Abstract
Total anomalous pulmonary venous connection (TAPVC) is a rare congenital heart defect where the pulmonary veins abnormally connect to the systemic venous circulation, rather than the left atrium (LA). This study retrospectively analyzed 48 children diagnosed with TAPVC from January 2014 to January 2024 to investigate how specific echocardiographic and morphological characteristics impact prognosis. Perioperative records, including echocardiography, computed tomography angiography (CTA) images, surgical details, and patient outcomes, were reviewed. Data were statistically analyzed using tools such as t-tests, Mann-Whitney U tests, χ² tests, and logistic regression. Key factors examined included age at surgery, anatomical subtype of TAPVC, presence of preoperative obstruction, surgical techniques used, and postoperative complications. By identifying the impact of these variables on survival rates and hospital recovery, the study aims to improve risk stratification and management strategies for better outcomes in children with TAPVC.
Similar content being viewed by others
Introduction
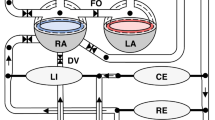
Total anomalous pulmonary venous connection (TAPVC) is a relatively rare and severe cyanotic congenital heart disease, occurring in approximately 0.5-2% of congenital heart diseases1. In this condition, all pulmonary veins fail to connect to the left atrium (LA); instead, they connect to the right atrium (RA) through systemic veins or drain directly into the RA. Based on the drainage pathways, TAPVC can be divided into four types: supracardiac, cardiac, infracardiac, and mixed. The supracardiac type is the most common, representing about 43% of all cases. Additionally, about 25-50% of patients with TAPVC have associated pulmonary venous obstruction (PVO)2,3,4.
The clinical symptoms of children with TAPVC are closely related to the presence of PVO, these symptoms can range from mild pulmonary hypertension to severe pulmonary congestion and acidosis5. In case where PVO is present, emergency surgery is required. Without timely intervention, most affected neonates do not survive the neonatal period.
Advancements in surgical techniques, diagnostic procedures, and perioperative care have significantly reduced mortality rates in TAPVC in recent years. Various studies have focused on prognostic factors such as preoperative general conditions (age, weight, oxygen saturation, pH value), surgical techniques, and cardiopulmonary bypass time6. While much of the research has concerned on the type and presence of PVO, less attention has been given to specific echocardiographic parameters.
This study aims to bridge that gap by retrospectively analyzing the correlation between detailed echocardiographic measurements and the prognosis of children with TAPVC. By understanding these relationships, better predictive models and tailored management strategies can be developed, potentially improving outcomes for patients with TAPVC.
Results
Clinical characteristics
In this cohort of 48 children diagnosed with TAPVC, the median age was 35 days (range 1-2615days), and the median weight was 3.8 kg (range 1.5–13.5 kg). The median surgical duration for these cases was 215 min, with a range from 135 to 485 min. The gender distribution included 34 males (approximately 70.8%) and 14 females (approximately 29.2%). Among of TAPVC connections, 15 cases (31.3%) were supracardiac, 20 cases (41.7%) were cardiac, 10 cases (20.8%) were infracardiac, and 3 cases (6.3%) had mixed connections. These details of the patient demographics and clinical characteristics in the study were shown in Table 1.
The drainage pathway of TAPVC
All 48 cases underwent echocardiographic examination to evaluate the pulmonary venous drainage pathways, the presence of obstruction, associated malformations, and cardiac function. Among the 15 supracardiac types, nine cases drained into the innominate vein and six into the superior vena cava (SVC) (Fig. 1). There were 13 single vertical vein and 2 cases of double vertical veins. In the 20 cardiac cases, 17 CPVs drained into the RA via the coronary sinus (CS) (Fig. 2), two CPV drained directly into the RA, and one had four pulmonary veins draining separately into the RA. Among the 10 infracardiac cases, eight cases had CPVs that drained into the portal vein via a vertical vein, one drained into the hepatic vein, and one drained into the inferior vena cava via ductus venous (Fig. 3). In the three cases mixed-type cases, all of them combines both supracardiac and cardiac types (Fig. 4).
The echocardiography of supracardiac TAPVC. (A) The left heart is extreme small. (B) The pulmonary veins drain into the CPV. (C) The CPV drains into the innominate vein via vertical vein. (D) An obstruction was detected at the drainage site.
The echocardiography of cardiac TAPVC. (A) The CS is dilated behind the small LA. (B) The CPV drains into the CS.
