tree growth and a uniform fruit size were selected and divided into a control (CK) group and a treatment (T) group, with 3 trees in each group. The fruits were sprayed 40 d after flowering (May 27) with water in the control group and 400 mg L−1 ABA in the treatment group, with a total of approximately 150 ml applied to all the fruits of each tree. The criterion for fruit maturity was that the size of the fruit did not change further, the fruit turned completely red, and the soluble solid content was stable.
The fruits were first collected 40 d after flowering and labeled OG. Each tree was treated as a biological replicate with 3 replicates and 20 fruits were collected from each replicate. After full mixing, 10 fruits were randomly selected, and their pulp was chopped, placed in a centrifuge tube and quickly frozen with liquid nitrogen. The samples were stored at -80℃ for RNA extraction, transcriptome sequencing, and the determination of endogenous hormone contents. The other 50 fruits were placed in an ice box and brought back to the laboratory for imaging and determinations of the single-fruit weight, transverse diameter, longitudinal diameter, soluble solid (TSS) content and titratable acid content (TA). Afterward, samples from the control group and treatment group were collected every 7 days (i.e., 47 days and 54 days after flowering, respectively).
Test methods
Determination of the fruit appearance quality
The single-fruit weight, transverse diameter and longitudinal stems were measured. Ten fruits were randomly selected from each treatment group and weighed with an electronic scale, and the average single-fruit weight was calculated. The transverse diameter of the fruit was measured along the largest transverse section of the fruit with Vernier calipers, and the longitudinal stem of the fruit was measured along the ventral suture of the fruit.
For the examination of fruit color, twenty fruits from each treatment group were randomly selected, arranged in order of color and photographed to record their color. Five samples were randomly selected form each treatment groups, and the color parameters of lightness (L), red-green value (a) and yellow-blue value (b) were measured using a Konica Minolta portable color difference meter (CR-400).
Fruit TSS and TA contents and sugar-acid ratio (TSS/TA)
A PAL-BX丨ACID 16 cherry sugar acidity meter was used for determination to measure these parameters.
Determination of endogenous hormone contents
ABA, gibberellin (GA3), ethylene (ACC), and salicylic acid (SA) levels in sweet cherry pulp samples were determined via liquid chromatography23.
Transcriptome sequencing
RNA was extracted from the samples using the polysaccharide and polyphenol plant total RNA extraction kit from Tiangen. The A260, A260/A230, A260/A280 and RNA concentrations (µg µL−1) of the RNA samples were measured with an IMPLEN Ultramicro UV spectrophotometer. RNA integrity was assessed using a Fragment Analyzer 5400 (Agilent Technologies, CA, USA).
After the extracted RNA samples were qualified, the total RNA was used as the input material for RNA sample preparation, sequencing libraries were generated using the Illumina® NEBNext®UltraTM RNA Library Preparation Kit, and index codes were added to attribute sequences to each sample. Fastp software (v0.19.7) was used for basic statistics and quality control of the raw sequencing data.
The fragments per kilobase per million mapped reads (FPKM) value was used to calculate gene expression. Then, TCC-GUI was used to screen DEGs between samples receiving different treatments, and the screening criterion was as a P value < 0.05. Gene heatmaps were generated using Cluster3.0. The false discovery rate (FDR) (Benjamini and Hochberg (BH)) method was used to ccorrect the results for multiple testing. KEGG pathways with corrected p values less than 0.05 were considered significantly enriched pathways.
iTAK software was used to predict the transcription factors (TFs) of ‘Luying 3’, and the TFs in the samples were identified based on the TFs and rules that had been classified and defined in the database.
qRT-PCR
Beacon Designer software was used to design the qRT‒PCR primers (Supplementary Table S1), which were synthesized by Shanghai Bioengineering Co., Ltd. qRT‒PCR was performed and amplified using the cDNA samples of the constructed RNA-seq library with CYP2 (GenBank search number: TC1916) as the internal reference gene according to the instructions of the SuperReal PreMix Plus (SYBR Green) Kit.
Result and analysis
Effects of exogenous ABA on the external morphology of sweet cherry fruits
Exogenous ABA promotes fruit coloration in sweet cherry
As shown in Fig. 1A,B, the application of exogenous ABA at 400 mg L−1 clearly promoted the coloration of the ‘Luying 3’ fruit and made its maturity stage more consistent in terms of appearance. Forty days after flowering, the fruits of ‘Luying 3’ were in the green‒green stage, and at 47 d after flowering, the fruits of the treatment group were more uniform than those of the control group. Most of the fruits in the control group were green-red (25, 50%), a small portion of the fruits were light-red (15, 30%), and the number of fruits in the red stage was the lowest (10, 20%), while the fruits in the treatment group were not green-red, but were light red or red, accounting for 40% and 60%, respectively. At 54 d after flowering, the fruits in the treatment group were nearly ripe and turned dark red (50, 100%), whereas those in the control group were still red (20, 40%).
Moreover, the results of the colorimeter measurements (Fig. 1C–E) also revealed that the standard deviations of the L, a and b values among individuals in the treatment group were significantly lower than those in the control group at 47 and 54 days after flowering; the L value was significantly lower than that in the control group, and the a and b values were significantly higher than those in the control group, indicating that the color in the treatment group was more uniform and that fruit maturity was more consistent.
