Abstract
The present study demonstrates a treatment approach that utilizes flower-shaped tin oxide nanoparticles for the removal of cadmium from synthetic wastewater by adsorption. Tin oxide nanoparticles were synthesized using a simple and eco-friendly method and employed for the removal of cadmium ions from wastewater. The nanoflowers were successfully characterized using Fourier Transform Infrared Spectroscopy, X-Ray Diffraction Analysis, Scanning Electron Microscope and Brunauer-Emmett-Teller (BET) surface area analysis. The batch adsorption process was optimized by response surface methods to investigate the influential parameters of the adsorption process. Optimal removal efficacy was achieved at a pH value of 9.0, a mixing time of 20 min, and an adsorbent amount of 15 mg. The results indicated that the adsorbent achieved a maximum removal efficiency of 99.14 ± 0.10% for Cd(II)ions in domestic wastewater. The adsorption equilibrium process was elucidated by Langmuir, Freundlich, Sips and Toth isotherm models using nonlinear regression. In addition, error functions such as Chi-square (X2, Average Relative Error (ARE), Root Mean Squared Error (RMSE) and HYBRID were used to test the validity of the nonlinear models. The results indicated that the Sips model, with an R2 value of 0.9894, accurately matched the experimental data and adsorption capacity of 57.12 mg g− 1 for Cd(II) was calculated respectively. The sorption kinetics were well characterized by a pseudo-second-order kinetic model. The results demonstrated that adsorption of cadmium onto the surface of SnO2 nanoparticles is influenced by both monolayer adsorption and multi-site interactions. These findings indicate that SnO2 nanoparticles are suitable for removing cadmium from aqueous solutions in batch processes.
Similar content being viewed by others
Introduction
Environmental pollution is an urgent concern that requires swift intervention. The environment has been extensively polluted by the vast discharge of various harmful inorganic and organic compounds resulting from contemporary industrial activity, leading to serious contamination of air, soil, and water1. Various industries release waste containing toxic metals including lead (Pb), cadmium (Cd), mercury (Hg), tin (Sn), chromium (Cr), copper (Cu), zinc (Zn), nickel (Ni), cobalt (Co), arsenic (As). Release of these metals occurs through activities such as mining, metalliferous operations, shipbuilding, energy production, fuel refining, synthetic fertilizer and pesticide manufacturing, steel production, leather processing, photography, and electronic manufacturing. Additionally, anthropogenic factors contribute to the discharge of heavy metals into the environment2.
Cadmium is a pliable, lustrous metal that is found naturally in the earth’s crust, as well as in the air and water. Cadmium has historically been utilized and continues to be employed in various human activities worldwide, albeit with significant regional variation. These consist of its application in the manufacturing of rechargeable batteries and dyes, as well as its utilization as coating and plating material, a stabilizer for plastics, and an electrical conductor (often in combination with other metals)3. It is a pervasive heavy metal in the environment that is extremely poisonous, causing detrimental effects on various human systems such as the kidneys, skeletal system, respiratory system, and reproductive system4. Moreover, cadmium is classified as a potential carcinogen, with studies associating its exposure to an increased risks of prostate and breast cancers5,6. Cadmium can be transported from soils to plants and eventually gets into the human food chain7,8. As to the World Health Organization (WHO) recommendation value of 2017, the allowable level of cadmium in drinking water is 0.003 ppm9. The elevated concentrations of cadmium reported in many natural resources including drinking water pose a hazardous danger to all living organisms. Furthermore, secondary contamination may occur due to the altered environmental factors in soil and water sources, causing the release of previously trapped cadmium in different chemical forms10. Given the importance of reducing cadmium contamination for drinking water and food safety, efforts are needed to prevent leachate from contaminated sites, reduce the use of cadmium containing fertilizers, and to treat contaminated water bodies.
