Abstract
Stringent emission regulations and the depletion of conventional fuel sources drive research on green fuels, additives, and the optimization of fuel injection and exhaust gas recirculation. This study analyzes the impact of butanol additives in diesel and cashew shell liquid biodiesel (CSLB) blends under optimal operating conditions. CSLB was produced with an 85.43% yield from waste cashew nut shell liquid under optimal conditions: a methanol/CSL molar ratio (MR) of 20:1, a process temperature (PT) of 70 °C, and a 4 wt% industrial waste-derived heterogeneous catalyst (IC), using the desirability function approach in the RSM-CCD model. The catalyst was characterized using XRD, FTIR, and BET analyses to confirm its catalytic activity. Engine performance improvements were achieved with specific modifications, including 4° CA timing retardation, 15% split injection, and a 20% exhaust gas recirculation rate when using CSLB blends. In common rail direct injection (CRDI) experimental investigations, diesel and CSLB blends were combined with butanol additives (2.5%, 5%, and 10%) and compared to the baseline test. Incorporating 10% butanol, with its higher latent heat, resulted in a lower combustion temperature, reducing NOx emissions by 47.09% in CSLB10. Additionally, the additive’s lower viscosity and higher oxygen content enhanced atomization, reducing CO (33%) and smoke (23.02%) emissions. However, a slight increase in CO2 (8.92%) and a decrease in HC emissions (27.14%) were observed in CSLB10. Improved combustion characteristics, reflected in higher peak pressure and heat release rate, resulted in a 4.75% increase in brake thermal efficiency and a 13.92% reduction in brake-specific energy consumption compared to ideal conditions. Overall, this study explores the impact of butanol additives on the performance and emissions of CRDI engines fuelled with CSLB blends derived from waste cashew nut shell liquids, providing insights for sustainable fuel optimization.
Similar content being viewed by others
Introduction
The shift towards alternative fuels from biomass and synthetic sources is driven by strict emission regulations and the need to reduce fossil fuel use in transportation. Biodiesel from non-edible biomass is gaining popularity in developing countries like India1,2. It offers a similar calorific value, a higher cetane rating, more O2, and lower sulfur and aromatics than diesel3. It is also renewable, non-toxic, and eco-friendly, derived from readily available resources that can reduce smoke emissions, hydrocarbons (HC), and carbon monoxide (CO)4,5. This study intends to explore the viability of cashew nut shell waste-derived liquid (CSL) as an inexpensive alternative fuel. Cashew trees are most often cultivated along India’s coast, and within each cashew nut shell is a thick reddish-brown liquid called CSL. The transesterification procedure can be employed to convert CSL into biodiesel. A calorific value and enhanced miscibility with diesel are some of CSL biodiesel’s positive traits6. However, CSL biodiesel makes achieving a constant air-fuel combination and efficient fuel atomization challenging because of its higher density and viscosity. Recent research7,8 recommends injection timing and split injection modifications to address this drawback, promoting effective miscibility, amended combustion, and minimizing emissions.
Response surface methodology (RSM) is a statistical and mathematical framework for modelling and optimizing response variables (input or output) influenced by multiple independent factors. It facilitates the identification of optimal process conditions by systematically adjusting control parameters within predefined constraints9. According to the literature, Ghorbani-Choghamarani et al.10 synthesized a novel molybdenum-based metal-organic framework (Mo-MOF) via a solvothermal method, exhibiting high catalytic efficiency for biodiesel production. Characterized by various techniques, the Mo-MOF had a 56 m2/g surface area, 300 °C thermal stability, and a rod-shaped configuration. It achieved 95% biodiesel yield under optimal conditions (60 °C, 13:1 methanol/oil ratio, 300 mg catalyst), with the fuel meeting ASTM standards. Wang11 produced biodiesel from spent coffee grounds oil using an innovative activated carbon-based green catalyst synthesized from pomegranate and orange peels. The maximum yield of 95.24% was obtained under optimized conditions: a molar ratio of 11:1, 2.60 wt% catalyst loading, 30.80 min of ultrasonic treatment, and a reaction temperature of 60 °C, as determined by RSM. Rao et al.12 developed a biochar-based heterogeneous catalyst (PJBC) from waste Prosopis juliflora biomass using RSM-genetic algorithm optimization, achieving a 46.31% yield under 790 °C, 60 min, and 60 mg biomass conditions. The various characterization techniques confirmed its catalytic activity. PJBC catalyzed waste Trichosanthes cucumerina seed biodiesel (WTSB) production via transesterification, reaching 97.42% yield. The WTSB met ASTM D6751 standards, proving its viability as a diesel alternative. Dubey et al.13 optimized transesterification parameters for converting waste cooking oil (WCO) into biodiesel. The process achieved a 92% biodiesel yield under 60 °C reaction temperature, 0.75 wt% catalyst loading, 80 min process time, and a 5:1 methanol/WCO molar ratio, utilizing RSM for optimization. Farouk et al.14 synthesized a magnesium oxide (MgO) nano-catalyst via the sol-gel method for waste cooking oil (WCO) biodiesel production through transesterification. Characterization techniques on MgO confirmed its high thermal stability, 67.13 nm particle size, and 60.2 m2/g surface area. Using RSM, the optimal conditions (7:1 methanol/WCO ratio, 1 wt% MgO catalyst, 50 °C, 60 min) achieved 92% biodiesel yield. The 95.3% fatty acid methyl ester content and 88% conversion efficiency met ASTM D6751 standards, validated through GC-MS and FTIR analysis, demonstrating the MgO catalyst’s efficiency for sustainable biodiesel production. Sun et al.15 examined the Fe3O4@UiO-66-NH2 catalyst for biodiesel synthesis from restaurant waste oil (RWO). Utilizing RSM, the study achieved a 98.72% biodiesel yield under optimized conditions: methanol/RWO ratio of 11.4:1, 8.84 min reaction time, 3.27% catalyst loading, and 723.4 rpm mixing speed.
Split injection methods have been successful in aiding complete combustion and achieving a homogeneous fuel mixture. Split injection with WCO biodiesel led to decreased NOx, PM emissions, and smaller mean particle diameters, according to Ganesan et al.16. How et al.17 independently suggested the conflict between NOx and smoke emissions might be resolved by adjusting injection timing and including split injection in biodiesel blends. When employing CSL biodiesel blends, Senthil Kumar and Thirumalini18 noticed lower NOx, smoke, and CO emissions by combining a 15% split injection with a 4° retarded CA. Another method used to reduce NOx emissions is exhaust gas recirculation (EGR). The utilization of W20P20 blends up to 20% EGR was shown to be a reduction in NOx and soot emissions, according to Venugopal et al.19. A mix of split injection, higher injection pressure, and exhaust gas recirculation may effectively reduce emissions, according to Rami Reddy et al.20. Using canola oil biodiesel blends, Ge et al.21 examined the effects of EGR and pilot injection timing on engine performance. It was found that EGR modestly increased brake-specific fuel consumption (BSFC) while dramatically reducing NOx emissions. With biodiesel blends, Ayhan et al.22 examined the effect of EGR on engine performance and emission characteristics. It was observed that performance parameters were unaffected. At the same time, there was a notable decrease in NOx, CO, and smoke emissions when a combination of delayed injection timing, 15% split injection, and 20% EGR was used. According to recent studies, incorporating EGR with fuel additives and split injection techniques may increase engine performance and lower emissions23,24,25.
