Abstract
We assessed the risk factors of neonatal seizures and their influences on neurodevelopment in very-low-birth-weight infants (VLBWIs). 16,279 VLBWIs born in 2013–2020 were included using Korean Neonatal Network data to assess the risk factors of neonatal seizures. 7,599 VLBWIs born in 2013–2018 and followed up at 18–24 months corrected age were included to assess the influence of seizures on mortality and any neurodevelopmental impairment (NDI) including blindness, deafness, cerebral palsy (CP), and mental and motor NDI based on Bayley test (NDI-Bayley). We also compared clinical factors between no NDI and any NDI groups after neonatal seizures. 1,430 infants (8.8%) had neonatal seizures. Significant risk factors for neonatal seizures were lower GA, male gender, congenital anomaly, small for GA, delivery room resuscitation, maternal chorioamnionitis, surfactant use, hypotension, severe intraventricular hemorrhage (IVH), post-hemorrhagic hydrocephalus, sepsis, meningitis, and necrotizing enterocolitis (NEC). In the subgroup analysis, lower GA and male gender increased the risk of seizures only in GA < 28 weeks group. Neonatal seizures increased the risk of deafness, CP, and NDI-Bayley. Significant risk factors for any NDI after seizures were severe IVH and NEC. Providing the best treatments for morbidities of VLBWIs in NICUs could prevent neonatal seizures and NDI after seizures.
Similar content being viewed by others
Introduction
The incidence of neonatal seizures varies from 9 to 58 per 1,000 very low birth weight infants (VLBWIs)1,2, higher than the incidence of neonatal seizures in term infants. The etiology of neonatal seizures usually includes fetal distress or hypoxic-ischemic brain injury during delivery, maternal infection, maternal diabetes, stroke, cerebral malformations or metabolic disturbances such as hypoglycemia and hypocalcemia in term infants3. However, many neonatal morbidities associated with preterm infants, including intra-ventricular hemorrhage (IVH) and infection, can also result in neonatal seizures during neonatal intensive care unit (NICU) admissions in VLBWIs4,5. Compared to the many reports about neonatal seizures in term infants, the information about neonatal seizures in preterm infants is limited, especially in Korea.
Regarding the long-term prognosis of neonatal seizures, neonatal seizures were reported to have a significant role in developing epilepsy, intellectual disability, and headache in the total population, including term infants, using the U.S. claim database6. In preterm infants, neonatal seizures were independently associated with higher odds of death or significant neurodevelopmental impairment (NDI) at 18–24 months of age in several reports1,7. However, we do not know which factors affect poor neurodevelopmental outcomes in VLBWIs with neonatal seizures.
In this study, we used a nationwide cohort to assess the risk factors of neonatal seizures in VLBWIs and whether there are any differences in the risk factors according to the gestational age (GA) at birth. We also assessed the long-term neurodevelopmental outcomes of neonatal seizures in VLBWIs and the risk factors of any major NDI in VLBWIs with neonatal seizures.
Methods
Study population
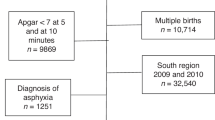
This was a prospective cohort study using the Korean Neonatal Network (KNN) database. For risk factor analysis of neonatal seizure during NICU admissions, VLBWIs born between 2013 and 2020 and admitted to the NICUs participating in the KNN with a GA ≥ 22 weeks were included in the analysis. Nineteen infants admitted in NICUs for more than 1 year and 5 infants whose gender was unknown were excluded; therefore, a total of 16,279 infants were included in the analysis (Fig. 1).
Study population for risk factor analysis of neonatal seizures.
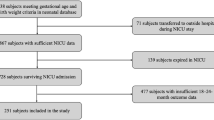
To assess the long-term neurodevelopmental outcomes of neonatal seizures at 18–24 months corrected age, VLBWIs born between 2013 and 2018 and admitted to the NICUs participating in the KNN with a GA ≥ 22 weeks were included. Eleven infants admitted for more than 1 year, 2 infants with unknown gender, 1,737 infants who died during hospitalization, and 3,131 infants whose follow-up data were not reported were excluded in the analysis; therefore, a total of 7,599 infants were included (Fig. 2).
Study population for the influence of neonatal seizures on long-term neurodevelopmental outcomes.
Risk factor analysis of neonatal seizures
Maternal, perinatal, and neonatal characteristics were compared between two groups according to the presence of neonatal seizures in the whole infant group, and subgroup analysis was done according to the GA at birth: < 28 weeks group and 28–36 weeks group. Maternal characteristics included maternal age, maternal education, multiple gestation, cesarean section, in-vitro fertilization (IVF), gestational diabetes, pregnancy-induced hypertension (PIH), histologic chorioamnionitis (HCA), and oligohydramnios. Perinatal and demographic characteristics were compared, including GA at birth, birth weight, gender, congenital anomalies, inborn, any dose of antenatal steroid use, 5-minute Apgar scores, cord pH, delivery room resuscitation, and small for GA (SGA). The following neonatal outcomes were collected for the risk factor analysis of neonatal seizures: surfactant use, patent ductus arteriosus (PDA) with treatment, hypotension within one-week-olds, IVH ≥ grade 3 for the Papile classification8, post-hemorrhagic hydrocephalus, sepsis within 14 days after birth, meningitis, and necrotizing enterocolitis (NEC) ≥ stage 2 for the modified Bell’s criteria9. We included sepsis only within 14 days after birth for the risk factor analysis of neonatal seizures in consideration of the possible time order.