The treatment of water sources commonly involves the utilization of adsorption, photocatalytic oxidation, coagulation-flocculation, separation processing, bioremediation, solvent extraction, reverse osmosis, and precipitation methods11,12,13. Adsorption, where ions or molecules attached to a solid surface, has been recognized to be more favorable than other approaches owing to its simplicity, potential for regeneration, affordability, and ability to be employed on industrial-scale processes. Adsorbents are available in several forms for adsorption applications, such as polymers, waste products, nano-scale materials, biological substances, graphene-based materials, silica, activated carbon, and zeolites. The adsorbent materials should possess a substantial surface area along with porosity to facilitate physio-chemical interactions, as well as complimentary functional groups to facilitate adsorption processes14.
Nanomaterials possess remarkable surface areas and exhibit unique features in several disciplines such as magnetism, electricity, optics, mechanics, and catalysis. Size, shape, synthesis conditions, and functionalization of these features can be tailored by meticulous control, utilizing different synthesis methods such as top-down and bottom-up approaches15. Numerous nanomaterials of various shapes and characteristics have been utilized in many studies for the removal of both inorganic and organic contaminants from environmental samples. Specific types of adsorption materials used include silica-based nanoparticles, clay-based nanomaterials, metal ferrites, carbon-based nanomaterials, and metal oxides16,17,18,19,20. As one of metal oxides, tin oxide distinguishes itself among other types of nanomaterials with its unique properties of porous structure, superior bandgap, thermal stability, cost-effectiveness, biocompatibility, and plentiful availability, exhibiting p-type conductivity, possesses a high theoretical specific capacity and significant hole mobility. The high capacity of tin oxide to interact with substances enables it to both detect and remove a broad range of compounds in complex matrices21. Numerous studies in the literature also provide evidence that tin oxide and functionalized nanomaterials based on tin oxide can be utilized to remove various contaminants from diverse environmental samples22,23,24.
This study introduces a novel method for cadmium removal from domestic wastewater through adsorption utilizing SnO2 nanoflowers, employing a multivariate optimization approach and utilizing nonlinear kinetic and isotherm models. In this study, flower-shaped tin oxide nanomaterial was obtained via hydrothermal synthesis and utilized for the first time for the removal of cadmium from domestic wastewater. Response surface methodology (RSM) and Box-Behnken design (BBD) were used to model and optimize the individual and combined effects of independent factors on adsorption efficacy, while minimizing the number of experimental runs. The factors including mixing time, pH of buffer solution, and adsorbent amount were optimized to achieve maximum percent removal of cadmium. The adsorption isotherm models including two-parameter (Langmuir, Freundlich, Temkin and Dubinin-Radushkevich) and three-parameter (Sips and Toth) models were evaluated using nonlinear regression with the help of Python software, in order to determine the model with the best simulation of the experimental data. Furthermore, the adsorption kinetics were studied using pseudo-first-order, pseudo-second-order and Weber-Morris intra-particle diffusion models. The performance of isotherm and kinetic models were investigated using coefficient of determination (R2 and four different error functions.
Materials and methods
Chemicals, standards, samples
The Cd(II) stock solution utilized for the studies was prepared from cadmium sulfate (CdSO4.3H2O, 98%) salt procured from Riedel-de-Haën (Germany). The solutions were prepared with deionized water acquired from an Elga Flex 3 (High Wycombe, England) Water Purification System (resistance of 18.2 MΩ.cm). Tin chloride (SnCl2·2H2O, 98–102%) and sodium hydroxide (NaOH) were purchased from Merck (Darmstadt, Germany). Trisodium citrate dihydrate (Na₃C₆H₅O₇.2H2O, 99%) was acquired from Isolab (Wertheim, Germany).
The isothermal and kinetic parameters of cadmium adsorption onto SnO2 nanoflowers were investigated using synthetic domestic wastewater prepared according to the procedure described by Turan et al. in their previously published research25. The stock solution was prepared at 1000.196 mg/kg using CdSO4.3 H₂O in deionized water. Dilutions were made with domestic wastewater (%90 v/v) in the range of 15–60 mg/L for equilibrium studies, while for kinetic experiments, adsorption was evaluated at 30 mg/L of Cd(II).