Methanol, ethanol, and butanol are common alcohol additives that improve engine performance. Butanol is a sustainable, less corrosive, and less volatile alternative with a lower auto-ignition temperature. Compared to other alcohols, butanol has a higher calorific value, cetane rating, flash point, and lower heat of vaporization26. Additionally, butanol has superior cold flow characteristics, blending stability, and better miscibility. Its characteristics are similar to diesel fuel, making it a desirable additive. Due to this, butanol was chosen for the current experimental investigation of its effects on engine performance traits27. Nanthagopal et al.28 examine 1-pentanol and 1-butanol as additives to Calophyllum Inophyllum biodiesel in a single-cylinder diesel engine using six binary blends. Results showed lower brake thermal efficiency (BTE) (27.60% for biodiesel vs. 31.56% for diesel) and increased BSFC (0.34 to 0.49 kg/kWh) with higher alcohol content. NOx emissions dropped by 10 to 23%, while CO, HC, and smoke emissions decreased by up to 35%. Peak in-cylinder pressure declined from 79.3 to 70.5 bar. The study confirms 1-pentanol and 1-butanol as partial diesel/biodiesel substitutes, improving emissions with slight efficiency trade-offs. The impact of butanol addition to diesel experimented with high-speed DI diesel engine parameters was studied by Tian et al.29. The results indicated decreased NOx, CO, and smoke emissions and a minor increase in BSFC and BTE compared to diesel. Yusri et al.30 investigated a diesel engine’s variables running with butanol-diesel blends. It was noted that lower cylinder pressure, a slower heat release rate (HRR), a slower rate of pressure rise, and a longer ignition delay. A reduction in NOx emissions of 34.7%, CO emissions of 30.9%, and HC emissions of 30.1% was achieved by blending 15% butanol with diesel. Singh et al.31 studied the impact of n-butanol as an additive in eucalyptus biodiesel-diesel blends on diesel engine performance and emissions. Engine tests with B20, B100, and n-butanol-blended fuels (B20 with 5%, 10%, and 15% n-butanol) at 1500 rpm revealed slight increases in brake power and BSFC, while BTE decreased. Emission analysis showed CO and HC reductions of up to 46%, though NOx and CO2 emissions were higher than neat diesel. However, adding n-butanol reduced NOx emissions by up to 25.16% due to its cooling effect. Zhu et al.32 analyzed the effect of n-butanol as an additive in diesel fuel on particulate matter (PM) emissions in a common-rail diesel engine using a pilot and main injection strategy. Three fuel blends were tested at 30% and 60% engine loads with 40 MPa and 60 MPa injection pressures. Experimental results showed NOx emissions reduced by 25% due to lower combustion temperatures from n-butanol’s lower heating value, while PM emissions dropped by 69%, with D50B50 under low load exhibiting the lowest emissions. Killol et al.33 thoroughly investigated the engine efficiency and emission trends of single-cylinder DI engines. They concluded that adding butanol to Karanja oil biodiesel improved BTE by 2.9% and decreased HC, CO and smoke emissions compared to biodiesel under full load. Additionally, it has been claimed that butanol-biodiesel blends have lower HRR with a higher value of latent heat of vaporization (LHV), so butanol addition is restricted to 10%. Jeevahan et al.34 found decreased BTE, BSFC, and lower NOx, CO, and HC emissions when employing waste cooking oil biodiesel and butanol blends. EL-Seesy et al.35 investigated the impact of n-butanol as an additive in diesel-jojoba oil blends on a CI engine’s combustion and emission characteristics under various loads. Jojoba oil’s high viscosity and NOx emissions were addressed by combining it with 10% n-butanol and 5–35% jojoba oil concentrations. Results showed that n-butanol reduced viscosity by up to 85% and improved combustion, with higher Pmax and HRR for blends up to 15% jojoba oil. However, Emission analysis revealed significant reductions in NOx by 50%, CO by 30%, and unburned HC by 40% compared to neat diesel.
Motivation, objective and novelty of the current research
According to the literature, CSLB is a practical alternative fuel with promising engine applications. Butanol also has many benefits and can improve biodiesel’s cold flow characteristics. Nevertheless, the performance and emission characteristics of engines using butanol additives and CSLB blends have received little attention from existing research works. As far as the available literature suggests, there has yet to be no prior research that has explored the optimization of CSLB input variables using the desirability function approach in the RSM-CCD model. Also, the CRDI engine characteristics operating on CSLB are derived from industrial waste heterogeneous catalysts with butanol additives. Additionally, most studies use a mechanically operated fuel injection, making it difficult to adjust the injection parameters accurately. The present experimental tests are carried out with butanol as an additive and CSLB blends to fill these research gaps. A research engine control unit regulates the EGR rate and injection parameters. According to the previous investigations in the literature, the ideal fuel injection settings are 10.2 degrees CA before TDC, 15% fuel injection, and 20% optimal EGR. When the CRDI engine is operated at ideal fuel injection settings and fueled with diesel and CSLB blends, emissions are reduced, and performance is marginally enhanced. This study also aims to determine the effect of adding 2.5%, 5% and 10% butanol as an additive to diesel and CSLB blends on CRDI engine characteristics under these ideal circumstances and compared with a baseline test.
Materials and methods
Preparation of industrial waste derived heterogeneous catalyst
In the latex industry, products like condoms and gloves are coated with coating powder, making them easier to unwrap. After the coating, these products are dipped in a water solution, and the powder that gets washed off becomes industrial waste. This waste, sometimes used in cement production, can also be processed into a useful dolomite catalyst (step 1). Converting this latex industry waste into a catalyst involves several stages, as presented in Fig. 1. In step 2, the waste solution, containing various chemicals and solid particles, first undergoes pre-treatment. This begins with filtration, which removes large particles and impurities, resulting in a cleaner, easier process solution. After that, neutralization is often necessary. The pH of the latex industry waste is tested, and adjustments are made to ensure the solution is at the right level for further processing, which is crucial for the catalyst preparation to proceed smoothly. After pre-treatment, the focus shifts to separating functional components in step 3. A standard method followed is precipitation, where chemicals are added to the solution to cause key ingredients, like magnesium (Mg) and calcium (Ca), to separate along with the dolomite powder. The solution undergoes centrifugation to concentrate further and isolate these materials, efficiently separating the solid components used as catalysts. Once the valuable components have been extracted, the next step 4 is calcination. During calcination, the mixture is heated to high temperatures, typically between 800 °C, to convert the carbonate forms of magnesium and calcium into their active oxide forms. This thermal treatment enhances the catalytic properties of the dolomite, preparing it for use in industrial applications. After calcination, the characterization of the dolomite catalyst is tested in step 5. These tests ensured the catalytic ability of the industrial waste. The dolomite is evaluated in transesterification in CSLB production to confirm its catalytic efficiency and ensure it performs as expected in its intended use.
Various steps involved in the preparation of industrial waste heterogeneous catalysts.
Major cultivation of cashew nut and use
India is one of the largest producers of cashew nuts, with major cultivation areas spread across states, as marked in Fig. 2. These regions provide optimal conditions for cashew cultivation, resulting in significant annual yields. However, alongside the valuable cashew nuts, the processing industry generates a substantial amount of agro-waste in the form of cashew nut shells. This byproduct, often discarded, represents an undeveloped resource with potential applications in various industries. The shells contain cashew nut shell liquid (CSL), which can be utilized in the synthesis of CSLB, among other benefits, offering an environmentally beneficial way to meet energy demands and support sustainable waste management.
Major cashew nut cultivation areas in India (map generated through https://www.mapchart.net/india.html).
Materials collection
The waste cashew nut shells in Fig. 3 were procured from local cashew processing units in Ariyalur, which are known for their abundant production of cashew nuts. These shells, a by-product of the cashew industry, were collected from processing facilities located in the Ariyalur, Cuddalore, Pudukkottai, Theni, Villupuram, and Sivaganga districts of Tamil Nadu, India, where cashew cultivation and processing are prevalent. The waste shells were chosen due to their high lignocellulosic content and the presence of cashew nut shell liquid (CSL), which contains valuable phenolic compounds. To ensure uniformity in raw material quality, the collected shells were thoroughly inspected, dried under sunlight to remove moisture, and stored in airtight containers to prevent contamination and degradation before use in subsequent CNSL-derived biodiesel (CSLB) synthesis experiments.
(a) Cashew tree (b) Cashew fruit (c) Cashew nut (d) Waste cashew nut-shell (all these images are captured by the corresponding author R. M).
CSL extraction
The collected waste cashew nut shells were used to extract CSL using a solvent extraction setup. This advanced technique involves recirculating the same hexane solvent through the extractor multiple times, as shown in Fig. 4. A 500 mL round-bottom conical flask was loaded with 100 g of waste cashew nut shells and filled with 250 mL of hexane. The solvent extraction took place for 6 h at 65 oC steady state temperature. The non-contact digital thermometer is used to measure and maintain the steady state temperature throughout the extraction process. The elliptical stirrer was used to stir the waste cashew nut shells plus hexane mixture at 500 rpm using an RPM controller. An allihn condenser tube is attached to the top of the round bottom flask for cooling water circulation from the cooling circulant. Once the extraction begins, the hexane solvent vapours are conveyed over the waste cashew nut shells liquid by using the allihn condenser tube and cooled. Siphoning action returns the cold vapours with CSL to the chamber and deflates them. During this extraction process, the liquid phase containing a mixture of hexane, CSL, and cashew nut shell waste was heated to 68 °C to evaporate the hexane, leaving behind CSL and cashew nut shell waste in a batch distillation process. After the complete evaporation of hexane, the mixture was transferred to a separating funnel. The cashew nut shell waste settled in the bottom layer, while the CSL formed the upper layer. After removing the cashew nut shell waste, the clean CSL was obtained in the flask.
Solvent extraction setup for CSL production.
