Abstract
Environmental variation has long been considered a key driver of evolutionary change, predicted to shape different strategies, such as genetic specialization, plasticity, or bet-hedging to maintain fitness. However, little evidence is available with regards to how the periodicity of stressors may impact fitness across generations. To address this gap, I conducted a reciprocal split-brood experiment using the freshwater crustacean, Daphnia magna, and an ecologically relevant environmental stressor, ultraviolet radiation (UVR). I exposed one group to constant and another group to fluctuating UVR conditions. Despite receiving the same dose of UVR, the first experimental generation displayed significant treatment-by-genotype interactions with respect to survival and reproductive output, as well as a delayed reproductive maturity under fluctuating UVR conditions. In the following experimental generation individuals exposed to fluctuating UVR exhibited higher fitness than those in a constant UVR regime. The ancestral conditions, i.e., maternal environment, however affected the survival probability and reproductive output, but did not significantly influence the maturation date. Overall, I demonstrate that the delivery of a stressor, not just its intensity, can have profound fitness consequences across generations, with important implications for seasonal succession of genotype–phenotype patterns in natural environments.
Similar content being viewed by others
Introduction
An organism’s phenotype reflects the developmental interplay between past and current environments1. Selection embeds information of past environments within the genotype2, and phenotypic plasticity allows for the assimilation of current environmental cues into locally adapted phenotypes3. Moreover, non-genetic inheritance mechanisms, such as maternal effects, have repeatedly been shown to contribute to the individual’s phenotype4,5. Despite the clear beneficial potential of integrating information across recent ancestry, from environmentally induced sources6,7, the adaptive nature of transgenerational plasticity (TGP) is not always evident8,9,10,11. In part this can be due to environmental unpredictability violating one of the fundamental assumptions of plasticity which is that organisms have the ability to accurately detect and forecast the effects of changing environmental conditions12.
This unpredictability may arise from the failure to detect stressors, unreliable cues, or stressors that vary at scales that preclude the advantages conferred through plasticity13. In response to perceived unpredictability, an alternative strategy that may evolve is bet-hedging. This is essentially the reduction in mean fitness at the individual level that then decreases the variation in fitness across generations14. In other words, so long as some individuals are able to reproduce in a given generation, then unpredictable fluctuations are unlikely to eliminate a genotype from the population. Although bet-hedging has been shown to be able to evolve through many models14,15, the ability to produce unpredictable experimental regimes have limited the number of empirical examples16,17,18.
Given that environmental variations then clearly have the potential to alter the evolutionary strategy utilised19, many studies have examined the effects of stressor introduction during specific developmental stages on fitness within and between generations9,20,21. However, these experiments have typically focused on the timing of stressor introduction rather than the temporal variability of stressors, particularly of those with a more ephemeral nature. Thus, our understanding of the responses of organisms to highly fluctuating environments remains limited22. Research has explored the impacts of temperature variability23,24, with some papers suggesting prior information of unpredictability ‘primed’ the organisms’ physiology to be better able to cope with extremes in temperature25. However, due to the complexity of the underlying physiological mechanisms that modulate the expressed phenotype, clear directional hypotheses of the effects of variability are often more challenging, as made clear by a recent meta-analysis which found longevity to decrease under fluctuating temperatures26. Even less knowledge is available about the effects of variability in other biotic and abiotic stressors. This is of increasing importance in light of the predicted increase in environmental variability due to climate change27, which directly effects a variety of abiotic factors such as temperature, precipitation and solar ultraviolet radiation (UVR).
In aquatic systems, UVR is a common and highly variable stressor for many organisms. It varies over seasonal scales, diel scales and even at shorter time scales, such as rapid changes in cloud cover (Supplementary Fig. S1). Zooplankton may be the best studied group of aquatic organisms in relation to the effects of UVR, with common negative consequences including reductions in longevity28, lower reproductive output29, reduced growth rates30 and even increased respiration rates31. In response to the direct negative consequences of UVR, zooplankton have a suite of phenotypic responses including the upregulation of photoprotective compounds, such as melanin or carotenoids32,33, alternative antioxidant stress responses34 or behavioural avoidance strategies such as diel vertical migration35,36. The relative expression of responses to a threat, such as UVR, will then inevitably depend on the current ecological trade-offs (such as predation risk and foraging opportunities)37,38 coupled with the information about past environments.
One of the most extensively studied zooplankton species is Daphnia magna. They represent a key species in shallow lakes and ponds across the northern hemisphere. Furthermore, they have become an invaluable model for all manner of evolutionary and ecological research questions due to their amenability to lab-based studies, in part because of their cyclical parthenogenetic mode of reproduction39. In particular, they are a powerful model for investigating transgenerational responses to stressors4,9,40,41, including towards UVR10. Despite the replete literature on threat response in the genus Daphnia, and the long history of them being studied in relation to UVR both within and across generations, very few studies consider the variable nature of the stressor. Those that do are limited to short-term exposure, or within-generation studies42,43. Furthermore, Daphnia often exhibit diverging responses due to the variation between genotypic lines43,44. As a result, we may make predictions regarding population viability that over- or underestimate a populations´ capacity to adapt. Therefore, it is crucial to understand empirically how organisms respond to either consistency or fluctuations in this environmental variable both within and across generations in multiple genetic lines.
