Abstract
Suppressing inflammation and promoting intestinal epithelial regeneration are the keys to mucosal healing in individuals with ulcerative colitis (UC). The upregulation of epithelial YAP and the induction of macrophages to polarize to the M2 phenotype in the mucosa can promote intestinal epithelial regeneration and alleviate ulcerative colitis. However, the role of YAP in macrophage polarization remains unclear. Here, we explored the effects of YAP on macrophage polarization and its biological role in a mouse DSS-induced colitis model. The results showed that YAP upregulation in macrophages could induce M2 polarization and increase the levels of anti-inflammatory cytokines such as IL-10 and IL-13. In addition, when mice were infused with YAP-overexpressing and empty vector-transfected macrophages, compared with control mice, YAP-overexpressing mice presented slower weight loss, a longer colon length, less intestinal inflammation, and a better arrangement of crypts. Moreover, macrophages in the lamina propria of the mouse colonic mucosa presented mainly the M2 phenotype in YAP-overexpressing macrophage-infused DSS-treated mice. Mechanistically, knockdown of the expression of the transcription factor TEAD4 in YAP-overexpressing macrophages inhibited macrophage M2 polarization and decreased anti-inflammatory cytokine expression, accompanied by the downregulated expression of C/EBPβ. Furthermore, silencing C/EBPβ following YAP overexpression suppressed M2 polarization. Chromatin immunoprecipitation revealed that TEAD4 was enriched at the C/EBPβ promoter region in YAP-overexpressing macrophages. Thus, YAP in macrophages regulates C/EBPβ expression through the transcription factor TEAD4, which mediates macrophage M2 polarization and inhibits the expression of inflammatory cytokines, thereby exerting inhibitory effects on intestinal inflammation and promoting mucosal healing in a colitis model.
Similar content being viewed by others
Introduction
Dysregulation of the immune response occurs in individuals with ulcerative colitis1, and macrophages are critical regulators of tissue repair, regeneration, and fibrosis2. Macrophages are divided into classically activated macrophages (M1 phenotype) and alternatively activated macrophages (M2 phenotype). M1 phenotype macrophages secrete proinflammatory cytokines involved in inflammation, such as IL-1β and IL-6. In contrast, M2 phenotype macrophages secrete anti-inflammatory and prorestorative cytokines involved in inflammation resolution and tissue repair, including TGF-β and IL-102,3,4. In the intestine, macrophages are considered to play a vital role in maintaining intestinal immunological homeostasis5. In recent years, the modulation of macrophage polarization has emerged as a potential therapeutic strategy to ameliorate and control ulcerative colitis5,6,7.
YAP is a critical cotranscription factor in the Hippo signalling pathway. Under homeostatic conditions in the intestinal epithelium, the Hippo signalling pathway negatively regulates intestinal epithelial cell proliferation and intestinal tumour development8,9. YAP also cross-regulates this process with Wnt/β-catenin, Notch, and other pathways9,10. Blocking the interaction between YAP and TEAD can inhibit cell proliferation and antitumour effects11. Among them, TEAD4 (TEA domain transcription factor 4), a member of the TEAD family, is recognized as a DNA anchor protein for the YAP transcription complex and is closely related to a variety of cancers, including colorectal cancer, gallbladder cancer and breast cancer12,13,14,15.
However, studies of the role of YAP in macrophage polarization have shown discrepancies. Previous research has indicated that YAP overexpression in macrophages promotes the proinflammatory response, whereas the downregulation of YAP restricts inflammation16. In individuals with inflammatory bowel disease and myocardial infarction, YAP impairs macrophage M2 polarization and promotes proinflammatory responses17,18. Conversely, some studies have indicated that YAP expressed in macrophages exerts anti-inflammatory effects. During acute lung injury, YAP in alveolar macrophages can downregulate NLRP3 through the YAP/β-catenin signalling axis, thereby inhibiting macrophage M1 polarization and lung inflammation19.
YAP and macrophages have been recognized as potential promising therapeutic targets for inflammatory bowel disease (IBD)6,7,20. In our study, we investigated the role of M2 macrophage polarization in ulcerative colitis and the mechanism by which YAP regulates macrophage polarization. We found that YAP in macrophages can protect against ulcerative colitis by promoting macrophage M2 polarization, which is achieved by modulating the YAP/TEAD4/C/EBPβ axis.
Methods
Reagents and antibodies
Dextran sodium sulfate (DSS) (MW 36,000–50,000) was purchased from MP Biomedicals. The following antibodies were used: CD86, CCR7, CD206, C/EBPβ, and Arg-1 antibodies were purchased from ABclonal; TEAD4, PCNA, cyclin D1, and Caspase3 antibodies were purchased from Proteintech; and Caspase4 and IL-1β antibodies were purchased from Cell Signaling Technology (CST). The primers used for real-time quantitative PCR were designed using the Primer Blast website and synthesized by Tsingke Biotechnology. RIPA buffer was purchased from Beyotime. The BCA protein concentration assay kit was purchased from NCM Biotech. The colour prestained protein marker, SDS‒PAGE Rapid Preparation Kit, 5 × SDS‒PAGE protein loading buffer and ECL reagent kit were purchased from Biosharp. Polyvinylidene fluoride (PVDF) membranes were purchased from Bio-Rad.