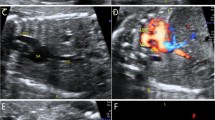
The echocardiography of infracardiac TAPVC. (A) The left heart is small, the posterior wall of the LA is smooth, and the CPV is behind the LA. (B) The pulmonary veins drain into the CPV. (C) The vertical vein goes down then drains into duct vein, the color doppler shows turbulent flow at the drainage site. (D) A non-phasic flow was detected at the drainage site.
The echocardiography of mixed TAPVC. (A) The CPV drains into RA via CS. (B) The other pulmonary vein drains into innominate vein via vertical vein.
Outcome of the surgery
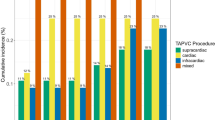
Postoperative death occurred in 14 (29%,14/48) cases, distributed as follows: supracardiac (26.7%,4/15), cardiac (20%,4/20), infracardiac (40%,4/10), mixed (66.7%,2/3). Postoperative PVO occurred in 14 cases (29%, 14/48), with the following distribution: supracardiac (20%,3/15), cardiac (10%,2/20), infracardiac (60%,6/10), mixed (100%,3/3). Among the 14 deaths, 11 patients had preoperative PVO, while three did not. Other postoperative complications included diaphragmatic paralysis in 2 cases, SVC obstruction in 1 case, and bronchial stenosis in 1 case.
The echocardiographic characteristics of TAPVC
The postoperative outcomes of patients with TAPVC were analyzed concerning various preoperative anatomical and physiological parameters, including drainage pathways, the diameter of the CPV, the size of the LA, the patent foramen ovale (PFO) or atrial septal defect (ASD), the presence of a patent ductus arteriosus (PDA), the degree of pulmonary hypertension, and the associated intracardiac malformations. The significant findings revealed that a smaller size of the LA and preoperative PVO were strongly associated with higher risks of postoperative PVO and death (P < 0.05) (Tables 2 and 3). When the LA is less than or equal to 0.85 cm, the risk of poor prognosis increases. Conversely, the drainage pathway, pulmonary arterial systolic pressure (PASP), CPV diameter, PFO, and the presence of PDA did not show significant associations with postoperative outcomes.
Discussion
Echocardiography is the first line diagnostic modality for congenital heart diseases, including TAPVC. However, there are some shortcomings of echocardiography. Due to the narrow diameter or tortuous pathways of the pulmonary veins or vertical veins, echocardiography may not visualize them. In contrast, computed tomography angiography (CTA) has advantages over echocardiography in visualizing the obstructed veins and displaying atypical vessels7. In our study, there were two cases misdiagnosed by echocardiography: one case of supracardiac was misdiagnosed as the mixed type (supracardiac and intracardiac), and another case of supracardiac TAPVC was misdiagnosed as partial supracardiac. Both were correctly diagnosed by CTA. Echocardiography offers distinct advantages, such as assessing blood flow velocity in pulmonary and vertical veins, evaluating cardiac function, and measuring pulmonary artery pressure without radiation exposure- capabilities CTA lacks. Additionally, echocardiography excels at observing the size of the PFO or ASD and detecting the presence of a PDA. However, CTA is superior in accurately evaluating the course and number of anomalous veins, making it particularly effective for visualizing atypical vessel structures8,9. Therefore, combining both echocardiography and CTA provides a comprehensive preoperative evaluation by leveraging the strengths of each modality.
The drainage pathway of pulmonary veins in TAPVC is a debated prognosis factor. Most studies indicate that infracardiac and mixed-type pathways pose higher risks for death and postoperative PVO, while supracardiac and cardiac types generately have better outcomes6,10,11,12. However, a retrospective in Toronto found that the cardiac type is a risk factor for postoperative death3. Additionally, patients with pulmonary veins draining into the coronary sinus (CS) are susceptible to postoperative PVO due to the accumulation of the neointimal tissue during intracardiac repair13. Specific morphological features are associated with postoperative PVO in patients with cardiac TAPVC, the confluence to total pulmonary vein ratio, length of drainage route, and pulmonary veins return to the RA roof are predictor for postoperative PVO14. This complexity underscores the need for tailored surgical and postoperative strategies to enhance patient prognosis. Mixed-type TAPVC features a wide anatomic variation including bilateral symmetrical asymmetrical connections, bilateral asymmetrical connections, and bizarre anatomic variants, with the later significantly increasing the risk of death, reportedly by 5.85 times15. Supracardiac TAPVC, the most common subtype, typically involves drainage into the innominate vein via left vertical vein (type-Ia), or into SVC or the right azygos vein (type-Ib). The type Ib connection is recognized as a risk factor for postoperative PVO and death16. Given this morphologic heterogeneity, individualized treatment and surgical planning are essential to mitigate risks and optimize patient outcomes.
