Abstract
Cerebral hemorrhage is a major cause of mortality and disability. This study investigates the association between glycemic variability (GV) and short- and long-term clinical outcomes such as poor outcomes at discharge, mortality at 90 days and 1 year and intensive care unit (ICU) /hospital length of stay (LOS) in ICU patients with critically ill cerebral hemorrhage. This retrospective analysis examined 732 ICU patients with non-traumatic cerebral hemorrhage from the Medical Information Mart for Intensive Care (MIMIC)-IV database. GV was quantified as the ratio of standard deviation to mean glucose during ICU stay. To assess associations between GV and clinical outcomes (poor outcomes at discharge, 90-day and 1-year mortality, ICU/hospital LOS), the study employed logistic regression, Cox proportional hazards models, and linear regression. Additionally, non-linear relationships were explored through restricted cubic spline analysis. The investigation further incorporated subgroup and sensitivity analyses to ensure robustness of findings. To evaluate the incremental predictive value of GV, the study utilized receiver operating characteristic (ROC) curve analysis, net reclassification improvement, and integrated discrimination improvement, thereby providing a comprehensive assessment of GV’s clinical utility. Higher GV was significantly associated with increased risk of poor outcomes at discharge and 90-day and 1-year mortality in both patient groups. GV showed a linear association with poor outcomes at discharge but a non-linear association with 90-day and 1-year mortality. GV thresholds of ≥ 0.11 for non-traumatic cerebral hemorrhage increased mortality risks. Cohort showed non-linear relationships between GV and ICU/hospital LOS. GV’s impact was stronger in non-hypertensive and male patients. Adding GV to existing severity scores improved their predictive ability for adverse outcomes. In patients with non-traumatic cerebral hemorrhage admitted to the ICU, GV demonstrates an independent association with poor outcomes over both short-term and long-term time horizons. Furthermore, GV is associated with extended durations of both ICU and overall hospital stays in this patient population. These findings underscore the importance of glycemic control in this patient population and suggest that GV could be a valuable prognostic indicator and potential therapeutic target.
Similar content being viewed by others
Introduction
Cerebrovascular diseases, such as cerebral hemorrhage and infarction, are major global causes of mortality and disability, impacting around 15 million individuals each year, resulting in 5 million deaths and 5 million cases of permanent disability1,2,3,4. Managing these patients in the intensive care unit (ICU) is challenging due to multi-organ dysfunction and complex treatment needs5, highlighting the need to identify factors affecting prognosis to improve outcomes and reduce healthcare burdens.
Insulin resistance (IR), common in cerebrovascular disease patients, reflects systemic inflammation and metabolic disturbances, leading to blood glucose fluctuations that exacerbate metabolic dysfunction and worsen clinical outcomes6,7,8. While prior research has extensively explored glycemic thresholds9,10—such as levels exceeding 10.5 mmol/L (190 mg/dL) being linked to increased in-hospital mortality11. Research on glycemic variability (GV)’s role in non-traumatic cerebral hemorrhage patients remains limited. GV, defined as fluctuations in blood glucose over time, a less-explored metric in non-traumatic cerebral hemorrhage, provides dynamic insight into glucose control and complements existing evidence on glycemic thresholds12. Studies consistently show that critically ill patients exhibit significant attenuation of insulin sensitivity, intensifying GV and complicating the metabolic landscape of severe illness13,14. Elevated GV has been associated with adverse outcomes, including increased mortality, prolonged hospital stays, and higher complications in critically ill patients15,16,17,18.
In evaluating ICU patient prognosis, both short- and long-term indicators are crucial. Short-term indicators, like poor discharge outcomes, provide immediate feedback, while long-term measures, such as survival rates, reveal sustained impacts of care19,20. ICU and hospital length of stay (LOS) also reflect medical resource use and recovery. While GV’s impact on outcomes has been linked to oxidative stress and inflammation, most studies focus on diabetic populations or short-term outcomes21,22. This study investigates GV’s association with discharge outcomes, 90-day and 1-year mortality, and LOS, aiming to complement existing evidence on glucose management in critically ill patients.