Neurodevelopmental outcomes of neonatal seizures
Infants registered in the KNN cohort were followed up at 18–24 months corrected age. At this visit, medical history and special service use interview after NICU discharge, measurement of weight, height, and head circumference, and comprehensive neurodevelopmental assessments, including Bayley scales of infant development (BSID) and Korean developmental screening test for infants and children (K-DST), were performed. Medical treatment after NICU discharge included oxygen treatment, ventilator care, tracheostomy, nasogastric tube feeding, ventriculoperitoneal shunt, anti-epileptic medication, rehabilitation and language treatment, and pulmonary hypertension treatment. We also searched for the history of readmission and neuroimaging, including computed tomography (CT)/ magnetic resonance imaging (MRI). The data collected at this age also included mortality after NICU discharge, blindness, deafness (hearing loss requiring hearing aids), and cerebral palsy (CP). BSID second or third edition (II or III) were used to assess long-term neurodevelopment. We evaluated the influence of seizures on post-discharge mortality, blindness, deafness, CP and NDI based on BSID (mental and motor developmental impairment) by multivariable logistic regression analysis with the backward elimination method adjusting for antenatal characteristics and perinatal and neonatal morbidities in NICUs.
We also compared clinical factors between no major NDI group and any major NDI group after neonatal seizures.
Statistical analysis
All the continuous variables were expressed as the median and interquartile range, and the categorical variables were expressed as the numbers and proportions. Comparisons of the maternal and neonatal characteristics between two groups according to the presence of neonatal seizures during NICU admissions and at 18–24 months follow-up were performed using the Mann-Whitney U test for continuous variables and chi-square test or Fisher’s exact test for categorical variables. Multivariable logistic regression analyses with backward elimination methods were done adjusting for GA, male gender, congenital anomaly, SGA, cesarean section, delivery room resuscitation, antenatal corticosteroid use, inborn, gestational diabetes, PIH, HCA, surfactant use, PDA requiring treatment, hypotension within the first week of life, IVH ≥ grade 3, post-hemorrhagic hydrocephalus, sepsis within 14 days after birth, meningitis, and NEC ≥ stage 2 to assess the risk factors of neonatal seizures in the whole group and the two different groups according to GA at birth: < 28 weeks and 28–36 weeks. We also performed multivariable logistic regression analysis with the backward elimination method to assess the influence of neonatal seizures on long-term neurodevelopmental outcomes adjusting for maternal age, maternal education ≥ college, gestational diabetes, PIH, HCA, oligohydramnios, antenatal steroid use, cesarean section, GA at birth, male gender, congenital anomalies, multiple births, IVF, SGA, delivery room resuscitation, surfactant use, PDA treatment, hypotension within one-week-old, NEC ≥ stage 2, sepsis, IVH ≥ grade 3, cystic periventricular leukomalacia (PVL), moderate to severe bronchopulmonary dysplasia (BPD), postnatal steroid use, retinopathy of prematurity (ROP) treatment, and days to full feeding. To assess the risk factors of any major NDI in infants after neonatal seizure in NICU admissions, multivariable logistic regression analysis with the backward elimination method was done, adjusting for variables statistically significant in the univariable analysis between no major NDI group and any major NDI group. All analyses were performed with R version 3.5.0 (http://www.r-project.org) with the statistical significance set at a P-value < 0.05.
Definitions
Clinical variables are defined according to the KNN operation manual. A seizure was defined as a seizure that required anti-epileptic medications during NICU admission. PIH was defined as any maternal diagnosis of preeclampsia, eclampsia, or hemolysis, elevated liver enzymes, and low platelet count syndrome. HCA was defined and classified using the grading by Salafia et al.10 Oligohydramnios was defined as amniotic fluid index < 5. Congenital anomaly was defined as a major anomaly registered in the KNN established according to the international classification of diseases (ICD) of the European Surveillance of Congenital Anomalies (EUROCAT) definitions. SGA was defined as a birth weight less than the 10th percentile for the GA and sex according to the Fenton growth curve. Surfactant use was defined as the administration of any surfactant, regardless of the purpose. PDA with treatment was defined as any pharmacologic or surgical treatment for preterm PDA. Hypotension was defined as any hypotension needed medications, including inotropics, hydrocortisone, and vasopressin. IVH was defined using Papile’s criteria8. NEC was defined as modified Bells’ stage 2 or greater9. Sepsis was defined as a culture-proven sepsis, and the patients should receive at least five days of antibiotics as a sepsis treatment. Meningitis was defined as a positive cerebrospinal fluid culture. Only cystic PVL was assessed in our analysis.