In order to address this gap in the available knowledge, I conducted a reciprocal split-brood experiment (Fig. 1) to investigate the responses of D. magna to constant and fluctuating UVR regimes for multiple generations. Theory predicts within-generation predictable variation should select for plastic responses to maintain fitness under stress. Building upon previous research, which demonstrated behavioural shifts in activity due to UVR fluctuations have negative reproductive consequences within a generation43, I hypothesised that constant exposure would yield fitness advantages within a generation. Specifically, I predicted earlier reproductive maturity and higher lifetime reproductive output, despite the trade-off with a shortened lifespan. Furthermore, given environmental consistency across generations i.e., maternal matching, is predicted to confer advantages41,45, I hypothesised that offspring experiencing the same environment as their mother would gain a fitness advantage in comparison to those that are mismatched. In this generation, I predicted the fitness advantage would manifest as greater survival in matched maternal environments.
Schematic of the experimental split brood design imposed on each of the three genotypes. Generation is denoted by the letter G and the number in the brackets denotes the maternal clutch from which the focal generation were obtained. Each individual from generation G3 and G4 was followed for the entirety of their lifetimes, which is denoted by the horizontal lines. Each vertical intersecting line represents a clutch, of which only four were followed from G3. All other clutches were removed and measured.
Methods
To ensure genotypic variation, Daphnia magna were collected from three source populations located in Southern Sweden. These localities were Lake Fnifahosjön (N 55.73907, E 13.20968; genotype ‘N’), Lake Bysjön (N 55.675393, E 13.545363; genotype ‘D’) and a Sydvatten pond (N 55.661140, E 13.541958; genotype ‘P’). The genotypes were cultured in the laboratory for over 50 generations at 18 °C, a 16:8 light:dark photoperiod, fed with a green alga live culture (Tetradesmus obliquus; formerly Scenedesmus obliquus46) and maintained at a density of ca. 100 individuals per litre. To establish the experiment (G0), one individual female per genotype was isolated and placed into separate 100 ml jars filled with 80 ml of Artificial Daphnia Medium (ADaM)47 and 240,000 cells ml−1 of T. obliquus. These jars were then placed under the same conditions with the addition of ultraviolet lamps (Sylvania F36W/GRO; 70 ± 10 μW cm−2) at ecologically relevant conditions of UVR (Supplementary Figs. S2–S3). These lamps were turned on for six hours each day between 10:00 and 16:00 and constituted a constant UVR exposure treatment.
This common-garden condition was maintained for two subsequent generations to remove potential maternal effects of moving to an increased UVR environment. It is important to highlight here, as copious research has investigated the stress response of UVR31,33,38,48,49,50,51, I have no treatment without exposure to UVR. My focus is the variation of the stress, therefore all differences detected will represent changes in the consistency of the stressor, and not dosage differences. From the moment that G0 individuals were isolated, every individual was transferred to new media and fresh food every other day. Furthermore, to ensure food availability and reduce any competition between either the mother and offspring, or between siblings, every individual was isolated and transferred to new media on the day that a brood was released, even if the mother had been transferred the day prior.
The experiment began when the G2 mothers had produced ten clonal offspring from their third brood. This brood was then divided into two treatments ‘Constant’ (as described above) and the ‘Fluctuating’, generating five replicates per treatment per genotype in generation G3. The fluctuating treatment also experienced six hours of UVR, however the schedule oscillated between on and off in 15-min intervals over a 12-h period during the photoperiod. To ensure that the oscillations themselves were unpredictable, I created multiple schedules each with six hours of UVR exposure, then alternated between the schedules randomly every 1–4 days (Supplementary Fig. S4). This scheduling prevented anticipatory modifications between broods and guaranteed that before maturity, each Daphnia in the fluctuating treatment was exposed to a variety of UVR schedules. Within both treatments the position of each individual was randomised to ensure that the location within the climate chamber did not influence the results.
To address how the variations of an environmental stressor may affect the life-history of Daphnia, I quantified longevity, reproductive maturity, as defined by the first day at which eggs were visible in the brood pouch52, and total reproductive output over each individual´s lifetime. Every individual was checked each day at the same time (± 0.5 h). In order to also take into account the offspring’s ‘fitness’, I repeated this design for a second generation using a split brood design. Specifically, I took the first, third, fifth and seventh broods forward to the next generation as to maximise the number of individuals in generation G4 while taking the other broods for size measurements of neonates (data not shown here). When the broods contained six or more offspring, each individual was randomly allocated to either the constant or fluctuating treatment group, generating four ‘Ancestry : Treatment’ combinations. If a brood had fewer than six individuals, then all individuals were removed from the analysis as to prevent unduly different sample sizes (see Table S1 for a breakdown of samples sizes per analysis).
Statistical analysis
All analyses were performed using R v.4.4.253. The two experimental generations (G3 and G4) were analysed independently. All models were subject to stepwise backward selection, removing any non-significant interaction terms. Post hoc tests were employed to investigate significant models, and p-values were adjusted using the Benjamini–Hochberg method for multiple comparisons, thereby decreasing the false discovery rate. Statistical figures were subsequently produced using the package ggplot2 v.3.5.154.
In order to assess how different categorical treatment groups affect lifespan, I utilised a Cox proportional hazard model using the coxme package55. As all individuals were followed for their lifetime, none were censored. Treatment, genotype and their interaction were used as the independent variables in the model for the G3 generation. The G4 generation was modelled using treatment, ancestral treatment, and their interaction as independent variables. When including genotype into the model, the assumptions of heteroscedasticity are violated. Therefore, I modelled each genotype separately. To account for the possibility that particular mothers invest differentially in offspring, the G4 generation was analysed using a mixed effects model with Mother ID as the random factor.