Methods
Data source and study population
This study used data from the Medical Information Mart for Intensive Care (MIMIC) IV (version 2.2, https://physionet.org/content/mimiciv/2.2/), a clinical database of 76,943 ICU admissions for 53,150 patients23. These patients received care at Beth Israel Deaconess Medical Center between 2008 and 2019, prior to the COVID-19 pandemic. To ensure confidentiality, all personal identifiers were removed. Due to the de-identification of patient data, the institutional review board waived the need for individual consent. Data extraction was performed by Shuhuai Zou (ID: 55774871).
Inclusion criteria
Study eligibility criteria included:
-
(a)
A confirmed diagnosis of non-traumatic cerebral hemorrhage, as defined by the International Classification of Diseases (ICD) coding system (Ninth or Tenth Revision).
-
(b)
Admission to an ICU during hospitalization.
Exclusion criteria
Patients were excluded if they met any of the following:
-
(a)
Insufficient glucose data (fewer than three measurements during ICU stay)24,25,26.
-
(b)
Multiple ICU admissions (two or more separate ICU stays).
-
(c)
Inability to obtain the modified Rankin Scale (mRS) score at discharge records.
-
(d)
Presence of extreme values in the collected data, which were considered outliers and could potentially skew the results.
This study adhered to the ethical principles of the Declaration of Helsinki (1964) and its amendments. Ethical approval was obtained from the institutional review board. This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines27, ensuring adherence to rigorous methodological standards for observational research.
Data collection and processing
Data for this study were extracted from the database using Structured Query Language (SQL) and processed with PostgreSQL and Navicat Premium. Extracted variables included demographics, vital signs, comorbidities, severity scores, laboratory parameters, medical history, and clinical outcomes. Baseline characteristics were derived from data collected within the first 24 h of ICU admission, while GV was calculated as the ratio of the standard deviation (SD) to the mean of all glucose measurements during ICU stays.
Patients were stratified into four groups based on GV quartiles (Q1–Q4) following established methods from prior research28. Missing data (< 20%) were addressed using the random forest algorithm, ensuring robust data processing. Detailed methods, including data extraction, variable definitions, missing data patterns (Additional File 1: Figure S1), and stratification procedures, are provided in Additional File 1.
Primary and secondary outcomes
The main outcome was poor outcomes at discharge29,30,31, including death or severe disability, defined as a mRS score of 3–6 (0 = no symptoms, 6 = death) at discharge. Two independent senior neurosurgical experts (associate professor level or above) evaluated each patient’s mRS score based on discharge records. Outcomes were dichotomized into ‘Yes’ (mRS 3–6, poor outcome at discharge) or ‘No’ (mRS 0–2, favorable outcome). In cases of disagreement, a third senior neurosurgeon (professor level) made the final decision, resulting in a definitive ‘Yes’ or ‘No’ outcome (Additional File 2: Table S1). Secondary outcomes were 90-day and 1-year mortality, as well as ICU and hospital LOS.
Statistical analyses
Baseline characteristics were summarized using appropriate descriptive statistics for continuous and categorical variables. Kaplan-Meier survival analysis and log-rank tests were used to compare survival probabilities across GV quartiles at 90 days and 1 year. Multiple regression models were applied to evaluate the associations between GV and clinical outcomes, including poor discharge outcomes, mortality, and LOS. Adjustments for potential confounders were made across three models, guided by clinical relevance and prior evidence. Non-linear relationships were explored using restricted cubic splines (RCS), and model performance was assessed using Area Under Curve (AUC), net reclassification improvement (NRI), and integrated discrimination improvement (IDI) metrics. Robustness was evaluated through sensitivity analyses, including multiple imputation methods and subgroup analyses. Statistical significance was set at two-tailed P < 0.05. Analyses were performed using R version 4.2.2. Detailed descriptions of statistical methods, variable selection, model specifications, and robustness testing are provided in Additional File 1 and Additional file 2: Tables S2-S7.
Results
Patient selection and cohort characteristics
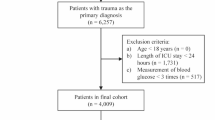
The study cohort included 732 patients with non-traumatic intracerebral hemorrhage, with a median age of 71 years (IQR: 60–81), comprising 396 males (396/732 [54.1%]) and 336 females (336/732 [45.9%]). The patient selection process is outlined in Fig. 1, and baseline characteristics stratified by GV quartiles are summarized in Table 1.