Effects of ABA on fruit coloration in sweet cherry. (A) color difference; (B) color distribution; (C) L value; (D) a value; (E) b value.
Effects of exogenous ABA on fruit morphology during fruit development
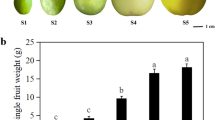
After the 400 mg L−1 ABA treatment, the fruit size and weight of ‘Luying 3’ changed during its growth and development. Under the influence of exogenous ABA, the average transverse diameter of the fruit in the treatment group (T) at 47 d after flowering increased by approximately 7.50%, the average vertical diameter of the fruit increased by approximately 6.37%, and the average single-fruit weight increased by approximately 5.98% compared with that of the control group (CK). At 54 d after flowering, the average transverse diameters of fruits in the treatment group (T) and the control group (CK) were 31.143 mm and 31.491 mm, respectively; the average vertical diameters of the fruits were 25.283 mm and 25.918 mm, respectively, and the average single-fruit weights were 11.411 g and 11.200 g, respectively (Fig. 2A–C). A significant difference was not observed between the two groups. In conclusion, ABA spraying at 400 mg L−1 did not affect the fruit shape or size of ‘Luying 3’.
Effects of exogenous ABA on the intrinsic quality of fruits at maturity
The taste attributes of sweet cherry fruit strongly affect consumer acceptance. Sweetness can be reported as the soluble solid content, acidity can be reported as the titratable acidity, and the sugar-acid ratio is considered an overall taste attribute. By measuring the TA content, soluble solid content and sugar-acid ratio of the fruits of the control group (CK) and the treatment group (T) at the mature stage at 54 d after flowering, we observed (Fig. 2D–F) that the average soluble solid contents were 20.99% and 20.72%, the average TA contents were 1.335% and 1.348%, and the sugar-acid ratios were 15.72 and 15.37, respectively. No significant differences in the TSS content, TA content and sugar-acid ratio between the control group and the treatment group. Our results indicated that spraying exogenous ABA at 400 mg L−1 had little effect on the intrinsic quality of the fruits of ‘Luying 3’.
Effects of ABA on fruit morphology and intrinsic quality. (A) average transverse diameters at different developmental stages; (B) average vertical diameters at different developmental stages; (C) average single-fruit weights at different developmental stages; (D) TSS content at 54 d after flowering (E) TA content at 54 d after flowering (F) sugar-acid ratio at 54 d after flowering.
Effects of exogenous ABA on the endogenous hormone contents in fruits
According to the determination of the endogenous hormone contents in the control group and the treatment group at 40 d, 47 d and 54 d after flowering (Fig. 3), the levels of IAA and ABA tended to increase during the fruit ripening process of ‘Luying 3’ from 40 d to 54 d after flowering. The levels of GA3 and SA decreased gradually, indicating that IAA and ABA played major roles in the ripening process of sweet cherry fruit after the developmental stage. Under the influence of exogenous ABA, the IAA content in ‘Luying 3’ was reduced by 42.77% and 45.34% at 47 d and 54 d after flowering, respectively. The endogenous ABA content increased by 70.34% at 47 d after flowering but decreased by 20.94% at 54 d after flowering. The endogenous GA3 content increased by 601.37% at 47 d after flowering and decreased by 19.64% at 54 d after flowering. The endogenous SA content increased by 7.88% and 24.45% at 47 d and 54 d after flowering, respectively. In summary, ABA treatment promoted the secretion of endogenous ABA, GA and SA but inhibited the secretion of IAA.
Levels of different endogenous hormones in sweet cherry fruits at different maturation stages. (A) Endogenous IAA content; (B) Endogenous ABA content; (C) Endogenous GA3 content; (D) Endogenous SA content. * P < 0.05, ** P < 0.01, and *** P < 0.001.
Transcriptome sequencing analysis of sweet cherry fruits treated with exogenous ABA
Statistical analysis of the sequencing results
A total of 213.94 G of raw data were generated by sequencing, and these raw data were submitted to the NCBI SRA database. After strict quality assessment and data filtering, 206.01 G of clean data were obtained. A total of 42,394,486 to 47,864,488 clean reads were obtained from each library, and the average base sequencing accuracy was greater than 94.92%. The Q20 rate, Q30 rate and GC content of the clean reads from each sample were greater than 96.67%, 91.68% and 45.38%, respectively (Supplementary Table S2). The results revealed that the clean reads obtained by sequencing were of good quality and could be used for subsequent research.
Transcriptome changes during the ripening of ABA-treated sweet cherry fruits
Gene expression in fruit was analyzed at 40 d, 47 d and 54 d after flowering and the results are shown in a heatmap (Supplementary Fig. S1), the gene expression levels at different ripening stages were clearly divided into three categories. At 40 d, 47 d and 54 d after flowering, gene expression in the control group and the treatment group was also basically divided into two categories. These results indicated that although ABA treatment affected gene expression in the fruits of ‘Luying 3’, the degree of similarity during the same period was still high.
