Abstract
RNA binding proteins (RBPs) are key factors regulating post-transcriptional events. Categorized as RBPs, RBM24 expression levels have been shown to be a prognosis-related pivotal gene in oral squamous cell carcinoma. We analyzed the binding targets and regulated post-transcriptional events of RBM24 in RBM24-overexpressing cell lines and controls using iRIP-seq and RNA-seq. RBM24-overexpressing cells showed significant changes in gene expression, which are involved in biological pathways related to apoptosis and immune inflammation. RBM24-regulated genes that undergo alternative splicing are primarily engaged in biological processes related to DNA damage repair and RNA metabolism. More notably, we found that RBM24 binds to lncRNAs in addition to pre-mRNAs. These results indicated that RBM24 could play a key role in cancer progression by finding clinical therapeutic targets for Oral squamous cell carcinoma.
Similar content being viewed by others
Introduction
Oral squamous cell carcinoma (OSCC) is one of the most common malignant tumors of the head and neck, causing more than 90% of all kinds of oral cancer worldwide. OSCC occurs through a complex mechanism in which normal oral keratin-forming cells can be regulated by multiple signaling pathways and thus transformed into precancerous lesions that further deteriorate into malignant tumors1. Frequent interactions with carcinogens can initiate a complicated, multi-step carcinogenic process made by accumulating genetic and epigenetic alterations in key genes, leading to oncogene activation, inactivation, or loss of tumor suppressor genes2,3,4. Epidemiological findings suggest that the incidence and mortality of OSCC will continue to increase in the next 20 years5. Therefore, there is an urgent need to develop early detection biomarkers for OSCC and to find highly credible therapeutic targets.
As proteins that regulate gene expression by binding RNA, RBPs are progressively accepted as critical mediators of molecular and cellular homeostasis, serving a critical role in increasing the functional diversity of the proteome6. Up to now, certain RBPs have been found to have a significant role in OSCC tumorigenesis, such as FXR1 and IGF2BPs7,8. As a highly conserved RBP, RNA binding motif protein 24 (RBM24) exhibits a remarkable tissue-specific expression pattern during embryonic development9. There is evidence that dysregulated expression levels of RBM24 are linked to a wide range of congenital diseases, including cardiomyopathy, skeletal muscle regeneration disorders, and hearing impairment10,11,12. As a splicing regulator, RBM24 can modulate the above diseases by broadly regulating alternative splicing(AS) of genes involved in a variety of biological processes. For example, excessive ablation of RBM24 during early embryonic development will lead to rapidly progressing dilated cardiomyopathy, heart failure and postnatal mortality events. Previous studies using the Cre-loxP method to obtain conditioned RBM24 knockout mice and removing 5-day postnatal mouse heart tissues for sequencing resulted in the identification of 590 ASEs in 292 genes13. Knockdown of RBM24 disrupts the normal myonodal structure in cardiomyocytes by directly binding to the precursor mRNA of the key gene for dilated cardiomyopathy, TTN, leading to abnormal splicing of the gene. Meanwhile, knockdown of RBM24 in mice resulted in sustained weight loss and impaired skeletal muscle function, which demonstrated that RBM24 deficiency would result in dysregulation of ASEs related to a variety of biological processes, such as myogenesis, muscle regeneration and muscle hypertrophy. Mechanistic studies have shown that RBM24 achieves these effects by regulating variable ASEs in the myogenesis-related genes Mef2d, Naca, Rock2, and Lrrfip111. AMPA-type glutamate receptors (AMPARs) mediate rapid excitatory neurotransmission in the mammalian CNS, and GluR2 subunits play a major role in this. The two best-known splice variants of GluR2 are the ‘flip’ splice variant, which contains exon 14, and the ‘flop’ splice variant, which contains exon 15, and in RBM24 knockout mice, exon 14 and exon 15 of GluR2 contain a reduced number of AMPARs. Exon 14 and exon 15 in RBM24 knockout mice showed reduced GluR2 exon 14 and exon 15 inclusion and cognitive dysfunction14. In addition to this, RBM24 has been shown to regulate multiple post-transcriptional events in different cancers, including (1) inhibition of translation-initiating complex assembly to regulate mRNA translation. In a single investigation, RBM24 bound to the 3′-untranslated region (3′-UTR) of p53 mRNA and interacted with eIF4E, thereby inhibiting p53 mRNA translation by blocking the assembly of the translation-initiating complexes15. (2) Binding to the 3′-UTR regulates mRNA stability. For example, RBM24 interacts with the 3′-UTR of PTEN mRNA to maintain its stability in colorectal cancer (CRC), thereby increasing PTEN expression and inhibiting CRC cell proliferation, migration, and invasion16. In bladder cancer (BC), RBM24 can drive BC progression by binding and enhancing the stability of Runx1t1 mRNA to form RBM24/Runx1t1/TCF4/miR-625-5p axes17. (3) regulates the expression of non-coding RNA (ncRNA). In nasopharyngeal carcinoma, RBM24 can inhibit tumor formation by downregulating miR-25 to target cancer-promoting lncRNA-MALAT24, reducing its expression levels18. These observations demonstrate the inhibitory effect of RBM24 on tumorigenesis. Although RBM24 has been reported to serve as an important prognostic indicator in head and neck squamous cell carcinoma, its specific function in OSCC is not clear, nor is it clear whether RBM24 as an RBP affects gene expression and AS19. We speculate that RBM24 may interact with mRNAs of key cancer-associated genes and modulate their AS patterns, thereby altering the proliferation, apoptosis, and motility of OSCC.
