Abstract
To explore the therapeutic efficacy of L-glutamine (L-Gln) on pathological progression and clinical symptoms of osteoarthritis (OA), and compare with glucosamine sulfate (GS), and celecoxib (CXB). Rats were administered sodium chloride, L-Gln, GS, or CXB via gavage for eight weeks starting from the fifth week after sham operation or Anterior Cruciate Ligament Transection (ACLT) + Medial Meniscectomy (MMx). Then the severity of knee OA in rats was evaluated by serological analysis, histological examination and imaging examination. In addition, patients with mild primary OA were administered L-Gln, GS, or CXB orally for 12 weeks in accordance with the randomization principle. The efficacy end points were the change from baseline to week 24 in the pain and physical function subscale scores of the Western Ontario and McMaster Universities OA Index (WOMAC), and Lequesne score. Treatment with L-Gln alleviated the increased concentration of serum cartilage degradation markers caused by OA in rats. Histological tests showed improvement in knee joint cartilage destruction after treatment. Three-dimensional CT scans and reconstructions revealed a reduction in osteophyte formation and subchondral bone loss. L-glutamine performed as well as or better than glucosamine sulfate and celecoxib in all comparative measures among the three treatment groups. In clinical trials, the WOMAC pain and physical function subscale scores, as well as the Lequesne score, decreased from baseline in all three patient groups during follow-up, with no significant differences observed between the groups. Our research indicates that L-Gln is comparable to GS and CXB in improving the pathological progression and clinical efficacy of OA, which makes it a promising drug for the treatment of osteoarthritis.
Similar content being viewed by others
Introduction
Osteoarthritis (OA) is now recognized as a multifactorial joint disease involving not only articular cartilage degeneration and bone remodeling but also persistent and chronic low-grade inflammation. Emerging evidence highlights the critical role of inflammatory pathways in the initiation and progression of OA, shifting the traditional view of OA as merely a “wear-and-tear” condition1,2,3,4. At present, the management of OA primarily centers around alleviating pain, which entails orally administering non-steroidal anti-inflammatory drugs or intra-articular injections of hyaluronic acid and steroids etc. while these medications often come with significant side effects and high costs5,6,7. Besides, Due to the complex pathogenesis of OA, there are still no effective disease-modifying therapies for OA. Therefore, a disease-modifying OA drug that could alleviate or reverse the progression of OA is urgently required8.
Amino acids are the fundamental building blocks of proteins in our cells and tissues; they are the second most abundant compound in mammals, following water. Among the 20 amino acids that constitute the human body, glutamine is the most abundant and widely utilized free amino acid9. Glutamine serves as a metabolic fuel and protein precursor and also participates in many biological processes, such as immune regulation, cell proliferation, apoptosis, redox potential, insulin resistance, and respiratory burst. Moreover, glutamine has been shown to regulate the expression of many genes associated with signal transduction, metabolism, and cellular defense and repair10,11. Furthermore, in addition to serving as a nutrient source for patients with burns or critically ill patients, glutamine has demonstrated clinical utility in the prevention of stomatitis/mucositis or peripheral neuropathy in patients with cancer following chemotherapy and the reduction of sickle cell-related pain attacks12,13,14. The high catabolism of glutamine does not result in any discernable side effects, even when administered at a dosage as high as 30 g/d15,16.
Glutamine is a crucial substrate for the biological processes in chondrocytes in both healthy and diseased states. Chondrocytes survive and function in a nonvascular environment; thus, they cannot meet their high biosynthetic requirements by relying only on a limited glucose supply. In this context, amino acids become the optimal choice. Glutamine has been shown to support chondrocyte proliferation and matrix synthesis via transaminase mediated aspartic acid synthesis17,18. In addition, Glutamine epigenetically regulates the expression of chondrogenesis genes through glutamate dehydrogenase dependent acetyl-CoA synthesis, which is essential for histone acetylation19.
Glutamine also plays a crucial role in protecting cartilage. It serves as a critical precursor in the synthesis of glutathione, which plays a crucial role in eliminating harmful reactive oxygen species within the body19. Glutamine exerts a protective effect against chondrocyte apoptosis in response to thermal or nitric oxide stimulation20. In addition, inflammatory stimulation affects the uptake of glutamine by chondrocytes, thereby regulating the inflammatory response through the metabolic reprogramming or autophagy system21,22.
Several studies have demonstrated that in patients with OA, glutamine concentrations in the joint fluid are altered23,24. Supplementation of glutamine can alleviate cartilage damage caused by osteoarthritis and delay its progression. Our previous study involving a rat model of OA elucidated that L-glutamine (L-Gln) reduces the expression of nitric oxide synthase, cyclooxygenase-2, and matrix metalloproteinase-13 (MMP13) in chondrocytes through the nuclear factor-κ B pathway, thereby reducing the degradation of extracellular matrix (i.e., aggregator and type II collagen). In addition, administering the rats with L-Gln via gavage significantly ameliorated the destruction of knee joint cartilage25.
In this study, we investigated the efficacy of L-Gln in comparison to two commonly used OA medications in a rat model of knee OA, namely glucosamine sulfate (GS), known for its chondroprotective properties, and the nonsteroidal anti-inflammatory drug celecoxib (CXB). Besides, we conducted a randomized, double-blind, multi-center, positive-controlled trial and evaluated the differences among the efficacy of L-Gln, GS, and CXB in treating early primary OA. We are striving to find a treatment drug for osteoarthritis that combines good efficacy, affordability, and minimal side effects.