CSLB production and its properties evaluation
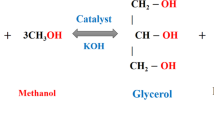
Using a transesterification process, the CSLB is extracted from agro-waste cashew nut shell-derived liquid (CSL), as represented in Fig. 5. Initially, the liquid was extracted from the waste cashew nut shells through mechanical pressing operation. After that, a magnetic stirrer is used to stir one litre of extracted CSL after being heated to 70 °C temperature. Then, 20:1 of methanol/CSL molar ratio and 4 wt% of industrial waste-derived heterogeneous catalyst are added to the CSL flask, stirring the mixture for 6 h. In the transesterification reaction, methyl ester from waste cashew nut shell forms the top layer, with glycerin forming beneath it after 12 h of settling time. A separating funnel is used to separate these layers. The contaminants are subsequently removed from the top layer of the ester by filtering and washing with distilled water. The ester is rinsed once more with sodium chloride to purify it further. The CSL methyl ester is once more rinsed with NaCl to purify it further. Now, the sanitized CSL methyl ester is vacuum-heated in an oven to eliminate remaining moisture, producing CSLB with a yield of 85.43%. The prepared CSLB is mixed through diesel fuel at 5% and 10% concentrations, denoted as CSLB5 and CSLB10 blends, due to its higher viscosity of 5.13 mm2/s and 856 kg/m3 density. Butanol is also added to diesel and CSLB blends to create a variety of test fuels, and their physio-chemical properties are listed in Table 1. These test fuel properties are compared with diesel and similar literature test fuels to confirm their suitability for CRDI engine applications.
Layout of CSLB production process.
Response surface methodology (RSM)
The statistical methodology seeks to determine the optimal combination of input elements to improve any number of outcome variables. This entails maximizing processing variables within predetermined bounds for the best final results38. In regression analysis, the least squares approach is used to estimate the relationship between input elements and outcome variables, employing either a linear or quadratic polynomial, as shown in Eq. (1).
Where Z = quadratic coefficient for result forecast; \(\:{x}_{o}\) = value of bias; \(\:{x}_{i}\) & \(\:{x}_{ij}\) = linear and interactive coefficients; n = number of factors; \(\:{a}_{c}\) & \(\:{a}_{d}\) = independent variables; C = random error.
Based on the previous literature39,40,41,42, the ranges of input variables involved in CSLB synthesis in the trans-esterification process were selected as 10:1 to 20:1 of Methanol/CSL molar ratio (MR), 60 oC to 80 oC of process temperature (PT), and 2 to 6 wt% of an industrial catalyst (IC). Table 2 details the experimental conditions, employing the CCD model to illustrate the three input factors at three distinct levels (-1 for low level, 0 for mid-level, and + 1 for high level).
Second-degree polynomial equations are frequently utilized to specify connections between elements and outcomes and simplify procedures. This process is facilitated through statistical and mathematical techniques provided by RSM. Design Expert 13.0.15 software was used to conduct the experiments, accurately recording responses and analyzing the results from a set of 20 different experiments with 6 centres per block designed using the CCD method. There are three key steps involved in CCD: (i) analysis of variance (ANOVA) to evaluate the significance of the CCD model terms, (ii) regression analysis to establish the relationship between input variables, and (iii) plotting the output results for CSLB production. The predicted CSLB yield obtained from the RSM-CCD model was compared with the experimental results, and the residual was presented in Table 3.
Experimental setup
(a) CRDI engine test setup and (b) Layout diagram.
Figure 6 (a) and (b) illustrate CRDI engine test setup and layout, respectively. The parameters of the CRDI engine, where the experiments are conducted, are given in Table 4. The twin-cylinder CRDI engine is coupled to Dynomerk controls eddy current dynamometer of 80 kW capacity to facilitate the experimentation. A sensor capable of taking 0 to 250 bar measurements and its sensitivity range − 20pC/bar is installed on the CRDI engine head to obtain Pmax signals. These signals are recorded throughout 100 cycles of operation, and the mean values are taken for analysis. A crank angle encoder acquired the CA position with a resolution of 0.1°. During the experiments, this encoder, which is installed on the engine crankshaft, allows for accurate measurement of crank angle position.
A research engine control unit (ECU) precisely controls split injection, injection timing and exhaust gas recirculation (EGR). An apparatus for gathering data (DAS) is employed to comprehensively gather temperature and pressure data across various lines, including the coolant, fuel, and exhaust lines. The temperature of the exhaust gas is recorded using a K-type thermocouple. A five-gas analyzer measures exhaust emissions such as nitrogen oxides, hydrocarbons, carbon monoxide and carbon dioxide, and an AVL smoke meter is used to obtain smoke emissions. The exhaust sample line is kept at a temperature of about 190 °C. Measurements are made three times for more accurate results, and the mean data are used for future investigation.
The experiments are conducted with diesel fuel, and CSLB blends under a range of loads at a persistent CRDI engine speed of 2200 rpm. At 14.2 degrees CA before TDC injection time with a single point fuel injection and without optimal EGR rate, the test engine parameters data are measured, and this value is considered the base reading. According to the authors’ prior experiments18, a CSLB blend-fueled engine has been found to demonstrate decreased emissions while using a combination of 15% split, 4° retarded CA (10.2 degrees CA before TDC) and 20% optimal EGR, which are regarded as the ideal running conditions. Then, under these ideal circumstances, experiments are carried out by blending diesel and CSLB blends with butanol at various concentrations (2.5%, 5%, and 10%). Investigation into the effects of butanol addition on engine performance and emission characteristics continues. Further research is being done on the impact of butanol addition on engine performance and emission characteristics. The Moffat43 approach is used to analyze errors, and Table 5 lists the percentage of uncertainty calculated for various measurements.
Results and discussion
Characterization study on industrial waste-derived heterogeneous catalyst
XRD pattern
The X-ray diffraction (XRD) pattern in Fig. 7a provides an extensive understanding of the crystalline phases of industrial waste catalyst, which is utilized as a heterogeneous catalyst in CSLB production. The peak at 30° in 2θ indicated the presence of calcium oxide (CaO), which plays an important role in the transesterification of waste cashew nut shell liquid, enabling the conversion of triglycerides into CSLB. Furthermore, many minor peaks between 30° and 65° in 2θ suggest the presence of MgO (magnesium oxide) and additional CaO phases, indicating that industrial waste powder is successfully calcined to form these oxides. MgO supports the catalyst’s thermal stability and durability. This XRD pattern reveals that the calcined industrial waste powder, which is largely constituted of CaO and MgO with minimal quartz inclusions, has the potential to serve as an efficient and long-lasting catalyst with a heterogeneous nature in CSLB production.
(a) XRD pattern of industrial waste catalyst. (b) FTIR analysis of industrial waste catalyst. (c) BET analysis of industrial waste catalyst.
FTIR analysis
Fourier Transform Infrared (FTIR) spectroscopy is a promising method for characterizing industrial waste powder, as depicted in Fig. 7b is utilized as a heterogeneous catalyst in CSLB production. The FTIR spectrum indicated different absorption bands for industrial waste catalyst functional groups, particularly carbonate ions (CO32−) peaks at 1634.17 cm−1 and 979.10 cm−1, indicating asymmetric and symmetric stretching vibrations. Furthermore, a sharp peak at 770.99 cm−1 is commonly connected with the out-of-plane bending vibration of carbonates present in industrial waste catalysts. In CSLB production, heterogeneous catalyst surface composition (CaO and MgO) is critical to increasing the reaction rate in transesterification.
BET analysis
BET surface area analysis is critical in assessing the porosity and surface characteristics of industrial waste powder used as a catalyst in CSLB production, as illustrated in Fig. 7c. A higher BET surface area (6.271 m²/g) of industrial waste powder generally indicates a more effective catalytic activity, as it provides greater active surface sites for the transesterification reaction in cashew nut shell liquid. The mesoporous structure of industrial waste-derived catalyst revealed through BET analysis enhances the interaction between the catalyst and the cashew nut shell liquid, improving the efficiency of CSLB conversion. Also, it provides insight into achieving sustainable CSLB production with minimal catalyst degradation.
RSM-CCD for CSLB prediction and optimization
The commercialization of biodiesel requires achieving maximum yield. Industrial waste-derived catalysts with heterogeneous nature in the transesterification process are widely employed because of their high efficiency and inexpensive cost44. The CSLB yield in this process is affected by factors such as the methanol/CSL molar ratio, process temperature, and industrial catalyst usage. Equation (2) predicts CSLB yield values for input variables within the range provided in Table 4. Using multiple regression analysis, the data was transformed into a second-order polynomial equation, yielding an empirical model that defines the connection between the response and the independent variables. Design-Expert version 13.0.15 was deployed to conduct an analysis of variance (ANOVA) to figure out the statistical significance of each term in the equations, assuring good fit and favourable adjustment values. Significant terms have an F value within 95% confidence and a p-value ≤ 0.05. The ideal values for the chosen variables were obtained by examining the response surfaces and solving the regression equation. The fitted polynomial equation was depicted as three-dimensional response surface plots to show the primary and interacting effects of the independent factors on the dependent variables.