Cell culture
FHC, RAW264.7 and THP-1 cell lines were obtained from the American Type Culture Collection (ATCC). The cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM) or RPMI-1640 basal medium supplemented with 10% foetal bovine serum (FBS) and 1% penicillin/streptomycin at 37 °C with 5% CO2.
THP-1 cell differentiation and polarization
THP-1 cells were incubated with 100 ng/mL phorbol myristate acetate (PMA; Sigma‒Aldrich) for 24 h to induce cell differentiation. The medium was then removed, followed by the addition of 20 ng/mL interferon-γ [IFN-γ; PeproTech] and 100 ng/mL lipopolysaccharide and an incubation for 48 h to induce M1 macrophage polarization, while 20 ng/mL interleukin-4 [IL-4; PeproTech] and interleukin-13 [IL-13; PeproTech] were added and incubated for 48 h to induce M2 macrophage polarization.
Coculture system
The coculture system used 6-well plates and coculture chambers with 0.4 μm pores [Corning]. Briefly, FHC cells were incubated in 6-well plates at a density of 2 × 105 cells/well and stimulated with 1 mg/ml LPS for 24 h to obtain inflammatory FHC cells. YAP-overexpressing and control vector-expressing THP-1 cells were also stimulated with 100 ng/ml PMA for 24 h to obtain YAP-overexpressing (YAPWT) and control vector-expressing (vector) macrophages. Then, YAPWT and vector THP-1-derived macrophages were incubated in the upper chamber and cocultured with inflammatory FHC cells for 48 h. Finally, FHC were collected for the CCK-8, scratch, and immunoblotting assays.
Lentiviral transfection and selection of stable clones
We purchased lentiviral expression vectors from GeneChem to overexpress YAP in human and mouse cells and used these vectors to generate YAP-overexpressing RAW264.7 and THP-1 cells, respectively. Forty-eight or 72 h after lentiviral transfection, the fluorescence of the RAW264.7 and THP-1 cells was observed under a fluorescence microscope. When the level of fluorescence was high, 2 μg/ml puromycin was added for screening; finally, the overexpression of YAP was confirmed in THP-1 cells and RAW264.7 macrophages using western blotting and qPCR.
Plasmid and siRNA transfection
Plasmids for overexpressing C/EBPβ and small interfering RNAs (siRNAs) for the knockdown of TEAD4, YAP, and C/EBPβ were acquired from GenePharma. Lipofectamine 3000 (Invitrogen) was used to transfect the siRNAs or plasmids according to the manufacturer’s instructions.
The sequences of the siRNAs and the sequence of the C/EBPβ plasmid used are listed in Supplementary Tables S4–5.
Cell proliferation and wound healing assay
Inflammatory FHC cells were cocultured with YAP-overexpressing THP-1 macrophages or vector control THP-1 macrophages for 48 h. Subsequently, the migration and proliferation of the cocultured inflammatory FHC cells were examined by a scratch assay and a CCK-8 assay.
A total of 1000 inflammatory FHC cells were incubated in each well of 96-well plates in a volume of 100 µl of medium per well. The original medium was removed at 12 h, 24 h, 48 h, or 72 h, and fresh medium containing 10 µl of CCK-8 reagent was added, followed by an incubation at 37 °C for 2 h. The light absorption value at 450 nm was determined using an enzyme-linked immunoassay plate reader.
The migratory activity of the cocultured inflammatory FHC cells was assessed by a scratch wound healing test. The original medium was removed after the cells were scraped vertically using a sterile 100-µl pipette tip, and an appropriate amount of fresh serum-free medium was added. Finally, the migrated cells were observed and imaged at 0 h and 48 h.
Western blot
Equal amounts of protein samples were separated via sodium dodecyl sulfate‒polyacrylamide gel electrophoresis (SDS‒PAGE) on 6–10% gels and then transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with 5% skim milk, cut and incubated overnight with the corresponding specific primary antibodies, followed by washes and an incubation with horseradish peroxidase (HRP)-conjugated secondary antibodies before being removed for chemiluminescence detection.
RNA extraction and quantitative PCR
Total RNA was extracted from tissues and cells using TRIzol reagent (Servicebio) according to the manufacturer’s instructions. Complementary DNA (cDNA) synthesis was performed using a cDNA synthesis kit [Vazyme]. Quantitative real-time PCR was performed using qPCR SYBR Green Master Mix [Vazyme]. GAPDH was used as an internal reference control. The primers used are listed in Supplementary Table S2.
Human samples
Paraffin-embedded sections of biopsy specimens were obtained from the Department of Pathology of the Second Xiangya Hospital (n = 6 UC tissues and n = 6 normal tissues) with approval from the Medical Ethics Committee of the Second Xiangya Hospital (2019S274). Informed consent was obtained from all the subjects. The patients’ characteristics are listed in Supplementary Table S1. These sections were used for immunohistochemical (IHC) staining, and the average optical density values of the IHC-stained sections were analysed using ImageJ software.
Mouse study
Six- to eight-week-old C57BL/6 J male mice were purchased from the Experimental Animal Center of the Second Xiangya Hospital and maintained in a specific pathogen-free (SPF) environment. All animal experiments were approved by the Animal Ethics Committee of the Second Xiangya Hospital, Central South University (20,220,505). All methods were performed in accordance with the relevant guidelines and regulations and reported in accordance with the ARRIVE guidelines (https://arriveguidelines.org) for the reporting of animal experiments.