To examine the effects of treatment, genotype and their interaction on individual reproductive success (the total number of neonates produced) of generation G3 I utilised a two-way ANOVA. Due to the large proportion of zeros within generation G4, this generation was analysed through a zero-inflated generalised linear mixed model with a Poisson error distribution using the ‘glmmTMB’ package56. Furthermore, due to the underrepresentation of genotypes ‘N’ and ‘P’, genotype was not included in this model. Treatment, ancestral treatment, and their interaction were assigned as fixed factors and the mother ID as a random effect. The assumptions for this model were confirmed using the package ‘DHARMa’57.
Age of reproductive maturity, as a numerical count, was analysed using a linear model for generation G3 with treatment, genotype and their interaction as independent variables. In order to account for the mother ID as a random factor in generation G4, I analysed age of reproductive maturity with a linear mixed effects model, using the ‘lme4’ package58. This meant treatment, ancestral treatment, the interaction between current and ancestral treatment were modelled as fixed effects and mother ID as the random effect. Similar to the analyses above, genotype was not included in this model due to underrepresentation of reproductively mature individuals from 2/3 of the initial genotypes. To analyse this disparity in the frequency of reproductive and unreproductive individuals, i.e., the reproductive status, I utilised a Cochran-Mantel–Haenszel test, which is an extension of a Chi-Squared, to explicitly test if this is dependent on genotype and ‘Treatment x Ancestry’ combination.
Results
Daphnia magna exhibit distinct life history responses to variation in UVR stress. Notably, significant interactions were observed in generation G3 for both survival and reproductive output. However, these interactions did not align with predictions based on prior experiments, nor the maternal matching hypothesis. Instead, the results suggest evidence for carryover effects of UVR variation across generations. Below, I provide a detailed account of the results for survival, reproductive output, and reproductive maturity, organized by generation.
Generation G3
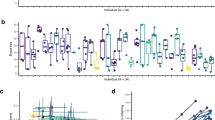
The interaction between genotype and treatment significantly impacted the survival probability (Table 1). Specifically, genotype P exhibited 3.5 times lower survival in a constant UVR regime, however when in a fluctuating environment they performed similar to genotype D at only 1.13 times higher risk of dying. Under constant stress genotype P were 3 times less likely to survive than genotype D but 12.6 times more likely than genotype N (Fig. 2; Table 2).
Survival curves of generation G3. Colours denote the genotype and the line type indicates the treatment group.
The two-way interaction between the treatment group and the genotype significantly affected the number of offspring produced within the G3 generation (Table 1; Fig. 3). More precisely, I found that genotype D was unaffected by the treatment differences, yet was significantly more productive than genotype P when under constant UVR but not under fluctuating UVR. When exposed to fluctuating UVR genotype D had significantly more offspring than genotype N (Table 3).
Model estimates (± 1 SE) for reproductive success (total number of neonates per female) for each treatment regime. Generation G3 displays the treatment group and genotype with the shape denoting the genotype and the colour representing the treatment factor. Generation G4 displays the effects of the current and ancestral treatments within genotype D.
Increased variability of the fluctuating UVR regime delayed the date of reproductive maturity in the G3 generation (Table 1; Fig. 4). Genotype also emerged as a significant determinant of reproductive maturity (Table 1; Supplementary Fig. S5). No interaction was found between the treatment and genotype. Averaging across all genotypes, individuals in the constant treatment achieved reproductive maturity at day 8.1 (± 0.56 SE), whereas those in the fluctuating treatment became reproductive at day 10.2 (± 0.56 SE). When averaging over treatment group, genotype D was the fastest to mature at 6.6 (± 0.69 SE) days and the genotype P was the slowest, taking nearly twice as long (12.6 ± 0.69 SE).
The average day at which individuals become reproductively mature in each treatment regime. The points display the mean ± 1 SE.
Generation G4
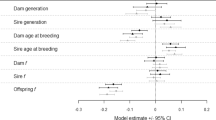
Survival in generation G4 was determined by the current treatment and ancestry independently, i.e., no interactions were detected (Table 1). There was an obvious disparity between genotypes in length of survival, which violated the models’ assumptions, therefore I modelled each genotype separately. Genotype D displayed that individuals under a currently fluctuating environment had a 54.07% higher survival rate irrespective of ancestry (Fig. 5; Table 1). Despite the lack of a significant interaction, ancestral treatment also increased the survival rate. Individuals that had a mother in a fluctuating environment had a 54.81% higher chance of survival (Fig. 5). There were no differences due to either current treatment or ancestry for both genotypes N and P (Fig. 5; Supplementary Table S2).
Survival curves of the G4 generation displaying the effects of genotype, current treatment and ancestral treatment in the respective panels from left to right. The centre and rightmost panels refer only to genotype D.
Treatment and Ancestry significantly interacted to determine the number of offspring produced in generation G4 (Table 1, Fig. 3). It is important to note here, that this only represents one genotype. In large part, this is due to no individuals of genotype N reproducing, and only two individuals of genotype P, both of which were in the fluctuating UVR treatment group. This means that genotype N and P were not significantly different from zero whereas genotype D averaged 67.9 (± 55.3 SE) offspring per individual. Within this genotype, those in the fluctuating UVR treatment group averaged 77.2 (± 62.9 SE) offspring per individual whereas those in a constant UVR environment averaged 59.7 (± 48.6 SE).