Materials and methods
Animals
Male Sprague-Dawley (SD) rats (300 ± 20 g) were obtained from Changsheng Biotechnology Co., Ltd. (Liaoning, China) and housed at 23 to 25 °C with a 12-hour light/dark cycle and allowed free access to water and standard laboratory diets. All animal experiments performed in this study strictly followed the ARRIVE guidelines and were approved by the Institutional Animal Care and Use Committee of Anhui Medical University (approval number LLSC20231140). All methods were performed in accordance with the relevant guidelines and regulations.
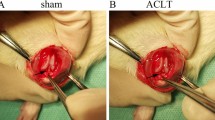
Following a one-week period of acclimation, the SD rats underwent either an Anterior Cruciate Ligament Transection (ACLT) + Medial Meniscectomy (MMx) or a sham operation for their right knees. Briefly, in the ACLT + MMx operation, the joint capsule of the rat was opened immediately post-anesthesia, and then the anterior cruciate ligament and part of the medial meniscus were excised to disrupt the stability of the knee joint without damaging other tissues. In the sham operation, the joint capsule was opened in the same way, but no additional damage was inflicted. The wound area was sterilized with a povidone-iodine solution, and cefazolin (100 mg/kg/d) was administered intramuscularly for three days to prevent wound infection.
In the fourth week post-ACLT + MMx surgery, we measured the concentration of L-Gln in the serum and synovial tissue at various time intervals following intragastric administration of L-Gln (250 mg/kg/d) to investigate the distribution and metabolism of L-Gln in the body of rats, especially in the knee joint synovium. To evaluate the efficacy of L-Gln and the drugs, we randomly divided the SD rats into five groups, namely sham (n = 5), OA (n = 5), OA + L-Gln (n = 5), OA + GS (n = 5), and OA + CXB (n = 5). Starting in the fifth week post-surgery, rats were administered sodium chloride (250 mg/kg/d), L-Gln (250 mg/kg/d), GS (130 mg/kg/d), or CXB (17 mg/kg/d) via gavage for eight weeks. In the 12th week post-surgery, the rats were euthanized under deep anesthesia with pentobarbital sodium, and the right knee joints and blood were obtained for subsequent experiments.
ELISA
Blood (3 mL) or synovial tissue of the knee joint (100 mg) was obtained immediately following the euthanization of the rats. Rat L-Gln, rat Cartilage Oligomeric Matrix Protein (COMP), rat interleukin-8 (IL-8), rat C-telopeptides of type II collagen (CTX-II) ELISA kits were purchased from Meimian Industrial Co., Ltd. (Jiangsu, China). The absorbance was measured at 450 nm using an Rayto-6100 microplate reader (Shenzhen, China), and a standard curve was prepared for the interpolation of the concentrations in each independent serum sample. All measurements were performed in duplicate according to the manufacturer’s instructions.
Micro-CT analysis
The right knee joints of rats were preserved in 10% formalin. The osteophytes and tibial plateausubchondral bone of the right knee joint were evaluated by micro-CT. Continuous tomographic scanning of the rat knee joints was performed at a voltage of 85 kV, a current of 200 mA, and a resolution of 10 μm using the SKYSCAN 1276 (Bruker, Belgium). Then, the ratio of the bone volume to the tissue volume (BV/TV), Trabecular Number (Tb. N) and Trabecular Separation (Tb. Sp) of the tibial subchondral bone was determined, and three-dimensional images of the knee joints were reconstructed using CTVOX software (Bruker, Belgium).
Histological examination
Following 48 h of fixation with Paraformaldehyde, the right knee joint of the rats was decalcified using a decalcifying solution containing 12.5% disodium ethylenediaminetetraacetate (pH = 7) for four weeks. The tissues were then embedded in paraffin, and histological Sect. (5-µm thick) were prepared. Hematoxylin and eosin (HE), safranin O-fast green, and immunohistochemistry staining against Matrix Metalloproteinase 13(MMP13) (AF5355, 1:100, Affinity, Jiangsu, China), Collagen II(COLII) (AF0135, 1:100, Affinity, Jiangsu, China)and Aggrecan (DF7561, 1:100, Affinity, Jiangsu, China) were performed. The stained sections were scanned with Axio Scan Z1 Microscope Slide Scanner (ZEISS, Jena, Germany). The mean cartilage degeneration severity (as per the Osteoarthritis Research Society International [OARSI] OA cartilage histopathology assessment system) were evaluated using the Zen Lite software (version 3.2; Carl Zeiss Microscope GmbH, Jena, Germany).
Patients
The inclusion criteria for this study were as follows: (1) Volunteer as a subject and sign the subject’s informed consent form; (2) Age between 40 and 80 years old, male or female; (3) diagnosed as knee OA according to the guidelines for the diagnosis and treatment of OA in China26; (4) Through imaging-verified diagnosis, the severity of osteoarthritis is classified using the Kellgren/Lawrence (K/L) grading system, with grades 1–2 out of a total of 0–427.