After one week of adaptation, the mice were randomly divided into four groups: the NC group, DSS group, YAPWT + DSS group, and vector + DSS group (n = 6 mice per group). For the YAPWT + DSS group and vector + DSS group, each mouse was infused with approximately 5 × 106 YAP-overexpressing RAW264.7 macrophages or control macrophages according to their respective groups. For the NC and DSS groups, equal volumes of PBS were injected. Three days after the injection, the NC group received normal water, whereas the mice in the DSS, YAPWT + DSS, and vector + DSS groups received 2.5% dextran sulfate sodium (DSS) (M.P. Biomedicals) dissolved in the drinking water for 7 consecutive days. The body weights and disease activity indices (DAIs) of the mice were monitored daily.
Extraction of lymphocytes from the mouse intestinal lamina propria
After being subjected to CO2 asphyxiation, the dissected colon was cut into 5 mm pieces. After washes with precooled PBS, epithelia were isolated by incubating the samples in DMEM containing 5 mM EDTA, 1 mM DTT, and 5% FBS at 37 °C for 25 min. Next, the samples were digested for 50 min in DMEM containing 1 mg/ml collagenase IV [Sigma], 1 mg/ml dispase II [Solarbio], and 5% FBS. Lymphocytes from the intestinal lamina propria were isolated using 30% and 70% Percoll isolation solutions [Solarbio].
HE staining and Immunohistochemistry
Mouse colon samples were fixed with 4% paraformaldehyde for 24 h and embedded in paraffin. Deparaffinized 5-μm-thick sections were used for HE or IHC staining. For IHC, the sections were subjected to endogenous peroxidase activity quenching, antigen retrieval, and subsequent blocking procedures. The sections were incubated overnight at 4 °C with the desired primary antibody and then with a biotinylated secondary antibody for 2 h at room temperature. The DAB kit was used to detect the signals.
Chromatin immunoprecipitation (ChIP) assay
Chromatin immunoprecipitation experiments were performed on YAP-overexpressing macrophages using the Protein A/G Magnetic Bead CHIP Kit (Beyotime) according to the manufacturer’s instructions. Briefly, the cells were crosslinked with 1% formaldehyde and disrupted with glycine, after which they were processed with a noncontact ultrasonic breaker. The supernatant was collected by centrifugation and incubated overnight with specific primary antibodies to obtain immunoprecipitated DNA‒protein complexes, followed by washing, elution, and reverse crosslinking steps. The immunoprecipitated DNA was subsequently analysed by qPCR. The primer sequences for the promoter regions used in this study are listed in Supplemental Table S3.
Data analysis
All the experiments were repeated at least three times, and the data are presented as the means ± SEMs. The data were statistically analysed and plotted using GraphPad Prism 8.0 software. Differences between two groups were determined by Student’s t test. p < 0.05 was considered statistically significant.
Statements
Human UC colon tissues were collected with the approval of the Institute Research Medical Ethics Committee of the Second Xiangya Hospital. Moreover, we confirmed that all the experiments were performed with the approval of the Animal Experimentation Committee of Southern Medical University (2019S274) and in accordance with institutional regulations.
Results
Ulcerative colitis enhanced M1 macrophage polarization and reduced M2 macrophage polarization in the mucosa
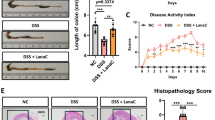
We examined the expression of different markers of macrophages in the colonic mucosa of UC patients to understand the role of macrophages in UC. We found that the expression of the M2 macrophage marker Arg-1 was decreased, whereas the expression of the M1 macrophage marker CD86 was increased in the inflamed colonic tissues of UC patients (Fig. 1A). Moreover, the body weights of the DSS-colitis group were significantly lower than those in the negative control group (Fig. 1C), and the colon lengths of the mice in the DSS-induced colitis group were significantly shorter (Fig. 1D). H&E staining revealed that the structure of the epithelium and villi of the colon of the mice in the DSS-induced group was extensively disrupted, resulting in prominent infiltration of inflammatory cells into the colonic mucosa (Fig. 1E). Immunohistochemical staining and western blotting revealed that the expression of the M2 macrophage marker Arg-1 was decreased in the colon tissues of the DSS group, whereas the expression of the M1 macrophage markers CD86 and CCR7 was increased, accompanied by a decrease in the expression of the proliferative marker PCNA and an increase in the expression of the apoptosis-associated factor Caspase-3 (Fig. 1B and 1F). Furthermore, we isolated lamina propria mononuclear cells of the mouse colon and found that the expression of the M1 macrophage markers CD86 and iNOS and the proinflammatory cytokine IL-6 was increased in the lamina propria of the colons of the mice in the DSS group (Fig. 1G). Overall, we suggest that the colonic mucosa exhibits impaired epithelial proliferation and increased apoptosis in a murine colitis model. Additionally, macrophages in the mucosa of UC patients present mainly with the M1 phenotype, accompanied by increased levels of proinflammatory cytokines.