Current treatment regime significantly delayed the date of reproductive maturity by 3.8 days in the fluctuating regime as compared to the constant regime (Fig. 4). Neither ancestry, nor the interaction between ancestry and the current treatment group had an impact on the date of reproductive maturity (Table 1). As date of maturity was determined only in the one genotype, I also examined the frequency of reproductive and unreproductive individuals across all genotypes. There was a strong association between the number of individuals that became reproductive in the generation G4, ancestry, current treatment and genotype (M2 = 21.15, d.f. = 6, p = 0.002) which is evident in Fig. 6. In the fluctuating treatment 16.4% of individuals became reproductive, in comparison with only 6.8% in the constant treatment. This was similar in magnitude to the ancestral treatment with 15.2% of individuals with a mother in the fluctuating environment becoming reproductive as opposed to 7.8% of those with mothers in the constant treatment group. Genotype, however, shows the largest disparity with 21.2% of genotype D, 2.6% of genotype P and no genotype N becoming reproductive. When binned according to whether the mothers and daughters experienced the same- or different regimes, those experiencing the same environments (irrespective of which) showed higher numbers of reproductive individuals (Supplementary Fig. S6).
Frequency of generation G4 individuals that became reproductively mature by genotype and ‘Ancestral regime : Current regime’ combinations. ‘C’ represents ‘Constant’ and ‘F’ represents ‘Fluctuating’ regimes.
Discussion
All threats, including predation risk, starvation and UVR exposure may affect an individual organism in either a more consistent or fluctuating manner. Whereas the theoretical impact of stressor variation on an organism’s evolutionary strategy is well-established1,15,22, our empirical understanding of the consequences of such fluctuations in stressors, independent of the intensity, remains limited. In this study, I show that exposure to different temporally variable UVR regimes have significant fitness consequences in multiple life history traits. I hypothesised that constant exposure would have the greatest fitness advantage, due to the predictable nature of the stressor. Contrary to my initial predictions, based upon prior evidence43, these results suggest that fluctuating UVR regimes lead to higher fitness compared to those exposed to an equally intense but more constant UVR regime. Furthermore, these effects carryover to the following generation which does not support the predicted fitness advantages from the maternal matching hypothesis.
Previous studies have found many potential adverse effects of UVR28,33,50, and have even shown how the effects span multiple generations10. In generation G3, my results indicate that survival and reproduction are strongly influenced by the interaction between UVR variability and the clonal line, whereas maturity was simply affected by the treatment and genotype independently. This implies that certain genotypes could be evolutionarily predisposed to differences in UVR variability, but this remains untested. Despite this, each line used here originated in a geographically similar area but the physical, chemical and biological properties of these lakes are likely to have defined differences. In particular, the UVR extinction depth is a viable proxy for the threat posed by UVR. Of the clones used, two of the three originated from ponds with a measured UV profile (genotype D from lake Bysjön and genotype N from lake Fnifahosjön). Lake Bysjön in comparison to lake Fnifahosjön is more transparent, thereby allowing UVR to penetrate deeper and potentially damage zooplankton, yet Daphnia in lake Bysjön were still visible at the water surface during mid-day (personal observations). As would be hypothesised, genotype D was better adapted to UVR stress than genotype N. However, this should be treated as anecdotal as there is no UVR profile for the native pond of genotype P, nor were multiple clones from each lake used. Future studies could directly address this question of UVR adaptation through comparisons of within- and between-lake clonal fitness, along a gradient of increasing transparency. Nonetheless, the various inter-clonal responses not only demonstrate the importance in utilising multiple genotypes in experimental studies, but also highlight genotype-by-environment interactions that may be a key to explain the incredibly rapid local adaptations by many zooplankton, including Daphnia59,60.
Generation G4 results reveal current treatment is a significant determinant of survival, reproduction and maturity, with those in the fluctuating environments exhibiting longer lifespans, increased offspring production, and delayed reproductive maturity. It is worth mentioning that genotype also impacts survival, but there were no a priori hypotheses as to which genotype would fare better. While ancestry did not exhibit any interactions with current treatment as predicted by the maternal matching hypothesis, it emerged as a significant contributor to survival and reproduction, following the same pattern as the current treatment. In addition to these significant differences in survival, it is evident that generation G4 suffered far greater mortality early in life across both ancestries as compared with the mortality in generation G3. Given generation G4 and G3 were largely contemporaneous, this indicates the presence of negative carryover effects of UVR, as opposed to a difference in the stress, covarying with time. Such carryover effects have been demonstrated in other multigenerational studies10. Taken together, this suggests that those mothers from a fluctuating environment were better able to allocate resources to all life history traits, which extends across generations to their offspring.
Trade-offs have been identified between various trait combinations, with one of the most extensively studied being the trade-off between early-life reproduction and survival61. As the reproductive value of an organisms is typically higher earlier in life, due to chance of death increasing as the organism ages, earlier maturation is often favoured. This investment into reproduction means less energy is available to repair the cellular damage accrued over life, which leads to earlier death62,63. As this relies on the perception of both the environmental conditions and the organism’s reproductive value, under a predictably stressful environment an organism may decide to produce offspring earlier. However, as animals likely accrue information through processes similar to Bayesian updating (the combination of prior information and new data leading to decision making)64, exposure to variably stressful environments may provide too little information to reliably initiate earlier reproduction. Therefore, a delayed reproduction may be favoured in order to assimilate more accurate information. In this study, I observed the same phenomena of an early maturation date coupled with shortened lifespan, which led to lower fitness values (fewer individuals produced towards the next generation) in the constant UVR environment. Evidently, this marginally earlier maturity did not compensate for the decrease in lifespan. This suggests that the ‘predictability’ of the constant treatment may be an ‘evolutionary trap’65 due to the timing of investment in reproduction coupled with the physiological costs of UVR exposure early in life. However, it is important to note here how despite the constant treatment group being potentially ‘predictable’, to accurately disentangle predictability of a stressor and the period of stress requires a carefully considered experimental design that also includes regular fluctuations. This would allow the disambiguation of predictable stress from the consistency of stress.