The model’s fitness was assessed using ANOVA with the least squares approach. ANOVA is a powerful statistical technique that divides the entire variation in model data into distinct sources of variation, allowing one to better understand individual contributions. Table 6 provides a detailed variation analysis of our model. The regression equation’s ANOVA yielded an R² value of 0.9806, suggesting a robust model fit. Values above 0.75 are regarded as good. The R² result indicates that the model can explain 98.06% of the observed variation in experimental parameters and interactions, leaving only 1.94% unexplained. The adjusted and forecast R² values were 0.9632 and 0.9084, respectively, indicating the model’s robustness. R² values vary from 0 to 1.0, with values near 1.0 indicating a better fit. The model’s F-value of 56.31 indicates its importance, and a p-value of less than 0.05 confirms the model’s statistical significance, with the lack of fit being negligible. A lower p-value emphasizes the significance of the interactions among factors. This investigation found significant individual and interaction terms, with the exception of PT*IC, at a p-value of less than 0.05. The anticipated and experimental CSLB production values have a significant correlation (R² = 0.9806). The yield estimation formula for CSLB, created by iterative statistical stepwise regression, is shown in Eq. (2).
Figure 8a–c displays the 3D surface plots illustrating the conversion of CSL to CSLB through the interaction between two independent variables like methanol/CSL molar ratio and process temperature (MR & PT), methanol/CSL molar ratio and industrial catalyst (MR & IC), process temperature and industrial catalyst (PT & IC) their impact on the dependent parameter, which is the CSLB yield. These plots are generated using the regression Eq. (2) and depict how each independent variable interacts with the response variable.
Effects of methanol/CSL molar ratio on CSLB yield
Increasing the methanol/CSL molar ratio (MR) from 10:1 to 20:1 greatly boosts CSLB yield due to the increased availability of methanol for the transesterification process, as shown in Fig. 8a and b. During transesterification, the triglycerides in CSL react with methanol to form methyl esters (CSLB) and glycerol. A higher methanol/CSL ratio shifts the reaction equilibrium towards producing more methyl esters, thereby enhancing CSLB conversion efficiency. Additionally, excess methanol helps overcome reaction kinetics limitations, reduces the viscosity of the reaction mixture, and improves the solubility of the reactants45. This leads to better interaction between CSL and methanol molecules, resulting in a more complete reaction and significantly increasing CSLB yield.
Effects of process temperature on CSLB yield
The CSLB yields a peak between 60 °C and 70 °C, as these temperatures provide ideal conditions for the transesterification reaction, resulting in the maximum conversion of triglycerides to CSLB, as depicted in Fig. 8a and c. At these temperatures, the reaction kinetics and solubility of methanol in CSL are optimal, allowing for efficient mixing and interaction of the reactants. However, when the temperature climbs to 80 °C, the CSLB yield decreases dramatically. This drop is most likely caused by methanol evaporation at higher temperatures, which reduces the alcohol available for the reaction. Thus, maintaining a temperature between 70 °C is critical for maximizing CSLB yield.
(a–c) 3D surface plots showing interaction effects of different input variables (MR, PT, and IC) on CSLB yield.
Effects of industrial catalyst concentration on CSLB yield
When the industrial waste-derived heterogeneous catalyst concentration (IC) is increased from 2 to 4 wt%, CSLB yield increases dramatically due to increased catalytic activity, which makes the transesterification process more efficient, as reported in Fig. 8b and c. At this ideal concentration, the catalyst provides adequate active sites for the process, resulting in a faster conversion of triglycerides to CSLB. However, increasing the catalyst weight from 4 to 6 wt% results in a drop in CSLB yield. This decline is caused by an overabundance of IC, which causes soap production and emulsification, preventing CSLB from being separated from glycerol and other byproducts. Furthermore, using too much IC might raise viscosity and impede mass transfer, limiting reaction efficiency and overall CSLB yield. As a result, determining the optimal IC concentration (4 wt%) is critical for increasing CSLB yield while minimizing negative effects46.
Figure 9 presents the normal plot of residuals, illustrating that nearly all points of CSLB yields align closely with a straight-line pattern. This alignment indicates no significant deviations from normality, thereby confirming the adequacy of the RSM-CCD model in representing the experimental data accurately.
Plot of externally studentized residuals of CSLB yield with normal % probability.
Figure 10 illustrates the optimal conditions for maximum CSLB production as determined by statistical regression Eq. (2). With optimized inputs of 20:1 MR, 70 °C PT, and 4 wt% IC, the transesterification process achieved a maximum CSLB yield of 85.54% using desirability function approach. The overall desirability value of the CCD model is 1, confirming that all optimized conditions are ideal for the CSLB production process.
Optimal conditions for maximum CSLB yield determined using the desirability function approach.
Characterization study on CSLB
GC-MS analysis on CSLB
GC-MS (Gas chromatograph coupled with mass spectrometer) is an analytical method performed to identify and quantify the CSLB’s chemical composition. In this test, the CSLB is vaporised and injected into the gas chromatograph, which separates the CSLB into individual components based on properties such as boiling point and polarity. These separated components were then introduced to the mass spectrometer, which was ionized into small charged particles using an FID detector. Then, it analyses the charged particles based on the mass-to-charge ratio, producing a mass spectrum that provides information on the number of individual components in the CSLB. The amount of each compound present in a CSLB is determined by comparing it with the resultant mass spectrum to the standard spectra. There are many peaks that are visible, hence the increased presence of ester compounds, which is listed in Table 7. The peak’s % area indicates the number of components contained in the CSLB. The maximum percentage of CSLB conversion from CSL using an industrial waste heterogeneous catalyst was primarily confirmed by the presence of 1-Methyl-2-hydroxybenzene (18.27%), 8-Octadecenoic acid, methyl ester (15.12%), Pentdecanoic acid, 14-methyl ester (12.87%), and Decane, 3,8-dimethyl- (11.24%) in the CSLB chromatogram.
FTIR analysis on CSLB
Fourier Transform Infra-Red (FTIR) spectroscopy is an analytical test carried out to identify and characterize the functional groups in diesel, CSL and CSLB. FTIR analysis works for a range of wavelengths in the infrared spectrum, which are transmitted through all test fuels. These fuels have the ability to transmit the IR ray’s energy at different wavelengths, determining the presence of different functional groups in the CSLB’s chemical composition. A graph (Fig. 11) is created by plotting the corresponding wavenumber (cm− 1) on the X-axis and the transmittance (%) of the diesel, CSL and CLSB on the Y-axis. Two bands within the mid-IR spectrum range from 4000 cm− 1 to 400 cm− 1. A noticeable peak at 3392 cm− 1 demonstrates the stretching vibration with weak absorption intensity of the hydroxyl (-OH) functional group, implying that WTSB substances contain phenols or alcohols. Stretching vibrations with substantial alkene and alkane absorption intensity are shown by the peaks at 2937 cm− 1 and 2852 cm− 1, respectively. These vibrations are thought to reflect the C-H functional groups in CSLB, resulting in methyl and methylene groups from transesterification. The appearance of a prominent absorption intensity peak at 1749 cm− 1 corresponding to a C = O stretching vibration validates the production of ester groups, such as mono-alkyl esters, in CSLB structures. Asymmetric and symmetric bending vibrations at 1460 cm− 1 and 1376 cm− 1 indicate the functional groups of CH2 and CH3. Similarly, the C-O functional groups formed at a sharp point of 1174 cm− 1, resulting in ester bonds or linkages with CSLB molecules. A small peak at 721 cm− 1 represents the out-of-plane bending (rocking) vibration of C-H bonds, indicating the presence of long-chain hydrocarbons in CSLB. The FTIR spectral analysis confirms the presence of oxygen functional groups in CSLB, highlighting its oxygenated nature. The spectral peaks of CSLB closely resemble those of conventional diesel, demonstrating comparable structural characteristics, as listed in Table 8. These findings suggest that CSLB is a viable and effective alternative to traditional diesel fuel.
FTIR spectra of diesel, CSL and CSLB.
CRDI engine emission characteristics
Nitrogen oxide (NOx) emissions
Figure 12 depicts the nitrogen oxide emission trends across multiple test fuels with optimal fuel injection circumstances and EGR rates at maximum load. When split injection, delayed timing, and EGR rate are combined, NOx emissions are significantly reduced across all load conditions. The delay in injection time causes the delayed combustion process, while exhaust gas recirculation, with its higher specific heat capacity, lowers the overall combustion temperature. As a result, peak cylinder pressure and temperature are reduced, resulting in decreased NOx emissions22. At full load, NOx emissions are reduced by 35.29% for diesel, 33.37% for CSLB5, and 29.14% for CSLB10 test fuels under ideal conditions. This is due to the reduced premixed burning rate after the delay period, which results in lower air entrainment and a slower fuel-air mixing rate, minimizing the NOx formation47.