Expression of macrophage markers and epithelial proliferation in the colonic tissues of humans and mice with colitis. The mice in the colitis group (DSS) were administered 2.5% dextran sodium sulfate for seven consecutive days, and the normal group (NC) was provided normal drinking water. (A) IHC detection of CD86 and Arg-1 expression in the inflamed or relatively normal colon tissue from patients with ulcerative colitis (UC). (B) Immunohistochemical (IHC) staining was performed to detect the expression of CD86, Arg-1, PCNA, and Caspase3 in the NC and DSS groups. (C) Comparison of body weight changes in the mice in the NC and DSS groups. (D) Comparison of colon length between the NC and DSS groups. (E) Histological morphology and structural changes in colon tissues from the NC and DSS groups were observed by H&E staining. (F) western blotting was performed to detect the expression of YAP, the macrophage markers Arg-1 and CCR7, the proliferation-associated factors PCNA and cyclin D1, and the apoptotic factor caspase3 in the mice from the NC and DSS groups. (G) Lamina propria mononuclear cells were extracted from the mouse colon for real-time fluorescence quantitative PCR to detect the expression of M1 macrophage markers and inflammatory cytokines. The asterisk represents statistical significance, * p < 0.05, ** p < 0.01, *** P < 0.001, **** P < 0.0001, ns, not significant; t test.
Overexpressing YAP in macrophages induced M2 polarization and promoted colonic epithelial cell proliferation
We stimulated THP-1 monocytes with PMA for 24 h to obtain nonpolarized M0 macrophages and then used LPS/IFN-γ to induce M1 macrophages and IL-4 to induce M2 macrophages to understand the functions of macrophages with different phenotypes. In M1-polarized macrophages, the expression of M1 macrophage markers, such as CD86, CCR7, and CD11c, was increased, accompanied by increased levels of the proinflammatory cytokines IL-6 and IL-1β. In contrast, in M2-polarized macrophages, the expression of M2 macrophage markers, including CD206, Arg-1, and Fizz1, was increased, accompanied by increased levels of the anti-inflammatory cytokines IL-10 and IL-13. Moreover, YAP expression was increased in M2-polarized macrophages and decreased in M1-polarized macrophages (Fig. 2A-B).
YAP promotes the M2 polarization of macrophages and repair of intestinal epithelial cells after inflammatory injury. (A) After THP-1 monocytes were stimulated with 100 ng/ml PMA for 24 h to obtain nonpolarized M0 macrophages, M1 macrophages were obtained by applying 100 ng/ml LPS and 20 ng/ml IFN-γ for 48 h, and the expression of macrophage markers and inflammatory factors was detected by western blotting and qPCR. (B) After nonpolarized M0 macrophages were obtained, M2 macrophages were induced with 20 ng/ml IL-4 for 48 h. Western blotting and qPCR were performed to detect the expression of macrophage markers and inflammatory factors. (C) Stable lentiviral transfection was used to construct YAP-overexpressing and empty control THP-1 cells, and 100 ng/ml PMA was incubated with the cells for 24 h to obtain YAP-overexpressing macrophages and control macrophages. Western blotting and qPCR were used to detect the expression of M1 and M2 macrophage markers and inflammatory factors. (D) After PMA-induced macrophages were generated, YAP expression in the macrophages was knocked down by transient siRNA transfection, and the corresponding target protein and mRNA expression levels were detected. (E) FHC cells were stimulated with 1 µg/ml lipopolysaccharide (LPS) for 24 h to establish an inflammatory intestinal epithelial cell model. The expression of the proinflammatory cytokines IL-6 and IL-1β and the anti-inflammatory cytokines IL-10 and IL-13 was detected by qPCR. (F) After LPS prestimulation, normal colonic epithelial cells (FHC cells) were cocultured with YAP WT macrophages or vector-transfected macrophages for 48 h, and the expression of YAP, the proliferation-associated factors PCNA and cyclin D1, and the apoptosis-associated factors caspase3 and caspase4 was detected by western blotting. (G-H) CCK-8 and scratch wound healing assays were performed to detect intestinal epithelial cell proliferation and migration after coculture. Asterisks indicate statistical significance, * p < 0.05, ** p < 0.01, *** p < 0.001, ****p < 0.0001; ns, not significant; t test.
Due to the critical role of YAP in intestinal mucosal repair21, we further investigated the role of YAP in macrophage polarization. We generated YAP-overexpressing macrophages by the stable transfer of a YAP-overexpressing lentivirus into THP-1 cells. We found that the expression of the M2 macrophage markers CD206, Arg-1, and Fizz1 and the anti-inflammatory cytokines IL-10 and IL-13 was increased in YAP-overexpressing THP-1 cells. However, the expression of the M1 macrophage markers CD86, CCR7, and iNOS and the proinflammatory cytokine IL-1β was decreased in YAP-overexpressing THP-1 cells (Fig. 2C). In contrast, the knockdown of YAP in THP-1 cells resulted in impaired M2 macrophage polarization and reduced expression of anti-inflammatory cytokines, whereas the expression of M1 macrophage markers and proinflammatory cytokines increased (Fig. 2D). These results suggest that upregulated YAP in macrophages mainly promotes M2 macrophage polarization. Increased YAP expression in macrophages might be a feature of M2 macrophages. Next, we constructed an inflammatory cell model to investigate the effect of macrophages on intestinal epithelial cells in the colonic mucosal microenvironment. We found that after LPS stimulation, the expression of the proinflammatory cytokines IL-1β and IL-6 was increased, and the expression of the anti-inflammatory cytokines IL-10 and IL-13 was decreased in FHC (Fig. 2F). However, when YAP-overexpressing (YAPWT macrophages) or vector-transfected THP-1 cells (control macrophages) were cocultured with LPS-prestimulated FHC cells, the expression of PCNA and cyclin D1 in FHC cells cocultured with YAPWT THP-1 cells was higher than that in the vector-transfected cells, while the expression of caspase-3 and caspase-4 was lower (Fig. 2E). In addition, the viability and “wound healing” ability of FHC cells were increased after coculture with YAPWT THP-1 cells compared with coculture with vector-transfected cells (Fig. 2G-H), suggesting that the overexpression of YAP in macrophages promoted the proliferation and wound healing of intestinal epithelial cells after inflammatory injury. Overall, YAP in macrophages may play an essential role in promoting intestinal epithelial repair by inducing macrophage polarization towards the M2 phenotype.