Analysis of differentially expressed genes at different maturation stages
A total of 7357 DEGs were identified in the control group (OG vs. CK-47 and CK-47 vs. CK-54) at different developmental stages, 919 of which were differentially expressed in both groups. A total of 8938 DEGs were identified in the treatment group at different developmental stages (OG vs. T-47 and T-47 vs. T-54), among which 1488 genes were differentially expressed in both groups. The expression of 766 different genes differed between the treatment and control groups (CK-47 vs. T-47 and CK-54 vs. T54) during the same period, 24 of which were differentially expressed in both groups (Supplementary Fig. S2A).
From 40 d to 47 d after flowering in ‘Luying 3’, the number of DEGs in the control group (OG vs. CK-47) was approximately the same as that in the treatment group (OG vs. T-47), and the number of upregulated and downregulated genes was also approximately the same. From 47 d to 54 d after flowering, the number of DEGs in the treatment group (T-47 vs. T-54) increased compared with that in the control group (CK-47 vs. CK-54), and the number of downregulated genes increased to a greater extent than the number of upregulated genes. The number of DEGs detected at 54 d after flowering (T-54 vs. CK-54) was greater than that at 47 d after flowering (T-47 vs. CK-47) between the control group and the treatment group (Supplementary Fig. S2B).
KEGG functional annotation and metabolic pathway enrichment analysis of DEGs
A total of 766 DEGs between the control group and the treatment group at 47 d and 54 d after flowering of ‘Luying 3’ were analyzed by determining enriched KEGG pathway, and different pathways of DEGs enriched in ‘Luying 3’ under the influence of 400 mg L−1 exogenous ABA were obtained (Supplementary Table S3). A total of 452 unigenes were annotated against the KEGG database, and the KEGG pathways of 452 sequences were enriched mainly in metabolic pathways, genetic information processing pathways and environmental information processing pathways. Among them, metabolic pathways contained the most unigenes (271), followed by environmental information processing pathways. A total of 32 terms were included. By analyzing 766 DEGs, some genes that may be affected by exogenous ABA and regulate sweet cherry fruit ripening, such as PavABF2 (FUN_007552-T1), PavPR1 (FUN_030157-T1), PavETR (FUN_039903-T1), PavTCH4 (FUN_000946-T1), PavPP2C (FUN_039354-T1), PavEBF1_2 (FUN_006443-T1), PavADH1 (FUN_027523-T1), PavADH5 (FUN_027515-T1), PavALDH (FUN_035255-T2), and PavALDO (FUN_038194-T1), were screened.
Among them, ‘Luying 3’ bloomed 47 d later, and the upregulated genes were enriched mainly in fatty acid biosynthesis, tyrosine metabolism, alpha-linolenic acid metabolism, glycolysis/gluconeogenesis, and biosynthesis of secondary metabolites (Fig. 4A). The downregulated genes were enriched mainly in the MAPK signaling pathway-plant, galactose metabolism, phosphatidylinositol signaling system, leucine and isoleucine biosynthesis (valine, leucine and isoleucine biosynthesis), pantothenate and CoA biosynthesis, and other pathways (Fig. 4B).
Bubble plot of the enriched KEGG pathways of the DEGs. (A) KEGG pathways of upregulated DEGs at 47 d; (B) KEGG pathways of downregulated DEGs at 47 d; (C) KEGG pathways of upregulated DEGs at 54 d; (D) KEGG pathways of downregulated genes at 54 d.
Fifty-four days after ‘Luying 3’ blossomed, the upregulated genes were enriched mainly in protein processing in the endoplasmic reticulum, glycolysis/gluconeogenesis and metabolic pathways, plant hormone signal transduction, sulfur metabolism, photosynthesis, the photosynthesis-antenna protein pathway, carbon metabolism, fructose and mannose metabolism, and purine metabolism (Fig. 4C). The downregulated genes were enriched mainly in phenylalanine, tyrosine and tryptophan biosynthesis, base excision repair, peroxisome, sesquiterpenoid and triterpenoid biosynthesis, plant hormone signal transduction, and other pathways (Fig. 4D).
Analysis of environmental information processing pathways in ‘Luying 3’ in response to ABA treatment
Analysis of DEGs related to the plant hormone signaling pathway
Through the KEGG enrichment analysis of the plant hormone signal transduction pathways in the KEGG database (Fig. 5), exogenous ABA was shown to affect the IAA, ABA, ethylene (ETH), brassinolide (BR) and SA signaling pathways in the fruits of ‘Luying 3’. Downregulation of the AUX cotransporter in the IAA signaling pathway and upregulation of the IAA-responsive gene SAUR increased cell size and fruit growth. The downregulation of PP2C, a negative regulator of the ABA signaling pathway, and the upregulation of SnRK2 protein kinase and ABA response element binding factor (ABF) promoted stomatal closure and fruit ripening. The expression of the ethylene receptor ETR on the endoplasmic reticulum was upregulated in the ethylene signaling pathway, and EBF1_2, which plays an important role in regulating the ethylene signaling pathway by regulating EIN3 protein degradation, was upregulated. The expression of TCH4 in the BR signaling pathway, which promotes cell elongation, was upregulated and ultimately promoted fruit ripening and senescence. PR-1 was upregulated in the SA signaling pathway. Since this pathway is involved in the fruit ripening process, the PavABF2 (FUN_007552-T1), PavPR1 (FUN_030157-T1), PavETR (FUN_039903-T1), PavTCH4 (FUN_000946-T1), PavPP2C (FUN_039354-T1), and PavEBF1_2 (FUN_006443-T1) genes, which are predicted to be the key genes affecting fruit ripening in this pathway, were screened according to changes in gene expression. Among them, the PavABF2, PavPR1, PavETR, PavTCH4, and PavEBF1_2 genes were upregulated, and PavPP2C was downregulated.