Patients with the following conditions were excluded: (1) Patients with end-stage osteoarthritis who require joint replacement; (2) Patients who participated in clinical trials of other drugs within three months before enrollment; (3) Patients who received unclear descriptions of treatment before enrollment; (4) Patients who took glucosamine, L-glutamine or non-steroidal anti-inflammatory drugs and other related OA therapies 2 weeks before enrollment; (5) Other osteoarthritis secondary to trauma, inflammation, joint instability, cumulative strain or congenital diseases; (6) Patients with other joint diseases, knee trauma or any other history of moderate to severe pain within 30 days; (7) Patients with severe primary diseases of the heart, brain, liver, kidney, lung and hematopoietic system; (8) People with allergies, such as those who are allergic to sulfonamides and those who induce asthma, urticaria or allergic reactions after taking aspirin or other non-steroidal anti-inflammatory drugs; (9) Psychiatric patients; (10) Pregnant or lactating women, unwilling to take contraceptive measures; (11) Known history of alcohol addiction or drug abuse; (12) Severe metabolic diseases and diabetic patients, etc; (13) People with peptic tract ulcers or active bleeding; (14) People who take drugs that are contraindicated in compatibility with L-Gln, GS, CXB.
Clinical study design
This phase II randomized, double-blind, positive control trial was conducted in five hospitals in Anhui Province, China, approved by the Ethics Committee of the Second Affiliated Hospital of Anhui Medical University (approval number YX2021-072). We conducted a retrospective registration for this study on 09/10/2023 at Chinese Clinical Trial Registry (URL: https://www.chictr.org.cn/. registration number: ChiCTR2300076450). All methods were carried out in accordance with relevant guidelines and regulations, and informed consent was obtained from all subjects and/or their legal guardians. All patients were randomly divided into three groups by drawing lots and received three different medication treatments for 12 weeks. Both the subjects and observers were unaware of the specific ingredients of the medication they were taking before unblinding. To ensure the implementation of a double-blind experiment, we used a double-dummy technique to design the placebos. Placebo I and L-glutamine, Placebo II and glucosamine sulfate (Zhejiang Haizheng Pharmaceutical Co., Ltd., Yisuojia, National Drug Approval Number: H20041316), and Placebo III and celecoxib (Pfizer Inc., Celebrex, National Drug Approval Number: J20140072) were identical in appearance, smell, and taste, preventing patients from identifying the active ingredients of the drugs by conventional means.The three treatment regimens were as follows:
Group 1: L-glutamine 1 g/qd + Placebo II 0.25 g/tid + Placebo III 0.2 g/qd (6 days on, 6 days off) for 12 weeks.
Group 2: Glucosamine sulfate 0.25 g/tid + Placebo I 1 g/qd + Placebo III 0.2 g/qd (6 days on, 6 days off) for 12 weeks.
Group 3: Celecoxib 0.2 g/qd (6 days on, 6 days off) + Placebo I 1 g/qd + Placebo II 0.25 g/tid for 12 weeks.
To enhance compliance, participants were provided with detailed instructions on medication use and regular reminders via phone calls or text messages.
The preliminary analysis of the therapeutic efficacy of L-Gln and the drugs was conducted in the 4th week, and subsequent follow-up assessments were conducted in the 12th and 24th weeks.
Efficacy endpoints
The primary efficacy endpoint was the alteration in the Western Ontario and McMaster Universities OA Index (WOMAC) pain subscale score from baseline to the 24th week. This score was determined by the total scores of the responses to five questions (a scale of 0–10) that assessed joint pain during walking, climbing stairs, resting in bed, sitting or lying, and standing. The minimal clinically important difference (MCID) for this score was 3.7528.
Secondary efficacy endpoints were alterations in the WOMAC physical function subscale score (the total scores of the responses to 17 questions; MCID, 11.3728) and Lequesne score ( the MCID have not been defined in publications29 ) from baseline to the 24th week.
Safety endpoints
Safety assessment was based on the frequency of treatment emergency adverse events (TEAEs) and imaging examinations. Adverse events were documented during each follow-up visit through structured interviews and patient self-reports. Magnetic resonance imaging (MRI) was performed at any time in the case of worsening joint pain that was inconsistent with the typical OA-related pain experienced by the patient. In the event that notable interval changes were observed in the follow-up X-ray film, additional MRI examination was performed.
Statistical analysis
All measurable data are presented as the mean + standard deviation. Efficacy variables were analyzed using a mixed-effects model repeated-measures (MMRM) approach. Data from all patients were used in the primary efficacy analysis according to intent-totreat principles with no imputation for missing data. The data were plotted using SPSS (version 26.0) GraphPad Prism (version 6.02). One-way analysis of variance followed by Student’s t-tests were employed to identify the statistical differences among the groups. All experiments were performed in triplicate, and P < 0.05 was deemed to indicate statistically significant differences.
Result
Concentration of L-Gln in the synovial tissue of the rat knee joint increased after Gavage administration
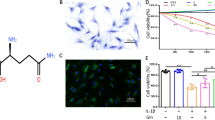
To investigate the metabolism of L-Gln in vivo, we measured the L-Gln concentrations in the serum and synovial tissue of knee joints at0, 10, 30, 60, 120, and 180 min after the rats were administered 250 mg/kg L-Gln via gavage (Fig. 1A). Ten minutes after administering L-Gln, its concentrations in the serum and synovial tissue were higher than the baseline, peaked between 30 and 60 min, then gradually declined and approached baseline levels after 180 min. L-Gln concentration in the serum and synovial tissue of the knee joint was lower in the rats of the ACLT + MMx group compared with those in the sham group. Although the metabolism of L-Gln was similar between the sham and OA groups, the concentration of L-Gln in the serum and synovial tissue of the knee joint of OA rats appeared to decrease more rapidly after reaching its peak (Fig. 1B and C).
