Abstract
In aquatic ecosystems, metal and ammonia pollution pose major concern as they contaminate the environment and induces toxicity in fish. The present study addresses the toxicity induced by chromium (Cr) and ammonia (NH3) toxicity in fish and investigates the potential of dietary curcumin in mitigating the effects of concurrent exposure to theses stressors in Pangasianodon hypophthalmus. Three isonitrogenous and isocaloric diets were formulated: a control diet (0% curcumin) and two curcumin-supplemented diets containing 0.1% and 0.2% curcumin. Four experimental groups were designed in a completely randomized design: (1) control, (2) concurrent exposure to Cr and NH3 toxicity and fed with control diet, (3) 0.1% curcumin with Cr and NH3 exposure, and (4) 0.2% curcumin with Cr and NH3 exposure. Fish fed with 0.2% curcumin diet, followed by the 0.1% curcumin diet under Cr and NH3 stress, exhibited significantly reduced cortisol levels compared to the control and Cr + NH3 groups. Similarly, the expression of HSP70 and iNOS genes in liver tissue was significantly downregulated in the 0.1% and 0.2% curcumin-fed groups compared to other groups. Concurrent exposure to Cr and NH3 led to a considerable increase in oxidative stress enzyme in liver and kidney tissues, including glutathione S-transferase (GST), catalase (CAT), and superoxide dismutase (SOD). However, dietary supplementation with 0.1% curcumin significantly reduced oxidative stress enzyme activities. The stressors markedly reduced acetylcholinesterase (AChE) activity, but supplementation with 0.1% curcumin restored AChE activity. The expression of stress-related genes such as cytochrome P450 (CYP450), caspase-3a (Cas3a), and tumor necrosis factor-alpha (TNF-α) was noticeably downregulated in the curcumin-fed groups, reducing the impact of Cr + NH3 stress. Furthermore, total immunoglobulin (Ig) levels and growth-related gene expression, including growth hormone (GH) and growth hormone receptor 1 (GHR1), were significantly upregulated in the 0.1% curcumin-fed group under Cr + NH3 stress compared to all other groups. Additionally, myostatin (MYST) gene expression was significantly downregulated in the 0.1% curcumin-fed group. Activities of cellular metabolic and digestive enzymes were significantly improved with curcumin supplementation, mitigating the adverse effects of Cr + NH3 stress compared to the control and other groups. Moreover, Cr bioaccumulation in different fish tissues was reduced in the 0.1% curcumin-fed group. This study highlights the potential of dietary curcumin in mitigating the adverse effects of concurrent Cr and NH3 exposure through gene regulation, thereby improving the physiological and productive performance of Pangasianodon hypophthalmus.
Similar content being viewed by others
Introduction
The aquatic ecosystem is house to various pollutants, including heavy metals, pesticides, ammonia, biological waste, and nano/microplastics1. These pollutants pose significantly threats to the life cycle of aquatic organism, including fish. Ammonia is a major water contaminant; primarily originating from agriculture runoff and decomposition of biological waste in aquatic ecosystem. It is highly toxic to aquatic animals because elevated NH4+ levels depolarizes neurons, displaces K+, activates the N-methyl-D-aspartate (NMDA) type glutamate receptor, and trigger an excessive influx of Ca2+, leading to cell death in the central nervous system2. Furthermore, total ammonia exists in two forms: ionized (NH4+) and un-ionized (NH3). The un-ionized form (NH₃) readily diffuses across biological membranes or induces toxicity by disrupting cellular processes3,4. According to Hoseini et al.5, uneaten feed and fish excrement are the primary sources of ammonia toxicity in aquaculture, impairing essential organs and leading to mass mortality in aquatic animals6.
Chromium (Cr) is a heavy metal found in water bodies that poses a significant harm to aquatic life, the environment, and human health. It is classified among 25 most hazardous substances due to its mutagenic, neurotoxic, and carcinogenic properties7,8. Cr is highly bioaccumulative, non-biodegradable, and toxic even at low concentration which can have a severe impact on aquatic life9. The primary source of Cr pollution in aquatic systems is textile, through industrial discharge of petroleum refining, printing, tanneries, fertilizer, mining, photography, pharmaceutical wastes, and electroplating10,11,12,13. The environmental waste management is often ineffective in controlling heavy metal pollution, particularly lithophilic or class B metals, which pose significant threats to ecosystems and aquatic life10. Chromium is especially concerning due to its long-term characteristics of biomagnification, bioaccumulation, and non-biodegradability14. According to Ahmed et al.10 and Velma et al.15, Cr exists in three oxidation states divalent (Cr2+), trivalent (Cr3+), and hexavalent (Cr6+). Among these, hexavalent (Cr6+) is the most hazardous due to its high membrane permeability, which facilitates cellular entry and toxicity. In contrast, trivalent chromium (Cr³⁺) is the predominant form found in biological systems and plays a crucial role in mammals, contributing to protein, lipid, and carbohydrate metabolism16. Trivalent chromium (Cr3+) has very low toxicity, limited membrane permeability, non-corrosiveness, and very little potential to biomagnify in the food chain. Due to its high membrane permeability, Cr⁶⁺ is significantly more toxic than other chromium forms10. In natural surface waters, Cr concentrations typically range from 1 to 10 ppb, while the provisional guideline limit is 50 ppb17. The maximum allowable intake of Cr is 0.05–0.15 mg per kg of body weight, as recommended by WHO18 and FEPA19.
Surprisingly, the adverse effects of pollution can be mitigated through dietary supplements such as curcumin. Curcumin (Diferuloylmethane), a polyphenol derived from Curcuma longa L, possess a wide range of bioactive properties, including immunostimulant, antioxidant, anti-inflammatory, anti-apoptosis, anti-stress, and antibacterial effects20,21. Additionally, curcumin has been shown to reduce metal and ammonia (NH3) toxicity in fish. Its application in aquaculture enhances fish immunity without the adverse effects associated with antibiotics and other medications22,23. Therefore, supplementing fish diets with curcumin can help minimize the risk of microbial infections and improve residue management in aquaculture. Curcumin also activates the transcription factor nuclear factor erythroid 2–related factor, or nrf2, which plays a crucial role in regulating cellular antioxidant defenses24. Stress-responsive genes in fish that react to low-dose Cr and NH3 stress include those associated with apoptosis, cytokines, nuclear factor kappa B (NF-κB), immune response, growth regulation and antioxidant defense. Nrf2 and NF-κB can actively respond to the intake of plant active ingredients, such as plant polysaccharides25,26. All species, including fish, rely on the crucial process known as “programmed” cell death, or apoptosis, which includes immunological and hormone-related gene development, regular cell repair, cellular function, and chemical cell death27. Cytokines, important signaling molecules, play a vital role in both normal and pathological conditions. Under stress, they modulate the host’s inflammatory response and immune system28.
Pangasianodon hypophthalmus is a robust, fast growing fish capable to adapting to a wide range of abiotic and biotic stressors29,30,31. According to Kumar et al.32, this fish species is most suited for studying abiotic-related gene expression. The present study employs gene regulation analysis to elucidate the protective role of dietary curcumin against chromium and ammonia toxicity in P. hypophthalmus.
Materials and methods
Ethics statement
NIASM aquaculture Central Wet Laboratory facilities and experimental protocols were approved under the Committee for the Purpose of Control and Supervision of Experiments on Animals (CCSEA) as 2190/GO/RReBi/SL/2022/CCSEA. This study also strictly adhered to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines. All methods were performed in accordance with the relevant guidelines and regulations.
Experimental animal and design
In this study, P. hypophthalmus (mean weight 47.15 ± 0.40 and length 16.30 ± 0.25 cm) was used. The fish were obtained in good health from NIASM farm pond and acclimatized for two weeks in a fiber-reinforced plastic (FRP) tank before the experiment commenced. Ten fish were kept in each replicate in 150-liter plastic rectangle tank. The experiment followed a completely randomized design (CRD) with four treatment groups, each replicated three times. The treatments were as follows:
-
1.