Complexed cardiac lesion that needs staged intervention is an independent risk factor for mortality. However, when the associated cardiac lesion was repaired during TAPVC surgery, it was not a risk factor for death17. The intracardiac malformations in our study were VSD, PDA and mild PS, which can typically be managed effectively during the correction of anomalous pulmonary venous connection. Additionally, the heterotaxy syndrome has been recognized as a predictor of postoperative mortality in TAPVC18. Therefore, in this study, the associated cardiac malformation is not a significant prognosis factor of postoperative mortality.
Our findings reaffirm previous research that preoperative PVO significantly impacts survival rates in patients undergoing surgical correction of TAPVC3,6,19,20,21. Specifically, with PVO, only 6 out of 15 cases survived (40%), whereas without PVO, 26 out of 33 cases survived (approximately 78.8%), a statistically significant difference underscoring the increased risk associated with PVO. These results highlight the necessity for early detection and management of PVO, meticulous preoperative evaluation, and possibly modified surgical techniques to improve outcomes. This data is crucial for clinicians to refine protocols and enhance patient care strategies, aiming for higher survival rates and better overall outcomes in TAPVC surgeries.
PVO, especially when manifested during the fetal period, critically impacts the pulmonary vascular bed, leading to increased pulmonary vascular pressure, vascular remodeling, or even atresia of the pulmonary veins22,23. The intricate anatomy of PVO, which can be due to intrinsic factors within the veins or extrinsic factors like surrounding structures or a dilated heart, adds complexity to its management24. PVO may present as segmental stenosis or diffuse narrowing, at various locations and involving single or multiple sites, making accurate diagnosis challenging13,25,26. Advanced imaging techniques such as CTA are superior to echocardiography in identifying these irregularities, underscoring the need for early and precise diagnostic interventions. To improve clinical outcomes, further research is necessary to optimize the assessment and management strategies for anomalous pulmonary veins in TAPVC patients.
In patients with TAPVC, the size of LA is crucial for assessing the development of the left heart, as blood flow to the left heart depends on right-to-left shunts, primarily including ASD, VSD, or PDA. While VSD and PDA often have bidirectional shunts, ASD typically involves a right-to-left shunt, with larger shunts facilitating better left heart filling and development. A small LA may lead to left heart overload postoperatively, poor pulmonary venous drainage, pulmonary congestion, and hypertension. Accurate preoperative evaluations of LA size and shunt sizes are necessary for surgical guidance and prognosis assessment. Our study highlighted that a small LA is an independent risk factor for death post-surgery, though the small sample size rendered ASD or PFO diameters non-significant as postoperative death risk factors.
Postoperative PVO is the major complication and the reason for reoperation, it also has impact on long term outcome. So it’s urgent to prevent PVO after TAPVC repairment. The sutureless repair has been adopted to reduce postoperative PVO since 1990. But the contribution of sutureless technique in preventing subsequent PVO remain controversial. Its benefits over conventional technique needs further confirmation6,27.
Methods
Study population
This retrospective study analyzed data from 48 children diagnosed with TAPVC by echocardiography and confirmed by surgery at Fujian Maternal and Children’s Hospital and Fujian Children’s Hospital from January 2014 to January 2024. The study included children potentially with ASD, PFO, VSD, PDA, and mild PS, while excluding those with functionally univentricular circulation or atrial isomerism. This study was approved by the local ethics committee at Fujian Maternal and Children’s Hospital.
Ethics statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Fujian Maternal and Child Health Hospital. The study subjects as well as their parents /legal guardians were informed about the study details and gave written consents to participate.
Echocardiographic equipment and assessment
Echocardiographic imaging was performed by Philips IPEIQ 7C and Philips IE33 ultrasound diagnostic system with a 3-8 MHz phased-array probe. The sonographers assessed the drainage pathways of pulmonary veins, the presence of any obstruction, and the associated cardiac lesions.
A diagnosis of preoperative PVO was made based on echocardiography data indicating a non-phasic flow velocity greater than 1.5 m/s13. LA maximum diameter was measured at the end systole on the parasternal long-axis view. The ASD or PFO diameter was measured on the subcostal long-axis view. The CPV was measured on the maximum section. The PASP was evaluated by tricuspid regurgitation and divided into mild (30-50mmHg), moderate (51-70mmHg) and severe (> 70mmHg) pulmonary hypertension.
