Abstract
Wig protectors produced via 3D printing were used in skull protection protocols following decompressive craniotomy (DC) surgery. This study is a single-center, prospective trial involving 25 patients who underwent DC. Patients admitted between February 1, 2023, and June 30, 2023, were assigned to the control group (receiving standard care), while those admitted between July 1, 2023, and December 31, 2023, comprised the experimental group (receiving 3D-printed wig protection). The study evaluated participants based on various metrics, including reinjury at the site of the skull defect, daily wear duration, skin indentation, pain levels, itching, discomfort, patient-reported aesthetics, and the Quality of Life (QLQ-C30). They completed both wear assessments and follow-up evaluations at 1, 4, 8, and 12 weeks. Post-intervention, the experimental group exhibited a lower incidence of secondary injuries compared to the control group (0 vs. 3, P = 0.206). At the three-month follow-up, the experimental group demonstrated better outcomes in self-assessed aesthetics (P = 0.025) and QLQ-C30 scores (P = 0.041). The 3D-printed wig protector had a weight of 132.3 ± 11.98 g (ranging from 111 to 146 g), and none of the patients in the experimental group reported pain, pressure, itching, or discomfort during the first 30-min wear period. On average, patients wore the device for 2.01 ± 0.22 h in week 1, 5.71 ± 0.40 h in week 4, 6.04 ± 0.10 h in week 8, and 5.93 ± 0.47 h in week 12. While some patients reported minor skin indentation, pain, itching, or a sensation of stuffiness, no serious adverse events were observed.
Similar content being viewed by others
Introduction
Decompressive craniectomy (DC) was the removal of a portion of the skull to alleviate pressure on the brain tissue and blood vessels, thereby reducing intracranial pressure, restoring cerebral blood flow, and potentially saving the patient’s life1,2. It is commonly employed in the treatment of severe traumatic brain injury, large cerebral hemorrhages, and significant cerebral infarction complicated by cerebral edema, among other conditions. Following a decompressive craniotomy, patients generally require cranial repair within 3 to 6 months.
In the weeks following DC, patients often experience impaired coordination and balance, which increases the risk of falls and secondary injuries3. These challenges can significantly impact the patient’s quality of life. Additionally, cranial defects may alter the patient’s appearance, leading to psychological issues such as feelings of inferiority4. As a result, it is essential to implement protective measures post-surgery and before cranial repair. Zhao Yin and colleagues5 developed a helmet-like design aimed at shielding the head to prevent reinjury. However, this device is bulky and aesthetically unappealing. Furthermore, each patient’s head has unique shape and size characteristics, meaning traditional protective devices may not offer an optimal fit, leading to discomfort. Several studies have explored the use of 3D-printed protective braces for patients with skull defects. These braces are designed to secure external elastic mesh caps or bandages, offering effective protection. However, these devices often suffer from poor comfort, inadequate fixation, and may negatively affect personal appearance2. Additionally, due to the rigidity of 3D-printed materials, 20% of patients reported temporary skin indentation. Given the limitations of 3D-printed materials, the safety of 3D printed headgear for the protection of patients with DC remains a critical consideration. therefor, we developed a personalized 3D-printed wig protection device with a positioning membrane for patients following DC. The feasibility, safety, and scalability of this approach will require further investigation.
Patients and methods
Study design
This single-center trial was conducted from February 1, 2023, to December 31, 2023. The study adheres to the ethical principles outlined in the Declaration of Helsinki and received approval from the Hospital Ethics Committee: [2023] Lun Shen Zi (LW-96). Trial registration: Chinese Clinical Trial Registry, ChiCTR2400092577 (19/11/2024). All participants were informed about the study and provided written informed consent prior to inclusion.
Selection of patients
Seventeen patients who underwent DC at the Department of Neurosurgery, Binzhou Medical University Hospital, Shandong Province, were selected as participants .
Inclusion criteria: (1)Age 18–80 years. (2)Diagnosis of acute ischemic stroke (AIS), cerebral hemorrhage (ICH), traumatic brain injury (TBI), subarachnoid hemorrhage (SAH), or benign or malignant brain tumors, as confirmed by CT or MRI.(3) Postoperative CT imaging data available. (4) Wound healing after DC surgery. (5) Patients who were able to communicate effectively. (6) Informed consent was obtained from either the patients or their family members.