Control: No exposure to chromium (Cr) or ammonia (NH₃) and fed a control diet.
-
2.
Cr + NH₃ Exposure: Concurrent exposure to ammonia (NH₃) and chromium (Cr) and fed a control diet.
-
3.
Curcumin 0.1% + Cr + NH₃: Concurrent exposure to Cr and NH₃ and supplemented with a 0.1% curcumin diet.
-
4.
Curcumin 0.2% + Cr + NH₃: Concurrent exposure to Cr and NH₃, and supplemented with a 0.2% curcumin diet.
Fish were fed the experimental diets twice a day, at 8:45 AM and 5:15 PM. Continuous aeration was provided using an aerator and faeces and uneaten food were removed daily through siphoning. Water quality parameters were monitored regularly following APHA33 guidelines and remained within the optimal range throughout the experiment. A stock solution of chromium trioxide (CrO3, Hi Media) (100 mg L− 1) was prepared, and ammonia toxicity (1/10th of the LC50; ammonia sulphate, 2.0 mg L− 1)34 was applied along with 1/20th of the LC50 of Cr (2.5 mg L− 1)13. The two third of the (2/3rd ) water were manually replaced every alternate day. During the trial, fish were fed curcumin diets that were iso-nitrogenous (35% crude protein) and iso-caloric (352.89 kcal/100 gram) in pellet form (Table 1). Gross energy was computed using the Halver method35.
Tissue homogenate preparation and blood collection
Under aseptic condition, fish were anaesthesia using clove oil (50 µl L− 1), after that, gills, muscles, brain, liver, and kidneys tissues were collected. To homogenize the tissue for enzyme analysis, a homogenizer(Omni Tissue Master Homogenize, Kennesaw, GA) was used with chilled sucrose (5% w/v, 0.25 M) and EDTA solution (1 mM). For gene expression analysis, muscle and liver tissues were processed using liquid nitrogen. The homogenized tissues were then centrifuged at 5,000 × g for 15 min at 4 °C to obtain homogenates for enzyme analysis. The collected supernatants were stored at -20 °C for further examination. Blood samples were drawn from five fish per tank during dissection and used for serum collection. Tissue protein analysis was conducted using the Lowry protein assay36.
RNA isolation and quantification
Total RNA was isolated from P. hypophthalmus liver tissue using the TRIzol method. The liver tissue was homogenized using liquid nitrogen. The homogenized samples were then mixed chloroform and allowed to phase separate for five minutes. RNA was separated by centrifugation with 75% ethanol, followed by air drying of the RNA pellet. The dried pellet was dissolved in nuclease free water and stored at -80 ˚C for further use. To assess RNA integrity, a 1% agarose gel was used, and RNA bands were visualized using the Gel documentation system (ChemiDocTM MP imaging system, Bio-Rad). Thermo-scientific’s nano-drop spectrophotometer was utilized to quantify RNA.
cDNA synthesis and quantitative PCR
First-strand cDNA synthesis was carried out using a cDNA synthesis kit (Thermo-scientific’s). The reaction mixture contained 12 µL of RNA template (100 ng) and 15 pmol of oligo dT primers. The mixture was heated to 65 °C for five minutes and then immediately cooled on ice. Subsequently, 1 µL of RiboLock RNase Inhibitor (20 U/µL), 2 µL of dNTP Mix (10 mM), and 1 µL of reverse transcriptase enzyme were added. The reaction mixture was briefly centrifuged and incubated at 60 °C for 42 min, followed by 70 °C for 5 min. The synthesized cDNA was stored at -20ºC for further analysis. The quality of synthesized cDNA was confirmed using β-actin as a housekeeping gene. Real-time PCR (qPCR) was performed using SYBR Green and gene-specific primers (Bio-Rad). The qPCR setup included an initial denaturation step at 95 °C for 10 min, followed by 39 cycles of amplification, with denaturation at 95 °C for 15 s and annealing at 60 °C for 1 min37. The details of the primers used are listed in Table 2.
Genes
The genes nitric oxide synthase (iNOS), heat shock protein (HSP 70), caspase 3a, cytochrome P450 (CYP 450), tumor necrosis factor (TNFα), growth hormone receptor (Ghr1), immunoglobulin (Ig), and growth hormone (GH) were examined in liver tissues and myostatin (MYST) in muscle tissue in this study in order to measure the data in real time.
Antioxidant enzyme activities
The Catalase (CAT) (EC 1.11.1.6) assay was carried out using the Takahara et al.38 method. The reaction mixture consisted of 1 mL of freshly prepared hydrogen peroxide substrate solution, 50 µL of tissue homogenate, and 2.45 mL of phosphate buffer (50 mM; pH 7). After through mixing the decrease in absorbance was recorded at 240 nm over three minutes. The Superoxide dismutase (SOD) (EC 1.15.1.1) assay was carried out using Misra and Fridovich’s39 method. Briefly, the reaction mixture contained 0.5 mL of freshly made epinephrine, 1.5 mL of phosphate buffer, and 50 µL of tissue homogenate. After complete mixing, absorbance was measured at 480 nm over three minutes using a UV spectrophotometer. The Glutathione-S-Transferase (GST) (EC 2.5.1.18) assay was performed spectrophotometrically following the protocol of Habig et al.40, using S-2,4-dinitrophenyl glutathione (CDNB) as the substrate. This method is based on the principle that glutathione (GSH), specifically S-2,4-dinitrophenyl glutathione, forms an adduct with CDNB. The formation of this adduct was monitored by measuring the increase in absorbance at 340 nm relative to a blank.
Neurotransmitter enzyme activities
The activity of acetylcholinesterase (AChE) (EC 3.1.1.7) was determined using the method of Hestrin modified from Augustinsson41. Briefly, the assay mixture consisted of 1 mL of phosphate buffer, 1 mL of acetylcholine buffer, and 0.2 mL of the sample. The mixture was then incubated at 37 °C for 30 min. After incubation, alkaline hydroxylamine, hydrochloric acid (HCl), and ferric chloride were added to the solution. The reaction was subsequently measured at 540 nm.
Cortisol
ELISA kit (commercially available Cortisol EIA kit, catalogue no. 500360, Cayman Chemicals, USA) was used to measure serum cortisol levels. The assay was carried out using an ELISA plate reader (Biotek India Pvt. Ltd.) in accordance with the instructions included with the kit.
Aspartate aminotransferase (AST) and Alanine aminotransferase (ALT), lactate dehydrogenase (LDH), and malate dehydrogenase (MDH)
Aspartate aminotransferase (AST) (EC 2.6.1.1) and alanine aminotransferase (ALT) (EC 2.6.1.2) activities were determined using the Wooten42 method. Briefly, the assay involved the preparation of sodium hydroxide (1 N), potassium dihydrogen phosphate, dipotassium hydrogen phosphate, and 2,4-dinitrophenylhydrazine (DNPH). For AST, the substrates used were DL-aspartic acid and β-ketoglutarate, while for ALT, DL-alanine and α-ketoglutarate were used. The tissue homogenate was mixed with the respective substrate and incubated at 37 °C for one hour. Following incubation, DNPH was added, and absorbance was measured at 540 nm. Lactate dehydrogenase (LDH) activity was assessed using the method of Wroblewski and Ladue43. In brief, 0.1 M sodium dihydrogen phosphate and disodium hydrogen phosphate were used to prepare a phosphate buffer. Freshly prepared NADH and sodium pyruvate were then added to this buffer. After introducing the sample/enzyme extract, the mixture was incubated for 20 min, and the absorbance was recorded at 320 nm. Similarly, malate dehydrogenase (MDH) activity was determined following the method of Ochoa44. The reaction mixture was similar to that used for LDH, except that oxaloacetate was used as the substrate instead of sodium pyruvate.