Comparative analysis of NOx emissions on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
The addition of butanol helps to reduce NOx emissions significantly. This effect is caused by the additives’ lower calorific content (33800 kJ/kg) and higher evaporation rate (626 kJ/kg), resulting in a delayed combustion process and subsequent decreases in maximum cylinder pressure and temperature. The lower combustion temperature inhibits reactivity among O2 and N2, reducing NOx emission. It is worth noting that the additives’ overall leaner combustion and cooling impact substantially influence minimizing NOx emissions48. While oxygen content in the additives may increase the premixed combustion temperature and hence increase NOx emissions, the cooling impact outweighs other effects, resulting in lower NOx emissions29. Adding butanol at 2.5%, 5%, and 10% concentrations to diesel, CSLB5 and CSLB10 at full load reduces NOx emissions. Notably, the 10% butanol addition significantly minimizes the NOx formations by 32.19%, 44.21%, and 46.37% for diesel, CSLB5 and CSLB10 fuels compared to ideal operating conditions. Similar trends are observed by Singh et al.31, who studied the 5%, 10%, and 15% n-butanol addition to eucalyptus biodiesel and diesel blends (B20), showing 21.90%, 23.60% and 25.16% decrease in NOx emission across top load conditions when compared to diesel, B100 and B20 blends. This is due to the high latent heat of vaporization of n-butanol, which induces a cooling effect during combustion, reducing in-cylinder pressure and temperature. In addition, the combustion duration of diesel/jojoba oil/butanol blends (DBJ15) is shorter than that of pure diesel fuel, which reduces the time available for NOx formation, thereby lowering NOx emissions as reported by EL-Seesy et al.35. These literature data indicate the beneficial effect of n-butanol additions on reducing NOx emissions in diesel and CSLB blends.
Smoke emissions
Internal combustion engine smoke emissions are primarily influenced by viscosity, uniform fuel-air mixture, availability of oxygen, and oxidation process time49,50. Figure 13 depicts the comparative analysis of smoke emissions among different test fuels at full load under ideal conditions. CSLB biodiesel blends, which have a lower sulfur concentration of 0.06%, emit less smoke than standard diesel fuel. Furthermore, for the investigated fuels, retarded timing and split injection strategies contribute to lower smoke emissions across all load conditions. However, as a result of the limited supply of oxygen, the use of EGR causes a modest increase in smoke emissions. The split injection improves fuel distribution and increases the regularity of the air-fuel mixture, reducing the presence of fuel-rich zones20. Due to this, smoke emissions are reduced by 15.52%, 2.72%, and 14.24% at full load under the ideal fuel injection and EGR conditions for diesel, CSLB5 and CSLB10 blends. Furthermore, the addition of butanol lowers smoke emissions even more. The enhanced oxygen concentration of the additions promotes oxidation by forming OH radicals, resulting in more complete combustion and lower smoke emissions27. At full load, blending 2.5%, 5%, and 10% butanol with diesel minimizes smoke emissions by 28.49%, 36.67%, and 45.48%. Similar trends are found across all load conditions when the additives are added to CSLB5 and CSLB10 blends. Notably, 26.56%, 28.98%, and 34.11% smoke reductions were observed for CSLB5, adding butanol by 2.5%, 5% and 10%, respectively. The further reduction in smoke was identified with the CSLB10 blend with 2.5%, 5% and 10% butanol additives by 5.46%, 9.57%, and 14.02% compared with ideal settings. These findings highlight the ability of butanol additives to reduce smoke emissions in diesel and CSLB blends effectively. Chen et al.51 observed a similar trend in smoke opacity emissions when n-butanol was added to a diesel mixture under full-load conditions, resulting in a 10.91% reduction in smoke opacity compared to other alcohols. This reduction is attributed to n-butanol’s lower viscosity, superior fuel atomization, and smaller fuel droplet size, contributing to decreased exhaust gas opacity. Pullagura et al.37 reported a 35.12% reduction in smoke opacity when using butanol in sea mango biodiesel/diesel blends compared to diesel fuel under higher load conditions.
Comparative analysis of Smoke emissions on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
Carbon monoxide (CO) emissions
Figure 14 compares CO emissions among various fuels at full load under ideal conditions. Implementation of delayed injection time, split fuel injection, and optimal EGR rate significantly reduces CO emissions. Retarded injection timing increases mixing duration, resulting in decreased CO emissions. Split injection results in a more homogenous mixture, eliminating fuel-rich zones and helping to reduce CO levels16. CO levels are minimized by 18.91% for diesel, 20.42% for CSLB5, and 26.92% for CSLB10 test fuels at full load under ideal conditions. Carbon monoxide emissions decreased considerably with butanol blends of 2.5%, 5%, and 10% in diesel fuel for 8.69%, 12%, and 16.82%, respectively. This reduction can be due to the increased spray characteristics, atomization, mixture quality, and combustion efficiency made possible by the additives’ lower density and viscosity52. The additives’ higher oxygen concentration improves combustion and reduces fuel-rich zones, which helps to reduce CO emissions29. Furthermore, when the amount of butanol in the mixture increases, the carbon content decreases, resulting in a lower carbon-to-hydrogen (C/H) ratio and reducing CO emissions. At full load, adding 2.5%, 5%, and 10% butanol with CSLB5 results in a 14%, 18.08%, and 26.86% reduction in CO emissions. The CSLB10 blend showed maximum CO reduction of 10.34%, 13.04%, and 18.18% under ideal conditions. Similar trends are found across all load conditions when the additives are added to CSLB blends. The inclusion of 10% isobutanol in the subabul seed biodiesel/diesel blend (B20) resulted in a 4.12% reduction in CO emissions at full load compared to diesel fuel. The additional oxygen content in isobutanol enhances combustion efficiency and promotes more complete fuel oxidation, potentially reducing CO emissions, as reported by Yamini et al.53.
Comparative analysis of CO emissions on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
Carbon dioxide (CO2) emissions
Figure 15 compares CO2 emissions among various fuels under ideal full-load conditions. With the application of split injection and EGR, there is a marginal rise in CO2 emissions. Split injection strategies contribute to somewhat greater CO2 emissions while enhancing mixture quality and boosting complete combustion. Similarly, using EGR raises the charge temperature, which causes carbon monoxide (CO) oxidation into carbon dioxide (CO2), contributing to higher CO2 emissions8. At maximum load, CSLB5 and CSLB10 blends emit around 9.27% and 9.79% more CO2 than the baseline value. Using butanol additives to the CSLB fuels results in a modest rise in CO2 emissions compared to the ideal base diesel tests. This rise may be due to the additives’ higher O2 availability, which promotes more efficient combustion26. Furthermore, the fuel’s lower viscosity helps the mixing process, allowing for a faster oxidation reaction and contributing to increased CO2 emissions. It is worth mentioning, however, that the percentage of additives used in fuels is constrained to 10% since a higher percentage can reduce carbon content, potentially compromising the combustion process54. Remarkably, adding additives to pure diesel has no discernible effect on CO2 emissions. However, adding 10% butanol to CSLB blends slightly increases CO2 emissions by 7.28% and 8.84% at full load. A similar result was reported by Hassan et al.36, where the presence of n-butanol in a biodiesel blend reduced CO2 emissions. This reduction is attributed to the lower carbon-to-hydrogen ratio of alcohol compared to neat biodiesel or diesel.
Comparative analysis of CO2 emissions on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
Hydrocarbon (HC) emissions
Figure 16 compares HC emissions among various fuels under ideal full-load conditions. HC emissions increase as injection timing is retarded and at various EGR rates. Because of the delayed timing, the combustion continues up to the initial stages of the power stroke. This influences the HRR and combustion of fuel temperature, resulting in increased HC formation22. Similarly, using EGR reduces combustion temperatures and decreases O2 availability, which influences hydrocarbon oxidation and contributes to increased HC emissions. Under ideal fuel injection and EGR settings, HC emissions rise by 9.02% for diesel, 7.78% for CSLB5, and 8.45% for CSLB10 test fuels at peak load. Notably, using butanol additives results in a slight reduction in HC emissions in diesel and CSLB blends. The maximum HC reduction was observed in the CSLB10 blend with 2.5%, 5% and 10%, butanol addition resulted in lower HC by 14.52%, 24.56%, and 39.21%, respectively. This reduction can be due to additives’ increased atomization, improved mixture quality, and improved flame propagation. The occurrence of oxygen atoms in the butanol additives accelerates hydrocarbon combustion55. Furthermore, the split fuel injection approach aids in producing a uniform blend, minimizing HC emissions substantially. However, HC emissions remain higher than the baseline values. The increase in HC emissions for the butanol/diesel mixture is more pronounced at low loads compared to high loads. This is due to the high latent heat of vaporization of the butanol/diesel mixture, which reduces droplet evaporation efficiency and leads to incomplete combustion near low-temperature walls, resulting in higher HC emissions at low loads. However, these effects are mitigated under high-load conditions, as reported by Chen et al.51.