Expanding on the physiological costs incurred by UVR, such as the increase in malonaldehyde concentration and catalase activity66, even repair mechanisms may negatively impact fitness if produced at high doses22. In zooplankton, UVR triggers a range of responses, including an increase in metabolism, engagement of antioxidant pathways, and upregulation of photorepair mechanisms/compounds, such as the photo-enzymatic repair (PER) process that utilizes the photolyase enzyme34,49. PER is specifically induced by UV-A radiation, which is the predominant wavelength emission used in this study, and acts by repairing cytotoxic photoproducts67,68. Although studies have demonstrated interspecific differences in PER activity and how various developmental stages rely on this mechanism to different degrees, no study has quantified how short-term fluctuations of UVR and photosynthetically active radiation (i.e., visible light) affect this response, and specifically the dose produced. Plainly, further research is needed in relation to the fluctuations and duration of UVR on the physiological repair mechanisms, in order to mechanistically link PER and the fitness outcomes observed. Despite this, and the fact that PER is a relatively slow reaction49, it has been stated that even short periods, on the minute-scale, can be enough to increase survival after UVR exposure33. Perhaps, the earlier onset of UVR coupled with short (15–60 min) periods of PER activating visible light in the fluctuating treatment in my study were then capable of offsetting negative effects of UVR, compared to the single block of UVR in the constant regime. Furthermore, this appears more critical in early life, in line with findings that report UVR-induced early life mortality28 and the differential effectivity of PER at distinct developmental stages69.
Such UVR-induced early life mortality, which is clear in generation G4, may explain the disparity in results and original hypothesis based on within generation fitness consequences of fluctuating UVR43. More specifically, Stábile et al.43 isolated juvenile (8 day) Daphnia magna for the study whereas I used day-old neonates. Therefore, the early life selection, which I show here is greater in the constant environment, was missed. This highlights the importance of timing of the novel environmental conditions; if the variable stressor arises in adulthood, it may reduce current reproductive output, yet if that stress continues into the next generation the offspring may do better than if they were exposed to a more consistently stressful environment. Despite differences between the study by Stábile et al.43 and this study regarding the within generation response, a common result is that the focal genotypes have distinctly different responses.
In a broader context, my study highlights that genotype-by-environment, i.e. eco-evolutionary, interactions have profound effects on the clonal representation in the environment, and I show here that among only three genotypes, there are highly different responses to the variability or periodicity of the mortal threat UVR. Even within the confines of this four-generation study one genotype became extinct, and another had only two individuals in generation G4 that reproduced (Fig. 6). As seasons change in natural ecosystems, the fluctuations of stressors like UVR may also vary, which could lead to local extinction of genotypes, as demonstrated here. Furthermore, as cloud cover is moving poleward due to climate change70 my results indicate that Daphnia magna will likely be under stronger selection due to increased UVR, especially considering the predicted increase in heat waves will bring a more consistent period of UVR exposure71. Alternatively, the serious fitness consequences of UVR may facilitate the evolution of specialised strategies, leading to patterns of seasonal clonal succession. In support of this notion is the documented clonal composition variation of zooplankton in response to variation in multiple stressors including toxic food and predation cues60,72,73.
In conclusion, I demonstrate that the variability of UVR exposure has fitness consequences that are independent of the total dose of irradiation. These results suggest that organisms experiencing fluctuating stressors may deploy adaptive strategies that mitigate fitness losses, in contrast to those exposed to constant stressors, which may amplify fitness consequences. Furthermore, these environments have intergenerational consequences, with prior selective pressures shaping genotypic responses to new challenges. This underscores the role of temporal variability in driving shifts in seasonal clonal successions and determining dominant genotype–phenotype combinations. By considering multiple generations, diverse genotypes, and both stable and variable environmental regimes, this study highlights the nuanced mechanisms through which organisms navigate variable stressors, advancing our understanding of evolutionary strategies in dynamic environments and providing empirical data for the further development of theory on dynamic environments.
Data availability
Data and code used in this study are available at the Dryad (https://doi.org/10.5061/dryad.m63xsj4cw) and Zenodo (https://doi.org/10.5281/zenodo.14500807) repositories, respectively.
References
West-Eberhard, M. J. Developmental Plasticity and Evolution (Oxford University Press, 2003).
McNamara, J. M., Dall, S. R. X., Hammerstein, P. & Leimar, O. Detection vs. selection: Integration of genetic, epigenetic and environmental cues in fluctuating environments. Ecol. Lett. 19, 1267–1276. https://doi.org/10.1111/ele.12663 (2016).
Goeppner, S. R., Roberts, M. E., Beaty, L. E. & Luttbeg, B. Freshwater snail responses to fish predation integrate phenotypic plasticity and local adaptation. Aquat. Ecol. 54, 309–322. https://doi.org/10.1007/s10452-019-09744-x (2020).
Agrawal, A. A., Laforsch, C. & Tollrian, R. Transgenerational induction of defences in animals and plants. Nature 401, 60–63. https://doi.org/10.1038/43425 (1999).