To test the above hypothesis, we established a human tongue squamous cell carcinoma cell line (CAL27) model of RBM24 overexpression (OE), and obtained transcriptome data on RBM24 regulation in CAL27 cells through RNA-seq. By analyzing transcriptomic data, we elucidated the characteristics of differential genes and alternative splicing events(ASEs) regulated by RBM24. We also utilized iRIP-seq to find RBM24 binding sites and discovered that RBM24 interacts with mRNA and lncRNA20. We have identified some key RBM24-binding genes that regulate cancer development, such as NEAT1 and NPM1. These studies have helped to provide new insights into the molecular mechanisms underlying the development of OSCC, which is important in the search for emerging loci for OSCC-targeted therapies.
Material and methods
Cloning and plasmid construction
pcDNA3.1-RBM24 (NM_001128148) was purchased from Youbio Biotech (Changsha, PRC).
Cell culture and transfections
DMEM media (Procell Life Sciences, China) added with 10% fetal bovine serum (FBS) and antibiotics (100 g/mL streptomycin, 100 U/mL penicillin) was used to cultrue the CAL-27 cell line (RRID:CVCL_1107, Procell Life Sciences, China). CAL27 cells were inoculated into 12-well culture dishes. We took 500 ng of plasmid per well, added serum-free medium for dilution, and incubated for five minutes. At the same time, we took 1ul of Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) per well and added the same serum-free medium for dilution, incubated it for 5 min, and then mixed it with the plasmid and added it to the 12-well plate. After resting for 20 min, we placed the 12-well plates in a 37 °C incubator containing 5% CO2 for 6 h. After replacing the complete medium, we continued to incubate the cells for 48 h. Subsequently, we tested the transfection efficiency using RT-qPCR and western blot.
RT-qPCR
We implemented standard methods to extract total RNA and synthesize cDNA. We employed Hieff qPCR SYBR® Green Master Mix (Low Rox Plus; YEASEN, China) to perform real-time QPCR after eliminating interfering DNA. According to the instruction manual, add 0.5 ul of primer F, 0.5 ml of primer R, 5 ul of Hieff® qPCR SYBR Green Master Mix, 3 ul of H2O, and 1ul of cDNA to each cDNA reaction system for a total volume of 10ul. Primer information is included in Table S1. By using the 2−ΔΔCT method, we quantified each transcript level with graphed mRNA levels.
Western blot
We homogenized the CAL27 cell samples with RIPA lysis buffer (Beyotime, China) and crushed them, lysed them on ice for 30 min, sonicated them in an ice bath for 3 min, and centrifuged them at 12,000 rpm at 4 °C for 10 min. The supernatant was extracted, and the concentration of the sample was measured by the BCA protein quantification kit (Beyotime, China). Samples were boiled for 10 min in boiling water with 1X SDS sample buffer. We used 12% SDS-PAGE to separate the protein samples and then used PVDF membranes for transmembrane. The membranes were placed in TBST buffer containing 5% skimmed milk and incubated for 1 h at room temperature. To bind the primary antibody to the target protein, we co-incubated the membrane with the primary antibodies, which included flag antibody (1:5,000, AE005, ABclonal) and Alpha Tublin (1:15000, 11224-1-AP, Proteintech), and incubated it overnight at 4 °C. We then incubated the PDVF membrane with HRP-labeled secondary antibodies (anti-mouse or anti-rabbit 1:10,000) (Abcam) for 1 h at room temperature. We detected the bands with an enhanced chemiluminescence (ECL) reagent (Bio-Rad, 170506).
Flow cytometric analysis of cell apoptosis
We used 0.25% trypsin without EDTA to digest CAL27 cells, and after terminating the digestion, we collected the above cells and rinsed them twice with PBS. We used the AnnexinV-APC/7-AAD Apoptosis Detection Kit(KeyGene BioTech, China)for apoptosis analysis. And then we detected apoptosis with flow cytometry (Cytoflex, Beckman).
CCK-8
The transfected cells were digested with trypsin, centrifuged and resuspended, and the cell density was adjusted to 1*104 cells/ml using a cell counting plate.Cells at the above concentrations were inoculated into 96-well plates, and three replicate wells were set up for each group. The old medium was removed from the wells under light-avoidance conditions, 10ul of CCK-8 solution was added to each well, and the absorbance at 450 nm was measured after incubation for 1.5 h under light-avoidance conditions, and the above steps were repeated at 0 h, 24 h, 48 h, and 72 h, respectively.
RNA-seq
Total RNA was extracted from the samples using Trizol reagent (Yeasan, China). These RNA samples were further purified using chloroform and isopropanol and then treated with RQ1 DNase (Promega) to remove DNA. Utilizing a Smartspec Plus (BioRad) instrument, the absorbance at 260/280 nm (A260/A280) was measured in order to assess the purity and quantity of the RNA. 1.5% agarose gel electrophoresis was used to further confirm the integrity of the RNA. The extracted total RNA was stored at − 80 °C before sequencing. For each sample, 1 μg of total RNA was used for RNA-seq library preparation. In accordance with Illumina’s protocols, high-quality RNA was processed for the preparation of cDNA libraries using a KAPA-stranded mRNA-seq kit. Following the manufacturer’s instructions, cDNA libraries were prepared for high-throughput sequencing and applied to the Illumina Novaseq 6000 system for 150 nt paired-end sequencing. RNA sequencing raw data were processed and alignment as described in previous studies21.
Differentially expressed genes (DEG) analysis and AS analysis
The R Bioconductor package DESeq2 was utilized to screen out the differentially expressed genes (DEGs)22. The P value < 0.05 and fold change ≥ 1.5 or ≤ 0.67 were set as the cut-off criteria for identifying DEGs. As previously mentioned, the ABLas pipeline was utilized to characterize and quantify the regulated alternative splicing events (RASEs) and ASEsamong the samples23. To assess RBP-regulated ASE, a Student’s t-test was performed to evaluate the significance of the ratio alteration of AS events. RBP-regulated ASEs were defined as events that were significant at a P-value cutoff corresponding to a false discovery rate cutoff of 5%.