Comparative analysis of HC emissions on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
CRDI engine performance characteristics
Brake-specific energy consumption (BSEC)
Comparative analysis of BSEC on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
Brake-specific energy consumption (BSEC), expressed in MJ/kW-hr, is an essential metric for evaluating engine performance since it measures the energy required to create 1 kW of BP. Figure 17 compares BSEC among various fuels under ideal full-load conditions. Ideal operating circumstances result in a large rise in BSEC. This increase might be attributed to delayed injection timing, which prolongs combustion56. Furthermore, the application of EGR has a diluting effect on the A/F ratio and fuel ignition, contributing to the reported increase in BSEC. Specifically, at maximum load and under ideal fuel injection conditions and EGR rate, BSEC rises by 22.06%, 25.21%, and 30.94% to the CSLB5, CSLB10, and diesel blends. The butanol additives’ lesser viscosity and enhanced oxygen concentration promote better atomization, resulting in complete combustion and reduced BSEC. Moreover, the split injection approach decreases cylinder heat losses and assures effective fuel distribution, resulting in a more uniform air-fuel mixture and increased combustion efficiency, contributing to the lower BSEC16. Notably, adding 10% butanol to diesel, CSLB5 and CSLB10 blend at full load results in a maximum reduction of BSEC by 17.81%, 18.11% and 16.18% than ideal conditions. Similar trends are observed across all load conditions with the usage of 2.5% and 5% butanol additives. Sayyed et al.57 confirmed that the addition of butanol to diesel/Karanja biodiesel blends resulted in lower brake-specific energy consumption (BSEC) compared to other biodiesel blends. This reduction is attributed to the optimized density and viscosity of the fuel blends, which enhance atomization and combustion efficiency, leading to reduced BSEC under peak load conditions.
Brake thermal efficiency (BTE)
Figure 18 compares BTE among various fuels under ideal full-load conditions. The split injection is advantageous because it encourages the development of a homogenous mixture, which reduces heat losses and increases BTE. In contrast, delayed injection timing and using EGR result in a minor decrease in thermal efficiency. This is due to the delayed start of injection caused by the retarded timing, which results in a decrease in total power developed20. Additionally, EGR affects flame temperature because exhaust gases have a larger heat capacity, and less oxygen is available, which affects ignition and, consequently, engine performance. Under ideal fuel injection and EGR conditions, BTE decreases for diesel, CSLB5, and CSLB10 blends, which are 7.91%, 6.24%, and 5.35%, respectively, compared to baseline. The use of butanol additives increases BTE when compared to ideal conditions. Notably, diesel with 2.5%, 5%, and 10% butanol gradually increment the BTE of 2.14%, 3.15% and 2.13%, respectively. Similar tendencies are found across all top load conditions when butanol additives are introduced into CSLB blends. It can be ascribed to many variables, including increased spray properties, enhanced fragmentation and oxygen content, contribute to more effective combustion and, as a result, higher BTE. Also, the lower combustion temperature caused by the presence of butanol reduces heat losses, improving total engine efficiency27. It is worth noting that the additive proportion is limited to 10% due to the potential detrimental impact on CRDI engine performance caused by the additives’ low energy content and higher latent heat. When 10% butanol is added to diesel at full load, BTE improves and reaches a level comparable to the baseline data. Although the addition of butanol improves the viscosity and density of the fuel, the resulting brake thermal efficiency (BTE) remains close to the baseline and ideal conditions. However, Singh et al.31 reported that the higher BTE observed in butanol-blended B20 fuels is attributed to their higher oxygen content and improved air-fuel atomization. Additionally, lower viscosity and density enhance fuel vaporization, facilitating better utilization of thermal energy.
Comparative analysis of BTE on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
CRDI engine combustion characteristics
Maximum cylinder pressure (Pmax)
Comparative analysis of Pmax on diesel, CSLB5 and CSLB10 fuel blends with different % butanol additive.
Figure 19 compares maximum cylinder pressure among various fuels under ideal full-load conditions. Retarded injection timing contributes to a delayed combustion process, resulting in lower peak Pmax and temperature56. However, using EGR in addition to split injection helps alleviate this disadvantage to some extent. As a result, peak Pmax decreases with diesel for 10.73%, CSLB5 for 10.06%, and CSLB10 for 11.89%, respectively, compared to baseline readings under the ideal fuel injection and EGR conditions. To address this issue, the use of butanol additives with different percentages (2.5%, 5%, and 10%) becomes desirable for all load conditions. However, using additives causes a slight rise in peak Pmax compared to the optimal conditions. The higher O2 availability and reduced viscosity of the additives improve combustion dynamics, resulting in higher maximum pressure and more heat output during the diluted phase29. A 10% addition of butanol to diesel, CSLB5 and CSLB10 rises peak Pmax by 5.62%, 5.46%, and 8.21%. Similar increasing Pmax trends are observed across all load conditions with the usage of 2.5% and 5% butanol additives. When fuel additives like butanol are blended with a B20 fuel blend, they can enhance the overall combustion process by reducing ignition delay. The addition of butanol has been shown to shorten ignition delay, promoting faster and more efficient combustion, which results in a higher peak pressure (Pmax), as reported by Yamini et al.53.
Heat release rate (HRR)
Comparative analysis of average HRR of three different test blends with different % butanol additive.
Figure 20 compares the average HRR values among various fuels under ideal full-load conditions. The HRR, represented in J/°CA, evaluates the engine’s capacity to transform accessible heat into usable power49. Results showed that the HRR increases through split fuel injection, indicating enhanced mechanisms for blending and ignition. In contrast, increasing EGR from 0 to 20% results in a drop in HRR of 74.16 J/°CA. This is due to EGR’s thermal, diluting, and chemical effects, which prolong the ignition delay and reduce O2 supply, consequently influencing combustion. Furthermore, the chemically inert nature of exhaust gas absorbs heat emitted during combustion, reducing the HRR8. When butanol additives are added by 2.5%, 5% and 10% to the test fuels, HRR slightly rises, and their peak HRR values are 76.59 J/°CA, 78.16 J/°CA, and 79.72 J/°CA, respectively. It is attributed to oxygen molecules promoting improved combustion and increased heat release during the premixed ignition phase26. The butanol additive has a higher latent heat, which may negatively influence combustion, potentially leading to a significant decrease in the maximum HRR. Butanol added percentage is kept at 10% for optimal combustion of CRDI engine performance. A similar trend was observed by Li et al.58. The increase in HRR is attributed to the higher butanol blending ratio, which extends the in-cylinder fuel’s physical and chemical preparation time. This prolongation allows for the formation of a larger homogeneous combustible mixture before ignition, thereby enhancing premixed combustion.
Conclusion
This study optimized CSLB production using industrial waste catalysts and the RSM-CCD technique. It also examined the impact of butanol additives in CSLB blends on CRDI engine performance under ideal conditions. The optimization and experimental results led to the following conclusions.
-
Characterization of the industrial waste-derived catalyst (XRD, FTIR, BET) revealed high catalytic compounds (CaO, MgO), carbonate ions (CO32−), and a surface area of 6.271 m²/g, confirming its effectiveness for CSLB production.
-
Optimized transesterification process (MR 20:1, PT 70 °C, IC 4 wt%) using RSM-CCD technique achieved high CSLB yields: 85.43% (experimental) and 85.54% (predicted) via the desirability function approach. GC-MS confirms hydrocarbon and ester formation, while FTIR reveals oxygen functional groups, emphasizing CSLB’s potential as a sustainable fuel.
-
Butanol additives improved atomization, air-fuel mixing, and combustion due to lower viscosity and higher oxygen content. A 10% addition with diesel, CSLB5, and CSLB10 reduced NOx by 44.08%, 48%, and 47.09%, smoke by 40.49%, 27.20%, and 23.02%, and CO by 27.89%, 34.63%, and 33% compared to the baseline test.
-
The 10% butanol additives improved oxidation, reducing HC by 31%, 26.67%, and 27.14% for diesel, CSLB5, and CSLB10. CO2 emissions slightly increased by 18.35%, 7.28%, and 8.92%, respectively, compared to ideal conditions. CRDI engine tests showed improved performance with 10% butanol additives, achieving 38.96% BTE and reducing BSEC by 15.12%. In addition, HRR trends indicated better combustion with peak cylinder Pmax.
-
Butanol additives should be limited to 10% to prevent CRDI engine performance degradation. Higher percentages enhance atomization and combustion but may affect combustion due to increased latent heat of vaporization and lower energy content.