Pulmonary hypertension requiring treatment was defined as any inhaled nitric oxide, sildenafil, iloprost, bosentan, remodulin or milrinone treatment for pulmonary hypertension. Abnormal CT/MRI neuroimaging findings included PVL, ventriculomegaly, hydrocephalus or others. CP was defined as permanent, not unchanging disorders of movement and/or posture and disorders of motor function caused by a non-progressive interference, lesion, or abnormality of the immature brain. Deafness was defined as hearing loss requiring hearing aids.
According to previous studies, mental NDI was defined as a mental developmental index < 70 on the BSID II test, a cognitive score < 85, or a language score < 85 on the BSID III test. Motor NDI was defined as a psychomotor developmental index < 70 on the BSID II test or a motor score < 85 on the BSID III test11.
Major NDI after neonatal seizure in a NICU was defined as any CP, blindness, deafness, and mental or motor NDIs based on BSID at the 18–24 months follow-up.
Ethics statement
The KNN registry was approved by the institutional review board (IRB) of each participating hospital, including the Seoul National University-Seoul Metropolitan Government (SNU-SMG) Boramae Medical Center (IRB number 26-2014-12). Informed consent was obtained from the parents and/or legal guardians of all the study participants at enrollment. All data collection was performed prospectively according to the guidelines and regulations of the KNN. All the researchers in each hospital put their data on digitally recorded online case record forms, and such data are monitored regularly by the KNN data management committee. The present study was approved by the KNN data management committee, and IRB approval was exempted by the SNU-SMG Boramae Medical Center (IRB number 07-2024-13). All research was performed in accordance with relevant guidelines/regulations, and also in accordance with the Declaration of Helsinki.
Results
Risk factor analysis of neonatal seizures in NICU
Among the 16,279 VLBWIs analyzed, 1,430 infants (8.8%) had neonatal seizures. In the whole group and subgroup analyses according to the GA at birth, the GA and birth weight were significantly lower in infants with neonatal seizures. There were no significant differences in maternal age and maternal education more than college graduation between the two groups. The proportion of male gender was significantly higher in infants with neonatal seizures among the whole group and GA < 28 weeks group. The proportion of congenital anomalies was higher in infants with neonatal seizures in the whole group and GA 28–36 weeks group but not in the GA < 28 weeks group. The proportion of PIH was lower, and the proportion of HCA was higher in infants with neonatal seizures among the whole group and infants with GA < 28 weeks. The 5-minute Apgar score was lower, and the proportion of delivery room resuscitation was higher in infants with neonatal seizures in all groups. The proportion of inborns was lower in infants with neonatal seizures. The proportion of SGA was lower in infants with neonatal seizures among the whole group; however, higher in infants with neonatal seizures among the GA < 28 weeks group (Table 1). The proportion of all neonatal in-hospital morbidities, as listed in Table 1 and Supplementary Table 1, was higher in infants with neonatal seizures in the whole, GA < 28 weeks, and 28–36 weeks groups, except for PDA with treatment, postnatal steroid use, and days of total respiratory care in the GA < 28 weeks group.
When we performed the multivariable logistic regression analysis on risk factors of neonatal seizures in NICU admission, lower GA at birth, male gender, congenital anomaly, SGA, delivery room resuscitation, maternal HCA, surfactant use, hypotension within 1-week-old, IVH ≥ grade 3, post-hemorrhagic hydrocephalus, sepsis within 14 days after birth, meningitis, and NEC ≥ stage 2 increased the risk of neonatal seizure. However, any antenatal steroid use, inborn delivery, and PDA requiring treatment decreased the risk of neonatal seizures (Table 2).
In the GA < 28 weeks group, lower GA at birth, male gender, and SGA were significant risk factors for neonatal seizures. However, congenital anomalies and delivery room resuscitation did not increase the risk of neonatal seizures. Neonatal morbidities such as hypotension within one-week-old, IVH ≥ grade 3, post-hemorrhagic hydrocephalus, meningitis and NEC ≥ stage 2 were also significant risk factors of neonatal seizures in the GA < 28 weeks group (Table 2).
In the GA 28–36 weeks group, GA at birth and male gender did not significantly increase the risk of neonatal seizures; however, congenital anomaly and delivery room resuscitation increased the risk of neonatal seizures. Surfactant use, hypotension within 1-week-old, IVH ≥ grade 3, post-hemorrhagic hydrocephalus, sepsis within 14 days after birth, meningitis, and NEC ≥ stage 2 significantly increased the risk of neonatal seizures (Table 2).