Plant hormone signal transduction pathways.
Analysis of DEGs related to the MAPK signal transduction pathway in plants
Through the KEGG enrichment analysis of MAPK plant signal transduction pathways in the KEGG database (Fig. 6), we found that pathogen infection, pathogen attack, plant hormones, and salt/drought/osmotic stress pathways were affected by exogenous ABA treatment. The expression of PR-1, which can be used for pathogen defense in the pathogen infection and pathogen attack pathways, was upregulated. The expression of the ethylene receptors ETR and EBF1_2 in the ethylene signaling pathway in the plant hormone pathway increased, the expression of the negative regulator PP2C in the ABA signaling pathway in the salt/drought/osmotic stress pathway was downregulated, and the expression of the mitogen-activated protein kinase MAPKKK17_18 decreased. Therefore, we screened PavPR1 (FUN_030157-T1), PavETR (FUN_039903-T1), PavTCH4 (FUN_000946-T1), PavPP2C (FUN_039354-T1), and PavEBF1_2 (FUN_006443-T1) as the key genes affecting fruit ripening in this pathway according to the changes in gene expression, and all of the genes were upregulated.
MAPK signal transduction pathways in plants.
Analysis of DEGs related to the glycolytic metabolic pathway in ‘Luying 3’ in response to ABA treatment
By annotating of glycolytic metabolic pathways via KEGG (Fig. 7), we found that both 6-phosphofructokinase (PFK) and fructose diphosphate aldolase (ALDO) were upregulated, which promoted the production of β-D-fructose-1,6-diphosphate through the pentose phosphate pathway. The expression of glyceraldehyde-3-phosphate dehydrogenase (gapN) was downregulated, and the expression of phosphoglycerate mutase (gpmI) was upregulated. These two enzymes are associated with light and carbon fixation in organisms. The expression of pyruvate kinase (PK) was upregulated, which can promote the production of pyruvate. The expression of pyruvate decarboxylase (PDC), aldehyde dehydrogenase (ALDH), alcohol dehydrogenase-1 (ADH1) and alcohol dehydrogenase-5 (ADH5) was upregulated, which could promote pyruvate metabolism, and this process is also connected to the tricarboxylic acid cycle. Among them, alcohol dehydrogenase-1 (ADH1) and alcohol dehydrogenase-5 (ADH5) can also promote aromatic biosynthesis. We screened the PavADH1 (FUN_027523-T1), PavADH5 (FUN_027515-T1), PavALDH (FUN_035255-T2), and PavALDO (FUN_038194-T1) genes related to this pathway based on the changes in gene expression; these genes were predicted to be the key genes promoting sweet cherry fruit ripening in this pathway, and all the genes were upregulated.
Glycolytic metabolic pathway.
TF analysis
iTAK software was used to predict TFs in the whole genome of ‘Luying 3’, and 1610 genes were predicted to encode TFs; these genes were further divided into 69 categories. Among them, the FAR1 was the most common (140, 8.69%), followed by the MYB (119, 7.39%), AP2/ERF (110, 6.83%), NAC (108, 6.71%), bHLH (104, 6.46%). We selected eight AP2/ERF (Supplementary Table S4) for a gene expression analysis by annotating gene functions and analyzing metabolic pathways combined with a transcriptomic assessment of the effect of the 400 mg L−1 exogenous ABA treatment on gene expression.
As shown in Fig. 8, the expression patterns of PavAP2-1, PavAP2-2, PavAP2-3 and PavAP2-4 were the same in the fruit ripening process of ‘Luying 3’ from 40 d to 54 d after flowering. Therefore, the expression levels of these four genes increased with the continuous ripening of ‘Luying 3’ fruit. The expression levels of all four genes were higher at 54 d after flowering. After ABA treatment, the expression levels of the PavAP2-1, PavAP2-2, PavAP2-3 and PavAP2-4 TFs were significantly different between the control group and the treatment group. Among them, the PavAP2-1 and PavAP2-4 TFs were not affected by ABA at 47 d after flowering. However, the gene expression level increased at 54 d after flowering. ABA inhibited the expression of the PavAP2-2 transcription factor, at 47 d and 54 d after flowering. Exogenous ABA delayed the expression of the PavAP2-3 gene, repressing it at 47 d but no longer inhibiting it at 54 d after flowering, restoring the normal gene expression level. In general, exogenous ABA promoted the expression of the PavAP2-1 and PavAP2-4 TFs while inhibiting the expression of the PavAP2-2 and PavAP2-3 TFs.
The expression patterns of the PavAP2-5 and PavAP2-6 genes during the fruit ripening process of ‘Luying 3’ from 40 d to 54 d after flowering showed an “up-down” trend, and the trend for PavAP2-6 was more obvious. The expression of these two genes first increased but then decreased with the continuous ripening of the fruit, and the expression level was higher in the fruit at 47 d after flowering, suggesting that these two genes play important roles in fruit ripening in ‘Luying 3’ at 47 d after flowering. The expression of PavAP2-5 increased significantly at 47 and 54 d after ABA treatment. The effects of PavAP2-6 were not obvious at 47 d after flowering, but the expression of PavAP2-6 was significantly inhibited by exogenous ABA at 54 d after flowering.