Exclusion criteria: (1) Patients with disturbances of consciousness. (2) Patients with severe organ dysfunction (heart, liver, kidney, etc.). (3) Active malignant tumour. (4) Severe anxiety or depression. (5) Presence of wound swelling or skull flap protrusion following DC surgery. (6) Scalp infection.
Study methods
(1) Fabrication of the 3D-Wig Protection Device (3D-WPD):
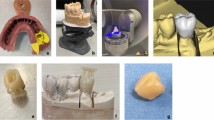
Initially, we acquired CT data of the patient’s head and imported the first postoperative CT image into computer software. The region of design for 3D head reconstruction was designated (Fig. 1). Along with the location, size, and shape of the skull resection, the shape and size of other parts of the head, as well as the thickness of the patient’s scalp, were considered. Based on scalp thickness measured during surgery and clinical experience, the original skull was expanded by 2 cm.Using the 3D Slicer image processing platform, the patient’s imaging data was processed and a 3D head model was digitally designed. The final model was saved in STL format. This file was imported into the 3D printing software (UltiMaker Cura), where model parameters, supports, and placement were adjusted. The model was then automatically sliced and sent to the 3D printer (CREALITY CR-M4, China). PLA (CREALITY) resin material was heated, melted, and extruded layer by layer. After printing, the model supports were removed, leaving the personalized head model (Fig. 2).
3D printed model of the patient’s head. Among them, the red is the skull defect of the patient.
Making process of 3D-WPD. Through processing, the simulated normal head of the patient is printed 3. Positioning film production process on 3D printing model: 4. Pruning of positioning film 5, 6 and 7. Respectively wig and positioning film inlay form the front, back and inside of the protective device.
The positioning film (R101, Guangzhou Coleridi Medical Equipment Co., Ltd.) was placed in water heated to 65–70 °C to soften. Once the film became transparent and soft, it was removed, excess moisture wiped off, and the film was pulled over the 3D printed head model and molded into shape. The film was then cured at room temperature, marked, and trimmed flat (Fig. 2 3 and 4). If the molding process was unsuccessful, it could be remolded in water. The average time for molding the positioning film was 9.90 ± 2.23 min, with a range of 7 to 14 min.
The positioning film is a special synthetic polymer polyester materials. It can be softened by heating for 2–5 min, allowing it to stretch with good compliance and adhesion. After molding, the film cures in 3–5 min at room temperature and exhibits properties such as light weight, minimal shrinkage, high strength, breathability, waterproofing, and biodegradability. The material is non-toxic, odorless, non-irritating to the skin, and safe for use. Finally, a wig was sewn by hand onto the positioning film, ensuring compatibility with the wig and meeting the aesthetic preferences of the patient (Fig. 2 5,6 and 7).
(2) Personalised 3D-WPD fitting.
From February 1, 2023, to June 30, 2023, nine patients who underwent DC were selected as the control group. Following suture removal post-surgery, patients and their families were advised to remain vigilant in protecting the skull defect.
Eight patients who underwent decompressive craniectomy from July 1, 2023, to December 31, 2023, were enrolled in the experimental group.The 3D-wig protection device (3D-WPD) was introduced 2–3 days after suture removal. During the first 30 min of wear, caregivers closely monitored the patients and inquired about any sensations of pain, pressure, or discomfort. The patients’ families were instructed to:(1) Learn proper techniques for donning and doffing the device.(2) Monitor the local skin condition.(3) Maintain the cleanliness of the 3D-WPD.(4) Gradually integrate the wearing of the WPD into outdoor activities according to the prescribed schedule. The wearing time began at 15 min, twice a day, and was gradually increased by 15–30 min per day until the wearing duration reached 2–3 h per session, to be sustained for 3 months. Patients were asked to document daily wear time and any occurrences of skin indentation, pain, itching, or a feeling of stuffiness. Upon discharge, patients received a written health education form, a prescribed wearing schedule, and the researcher’s contact details (phone number or WeChat) for further inquiries.
Baseline demographic data for both groups were recorded following suture removal.
(3) Follow-up Period (12 Weeks).