Instrumentation
A flame atomic absorption spectrometry instrument (FAAS, ATI UNICAM 929 AA, UNICAM, England) was utilized for analytical purposes. The instrument was equipped with a burner head, a cadmium hollow cathode lamp (Photron) with a working current of 8.0 mA, a spectral bandwidth of 0.50 nm, and an analytical line at 228.8 nm. Additionally, a deuterium lamp was employed to provide background correction. The primary objective of using the instrument was to assess and measure the pollutant levels before and after the adsorption-based removal process. A stoichiometric flame was generated utilizing an acetylene/air mixture for the atomization process. The acetylene and air were pressurized to 1.0 and 5.0 bar, respectively, using pressure gauges.
Characterization of flower-shaped nanoparticles was conducted using Fourier Transform Infrared (FTIR) Spectrum, X-Ray Diffraction (XRD) Analysis, Brunauer-Emmett-Teller (BET) Surface Area Analysis and Scanning Electron Microscope (SEM) images. The FTIR spectrum of SnO2 nanoparticles were acquired in the wavelength range between 500 and 4000 cm− 1 using a Bruker Tensor 27 model Attenuated Total Reflection (ATR) spectrometer with a wavenumber accuracy of ± 0.01 cm− 1. XRD analysis was carried out using PANanalytical X’Pert Pro XRD system equipped with a Cu-Kα radiation source. XRD spectrum was obtained in a scanning angle 2θ adjusted between 2° to 90° with a scanning rate of 2°/min at 40 mA and 45 kV. A Quantachrome Quadrosorb SI BET was used to analyze the specific surface area of metal oxide nanoflowers. The synthesized nanoflowers were degassed at 393 K for 2 h prior to analysis. SEM images were taken using a Zeiss EVO LS10 model SEM in beam mode at 20 kV. The nanoflowers were coated by gold-palladium under argon plasma using a Quorum SC7620 Sputter Coater.
Synthesis of Tin oxide nanoparticles (SnO2)
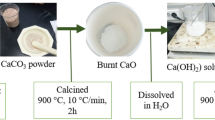
The synthesis of flower-shaped tin oxide nanoparticles was achieved according to the method by Jin et al. with a small modification26 .Initially, a quantity of 0.358 g of SnCl2·2H2O was dissolved in a mixture of 20 mL of glycol and water (with a ratio of 1:1, v/v), applying vigorous stirring. Following that, a quantity of 2.406 g of sodium citrate (Na3C6H5O7·2H2O) was dissolved in the SnCl2 solution and the mixture was then agitated using a magnetic stirrer until a uniform solution was obtained. Furthermore, NaOH was gradually introduced into the uniform solution dropwise until the pH reached a value of 10. After approximately one hour of stirring, the solution was transferred to a 50 mL autoclave made of stainless steel and lined with Teflon. The autoclave was then sealed and heated in a laboratory oven at a temperature of 433.15 K for a duration of 12 h.
Determination of point of zero charge
The solid addition approach was employed for calculating the point of zero charge (pHPZC) of SnO2 nanoflowers. The method was carried out by preparing a set of Erlenmeyer flasks with 25 mL of 0.1 M KNO3. The initial pH (pH0) of the solutions was altered to range from 2.0 to 12 through the addition of 0.1 N HCl or NaOH solutions. Each solution consisted of 25 mg of SnO2 nanoparticles and allowed to equilibrate at room temperature for 24 h. Subsequent to this period, the final pH (pHf) was recorded, and the pHPZC was established by graphing the difference between the initial and final pH (ΔpH) values in relation to the initial pH values.
Optimization of influential parameters in batch adsorption
The effects of experimental variables on the adsorption capacity of nanoflowers were examined by response surface methodology. A Box-Behnken design (BBD) was utilized to optimize three variables: agitation period, buffer type, and nanoflower amount. The experimental data, obtained from the averages of three replicate analyses, was evaluated using Design Expert software (trial version 13, stat-ease).
BBD was conducted with three factors and three levels (-1, 0, and + 1) for the input variables: SnO2 NP amount (15–35 mg), agitation duration (5–35 min), and pH of buffer (5.0–9.0). The design comprised 29 tests with 5 replicates at the center point. The response value for each run was determined by averaging the results of three technical replicates. The system’s behavior is described by the equation presented in reference27.