Ehrenreich, I. M. & Pfennig, D. W. Genetic assimilation: A review of its potential proximate causes and evolutionary consequences. Ann. Botany 117, 769–779. https://doi.org/10.1093/aob/mcv130 (2016).
Walsh, M. R., Whittington, D. & Funkhouser, C. Thermal transgenerational plasticity in natural populations of Daphnia. Integr. Comp. Biol. 54, 822–829. https://doi.org/10.1093/icb/icu078 (2014).
Bernhardt, J. R., O’Connor, M. I., Sunday, J. M. & Gonzalez, A. Life in fluctuating environments. Philos. Trans. R. Soc. B Biol. Sci. 375, 20190454. https://doi.org/10.1098/rstb.2019.0454 (2020).
Uller, T., Nakagawa, S. & English, S. Weak evidence for anticipatory parental effects in plants and animals. J. Evolut. Biol. 26, 2161–2170. https://doi.org/10.1111/jeb.12212 (2013).
Radersma, R., Hegg, A., Noble, D. W. A. & Uller, T. Timing of maternal exposure to toxic cyanobacteria and offspring fitness in Daphnia magna: implications for the evolution of anticipatory maternal effects. Ecol. Evolut. 8, 12727–12736. https://doi.org/10.1002/ece3.4700 (2018).
Sha, Y. C. & Hansson, L. A. Ancestral environment determines the current reaction to ultraviolet radiation in Daphnia magna. Evolution 76, 1821–1835. https://doi.org/10.1111/evo.14555 (2022).
Yin, J. J., Zhou, M., Lin, Z. R., Li, Q. S. Q. & Zhang, Y. Y. Transgenerational effects benefit offspring across diverse environments: A meta-analysis in plants and animals. Ecol. Lett. 22, 1976–1986. https://doi.org/10.1111/ele.13373 (2019).
Pigliucci, M. Beyond Nature and Nurture (Johns Hopkins University Press, 2001).
Innes-Gold, A. A., Zuczek, N. Y. & Touchon, J. C. Right phenotype, wrong place: Predator-induced plasticity is costly in a mismatched environment. Proc. R. Soc. B-Biol. Sci. 286, 20192347. https://doi.org/10.1098/rspb.2019.2347 (2019).
Haaland, T. R., Wright, J. & Ratikainen, I. I. Bet-hedging across generations can affect the evolution of variance-sensitive strategies within generations. Proc. R. Soc. B Biol. Sci. 286, 20192070. https://doi.org/10.1098/rspb.2019.2070 (2019).
Starrfelt, J. & Kokko, H. Bet-hedging—A triple trade-off between means, variances and correlations. Biol. Rev. 87, 742–755. https://doi.org/10.1111/j.1469-185X.2012.00225.x (2012).
Childs, D. Z., Metcalf, C. J. E. & Rees, M. Evolutionary bet-hedging in the real world: Empirical evidence and challenges revealed by plants. Proc. R. Soc. B Biol. Sci. 277, 3055–3064. https://doi.org/10.1098/rspb.2010.0707 (2010).
Kain, J. S. et al. Variability in thermal and phototactic preferences in Drosophila may reflect an adaptive bet-hedging strategy. Evolution 69, 3171–3185. https://doi.org/10.1111/evo.12813 (2015).
Pinceel, T. et al. An empirical confirmation of diversified bet hedging as a survival strategy in unpredictably varying environments. Ecology https://doi.org/10.1002/ecy.3496 (2021).
Lind, M. I. et al. Environmental variation mediates the evolution of anticipatory parental effects. Evolut. Lett. 4, 371–381. https://doi.org/10.1002/evl3.177 (2020).
Fawcett, T. W. & Frankenhuis, W. E. Adaptive explanations for sensitive windows in development. Front. Zool. 12, S3. https://doi.org/10.1186/1742-9994-12-s1-s3 (2015).
Hoverman, J. T. & Relyea, R. A. How flexible is phenotypic plasticity? Developmental windows for trait induction and reversal. Ecology 88, 693–705. https://doi.org/10.1890/05-1697 (2007).
Taborsky, B. et al. An evolutionary perspective on stress responses, damage and repair. Horm. Behav. 142, 105180. https://doi.org/10.1016/j.yhbeh.2022.105180 (2022).
Kielland, O. N., Bech, C. & Einum, S. Is there plasticity in developmental instability? The effect of daily thermal fluctuations in an ectotherm. Ecol. Evolut. 7, 10567–10574. https://doi.org/10.1002/ece3.3556 (2017).
Burton, T., Lakka, H. K. & Einum, S. Measuring phenotypes in fluctuating environments. Funct. Ecol. 34, 606–615. https://doi.org/10.1111/1365-2435.13501 (2020).
Drake, M., Miller, N. & Todgham, A. The role of stochastic thermal environments in modulating the thermal physiology of an intertidal limpet, <i>Lottia digitalis</i>. J. Exp. Biol. 220, 3072–3083. https://doi.org/10.1242/jeb.159020 (2017).
Stocker, C. et al. The effect of temperature variability on biological responses of ectothermic animals—A meta-analysis. Ecol. Lett. https://doi.org/10.1111/ele.14511 (2024).
IPCC. in Climate change 2023: Synthesis Report. Contribution of working groups I, II and III to the sixth assessment report of the intergovernmental panel on climate change (ed Lee Core writing team, H. and Romero, J.) 35–115 (IPCC, Geneva, Switzerland, 2023).