Video follow-up sessions were conducted by the researchers at 1, 4, 8, and 12 weeks post-fitting, during which both objective and subjective results were documented at each visit, including:(1)Presence or Absence of Re-injury to the Cranial Defect. (2)Daily wearing time (in hours).(3)Severity of skin indentation, pain, itching, and sulkiness (scored from 0 to 10). (4)Patient’s subjective assessment of aesthetics, rated from 0 to 10, with higher scores indicating a more favorable evaluation.(5)Quality of life, assessed using the Quality of Life Scale (QLQ-C30), which includes 30 items across 4 domains: symptoms, function, general health, and single items.The first 28 items were scored on a scale from 1 to 4, while the last 2 items were scored from 1 to 7, resulting in a total score of 126. Lower scores indicate poorer quality of life6.
Outcome measures
The primary outcomes included:
-
Secondary injury to the cranial defect,
-
Subjective assessment of aesthetics,
-
Quality of life in both groups.
Secondary outcomes included:
-
Pain, pressure, or other discomfort within the first 30 min of initial wear,
-
later: Pain, itching, and swelling.
Quality assurance
-
1.
Study Design: This is a non-randomized controlled study in which participants are divided into control and experimental groups based on time nodes to minimize interference from other factors.
-
2.
Standardized Data Collection Tools: Uniform, standardized assessment tools, including aesthetic subjective assessment scales and the Quality of Life Measurement Scale (QLQ-C30), are employed to ensure the reliability and validity of the collected data.
-
3
Unified Training: Participants involved in data collection are given standardized training to ensure they are familiar with the assessment tools, including proper usage methods and scoring criteria.
-
4.
Regular Supervision and Evaluation: Ongoing supervision is conducted to ensure the consistency and accuracy of data collection across all phases of the study.
-
5.
Support and Communication: Participants are provided with the researcher’s contact information (phone number or WeChat). A unified record book is developed to guide patients or their family members in documenting data accurately and in a timely manner. Regular monitoring and evaluation of patient wear is performed during follow-ups to ensure compliance with wearing protocols.
-
6.
Adverse Event Documentation: Any adverse events are carefully recorded and analyzed to inform potential improvements in future stages of the study.
Statistical analysis
Data analysis and processing were performed using SPSS 22.0 software. Count data were expressed as frequencies and percentages, and comparisons between the two groups were conducted using either the chi-squared test or Fisher’s exact test, depending on the data characteristics. A homogeneity of variance test was conducted for the measurement data, which were expressed as ¯χ ± s. The rank sum test was used for assessing heterogeneity of variance. Statistical significance was defined as P < 0.05.
Results
Patient recruitment
From February 1, 2023, to December 31, 2023, a total of 25 patients underwent DC. Of these, 17 patients met the inclusion and exclusion criteria and completed follow-up, with 9 patients in the control group and 8 patients in the experimental group (Fig. 3).
Flow diagram of the study protocol.
Comparison of baseline patient characteristics
The average age of the patients was 53.18 ± 8.26 (37–66)years. Among the participants, 13 were male (76.47%) and 4 were female (23.53%). The causes of the skull defect were as follows: acute ischemic stroke in 2 patients (11.76%), cerebral hemorrhage in 3 patients (17.65%), traumatic brain injury in 7 patients (41.18%), and subarachnoid hemorrhage in 5 patients (29.41%).
A comparison of the baseline data between the two groups (Table 1) showed no significant differences in age, gender, cause, or location of the cranial defect (P > 0.05).
Secondary skull defect injury in the two groups
During the three-month follow-up period, three patients in the control group suffered from secondary injuries of varying degrees of severity. One patient fell while out unaccompanied by a family member, caused by dizziness. Another patient had a head collision with a family member, while the third fell and struck their head against a wall, also due to dizziness. All three sustained minor injuries and reported some discomfort, but it was manageable. None of the three patients sought medical evaluation or underwent imaging. In contrast, none of the eight patients in the experimental group experienced any re-injury (Table 2).
Aesthetic subjective assessment of patients in the two groups
Before wearing the 3D-WPD, there was no significant difference in the subjective aesthetic ratings between the two groups (P = 0.149). After using the 3D-WPD, the experimental group reported a higher aesthetic self-rating (7.88 ± 0.64 min) compared to the control group (2.33 ± 1.22 min), with this difference being statistically significant (P = 0.025) (Table 2).
Quality of life scores of patients in the two groups
The Quality of Life was assessed using the QLQ-C30 at 7 days and 3 months post-surgery. There were no statistically significant differences between the two groups at 7 days after surgery (P = 0.798). However, a significant difference was observed between the two groups at three months (P = 0.041) and six months after surgery (P < 0.001) (Table 2).