YAP overexpression in macrophages attenuated DSS-induced colitis in mice
We further validated the function of YAP-overexpressing macrophages in colitis by infusing YAP-overexpressing RAW264.7 macrophages (YAPWT + DSS) and control RAW264.7 macrophages (vector + DSS) into DSS-induced colitis model mice (Fig. 3A). The results showed that the colon length of the DSS group was significantly shorter than that of the NC group, and the body weight was significantly lower (Fig. 1D-E), whereas the colon length of the mice infused with YAP-overexpressing RAW264.7 macrophages was longer than that of the control RAW264.7 macrophage-injected mice, and the body weight was greater on Day 7 (Fig. 3B-C). Moreover, H&E staining revealed that the colonic tissues of the vector + DSS group exhibited extensive destruction of epithelial and villous structures and prominent infiltration of inflammatory cells. In contrast, the colonic tissues of the YAPWT + DSS group had more intact villous structures and less inflammatory cell infiltration in the mucosal layer (Fig. 3D). Moreover, the expression of the M1 macrophage markers CD86 and CCR7 and the apoptosis-associated gene Caspase3 decreased in the colons of the mice in the YAPWT + DSS group compared with those in the vector + DSS group. Moreover, the expression of the M2 macrophage marker Arg-1 and the proliferation-associated factors PCNA cyclin D1 was increased in the colons of the mice in the YAPWT + DSS group (Fig. 3E-F). More importantly, we isolated primary lymphocytes from the lamina propria of the colons of mice from different groups and found that, compared with the vector + DSS group of mice, the YAPWT + DSS group of mice presented increased expression of YAP and M2 macrophage markers, such as CD206, Arg-1, Fizz1, and the anti-inflammatory cytokine IL-10, and decreased expression of the M1 macrophage marker iNOS. Moreover, the transcription factor C/EBPβ, which regulates Arg-1 expression, was also upregulated in lamina propria lymphocytes from YAPWT + DSS mice, which illustrated that macrophages in the colonic lamina propria of mice injected with YAP-overexpressing RAW264.7 macrophages presented mainly the M2 phenotype (Fig. 3G). Overall, we believe that M2 macrophages can protect mice from DSS-induced colitis. Elevated YAP expression in macrophages can promote macrophage M2 polarization and increase the expression of anti-inflammatory cytokines in the colonic mucosa to attenuate mucosal inflammation and facilitate epithelial wound healing, and YAP-induced macrophage M2 polarization may be associated with the expression of C/EBPβ.
M2 macrophages attenuate DSS-induced colitis in mice in vivo. A YAP-overexpressing lentivirus was used to transfect RAW 264.7 macrophages and generate YAP-overexpressing RAW 264.7 macrophages. We subsequently divided the mice into four groups of six mice each; the YAP WT + DSS group and the vector + DSS group were infused with YAP-overexpressing RAW 264.7 macrophages and control RAW 264.7 macrophages, respectively, and the NC group and the DSS group were injected with an equal volume of PBS. After three days, except for the mice in the NC group, the mice were administered DSS for seven days. (A) Diagram of the mouse modelling process. (B) Comparison of the colon length between the YAP WT + DSS and vector + DSS groups of mice. (C) Comparison of body weight changes between the YAP WT + DSS and vector + DSS groups of mice. (D) H&E staining was performed to observe the histological morphology and structural changes in the colon tissues. (E) WB detection of the expression of target proteins in colon tissues. (F) Immunohistochemical (IHC) staining was performed to detect the expression of CD86, Arg-1, PCNA, and Caspase3 in colon tissues from the YAP WT + DSS group and the vector + DSS group. (G) Extraction of primary lamina propria lymphocytes from the mouse colon and qPCR detection of the expression levels of target mRNAs. Asterisks indicate statistical significance, * p < 0.05, ** p < 0.01; *** p < 0.001, ****p < 0.0001; ns, not significant; t test.