Growth performance
The following procedure was evaluated in order to determine the growth performance. The fish was sampled and weighed every 15 to 40 days on average.
Body weight gain (BWG) = Final weight (g) – Initial weight (g)
Feed conversion ratio (FCR) = Total dry feed intake (g)/Wet weight gain (g)
Specific growth rate (SGR) = 100 (ln FBW-ln IBW)/ number of days.
Weight gain (%) = Final body weight (FBW)-Initial body weight (IBW)/Initial body weight (IBW) ×100
Relative feed intake, (FI) (%/d) = 100 × (TFI/ΙBW)
Protein efficiency ratio (PER) = Total wet weight gain (g)/crude protein intake (g)
Thermal growth coefficient, (TGC) = (FBW1/3 – IBW1/3) × (ΣD0)−1, where ΣD0 is the thermal sum (feeding days × average temperature, ºC)
Daily growth index, DGI (%) = (FBW1/3 – IBW1/3)/days × 100
Chromium (Cr) analysis in fish tissues and experimental water
The Cr bioaccumulation in the different fish tissues such as brain, kidney, gills, liver, and muscles were measured. The tissues were processed according to Kumar et al.13 method utilizing Inductively Coupled Plasma Mass Spectrometry (ICP-MS) (Agilent 7700 series, Agilent Technologies, USA). The samples were digested in a microwave digestion system (Microwave Reaction System, Multiwave PRO, Anton Paar GmbH, Austria, Europe).
Statistics
Statistical Package for the Social Sciences (SPSS) version 16 was used to analyze the data. Shapiro-Wilk and Levene’s test and Shapiro-Wilk’s test, respectively, were used to assess the data for normality and homogeneity of variance. The current study used a one-way ANOVA (analysis of variance) with Duncan’s multiple range tests. After analysis, the data was significant at p < 0.05.
Results
Cortisol, heat shock protein (HSP 70) and inducible nitric oxide synthase (iNOS)
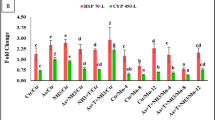
In the present study, cortisol levels were significantly elevated under concurrent exposure to low dose of Cr (1/20th of LC50 of Cr) and NH3 stress compared to the control and curcumin-supplemented diets. However, cortisol levels significantly decreased (p = 0.0051) with dietary curcumin at 0.2%, followed by 0.1% with exposure to stressor (Fig. 1A). Similarly, concurrent exposure to Cr and NH3 resulted in a significant upregulation of HSP 70 (p = 0.001) and iNOS (p = 0.0021) expression compared to the control and curcumin supplemented groups. Conversely, dietary curcumin supplementation at 0.1% and 0.2% significantly downregulated HSP70 and iNOS expression compared to the control and other groups (Fig. 1A,B).
(A, B) Effect of dietary curcumin on cortisol (p = 0.0051), and gene expression of heat shock protein (HSP, P = 0.001) and inducible nitric oxide synthase (iNOS, P = 0.0021) of P. hypophthalmus reread under chromium and ammonia toxicity for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 3).
Anti-oxidative enzymes
The liver and kidney tissues of P. hypophthalmus were to assess anti-oxidative enzymes, including as catalase (CAT), superoxide dismutase (SOD), and glutathione-s-transferase (GST) (Fig. 2A–C). Upon exposure to a low dose of concurrent exposure to Cr and NH3, CAT activity significantly increased in the liver (p = 0.0016) and kidney (p = 0.0013) tissues. However, CAT activity was considerably reduced in the group provided dietary curcumin at 0.1% and exposed to Cr + NH3 compared to the control and the group fed curcumin at 0.2%. In contrast, the group fed curcumin at 0.2% and subjected to Cr + NH3 exhibited significantly higher CAT gene expression than the control and the 0.1% curcumin group (Fig. 2A). SOD activities in the kidney (p = 0.022) and liver (p = 0.0032) was significantly upregulated upon concurrent exposure to low dose of Cr + NH3 compared to control and curcumin supplemented groups. Furthermore, dietary curcumin at 0.1% significantly reduced (p < 0.05) SOD activity in liver and kidney compared to the control and 0.2% curcumin fed group (Fig. 2B). Similarly, GST activity was significantly elevated in the liver (p = 0.0018) and kidney (p = 0.0025) under concurrent Cr and NH3 stress compared to 0.1% curcumin group. However, dietary, curcumin at 0.1% significantly reduced GST activity in liver and kidney tissue compared to control and 0.2% curcumin fed group at 0.2%. However, dietary curcumin at 0.1% significantly reduced GST activity in both tissues compared to the control and the 0.2% curcumin-fed group. Additionally, GST activity in the kidney was significantly lower in the 0.2% curcumin group compared to the control, while GST activity in the liver was also markedly reduced in the 0.2% curcumin group (Fig. 2C).
(A–C) Effect of dietary curcumin on CAT (Liver, p = 0.0016; Kidney, p = 0.0013), SOD (Liver, p = 0.0032; Kidney, p = 0.022) and GST (Liver, p = 0.0018, kidney, p = 0.0025) in liver and kidney tissues of P. hypophthalmus reread under chromium and ammonia toxicity for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 3).
Neurotransmitter enzymes
Figure 3A shows the neurotransmitter enzyme in the brain tissue of P. hypophthalmus reared under control or stressors group (Cr + NH3). Compared to dietary curcumin fed groups, the acetylcholinesterase activity (AChE) in the brain was significantly decreased (p = 0.0046) in the group fed a control diet and concurrently exposed to Cr and NH3. Furthermore, as compared to the control group that also had concomitant exposure to Cr and NH3 stress, the AChE activity was noticeably enhanced with dietary curcumin at 0.1% followed by 0.2% fed group.
(A–C) Effect of dietary curcumin on acetylcholinesterase (AChE, p = 0.0046) in brain, lactate dehydrogenase (LDH, liver, p = 0.017, gill p = 0.019) and malate dehydrogenase (MDH, liver, p = 0.002, gill, p = 0.001) in liver and gill tissues of P. hypophthalmus reread under chromium and ammonia toxicity for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
Cellular metabolic enzymes
The activities of lactate dehydrogenase (LDH) and malate dehydrogenase (MDH) in liver and gill tissues of P. hypophthalmus are presented in Fig. 3B,C. Upon concurrent exposure to Cr + NH3, LDH activity was significantly higher in the liver (p = 0.017) and gill (p = 0.019) compared to the control and curcumin-supplemented fed groups. However, in the group supplemented with dietary curcumin at 0.1% and exposed to Cr + NH3, LDH activity in the liver and gill was significantly lower than the control and 0.2% curcumin fed group. Additionally, a substantial difference in LDH activity was observed in the liver and gill tissues (Fig. 3B), with liver LDH level being significantly higher in the 0.2% curcumin group compared to the control. Moreover, compared to the control, stressors group (Cr + NH3) and 0.2% curcumin fed group, MDH activity was significantly lower in the liver (p = 0.002) and gill (p = 0.001) tissue of P. hypophthalmus in the 0.1% curcumin fed group exposed to Cr + NH3. Conversely, the 0.2% curcumin group exhibited significantly higher MDH activity in the gill tissue compared to all other groups (Fig. 3C). Similarly, ALT (liver, p = 0.0026; gill, p = 0.0018) and AST (liver, p = 0.0031; gill, p = 0.0011) activities in both liver and gill tissues were significantly higher in the control diet group subjected to Cr + NH3 exposure compared to the control and curcumin-supplemented diet groups. However, supplementation with curcumin at 0.1% significantly reduced ALT activity in both tissues compared to the control, stressor-exposed (Cr + NH3), and 0.2% curcumin-fed groups. While curcumin at 0.1% effectively reduced ALT activity in both tissues, curcumin at 0.2% was less efficient in modulating ALT levels (Fig. 4A). Furthermore, supplementation with curcumin at 0.1% and 0.2% significantly reduced AST activity in the liver (p = 0.016) and gill (p = 0.002) compared to the control diet group concurrently exposed to Cr + NH3 (Fig. 4B).