In this equation, \(\:y\) represents the response (dependent variable), \(\:\epsilon\:\) is the residual connected with the experiments or random error, \(\:{\beta\:}_{0}\), \(\:{\beta\:}_{i}\) and \(\:{\beta\:}_{ij\:}\)refer to the constant term, the linear effect, and the coefficient of the interaction parameters, respectively27. Table 1 presents the factors, their corresponding levels and the observed responses in the design.
Analysis of variance (ANOVA) test was applied to evaluate the adequacy and significance of the model. R-squared values including the coefficient of determination (R2 and the adjusted-R2 were used to examine the model adequacy. The probability values (p-value) below 0.05 were regarded as statistically significant.
Determination of cadmium by FAAS system
The concentration of Cd in the effluents from batch removal studies has been determined following the assessment of the analytical performance of the FAAS system for Cd element. The FAAS system’s ability to measure a broad range of Cd concentrations was evaluated through the analysis of various concentrations of Cd spiked into synthetic domestic wastewater. The LOD (limit of detection) and LOQ (limit of quantification) were determined by calculating the standard deviation (SD) by employing 6 replicates of the lowest concentration in a linear calibration plot, with a signal to noise ratio of 3. The LOD and LOQ values were determined using 3xSD/m and 10xSD/m formulas, respectively. The symbol “m” represents the slope of the calibration plot, which has a determination coefficient (R2 greater than 0.99.
Linear range of concentrations of Cd in synthetic domestic wastewater varied from 0.10 to 5.0 mg/L, with a coefficient of determination (R2 of 0.9993. LOD and LOQ values were found to be 0.020 and 0.067 mg/L, respectively. LOQ values were employed to quantify the signals of undetectable samples, while the samples with saturated signals were diluted until achieving quantitative signals. The equation of y = 0.1249x + 0.0057 was utilized to determine the quantity of Cd in the effluents of the adsorption process. In this equation, y represents the absorbance of the sample signal, and x represents the Cd content (mg/L) in the sample.
Batch adsorption procedure for the removal of cadmium (II) ions
10 mL of a standard/sample solution with different concentrations were placed into an Erlenmeyer flask. 15 mg of tin oxide (SnO2) nanoparticles and a 1.0-mL solution of borax buffer with a pH of 9.0 were added to each Erlenmeyer flask and sonicated for 30 s. Then, samples were subjected to agitation in an orbital shaker at an average speed of 250 rpm for a duration of 20 min. After mixing, nanoflowers were settled down at the bottom of the tube using centrifugation at 3000 rpm for 2.0 min. Subsequently, the resulting clear upper-phase solution was transferred to the FAAS system. Figure 1 provides a comprehensive depiction of all the experimental stages.
Schematic representation of the experimental procedure for the SnO2 based removal method.
After performing the FAAS analysis, Eqs. (2) and (3) were used to compute the amounts of adsorbates collected by the adsorbent (\(\:{q}_{e}\)) and the percent removal efficiency, respectively28.
where \(\:{C}_{i}\) and \(\:{C}_{e}\) are the concentrations of cadmium ion in mg/L at the initial and equilibrium, respectively. \(\:{q}_{e}\) is the amount of cadmium adsorbed onto nanoflowers in mg/g, m is the mass of nanoflowers (g) and V is the volume of sample solution (L).
Adsorption isotherm and kinetic models, error functions
The equations of isotherm models, kinetic models and error functions used in this study are grouped in Table 2. Isotherm models including Langmuir, Freundlich, Dubinin-Radushkevich (D-R), Temkin, Sips and Toth were used to evaluate the fitness of the experimental data. Synthetic domestic water was spiked with different initial concentrations of Cd (15–60 mg/L) and batch adsorption experiments were carried out for all samples. The experimental data was obtained by the determination of effluent Cd concentration. The parameters in isotherm equations are defined in Table 2.