Comparison of the therapeutic effects of L-Gln, GS, and CXB in the rat model of knee OA
Serological analysis
Starting in the fifth week post-ACLT + MMx surgery, we administered rats with L-Gln (250 mg/kg/d), GS (130 mg/kg/d), or CXB (17 mg/kg/d) via gavage for eight weeks (Fig. 1D). Subsequently, we found that the serum L-Gln concentration in the rats of the OA group was significantly lower than that in the rats of the sham group (P = 0.0062). Intragastric administration of L-Gln resulted in increased serum L-Gln concentrations in rats (P = 0.0105), whereas administration of GS or CXB did not prevent the decrease in serum L-Gln concentration caused by arthritis (Fig. 1E). In addition, following L-Gln treatment, the serum CTX-II (P = 0.0047) and COMP (P = 0.0001) concentrations were significantly decreased in the rats, similar to the effect of GS treatment. However, compared to L-Gln treatment, CXB treatment led to higher concentrations of CTX-II (P = 0.0093) and COMP (P = 0.0018) in the rats (Fig. 1F and G). Regarding serum inflammatory markers, we found that only GS treatment reduced serum IL-8 concentrations in OA rats (P = 0.0006) (Fig. 1H).
L-Gln reduces serum cartilage degradation markers in rats with OA. A Experimental process of drug distribution and metabolism in rats. B, C Serum and synovium L-Gln concentration in rats (n = 5 per group). D Experimental protocol for comparative efficacy assessment of three drugs in OA rats. E–H Serum L-Gln, CTX-II, COMP, and IL-8 concentration in rrats at 12 weeks post-surgery (n = 5 per group). All data are represented as mean ± SD. ns = no significance * P < 0.05, ** P < 0.01, *** P < 0.001.
Histological examination
The anatomical observation of the knee joint showed that at 12 weeks after receiving ACLT + MMx surgery, the cartilage surface of the right knee in rats was rough and dark in color, and the cartilage was significantly damaged, while the thickness of the cartilage increased and the surface became smoother after L-Gln and the two drugs treatment (Fig. 2A). HE staining showed that the cartilage surface of the control group was smooth and flat, the number of chondrocytes was normal, and the tide line was complete. In the OA model group, the cartilage was defective, the number of chondrocytes was significantly increased, the tide line was blurred, and synovial tissue hyperplasia was seen. After treatment with three drugs, the manifestations of osteoarthritis were reduced (Fig. 2B). Safranin O-Fast green staining was performed to further evaluate the loss of proteoglycan in cartilage. As shown in Fig. 2B, the knee cartilage of rats was severely damaged in the 12th week post-ACLT + MMx surgery; however, L-Gln and GS treatment effectively mitigated cartilage destruction caused by OA and restored the levels of proteoglycan in cartilage. Compared to those in the L-Gln groups, rats in the CXB group exhibited more cartilage damage. The OARSI scores further validated the aforementioned results. The OARSI scores of the rats in the L-Gln and GS groups were significantly lower than those of the OA group, whereas the OARSI scores of the rats in the CXB group were only slightly decreased (Fig. 2D). Immunohistochemical staining of the right knee joint of rats showed that the expression of MMP13 (L-Gln: P = 0.0014, GS: P = 0.0101, CXB: P = 0.0005) in the treatment group was lower than that in the OA group, and the expression of aggrecan(L-Gln: P = 0.0034, GS: P = 0.049, CXB: P = 0.008) and COLII (L-Gln: P = 0.0019, GS: P = 0.0213, CXB: P = 0.0259) was higher than that in the OA group. In comparison between the three treatment groups, aggrecan expression was higher in the L-Gln group than in the other two groups (GS: P = 0.0396, CXB: P = 0.0188). The expression of COL II was higher than that in the GS group (P = 0.0106) and the same as that in the CXB group (P = 0.3316). However, the expression of MMP13 in the GS group (P = 0.8915) and the CXB group (P = 0.7869) was not significantly different from that in the L-Gln group (Fig. 2C, E–G).
L-Gln, GS and CXB attenuated cartilage degradation in ACLT rats. A photos of rat femur and tibial plateau at 8 weeks after treatment. B Safranin O-Fast Green Staining and H-E Staining of rat knee samples. C immunohistochemistry staining against Aggrecan, Collagen II and MMP13 of rat knee samples. D OARSI scores of rats at 12 weeks after surgery. E–G Determination of grayscale values of immunohistochemistry results. All data are represented as mean ± SD. ns = no significance * P < 0.05, ** P < 0.01, *** P < 0.001.
Imaging examination
To investigate the effects of L-Gln and the two drugs on the osteophytes in the knee joint and subchondral bone of the tibia plateau in OA rats, we performed micro-CT scanning and three-dimensional reconstruction of the right knee joint of rats treated for eight weeks. As shown in Fig. 3A, compared to those in the sham group, rats in the OA group exhibited an increase in osteophyte formation in the knee joint and significant local subchondral osteosclerosis and extensive bone loss in the tibia plateau. The administration of L-Gln and the two drugs notably improved this condition.