Flow chart of patient selection process. ICD-9/10 International Classification of Diseases, Ninth and Tenth Revision; MIMIC-IV Medical Information Mart for Intensive Care IV; ICU intensive care unit; mRS modified ranking scale.
Patients in higher GV quartiles had a significantly higher prevalence of comorbidities such as diabetes, respiratory failure, chronic kidney disease, kidney failure, and sepsis (all P < 0.05). Additionally, higher GV was associated with increased disease severity scores (APSIII, OASIS, SAPSII, and CCI) and elevated BUN levels (all P < 0.001).
Clinical outcomes progressively worsened with increasing GV levels. Poor outcomes at discharge and mortality rates at 90 days and 1 year were all significantly higher in patients with elevated GV, as shown in Table 1. ICU and hospital length of stay were also prolonged in higher GV quartiles (P < 0.001).
Further stratification by gender and clinical outcomes is presented in Additional File 2: Tables S8 and S9, respectively. Notably, GV was significantly higher in patients with poor outcomes at discharge, 90-day mortality, and 1-year mortality compared to those without these outcomes, consistent with findings illustrated in Fig. 2.
Impact of glycemic variability on outcomes. ** P < 0.01; ***P < 0.001. POD poor outcomes at discharge, CI cerebral infarction; Yes, Survivor; No, Non-survival.
Survival analysis
Kaplan-Meier survival analysis demonstrated significant differences in cumulative mortality across GV quartiles at both 90-day and 1-year time points, with higher GV quartiles associated with significantly lower survival probabilities (Fig. 3, P < 0.05). Detailed survival curves and log-rank test results are provided in Additional File 1: Figure S2.
Kaplan–Meier survival analysis curves for 90-day and 1-year mortality. Kaplan–Meier curves illustrating the cumulative probability of all-cause mortality stratified by glycemic variability (GV) quartiles: (A) 90-day mortality, (B) 1-year mortality. GV Quartile Ranges: Q1: [0.0045, 0.105) ; Q2: [0.105, 0.15) ; Q3: [0.15, 0.215) ; Q4: [0.215, 1.3]. GV glycemic variability.
Regression analyses
The regression analyses identified significant associations between GV and poor outcomes at discharge, as well as 90-day and 1-year mortality, as summarized in Table 2. These associations were consistent across all three models, even after adjusting for demographic and clinical factors. For poor outcomes at discharge, higher GV was significantly associated with increased risk, with an OR per standardized unit increase in GV of 1.43 (95% CI: 1.08–1.97, P = 0.020) in Model 3. Quartile analysis indicated a clear trend (P for trend = 0.005), with patients in the highest quartile (Q4) showing more than double the risk compared to the lowest quartile (Q1) (OR: 2.37, 95% CI: 1.31–4.39). Similarly, GV was significantly associated with 90-day mortality, with a HR per standardized unit increase of 1.19 (95% CI: 1.08–1.30, P < 0.001) in Model 3, and quartile analysis showed a significant trend (P for trend < 0.001), with Q4 demonstrating almost double the mortality risk compared to Q1 (HR: 1.94, 95% CI: 1.35–2.79). For 1-year mortality, GV continued to show significant associations, with an HR per standardized unit increase of 1.15 (95% CI: 1.05–1.26, P = 0.002) in Model 3, and patients in Q4 exhibited a significantly higher risk compared to Q1 (HR: 1.78, 95% CI: 1.27–2.48, P < 0.001).
Additional File 2: Table S10 shows the relationship between GV and LOS in ICU and hospital across three models. GV as a continuous variable was not significantly associated with ICU LOS, but higher quartiles (Q2, Q3, Q4) were linked to longer ICU stays (Model 3: β for Q2 = 2.73, Q3 = 3.34, Q4 = 3.09, all P < 0.001). Higher quartiles of GV were also linked to longer hospital stays, though the association was weaker for Q4 in Model 3 (β for Q2 = 3.86, P = 0.014; Q3 = 5.11, P = 0.001; Q4 = 3.11, P = 0.054). These results indicate a clear relationship between higher GV and increased LOS in ICU and hospital settings, highlighting the importance of managing GV to reduce LOS and improve outcomes.
Restricted cubic spline analysis
We conducted RCS analysis to investigate potential nonlinear associations between GV and clinical outcomes. This analysis employed four knots (5th, 35th, 65th, and 95th percentiles). Significant associations were consistently observed across all models in all examined outcomes (P < 0.05).