When we searched for the association between the types of congenital anomalies and the development of neonatal seizures, the incidence of central nervous system (CNS) anomalies, cardiac anomalies, and gastrointestinal anomalies was significantly higher in infants with neonatal seizures (Supplemental Table 2).
Neurodevelopmental outcomes at 18–24 months follow-up in infants according to the presence of neonatal seizure
The total follow-up rate of VLBWIs at 18–24 months of corrected age registered in the KNN between 2013 and 2018 was 76.9% (7,599/9,880), and there was no significant difference in the follow-up rate between the two groups (76.8% in no seizure group and 79.3% in neonatal seizure group). However, the rates of visits to the initially registered KNN hospital were 83.6% in the no seizure group and 79.3% in the seizure group, slightly lower in the seizure group. Infants with neonatal seizures in the NICU had a lower GA at birth and lower birth weight, and male gender was predominant at the 18–24 months follow-up. When we compared the growth parameters at the 18–24 months follow-up, the proportions of weight, height, and head circumference < 10 percentile were higher in the neonatal seizure group when compared with the no seizure group. The frequency of any medical treatment after NICU discharge such as oxygen treatment, ventilator care, tracheostomy, nasogastric tube feeding, ventriculo-peritoneal shunt, rehabilitation and language treatment, pulmonary hypertension treatment, and anti-epileptic medications was higher in the neonatal seizure group. The readmission rate was also higher in infants with neonatal seizures. Regarding the CT/ MRI check after NICU discharge, the performance rate was higher in the neonatal seizure group when compared with the no-seizure group. Abnormal results of brain CT/MRI were also higher in infants with neonatal seizures (Table 3).
In the univariable analyses, the rate of post-discharge mortality, CP, hearing loss requiring hearing aids, and blindness was higher in infants with neonatal seizures. Mental and motor BSID scores were lower in infants with neonatal seizures, and the proportion of the K-DST scores as follow-up needed or further evaluation needed was higher in infants with neonatal seizures (Table 4).
When we performed multivariable logistic regression analysis with the backward elimination method on the influence of neonatal seizures on long-term neurodevelopmental outcomes at 18–24 months follow-up adjusting for maternal age, maternal education ≥ college, gestational diabetes, PIH, HCA, oligohydramnios, antenatal steroid use, cesarean section, GA at birth, male gender, congenital anomalies, multiple births, IVF, SGA, delivery room resuscitation, surfactant use, PDA treatment, hypotension, NEC ≥ stage 2, sepsis, IVH ≥ grade 3, cystic PVL, moderate to severe BPD, postnatal steroid use, ROP with treatment, and days to full feeding, seizure increased the risk of hearing loss requiring hearing aids [adjusted odds ratio (aOR) 4.28, 95% confidence interval (CI) 1.80-10.14], CP (aOR 4.38, 95% CI 2.78–6.90), and mental and motor NDIs based on BSID scores (aOR 2.41, 95% CI 1.57–3.71 for mental developmental impairments; aOR 3.30, 95% CI 2.10–5.18 for motor developmental impairments). However, neonatal seizure did not increase the risk of post-discharge mortality (aOR 1.95, 95% CI 0.82–4.63) and blindness (aOR 1.47, 95% CI 0.56–3.78, Supplemental Table 3).
When we compared the clinical factors of any major NDIs in infants suffering from neonatal seizures during NICU admission, there were no significant differences in maternal age, maternal education ≥ college, GA, birth weight, the proportion of male gender and congenital anomaly. However, sepsis, NEC ≥ stage 2, IVH ≥ grade 3, and moderate to severe BPD increased the risk of any major NDIs in the univariate analysis among infants with neonatal seizures. Multiple gestation and IVF decreased the risk of any major NDIs in infants with neonatal seizures. After the multivariable logistic regression analysis with the backward elimination method was done adjusting for GA, male gender, multiple gestation, IVF, sepsis, NEC ≥ stage 2, IVH ≥ grade 3, and moderate to severe BPD, NEC ≥ stage 2 and IVH ≥ grade 3 significantly increased the risk of any major NDIs in infants with neonatal seizures (Table 5).
Discussion
When we assessed the risk factors of neonatal seizures, lower GA at birth, male gender, congenital anomaly, SGA, delivery room resuscitation, maternal HCA, surfactant use, hypotension within one-week old, IVH ≥ grade 3, post-hemorrhagic hydrocephalus, sepsis within 14 days after birth, meningitis, and NEC ≥ stage 2 increased the risk of neonatal seizure. In subgroup analysis, demographic factors such as lower GA at birth, male gender, and SGA were significant risk factors for neonatal seizures only in the lower GA group. However, congenital anomalies and delivery room resuscitation were significant risk factors for neonatal seizures only in the higher GA group. When we assessed the influence of neonatal seizures on long-term neurodevelopmental outcomes at the 18–24 months follow-up, neonatal seizures increased the risk of hearing loss requiring hearing aids, CP, and mental and motor NDI based on BSID scores. When we assessed the risk factors of any major NDI after neonatal seizures, NEC ≥ stage 2 and IVH ≥ grade 3 significantly increased the risk of any major NDI.