The YAP/TEAD4 transcriptional complex promotes M2 macrophage polarization through the regulation of C/EBPβ
High expression of YAP in macrophages promotes M2 macrophage polarization and exerts anti-inflammatory effects on mice with colitis. Finally, we explored the molecular mechanisms by which YAP expressed in macrophages regulates M2 macrophage polarization. TEAD is the most important transcription factor in the YAP family22. The combination of YAP and TEAD4 can promote cell proliferation, thereby promoting the occurrence of colorectal tumours15. TEAD4 can also promote the regeneration of intestinal crypts after radiation injury by acting alone or in combination with the transcription factor TCF4 through Wnt signalling23. Our study revealed that silencing TEAD4 in YAP-overexpressing THP-1 cells decreased the expression of the M2 macrophage markers CD206, Arg-1, and Fizz1 compared with that in the control group, whereas the expression of the M1 markers CD86 and CD11c was increased. Moreover, YAP-overexpressing THP-1 cells presented decreased expression of the anti-inflammatory cytokines IL-4, IL-10, and IL-13 combined with increased expression of proinflammatory cytokines such as IL-6 and IL-1β. These results indicated that YAP may regulate M2 macrophage polarization in a TEAD4-dependent manner (Fig. 4A-B). Then, we performed an iTRAQ proteomic analysis and found that C/EBPβ expression was significantly upregulated in YAP-overexpressing THP-1 cells (Fig. 4C). C/EBPβ is an essential factor that regulates Arg-1 expression24,25. Our study revealed that silencing CEBP/β in YAP-overexpressing THP-1 cells resulted in the downregulation of the M2 macrophage markers CD206, Arg-1, and anti-inflammatory cytokines, whereas CD86, IL-6, and IL-1β were upregulated (Fig. 4A-B). Furthermore, the overexpression of C/EBPβ in THP-1 cells resulted in increased expression of the M2 macrophage markers CD206, Arg-1, and Fizz1 and the anti-inflammatory cytokines IL-4, IL-10, and IL-13 compared with the levels in the control group, accompanied by reduced CD86 and IL-1β expression, indicating that CEBP/β mediates the function of YAP in regulating M2 macrophage polarization (Fig. 4D). We further investigated the interaction between the YAP/TEAD4 transcriptional complex and CEBP/β by performing chromatin immunoprecipitation and found that TEAD4 was strongly enriched at the C/EBPβ promoter region in YAP-overexpressing macrophages compared with control macrophages, suggesting that CEBP/β is a transcriptional target gene of the YAP/TEAD4 transcriptional complex (Fig. 4E). Therefore, the interaction of YAP with the transcription factor TEAD4 in macrophages promotes M2 macrophage polarization by transcriptionally regulating the target gene CEBP/β, thus inhibiting intestinal inflammation and promoting healing of the damaged mucosa.
The molecular mechanism by which YAP regulates the M2 polarization of macrophages. (A) In YAP-overexpressing macrophages, siRNA-mediated knockdown of TEAD4 or C/EBPβ was performed, and the expression of macrophage markers and inflammatory cytokines was detected by western blotting. (B) After the siRNA-mediated knockdown of TEAD4 or C/EBPβ in YAP-overexpressing THP-1-derived macrophages, the expression of macrophage markers and inflammatory cytokines was detected by real-time fluorescence quantitative PCR. (C) YAP-overexpressing and control macrophages were induced in vitro, followed by an iTRAQ proteomic analysis, and clustering heatmaps were produced using the Bioscience Cloud platform. (D) A plasmid was used to overexpress C/EBP β in macrophages, after which the expression of macrophage markers and inflammatory cytokines at the mRNA level was detected by real-time fluorescence quantitative PCR. (E) Chromatin immunoprecipitation (ChIP). Real-time fluorescence quantitative PCR was performed after immunoprecipitation, and the fold enrichment and percent input (% of input) of TEAD4 at the C/EBPβ promoter region were statistically analysed using GraphPad Prism 8.0 software. Asterisks indicate statistical significance, * p < 0.05, ** p < 0.01; *** p < 0.001; ****p < 0.0001; ns, not significant; t test.
Discussion
The development of ulcerative colitis (UC) is associated with many factors, among which the dysregulation of the immune system is an important feature. As a critical component of intrinsic immunity, macrophages are essential in ulcerative colitis. In individuals with colitis, the promotion of macrophage M2 activation or an increase in the M2/M1 macrophage ratio can play an essential role in alleviating intestinal inflammation and promoting intestinal tissue repair. The molecular mechanisms controlling the M2 polarization of macrophages in the lamina propria of the intestinal mucosa are unclear. Our study revealed that increased M1 macrophage activation and impaired M2 macrophage polarization occur in individuals with ulcerative colitis. M2-phenotype macrophages highly express YAP, and elevated YAP expression in macrophages can induce M2 polarization by binding to TEAD4 to form a transcriptional complex that transcriptionally upregulates the expression of the target gene C/EBPβ, which can upregulate Arg-1 expression. An infusion of macrophages overexpressing YAP protected mice from DSS-induced inflammatory injury.
YAP is involved in promoting cell proliferation, tissue repair, fibrosis, and tumorigenesis, but the role of YAP in macrophage polarization is controversial. In LPS-induced macrophages, NF-κB activation and TNF-α production require YAP activation and nuclear translocation. YAP can interact with the NF-κB subunit p65 to promote proinflammatory cytokine production26. In liver macrophages, YAP synergizes with Notch to promote M1 macrophage polarization27. In alveolar macrophages (AMs), activation of the YAP/β-catenin signalling pathway after acute lung injury inhibits macrophage M1 polarization and the secretion of proinflammatory cytokines, thus playing a protective role in acute lung injury19. In various types of cancer, high expression of YAP in macrophages promotes the polarization of macrophages to the M2 phenotype, thereby regulating tumorigenesis, progression, and metastasis28,29,30. In our study, M2 macrophages presented increased expression of YAP. YAP overexpression in macrophages is a characteristic of M2 macrophages, which highly express anti-inflammatory cytokines and promote epithelial wound healing. When YAP is knocked down in macrophages, they predominantly exhibit an M1 phenotype with increased expression of proinflammatory cytokines.