The relationship between GV and poor outcomes at discharge was primarily linear (Fig. 4), with no significant non-linear relationships detected (P-nonlinear > 0.05). This linear association remained after adjusting for confounders. In contrast, GV and 90-day mortality showed significant non-linear components (P-nonlinear < 0.05), indicating a complex association. The association between GV and 1-year mortality is demonstrated, with all models showing significant non-linear relationships (P-nonlinear < 0.05). Inflection points defining critical thresholds for GV and mortality are provided in Additional File 2: Table S11. Model 3 identified an inflection point at 0.11 GV for 90-day mortality. For GV ≥ 0.11, the adjusted HR per SD was 1.21 (95% CI: 1.10–1.32, P < 0.001), indicating increased 90-day mortality risk. For 1-year mortality, the adjusted HR per SD was 1.17 (95% CI: 1.07–1.29, P < 0.001), indicating increased long-term mortality risk.
RCS curves for the relationship between GV and clinical outcomes. RCS Curves of GV and OR/HR: (A–C) RCS curves for Poor Outcomes at Discharge in Model 1, Model 2, and Model 3, respectively; (D–F) RCS curves for 90-day mortality in Model 1, Model 2, and Model 3, respectively; (G–I) RCS curves for 1-year mortality in Model 1, Model 2, and Model 3, respectively; GV glycemic variability, OR odds ratio, HR hazard ratio, RCS restricted cubic spline, ICU intensive care unit.
The analysis was extended to hospital outcomes (Additional File 1: Figure S3). The RCS curves showed significant nonlinear relationships between GV and both ICU and hospital LOS (P-nonlinear < 0.05). These findings highlight the importance of precise GV management to improve survival, with identified thresholds guiding clinical vigilance.
Predictive capacity and incremental value of GV
The predictive capacity and incremental value of GV for poor discharge outcomes, 90-day, and 1-year mortality were evaluated using AUC, NRI, and IDI metrics. GV demonstrated modest predictive performance and provided incremental improvement when incorporated into existing scoring tools. Detailed results are provided in Additional File 1: Figure S4 and Additional File 2: Table S12-S13.
Subgroup analyses
We assessed the risk stratification capacity of GV across subgroups. Subgroups were stratified by gender, age, hypertension, diabetes, and AF. Increased GV was linked to higher risk of adverse outcomes across subgroups (Fig. 5). The impact of GV on poor discharge outcomes appeared more pronounced in patients without hypertension [adjusted OR (95% CI): 2.03 (1.14–3.61), P = 0.017] compared to those with hypertension [adjusted OR (95% CI): 1.17 (0.85–1.61), P = 0.330]. However, given the multiple comparisons and the absence of consistent interactions across other analyses, this finding should be interpreted with caution (P for interaction = 0.046). Similar results were seen in males for 90-day mortality [adjusted HR (95% CI): 1.41 (1.21–1.65), P < 0.001 for males vs. 1.12 (0.98–1.29), P = 0.099 for females; P for interaction = 0.035]. These findings highlight the differential impact of GV on outcomes across subgroups, providing insights for tailoring glucose management in clinical practice.
Subgroup analyses for the association of glycemic variability with clinical outcomes. (A) Association between GV and Poor Outcomes at Discharge; (B) Association between GV and 90-day mortality; (C) Association between GV and 1-year mortality. GV glycemic variability; HR hazard ratio; CI confidence interval; BMI body mass index; AF atrial fibrillation.
Sensitivity analyses
Sensitivity analyses demonstrated consistent associations between GV and poor discharge outcomes, 90-day and 1-year mortality, as well as ICU/hospital LOS across multiple imputation methods, confirming the robustness of our findings. Detailed results, including subgroup analyses and confidence intervals, are provided in Table 2, Additional File 1: Figures S5-S9 and Additional File 2: Tables S10, S14–S21.
Discussion
This investigation uniquely examines the relationship of GV with short-term outcomes (poor discharge outcomes, 90-day mortality), long-term outcomes (1-year mortality), and hospital resource utilization (ICU and hospital LOS). This approach provides a holistic perspective on the impact of GV across temporal and clinical dimensions in this critical patient population.