Huebner, J. D., Young, D. L. W., Loadman, N. L., Lentz, V. J. & Wiegand, M. D. Age-dependent survival, reproduction and photorepair activity in Daphnia magna (Straus, 1820) after exposure to artificial ultraviolet radiation. Photochem. Photobiol. 82, 1656–1661. https://doi.org/10.1562/2006-05-03-ra-890 (2006).
Fernández, C. E., Campero, M., Uvo, C. & Hansson, L.-A. Disentangling population strategies of two cladocerans adapted to different ultraviolet regimes. Ecol. Evolut. 8, 1995–2005. https://doi.org/10.1002/ece3.3792 (2018).
Wolinski, L., Souza, M. S., Modenutti, B. & Balseiro, E. Effect of chronic UVR exposure on zooplankton molting and growth. Environ. Pollut. 267, 115448. https://doi.org/10.1016/j.envpol.2020.115448 (2020).
Fischer, J. M., Fields, P. A., Pryzbylkowski, P. G., Nicolai, J. L. & Neale, P. J. Sublethal exposure to UV radiation affects respiration rates of the freshwater cladoceran Daphnia catawba. Photochem. Photobiol. 82, 547–550. https://doi.org/10.1562/2005-08-30-ra-664 (2006).
Hansson, L.-A. Induced pigmentation in zooplankton: A trade-off between threats from predation and ultraviolet radiation. Proc. R. Soc. B Biol. Sci. 267, 2327–2331. https://doi.org/10.1098/rspb.2000.1287 (2000).
Hansson, L.-A. & Hylander, S. Effects of ultraviolet radiation on pigmentation, photoenzymatic repair, behavior, and community ecology of zooplankton. Photochem. Photobiol. Sci. 8, 1266–1275. https://doi.org/10.1039/b908825c (2009).
Oexle, S. et al. Rapid evolution of antioxidant defence in a natural population of Daphnia magna. J. Evolut. Biol. 29, 1328–1337. https://doi.org/10.1111/jeb.12873 (2016).
Rhode, S. C., Pawlowski, M. & Tollrian, R. The impact of ultraviolet radiation on the vertical distribution of zooplankton of the genus Daphnia. Nature 412, 69–72. https://doi.org/10.1038/35083567 (2001).
Leach, T. H., Williamson, C. E., Theodore, N., Fischer, J. M. & Olson, M. H. The role of ultraviolet radiation in the diel vertical migration of zooplankton: An experimental test of the transparency-regulator hypothesis. J. Plankton Res. 37, 886–896. https://doi.org/10.1093/plankt/fbv061 (2015).
Hansson, L. A., Hylander, S. & Sommaruga, R. Escape from UV threats in zooplankton: A cocktail of behavior and protective pigmentation. Ecology 88, 1932–1939. https://doi.org/10.1890/06-2038.1 (2007).
Lee, M. & Hansson, L.-A. Daphnia magna trade-off safety from UV radiation for food. Ecol. Evolut. 11, 18026–18031. https://doi.org/10.1002/ece3.8399 (2021).
Walsh, M. R. et al. in Life Histories: Volume 5 (eds Martin Thiel & Gary A. Wellborn) (Oxford University Press, 2018).
Gustafsson, S., Rengefors, K. & Hansson, L. A. Increased consumer fitness following transfer of toxin tolerance to offspring via maternal effects. Ecology 86, 2561–2567. https://doi.org/10.1890/04-1710 (2005).
Coakley, C. M., Nestoros, E. & Little, T. J. Testing hypotheses for maternal effects in Daphnia magna. J. Evolut. Biol. 31, 211–216. https://doi.org/10.1111/jeb.13206 (2018).
Zagarese, H. E., Cravero, W., Gonzalez, P. & Pedrozo, F. Copepod mortality induced by fluctuating levels of natural ultraviolet radiation simulating vertical water mixing. Limnol. Oceanogr. 43, 169–174. https://doi.org/10.4319/lo.1998.43.1.0169 (1998).
Stábile, F., Brönmark, C., Hansson, L.-A. & Lee, M. Fitness cost from fluctuating ultraviolet radiation in Daphnia magna. Biol. Lett. https://doi.org/10.1098/rsbl.2021.0261 (2021).
Miner, B. E., Kulling, P. M., Beer, K. D. & Kerr, B. Divergence in DNA photorepair efficiency among genotypes from contrasting UV radiation environments in nature. Mol. Ecol. 24, 6177–6187. https://doi.org/10.1111/mec.13460 (2015).
Galloway, L. & Etterson, J. Transgenerational plasticity is adaptive in the wild. Science 318, 1134–1136. https://doi.org/10.1126/science.1148766 (2007).
Oliveira, C. Y. B. et al. A multidisciplinary review of Tetradesmus obliquus: A microalga suitable for large-scale biomass production and emerging environmental applications. Rev. Aquacult. 13, 1594–1618. https://doi.org/10.1111/raq.12536 (2021).
Klüttgen, B., Dülmer, U., Engels, M. & Ratte, H. T. ADaM, an artificial fresh-water for the culture of zooplankton. Water Res. 28, 743–746. https://doi.org/10.1016/0043-1354(94)90157-0 (1994).
Williamson, C. E. & Rose, K. C. When UV meets fresh water. Science 329, 637–639 (2010).
Grad, G., Williamson, C. E. & Karapelou, D. M. Zooplankton survival and reproduction responses to damaging UV radiation: A test of reciprocity and photoenzymatic repair. Limnol. Oceanogr. 46, 584–591. https://doi.org/10.4319/lo.2001.46.3.0584 (2001).