Observation index results of the experimental group
Basic situation of patients wearing 3D-WPD in the experimental group
All 10 patients in the experimental group completed the initial 30-min wear period with the 3D-WPD. None of the patients reported experiencing pain, pressure, itching, or discomfort during this time. One patient voluntarily withdrew from the study at week 4, and another patient was lost to follow-up after week 8. Ultimately, 8 patients successfully completed 12 weeks of follow-up. The ages of the 8 patients ranged from 37 to 62 years, with a mean age of 53.13 ± 8.92 years. The group consisted of 1 female (12.5%) and 7 males (87.5%). The causes of the skull defects were as follows: 1 case of acute ischemic stroke (12.25%), 1 case of intracerebral hemorrhage (12.25%), 4 cases of traumatic brain injury (50%), and 2 cases of subarachnoid hemorrhage (25%). Regarding the location of the skull defects, 4 patients had left-sided defects (50%) and 4 had right-sided defects (50%). Among them, 5 underwent cranial repair surgery three months later. Three patients continued wearing the 3D-WPD beyond the 3-month follow-up period. The average weight of the headgear with the wig was 132.3 ± 11.98 g, with a range from 111 to 146 g. Detailed information about the patients and their use of the wig headgear is presented in Table 3.
Experimental group patients wearing wig protection device time
The mean wearing times for the 3D-WPD among the eight patients who completed the 12-week follow-up were 2.01 ± 0.22 h (Week 1), 5.71 ± 0.40 h (Week 4), 6.04 ± 0.10 h (Week 8), and 5.93 ± 0.47 h (Week 12) (Table 4 and Fig. 4). Additionally, one patient reported 0 h of wearing time after week 8, citing summer outdoor activities, head sweating, and intolerance to the wig as reasons for non-compliance. Another subject reported 0 h of wear after week 4, though the reasons were not explained in detail.
Average daily wearing time of wig protection device for patients in different time periods.
Adverse events associated with wig wear
Throughout the study, some patients in the experimental group experienced mild adverse events such as skin indentation, pain, itching, and a burning sensation (Table 5). However, no serious adverse events were reported. The occurrence of these mild adverse events may be attributed to factors such as excessive wearing time, hot summer weather, and other related conditions.
Discussion
The rapid development of 3D printing technology has significantly impacted various fields of medicine, especially in neurosurgery, where it has shown great promise in improving patient outcomes. The primary objective of this study was to assess the efficacy of the 3D-WPD in patients who had undergone DC. The results suggest that the 3D-WPD not only effectively protects the cranial defect site but also improves patients’ aesthetic self-ratings and quality of life.
3D printing technology has revolutionized medical practice due to its precision, personalized approach, and ability to rapidly produce customized solutions. In neurosurgery, it has been used extensively for surgical planning, creating models for preoperative simulations7,8,9, and designing custom implants that improve surgical outcomes and patient satisfaction. These advancements have allowed for more personalized, effective treatments and enhanced patient care.
In the field of surgery, 3D printing technology has been extensively utilized in surgical planning and simulation. Utilizing a patient’s medical image data, doctors can create precise three-dimensional models. These models enable the design and printing of surgical navigation molds, which assist in hematoma puncture and drainage, thereby achieving personalized and precise medical treatment10. Furthermore, 3D printing technology has found preliminary application in preoperative localization of meningiomas in primary hospitals. Following surgery, patients exhibit no significant hematoma, brain edema, or nervous system symptoms, indicating favorable surgical outcomes11. Additionally, 3D-printed, customized implants, such as vascular grafts, significantly enhance surgical results and patient satisfaction due to their seamless anatomical fit12.
In the context of head protection following decompressive craniectomy, traditional methods of protecting the skull defect are often inadequate. This study employed 3D printing technology to create a more personalized, lightweight, and effective protective device that not only shields the defect but also addresses the aesthetic concerns of patients.
The 3D-WPD is made from safe, non-irritating materials, ensuring patient comfort. Its lightweight, breathable, and biodegradable properties offer both functional and environmental benefits. The device conforms to the patient’s unique cranial contours, ensuring a secure fit while providing adequate protection against external impacts, preventing re-injury. The customization of the device, including wig selection based on the patient’s preferences, enhances both protection and aesthetics, contributing to an improved quality of life.
The results showed that the control group experienced secondary injuries during the follow-up period, while none of the patients in the experimental group experienced re-injury. This suggests that the 3D-WPD is effective in protecting the cranial defect and minimizing the risk of further harm, which is consistent with previous research demonstrating the protective benefits of such devices3.