Our findings show that in critically non-traumatic cerebral hemorrhage patients, higher GV levels, especially in the highest quartile, were significantly linked to increased risk of poor outcomes at discharge, 90-day and 1-year mortality, and prolonged ICU and hospital LOS, even after adjusting for confounders. GV had independent associations with these adverse outcomes, highlighting its potential as a crucial prognostic indicator. GV showed a linear association with poor discharge outcomes and non-linear relationships for mortality. Significant non-linear relationships were found between GV and both ICU and hospital LOS. Adding GV to baseline severity scores improved predictive value for adverse outcomes, as shown by enhancements in clinical scoring tools. These findings were robust across multiple sensitivity analyses.
Expanding the Understanding of glycemic variability in non-traumatic cerebral hemorrhage
Cerebrovascular diseases are major contributors to long-term disability and mortality globally, imposing significant economic burdens and affecting patients’ quality of life32. GV has recently emerged as a crucial prognostic factor in critical illnesses, including non-traumatic cerebral hemorrhage. Its significance has been demonstrated in various ICU populations. For example, Lu et al.33 reported that each SD increase in GV was linked to a 5% increase in mortality risk in sepsis patients. GV is also an independent predictor of diabetes-related complications34. In cerebrovascular patients, Cai et al.26 found significant associations between GV and severe consciousness disturbance, in-hospital mortality, and cognitive decline. While these studies highlighted GV’s importance, they focused mainly on short-term outcomes. Our study extends this by investigating GV’s relationship with poor outcomes, 90-day and 1-year mortality, and LOS in ICU and hospital settings, providing a deeper understanding of GV’s immediate and long-term impacts on this high-risk population.
Comparison with previous studies and novel insights into glycemic variability -outcome relationships
Our study extends Cai et al.26 by elucidating complex relationships between GV and outcomes in non-traumatic cerebral hemorrhage patients. GV showed a linear relationship with poor outcomes and non-linear associations with mortality. Cohort showed significant non-linear relationships between GV and ICU/hospital LOS. Methodological differences, including direct GV analysis versus logarithmic CV and variations in outcome definitions, may explain the divergence from previous findings. These results suggest the need for tailored glucose control approaches based on disease type and outcomes. Both studies highlight the complexity of GV-outcome relationships, emphasizing the need for further research in diverse populations.
This study identifies a significant non-linear association between GV and hospital stay in non-traumatic cerebral hemorrhage patients (P < 0.001, P-nonlinear < 0.05), with distinct inflection points that provide clinically meaningful thresholds for risk stratification. Additionally, GV ≥ 0.11 was associated with significantly increased 90-day and 1-year mortality risks, extending previous findings on the positive correlation between LogGV and hospital stay26. Our findings align with those from a study on the Triglyceride-glucose (TyG) index in critically ill patients35, where TyG was positively associated with LOS for hospital survivors (β = 1.36, P = 0.008) and ICU survivors (β = 0.87, P = 0.004). These findings suggest that glucose metabolism dysregulation, including GV and TyG, may be linked to prolonged hospitalization, possibly involving oxidative stress, inflammation, and increased complications36,37. Consistent results across different metrics and populations underscore the importance of glucose homeostasis in critical illness outcomes and hospital resource use.
Based on the data presented in Fig. 3; Table 2, and Additional File 2 Table S11, the observed non-linear increase in the RCS plots may be influenced by variability in data density and measurement frequency at the extremes of the GV distribution. This suggests that the primary findings likely reflect a linear association between higher GV and increased mortality risk, rather than a true hockeystick-like relationship. Notably, a similar nonlinear pattern was reported in Cai et al.’s study26, highlighting the need for careful interpretation of RCS analyses, particularly when variability at extreme values may impact the observed trends.
Subgroup analysis suggested that GV may have a greater impact on adverse outcomes in non-hypertensive hemorrhage patients. This finding could be partially explained by the potential link between insulin resistance, hyperinsulinemia, and aldosterone regulation in hypertensive patients38, as well as more pronounced glucose fluctuations in individuals with insulin resistance7. However, given the exploratory nature of this analysis and the possibility of chance findings due to multiple comparisons, these results should be interpreted with caution. It is also plausible that hypertensive patients benefit from regular interventions to mitigate glucose fluctuations, while normotensive patients may be more susceptible to the adverse effects of GV. Similarly, GV appeared to have a greater impact on 90-day mortality in males, which might be attributed to estrogen’s protective effect on insulin sensitivity in women and higher insulin resistance in men39,40,41. Nonetheless, further studies are needed to validate these observations and interactions in independent cohorts and to explore potential mechanisms underlying the differential impact of GV in hypertensive and non-hypertensive patients, as well as between males and females.