(A–C) Effect of dietary curcumin on alanine aminotransferase (ALT, liver, p = 0.0026, gill, p = 0.0018), aspartate aminotransferase (AST, liver, p = 0.0031, gill, p = 0.0011) in liver and gill and protease (p = 0.0028) and amylase (p = 0.016) activity of P. hypophthalmus reread under chromium and ammonia toxicity for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 6).
Digestive enzymes
The intestinal tissue of P. hypophthalmus was analysed under controlled conditions and concurrent exposure to Cr and NH3, to assess protease and amylase activities (Fig. 4C). Protease activity was significantly increased (p = 0.0028) in the groups supplemented with curcumin at 0.1% and 0.2% compared to the control group and the group exposed to Cr + NH3 while receiving a control diet. Conversely, protease activity was significantly lower in the control diet group exposed to Cr + NH3 compared to the control and curcumin-supplemented groups. Similarly, amylase activity was significantly enhanced (p = 0.016) in the group supplemented with curcumin at 0.1% and exposed to Cr + NH3 compared to the control, 0.2% curcumin, and Cr + NH3-exposed groups. Amylase activity was comparable between the control and 0.2% curcumin-fed groups, while the lowest amylase activity was observed in the control diet group concurrently exposed to Cr and NH3.
Apoptotic and detoxifying gene
Compared to the control and curcumin-supplemented groups, the gene expression of cytochrome P450 (CYP450) in liver tissue was significantly upregulated (p = 0.0018) in the control diet group concurrently exposed to Cr and NH3. Notably, CYP450 gene expression was significantly downregulated in the group supplemented with 0.1% curcumin relative to the stressor-exposed (Cr + NH3) group and in the stressor-exposed group fed a control diet (Fig. 5A). Similarly, caspase (Cas 3a) expression was significantly downregulated (p = 0.0013) in the 0.1% curcumin-supplemented group exposed to Cr + NH3 compared to the control, stressor-exposed, and 0.2% curcumin-fed groups. Among all groups, the control diet group concurrently exposed to NH3 exhibited the highest Cas 3a expression in liver tissues (Fig. 5A).
(A, B) Effect of dietary curcumin on gene expression of CYP P450 (p = 0.0018), Cas 3a (0.013), Ig (p = 0.011), and TNFα (p = 0.0027) in liver tissue of P. hypophthalmus reread under chromium and ammonia toxicity for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 3).
Total immunoglobulin and tumor necrosis factors genes
Figure 5B presents the gene expression levels of total immunoglobulin (Ig) and tumor necrosis factor-alpha (TNFα) in the liver tissue of P. hypophthalmus exposed to low doses of Cr and NH3. Compared to the control and stressor-exposed (Cr + NH3) groups, Ig gene expression was significantly upregulated (p = 0.0011) in the group supplemented with 0.1% curcumin, followed by the 0.2% curcumin group exposed to stressors. Conversely, Ig expression was markedly downregulated in the group fed a control diet and concurrently exposed to Cr + NH3. Similarly, TNFα expression exhibited an inverse correlation with Ig expression. In the presence of stressors, dietary supplementation with 0.1% curcumin resulted in a significant downregulation of TNFα (p = 0.0011) compared to the control diet group concurrently exposed to Cr and NH3.
Growth performance
The growth parameters of P. hypophthalmus reared under control conditions, with or without exposure to Cr and NH3, were assessed, including weight gain percentage, feed conversion efficiency (FCE), specific growth rate (SGR), protein efficiency ratio (PER), daily growth index (DGI), and relative feed intake (RFI). The results are presented in Table 3. Compared to the control group and the group fed a control diet while concurrently exposed to Cr and NH3, the weight gain percentage (p = 0.0042), FCE (p = 0.0031), SGR (p = 0.0027), PER (p = 0.0023), DGI (p = 0.016), and RFI (p = 0.026) were significantly higher in the group supplemented with 0.1% curcumin, followed by the 0.2% curcumin group. Conversely, the group exposed to Cr and NH3 while fed a control diet exhibited a significant reduction in all growth-related parameters, including weight gain percentage, FCE, SGR, PER, DGI, and RFI, compared to the other groups. Additionally, Fig. 6A,B present the expression of growth-related genes (GH, GHR1, and MYST) in P. hypophthalmus reared under control conditions or exposed to Cr and NH3 toxicity and fed curcumin-supplemented diets. Compared to the control and stressor-exposed (Cr + NH3) groups, the genes regulating growth hormone (GH) (p = 0.0034) and growth hormone receptor 1 (GHR1) (p = 0.0018) were significantly upregulated in the groups fed 0.1% curcumin and subjected to Cr + NH3. Additionally, GH and GHR1 were significantly downregulated in the group fed a control diet and exposed to Cr and NH3 compared to the control and curcumin-supplemented groups (Fig. 6A). Furthermore, dietary supplementation with 0.1% curcumin significantly downregulated MYST expression (p = 0.0037) compared to the other groups. In contrast, MYST expression was significantly elevated in the group exposed to Cr and NH3 and fed a control diet compared to the control and curcumin-supplemented groups (Fig. 6B).
(A, B) Effect of dietary curcumin on gene expression of GH (p = 0.0034), GHR1 (p = 0.0018) and MYST (p = 0.0037) in liver tissue of P. hypophthalmus reread under chromium and ammonia toxicity for 40 days. Within endpoints and groups, bars with different superscripts differ significantly (a–d). Data expressed as Mean ± SE (n = 3).
Chromium bioaccumulation
The chromium (Cr) concentration in water samples and its bioaccumulation in various tissues, including muscle, liver, kidney, gill, and brain, are presented in Table 4. The Cr concentrations in water varied across different groups, with levels of 0.01 µg L− 1 in the control group, 2352 µg L− 1 in the Cr+NH3-exposed group, and 492 µg L− 1 and 580 µg L− 1 in the groups supplemented with 0.1% and 0.2% curcumin, respectively. Furthermore, the highest Cr bioaccumulation was observed in the liver (3.40 mg kg− 1), followed by the kidney (3.17 mg kg− 1) and gill (2.47 mg kg− 1). The results also indicate that dietary supplementation with 0.1% curcumin led to the lowest Cr bioaccumulation across all tissues.
Discussion
In the present investigation, concurrent exposure to chromium (Cr) and ammonia stress led to an elevated cortisol level in P. hypophthalmus. The Cr has strong association with cortisol secretion in stress condition, activating metabolic process in response to Cr toxicity45. It has also been demonstrated that the elevated cortisol levels may be physiological response to restore homeostasis in fish, protecting them against Cr toxicity46. Surprisingly, concurrent exposure to Cr and NH3 disturbs the central nervous system, affecting glycogenolysis and gluconeogenesis13,47. Interestingly, dietary curcumin at 0.1% effectively reduced cortisol levels, likely due to polyphenolic nature of curcumin. Polyphenol in curcumin exhibit antioxidant properties, neutralizing lipid radicals in the cell membrane and converting them into phenoxyl radicals, ultimately reducing cortisol levels48,49,50. Abdel-Ghany et al.51 reported that nano-curcumin supplementation at 50 and 100 mg kg− 1 significantly reduces cortisol levels in Tilapia reared under heat stress. Furthermore, the cortisol levels observed in this study were consistent with HSP 70 gene expression in P. hypophthalmus liver tissue. The current investigation found that concurrent exposure to low doses of Cr and NH3 upregulated HSP 70 gene expression likely due to Cr and NH3 altering catecholamine levels, leading to excessive HSP 70 expressions13. Interestingly, dietary curcumin at 0.1% protects the chaperon’s family HSP 70 by downregulating its production, thereby preventing protein degradation23. Curcumin also facilitates HSP 70 binding to denatured proteins, preventing their further misfolding and promoting their recovery to maintain tissue function52 (Heredia-Middleton et al., 2008).