Aesthetic outcomes are particularly important for patients following cranial surgeries, as changes in appearance can affect self-esteem and social relationships5. In this study, patients in the experimental group rated their appearance significantly higher than those in the control group(P < 0.05), indicating that the 3D-WPD offers a meaningful improvement in aesthetic outcomes. This finding is significant as it highlights the potential for 3D-WPDs to not only protect the skull but also positively influence the patient’s emotional and psychological well-being.
Furthermore, the quality of life, as measured by the QLQ-C30 scale, was significantly better in the experimental group, particularly three months post-surgery.Patients often experience physical and psychological stress following surgery, which may manifest in the form of unexpected injuries, discomfort, and changes in appearance13. This indicates that the device has a positive impact on patients’ overall well-being, providing both functional and psychological benefits.
Three patients in the experimental group continued to wear the device for three months, until skull repair at six months. Consequently, we collected quality of life (QOL) scores for these three patients at the six-month mark and compared them with those of the control group. Significant differences in QOL were observed between the two groups. However, due to the small sample size, there may be some bias in our results. In future studies, we plan to further expand the sample size and conduct long-term follow-up.
In this study, the experimental group consisted of 10 patients, and the weight of the wearable protection device (WPD) was 132.3 ± 11.98 g, significantly lighter than that of the rigid helmet4. Despite the promising results, some patients did experience minor adverse effects such as skin indentation, pain, itching, and a burning sensation. These adverse events were likely related to prolonged wear, sweating, and temperature variations. Future refinements to the device, particularly in terms of improving breathability and comfort, could help mitigate these issues. Additionally, longer follow-up periods will be necessary to assess the long-term effects of the 3D-WPD.
One limitation of the study is the small sample size, which may have affected the generalizability of the findings. The study was also short-term, and the long-term safety and effectiveness of the device were not fully explored. Expanding the sample size and extending the follow-up period in future studies would provide a more comprehensive evaluation of the device’s impact.
In terms of cost, the estimated cost of producing a customized 3D-WPD is around $800. While this may seem high initially, it is important to consider the potential benefits the device provides in terms of protecting the patient from secondary injury, improving their aesthetics, and enhancing their overall quality of life. Moreover, as the design and production of these devices become more standardized, costs are likely to decrease, making them more accessible to a larger number of patients.
The 3D-WPD represents an effective, personalized solution for protecting patients following decompressive craniectomy. It improves both aesthetic outcomes and quality of life while preventing secondary head injuries. Future studies with larger sample sizes and longer follow-up periods are needed to further evaluate the long-term benefits and refine the device for broader clinical use.
Conclusion
In patients who have undergone decompressive craniotomy surgery, the 3D printing-based wig protection device demonstrates a significant effect in reducing the risk of secondary injury to cranial defects while also enhancing the appearance and overall quality of life. This study offers a safe and effective treatment option, highlighting the practical implications for patient care in the medical field. However, the study’s results may be constrained by the small sample size and limited follow-up period. Future research with larger sample sizes and extended follow-up durations is necessary to further validate these findings. Additionally, for patients who experience issues like excessive sweating or discomfort from heat, further refinements to the device may be needed. Long-term use of the device may also result in mild adverse events, such as skin indentation or pain, which should be addressed in future developments to improve patient comfort and prevent complications.
Data availability
Data are available on request to the authors. Contact the first author (Shasha Ma), E-mail: byfymss@126.com.
References
Zhang, C. & Wang, Y. H. Research status of surgical treatment of severe craniocerebral trauma based on the concept of controlled decompression. Chin. J. Neurosurg. 37(3), 322–324. https://doi.org/10.3760/cma.j.cn112050-20200601-00312 (2021).
Hatamleh, M. M. Contemporary review on craniectomy and cranioplasty; Part 1: Decompressive craniectomy. J. Craniofac. Surg. 33(3), 838–841. https://doi.org/10.1097/SCS.0000000000008041 (2022).
Shi, Z. J. et al. Application value of personalized 3D printing in brain protection after decompressive craniotomy. Natl. Med. J. China https://doi.org/10.3760/cma.j.cn112137-20211019-02314 (2022).
Chua, K. S. G. et al. 3D-printed external cranial protection following decompressive craniectomy after brain injury: A pilot feasibility cohort study. PLoS One. 16(10), e0258296 (2021).