Pathophysiological mechanisms of glycemic variability in poor outcomes among patients with non-traumatic cerebral hemorrhage
The brain’s vulnerability to glucose fluctuations is due to its high energy demands and metabolic rate, increasing sensitivity to glycemic variations and oxidative stress42. In non-traumatic cerebral hemorrhage patients, especially in ICU, GV can negatively impact prognosis through various interconnected mechanisms.
At the subcellular level, GV affects mitochondrial function, including the electron transport chain, ATP production, and the balance between fusion and fission, leading to mitochondrial fragmentation and reduced energy efficiency43. Additionally, GV induces endoplasmic reticulum (ER) stress, activating the unfolded protein response (UPR). Sustained ER stress may exacerbate cellular injury, intensify inflammation, and oxidative stress via JNK and NF-κB pathways22. These mechanisms interact, creating a complex network in which oxidative stress and inflammation amplify endothelial dysfunction and blood-brain barrier disruption. GV also promotes plaque formation, affects platelet function, and coagulation factor activity in atherosclerosis21,22. In ICU non-traumatic cerebral hemorrhage patients, the critical condition activates the sympathetic nervous system, raising blood glucose levels. Factors like therapeutic interventions, stress, infections, and nutritional support further influence glucose fluctuations, complicating glucose management and necessitating more frequent monitoring and individualized treatment strategies.
Strengths and limitations
This study found an independent association between GV and both short-term outcomes and long-term (90-day and 1-year mortality) in ICU patients with non-traumatic cerebral hemorrhage, emphasizing its prognostic value. Limitations of this study include its retrospective design, which may introduce potential selection bias, and the reliance on the MIMIC-IV database, which lacks key ICH-specific characteristics such as size, location, presence of intraventricular extension, and pre-ICH mRS scores. The absence of these critical variables limits our ability to fully adjust for potential confounders, potentially affecting the robustness of the observed associations between GV and clinical outcomes. Additionally, the use of non-continuous glucose measurements may bias GV calculations and limit their precision. Future research should focus on prospective studies incorporating standardized, continuous glucose monitoring and datasets that include comprehensive ICH-specific variables. Such studies could also investigate the underlying mechanisms linking GV to adverse outcomes, including oxidative stress, inflammation, and endothelial dysfunction, to provide deeper insights into its role in critically ill patients with ICH.
Conclusion
This study shows GV’s role in cerebral hemorrhage prognosis, with higher GV linked to increased adverse events, mortality, and longer stays. Established GV thresholds (0.11) aid risk stratification, and adding GV improves scoring systems’ predictive power. GV monitoring and individualized treatment are crucial, and future research should validate these findings, explore mechanisms, and assess targeted GV interventions.
Data availability
The datasets generated and analyzed in this study are presented within the manuscript or supplementary materials, with additional information available from the corresponding author upon reasonable request.
References
Mola-Caminal, M. et al. PATJ low frequency variants are associated with worse ischemic stroke functional outcome. Circ. Res. 124, 114–120 (2019).
Zhang, H. et al. Associations of Chinese visceral adiposity index and new-onset stroke in middle-aged and older Chinese adults: an observational study. Lipids Health Dis. 22, 74 (2023).
Zhang, H. et al. Brain-computer interfaces: the innovative key to unlocking neurological conditions. Int. J. Surg. 110, 5745–5762 (2024).
Correa-Paz, C. et al. Sonosensitive capsules for brain thrombolysis increase ischemic damage in a stroke model. J. Nanobiotechnol. 20, 46 (2022).
Fläring, U. B. et al. Temporal changes in whole-blood and plasma glutathione in ICU patients with multiple organ failure. Intensive Care Med. 31, 1072–1078 (2005).
Saxena, M., Agrawal, C. G., Bid, H. K. & Banerjee, M. An interleukin-10 gene promoter polymorphism (-592A/C) associated with type 2 diabetes: a North Indian study. Biochem. Genet. 50, 549–559 (2012).