The mechanisms that regulate macrophage polarization have not been fully clarified. Currently, researchers believe that transcription factors such as NF-κB, activator protein-1 (AP-1), and STAT1 regulate macrophage M1 polarization. In contrast, interferon regulatory factor 4 (IRF4), CCAAT/enhancer-binding protein β (C/EBPβ), STAT6, and peroxisome proliferator-activated receptor γ (PPAR-γ) promote M2 activation31. Arginase 1 (Arg-1) is a significant marker of M2 macrophages, and its expression indicates the predominance of the M2 anti-inflammatory phenotype. The transcription factor C/EBP β is believed to mediate the expression of Arg-1 in macrophages24,32. In our study, the knockdown of C/EBPβ in YAP-overexpressing THP-1-derived macrophages resulted in decreased Arg-1 expression, and the overexpression of C/EBPβ promoted macrophage M2 polarization and increased the expression of anti-inflammatory cytokines. As both YAP and C/EBPβ are involved in M2-type macrophage polarization, we explored whether they constitute an internal regulatory mechanism that promotes M2 polarization. In this study, we screened for C/EBPβ by iTRAQ sequencing in YAP-overexpressing and control macrophages. Moreover, a positive correlation was observed between YAP and C/EBPβ expression in macrophages. The TEAD family is the most classic synergistic transcription factor of YAP, and YAP/TEAD4 promote cell proliferation and tumorigenesis, such as in colorectal and hepatocellular carcinomas15,29,33. Additionally, we explored whether YAP regulates C/EBPβ expression through TEAD4. High expression of YAP promoted M2 macrophage polarization and increased the expression of anti-inflammatory cytokines. When TEAD4 was knocked down in YAP-overexpressing macrophages, they mainly exhibited the M1 phenotype and high levels of proinflammatory cytokines. Moreover, TEAD4 could bind to the C/EBPβ promoter, and TEAD4 knockdown resulted in a decrease in C/EBPβ expression, suggesting that YAP interacted with TEAD4 to transcriptionally regulate the expression of C/EBPβ and induce M2 polarization.
In summary, our study reveals a new molecular mechanism by which YAP regulates macrophage polarization, which may facilitate the development of potential therapeutic strategies for UC. We showed that YAP in macrophages regulates the expression of C/EBPβ by binding to the transcription factor TEAD4, which promotes macrophage M2 polarization, inhibits inflammation, and ultimately controls and ameliorates ulcerative colitis.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Ordás, I., Eckmann, L., Talamini, M., Baumgart, D. C. & Sandborn, W. J. Ulcerative colitis. Lancet 380, 1606–1619 (2012).
Wynn, T. A. & Vannella, K. M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity 44, 450–462 (2016).
Brazil, J. C., Quiros, M., Nusrat, A. & Parkos, C. A. Innate immune cell-epithelial crosstalk during wound repair. J. Clin. Invest. 129, 2983–2993 (2019).
Shapouri-Moghaddam, A. et al. Macrophage plasticity, polarization, and function in health and disease. J. Cell Physiol. 233, 6425–6440 (2018).
Pan, X., Zhu, Q., Pan, L. L. & Sun, J. Macrophage immunometabolism in inflammatory bowel diseases: From pathogenesis to therapy. Pharmacol. Ther. 238, 108176 (2022).
Deng, F. et al. A molecular targeted immunotherapeutic strategy for ulcerative colitis via dual-targeting nanoparticles delivering miR-146b to intestinal macrophages. J. Crohns Colitis 13, 482–494 (2019).
Lu, J. et al. Thalidomide attenuates colitis and is associated with the suppression of M1 macrophage polarization by targeting the transcription factor IRF5. Dig. Dis. Sci. 66, 3803–3812 (2021).
Meng, Z., Moroishi, T. & Guan, K. L. Mechanisms of Hippo pathway regulation. Genes Dev. 30, 1–17 (2016).
Moya, I. M. & Halder, G. Hippo-YAP/TAZ signalling in organ regeneration and regenerative medicine. Nat. Rev. Mol. Cell Biol. 20, 211–226 (2019).
Yu, F. X., Meng, Z., Plouffe, S. W. & Guan, K. L. Hippo pathway regulation of gastrointestinal tissues. Annu. Rev. Physiol. 77, 201–227 (2015).
Hagenbeek, T. J. et al. An allosteric pan-TEAD inhibitor blocks oncogenic YAP/TAZ signaling and overcomes KRAS G12C inhibitor resistance. Nat. Cancer 4, 812–828 (2023).
Kim, J. Y., Sung, Y. N. & Hong, S. M. High YAP and TEAD4 immunolabelings are associated with poor prognosis in patients with gallbladder cancer. APMIS 129, 729–742 (2021).
Chen, M. et al. Structural and functional overview of TEAD4 in cancer biology. Onco Targ. Ther. 13, 9865–9874 (2020).
Hsu, S. C. et al. TEAD4 as an oncogene and a mitochondrial modulator. Front. Cell Dev. Biol. 10, 890419 (2022).