Improved RNA immunoprecipitation and sequencing
The RBM24 co-immunoprecipitation was performed as described24. CAL27 cells were irradiated once with 400 mJ/cm2 of UV light to crosslink RNA-binding proteins (RBPs) to their bound RNAs. Cells were lysed in ice pre-cooled lysis buffer (1× PBS, 0.5% SDS, 0.5% NP-40 and 0.5% sodium deoxycholate) for approximately 30 min, followed by centrifugation at 10,000 rpm for 10 min at 4 °C to remove cell debris. The lysate was treated with RQ I DNase (1 U/μl, final concentration 0.1 U/μl) for 30 min at 37 °C to remove DNA interferences and the reaction was subsequently terminated. The mixture was then vibrated vigorously and centrifuged at 13,000 × g at 4 °C for 15 min to remove cell debris. Then RNA digestion by MNase (Thermo Scientific,USA) was performed. For immunoprecipitation, the supernatant was incubated overnight at 4 °C with 10 μg Flag-antibody (Proteintech,China) and control IgG-antibody (AB clonal,China). The immunoprecipitates were further incubated with Protein A/G Dynabeads for 2 h at 4 °C to capture the antibody-bound complexes. The beads were then washed sequentially with washing buffer (1× PBS, 0.1% SDS, 0.5% NP-40 and 0.5% sodium deoxycholate), high-salt buffer (250 mM Tris 7.4, 750 mM NaCl, 10 mM EDTA, 0.1% SDS, 0.5% NP-40 and 0.5 deoxycholate), and PNK buffer (50 mM Tris, 20 mM EGTA and 0.5% NP-40) for two times, respectively. Resuspend the beads in of Elution buffer (50 mM Tris 8.0, 10 mM EDTA and 1% SDS). Incubate the suspension for 30 min in a heat block at 70 °C to release the immunoprecipitated RBP with crosslinked RNA and vortex. Transfer the supernatant to a 1.5 ml EP tube. Proteinase K (final concentration 1.2 mg/ml) was added to both the input (non-immunoprecipitated) and immunoprecipitated samples to digest proteins and release crosslinked RNA (Sangon Biotech, China). The samples were incubated at 55 °C for 2 h. The RNA was purified with Trizol reagent(Accurate Biology, China). cDNA libraries were prepared with the KAPA RNA Hyper Prep Kit (KAPA, KK8541). According to the manufacturer’s instructions, the libraries were constructed and used for 150nt paired-end sequencing through the Illumina NovaSeq system.
Data Analysis
After reads were aligned onto the genome with HISAT2, the unique comparison on the genome was finally obtained, and the comparison result of PCR duplicate was removed. And then two software programs, Piranha and ABLIRC, were used to perform peak calling25. At the same time, reads on each gene were randomly distributed to each gene for 500 times. We counted the frequency of observed peak depths and analyzed the identified peaks for significance analysis. Significant peaks were identified using a P-value < 0.05 or peak maximum depth ≥ 10 as a threshold. With the input samples as the control, simulated independence analyses were performed on the two samples, and the peak with IP abundance greater than 4 times (adjustable parameter) of input abundance was screened as the final combination peak. The RBM24 target genes were ultimately determined by the peaks, and the binding motifs of the RBM24 protein were identified using HOMER software26.
Functional enrichment analysis
To classify peak-associated genes (target genes) into functional categories, Gene Ontology (GO) terms were identified using the KOBAS 2.0 server27. Hypergeometric test and Benjamini–Hochberg FDR controlling procedure were used to define the enrichment of each term.
Statistical analysis
The data from at least three independent experiments were presented as mean Standard Deviation (SD). Data were analyzed using either Student’s t-test(two-group comparison)or two- way ANOVA (more than two groups). A p-value of < 0.05 was considered statistically significant (*p 0.05; **p < 0.01; ***p < 0.001). All graphs were made using the GraphPad Prism software (Version number8.0, San Diego, CA).
Results
In vitro functional analysis of RBM24 in CAL27 cells
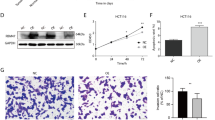
It has been shown that RBM24 can be one of the biomarkers for the prognostic assessment of OSCC. To determine the detailed mechanism of RBM24 in OSCC, we constructed and tested a model of RBM24 overexpression (RBM24-OE) in CAL27 cells. The results of RT-qPCR combined with western blotting showed that plasmids can effectively increase the mRNA (P < 0.0001) and protein expression levels of RBM24 (Fig. 1A and B). On the basis of apoptosis detection by flow cytometry, apoptosis levels were significantly increased after RBM24 overexpression (Fig. 1C and D, P < 0.001). In the CCK-8 experiment, CCK-8 was added to the NC and OE groups at 0 h, 24 h, 48 h and 72 h. The OD values of each group were detected at each time point, as shown in Fig. 1E, and it was observed that the relative proliferation ratio of the OE group was lower than that of the NC group at all stages, and the difference in proliferation rate between the two groups gradually increased with time, which was statistically significant. In summary, RBM24 can inhibit OSCC malignant progression.
RBM24 inhibits apoptosis in CAL27 cell. (A, B) RT-PCR (A) and Western blotting. (B) analysis of LTA4H expression. (C, D) Flow-type apoptosis results of CAL27 cells after RBM24 overexpression. (E) Cell proliferation was determined by CCK-8 assay after RBM24 overexpression. Error bars represent mean ± SEM. **** P-value < 0.0001, *** P-value < 0.001.