The analysis of BV/TV, Tb. N and Tb. Sp of the subchondral bone of the tibia validated the aforementioned results. OA resulted in a significant decrease in BV/TV (P = 0.0169) and Tb. N (P = 0.0017) of the tibial plateau of the knee joint in rats, and an increase in trabecular bone separation (P = 0.0076). Compared with the OA group, the rats in the L-Gln group (BV/TV: P = 0.0074, Tb. N: P = 0.0089, Tb. Sp: P = 0.0087), the GS group (BV/TV: P = 0.0122, Tb. N: P = 0.056, Tb. Sp: P = 0.086) and the CXB group (BV/TV: P = 0.0253, Tb. N: P = 0.0122, Tb. Sp: P = 0.0486) had increased bone mass and trabecular bone number in the tibial plateau of rats. (Fig. 3B–D). However, there are no significant difference between the effect of L-Gln on subchondral bone mass of tibial plateau and that of the two drugs.
Micro-CT analysis of L-Gln, GS and CXB effects on the knees of OA rats. A CT images and three-dimensional reconstruction of the right knee joint in rats B–D BV/TV, Tb. N and Tb. Sp were measured in the subchondral bone of of the rat tibial plateau at 12 weeks after surgery (n = 3 per group). All data are represented as mean ± SD. ns = no significance * P < 0.05, ** P < 0.01, *** P < 0.001.
Clinical efficacy
Disposition of the patients
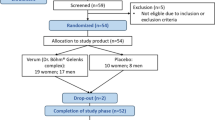
This study was conducted from October 2021 to November 2022. A total of 99 patients were randomly treated with L-Gln (n = 37), GS (n = 33), or CXB (n = 29). Subsequently, 93 patients were followed up over a period of six months [ L-Gln group, n = 37 (100%); GS group, n = 29 (87.8%); CXB group, n = 27 (93.1%)] (Fig. 4).
Flow diagram of clinical trials.
Demographic and baseline clinical characteristics of the patients
The demographic and baseline clinical characteristics of the patients in each treatment group were generally comparable (Table 1). The average age of the patients was 57.6, female patients accounted for 80.8% of the total patients. The K/L severity grade of 57% patients was 1 and the rest was 2. Most of the patients (73.7%) had not received drugs or had solely received informal drug treatment.
Efficacy
Using a mixed linear model analysis, statistically significant differences were found between the four time points for both the WOMAC pain subscale scores (F = 43.705, P < 0.001) and the WOMAC function subscale scores (F = 31.534, P < 0.001). The WOMAC pain and physical function scores of the three groups of patients were significantly lower than the baseline levels (Table 2; Fig. 5A, B). The differences in WOMAC pain scores and WOMAC function scores at three time points (4 weeks, 12 weeks, and 24 weeks) were calculated by subtracting the baseline scores. After normality tests, the differences at all time points followed a normal distribution, One-way ANOVA was used to compare the differences in scores between the three groups at the three time points from the baseline scores. Among all the differences, the L-Gln group had the highest values, but the differences between the three groups were not statistically significant (Table 3). We also assessed whether the differences in WOMAC pain and function scores met the minimal clinically important difference (MCID). As shown in Table 4, at 24 weeks, 70.30% of patients in the L-Gln group had differences in WOMAC pain scores from baseline that reached the MCID, compared to 52.00% in the GS group and 55.56% in the CXB group. For WOMAC function scores at the same time point, 73.00% of patients in the L-Gln group achieved the MCID, compared to 75.90% in the GS group and 59.30% in the CXB group.
We also compared the changes in Lequesne scores among the three groups of patients. The mixed linear model analysis showed that the differences in Lequesne scores between the four time points were statistically significant (F = 48.475, P < 0.001). Throughout the follow-up period, the Lequesne scores of OA patients treated with L-Gln and the other two drugs were significantly lower than their baseline levels (Table 2; Fig. 5C). Additionally, the effects of L-Gln at any time point were not significantly different from those of the other two drugs (Table 3).
Analyses were based on a mixed-effects model repeated-measures approach. 95% CI = 95% confidence interval.
Change from baseline in WOMAC pain score (A), WOMAC function score (B) and Lequesne score (C).
Safety
During the study period, the occurrence of Treatment-Emergent Adverse Events (TEAEs) was recorded and analyzed. It was noted that 4 patients in the GS group and 2 patients in the CXB group discontinued the study due to severe gastrointestinal adverse reactions. In contrast, no adverse reactions related to gastrointestinal issues were reported in the L-Gln group.
Discussion
OA is a notable degenerative disease of the joint with a high incidence rate; OA imposes a remarkable economic burden on society1,2,3,4. Despite following international guidelines for the treatment of OA, the prevention of its progression remains elusive8. Commonly used drugs in clinical practice, such as GS and CXB, primarily alleviate symptoms such as knee joint pain and stiffness in patients with OA30. Chondrocyte apoptosis and cartilage matrix degradation induced by inflammation play central roles in the pathogenesis of OA31,32. Oral administration of L-Gln, an anti-inflammatory amino acid, has been used as a therapeutic approach for various diseases12,33,34. Our previous study elucidated that L-Gln effectively inhibits the degradation of the chondrocyte matrix, thereby treating OA25.
In this study, we found that the administration of L-Gln via gavage effectively increases its concentration in the synovial tissue of the rat knee joint and that its concentration peaks within 60 min. This finding provides empirical evidence for the potential application of L-Gln for the treatment of OA. However, 180 min after L-Gln administration, the concentration of L-Gln in both the serum and synovial tissue reverted to their initial baseline levels. Therefore, extending the elimination half-life of L-Gln through suitable methods may enhance the therapeutic effect of L-Gln, making this a meaningful direction for future work.