Surprisingly, Cr and NH3 stress significantly upregulated iNOS expression, whereas dietary curcumin supplementation downregulated it. Cr and NH3 exposure stimulate nitric oxide (NO) production, leading to oxidative damage by nitrating biomolecules such as DNA53. Additionally, ammonia accumulation in various tissues contributes to NO production, with ammonia being a key factor influencing iNOS gene expression in muscle tissue54. Interestingly, dietary curcumin may play a crucial role in suppressing constitutive NF-kappaB (NF-κB) and IKK activity55. Furthermore, curcumin inhibits iNOS expression by reducing NF-κB translocation56. The findings suggest that curcumin mitigates the harmful effects of Cr and NH3 by modulating NF-κB signaling pathways57.
The activities of CAT, SOD and GST in liver and kidney were significantly elevated under Cr and NH3 toxicity. However, dietary curcumin effectively substantiated the CAT, SOD and GST in liver and kidney tissues. Concurrent exposure to Cr and NH3 accelerates free radical production in these tissues. Cr is generally required as a catalyst for reversible oxidation using hydrogen oxide (HO)58. Cr (VI) is particularly responsible for production of reactive oxygen species (ROS) through a Fenton-like redox cycling mechanism58. It also interferes with mitochondria regulation59. ROS attack biomolecules such as lipids, protein, nucleic acids, disrupting cellular function and integrity60. Interestingly, 0.1% dietary curcumin supplementation significantly enhanced the anti-oxidative status of P. hypophthalmus by modulating CAT, SOD, and GST activities in liver and kidney tissues. This effect is most likely attributed to curcumin’s antioxidant properties as it functions both as a direct radical scavenger and an inducer of anti-oxidant responses61. Additionally, curcumin contributes to reducing oxidative damage by enhancing various anti-oxidant defences, thereby protecting fish from Cr and NH3 stress62,63,64.
Acetylcholinesterase (AChE) activity in brain tissue of P. hypophthalmus was significantly inhibited under Cr and NH3 stress. AChE inhibition is a key indicator of nervous systems damage, leading to behavioural, feeding, and respiration alteration in the organism60,65. The combined exposure also blocks the enzyme’s active site, alter its structure and affects the amino acid sequence13,66. Surprisingly, dietary curcumin protects neurotransmitter from damage and blockage by enhancing AChE activity in brain tissue. Moreover, curcumin has neuroprotective nature due to their role in cross blood-brain barrier67. Many investigations have shown that curcumin lowers AChE activity following exposure to hazardous chemicals67,68.
The supplementation of curcumin diet improved AChE activity. The current investigation found that exposure to Cr and NH3 significantly increased LDH and MDH activities in liver and gill tissues. However, dietary curcumin at 0.1% significantly reduced LDH and MDH activity. It has been shown that under condition of oxygen and/or glucose deprivation, cells containing LDH are damaged or killed, leading to increased cell membrane permeability or rupture causing enzyme leakage69,70,71,72.
ALT and AST activities in liver and gill tissues were significantly elevated under concurrent exposure to Cr and NH3. However, dietary curcumin at 0.1% noticeably reduces the ALT and AST activities. This is likely due to ability of curcumin to protect the liver and gill tissue against oxidative stress caused by hydrogen peroxide72. Curcumin also acts as anti-inflammatory agent. Its ability to reduce ALT and AST activities suggests a hepatoprotective effect against toxic materials73. Additionally, curcumin may play a crucial role in stabilizing tissues cell membrane, preventing the leakage of the intracellular enzymes74. The present result of ALT and AST activities suggest that curcumin protects liver and gill tissues from the stress (Cr + NH3) while maintaining cell membranes integrity and function75.
The present study also revealed that concurrent exposure to Cr and NH3 reduced the activity of digestive enzymes such as protease and amylase. However, curcumin supplementation improved digestive enzymes activity by enhancing intestinal enzymes function and increasing the number of mature intestinal cells76. Digestive enzyme activity directly influences nutrient absorption in fish77. A study on Oreochromis niloticus demonstrated that diets supplemented with 0.5 and 1.0% curcumin significantly improved the activities of digestive enzymes, including protease, amylase and lipase78. Furthermore, digestive enzymes activity is closely associated with fish growth performance. Interestingly, the present study indicates that curcumin has the potential to enhance digestive enzymes activity, intestinal absorption efficiency and overall nutrient utilization79.
In this study, concurrent exposure to Cr and NH3 upregulated CYP450 gene expression. In contrast, dietary curcumin supplementation reduced CYP450 activity in liver tissue. CYP450 plays a key role in metabolic pathway involving lipoxygenase, arachidonic acid, and cyclooxygenase. Moreover, it contributes to the formation of carcinogenic electrophilic intermediates from naturally occurring compounds like curcumin and is involved in detoxification and xenobiotic metabolism. However, our findings indicate that Cr and NH₃ exposure significantly upregulated CYP450 gene expression. Previous studies have also reported that exposure to arsenic, ammonia, and high temperature, upregulates CYP450 expression80,81,82. Notably, Giri et al.83 found that curcumin downregulates CYP450 gene expression in fish, suggesting its protective role against oxidative stress.
Cas 3a plays a crucial role in apoptosis, which is closely linked to oxidative stress and inflammatory responses84. In this study, Cr and NH3 stress induced apoptosis via p53, a key regulator of apoptosis85. The findings were correlated with CYP450, which is implicated in stress-induced apoptosis and up-regulates the transcription of bcl2-associated X protein (Bax)86,87. Surprisingly, Bax is a major pro-apoptotic gene in fish, triggering the release of cytochrome C and caspase activation88. Furthermore, dietary curcumin inhibited the Cas 3a gene expression in liver tissue by activating caspase-3 and cleaving poly (ADP-ribose) polymerase 1 (PARP)89.
Curcumin is effective anti-inflammatory agent, protecting tissues from damage caused by toxin substances. In this study, Cr and NH3 exposure downregulated TNFα gene expression in liver tissue indicating damage. NFκB regulates genes involved in the production of proinflammatory cytokines, including TNF-α, thereby contributing to inflammation90. Curcumin is known to reduce inflammation and protect tissues from injury57.
Interestingly, Cr and NH3 stress significantly impaired fish growth performance However, dietary curcumin at 0.1 and 0.2%, dramatically improved growth performance and mitigated the toxic effect of Cr and NH3 toxicity. Curcumin’s growth-promoting effects may be attributed to its role in enhancing digestive enzyme activity91, modulating immunity92, and exerting anti-oxidative and anti-stress properties93,94. The growth promoting benefits of curcumin are primarily due to its ability to enhance digestive enzymes, particularly those involved in protein metabolism. Additionally, curcumin contributes to overall health by acting as a selective growth factor and substrate for beneficial gut microbiota while inhibiting harmful intestinal bacteria. Previous studies have also reported curcumin as a growth enhancer in fish93,94.
The present findings are further supported by the expression of MYST, GH, and GHR1 genes. While NH₃ exposure upregulated MYST gene expression in liver tissue, curcumin supplementation inhibited its expression. Furthermore, curcumin upregulated GH and GHR1 gene expression, whereas NH₃ stress downregulated them. Curcumin’s ability to enhance growth-related gene expression and improve growth performance may be attributed to its role in RNA, DNA, and protein synthesis, particularly the regulation of GH gene production. Additionally, curcumin modulates the insulin-like growth factor signaling pathway to promote growth95. Interestingly, curcumin has been shown to enhance Cr detoxification in muscle, liver, kidney, gill, and brain tissues. The findings related to CYP450 and caspase-3 corroborate these results. Overall, curcumin exhibits significant potential as a detoxifying agent, effectively reducing Cr bioaccumulation in various organs.