There have not been many reports of neonatal seizures in preterm infants or VLBWIs. An epidemiologic study of only EEG-confirmed neonatal seizures confirmed a 5.0/1,000 incidence for neonates with a GA of 31–36 weeks, 54.9/1,000 for those with a GA of 28–30 weeks, and 85.6/1,000 for neonates with a GA < 28 weeks, indicating an increasing incidence of neonatal seizures with decreasing GA12. When amplitude electroencephalogram (EEG) monitoring was done for infants under 28 weeks of gestation, 20% had at least one seizure episode, and the presence of seizures in the first three days of life tended to predict an increased risk of mortality significantly13. In our study, 8.8% of VLBWIs suffered from neonatal seizures during their NICU admissions.
The etiology of neonatal seizures differs between term and preterm infants. In term infants, hypoxic ischemic encephalopathy (HIE), stroke, cerebral malformation, and metabolic disorders are the leading causes of neonatal seizures. In preterm infants, IVH, PVL, and infection are the major risk factors for neonatal seizures. However, many other severe morbidities associated with preterm infants also can be a significant risk factor for neonatal seizures in VLBWIs, which have not been thoroughly evaluated. In extremely preterm infants with a GA < 28 weeks, the incidence of preterm morbidities, including IVH and infection, is significantly higher compared to preterm infants with a GA of 28–36 weeks. Due to the significantly higher incidence of neonatal seizures, the increased prevalence of preterm morbidities associated with the development of neonatal seizures in extremely preterm infants, and the different etiologies of neonatal seizures according to the GA, we performed a subgroup analysis to assess whether there are any differences in the risk factors for neonatal seizures between extremely preterm infants with a GA < 28 weeks and those with a GA of 28–36 weeks. When we searched for the different risk factors of neonatal seizures according to the GA, GA and male gender increased the risk of neonatal seizures only in the lower GA group, not in the higher GA group. However, congenital anomalies and delivery room resuscitation did not increase the risk of neonatal seizures in infants with lower GA. However, they were meaningful factors in infants with higher GA. Neonatal morbidities such as hypotension, severe IVH, post-hemorrhagic hydrocephalus, meningitis, and NEC were significant risk factors for neonatal seizures regardless of GA. Therefore, especially in infants with higher GA, improving neonatal care to reduce such preterm morbidities associated with the development of neonatal seizures will be very important in the prevention of neonatal seizures.
Since we used the KNN data only registered VLBWIs, we could not get sufficient information on neonatal seizures in older preterm infants. We searched for other studies comparing the characteristics of neonatal seizures and the outcomes of neonatal seizures between VLBWIs and the older preterm infants. Glass et al.14, using the neonatal seizure registry database in seven pediatric centers in the U.S., searched for the different characteristics of neonatal seizures according to the GA subgroups (GA < 28 weeks, 28–32 weeks, and 32–37 weeks) in preterm infants and full-term infants. HIE was the most common etiology of neonatal seizures among moderate to late preterm and term infants. In contrast, intracranial hemorrhage was more common among extremely and very preterm infants, which was compatible with our results. The presence of subclinical seizures, monotherapy treatment failure, and distribution of seizure burden including status epilepticus was similar in preterm and term neonates. Mortality was similar among the three preterm GA groups; however, preterm mortality was more than twice that of term infants. Schussler SC et al.15 also investigated the long-term outcomes of VLBW and LBW (birthweight 1,500-2,499 g) preterm infants with neonatal seizures. The postnatal age at seizure onset was earlier in LBW infants when compared with VLBWIs (13 ± 11 days in VLBWIs and 9 ± 8 days in LBW infants). Higher-grade intracranial hemorrhage was the predominant etiology in VLBWIs, while the etiology in the LBW infants was more heterogeneous, including CNS malformations and genetic syndromes. At the mean age of 6.2 ± 2.0 years, 44.4% of VLBWIs and 71.4% of LBW infants showed intellectual impairment, and 22% of VLBWIs and 42.9% of LBW infants had infantile CP.
Regarding the association between antenatal maternal characteristics and the development of neonatal seizures, in one study in late preterm infants, chorioamnionitis and cesarean section were reported to be significant risk factors for neonatal seizures16. In our study, chorioamnionitis was a significant risk factor for neonatal seizures in the whole infant group and also in the GA 28–36 weeks group. However, cesarean section was not a significant risk factor. PDA, especially PDA ligation, was a significant risk factor for neonatal seizures in another study5. However, PDA was a protective factor in our study.
When we searched for the associated type of congenital anomalies, not only CNS anomalies but also serious anomalies such as cardiac and gastrointestinal anomalies were significant anomalies in the development of neonatal seizures. In infants with cardiac and gastrointestinal anomalies, surgeries and hemodynamic instabilities, including hypotension and hypoxic events, are more frequent, which could impact the development of neonatal seizures.