RBM24 regulates gene expression in CAL27 cells
In order to obtain transcriptome data regulated by RBM24, transcriptome sequencing was conducted between the RBM24 overexpression group and the negative control group to identify RBM24-related downstream signals in CAL27. We prepared cDNA libraries for the RBM24-OE group and the control group. We evaluate the RNA-seq libraries for quality control and then complete the sequence of six RNA-seq libraries. Effective overexpression of RBM24 was verified in the RNA-seq evaluation (Fig. 2A, p < 0.001). By principal component analysis, we found substantial differences between the above two groups (Fig. 2B). Through using the edgeR package to identify differentially expressed genes (DEGs), we screened 301 significant DEGs with a threshold of fold change ≥ 3/2 or ≤ 2/3 and a P-value < 0.05. We constructed a volcano plot to show significant DEGs between the RBM24-OE and control groups. The plot included 188 up-regulated genes and 113 down-regulated genes (Fig. 2C). According to heat map analysis of the DEG expression patterns in the RNA-seq samples, RBM24-regulated transcription was very consistent in both groups, demonstrating the validity of RNA-seq-based transcriptional profile assessments (Fig. 2D). Gene Ontology (GO) functional enrichment analysis was performed on the DEGs to investigate their potential biological functions. Then, we observed that the up-regulated genes were functionally concentrated on biological processes connected to apoptosis, such as positive regulation of the apoptotic process and the apoptosis process. The down-regulated genes, on the other hand, were associated with biological processes connected to immunological inflammation such as neutrophil tropism, inflammatory response, chemotaxis, chemokine-mediated signaling pathway, antimicrobial peptide-mediated antimicrobial humoral immune response, immune response, the cytokine-mediated signaling pathway, lymphocyte tropism, and monocyte tropism (Fig. 2E).
Analysis of RBM24-regulated differential genes by RNA-seq. (A) The expression pattern of DEGs for RBM24. (B) PCA base on FPKM value of all detected genes RBM24 overexpression. (C) All DEGs between RBM-OE and NC samples. (D) Hierarchical clustering of DEGs in RBM24-OE and NC samples. (E)The top 10 representative GO Biological Process results of the up-regulated and down-regulated DEGs. (F) RNA-seq and RT-qPCR showed that RBM24 regulates several differentially expressed genes. Error bars represent mean ± SEM.*** P-value < 0.001.
These data imply that RBM24 may be involved in apoptosis and immunological inflammatory pathways in OSCC, which are consistent with the results of flow cytometry apoptosis assays. Among them, pro-apoptotic genes such as GADD45B, HMOX1, ANKRD1, and GADD45G were upregulated, and immune inflammation-related genes such as S100A9, S100A8, S100A7, IL1A, CXCL3, CXCL10, CXCL11, CCL2, CCL5, and CCL20 were downregulated. Through searching the TCGA database (the Cancer Genome Atlas), we evaluated the expression levels of the aforementioned DEGs in HNSC and normal tissues. The expression levels of the mentioned genes (transcripts per million; TPM) are depicted using box-line plots (Figure S1). ANKRD1 was expressed at a high level in HNSC compared to normal tissues, while IL1A, CCL5, CCL20, CXCL10, and CXCL11 were at low levels, which are highly consistent with the sequencing data. To further verify whether RBM24 affected the expression of the above DEGs, we performed RT-qPCR to determine their mRNA levels. In the RBM24 overexpression group, all of these DEGs’ expression levels proved to be significantly changed, consistent with RNA-seq analysis (Fig. 2F).
RBM24 regulates gene AS in CAL27 cells
To investigate the regulatory mechanism of RBM24 on ASEs, we used RNA-seq data for ASEs analysis in CAL27 cells. Then we used the ABLas software tool and detected a total of 498,615 ASEs, including 215,826 known and 282,789 new ones. In addition, by setting a criterion of p-value ≤ 0.05, we identified ASEs regulated by RBM24 in CAL27 cells. 2900 regulatory RASEs were obtained. As shown in Fig. 3A,the above RASEs mainly included 418 cassette exons, 166 mutually exclusive exons (MXE), and 450 exon skipping (ES), 130 A5SS&ES, 692 alternative 5’splice site (A5SS), 106 A3SS&ES, 668 alternative 3’splice site (A3SS), 188 mutually exclusive 5’UTRs (5pMXE), and 182 mutually exclusive 3’UTRs (3pMXE). We can know that A5SS, A3SS, ES, and cassette exon are the RBM24-regulated major AS events. These findings imply that RBM24 modulates global ASEs in CAL27 cells with a biased regulation at A5SS. Principal component analysis showed significant differences in RASEs between the RBM24 overexpression group and the control group (Fig. 3B). We further analyzed the correlation between samples using heatmap analysis based on RNA-seq data, which revealed high consistency and comparability of RBM24-regulated transcripts in both groups (Fig. 3C). Using the GO database, we analyzed the RASG related to RBM24 and found that their main functions focused on cellular responses to DNA damage stimuli, DNA repair, viral processes, cell division, cell cycle, autophagy, RNA splicing, translation, transcription-coupled nucleotide excision repair, protein translocation, and other biological processes related to DNA damage repair and RNA metabolism (Fig. 3D).
RBM24 regulates multiple RASE. (A) Alternative splicing event regulated by RBM24. (B) PCA based on PSI value of all different expression levels of RBM24 regulated RASE. (C) Hierarchical clustering of RASE in RBM-OE and NC samples. (D) The top 10 representative pathways of the alternative splicing genes regulated by RBM24 in GO database. (E) Validation of gene expression of RASE using RNA-seq and RT-qPCR. Error bars represent mean ± SEM.*** P-value < 0.001,** P-value < 0.01.