The expression patterns of PavAP2-7 and PavAP2-8 were the same during the fruit ripening process of ‘Luying 3’ from 40 d to 54 d after flowering, and both showed a decreasing trend. Therefore, the expression levels of these two genes decreased with the continuous ripening of the ‘Luying 3’ fruit. We speculated that the regulatory effects of these two genes on the ripening of ‘Luying 3’ fruit mainly occurred before 40 d after flowering. Exogenous ABA had little effect on the PavAP2-7 TFs at 47 d after flowering but promoted their expression at 54 d after flowering, and for the PavAP2-8 TFs, exogenous ABA promoted expression at both 47 d and 54 d after flowering.
Effects of ABA treatment on the expression levels of AP2/ERF transcription factors.
Real-time quantitative PCR
Ten DEGs related to the plant hormone signal transduction pathway, MAPK plant signal transduction pathway and glycolysis metabolism pathway and 8 AP2/ERF TFs related to sweet cherry fruit ripening were selected from the RNA-seq data, and the selected genes were tested via qRT-PCR, as shown in Fig. 9. The relative expression levels of 18 genes were similar to the results of the RNA-seq analysis.
The PavABF2 gene is involved in the plant hormone signal transduction pathway, and its expression was significantly promoted by exogenous ABA at 54 d after flowering. The PavPR1 gene is involved in the plant hormone signal transduction pathway and the MAPK signal transduction pathway. ABA inhibited PavPR1 expression at 47 d after flowering but promoted PavPR1 expression at 54 d after flowering. The PavETR gene is involved in the plant hormone signal transduction pathway and MAPK signal transduction pathway, and its expression was promoted by ABA at 54 d after flowering. The PavTCH4 gene is involved in the plant hormone signal transduction pathway, and its expression level was increased nearly 10-fold by ABA treatment at 54 d after flowering. PavPP2C is involved in the plant hormone signal transduction pathway and the MAPK signal transduction pathway. ABA inhibited PavPP2C expression at 47 d and 54 d after flowering. The PavEBF1_2 gene is involved in the plant hormone signal transduction pathway and MAPK signal transduction pathway, and its expression was significantly promoted by ABA at 54 d after flowering. The PavADH1 and PavADH5 genes are involved in the glycolytic metabolic pathway and were stimulated by ABA at 47 d and 54 d after flowering. The PavALDH gene is involved in the glycolytic metabolic pathway, and its expression was inhibited at 47 d after anthesis and promoted at 54 d after anthesis by ABA. The expression of the PavALDO gene increased at both 47 d and 54 d after flowering, and its expression increased nearly 6-fold at 54 d after flowering under the influence of exogenous ABA.
Plots of trends in the real-time fluorescence quantitative PCR expression and transcriptomic.
Discussion
Effects of exogenous ABA on fruit shape and endogenous hormone levels in sweet cherry
The ripening process of sweet cherry involves changes in skin color, softening of the fruit texture, fructose and glucose accumulation in the pulp, and darkening of the pulp color. In sweet cherry, ABA is associated with the regulation of anthocyanin synthesis and sensory properties (TSS/TA ratio)15. Time et al.‘s21 canopy spraying test on 4 cherry varieties with 400 mg/l ABA revealed that the treatment 1) enhanced sweet cherry fruit color in the ‘Glenred’, ‘Lapins’ and ‘Bing’ cultivars, but not in the ‘Royal Rainier’ cultivar (a bi-colored cultivar), and 2) decreased fruit size and firmness in the ‘Lapins, ‘Bing’ and ‘Royal Rainier’ cultivars and increased Lapins’ fruit color without reducing size. Moreover, in addition to ABA, cytokinins, JA, GA, and IAA play roles in the ripening of sweet cherry fruit4,5. In this study, the effects of exogenous ABA on sweet cherry fruits were studied. Based on appearance, exogenous ABA significantly enhanced the color of ‘Luying 3’ fruit during the development stage but had no significant effects on the transverse diameter, vertical stem, and single-fruit weight at the mature stage. By measuring the soluble sugar content, soluble acid content and sugar-acid ratio of ‘Luying 3’ fruit in the mature stage, exogenous ABA had no significant effect on the taste attributes of ‘Luying 3’ fruit after reaching full maturity. In addition, the effects of exogenous ABA on the levels of endogenous hormones in sweet cherry fruit of ‘Luying 3’ were measured during the development period were measured. Exogenous ABA treatment promoted the secretion of endogenous ABA, GA and SA in sweet cherry, which was consistent with previous results14. In summary, exogenous ABA spraying accelerated the ripening of ‘Luying 3’ fruit during the developmental period, resulting in more consistent fruit maturity stages but having little effect on the final fruit size, weight, and taste attributes.