Statistical analysis
The Shapiro-Wilk test was used to determine if the data followed a normal distribution. For normally distributed data, the mean ± standard deviation was used, and t-tests were performed to compare differences between groups. For non-normally distributed data, the median and interquartile range were used, and the Mann-Whitney U test was performed to compare differences between groups. Count data were expressed as frequencies (percentages) and compared between groups using χ2 tests or Fisher’s exact tests. Univariate and multivariate logistic regression analyses were conducted to assess the association between various factors and adverse outcomes. All statistical analyses were performed using R 4.3.2 software, with α = 0.05 as the test level and P < 0.05 considered statistically significant.
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Abbreviations
- TAPVC:
-
Total anomalous pulmonary venous connection
- LA:
-
Left atrium
- RA:
-
Right atrium
- PVO:
-
Pulmonary venous obstruction
- CPV:
-
Common pulmonary vein
- CS:
-
Coronary sinus
- SVC:
-
Superior vena cava
- ASD:
-
Atrial septal defect
- PFO:
-
Patent foramen ovale
- VSD:
-
Ventricular septal defect
- PDA:
-
Patent ductus arteriosus
- PS:
-
Pulmonary stenosis
- PASP:
-
Pulmonary arterial systolic pressure
- CTA:
-
Computed tomography angiography
References
Hoffman, J. I. Incidence of congenital heart disease: II. Prenatal incidence. Pediatr. Cardiol. 16, 155–165. https://doi.org/10.1007/BF00794186 (1995).
Husain, S. A. et al. Total anomalous pulmonary venous connection: factors associated with mortality and recurrent pulmonary venous obstruction. Ann. Thorac. Surg. 94, 825–831. https://doi.org/10.1016/j.athoracsur.2012.04.026 (2012). discussion 831 – 822.
Karamlou, T. et al. Factors associated with mortality and reoperation in 377 children with total anomalous pulmonary venous connection. Circulation 115, 1591–1598. https://doi.org/10.1161/CIRCULATIONAHA.106.635441 (2007).
Horer, J. et al. What to expect after repair of total anomalous pulmonary venous connection: data from 193 patients and 2902 patient years. Eur. J. Cardiothorac. Surg. 44, 800–807. https://doi.org/10.1093/ejcts/ezt129 (2013).
Files, M. D. & Morray, B. Total anomalous pulmonary venous connection: preoperative anatomy, physiology, imaging, and interventional management of postoperative pulmonary venous obstruction. Semin Cardiothorac. Vasc Anesth. 21, 123–131. https://doi.org/10.1177/1089253216672442 (2017).
Shi, G. et al. Total anomalous pulmonary venous connection: the current management strategies in a pediatric cohort of 768 patients. Circulation 135, 48–58. https://doi.org/10.1161/CIRCULATIONAHA.116.023889 (2017).
Shen, Q., Pa, M., Hu, X. & Wang, J. Role of plain radiography and CT angiography in the evaluation of obstructed total anomalous pulmonary venous connection. Pediatr. Radiol. 43, 827–835. https://doi.org/10.1007/s00247-012-2609-6 (2013).
Schmiady, M., Valsangiacomo-Buechel, E., Hubler, M. & Dave, H. Significance of preoperative imaging in children with total anomalous pulmonary venous connection. Eur. Heart J. 38, 2040. https://doi.org/10.1093/eurheartj/ehw515 (2017).
Matsuhisa, H. et al. Computed tomography-based surgical strategy for total anomalous pulmonary venous connection. Eur. J. Cardiothorac. Surg. 58, 237–245. https://doi.org/10.1093/ejcts/ezaa028 (2020).
Ji, E. et al. The outcomes of total anomalous pulmonary venous connection in neonates-10-year experience at a single center. Front. Cardiovasc. Med. 8, 775578. https://doi.org/10.3389/fcvm.2021.775578 (2021).
Kelle, A. M., Backer, C. L., Gossett, J. G., Kaushal, S. & Mavroudis, C. Total anomalous pulmonary venous connection: results of surgical repair of 100 patients at a single institution. J. Thorac. Cardiovasc. Surg. 139, 1387–1394e1383. https://doi.org/10.1016/j.jtcvs.2010.02.024 (2010).
Chen, L., Qiu, Z., Xu, F., Chen, X. & Chen, L. Clinical nomogram for predicting the prognosis of patients with pulmonary venous obstruction after total anomalous pulmonary venous connection repair. Front. Cardiovasc. Med. 9. https://doi.org/10.3389/fcvm.2022.733253 (2022).
Ricci, M. Management of pulmonary venous obstruction after correction of TAPVC: risk factors for adverse outcome. Eur. J. Cardiothorac. Surg. 24, 28–36. https://doi.org/10.1016/s1010-7940(03)00180-5 (2003).
Shi, G. et al. Cardiac-type total anomalous pulmonary venous return is not benign. J. Thorac. Cardiovasc. Surg. 165, 449–459e444. https://doi.org/10.1016/j.jtcvs.2022.03.036 (2023).