Conclusion.
The current study revealed that incorporating a 0.1% curcumin diet significantly enhanced the antioxidative capacity, growth performance, and digestive efficiency of P. hypophthalmus reared under chromium and ammonia toxicity (Cr + NH3). Moreover, curcumin supplementation at 0.1% positively influenced the regulation of key genes, including HSP 70, iNOS, TNFα, and Ig, particularly in fish exposed to when fish were subjected to simultaneous exposure to Cr and ammonia toxicity. Significant improvements were also observed in the expression of detoxification-associated genes, such as CYP450 and caspase, when dietary curcumin at 0.1% was introduced, strengthening the fish’s resilience against the combined effects of Cr and ammonia toxicity. Additionally, growth-related genes such as GH and GHR1 exhibited upregulation, while MYST showed downregulation with 0.1% curcumin supplementation, contributing to an overall enhancement in the fish’s growth performance. Furthermore, the supplementation of curcumin at 0.1% in the diet led to a substantial reduction in Cr accumulation. Notably, this study is the first to highlight the role of curcumin in mitigating the adverse effects of simultaneous Cr and NH₃ stress in fish.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Mishra, R. K., Mentha, S. S., Misra, Y. & Dwivedi, N. Emerging pollutants of severe environmental concern in water and wastewater: A comprehensive review on current developments and future research. Water Energy Nexus 6, 74–95 (2023).
Randall, D. J. & Tsui, T. K. N. Ammonia toxicity in fish. Mar. Pollut. Bull. 45 (1–12), 17–23 (2002).
Sun, S-X. et al. High dissolved oxygen exacerbates ammonia toxicity with sex-dependent manner in zebrafish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 266, 109549 (2023).
Benli, A. C. K., Oksal, G. K. & Ozkul, A. Sublethal ammonia exposure of nile tilapia (Oreochromis niloticus L.): effects on Gill, liver and kidney histology. Chemosphere 72, 1355–1358 (2008).
Hoseini, S. M., Yousei, M., Hoseinifar, S. H. & Van Doan, H. Antioxidant, enzymatic and haematological responses of common carp (Cyprinus carpio) fed with myrcene- or menthol- supplemented diets and exposed to ambient ammonia. Aquac 506, 246–255 (2019).
Kim, J. H., Kang, Y. J., Kim, K. I., Kim, S. K. & Kim, J. H. Toxic effects of nitrogenous compounds (ammonia, nitrite, and nitrate) on acute toxicity and antioxidant responses of juvenile olive lounder, Paralichthys olivaceus. Environ. Toxicol. Pharmacol. 67, 73–78 (2019).
Li, M-Y., Shi, Y-C., Xu, W-X., Zhao, L. & Zhang, A-Z. Exploring Cr(VI)-induced blood-brain barrier injury and neurotoxicity in zebrafish and snakehead fish, and inhibiting toxic effects of astaxanthin. Environ. Pollut. 355, 124280 (2024).
Zhao, L. et al. Long-term exposure of zebrafish (Danio rerio) to Cr(VI): reproductive toxicity and neurotoxicity. Reg. Stud. Mar. Sci. 74, 103559 (2024).
Yu, Z. et al. Toxic effects of hexavalent chromium (Cr6+) on bioaccumulation, apoptosis, oxidative damage and inflammatory response in channa asiatica. Environ. Toxicol. Pharmacol. 87, 103725 (2021).
Ahmed, M. K. et al. Chromium (VI) induced acute toxicity and genotoxicity in freshwater stinging catfish, heterpneustes fossilis. Ecotoxicol. Environ. Saf. 1–7 (2013).
Shi, Y-C. et al. Hexavalent chromium-induced toxic effects on the hematology, redox State, and apoptosis in cyprinus carpio. Reg. Stud. Mar. Sci. 56, 102676 (2022).
Zhao, L. et al. Polysaccharide from dandelion enriched nutritional composition, antioxidant capacity, and inhibited bioaccumulation and inflammation in Channa Asiatica under hexavalent chromium exposure. Int. J. Biol. Macromol. 15, 201 557–568 (2022).
Kumar, N., Bhushan, S., Patole, P. B. & Gite, A. Multi-biomarker approach to assess chromium, pH and temperature toxicity in fish. Comp. Biochem. Physiol. Part (C) Toxicol. Pharmacol. 254 109264 (2022).
Lodhi, H. S., Khan, M. A., Verma, R. S. & Sharma, U. D. Acute toxicity of copper sulphate to freshwater prawns. J. Environ. Biol. 27, 585–588 (2006).
Velma, V., Vutukuru, S. S. & Tchounwou, P. B. Ecotoxicology of hexavalent chromium in freshwater fish: a critical review. Rev. Environ. Health 24 (2), 129–145 (2009).
Steven, J. D. et al. Effects of chromium in the Canadian environment. Nat. Res. Coun Can. 168 NRCC N. (1976).
WHO World Health Organisation; Geneva, Switzerland. Guidelines for Drinking Water Quality. (2003).
WHO (World Health Organization). Recommendation WHO; Geneva. (Guidelines for Drinking Water Quality). 1 130. (1985).
FEPA (Federal Environmental Protection Agency). Guidelines and Standards for Environmental Pollution Control in Nigeria. 238. (2003).
Elabd, H. et al. Nano Curcumin/chitosan modulates growth, biochemical, immune and anti-oxidative profiles with related-gene expression in nile tilapia. Fishes 8 (7), 333 (2023).
Alagawany, M. et al. Curcumin and its different forms: A review on fish nutrition. Aquaculture 532, 736030 (2021).
Li, M. et al. Effects of dietary Curcumin on growth performance, lipopolysaccharide-induced immune responses, oxidative stress and cell apoptosis in Snakehead fish (Channa argus). Aquac. Rep. 22, 100981 (2022).
Xavier, M. J. et al. Effects of dietary curcumin in growth performance, oxidative status and gut morphometry and function of gilthead seabream postlarvae. Aquac. Rep. 24, 101128 (2022).
Giri, S. S., Sukumaran, V. & Park, S. C. Effects of bioactive substance from turmeric on growth, skin mucosal immunity and antioxidant factors in common carp, cyprinus carpio. Fish. Shellfish Immun. 92, 612–620 (2019).
Yu, Z. et al. Dietary taraxacum Mongolicum polysaccharide ameliorates the growth, immune response, and antioxidant status in association with NF-κB, Nrf2 and TOR in Jian carp (Cyprinus Carpio var. Jian). Aquaculture 547, 737522 (2022).
Niu, X-T. et al. The major role of glucocorticoid receptor (GR) in astaxanthin alleviates immune stress in channa argus lymphocyte. Aquaculture 584, 740637 (2024).
Elmore, S. & Apoptosis A review of programmed cell death. Toxicol. Pathol. 35 (4), 495–516 (2007).
Ahmadian, E. et al. The role of cytokines in nephrotic syndrome. Mediat. Inflamm. 6499668 (2022).
Kumar, N., Krishnani, K. K., Gupta, S. K. & Singh, N. P. Selenium nanoparticles enhanced thermal tolerance and maintain cellular stress protection of pangasius hypophthalmus reared under lead and high temperature. Respir. Physiol. Neurobiol. 246, 107–116 (2017a).
Kumar, N. et al. Immuno-protective role of biologically synthesized dietary selenium nanoparticles against multiple stressors in pangasianodon hypophthalmus. Fish. Shellfish Immunol. 78, 289–298 (2018).