Zhao, Y. & Hu, X. Y. Preliminary study on clinical application effect of a skull defect cap. Med. Higher Voc. Ed. Mod. Nurs. 4(05), 447–449. https://doi.org/10.3969/j.issn.2096-501X.2021.05.020 (2021).
Reygagne, E., Du Boisgueheneuc, F., Berger, A. & Ingrand, P. Examining the inter hemispheric transfer time test: A new computerized cognitive test to incorporate into therapeutic strategy for patients with brain metastases? A pilot study. Clin. Transl. Radiat. Oncol. https://doi.org/10.1016/j.ctro.2018.11.006 (2018).
Ganapathy, A. et al. Guide for starting or optimizing a 3D printing clinical service. Methods 206, 41–52. https://doi.org/10.1016/j.ymeth.2022.08.003 (2022).
Li, Z. et al. Three-dimensional printing models improve understanding of spinal fracture–A randomized controlled study in China. Sci. Rep. 5, 11570 (2015).
Vukicevic, M., Mosadegh, B., Min, J. K. & Little, S. H. Cardiac 3D printing and its future directions. JACC Cardiovasc. Imaging 10(2), 171–184. https://doi.org/10.1016/j.jcmg.2016.12.001 (2017).
Wang, Q. et al. Application of a 3D-printed navigation mold in puncture drainage for brainstem hemorrhage. J Surg. Res. 245, 99–106. https://doi.org/10.1016/j.jss.2019.07.026 (2020).
Chen, Z. et al. Preliminary application of three-dimensional printing technique in preoperative localization of meningioma in primary hospitals. J. Craniofac. Surg. 32(5), 1796–1799. https://doi.org/10.1097/SCS.0000000000007509 (2021).
Sonaye, S. Y. et al. Patient-specific 3D printed Poly-ether-ether-ketone (PEEK) dental implant system. J. Mech. Behav. Biomed. Mater. 136, 105510. https://doi.org/10.1016/j.jmbbm.2022.105510 (2022).
Lin, G. F. et al. Application effect of 3D printing individualized protective brace in patients with skull defect. China Mod. Med. https://doi.org/10.3969/j.issn.1674-4721.2021.16.065 (2021).
Acknowledgements
We would like to thank DeepL (www.deepl.com/) for English language editing.
Funding
This work was supported by the Natural Science Foundation of Shandong Province (Grant no. ZR2023MH378) and the Medicine and Health Project of Shandong Province (grant no. 202314050473).
Author information
Authors and Affiliations
Contributions
Research design: S.S.M and H.M.X, Model making: C.L.L and Q.B.W, Picture preparation: C.L.L, Y.G and X.X.L, Data collection and analysis: Y.G and X.X.L, Thesis writing: S.S.M and Y.G, Paper revision: H.M.X and C.L.L. All authors provided critical feedback and contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study was submitted for review and received approval from the Hospital Ethics Committee: [2023] Lun Shen Zi (LW-96). The committee conducted a comprehensive evaluation of the study protocol, informed consent form, data collection, and analysis methods to ensure compliance with ethical standards and regulations. Strict measures will be in place to protect participants’ personal information and research data, ensuring that these are not disclosed to unauthorized third parties. Study Risks: While the study aims to evaluate the safety and efficacy of 3D-printed wig protection devices, certain risks may arise. The investigators will closely monitor participants’ health and intervene as necessary to safeguard their well-being. Use and Release of Study Results: The investigators ensure that the study results will be used and released ethically, avoiding any misleading conclusions or harm to participants’ rights and interests. For detailed research steps, refer to the Research Protocol and the Chinese Clinical Trial Registry link https://www.chictr.org.cn/bin/project/edit?pid=228588.
Informed Consent
It involves details about the study’s purpose, methods, risks, potential benefits, participants’ rights, data confidentiality measures, and the right to withdraw from the study. The investigator provides a thorough explanation of the study and the consent form to potential participants and their families to ensure they fully understand the study and voluntarily sign the consent form.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ma, S., Li, C., Gu, Y. et al. Application of a 3D printed wig protection device after decompressive craniectomy. Sci Rep 15, 9545 (2025). https://doi.org/10.1038/s41598-025-94246-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94246-0
Keywords
This article is cited by
-
A personalized 3D printed cranial shield using mirror-image modeling: design and clinical assessment
3D Printing in Medicine (2025)