Tang, J. Y. et al. TEAD4 promotes colorectal tumorigenesis via transcriptionally targeting YAP1. Cell Cycle 17, 102–109 (2018).
Meli, V. S. et al. YAP-mediated mechanotransduction tunes the macrophage inflammatory response. Sci. Adv. https://doi.org/10.1126/sciadv.abb8471 (2020).
Mia, M. M. et al. YAP/TAZ deficiency reprograms macrophage phenotype and improves infarct healing and cardiac function after myocardial infarction. PLoS Biol. 18, e3000941 (2020).
Zhou, X. et al. YAP aggravates inflammatory bowel disease by regulating M1/M2 macrophage polarization and gut microbial homeostasis. Cell Rep. 27, 1176-1189.e5 (2019).
Zhang, T. et al. Exosome from BMMSC attenuates cardiopulmonary bypass-induced acute lung injury Via YAP/β-catenin pathway: Downregulation of pyroptosis. Stem. Cell. 40, 1122–1133 (2022).
Daskalaki, M. G. et al. Neorogioltriol and related diterpenes from the red alga laurencia inhibit inflammatory bowel disease in mice by suppressing M1 and promoting M2-like macrophage responses. Mar. Drug. https://doi.org/10.3390/md17020097 (2019).
Deng, F. et al. YAP triggers the Wnt/β-catenin signalling pathway and promotes enterocyte self-renewal, regeneration and tumorigenesis after DSS-induced injury. Cell. Death Dis. 9, 153 (2018).
Zhao, B. et al. TEAD mediates YAP-dependent gene induction and growth control. Genes Dev. 22, 1962–1971 (2008).
Zhu, X. et al. REGγ drives Lgr5(+) stem cells to potentiate radiation induced intestinal regeneration. Sci. China Life Sci. 65, 1608–1623 (2022).
Lamkin, D. M. et al. C/EBPβ regulates the M2 transcriptome in β-adrenergic-stimulated macrophages. Brain Behav. Immun. 80, 839–848 (2019).
Ruffell, D. et al. A CREB-C/EBPbeta cascade induces M2 macrophage-specific gene expression and promotes muscle injury repair. Proc. Natl. Acad. Sci. U. S. A. 106, 17475–17480 (2009).
Yang, K. et al. Lactate suppresses macrophage pro-inflammatory response to LPS stimulation by inhibition of YAP and NF-κB activation via GPR81-mediated signaling. Front. Immunol. 11, 587913 (2020).
Yang, Y. et al. Targeting notch1-YAP circuit reprograms macrophage polarization and alleviates acute liver injury in mice. Cell. Mol. Gastroenterol. Hepatol. 15, 1085–1104 (2023).
Zhang, Y. et al. OTUD5-mediated deubiquitination of YAP in macrophage promotes M2 phenotype polarization and favors triple-negative breast cancer progression. Cancer Lett. 504, 104–115 (2021).
Thomann, S. et al. YAP-induced Ccl2 expression is associated with a switch in hepatic macrophage identity and vascular remodelling in liver cancer. Liver Int. 41, 3011–3023 (2021).
Huang, Y. J. et al. Ovatodiolide suppresses colon tumorigenesis and prevents polarization of M2 tumor-associated macrophages through YAP oncogenic pathways. J. Hematol. Oncol. 10, 60 (2017).
Lawrence, T. & Natoli, G. Transcriptional regulation of macrophage polarization: Enabling diversity with identity. Nat. Rev. Immunol. 11, 750–761 (2011).
Barilli, A. et al. Gluten peptides drive healthy and celiac monocytes toward an M2-like polarization. J. Nutr. Biochem. 54, 11–17 (2018).
Cai, J. et al. Bulk and single-cell transcriptome profiling reveal extracellular matrix mechanical regulation of lipid metabolism reprograming through YAP/TEAD4/ACADL axis in hepatocellular carcinoma. Int. J. Biol. Sci. 19, 2114–2131 (2023).
Funding
This work was supported by the Natural Science Foundation of Hunan Province [Project No. 2020JJ5].
Author information
Authors and Affiliations
Contributions
S.W: study concept, experimental manipulation, data analysis, illustration drawing and manuscript draft; F.Z: technological support and manuscript revision; MM.X, ZR.W and PP.X: data analysis and manuscript revision; FH.D: study design, manuscript revision, study supervision and obtained funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflicts of interests
The authors declare that the research was conducted without any commercial or financial relationships that could be construed as potential conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, S., Zou, F., Xu, M. et al. The YAP/TEAD4 transcriptional complex in intestinal macrophages promotes M2 polarization and alleviates DSS-induced colitis via the regulation of C/EBPβ. Sci Rep 15, 11796 (2025). https://doi.org/10.1038/s41598-025-95933-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-95933-8
Keywords
This article is cited by
-
TEAD4 and RXRA Regulate the Function of Nucleus Pulposus Cells in Intervertebral Disc Degeneration Via the TNF-α/NF-κB Pathway: An Integrated Analysis of Single-Cell RNA-Seq, Bulk RNA-Seq, and In Vitro Validation
Applied Biochemistry and Biotechnology (2026)
-
M2 macrophage polarization in allogeneic skin transplantation: from intrinsic mechanisms to clinical prospects for immune tolerance
Molecular Biology Reports (2026)