Kumar, N., Singh, D. K., Bhushan, S. & Jamwal, A. Mitigating multiple stresses in pangasianodon hypophthalmus with a novel dietary mixture of selenium nanoparticles and Omega-3-fatty acid. Sci. Rep. 11 (1), 1–20 (2021).
Kumar, N., Krishnani, K. K. & Singh, N. P. Effect of zinc on growth performance and cellular metabolic stress of fish exposed to multiple stresses. Fish. Physiol. Biochem. 46, 315–329 (2020).
APHA-AWWA-WEF. Standard Methods for the Estimation of Water and Waste Water, Twentieth Ed. American Public Health Association. (eds Clesceri, L. S., Greenberg, A. E. & Eaton, A. D.) (American Water Works Association, Water Environment Federation, 1998).
Kumar, N. et al. Exploring mitigating role of zinc nanoparticles on arsenic, ammonia and temperature stress using molecular signature in fish. J. Trace Elem. Med. Biol. 74, 127076 (2022).
Halver, J. E. The nutritional requirements of cultivated warm water and cold water fish species in report of the FAO technical conference on aquaculture, Kyoto, Japan. FAO Fisheries Report No. 188 FI/ R188 (En), 9 (1976).
Lowry, O. H., Ronebrough, N. J., Farr, A. L. & Randall, R. J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT-PCR. Nucl. Acids Res. 29 (9), e45 (2001).
Takahara, S. et al. Hypocatalesemia, a new generis carrier state. J. Clin. Investig. 29, 610–619 (1960).
Misra, H. P. & Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 247, 3170–3175 (1972).
Habing, W. H., Pabst, M. N., Bjacoby, W. & Glutathion, S. Transferase, the first enzymatic step in mercatpopunc acid formation. J. Biol. Chem. 249, 7130–7138 (1974).
Hestrin, S. The reaction of acetyl choline esters and other carboxylic acid derivatives with hydroxyline and its analytical application. J. Bio Chem. 180, 249–261 (1949).
Wootton, I. D. P. Microanalysis in Medical Biochemistry 101–103 (J & A Churchill Ltd., 1964).
Wroblewski, L. & LaDue, J. S. Lactic dehydrogenase activity in blood. Proc. Soc. Exp. Biol. Med. 90 210–213 (1955).
Ochoa, S. Malic dehydrogenase and ‘malic’ enzyme. In (eds Coloric, S. P. & Kaplan, N.) Methods of Enzymology. I. 735–745. (Academic, New York, 1955).
Kim, J. H. & Kang, J. C. The immune responses and expression of metallothionein (MT) gene and heat shock protein 70 (HSP 70) in juvenile rockfish, Sebastes schlegelii, exposed to waterborne arsenic (As3+) environ. Toxicol. Pharmacol. 47, 136–141 (2016).
Mishra, A. K. & Mohanty, B. Effect of hexavalent chromium exposure on the pituitary–interrenal axis of a teleost, Channa punctatus (Bloch). Chemosphere 76, 982–988 (2009).
Kim, J. & Kang, J. The toxic effects on the stress and immune responses in juvenile rockfish, Sebastes schlegelii exposed to hexavalent chromium. Environ. Toxicol. Pharmacol. 43, 128–133 (2016).
Ak, T. Antioxidant and radical scavenging properties of curcumin. Chem. Biol. Interact. 174, 27–37 (2008).
Yuan, J., Liu, R., Ma, Y., Zhang, Z. & Xie, Z. Curcumin attenuates airway inflammation and airway remolding by inhibiting NF-kB signaling and COX-2 in cigarette smoke-induced COPD mice. Inflammation 41, 1804–1814 (2018).
Husni, A., Lailatussifa, R. & Isnansetyo, A. Sargassum hystrix as a source of functional food to improve blood biochemistry profiles of rats under stress. Prev. Nutr. Food Sci. 24, 150–158 (2019).
Abdel-Ghany, H. M., El-Sisy, D. M. & Salem, M. E. S. A comparative study of effects of Curcumin and its nanoparticles on the growth, immunity and heat stress resistance of nile tilapia (Oreochromis niloticus). Sci. Rep. 13, 2523 (2023).
Heredia-Middleton, P., Brunelli, J., Drew, R. E. & Thorgaard, G. H. Heat shock protein (HSP70) RNA expression differs among rainbow trout (Oncorhynchus mykiss) clonal lines. Comp. Biochem. Physiol. B 149(4) (2008).
Pinlaor, S. et al. Nitroguanine formation in the liver of hamsters infected with opisthorchis viverrini. Biochem. Biophys. Res. Commun. 309 (3), 8 (2003).
Kumari, S., Choudhury, M. G. & Saha, N. Hyper-ammonia stress causes induction of inducible nitric oxide synthase gene and more production of nitric oxide in air-breathing Magur Catish, Clarias Magur (Hamilton). Fish. Physiol. Biochem. 45 (3), 907–920 (2019).
Aggarwal, B. B. et al. Curcumin suppresses the paclitaxel-induced nuclear factor-kappaB pathway in breast cancer cells and inhibits lung metastasis of human breast cancer in nude mice. Clin. Cancer Res. 11 (20), 7490–7498 (2005).
Karin, M. & Greten, F. R. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat. Rev. Immunol. 5, 749 – 59. (2005).
Nakatake, R. et al. Curcumin protects liver inflammation by suppressing expression of inducible nitric oxide synthase in primary cultured rat hepatocytes. Funct. Foods Health Dis. 7 (9), 716–734 (2017).
Lushchak, V. I. Contaminant-induced oxidative stress in fish: a mechanistic approach. Fish. Physiol. Biochem. 42 (2), 711–747 (2016).
Pourahmad, M., Brien, P. J. O., Jokar, F. & Daraei, B. Carcinogenic metal induced sites of reactive oxygen species formation in hepatocytes. Toxicol. Vitro 17 (5–6), 803–810 (2003).
Mansour, A. T. et al. Ameliorative effect of Quercetin against abamectin induced hemato-biochemical alterations and hepatorenal oxidative damage in nile tilapia, Oreochromis niloticus. Animals 12 (23), 3429 1–16 (2022).
Kidd, P. M. Bioavailability and activity of phytosome complexes from botanical polyphenols: the silymarin, curcumin, green tea, and grape seed extracts. Altern. Med. Rev. 14 (3), 226–246 (2009).
Awasthi, Y. et al. A protective study of Curcumin associated with Cr6 + induced oxidative stress, genetic damage, transcription of genes related to apoptosis and histopathology of fish, Channa punctatus (Bloch, 1793). Environ. Toxicol. Pharmacol. 71, 103209 (2019).
Hoseini, S. M. et al. Mitigation of transportation stress in common carp, Cyprinus Carpio, by dietary administration of turmeric. Aquac 546, 737380 (2022).
Ming, J. H. et al. Optimal dietary Curcumin improved growth performance, and modulated innate immunity, antioxidant capacity and related genes expression of NF-κB and Nrf2 signaling pathways in grass carp (Ctenopharyngodon idella) after infection with Aeromonas hydrophila. Fish. Shellfish Immunol. 97, 540–553 (2020).
Cunha, I., Mangas-Ramirez, E. & Guilhermino, L. Effects of copper and cadmium on cholinesterase and glutathione S-transferase activities of two marine gastropods (Monodonta lineata and Nucella lapillus). Comp. Biochem. Physiol. C 145, 648–657 (2007).
Sabullah, M. K. et al. The assessment of cholinesterase from the liver of puntius javanicus as detection of metal ions. Sci. World J. 571094 (2014).
Jaques, J. A. S. et al. Curcumin protects against cigarette smoke-induced cognitive impairment and increased acetylcholinesterase activity in rats. Physiol. Behav. 106, 664–669 (2012).