Freedman, B. I. et al. Genome-wide scans for heritability of fasting serum insulin and glucose concentrations in hypertensive families. Diabetologia 48, 661–668 (2005).
Hu, H. et al. Silicon dioxide nanoparticles induce insulin resistance through Endoplasmic reticulum stress and generation of reactive oxygen species. Part. Fibre Toxicol. 16, 41 (2019).
Saxena, A. et al. Prognostic significance of hyperglycemia in acute intracerebral hemorrhage: the INTERACT2 study. Stroke 47, 682–688 (2016).
Kreutziger, J. et al. Blood glucose concentrations in prehospital trauma patients with traumatic shock: A retrospective analysis. Eur. J. Anaesthesiol. 35, 33–42 (2018).
Wang, Y. et al. Threshold of hyperglycaemia associated with mortality in critically ill patients: a multicentre, prospective, observational study using continuous glucose monitoring. Diabetologia 67, 1295–1303 (2024).
Rodbard, D. Glucose Variability. A review of clinical applications and research developments. Diabetes Technol. The. 20 (2-5-), 15 (2018). S2-.
Black, P. R., Brooks, D. C., Bessey, P. Q., Wolfe, R. R. & Wilmore, D. W. Mechanisms of insulin resistance following injury. Ann. Surg. 196, 420–435 (1982).
Zauner, A. et al. Severity of insulin resistance in critically ill medical patients. Metabolis 56, 1–5 (2007).
Mendez, C. E. et al. Increased glycemic variability is independently associated with length of stay and mortality in noncritically ill hospitalized patients. Diabetes Care. 36, 4091–4097 (2013).
Hermanides, J. et al. Glucose variability is associated with intensive care unit mortality. Crit. Care Med. 38, 838–842 (2010).
Gerbaud, E. et al. Glycemic variability is a powerful independent predictive factor of midterm major adverse cardiac events in patients with diabetes with acute coronary syndrome. Diabetes Care. 42, 674–681 (2019).
Su, Y., Fan, W., Liu, Y. & Hong, K. Glycemic variability and in-hospital death of critically ill patients and the role of ventricular arrhythmias. Cardiovasc. Diabetol. 22, 134 (2023).
Pollack, M. M. et al. PICU length of stay: factors associated with bed utilization and development of a benchmarking model. Pediatr. Crit. Care Me. 19, 196–203 (2018).
Caruso, P. et al. Short- and long-term survival of patients with metastatic solid cancer admitted to the intensive care unit: prognostic factors. Eur. J. Cancer Care. 19, 260–266 (2010).
Bornfeldt, K. E. & Tabas, I. Insulin resistance, hyperglycemia, and atherosclerosis. Cell. Metab. 14, 575–585 (2011).
Mannucci, E., Dicembrini, I., Lauria, A. & Pozzilli, P. Is glucose control important for prevention of cardiovascular disease in diabetes? Diabetes Care. 36, S259–S263 (2013).
Johnson, A. E. W. et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data. 10, 1 (2023).
He, H. M. et al. Simultaneous assessment of stress hyperglycemia ratio and glycemic variability to predict mortality in patients with coronary artery disease: a retrospective cohort study from the MIMIC-IV database. Cardiovasc. Diabetol. 23, 61 (2024).
Zhou, Z., Zhang, H., Gu, Y., Zhang, K. & Ouyang, C. Relationship between glycemic variability and the incidence of postoperative atrial fibrillation following cardiac surgery: A retrospective study from MIMIC-IV database. Diabetes Res. Clin. Pract. 219, 111978 (2024).
Cai, W. et al. Association of glycemic variability with death and severe consciousness disturbance among critically ill patients with cerebrovascular disease: analysis of the MIMIC-IV database. Cardiovasc. Diabetol. 22, 315 (2023).
von Elm, E. et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int. J. Surg. 12, 1495–1499 (2014).
Chen, T., Qian, Y. & Deng, X. Triglyceride glucose index is a significant predictor of severe disturbance of consciousness and all-cause mortality in critical cerebrovascular disease patients. Cardiovasc. Diabetol. 22, 156 (2023).
Anderson, C. S. et al. Rapid blood-pressure Lowering in patients with acute intracerebral hemorrhage. N Engl. J. Med. 368, 2355–2365 (2013).