Analysis of ripening pathways associated with exogenous ABA treatment in sweet cherry
The DEGs associated with the response to ABA in ‘Luying 3’ were enriched mainly in the plant hormone signal transduction pathway, the MAPK plant signal transduction pathway and the glycolysis metabolism pathway, and these three pathways are related to the ripening process of cherry fruit24,25. In the plant hormone signal transduction pathway, exogenous ABA affected the IAA, ABA, ETH, BR and SA signaling pathways in ‘Luying 3’ fruit. We found that PavABF2 (FUN_007552-T1), PavPR1 (FUN_030157-T1), PavETR (FUN_039903-T1), PavTCH4 (FUN_000946-T1), PavPP2C (FUN_039354-T1), and PavEBF1_2 (FUN_006443-T1) were strongly affected by ABA and regulated endogenous hormone secretion in fruit, thereby affecting fruit ripening. The expression of PavABF2, PavPR1, PavETR, PavTCH4 and PavEBF1_2 was upregulated, whereas that of PavPP2C was downregulated. The MAPK signal transduction pathway, pathogen infection, pathogen attack, plant hormones, and salt/drought/osmotic stress pathways were affected by exogenous ABA, and the plant hormone pathway was the main pathway affected. The PavPR1 (FUN_030157-T1), PavETR(FUN_039903-T1), PavTCH4 (FUN_000946-T1), PavPP2C (FUN_039354-T1), and PavEBF1_2(FUN_006443-T1) genes were significantly upregulated by ABA, which caused a stress response and changes in endogenous hormone secretion and subsequently affected fruit ripening. In the glycolytic pathway, the PavADH1 (FUN_027523-T1), PavADH5 (FUN_027515-T1), PavALDH (FUN_035255-T2) and PavALDO (FUN_038194-T1) genes were significantly upregulated following ABA treatment. ABA can accelerate respiratory metabolism in sweet cherry fruit and subsequently promote the ripening of sweet cherry fruit. Our results suggested that exogenous ABA promoted the ripening of sweet cherry fruit mainly by affecting endogenous hormone secretion and the metabolic rate in the plants.
Analysis of AP2/ERF TF gene expression levels in response to exogenous ABA treatment
AP2/ERF transcription factors act as important regulators of many biological and physiological processes, such as hormone signaling, responses to various stresses, plant morphogenesis, and metabolite regulation26,27, and an increasing number of studies have shown that the AP2/ERF transcription factor family plays a key regulatory role in fruit ripening and is involved in the taste change, pigmentation and flavor formation of fruits by regulating downstream ripening-related genes28,29,30. In tomato, 55 ERF subfamily members presented a ripening-related expression pattern, of which 27 members were upregulated and 28 members were downregulated. Five AP2-like genes were also identified in tomato, among which SL4P2a was expressed at a low level in green immature fruits and increased at the fruit color transition stage. The expression level of this gene in the red ripening stage of fruit maintained is high, indicating that it is closely associated with fruit ripening31. In strawberry, FaRAV1 (an AP2/ERF transcription factors) activates the expression of FaMYB10, a gene important for anthocyanin biosynthesis, by binding to its promoter, which in turn promotes strawberry coloration32.
In this study, a total of 8 AP2/ERF TFs affected by exogenous ABA were screened. Among them, PavAP2-1, PavAP2-2, PavAP2-3, and PavAP2-4 exhibited the same expression pattern during the fruit ripening process of ‘Luying 3’ from 40 d to 54 d after flowering, and they all presented an increasing trend. The expression levels of these four genes were higher at 54 d after flowering, suggesting that the effects of these four genes on promoting sweet cherry fruit mainly ripening occurred 54 d after flowering. The expression patterns of PavAP2-5 and PavAP2-6 were the same during the fruit ripening process of ‘Luying 3’ from 40 d to 54 d after flowering, with both showing an “up-down” trend, and the expression level was higher in the fruit at 47 d after flowering. We speculated that these genes had a significant effect on the fruit ripening of ‘Luying 3’ fruit at approximately 47 d after flowering. The expression patterns of PavAP2-7 and PavAP2-8 were the same during the fruit ripening process of ‘Luying 3’ from 40 d to 54 d after flowering, with both showing a decreasing trend. We speculated that the regulatory effects of these two genes on ‘Luying 3’ fruit ripening occurs mainly before 40 d after flowering or that they are not involved in the ripening process of sweet cherry fruit. An analysis of the gene expression of multiple AP2/ERF TFs following exogenous ABA treatment revealed that exogenous ABA promoted the expression of the PavAP2-1, PavAP2-4, PavAP2-5, PavAP2-7 and PavAP2-8 TFs. However, the expression of the PavAP2-2, PavAP2-3 and PavAP2-6 TFs was inhibited. Therefore, further exploration, such as via bioinformatics analysis, of genes whose effects differ greatly should be performed.