Several reports using a nationwide network database support the influence of neonatal seizure on significant long-term NDIs in both term and preterm infants. According to a nationwide claim database, seizures increased the risk of epilepsy, intellectual disability, and headache after controlling for sex, birth weight, preterm birth status, and underlying etiologies6. In a report from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network (NICHD-NRN) on the influence of seizures in extremely low birth weight infants, clinical seizures remained significantly associated with later death or NDIs at 18–24 months corrected age7. A population-based cohort study using the Canadian Neonatal Network database showed that neonatal seizures were independently associated with higher odds of death or significant NDIs at 18 to 24 months of age in infants with a GA < 29 weeks1. However, there were no reports comparing clinical factors associated with any major NDI in infants suffering from neonatal seizures in NICU admissions. In our study, among infants with neonatal seizures, any major NDI were not dependent on demographic characteristics such as GA at birth, birth weight, and male gender but on the in-hospital morbidities in NICUs. This result implies that neonatal care after birth in VLBWIs is important to prevent any major NDI in infants with neonatal seizures.
Although infants with a medically complex history generally have more hospital visits, the rate of in-person visits to the registered KNN hospital was higher in infants without neonatal seizures in our study. In Korea, a nationwide long-term follow-up project encouraged outpatient in-person visits for VLBWIs at 18–24 months corrected age by telephone prior to hospital visits. The in-person follow-up visits to the registered hospital increased particularly for infants with fewer medical problems. Additionally, due to the unavailability of certain treatments, such as rehabilitation and anti-epileptic therapy by pediatric neurologists, more infants with neonatal seizures visited follow-up clinics at other hospitals. When we assessed the reasons for the lack of in-person follow-up visits to the registered hospital in the two groups, we found that the proportion of post-discharge mortality was significantly higher in infants with neonatal seizures, and more infants with neonatal seizures attended a follow-up visit at another hospital. When considering both in-person visits to the registered KNN hospital and other hospitals, we found that 85.1% (6,106/7,174) of infants without neonatal seizures and 85.4% (363/425) of those with neonatal seizures attended their 18–24 month in-person follow-up at clinics. This suggests a slightly higher number of in-person visits among infants with neonatal seizures. We considered that all of these factors collectively contribute to the lower frequency of in-person follow-up visits to the registered hospital among infants with neonatal seizures.
NEC was a significant risk factor for any major NDIs in infants with neonatal seizures. Several reports confirm the association between NEC and long-term NDI17,18. Malnutrition, impaired perfusion and oxygen supply, and inflammation by cytotoxic mediators combined with NEC in critical periods of brain development primarily affect the neurodevelopment in preterm infants. Those insults can result in white matter injury, cognitive impairment, and motor impairment17. Especially, surgical NEC was significantly associated with significant growth delay and adverse neurodevelopmental outcomes at 18–24 months follow-up when compared with medial NEC or unaffected19,20. In studies on the association between systemic inflammation caused by NEC and neurodevelopment, systemic inflammation induced by NEC was associated with NDI in preterm infants, and the seizure spike on the amplitude EEG during inflammatory episodes was associated with poor neurodevelopment21.
IVH is a well-known risk factor for neonatal seizures and long-term NDI. Meta-analysis supports that severe IVH, compared with mild IVH, increased the susceptibilities of children to NDI, motor delay, CP, and hearing and visual impairments. Moreover, mild IVH was not associated with seizures or epilepsy22. In VLBWIs, refractory hypotension within a week of life and seizures were associated with severe IVH and developmental delay at 18–24 months corrected age23. In our study, severe IVH was a significant risk factor for the development of neonatal seizures, and it increased the risk of post-discharge mortality, blindness, CP, and mental and motor developmental impairment based on BSID tests. It also increased the risk of any major NDIs in infants with neonatal seizures.
In this study, we reported a difference in long-term neurodevelopmental outcomes of neonatal seizures based on the K-DST and the Bayley test at 18–24 months follow-up. The K-DST is a developmental screening test developed through the National Health Screening Program for infants and children in 201424. The K-DST examine early childhood development in the gross motor, fine motor, cognition, language, social skills, and self-help domains. For each of the 6 domains, a set of 8 questions was asked, and the total score was calculated based on the answers. Based on the total score in each domain, the K-DST results were grouped into 4 categories: good, continued care, requires follow-up examination, and requires further detailed assessment. In the study comparing the efficacy of the K-DST and the Bayley-II test in VLBWIs using the KNN database, the frequency of a failed psychomotor developmental index < 85 was similar to that in at least one domain of the K-DST < 1 standard deviation, revealing a useful screening tool for predicting mental developmental delay in VLBWIs25.