Yu, H. et al. The clinical value of serum Xanthine oxidase levels in patients with acute ischemic stroke. Redox Biol. 60, 102623 (2023).
Denti, L. et al. The poor outcome of ischemic stroke in very old people: a cohort study of its determinants. J. Am. Geriatr. Soc. 58, 12–17 (2010).
Feigin, V. L. et al. Global, regional, and National burden of stroke and its risk factors, 1990–2019: a systematic analysis for the global burden of disease study 2019. Lancet Neurol. 20, 795–820 (2021).
Lu, Z. et al. Association of blood glucose level and glycemic variability with mortality in Sepsis patients during ICU hospitalization. Front. Public. Health. 10, 857368 (2022).
Raj, R. et al. Time in range, as measured by continuous glucose monitor, as a predictor of microvascular complications in type 2 diabetes: a systematic review. BMJ Open. Diabetes Res. Care. 10, e002573 (2022).
Liao, Y. et al. Triglyceride-glucose index linked to all-cause mortality in critically ill patients: a cohort of 3026 patients. Cardiovasc. Diabetol. 21, 128 (2022).
Adams, J. A., Lopez, J. R., Banderas, V. & Sackner, M. A. A single arm trial using passive simulated jogging for blunting acute hyperglycemia. Sci. Rep. 11, 6437 (2021).
Hajime, M. et al. Twenty-four-hour variations in blood glucose level in Japanese type 2 diabetes patients based on continuous glucose monitoring. J. Diabetes Investig. 9, 75–82 (2018).
Sechi, L. A., Colussi, G. & Catena, C. Metabolic dysfunction in primary aldosteronism. Hypertension 53, e37 (2009). author reply e38.
Konrad, K. et al. Comparison of cystic fibrosis-related diabetes with type 1 diabetes based on a German/Austrian pediatric diabetes registry. Diabetes Care. 36, 879–886 (2013).
Huffman, K. M. et al. Relationships between Circulating metabolic intermediates and insulin action in overweight to obese, inactive men and women. Diabetes Care. 32, 1678–1683 (2009).
Tan, K. C. B., Shiu, S. W. M., Wong, Y. & Tam, X. Serum advanced glycation end products (AGEs) are associated with insulin resistance. Diabetes Metab. Res. Rev. 27, 488–492 (2011).
Waiwut, P. et al. Toxicity profiles of Kleeb Bua Daeng formula, a traditional Thai medicine, and its protective effects on memory impairment in animals. Pharmaceuticals 15, 988 (2022).
Mordel, P., Fontaine, F., Dupas, Q., Joubert, M. & Allouche, S. Glucose fluctuation promotes mitochondrial dysfunctions in the cardiomyocyte cell line HL-1. PLOS One. 18, e0289475 (2023).
Acknowledgements
We express our heartfelt gratitude to the contributions of all staff who work on the Medical Information Mart for Intensive Care.
Author information
Authors and Affiliations
Contributions
Dong Wang: Conceptualization, Methodology, Writing - Original Draft Preparation; Chang He and Shuhuai Zou: Data Curation, Formal Analysis, Visualization; Lizheng Yu and Biyuan Han: Investigation, Resources, Validation; Liming He, Ankang Liu, and Yingying Hong: Software, Writing - Review & Editing; Qianfeng Li: Writing - Review & Editing, Supervision, Project Administration, Funding Acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was conducted in full compliance with the principles outlined in the Declaration of Helsinki. The utilization of the MIMIC-IV database (https://physionet.org/content/mimiciv/2.2/) was sanctioned by the Institutional Review Board of the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center. Given that the MIMIC-IV database is a publicly accessible resource, the requirement for specific ethical approval and individual informed consent for this study was waived. This waiver is in accordance with standard procedures for research involving de-identified, publicly available datasets.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, D., He, C., Zou, S. et al. Glycemic variability and its association with short and long term clinical outcomes in critically ill patients with cerebral hemorrhage. Sci Rep 15, 7820 (2025). https://doi.org/10.1038/s41598-025-92415-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-92415-9
Keywords
This article is cited by
-
Differential prognostic value of glycemic dysregulation markers in critically ill individuals with ischemic stroke, with and without diabetes
Diabetology & Metabolic Syndrome (2025)