Applications and prospects of ABA in the cherry fruit industry
Exogenous ABA also plays a crucial role in regulating plant growth and development, improving fruit quality, and fruit ripening. It promotes fruit ripening by regulating the synthesis of the endogenous hormone ETH and the accumulation of sugars, carotenoids, and anthocyanins7,14,33,34. Exogenous ABA can regulate genes related to endogenous ABA synthesis (Fc NCED2, etc.) and ETH synthesis (Fc ACO2, Fc ACS4, etc.), thereby affecting the secretion of endogenous ABA and ETH and promoting the ripening of fig fruit. In tomato, banana and other fruits, ABA enhances color, softens fruit, and increase the soluble sugar content17,18. Ren and Leng19 reported that exogenous ABA promoted the synthesis of endogenous ABA in light red sweet cherry. By treating ‘Luying 3’ fruit, we also found that exogenous ABA promoted the ripening of sweet cherry fruit by affecting endogenous hormone secretion and the metabolic rate of cherry fruit, making the fruit maturity stage more consistent but having no significant effect on the final size, weight, and taste attributes of the fruit. Therefore, exogenous ABA can be applied as appropriate to promote the consistent maturation of the sweet cherry fruit ‘Luying 3’, reduce labor in the process of fruit picking, and promote the development of the cherry industry. However, further verification of these results in different sweet cherry varieties and the appropriate concentrations for different varieties is needed. Moreover, affordable products containing ABA have been developed and applied to the agricultural production of sweet cherry.
Conclusion
In this study, exogenous ABA (400 mg L−1) promoted the consistency of cherry fruit maturity but had no significant effect on the final size, weight and taste attributes of the fruit. Moreover, the application of ABA promoted the secretion of endogenous ABA, GA and SA. In the analysis of changes in the transcriptome, differentially expressed genes were significantly enriched in the plant hormone signal transduction pathway, MAPK plant signal transduction pathway and glycolysis pathways, and 9 key differentially expressed genes and 8 PavAP2 transcription factors were screened and shown to be involved in the effect of exogenous ABA on sweet cherry fruit ripening. This study provides a theoretical foundation for the application of ABA in promoting the consistency of cherry fruit maturity.
Data availability
Data is provided within the manuscript or supplementary information files.
References
Faust, M. & Surányi, D. Origin and dissemination of cherry. Hortic. Rev. 19, 263–317. https://doi.org/10.1002/9780470650622.ch5 (1997).
Jung, S. et al. GDR (Genome Database for Rosaceae): Integrated web-database for Rosaceae genomics and genetics data. Nucleic Acids Res. 36, D1034–D1040. https://doi.org/10.1186/1471-2105-5-130 (2008).
Rademacher, W. Plant growth regulators: backgrounds and uses in plant production. Plant. Growth Regul. 34, 845–872. https://doi.org/10.1007/s00344-015-9541-6 (2015).
Castro, R. I. et al. Effect of exogenous auxin treatment on cell wall polymers of strawberry fruit. Int. J. Mol. Sci. 22(12), 6294. https://doi.org/10.3390/IJMS22126294 (2021).
Liu, C. et al. Exogenous gibberellin treatment improves fruit quality in self-pollinated apple. Plant. Physiol. Biochem. 174, 11–21. https://doi.org/10.1016/j.plaphy.2022.01.029 (2022).
Perotti, V. E., Moreno, A. S. & Podestá, F. E. Physiological aspects of fruit ripening: the mitochondrial connection. Mitochondrion 17, 1–6. https://doi.org/10.1016/j.mito.2014.04.010 (2014).
Alexander, L. & Grierson, D. Ethylene biosynthesis and action in tomato: a model for climacteric fruit ripening. J. Exp. Bot. 53(377), 2039–2055. https://doi.org/10.1093/jxb/erf072 (2002).
Pandey, P. V. & Srivastava, R. The fading distinctions between classical patterns of ripening in climacteric and non-climacteric fruit and the ubiquity of ethylene-An overview. J. Food Sci. Technol. 49, 1–21. https://doi.org/10.1007/s13197-011-0293-4 (2012).
Fukano, Y. & Tachiki, Y. Evolutionary ecology of climacteric and non-climacteric fruits. Biol. Lett. 17, 20210352. https://doi.org/10.1098/rsbl.2021.0352 (2021).
Giovannoni, J. J. Genetic regulation of fruit development and ripening. Plant. Cell. 16(Suppl. 1), S170–S180. https://doi.org/10.2307/3872309 (2004).
Klee, H. J. & Giovannoni, J. J. Genetics and control of tomato fruit ripening and quality attributes. Annu. Rev. Genet. 45, 41–59. https://doi.org/10.1146/annurev-genet-110410-132507 (2011).
Li, C., Jia, H., Chai, Y. & Shen, Y. Abscisic acid perception and signaling transduction in strawberry: a model for non-climacteric fruit ripening. Plant. Signal. Behav. 6(12), 1950–1953. https://doi.org/10.4161/psb.6.12.18024 (2011).
Li, B. J., Grierson, D., Shi, Y. & Chen, K. S. Roles of abscisic acid in regulating ripening and quality of strawberry, a model non-climacteric fruit. Hortic. Res. 9, uhac089. https://doi.org/10.1093/hr/uhac089 (2022).
Qiao, H., Zhang, H., Wang, Z. & Shen, Y. Fig fruit ripening is regulated by the interaction between ethylene and abscisic acid. J. Integr. Plant. Biol. 63(3), 553–569. https://doi.org/10.1111/jipb.13065 (2021).
Kou, X. et al. Different regulatory mechanisms of plant hormones in the ripening of climacteric and non-climacteric fruits: a review. Plant. Mol. Biol. 107(6), 477–497. https://doi.org/10.1007/s11103-021-01199-9 (2021).
Jiang, Y., Joyce, D. C. & Macnish, A. J. Effect of abscisic acid on banana fruit ripening in relation to the role of ethylene. J. Plant. Growth Regul. 19(1), 106–111. https://doi.org/10.1007/s003440000011 (2000).