Kuhad, A., Pilkhwal, S., Sharma, S., Tirkey, N. & Chopra, K. Effect of Curcumin on inflammation and oxidative stress in cisplatin induced experimental nephrotoxicity. J. Agric. Food Chem. 55, 10150–10155 (2007).
Kumar, N., Gupta, S. K., Bhushan, S. & Singh, N. P. Impacts of acute toxicity of arsenic (III) alone and with high temperature on stress biomarkers, immunological status and cellular metabolism in fish. Aquat. Toxicol. 214, 105233 (2019).
Kumar, N., Thorat, S. T. & Chavhan, S. R. Multifunctional role of dietary copper to regulate stress-responsive gene for mitigation of multiple stresses in pangasianodon hypophthalmus. Sci. Rep. 14 (1), 2252 (2024).
Prince, P. S. M., Suman, S., Devika, P. T. & Kithianathan, M. Cardioprotective effect of ‘marutham’ a polyherbal formulation on isoproterenol induced myocardial infarction in Winstar rats. Fitoterapia 79, 433–438 (2008).
Al-Rubaei, Z. M., Mohammad, T. U. & Ali, L. K. Effects of local curcumin on oxidative stress and total antioxidant capacity in vivo study. Pak. J. Biol. Sci. 17, 1237–1241 (2014).
Kong, Y. D. et al. The positive effects of single or conjoint administration of lactic acid bacteria on channa argus: digestive enzyme activity, antioxidant capacity, intestinal microbiota and morphology. Aquac 531, 735852 (2021).
Yousef, M. I., El-Demerdash, F. M. & Radwan, F. M. Sodium arsenite induced biochemical perturbations in rats: ameliorating effect of curcumin. Food Chem. Toxicol. 46, 3506–3511 (2008).
Kumar, N., Jadhao, S. B., Chandan, N. K., Aklakur, M. & Rana, R. S. Methyl donors potentiates growth, metabolic status and neurotransmitter enzyme in Labeo Rohita fingerlings exposed to endosulfan and temperature. Fish. Physiol. Biochem. 38, 1343–1353 (2012).
Zhu, H. Effects of dietary Curcumin supplementation on growth, intestinal and liver health of juvenile American eel (Anguilla Rostrata) (2021).
Shinde, A. et al. N., Combined effect of mercury and ammonia toxicity and its mitigation using selenium nanoparticles in fish. Aquat. Toxicol., 107270 (2025).
Midhun, S. J. et al. Modulation of digestive enzymes, GH, IGF-1 and IGF-2 genes in the teleost, tilapia (Oreochromis mossambicus) by dietary curcumin. Aquac. Int. 24, 1277–1286 (2016).
Wen, Z. P., Zhou, X. Q., Feng, L., Jiang, J. & Liu, Y. Effect of dietary pantothenic acid supplement on growth, body composition and intestinal enzyme activities of juvenile Jian carp (Cyprinus Carpio Var. Jian). Aquacult. Nutr. https://doi.org/10.1111/J.1365-2095.2008.00612.X
Kumar, N. Dietary riboflavin enhances immunity and anti-oxidative status against arsenic and high temperature in pangasianodon hypophthalmus. Aquaculture 533, 736209
Kumar, N. et al. Nano–zinc enhances gene regulation of non–specific immunity and antioxidative status to mitigate multiple stresses in fish. Sci. Rep. 13, 5015 (2023).
Kumar, N., Thorat, S. T. & Reddy, K. S. Multi biomarker approach to assess manganese and manganese nanoparticles toxicity in pangasianodon hypophthalmus. Sci. Rep. 13 (1), 8505 (2023).
Giri, S. S. et al. Role of dietary curcumin against waterborne lead toxicity in common carp cyprinus carpio. Ecotoxicol. Environ. Saf. 219, 112318 (2021).
Mckenna, M. C., Waagepetersen, H. S., Schousboe, A. & Sonnewald, U. Neuronal and astrocytic shuttle mechanisms for cytosolic-mitochondrial transfer of reducing equivalents: current evidence and pharmacological tools. Biochem. Pharmacol. 71, 399–407 (2006).
Soengas, M. S. et al. Apaf-1 and caspase-9 in p53-dependent apoptosis and tumor inhibition. Science 284, 156–159 (1999).
Kumar, N., Thorat, S. T., Gite, A. & Patole, P. B. Nano-copper enhances gene regulation of non-specific immunity and antioxidative status of fish reared under multiple stresses. Biol. Trace Elem. Res. 201 (10), 4926–4950 (2023).
Zeng, C. et al. The role of apoptosis in MCLR-induced developmental toxicity in zebrafish embryos. Gig. Sanit. 149, 25–32 (2014).
Wei, M. C. et al. Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292, 727–730 (2001).
Hussain, A. R. et al. Curcumin induces apoptosis via inhibition of PI3′-kinase/AKT pathway in acute T cell leukemias. Apoptosis 11, 245–254 (2006).
Lawrence, T. The nuclear factor NF-κB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 1, a001651 (2009).
Jiang, J. et al. Glutamate attenuates lipopolysaccharide-induced oxidative damage and mRNA expression changes of tight junction and defensin proteins, inflammatory and apoptosis response signaling molecules in the intestine of fish. Fish. Shellfish Immunol. 70, 473–484 (2017).
Ming, J. et al. Optimal dietary curcumin improved growth performance, and modulated innate immunity, antioxidant capacity and related genes expression of NF-kappaB and Nrf2 signaling pathways in grass carp (Ctenopharyngodon idella) after infection with Aeromonas hydrophila fish. Shellfish Immun. 97, 540–553 (2020).
Li, L., Huang, Y. & Zhang, Z. Expression profile of mirnas involved in the hepatoprotective effects of curcumin against oxidative stress in nile tilapia. Aquat. Toxicol. 237, 105896 (2021).
Akdemir, F. et al. The efficacy of dietary Curcumin on growth performance, lipid peroxidation and hepatic transcription factors in rainbow trout Oncorhynchus Mykiss (Walbaum) reared under different stocking densities. Aquac. Res. 48, 4012–4021 (2017).
Cek, S., Turan, F. & Atik, E. The effects of gokshura, tribulus terrestris on sex differentiation of Guppy, Poecilia reticulata. Pak. J. Biol. Sci. 10, 718–725 (2007).
Acknowledgements
The present research was supported by institute in-house project (#IXX15014). The present work was conducted for completing master degree for first author. The authors are thankful to the Director, ICAR-National Institute of Abiotic Stress Management, Baramati, Pune for providing all the facilities for this study. The authors also acknowledge to Vidya Pratishthan’s Arts, Commerce and Science College Baramati, Pune for providing student for Master degree dissertation.
Funding
Institutional project (#IXX15014): Indian Council of Agricultural Research (ICAR), New Delhi, India has been provided financial and infrastructure support.
Author information
Authors and Affiliations
Contributions
Amir Najir Mulla: Data curation; Formal analysis, MethodologySupriya Tukaram Thorat: Data curation; Formal analysis, MethodologyKalpana Chandramore: ResourcesPrem Kumar: Validation; VisualizationK Sammi Reddy: Supervision; Neeraj Kumar: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Roles/Writing - original draft; and Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Institute Research Advisory Committee (RAC) has approved the experimental procedures. The present study was in complied with ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines. The methodology and care and maintenance of the fish were conducted in accordance with the relevant guidelines and regulations.
Consent for publication
Taken from PME, ICAR-NIASM, Baramati, Pune, Maharashtra, India.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mulla, A.N., Thorat, S.T., Chandramore, K. et al. Curcumin as a protective agent against chromium and ammonia toxicity using molecular and biochemical approaches in fish. Sci Rep 15, 12023 (2025). https://doi.org/10.1038/s41598-025-95369-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-95369-0