Rautio, M. & Tartarotti, B. UV radiation and freshwater zooplankton: Damage, protection and recovery. Freshw. Rev. J. Freshw. Biol. Assoc. 3, 105–131 (2010).
Connelly, S. J. et al. UV-stressed Daphnia pulex increase fitness through uptake of vitamin D-3. Plos One https://doi.org/10.1371/journal.pone.0131847 (2015).
Harney, E., Paterson, S. & Plaistow, S. J. Offspring development and life-history variation in a water flea depends upon clone-specific integration of genetic, non-genetic and environmental cues. Funct. Ecol. 31, 1996–2007. https://doi.org/10.1111/1365-2435.12887 (2017).
R Core Team. (R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/, (2021).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, 2016).
Therneau, T. M. A package for survival analysis in R. See https://cran.r-project.org/package=survival (2022).
Brooks, M. et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J. https://doi.org/10.32614/RJ-2017-066 (2017).
Hartig, F. DHARMa: residual diagnostics for hierarchical (multi-level/mixed) regression models. R package version 0.3 3 (2020).
Bates, D., Machler, M., Bolker, B. M. & Walker, S. C. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Stoks, R., Govaert, L., Pauwels, K., Jansen, B. & De Meester, L. Resurrecting complexity: the interplay of plasticity and rapid evolution in the multiple trait response to strong changes in predation pressure in the water flea Daphnia magna. Ecol. Lett. 19, 180–190. https://doi.org/10.1111/ele.12551 (2016).
Schwarzenberger, A., D’Hondt, S., Vyverman, W. & von Elert, E. Seasonal succession of cyanobacterial protease inhibitors and Daphnia magna genotypes in a eutrophic Swedish lake. Aquatic Sci. 75, 433–445. https://doi.org/10.1007/s00027-013-0290-y (2013).
Kirkwood, T. B. L. & Rose, M. R. Evolution of senescence—Late survival sacrificed for reproduction. Philos. Trans. R. Soc. B Biol. Sci. 332, 15–24. https://doi.org/10.1098/rstb.1991.0028 (1991).
Daan, S., Deerenberg, C. & Dijkstra, C. Increased daily work precipitates natural death in the kestrel. J. Anim. Ecol. 65, 539–544. https://doi.org/10.2307/5734 (1996).
Maklakov, A. A. et al. Antagonistically pleiotropic allele increases lifespan and late-life reproduction at the cost of early-life reproduction and individual fitness. Proc. R. Soc. B Biol. Sci. https://doi.org/10.1098/rspb.2017.0376 (2017).
Stamps, J. A. & Krishnan, V. V. Combining information from ancestors and personal experiences to predict individual differences in developmental trajectories. Am. Nat. 184, 647–657. https://doi.org/10.1086/678116 (2014).
Schlaepfer, M. A., Runge, M. C. & Sherman, P. W. Ecological and evolutionary traps. Trends Ecol. Evolut. 17, 474–480. https://doi.org/10.1016/s0169-5347(02)02580-6 (2002).
Vega, M. P. & Pizarro, R. A. Oxidative stress and defence mechanisms of the freshwater cladoceran Daphnia longispina exposed to UV radiation. J. Photochem. Photobiol. B-Biol. 54, 121–125. https://doi.org/10.1016/s1011-1344(00)00005-1 (2000).
Mitchell, D. L. & Karentz, D. in Environmental UV Photobiology (eds Antony R. Young, Johan Moan, Lars Olof Björn, & Wilhelm Nultsch) 345–377 (Springer, 1993).
MacFadyen, E. J. et al. Molecular response to climate change: temperature dependence of UV-induced DNA damage and repair in the freshwater crustacean Daphnia pulicaria. Global Change Biol. 10, 408–416. https://doi.org/10.1111/j.1529-8817.2003.00750.x (2004).
Ramos-Jiliberto, R., Dauelsberg, P. & Zúñiga, L. R. Differential tolerance to ultraviolet-B light and photoenzymatic repair in cladocerans from a Chilean lake. Marine Freshw. Res. 55, 193–200. https://doi.org/10.1071/mf03027 (2004).
Norris, J. R. et al. Evidence for climate change in the satellite cloud record. Nature 536, 72. https://doi.org/10.1038/nature18273 (2016).
Ha, K. J. et al. Dynamics and characteristics of dry and moist heatwaves over East Asia. Npj Clim. Atmosph. Sci. https://doi.org/10.1038/s41612-022-00272-4 (2022).
Carvalho, G. R. & Crisp, D. J. The clonal ecology of Daphnia magna (Crustacea, Cladocera) 1. Temporal changes in the clonal structure of a natural population. J. Anim. Ecol. 56, 453–468. https://doi.org/10.2307/5060 (1987).
Stibor, H. & Lampert, W. Components of additive variance in life-history traits of Daphnia hyalina: Seasonal differences in the response to predator signals. Oikos 88, 129–138. https://doi.org/10.1034/j.1600-0706.2000.880115.x (2000).
Acknowledgements
I would like to thank Alexander Hegg for invaluable assistance during the experimental work. I would also like to thank Lars-Anders Hansson and Matthew Walsh for constructive feedback on both the design and writing of this manuscript. This study was financed by the Swedish Research Council (VR; grant #2016-03552) and the Royal Physiographic Society. I am currently financed by the Swedish Research Council (VR; grant #2024-00429).
Funding
Open access funding provided by Lund University.
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, M. Variable stressor exposure shapes fitness within and across generations. Sci Rep 15, 3626 (2025). https://doi.org/10.1038/s41598-025-87334-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-87334-8