Jia, H. et al. Abscisic acid and sucrose regulate tomato and strawberry fruit ripening through the abscisic acid-stress-ripening transcription factor. Plant. Biotechnol. J. 14(10), 2045–2065. https://doi.org/10.1111/pbi.12563 (2016).
Tao, X. et al. Effects of exogenous abscisic acid on bioactive components and antioxidant capacity of postharvest tomato during ripening. Molecules 25(6), 1346. https://doi.org/10.3390/molecules25061346 (2020).
Ren, J. & Leng, P. Role of abscisic acid and ethylene in fruit maturation of sweet cherry. Act. Hortic. Sin. 37(02), 199–206. https://doi.org/10.16420/j.issn.0513-353x.2010.02.007 (2010).
Kuhn, N. ABA influences color initiation timing in P. Avium L. fruits by sequentially modulating the transcript levels of ABA and anthocyanin-related genes. Tree Genet. Genomes. https://doi.org/10.1007/s11295-021-01502-1 (2021).
Time, A. et al. Canopy spraying of abscisic acid to improve fruit quality of different sweet cherry cultivars. Agronomy 11, 1947. https://doi.org/10.3390/agronomy11101947 (2021).
Kondo, S. & Gemma, H. Relationship between abscisic acid (ABA) content and maturation of the sweet cherry. Engei Gakkai Zasshi. 62, 63–68. https://doi.org/10.2503/jjshs.62.63 (1993).
Blum, F. High performance liquid chromatography. Br. J. Hosp. Med. (Lond) 75(2), C18–21. https://doi.org/10.12968/hmed.2014.75.Sup2.C18 (2014).
Lunt, S. Y. & Vander Heiden, M. G. Aerobic glycolysis: meeting the metabolic requirements of cell proliferation. Annu. Rev. Cell. Dev. Biol. 27, 441–464. https://doi.org/10.1146/annurev-cellbio-092910-154237 (2011).
Meng, X. & Zhang, S. MAPK cascades in plant disease resistance signaling. Annu. Rev. Phytopathol. 51, 245–266. https://doi.org/10.1146/annurev-phyto-082712-102314 (2013).
Gu, C. et al. Multiple regulatory roles of AP2/ERF transcription factor in angiosperm. Bot. Stud. 58(1), 6. https://doi.org/10.1186/s40529-016-0159-1 (2017).
Feng, K. et al. Advances in AP2/ERF super-family transcription factors in plant. Crit. Rev. Biotechnol. 40(6), 750–776. https://doi.org/10.1080/07388551.2020.1768509 (2020).
Liu, M., Diretto, G., Roustan, J. P. & Li, Z. The chimeric repressor version of an ethylene response factor (ERF) family member, sl-erf.b3, shows contrasting effects on tomato fruit ripening. New Phytol. 203(1), 206–218. https://doi.org/10.1111/nph.12771 (2014).
Zhang, C. & Whiting, M. Plant growth regulators improve sweet cherry fruit quality without reducing endocarp growth. Sci. Hortic. 150, 73–79 (2013).
Zhang, L. et al. Function analysis of the erf and dreb subfamilies in tomato fruit development and ripening. Front. Plant. Sci. 13, 849048. https://doi.org/10.3389/fpls.2022.849048 (2022a).
Zhang, X. et al. Transcriptome analysis reveals roles of sucrose in anthocyanin accumulation in ‘kuerle xiangli’ (Pyrus sinkiangensis Yü). Genes (Basel) 13(6), 1064. https://doi.org/10.1016/j.gene.2019.144284 (2022).
Zhang, Y. et al. Genome-wide analysis of the erf family and identification of potential genes involved in fruit ripening in octoploid strawberry. Int. J. Mol. Sci. 23(18), 10550. https://doi.org/10.3390/ijms231810550 (2022).
Leng, P. The role of abscisic acid in fruit ripening and responses to abiotic stress. J. Exp. Bot. 65, 4577–4588. https://doi.org/10.1093/jxb/eru204 (2014).
Bai, Q., Huang, Y. & Shen, Y. The physiological and molecular mechanism of abscisic acid in regulation of fleshy fruit ripening. Front. Plant. Sci. 11, 619953. https://doi.org/10.3389/fpls.2020.619953 (2021).
Funding
This study was supported by the Key R&D Program of Shandong Province, China (2022TZXD006 and 2020LGGC008) and the Shandong Provincial Natural Science Foundation (ZR2021MC117), and the Yantai Seed Project ‘Creation of the sweet cherry Breeding Team’.
Author information
Authors and Affiliations
Contributions
JW, QL and QQ conducted the experiments, analyzed the data. QQ and BS wrote the main manuscript text. KL, DZ, PH, LZ, JS, SS, QL, YG and SZ prepared Figs. 1, 2, 3, 4, 5, 6, 7, 8 and 9. QQ, BS, JW and QL revised and checked the manuscript.All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.


Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qiao, Q., Shen, B., Lin, K. et al. Detecting the physiological and molecular mechanisms by which abscisic acid (ABA) regulates the consistency of sweet cherry fruit maturity. Sci Rep 15, 6311 (2025). https://doi.org/10.1038/s41598-025-85821-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-85821-6