The disruption of the dynamic balance of extracellular matrix degradation is a notable feature of the pathogogenesis of OA35. In this study, safranin O-fast green staining of tissue sections of the knee joints of rats revealed that L-Gln significantly restored the proteoglycan levels in cartilage. Immunohistochemistry validated an increase in aggrecan and col-2. In addition, MMP13, which plays a crucial role in the degradation of extracellular matrix36, was significantly decreased in the knee joints following L-Gln treatment. The reduction of CTX-IIand COMP, which are two types of cartilage degradation products, further validated that L-Gln reduces cartilage damage37,38. L-Gln has demonstrated a substantial impact in alleviating cartilage damage associated with OA. When compared to GS and CXB, it exhibits a slight advantage in some indicators of therapeutic effectiveness.
Osteophyte, which is a bone growth product derived from mesenchymal stem cells (MSCs) and limits joint movement and causes pain, is a notable sign of OA39,40,41. The three-dimensional reconstruction of rat knee joints provides evidence that L-Gln has the potential to decrease the development of knee joint osteophytes and enhance the subchondral bone mass in the tibia. However, it should be noted that L-Gln does not exhibit a significant advantage in this particular aspect.
In the context of our phase II study involving patients with early OA, our findings pertaining to the improvement of pain and function warrant the contextualization of existing literature on pain management treatments. Several studies have shown that, compared to placebo, acetaminophen, CXB, naproxen, ibuprofen, and diclofenac reduce the pain score from − 0.45 to −2.4542,43. There exists some discourse regarding the utilization of glucosamine for the treatment of OA. The European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases recommends the use of acetaminophen in the short term and at a low dosage, drug-grade glucosamine, and chondroitin sulfate, whereas the OARSI strongly advises against their use (including all glucosamine or chondroitin preparations)30,44,45,46. This study revealed that L-Gln, GS, and CXB all significantly reduce patients’ WOMAC pain and physical function scores, as well as Lequesne scores. Despite the lack of significant differences in efficacy among the three treatments. Notably, of the 99 participants in this study, 26 had previously been treated with NSAIDs or other medications but did not exhibit a satisfactory therapeutic response. During the subsequent follow-up, we paid special attention to these patients. 11 of these subjects were assigned to the L-Gln group. It is worth noting that 8 of these patients had significantly reduced WOMAC pain subscale scores and WOMAC function subscale scores after 12 weeks of treatment with L-Gln, which differed more than MCID from baseline. However, it is important to conduct larger-scale studies to provide additional evidence and validate these findings.
In this study, patients exhibited good tolerance to L-Gln, as evidenced by the reduced overall incidence of TEAEs compared to the two drugs and no cases of discontinuation of L-Gln due to TEAEs. In addition, the incidence of gastrointestinal diseases in patients who were administered L-Gln was significantly lower, which may be associated with glutamate receptors in the intestine that regulate gastric motility and mucosal function47.
The limitations of this study should be acknowledged. First, the relatively small sample size may limit the generalizability of the findings to broader populations. Second, the lack of long-term follow-up data prevents an assessment of the sustainability of the observed treatment effects over time. Third, although measures were taken to ensure patient compliance, potential variability in adherence to treatment protocols could have influenced the outcomes. Finally, the study did not incorporate certain biomarkers or advanced imaging techniques in the post-treatment evaluation of all participants, which could have provided deeper insights into the mechanisms and efficacy of the treatments. These limitations should be considered when interpreting the results and designing future studies.
Conclusion
In this study, we compared the therapeutic effects of L-Gln, GS, and CXB on OA from two aspects: pathological alterations and clinical efficacy. Our study establishes that L-Gln shows equivalent or superior efficacy in certain indicators compared to GS and CXB in terms of pathological progression and clinical efficacy in OA. However, L-Gln exhibits a lower occurrence of gastrointestinal adverse reactions. These findings indicate that L-Gln holds promise as a potential new clinical option for treating osteoarthritis.
Data availability
The authors declare that all the data supporting the findings of this study are available within the article and its Supplementary Information files.
Abbreviations
- OA:
-
Osteoarthritis
- L-Gln:
-
L-glutamine
- MMP13:
-
Matrix metalloproteinase-13
- GS:
-
Glucosamine sulfate
- CXB:
-
Celecoxib
- SD rats:
-
Sprague-Dawley rats
- ACLT:
-
Anterior Cruciate Ligament Transection
- MMx:
-
Medial Meniscectomy
- COMP:
-
Cartilage Oligomeric Matrix Protein
- IL-8:
-
Interleukin-8
- CTX-II:
-
C-telopeptide of Type II collagen
- BV/TV:
-
Bone volume to the tissue volume
- Tb. N:
-
Trabecular Numbe
- Tb. Sp:
-
Trabecular Separation
- HE:
-
Hematoxylin and eosin
- COLII:
-
Collagen II
- OARSI:
-
Osteoarthritis Research Society International
- K/L:
-
Kellgren/Lawrence
- WOMAC:
-
Western Ontario and McMaster Universities OA Index
- MCID:
-
Minimal clinically important difference
- TEAEs:
-
Treatment emergency adverse events
- MRI:
-
Magnetic resonance imaging
- MMRM:
-
Mmixed-effects model repeated-measures
References
Abramoff, B. & Caldera, F. E. Osteoarthritis. Med. Clin. N. Am. 104, 293–311 (2020).
Sharma, L. Osteoarthritis of the knee. N Engl. J. Med. 384, 51–59 (2021).