There are some limitations in our analyses. First, we only included infants with neonatal seizures, which required anti-epileptic medications, without confirming the EEG findings. Recently, the International League Against Epilepsy (ILAE) presented a new classification and framework for seizures in the neonatal period, emphasizing the key role of EEG in diagnosing seizures in neonates26. Most neonatologists in Korea give anti-epileptic medications when clinical seizures are confirmed by EEG or any electrographic seizure detected by EEG, including amplitude EEGs. That is why the KNN defined neonatal seizures based on the anti-epileptic medication treatment. However, there can still be a small chance of missing only electrographic seizures. When we compare the definition of neonatal seizures to other nationwide neonatal networks, we see that neonatal seizures are defined as clinical or EEG-confirmed seizures on the California Perinatal Quality Care Collaborative network. In contrast, it is defined as only EEG-confirmed seizures on the NICHD-NRN network.
Second, we could not know the exact time order of neonatal morbidities and neonatal seizures, and that is why we only included sepsis within 14 days after birth and hypotension within one week in the risk factor analysis of neonatal seizures. According to the previous studies on the onset of neonatal seizures in preterm infants, mean seizure onset was 10.8 days after birth (range 1-120 days), and when divided according to GA, mean seizure onset was 14.6 days after birth (range 1-120 days) in preterm infants with a GA ≤ 29 weeks and 5.2 days after birth (range 1–37 days) in infants with a GA > 29 weeks27.
In our study, a lower GA and male gender were significant risk factors for neonatal seizures in infants with lower GA at birth. However, congenital anomalies and delivery room resuscitation significantly increased the risk of neonatal seizures in higher GA infants. In the whole group, serious neonatal morbidities such as severe IVH, NEC, post-hemorrhagic hydrocephalus, sepsis within 14 days after birth, meningitis, and hypotension within one week were significant risk factors for neonatal seizures. Neonatal seizures increased the risk of CP, deafness, and the mental and motor NDIs based on the Bayley tests. In VLBWIs with neonatal seizures, severe NEC and IVH were significant risk factors for any major NDI at 18–24 months corrected age. Thus, it is important to perform the best treatment to reduce serious neonatal morbidities of preterm infants, such as severe NEC and IVH in NICUs, which will prevent neonatal seizures and also help with no major NDI even after neonatal seizures.
Data availability
The dataset analyzed in this study is not publicly available due to the Korea Center for Disease Control and Prevention research policy. However, the dataset is available from the corresponding author upon reasonable request.
References
Iwami, H. et al. Outcomes after neonatal seizures in infants less than 29 weeks’ Gestation: A Population-based Cohort Study. Am. J. Perinatol. 36, 191–199. https://doi.org/10.1055/s-0038-1667107 (2019).
Vasudevan, C. & Levene, M. Epidemiology and aetiology of neonatal seizures. Semin. Fetal Neonatal. Med. 18, 185–191. https://doi.org/10.1016/j.siny.2013.05.008 (2013).
Glass, H. C. et al. Antenatal and intrapartum risk factors for seizures in term newborns: A population-based study, California 1998–2002. J. Pediatr. 154, 24–28e21. https://doi.org/10.1016/j.jpeds.2008.07.008 (2009).
Kohelet, D., Shochat, R., Lusky, A. & Reichman, B. Risk factors for seizures in very low birthweight infants with periventricular leukomalacia. J. Child. Neurol. 21, 965–970. https://doi.org/10.1177/08830738060210111301 (2006).
Kohelet, D., Shochat, R., Lusky, A. & Reichman, B. Risk factors for neonatal seizures in very low birthweight infants: Population-based survey. J. Child. Neurol. 19, 123–128. https://doi.org/10.1177/08830738040190020701 (2004).
Oh, A., Thurman, D. J. & Kim, H. Independent role of neonatal seizures in subsequent neurological outcomes: A population-based study. Dev. Med. Child. Neurol. 61, 661–666. https://doi.org/10.1111/dmcn.14174 (2019).
Davis, A. S. et al. Seizures in extremely low birth weight infants are associated with adverse outcome. J. Pediatr. 157, 720–725.e721-722 https://doi.org/10.1016/j.jpeds.2010.04.065 (2010).
Papile, L. A., Burstein, J., Burstein, R. & Koffler, H. Incidence and evolution of subependymal and intraventricular hemorrhage: A study of infants with birth weights less than 1,500 gm. J. Pediatr. 92, 529–534. https://doi.org/10.1016/s0022-3476(78)80282-0 (1978).
Bell, M. J. et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann. Surg. 187, 1–7. https://doi.org/10.1097/00000658-197801000-00001 (1978).
Salafia, C. M., Weigl, C. & Silberman, L. The prevalence and distribution of acute placental inflammation in uncomplicated term pregnancies. Obstet. Gynecol. 73, 383–389 (1989).
Johnson, S., Moore, T. & Marlow, N. Using the Bayley-III to assess neurodevelopmental delay: which cut-off should be used? Pediatr. Res. 75, 670–674. https://doi.org/10.1038/pr.2014.10 (2014).