Chowdhury, U. K. et al. Mixed total anomalous pulmonary venous connection: anatomic variations, surgical approach, techniques, and results. J. Thorac. Cardiovasc. Surg. 135, 106–116. https://doi.org/10.1016/j.jtcvs.2007.08.028 (2008). 116 e101-105.
Liu, X. et al. Supracardiac total anomalous pulmonary venous connection type Ib: Morphology and outcomes. J. Thorac. Cardiovasc. Surg. 166, 193–200 e191. https://doi.org/10.1016/j.jtcvs.2022.10.008 (2023).
Seale, A. N. et al. Total anomalous pulmonary venous connection: morphology and outcome from an international population-based study. Circulation 122, 2718–2726. https://doi.org/10.1161/CIRCULATIONAHA.110.940825 (2010).
Domadia, S., Kumar, S. R., Votava-Smith, J. K. & Pruetz, J. D. Neonatal outcomes in total anomalous pulmonary venous return: the role of prenatal diagnosis and pulmonary venous obstruction. Pediatr. Cardiol. 39, 1346–1354. https://doi.org/10.1007/s00246-018-1901-0 (2018).
White, B. R. et al. Venous flow variation predicts preoperative pulmonary venous obstruction in children with total anomalous pulmonary venous connection. J. Am. Soc. Echocardiogr. 34, 775–785. https://doi.org/10.1016/j.echo.2021.02.007 (2021).
Kirshbom, P. M. et al. Preoperative pulmonary venous obstruction affects long-term outcome for survivors of total anomalous pulmonary venous connection repair. Ann. Thorac. Surg. 74, 1616–1620. https://doi.org/10.1016/s0003-4975(02)03935-8 (2002).
Sinzobahamvya, N., Arenz, C., Brecher, A. M., Blaschczok, H. C. & Urban, A. E. Early and long-term results for correction of total anomalous pulmonary venous drainage (TAPVD) in neonates and infants. Eur. J. Cardiothorac. Surg. 10, 433–438. https://doi.org/10.1016/s1010-7940(96)80111-4 (1996).
Seale, A. N. et al. Pulmonary vein stenosis: the UK, Ireland and Sweden collaborative study. Heart 95, 1944–1949. https://doi.org/10.1136/hrt.2008.161356 (2009).
Haworth, S. G. Total anomalous pulmonary venous return. Prenatal damage to pulmonary vascular bed and extrapulmonary veins. Br. Heart J. 48, 513–524. https://doi.org/10.1136/hrt.48.6.513 (1982).
Hoashi, T. et al. Intrinsic obstruction in pulmonary venous drainage pathway is associated with poor surgical outcomes in patients with total anomalous pulmonary venous connection. Pediatr. Cardiol. 36, 432–437. https://doi.org/10.1007/s00246-014-1031-2 (2015).
van de Wal, H. J. et al. Pulmonary venous obstruction following correction for total anomalous pulmonary venous drainage: a challenge. Eur. J. Cardiothorac. Surg. 6, 545–549. https://doi.org/10.1016/1010-7940(92)90006-j (1992).
van Son, J. A., Danielson, G. K., Puga, F. J., Edwards, W. D. & Driscoll, D. J. Repair of congenital and acquired pulmonary vein stenosis. Ann. Thorac. Surg. 60, 144–150. https://doi.org/10.1016/0003-4975(95)00325-f (1995).
Caldarone, C. A. et al. Relentless pulmonary vein stenosis after repair of total anomalous pulmonary venous drainage. Ann. Thorac. Surg. 66, 1514–1520. https://doi.org/10.1016/s0003-4975(98)00952-7 (1998).
Funding
This study was funded by Joint Funds for the innovation of science and Technology, Fujian province (Grant number:2023Y9377), Joint Funds for the innovation of science and Technology, Fujian province (Grant number:2023Y9378) and Sponsored by Fujian provincial health technology project (Grant number: 2023QNA057).
Author information
Authors and Affiliations
Contributions
Zongjie Weng designed and Shan Guo supervised the study. Xiaoying Xue worked on the literature review. Wen Ling focused on imaging acquisition, data collection, and analysis. Xiaoying Xue and Biying Huang performed the statistical analysis. Xiaoying Xue drafted the manuscript. Shan Guo worked on the editing. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xue, X., Ling, W., Wu, Q. et al. Prognostic factors in pediatrics TAPVC: a 10-year retrospective study. Sci Rep 15, 11734 (2025). https://doi.org/10.1038/s41598-025-94619-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94619-5