Lotz, M. K. & Caramés, B. Autophagy and cartilage homeostasis mechanisms in joint health, aging and OA. Nat. Rev. Rheumatol. 7, 579–587 (2011).
Glyn-Jones, S. et al. Osteoarthritis. Lancet 386, 376–387 (2015).
Bannuru, R. R. et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthr. Cartil. 27, 1578–1589 (2019).
McGrory, B. et al. The American academy of orthopaedic surgeons evidence-based clinical practice guideline on surgical management of osteoarthritis of the knee. J. Bone Jt. Surg. 98, 688–692 (2016).
Hunter, D. J. Pharmacologic therapy for osteoarthritis—the era of disease modification. Nat. Rev. Rheumatol. 7, 13–22 (2011).
Guo, H. et al. Quercitrin alleviates cartilage extracellular matrix degradation and delays ACLT rat osteoarthritis development: an in vivo and in vitro study. J. Adv. Res. 28, 255–267 (2021).
Cruzat, V., Macedo Rogero, M., Noel Keane, K., Curi, R. & Newsholme, P. Glutamine: metabolism and immune function, supplementation and clinical translation. Nutrients 10, 1564 (2018).
Roth, E. Nonnutritive effects of glutamine. J. Nutr. 138, 2025S-2031S (2008).
Curi, R. et al. Molecular mechanisms of glutamine action. J. Cell. Physiol. 204, 392–401 (2005).
Choi, K. et al. The effect of oral glutamine on 5-fluorouracil/leucovorin-induced mucositis/stomatitis assessed by intestinal permeability test. Clin. Nutr. 26, 57–62 (2007).
Peterson, D. E., Jones, J. B. & Petit, R. G. Randomized, placebo-controlled trial of Saforis for prevention and treatment of oral mucositis in breast cancer patients receiving anthracycline-based chemotherapy. Cancer 109, 322–331 (2007).
Niihara, Y. et al. A phase 3 trial of L -Glutamine in sickle cell disease. N Engl. J. Med. 379, 226–235 (2018).
Senkal, M. Early enteral gut feeding with conditionally indispensable pharmaconutrients is metabolically safe and is well tolerated in postoperative cancer patients? A pilot study*1. Clin. Nutr. 23, 1193–1198 (2004).
Garlick, P. J. Assessment of the safety of glutamine and other amino acids. J. Nutr. 131, 2556S–2561S (2001).
Kronenberg, H. M. Developmental regulation of the growth plate. Nature 423, 332–336 (2003).
Long, F. & Ornitz, D. M. Development of the endochondral skeleton. Cold Spring Harbor Perspect. Biol. 5, a008334–a008334 (2013).
Stegen, S. et al. Glutamine metabolism controls chondrocyte identity and function. Dev. Cell. 53, 530–544e8 (2020).
Tonomura, H. et al. Glutamine protects articular chondrocytes from heat stress and NO-induced apoptosis with HSP70 expression. Osteoarthr. Cartil. 14, 545–553 (2006).
Piepoli, T. et al. Glutamate signaling in chondrocytes and the potential involvement of NMDA receptors in cell proliferation and inflammatory gene expression. Osteoarthr. Cartil. 17, 1076–1083 (2009).
Arra, M. et al. Glutamine metabolism modulates chondrocyte inflammatory response. eLife 11, e80725 (2022).
Anderson, J. R. et al. 1 H NMR metabolomics identifies underlying inflammatory pathology in osteoarthritis and rheumatoid arthritis synovial joints. J. Proteome Res. 17, 3780–3790 (2018).
Akhbari, P. et al. Can joint fluid metabolic profiling (or metabonomics) reveal biomarkers for osteoarthritis and inflammatory joint disease? A systematic review. Bone Jt. Res. 9, 108–119 (2020).
Ma, X. et al. L-G Lutamine alleviates osteoarthritis by regulating lncRNA- NKILA expression through the TGF-β1/SMAD2/3 signalling pathway. Clin. Sci. 136, 1053–1069 (2022).
Zhang, Z. et al. Guidelines for the diagnosis and treatment of osteoarthritis in China (2019 edition). Ann. Transl Med. 8, 1213–1213 (2020).
Kellgren, J. H. & Lawrence, J. S. Radiological assessment of osteo-arthrosis. Ann Rheum Dis. 16, 494–502 (1957).
Angst, F., Aeschlimann, A. & Stucki, G. Smallest detectable and minimal clinically important differences of rehabilitation intervention with their implications for required sample sizes using WOMAC and SF-36 quality of life measurement instruments in patients with osteoarthritis of the lower extremities. Arthr. Rhuem. 45, 384–391 (2001).
Singh, J. A., Noorbaloochi, S., MacDonald, R. & Maxwell, L. J. Chondroitin for osteoarthritis. Cochrane Database Syst Rev. ;2016. (2015).
Hochberg, M. C. et al. Combined chondroitin sulfate and glucosamine for painful knee osteoarthritis: a multicentre, randomised, double-blind, non-inferiority trial versus celecoxib. Ann. Rheum. Dis. 75, 37–44 (2016).
Sokolove, J. & Lepus, C. M. Role of inflammation in the pathogenesis of osteoarthritis: latest findings and interpretations. Ther. Adv. Musculoskelet. 5, 77–94 (2013).
Scanzello, C. R. Role of low-grade inflammation in osteoarthritis. Curr. Opin. Rheumatol. 29, 79–85 (2017).