We hypothesized that RBM24 could influence the progression of OSCC by regulating AS events in downstream target genes. To verify whether RBM24 overexpression affects the expression of these RASGs, we arranged the RASGs in ascending order of P-value and screened out genes that undergo significant ASEs, such as MAGEA4, CBX4, DEPDC1, EZH2, EGFR, and HMGA2. We used qPCR to validate the above genes, and we found that the rate of ASEs was increased for EZH2 and EGFR and decreased for DEPDC1 in the RBM24-OE group, which was consistent with RNA-seq analysis (Fig. 3E). These genes have been shown to be associated with apoptosis and migration potential of OSCC cells. Collectively, these data indicate that the number of splicing events significantly exceeds the number of differentially expressed genes, suggesting that RBM24 may function in CAL27 mainly by regulating AS of pre-mRNA and that A5SS is the main splicing event regulated by RBM24.
RBM24 binds to mRNAs associated with OSCC in CAL27 cells
Classified as an RBP, RBM24 can regulate post-transcriptional events by binding multiple target RNAs. Using the iRIP-seq method, we further constructed a transcriptome-binding profile of RBM24 to elucidate the mechanism of RBM24-RNA interactions in CAL27 cells. Immunoprecipitation was performed using RBM24 with flag tags, and two independent iRIP replicates were performed. Flag-RBM24 protein was identified through western blotting of both IP samples but not in the IgG control (Fig. 4A). After removing adaptor self-links and poor-quality bases and reads, we carried out paired-end sequencing on the IP and input group cDNA libraries using the Illumina Novaseq 6000 sequencing platform to get high-quality transcriptome data. The two IP samples were highly associated and statistically different from the input group, according to a correlation analysis of the mapped reads of each gene in the four samples at the whole transcriptome level (Fig. 4B). These results indicate that iRIP-seq experiments are reliable and that RBM24 binds RNA with good specificity. Mapped to the reference genomic region, the distribution of reads in the IP group was enriched in the intergenic region relative to the input group (Fig. 4C). This implies that RBM24 prefers to bind non-coding RNAs. Peak calling using the ABlife and piranha methods to obtain binding peaks for RBM24 and motif analysis of the peaks showed that RBM24 preferentially binds AU-enriched regions, consistent with earlier findings (Fig. 4D). The RBM24 binding peaks obtained from both experiments were arranged in descending order according to the number of reads covered by the vertices of the binding peaks, and the results are shown in the accompanying figure, with PTPRD, NEAT1, and NPM1 appearing in both experiments and warranting attention. In addition, SMG1, which appeared in IP1, and SQSTM1, which appeared in IP2, were associated with OSCC. To further confirm the binding of RBM24 to target genes, we employed sashimi plots for visual analysis of peak-associated genes. (Fig. 4E and F, left panel) The results across NEAT1 and NPM1 showed consistent results for both replicates, and the distribution of reads in the IP group was enriched in the intergenic region relative to the input group. In addition, we verified that RBM24 directly interacted with NEAT1 and NPM1 by RIP-PCR. Compared with the input group, mRNA NEAT1 and NPM1 were significantly enriched in the IP group, which was consistent with the expected results (Fig. 4E and F, right panel). Taken as a whole, we hypothesized that in CAL27 cells, RBM24 could closely interact with mRNAs and LncRNAs. These findings helped us construct the RBM24-binding RNA profiles in CAL27 cells.
Analysis of the targets bound by RBM24 in iRIP-seq. (A) Western blot experiment to validate IP efficiency. (B) Heat map showing two immunoprecipitated samples clustered (C) Reads distribution across reference genome. (D) Motif enrichment of RBM24-bound peaks by HOMER (E) RBM24 binding peak genes of NPM1. (F) RBM24 binding peak genes of NEAT1. Error bars represent mean ± SEM.*** P-value < 0.001.
RBM24 binds to mRNA and regulates its AS
To investigate whether RBM24 regulates AS events through binding mRNA, 1898 genes corresponding to the 2900 significantly different RASEs (p value < 0.05) obtained from RNA-seq data were analyzed, overlapping with 1285 RBM24-binding genes from the iRIP-seq data, and we obtained 266 overlapping genes (Fig. 5A). The above evidence shows a significant association between RBM24-binding mRNA and its regulation of AS events. In the statistical list of 266 genes with variable splicing events, we found plausible variable splicing events for tumor-associated genes ENOSF1, SQSTM1, and KDM2A based on AS events with significant changes in ratio values between experimental and control groups. We show the RNA-seq peaks and the iRIP-seq of associated genes, and we further validated the potential binding of RBM24 to the above genes using RIP-PCR (Fig. 5B and C, Figure S2 and S3). The results indicate that the RBM24 protein directly binds ENOSF pre-mRNAs to regulate their A5SS. However, in the validation experiments, KDM2A was significantly enriched in IP group, which means that RBM24 can directly bind the mRNA of KDM2A. But KDM2A A5SS was enriched in the control group, showing a contrast trend to our expectations. In the RBM24-OE group, we found that SQSTM1 only enriched in IP2, and the enrichment patterns of SQSTM1 A5SS were not consistent with the RNA-seq date. This result could be caused by experimental error or due to the fact that RBM24 did not specifically bind to SQSTM1. The above results suggest that the specific mechanism of SQSTM1 and KDM2A binding to RBM24 has not been elucidated and that more research is needed.