Pisani, F. et al. Incidence of neonatal seizures, perinatal risk factors for epilepsy and mortality after neonatal seizures in the province of Parma, Italy. Epilepsia 59, 1764–1773. https://doi.org/10.1111/epi.14537 (2018).
Meledin, I. et al. Seizures in premature infants born at less than 28 weeks’ Gestation. Neonatology 115, 247–255. https://doi.org/10.1159/000494626 (2019).
Glass, H. C. et al. Seizures in Preterm neonates: Aa Multicenter Observational Cohort Study. Pediatr. Neurol. 72, 19–24. https://doi.org/10.1016/j.pediatrneurol.2017.04.016 (2017).
Schüssler, S. C. et al. Long-term outcomes of very-low-birth-weight and low-birth-weight preterm newborns with neonatal seizures: A single-center perspective. Eur. J. Paediatr. Neurol. 36, 137–142. https://doi.org/10.1016/j.ejpn.2021.12.013 (2022).
McLaren, R. Jr., Clark, M., Narayanamoorthy, S. & Rastogi, S. Antenatal factors for neonatal seizures among late preterm births*. J. Matern Fetal Neonatal Med. 35, 9544–9548. https://doi.org/10.1080/14767058.2022.2047924 (2022).
Hickey, M., Georgieff, M. & Ramel, S. Neurodevelopmental outcomes following necrotizing enterocolitis. Semin. Fetal Neonatal Med. 23, 426–432. https://doi.org/10.1016/j.siny.2018.08.005 (2018).
Adams-Chapman, I. Necrotizing enterocolitis and Neurodevelopmental Outcome. Clin. Perinatol. 45, 453–466. https://doi.org/10.1016/j.clp.2018.05.014 (2018).
Hintz, S. R. et al. Neurodevelopmental and growth outcomes of extremely low birth weight infants after necrotizing enterocolitis. Pediatrics 115, 696–703. https://doi.org/10.1542/peds.2004-0569 (2005).
Fullerton, B. S. et al. Severe neurodevelopmental disability and healthcare needs among survivors of medical and surgical necrotizing enterocolitis: A prospective cohort study. J. Pediatr. Surg. https://doi.org/10.1016/j.jpedsurg.2017.10.029 (2017).
Lee, E. S. et al. Factors associated with neurodevelopment in preterm infants with systematic inflammation. BMC Pediatr. 21, 114. https://doi.org/10.1186/s12887-021-02583-6 (2021).
Zhou, M., Wang, S., Zhang, T., Duan, S. & Wang, H. Neurodevelopmental outcomes in preterm or low birth weight infants with germinal matrix-intraventricular hemorrhage: A meta-analysis. Pediatr. Res. 95, 625–633. https://doi.org/10.1038/s41390-023-02877-8 (2024).
Hwang-Bo, S., Seo, Y. M., Oh, M. Y., Im, S. A. & Youn, Y. A. The prognosis of refractory hypotension and severe intraventricular hemorrhage in very low birth weight infants. Med. (Baltim). 101, e29598. https://doi.org/10.1097/md.0000000000029598 (2022).
Kim, D. et al. Korean developmental screening test for infants and children (K-DST): development, applications, and implications for future early childhood development interventions. Clin. Exp. Pediatr. 66, 288–293. https://doi.org/10.3345/cep.2022.00906 (2023).
Kim, C. Y., Jung, E., Lee, B. S., Kim, K. S. & Kim, E. A. Validity of the Korean developmental screening test for very-low-birth-weight infants. Korean J. Pediatr. 62, 187–192. https://doi.org/10.3345/kjp.2018.07381 (2019).
Pressler, R. M. et al. The ILAE classification of seizures and the epilepsies: Modification for seizures in the neonate. Position paper by the ILAE Task Force on neonatal seizures. Epilepsia 62, 615–628. https://doi.org/10.1111/epi.16815 (2021).
Pisani, F. et al. Neonatal seizures in preterm newborns: A predictive model for outcome. Eur. J. Paediatr. Neurol. 20, 243–251. https://doi.org/10.1016/j.ejpn.2015.12.007 (2016).
Acknowledgements
This research was supported by the “Korea National Institute of Health” (KNIH) research project (2022-ER0603-02#).
Author information
Authors and Affiliations
Contributions
J.A.L. conceived and designed the work, acquired the data, and analyzed it. S.O. performed statistical analysis and analyzed the data. J.A.L. wrote the manuscript, and J.A.S. and J.A.L. added the figures and tables. J.A.S. and S.O. critically reviewed the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, J.A., Sohn, J.A. & Oh, S. Risk factors and influence on neurodevelopmental outcomes of neonatal seizures in very low birth weight infants based on nationwide cohort. Sci Rep 15, 10875 (2025). https://doi.org/10.1038/s41598-025-86224-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86224-3