Cruzat, V. F., Pantaleão, L. C., Donato, J., de Bittencourt, P. I. H. & Tirapegui, J. Oral supplementations with free and dipeptide forms of l-glutamine in endotoxemic mice: effects on muscle glutamine-glutathione axis and heat shock proteins. J. Nutr. Biochem. 25, 345–352 (2014).
Wang, W-S. et al. Oral glutamine is effective for preventing oxaliplatin-induced neuropathy in colorectal cancer patients. Oncologist 12, 312–319 (2007).
Xia, B., Di Chen, Zhang, J., Hu, S., Jin, H. & Tong, P. Osteoarthritis pathogenesis: A review of molecular mechanisms. Calcif Tissue Int. 95, 495–505 (2014).
Fosang, A. J., Last, K., Knäuper, V., Murphy, G. & Neame, P. J. Degradation of cartilage Aggrecan by collagenase-3 (MMP-13). FEBS Lett. 380, 17–20 (1996).
Christgau, S. et al. Collagen type II C-telopeptide fragments as an index of cartilage degradation. Bone 29, 209–215 (2001).
Lohmander, L. S., Saxne, T. & Heinegard, D. K. Release of cartilage oligomeric matrix protein (COMP) into joint fluid after knee injury and in osteoarthritis. Ann. Rheum. Dis. 53, 8–13 (1994).
Van Der Kraan, P. M. & Van Den Berg, W. B. Osteophytes: relevance and biology. Osteoarthr. Cartil. 15, 237–244 (2007).
Schelbergen, R. F. P. et al. Alarmins S100A8/S100A9 aggravate osteophyte formation in experimental osteoarthritis and predict osteophyte progression in early human symptomatic osteoarthritis. Ann. Rheum. Dis. 75, 218–225 (2016).
Kaneko, H. et al. Synovial Perlecan is required for osteophyte formation in knee osteoarthritis. Matrix Biol. 32, 178–187 (2013).
Stam, B. Efficacy of Etoricoxib, celecoxib, lumiracoxib, non-selective NSAIDs, and acetaminophen in osteoarthritis: a mixed treatment comparison. TORJ 6, 6–20 (2012).
Machado, G. C. et al. Efficacy and safety of Paracetamol for spinal pain and osteoarthritis: systematic review and meta-analysis of randomised placebo controlled trials. BMJ 350 (mar31 2), h1225–h1225 (2015).
Arden, N. K. et al. Non-surgical management of knee osteoarthritis: comparison of ESCEO and OARSI 2019 guidelines. Nat. Rev. Rheumatol. 17, 59–66 (2021).
Reginster, J-Y-L., Bruyere, O. & Cooper, C. Different glucosamine sulfate products generate different outcomes on osteoarthritis symptoms. Ann. Rheum. Dis. 77, e39–e39 (2018).
Clegg, D. O. et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N. Engl. J. Med. 354, 795–808 (2006).
Tsai, L. H. Function of GABAergic and glutamatergic neurons in the stomach. J. Biomed. Sci. 12, 255–266 (2005).
Funding
This work was supported by the Anhui Provincial Department of Science and Technology [Grant number 202007d07050007 and 2208085MH214. Fund recipient: Wendan Cheng] and Anhui Medical University [Grant number: 2021xkj041. Fund recipient: Chun Zhang]. All funds are managed by the Finance Department of Anhui Medical University and the Second Affiliated Hospital of Anhui Medical University.
Author information
Authors and Affiliations
Contributions
Author contributions: Conceptualization, Haoran Yu; Data curation, Zhongyao Hu, Changming Wang, Chen Wang and Haoran Yu; Formal analysis, Chen Wang; Funding acquisition, Chun Zhang and Wendan Cheng ; Investigation, Zhongyao Hu, Changming Wang, Chen Wang, Junyan He, Yiqun Yan, Zelin xu, Yangmang Yu, Ya Yu, Huan Cheng, Lei Liu and Miao Tang; Methodology, Zhongyao Hu, Haoran Yu, Juehua Jing and Wendan Cheng ; Project administration, Wendan Cheng ; Supervision, Haoran Yu and Juehua Jing; Validation, Changming Wang; Visualization, Zhongyao Hu and Chen Wang; Writing – original draft, Zhongyao Hu; Writing – review & editing, Wendan Cheng . Zhongyao Hu, Changming Wang, Chen Wang are considered to be co-first authers
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All animal experiments performed in this study were approved by the Institutional Animal Care and Use Committee of Anhui Medical University (approval number LLSC20231140). The clinical trial was approved by the Ethics Committee of the Second Affiliated Hospital of Anhui Medical University (approval number YX2021-072). We conducted a retrospective registration for this study on 09/10/2023. Name of the registry: Chinese Clinical Trial Registry. URL: https://www.chictr.org.cn/. registration number: [ChiCTR2300076450].
Consent for publication
All authors agree to publish the manuscript in this journal.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hu, Z., Wang, C., Wang, C. et al. The comparative efficacy of L-glutamine, celecoxib, and glucosamine sulfate in osteoarthritis management. Sci Rep 15, 8992 (2025). https://doi.org/10.1038/s41598-025-93357-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93357-y
Keywords
This article is cited by
-
Formulation and Optimisation of Mucoadhesive Nasal Powder of Celecoxib for Management of Pain Associated with Osteoarthritis
Journal of Pharmaceutical Innovation (2025)