Finally, the study showed that CSLB with optimal conditions and butanol in CSLB5 and CSLB10 improved CRDI engine performance and emissions. The experimental results offer valuable information for the automotive industry’s optimization of CSLB blends and CRDI engine operating conditions to achieve greater sustainability and less environmental harm.
Potential future directions of this research work
-
Improve industrial waste-derived catalysts for better stability, reusability, and efficiency using advanced activation methods, such as microwave or ultrasonic-assisted techniques.
-
Conduct life cycle assessment and cost analyses to evaluate the commercial viability and environmental benefits of CSLB production.
-
Research on advanced emission control technologies, such as after-treatment systems (catalytic converters and particulate filters), combined with CSLB/butanol blends, could further minimize environmental impact.
-
Investigate the feasibility of using a higher-level percentage of butanol in CSLB and its blends while mitigating energy loss and combustion problems.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- ASTM:
-
American society for testing and materials
- BTE:
-
Brake thermal efficiency
- BET:
-
Brunauer-Emmett-Teller
- BSEC:
-
Brake-specific energy consumption
- CSLB5:
-
5% CSLB
- CSLB10:
-
10% CSLB
- CCD:
-
Central composite design
- CSL:
-
Cashew nut shell liquid
- CA:
-
Crank angle
- CO:
-
Carbon monoxide
- CO2 :
-
Carbon dioxide
- CRDI:
-
Common rail direct injection
- CSLB:
-
Cashew nut shell liquid biodiesel
- DI:
-
Direct injection
- ECU:
-
Electronic control unit
- EGR:
-
Exhaust gas recirculation
- FTIR:
-
Fourier transform infrared spectroscopy
- GC-MS:
-
Gas chromatography-mass spectrometry
- HC:
-
Hydrocarbon
- HRR:
-
Heat release rate
- NOx :
-
Nitrogen oxide
- Pmax :
-
Maximum cylinder pressure
- RSM:
-
Response surface methodology
- XRD:
-
X-ray diffraction
References
Kaya, C. & Kökkülünk, G. Environmental effects biodiesel as alternative additive fuel for diesel engines: an experimental and Theoretical investigation on emissions and performance characteristics biodiesel as alternative additive fuel for diesel engines : an experimental and Th. Energy Sour. Part A Recover. Utilization Environ. Eff. 00, 1–23 (2020).
Nayak, S. K. & Mishra, P. C. Emission characteristics of Jatropha oil blends using waste wood producer gas. Energy Sources Part. A: Recovery Utilization Environ. Eff. 38, 2153–2160 (2016).
Nayak, S. K., Mishra, P. C. & Behera, G. R. Characterization of coconut shell imitatived producer gas in adiesel engine. Energy Sources Part. A: Recovery Utilization Environ. Eff. 39, 1718–1724 (2017).
Imdadul, H. K. et al. Experimental assessment of non-edible candlenut biodiesel and its blend characteristics as diesel engine fuel. Environ. Sci. Pollut. Res. 24, 2350–2363 (2017).
Nayak, S. K. & Mishra, P. C. Emission characteristics of diesel fuel composed of linseed oil (Linum Usitatissium) blends utilizing rice husk producer gas. Energy Sources Part. A: Recovery Utilization Environ. Eff. 38, 2001–2008 (2016).
Kandaswamy, S. et al. Cashew nut shell oil as a potential feedstock for biodiesel production: An overview. Biotechnol. Bioeng. 120, 3137–3147 https://doi.org/10.1002/bit.28515 (2023).
Kasiraman, G., Edwin Geo, V. & Nagalingam, B. Assessment of cashew nut shell oil as an alternate fuel for CI (Compression ignition) engines. Energy 101, 402–410 (2016).
Edara, G., Satyanarayana Murthy, Y. V. V., Nayar, J., Ramesh, M. & Srinivas, P. Combustion analysis of modified light duty diesel engine under high pressure split injections with cooled EGR. Eng. Sci. Technol. Int. J. 22, 966–978 (2019).
Manimaran, R., Mohanraj, T., Venkatesan, M., Ganesan, R. & Balasubramanian, D. A computational technique for prediction and optimization of VCR engine performance and emission parameters fuelled with trichosanthes cucumerina biodiesel using RSM with desirability function approach. Energy 254, (2022).
Ghorbani-Choghamarani, A., Taherinia, Z. & Tyula, Y. A. Efficient biodiesel production from oleic and palmitic acid using a novel molybdenum metal–organic framework as efficient and reusable catalyst. Sci. Rep. 12, (2022).
Wang, J. Biodiesel production from spent coffee grounds utilizing pomegranate/orange Peel Biochar as a green and renewable nanocatalyst: compression ignition engine performance and emission. Ind. Crops Prod. 218, 118890 (2024).
Rao, T. S. S. B. et al. Sustainable synthesis and advanced optimization of prosopis Juliflora biomass catalyst for efficient biodiesel production and environmental impact assessment. Sci. Rep. 15, 4472 (2025).
Dubey, A., Prasad, R. S. & Singh, J. K. An analytical and economical assessment of the waste cooking oil based biodiesel using optimized conditions on the process variables. Energy Sources Part. A: Recovery Utilization Environ. Eff. https://doi.org/10.1080/15567036.2020.1839600 (2020).
Farouk, S. M., Tayeb, A. M., Osman, R. M. & Abdel-Hamid, S. M. S. Sustainable production of biodiesel from waste cooking oil using magnesium oxide nano catalyst: an optimization study. Sci. Rep. 14, (2024).
Sun, X., Xu, L., Yuan, H., Wang, G. & Maleki, B. Boosted conversion of restaurant waste oil into biodiesel using Fe3O4@UiO-66-NH2 magnetic heterogeneous nanocatalyst and its application on the diesel engine: optimization via RSM. Renew. Energy. 223, 120007 (2024).
Ganesan, N. et al. Split injection strategies based RCCI combustion analysis with waste cooking oil biofuel and methanol in an open ECU assisted CRDI engine. Fuel 319, (2022).
How, H. G., Masjuki, H. H., Kalam, M. A. & Teoh, Y. H. Influence of injection timing and split injection strategies on performance, emissions, and combustion characteristics of diesel engine fueled with biodiesel blended fuels. Fuel 213, 106–114 (2018).
Senthil kumar, D. & Thirumalini, S. Investigations on effect of split and retarded injection on the performance characteristics of engines with cashew nut shell biodiesel blends. Int. J. Ambient Energy. 43, 2251–2259 (2022).
Venugopal, I. P., Balasubramanian, D., Rajarajan, A. & Suresh, K. Quantification of φ-operating range with impact of exhaust gas recirculation under low-temperature combustion mode with polyoxymethylene dimethyl ether: A perspective study. J. Clean. Prod. 411, (2023).
Rami Reddy, S., Murali, G., Ahamad Shaik, A., Dhana Raju, V. & Sreekara Reddy, M. B. S. Experimental evaluation of diesel engine powered with waste Mango seed biodiesel at different injection timings and EGR rates. Fuel 285, 119047 (2021).
Ge, J. C., Kim, M. S., Yoon, S. K. & Choi, N. J. Effects of pilot injection timing and EGR on combustion, performance and exhaust emissions in a common rail diesel engine fueled with a Canola oil Biodiesel-Diesel blend. Energies. 8, 7312–7325 (2015).
Ayhan, V., Çangal, Ç., Cesur, İ. & Safa, A. Combined influence of supercharging, EGR, biodiesel and ethanol on emissions of a diesel engine: proposal of an optimization strategy. Energy 207, (2020).
Jain, A., Singh, A. P. & Agarwal, A. K. Effect of fuel injection parameters on combustion stability and emissions of a mineral diesel fueled partially premixed charge compression ignition (PCCI) engine. Appl. Energy. 190, 658–669 (2017).
Edara, G., Satyanarayana Murthy, Y. V. V., Srinivas, P., Nayar, J. & Ramesh, M. Effect of cooled EGR on modified light duty diesel engine for combustion, performance and emissions under high pressure split injection strategies. Case Stud. Therm. Eng. 12, 188–202 (2018).
Gürbüz, H. & Sandalcı, T. Numerical analysis of diesel injection strategies on emissions and performance in CH4/diesel powered RCCI diesel engine with high ratio EGR. Alexandria Eng. J. 64, 517–526 (2023).
Ning, L. et al. A comparative study on the combustion and emissions of a non-road common rail diesel engine fueled with primary alcohol fuels (methanol, ethanol, and n-butanol)/diesel dual fuel. Fuel 266, (2020).
Deng, X., Li, J., Liang, Y. & Yang, W. Dual-fuel engines fueled with n-butanol/n-octanol and n-butanol/DNBE: A comparative study of combustion and emissions characteristics. Energy 263, (2023).