Comprehensive analysis of downstream targets and alternative splicing genes regulated by RBM24. (A) The overlapped genes between RASGs and peak genes from the two biological replicates. (B) RBM24 regulates alternative splicing of ENOSF1. Error bars represent mean ± SEM. * * * P-value < 0.001, * P-value < 0.05. (C) RBM24 binding peak genes of ENOSF1. Error bars represent mean ± SEM. * * * P-value < 0.001.
Discussion
Recent studies have shown that RBPs regulate cell differentiation and survival in different tissues by regulating the transcription of downstream target RNAs28. The HPA (Human Protein Atlas) database revealed that RBM24 mRNA is highly expressed in skeletal muscle, cardiac muscle, and tongue tissues but has low or no expression in other tissues. RBM24 expression varies significantly in different tissues, which may imply its potential physiological functions. RBM24 has been linked to malignant transformation in several kinds of tumors. Furthermore, in different tumor types, RBM24 may regulate different target genes in an environment- or context-dependent manner, exerting anti-tumor or pro-tumor activity29. The TCGA database showed low expression of RBM24 in OSCC tissues, which is consistent with our previous experimental results. We speculate that RBM24 may be closely associated with cell survival in OSCC.
In this study, we found that RBM24 promotes apoptosis and clarified the function of RBM24 in OSCC. In addition, by using RNA-seq and iRIP-seq, this research for the first time determined the interaction profile of RBM24 with RNAs in CAL27 cells. Through high-throughput sequencing, we discovered 188 up-regulated genes against 133 down-regulated genes. More importantly, functional enrichment analysis was carried out to assess these DEGs, and we found that the up-regulated genes were mainly enriched in apoptotic processes, which was consistent with our functional assay results. The results suggest that RBM24 may exert antitumor effects by promoting apoptosis in tumor cells.
By evaluating transcriptome data obtained by high-throughput sequencing, we found that RBM24 affects 2900 ASEs after overexpression in CAL27 cells, with significantly more splicing events than the number of differential genes, suggesting that RBM24 may function in CAL27 mainly by regulating AS of pre-mRNA. GO annotation of functional enrichment analysis showed that these ASEs affect DNA damage repair and RNA metabolism-related biological processes. Genomic instability is a potential hallmark of cancer, and defective DNA damage is one of the main causes of genomic instability30. Meanwhile, DNA damage can affect tumor progression by regulating the expression or subcellular localization of splicing factors, and AS of genes associated with the DNA damage response, in accordance with our findings31. At the same time, we verified that significant differential ASEs occurred in the pre-mRNA of EZH2, DEPDC1, and EGFR. Numerous studies have demonstrated that EZH2 is overexpressed in OSCC and that knockdown of EZH2 inhibits migration and metastasis of oral squamous cells32. DEPDC1 is also overexpressed in OSCC and silencing DEPDC1 significantly suppresses the growth of OSCC by inhibiting cell proliferation and inducing apoptosis in vivo33. Another study showed that the main mechanism by which DEPDC1 promotes OSCC progression may be to promote aerobic glycolysis, migration, and invasion of oral carcinoma through the WNT/β-catenin pathway34. As an important target for cancer therapy, overactivation of EGFR is associated with key processes of tumor growth and progression35. Overexpression or carriage of mutations in EGFR can be observed in 80–90% of head and neck cancer patients and is associated with poorer overall survival in clinical practice36,37. Furthermore, it was shown that the expression of EGFRvA, an EGFR heterodimer generated by AS, correlates more significantly with glioma grade and poor prognosis in glioma patients compared to EGFR expression38,39. These studies contribute to unraveling the physiological mechanisms of OSCC tumorigenesis and cancer progression.
Furthermore, by using iRIP-seq, we clarify the map of RBM24-RNA interactions in CAL27 cells. The iRIP-seq data showed that RBM24 binding sites in CAL27 cells are enriched in the intergenic region. The intergenic region contains many potentially important DNA sequences that generate a variety of non-coding RNAs that play important roles in regulating gene expression and epigenetic regulation in specific tissues and cells. Further clarification of the binding sites of RBM24 in the intergenic region will provide new clues to the specific mechanisms of RBM24 regulation in OSCC40. Furthermore, we verified by iRIP-PCR that NPM1 and LncRNA-NEAT1 are the targets of the direct action of RBM24. NPM1 is an independent marker of OSCC progression and prognosis, and acetylated NPM1 enhances its oncogenic potential by regulating genes involved in cell survival and proliferation during carcinogenesis38,39. Furthermore, by using iRIP-seq, we clarify the map of RBM24-RNA interactions in CAL27 cells. iRIP-seq data showed that RBM24-related RNA sequencing sequences were enriched in the intergenic region. Notably, RBM24 binds non-coding RNAs in addition to mRNA, suggesting that RBM24 is also involved in non-coding processes. It has been reported that many lncRNAs can bind to RBPs to exert transcriptional regulation. Upregulation of NEAT1 expression plays an oncogenic role in most solid tumors41. In OSCC, NEAT1 downregulation reduced the survival of cancer patients, even when patients had received radiotherapy and chemotherapy42. In addition, NEAT1 can regulate OSCC progression by activating VEGF-A and Notch signaling pathways to promote cell proliferation and EMT in vitro43. These results suggest that RBM24 can regulate OSCC cancer progression by modulating ASEs of coding and non-coding RNAs. We need to further explore the specific mechanisms by which RBM24 interacts with the above RNAs to provide new possible targets for the clinical treatment of OSCC.