Nanthagopal, K. et al. An assessment on the effects of 1-pentanol and 1-butanol as additives with Calophyllum Inophyllum biodiesel. Energy Convers. Manag. 158, 70–80 (2018).
Tian, W., Zhang, H., Wang, L., Han, Z. & Yu, W. Effect of premixed n-butanol ratio on the initial stage of combustion in a light-duty butanol/diesel dual-fuel engine. Energies (Basel) 13, (2020).
Yusri, I. M., Mamat, R., Akasyah, M. K., Jamlos, M. F. & Yusop, A. F. Evaluation of engine combustion and exhaust emissions characteristics using diesel/butanol blended fuel. Appl. Therm. Eng. 156, 209–219 (2019).
Singh, R., Singh, S. & Kumar, M. Impact of n-butanol as an additive with Eucalyptus biodiesel-diesel blends on the performance and emission parameters of the diesel engine. Fuel 277, (2020).
Zhu, Q. et al. Evaluating the effect of n-butanol additive on particulate matter emission in diesel engine. Fuel 332, (2023).
Killol, A., Reddy, N., Paruvada, S. & Murugan, S. Experimental studies of a diesel engine run on biodiesel n-butanol blends. Renew. Energy. 135, 687–700 (2019).
Jeevahan, J., Sriramanjaneyulu, G., Durairaj, R. B. & Mageshwaran, G. Experimental investigation of the suitability of 1-butanol blended with biodiesel as an alternative biofuel in diesel engines. Biocatal. Agric. Biotechnol. 15, 72–77 (2018).
EL-Seesy, A. I., He, Z., Hassan, H. & Balasubramanian, D. Improvement of combustion and emission characteristics of a diesel engine working with Diesel/jojoba oil blends and butanol additive. Fuel 279, (2020).
Hassan, T. et al. Effect of Ni and al nanoadditives on the performance and emission characteristics of a diesel engine fueled with diesel-castor oil biodiesel-n-butanol blends. Case Stud. Chem. Environ. Eng. 8, (2023).
Pullagura, G. et al. Performance, combustion and emission reduction characteristics of metal-based silicon dioxide nanoparticle additives included in ternary fuel (diesel-SMME-iso butanol) on diesel engine. Heliyon 10, (2024).
Manimaran, R., Mohanraj, T. & Prabakaran, S. Biodegradable waste-derived biodiesel as a potential green fuel: optimization of production process and its application in diesel engine. Ind. Crops Prod. 192, (2023).
Devasan, R. et al. Microwave-assisted biodiesel production using bio-waste catalyst and process optimization using response surface methodology and kinetic study. Sci. Rep. 13, (2023).
Hanif, M., Bhatti, I. A., Zahid, M. & Shahid, M. Production of biodiesel from non-edible feedstocks using environment friendly nano-magnetic Fe/SnO catalyst. Sci. Rep. 12, (2022).
Tamoradi, T., Kiasat, A. R., Veisi, H., Nobakht, V. & Karmakar, B. RSM process optimization of biodiesel production from rapeseed oil and waste corn oil in the presence of green and novel catalyst. Sci. Rep. 12, (2022).
Varalakksmi, V., Hariharan, R., Rahul, S., Prabakaran, S. & Arumugam, A. Sustainable design of one-pot synthesis of Madhuca indica Methyl ester via reactive extraction using a sulfonated heterogeneous catalyst and techno-economic analysis. Ind. Crops Prod. 223, (2025).
Moffat, R. J. Using Uncertainty Analysis in the Planning of an Experiment. https://fluidsengineering.asmedigitalcollection.asme.org (1985).
Monika, Banga, S. & Pathak, V. V. Biodiesel production from waste cooking oil: A comprehensive review on the application of heterogenous catalysts. Energy Nexus. 10, 100209 (2023).
Sendzikiene, E. & Makareviciene, V. Synthesis of biodiesel by interesterification of triglycerides with Methyl formate. Appl. Sci. 12, (2022).
Al-Muhtaseb, A. H. et al. State-of-the-art novel catalyst synthesised from waste glassware and eggshells for cleaner fuel production. Fuel 330, 125526 (2022).
Nayak, S. K., Mishra, P. C., Kumar, A., Behera, G. R. & Nayak, B. Experimental investigation on property analysis of Karanja oil Methyl ester for vehicular usage. Energy Sources Part. A: Recovery Utilization Environ. Eff. 39, 306–312 (2017).
Tamilvanan, A. et al. Chapter 5 - Effect of engine operating parameters in NOx reduction. In NOx Emission Control Technologies in Stationary and Automotive Internal Combustion Engines (ed. Ashok, B.) 125–153 https://doi.org/10.1016/B978-0-12-823955-1.00005-X (Elsevier, 2022).
R., M., K., M. M. K. & V., H. S. Study on novel nano Mahua Methyl ester powered DI diesel engine preheated with a thermoelectric waste heat recovery system. Energy Sour. Part A Recover. Utilization Environ. Eff. 43, 3251–3265 (2021).
Nayak, S. K., Behera, G. R., Mishra, P. C. & Sahu, S. K. Biodiesel vs diesel: A race for the future. Energy Sources Part. A: Recovery Utilization Environ. Eff. 39, 1453–1460 (2017).
Chen, Q. et al. Analyzing the combustion and emissions of a DI diesel engine powered by primary alcohol (methanol, ethanol, n-butanol)/diesel blend with aluminum nano-additives. Fuel 328, (2022).
Tamilvanan, A., Mohanraj, T., Ashok, B. & Santhoshkumar, A. Enhancement of energy conversion and emission reduction of Calophyllum Inophyllum biodiesel in diesel engine using reactivity controlled compression ignition strategy and TOPSIS optimization. Energy 264, (2023).
YAMINI, K., KISHORE, P. S. & DHANA RAJU, V. Effect of diethyl ether and Isobutanol as fuel additives on the diesel engine attributes fueled with subabul seed biodiesel. J. Therm. Eng. 11, 215–225 (2025).
Xiao, H., Guo, F., Li, S., Wang, R. & Yang, X. Combustion performance and emission characteristics of a diesel engine burning biodiesel blended with n-butanol. Fuel 258, 115887 (2019).
Goga, G., Chauhan, B. S., Mahla, S. K. & Cho, H. M. Performance and emission characteristics of diesel engine fueled with rice Bran biodiesel and n-butanol. Energy Rep. 5, 78–83 (2019).
Karthic, S. V., Kumar, S., Nataraj, M., Pradeep, P. & G. & An assessment on injection pressure and timing to reduce emissions on diesel engine powered by renewable fuel. J. Clean. Prod. 255, 120186 (2020).
Sayyed, S., Kulkarni, K., Das, R. K., Alam, T. & Eldin, S. M. Direct injection diesel engine characteristics fuelled with diesel, biodiesel and 1-butanol blends. Case Stud. Therm. Eng. 53, (2024).
Li, G. et al. Effects of isopropanol-butanol-ethanol on the performance, combustion and emission characteristics of a diesel-methanol dual-fuel engine. Renew. Energy 237, (2024).
Acknowledgements
The authors want to express their gratefulness to Amrita School of Engineering, Amrita Vishwa Vidyapeetham, Coimbatore, and SRM Institute of Science and Technology, Tiruchirappalli campus, Tamil Nadu, India, Madanapalle Institute of Technology and Science, Madanapalle, Andhra Pradesh, India and Yeshwantrao Chavan College of Engineering, Nagpur, Maharashtra, India for completing the present study. Also, the authors extend their appreciation for financial assistance provided by the College of Engineering, University of Business and Technology, Jeddah, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
S. D.: Supervision, Validation. J.G.: Writing - review & editing. T. S. S. B. R.: Software, Resources. M. K.: Data curation, Funding acquisition. R. M.: Conceptualization, Methodology, Investigation, Writing - original draft.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Duraisamy, S., Giri, J., Rao, T.S.S.B. et al. Optimization of cashew nut shell biodiesel production with industrial waste catalysts and butanol additives for ecofriendly CRDI engine applications. Sci Rep 15, 9573 (2025). https://doi.org/10.1038/s41598-025-93805-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93805-9
Keywords
This article is cited by
-
Performance, combustion, emission and optimization characteristics of biodiesel–n-butanol blends enriched with Ni2O3 nanoparticles in a diesel engine
Scientific Reports (2026)
-
Design and Development of a Compact Pyro-Gasifier System for Bio-Oil and Biochar Production Using Coconut Shell Biomass
BioEnergy Research (2026)
-
Parametric analysis of biodiesel synthesis from palm oil using homogenous base catalyst: experimental and numerical investigation
Scientific Reports (2025)
-
Advanced prediction and optimization of VCR engine characteristics using RSM with DFA for sustainable biofuel derived from waste lemon Peel
Scientific Reports (2025)