Finally, we overlaid the RBM24 binding peaks with genes corresponding to significantly different AS events, yielding 266 genes that are thought to be directly bound and regulated by RBM24. By RIP-PCR, we verified that RBM24 directly binds to the pre-mRNA of ENOSF1 to regulate splicing events. ENOSF1β (rTSβ) is a shear isoform of human ENOSF1, and transient knockdown of ENOSF1 in zebrafish promotes apoptosis, suggesting that the level of ENOSF1 expression has an impact on cancer therapy44. Another study showed that ENOSF1β overexpression or phosphorylation is a marker of resistance to the drug fluorouracil (5-FU) in human colorectal cancer45,46. RBM24 overexpression increased apoptosis in 5-FU or cisplatin-treated CRC cells compared to 5-FU or cisplatin-treated cells alone16. We hypothesized that in CAL27 cells, RBM24 could mediate cancer cell resistance by binding to the mRNA of ENOSF1 and producing a shear isoform. Although our experiments demonstrated that RBM24 can bind and regulate the ASEs of ENOSF1, the specific mechanism involved has not been elucidated and requires further study. In summary, our research offers novel perspectives on the clinical diagnosis and therapy for OSCC, and targeting the RBM24-mediated dysregulated splicing events in cancer progression may be a novel therapeutic approach for treating OSCC patients.
In the present study, we discovered that RBM24 exerts anti-tumor effects by promoting apoptosis in CAL27 cells. Furthermore, using RNA-seq and iRIP-seq experiments, we have firstly found that RBM24 prioritizes binding cancer-related genes and regulating ASEs of target genes in CAL27 cells. We hypothesize that RBM24 can control AS processes by binding to specific regions of target genes to generate different protein isoforms and thus affect the proliferation, migration, and apoptosis of OSCC. In conclusion, our work paves the way for finding novel biological functions in OSCC development and provides new insights into targeted therapy for OSCC.
Data availability
The iRIP-seq and RNA-seq data have been uploaded to the GEO database with the accession number GSE283216 and GSE283217.
Abbreviations
- AS:
-
Alternative splicing
- ASEs:
-
Alternative splicing events
- BC:
-
Bladder cancer
- CRC:
-
Colorectal cancer
- eIF4E:
-
Eukaryotic initiation factor 4E
- DEGs:
-
Differentially expressed genes
- GO:
-
The Gene Ontology
- HPA:
-
Human Protein Atlas
- HPV:
-
Human papillomavirus
- ncRNA:
-
Non-coding RNA
- OE:
-
Overexpression
- OSCC:
-
Oral squamous cell carcinoma
- RASEs:
-
Regulatory alternative splicing events
- RBPs:
-
RNA binding proteins
- RBM24:
-
RNA binding motif protein 24
- 3′-UTR:
-
3′-Untranslated region
References
Zhang, L. et al. Long non-coding RNAs in Oral squamous cell carcinoma: Biologic function, mechanisms and clinical implications. Mol. Cancer 18, 102 (2019).
Gandini, S. et al. Tobacco smoking and cancer: a meta-analysis. Int. J. Cancer 122, 155–164 (2008).
Lee, J., Taneja, V. & Vassallo, R. Cigarette smoking and inflammation: Cellular and molecular mechanisms. J. Dent. Res. 91, 142–149 (2012).
Adil, N. et al. Evaluation of cytotoxicity of areca nut and its commercial products on normal human gingival fibroblast and oral squamous cell carcinoma cell lines. J. Hazard Mater 403, 123872 (2021).
Miranda-Filho, A. & Bray, F. Global patterns and trends in cancers of the lip, tongue and mouth. Oral Oncol. 102, 104551 (2020).
Zhang, L.-W. et al. Incidence and mortality trends in oral and oropharyngeal cancers in China, 2005–2013. Cancer Epidemiol. 57, 120–126 (2018).
McClure, J. J. & Palanisamy, V. Muscle-specific FXR1 isoforms in squamous cell cancer. Trends Cancer 5, 82–84 (2019).
Wu, K., Chang, F., Li, W., Su, T. & Lei, D. Role of IGF2BPs in head and neck squamous cell carcinoma. Front. Oncol. 12, 1003808 (2022).
Grifone, R. et al. The RNA-binding protein Rbm24 is transiently expressed in myoblasts and is required for myogenic differentiation during vertebrate development. Mech. Dev. 134, 1–15 (2014).
Lu, S.H.-A. et al. Alternative splicing mediated by RNA-binding protein RBM24 facilitates cardiac myofibrillogenesis in a differentiation stage-specific manner. Circ. Res. 130, 112–129 (2022).
Zhang, M. et al. Rbm24 modulates adult skeletal muscle regeneration via regulation of alternative splicing. Theranostics 10, 11159–11177 (2020).
Zheng, L. et al. Rbm24 regulates inner-ear-specific alternative splicing and is essential for maintaining auditory and motor coordination. RNA Biol. 18, 468–480 (2021).
Liu, J., Kong, X., Zhang, M., Yang, X. & Xu, X. RNA binding protein 24 deletion disrupts global alternative splicing and causes dilated cardiomyopathy. Protein Cell 10, 405–416 (2019).
Yao, D., Wang, X., Liu, J. & Xu, X. Q. Rbm24 modulates neuronal RNA splicing to restrict cognitive dysfunction. Int. J. Biol. Macromol. 276, 133853 (2024).
Zhang, M. et al. Rbm24, a target of p53, is necessary for proper expression of p53 and heart development. Cell Death Differ. 25, 1118–1130 (2018).
Xia, R. M., Liu, T., Li, W. G. & Xu, X. Q. RNA-binding protein RBM24 represses colorectal tumourigenesis by stabilising PTEN mRNA. Clin. Transl. Med. 11, e383 (2021).
Yin, Y.-W. et al. RBM24 exacerbates bladder cancer progression by forming a Runx1t1/TCF4/miR-625-5p feedback loop. Exp. Mol. Med. 53, 933–946 (2021).
Hua, W.-F. et al. RBM24 suppresses cancer progression by upregulating miR-25 to target MALAT1 in nasopharyngeal carcinoma. Cell Death Dis. 7, e2352 (2016).
Jin, Y. & Qin, X. Comprehensive analysis of the roles and prognostic value of RNA-binding proteins in head and neck squamous cell carcinoma. DNA Cell Biol. 39, 1789–1798 (2020).
Ke, J. et al. PARP1-RNA interaction analysis: PARP1 regulates the expression of extracellular matrix-related genes in HK-2 renal proximal tubular epithelial cells. FEBS Lett. 595, 1375–1387 (2021).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 12, 357–360 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Jin, L. et al. Transcriptome analysis reveals the complexity of alternative splicing regulation in the fungus Verticillium dahliae. BMC Genomics 18, 130 (2017).
Xueqing, H. et al. IGF2BP3 may contributes to lung tumorigenesis by regulating the alternative splicing of PKM. Front. Bioeng. Biotechnol. 8, 679 (2020).
Wang, N. et al. RNA-binding protein ENO1 promotes the tumor progression of gastric cancer by binding to and regulating gastric cancer-related genes. J. Gastrointest. Oncol. 14, 585–598 (2023).
Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 38, 576–589 (2010).
Xie, C. et al. KOBAS 2.0: A web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 39, W316-322 (2011).
Weiße, J. et al. RNA-binding proteins as regulators of migration, invasion and metastasis in oral squamous cell carcinoma. Int. J. Mol. Sci. 21, 6835 (2020).
Shi, D.-L. RBM24 in the post-transcriptional regulation of cancer progression: Anti-tumor or pro-tumor activity?. Cancers 14, 1843 (2022).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Shkreta, L. & Chabot, B. The RNA Splicing Response to DNA Damage. Biomolecules 5, 2935–2977 (2015).
Zhao, L. et al. Role of EZH2 in oral squamous cell carcinoma carcinogenesis. Gene 537, 197–202 (2014).
Guo, J. et al. NNK-mediated upregulation of DEPDC1 stimulates the progression of oral squamous cell carcinoma by inhibiting CYP27B1 expression. Am. J. Cancer Res. 10, 1745–1760 (2020).
Huang, G. et al. Glycolysis-related gene analyses indicate that DEPDC1 promotes the malignant progression of oral squamous cell carcinoma via the WNT/β-catenin signaling pathway. Int. J. Mol. Sci. 24, 1992 (2023).
Siena, S., Sartore-Bianchi, A., Di Nicolantonio, F., Balfour, J. & Bardelli, A. Biomarkers predicting clinical outcome of epidermal growth factor receptor-targeted therapy in metastatic colorectal cancer. J. Natl. Cancer Inst. 101, 1308–1324 (2009).
Johnson, D. E. et al. Head and neck squamous cell carcinoma. Nat. Rev. Dis. Primers 6, 92 (2020).
Kalyankrishna, S. & Grandis, J. R. Epidermal growth factor receptor biology in head and neck cancer. J. Clin. Oncol. 24, 2666–2672 (2006).
Shandilya, J. et al. Acetylated NPM1 localizes in the nucleoplasm and regulates transcriptional activation of genes implicated in oral cancer manifestation. Mol. Cell Biol. 29, 5115–5127 (2009).
Peng, H.-H. et al. Upregulated NPM1 is an independent biomarker to predict progression and prognosis of oral squamous cell carcinomas in Taiwan. Head Neck 42, 5–13 (2020).
Her, Y. R. et al. Genome-wide chromatin occupancy of BRDT and gene expression analysis suggest transcriptional partners and specific epigenetic landscapes that regulate gene expression during spermatogenesis. Mol. Reprod. Dev. 88, 141–157 (2021).
Yu, X., Li, Z., Zheng, H., Chan, M. T. V. & Wu, W. K. K. NEAT1: A novel cancer-related long non-coding RNA. Cell Prolif. 50, e12329 (2017).
Lin, N.-C. et al. The relation between NEAT1 expression level and survival rate in patients with oral squamous cell carcinoma. J. Dent. Sci. 17, 361–367 (2022).
He, K., Zhu, Z.-B., Shu, R. & Hong, A. LncRNA NEAT1 mediates progression of oral squamous cell carcinoma via VEGF-A and Notch signaling pathway. World J. Surg. Oncol. 18, 261 (2020).
Finckbeiner, S. et al. Transient knockdown and overexpression reveal a developmental role for the zebrafish enosf1b gene. Cell Biosci. 1, 32 (2011).
Dolnick, R. et al. Enhancement of 5-fluorouracil sensitivity by an rTS signaling mimic in H630 colon cancer cells. Cancer Res. 65, 5917–5924 (2005).
Lin, Y.-L. & Chow, K.-C. rTSbeta as a novel 5-fluorouracil resistance marker of colorectal cancer: A preliminary study. Ann. Acad. Med. Singap. 39, 107–111 (2010).
Funding
This work was mainly supported by a grant from the National Natural Science Foundation of China (Grant numbers 81901039) and Henan Provincial Medical Science and Technology Tackling Project (LHGJ20220362).
Author information
Authors and Affiliations
Contributions
Z.G and J.S designed the study. Z.G and J. S and S. W and S. P performed the experiments. J. S and S.W and T.G analyzed the data and wrote the manuscript. All authors discussed the data and refined the manuscript. The work reported in the paper has been performed by the authors, unless clearly specified in the text.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Zhengzhou University and was performed according to the institutional guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, J., Wang, S., Peng, S. et al. RBM24 regulates apoptosis rates by modulating global transcriptome profile in CAL27 cells. Sci Rep 15, 12069 (2025). https://doi.org/10.1038/s41598-025-96932-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-96932-5