In order to evaluate adsorption kinetics, batch adsorption experiments were employed for different process periods (between 5.0 and 20 min) with constant initial concentration of Cd (30.0 mg/L). The adsorption period (t, min) and the adsorption capacity at time t (\(\:{q}_{t})\) were measured to study three different kinetic models such as pseudo-first-order (PFO), pseudo-second-order (PSO) and Weber-Morris (WM) kinetic models. The rate constants of models were defined as \(\:{k}_{1}\) (1/min) for PFO, \(\:{k}_{2}\) (g/min·mg) for PSO and \(\:{k}_{wm}\) (mg/g·min0.5) for WM models.
The nonlinear regression was applied to find the unknown parameters of models through trial-and-error iteration approach. Curve fitting was employed using the nonlinear functions of the models by scipy.optimize.curve_fit from Scipy library of Python. The iteration was employed to achieve the minimum value of the sum of square of the errors and, coefficient of determination (R2 and four additional error functions including Chi-square (X2, Average Relative Error (ARE), Root Mean Squared Error (RMSE) and HYBRID were also calculated to determine the model with best fit to experimental data. In the error equations, n refers to the number of data points, p is the number of parameters, \(\:{q}_{e,exp}\) and \(\:{q}_{e,calc}\) are the measured and calculated adsorption capacity values at the equilibrium.
Results and discussion
Characterization of tin oxide nanoflowers
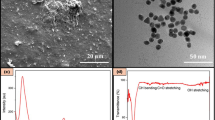
Formation of tin oxide nanoflowers was confirmed by SEM, XRD, FTIR, particle size distribution and point of zero charge (pHPZC) analysis. Figure 2a shows the FTIR spectrum of tin oxide nanoflowers between 4000 and 500 cm− 1. From Fig. 2a, it can be seen that two absorption bands appeared in the low wavenumber region at 548 cm− 1 and 576 cm− 1. These bands can be attributed to the antisymmetric vibration of O-Sn-O bridge functional groups. The weak peak at 1632 cm− 1 might be assigned to the Sn-OH bond. The broad band around 3300 cm− 1 corresponds to stretching vibration of hydroxyl groups present in the surface of nanoflowers29. The remaining weak bands at 2160 cm− 1, 1367 cm− 1, and 1245 cm− 1 may be related to the presence of adsorbed CO₂ or organic residues from the synthesis process.
(a) FT-IR, (b) XRD spectra and c-d) SEM images and e) pHpzc analysis of of tin oxide nanoflowers.
The crystal structure and phase composition of tin oxide nanoflower were confirmed by XRD pattern. As given in Fig. 2b, the diffractogram obtained at a 2θ scanning angle showed high peak intensities at the values of 26.49°, 32.38°, 37.24°, 51.64°, 54.14° and 65.69° which can be assigned to the tetragonal rutile structure of SnO2 (JCPDS: 41-1445). Additionally, XRD pattern obtained for the synthesized product in this study closely resembles the patterns observed in previous studies documented in the literature30. The average crystallite size of tin oxide nanoflowers was calculated using the Debye-Scherer equation: \(\:D=\:0.89\lambda\:/\beta\:cos\theta\:\) and found to be 72.13 nm. The average particle size measured as 78.8 nm. The surface area of the produced nanoflowers was evaluated using BET analysis, and a surface area of 31.31 m2/g was obtained.
The morphology of tin oxide structure was studied by SEM images (Fig. 2c). The nanoparticles originated in a flower-like shape and exhibited a heterogeneous disparity. The presence of this characteristic can be attributed to the extensive surface area of the produced nanoflowers. SEM pictures closely resembled the SnO2 nanoparticles synthesized in a previous study31.
The adsorbent’s surface charge might be affected by the pH level, as the pH at the point of zero charge plays a crucial role in determining adsorption behavior32. The pHpzc of SnO₂ nanoparticles were determined utilizing the solid addition method. Figure 2d demonstrates that the pHPZC of SnO2 nanoparticles is about 5.8, indicating that the SnO₂ nanoparticles exhibit surface charge transitions from positive to negative depending on the environment’s pH. At pH values below the pHPZC (pH < 5.8), the surface of SnO2 nanoparticles is positively charged, while at pH values above the pHPZC (pH > 5.8), the surface becomes negatively charged. The negatively charged surface is more likely interact with positively charged ions such as Cd(II) cations, which explains the increased adsorption efficiency of SnO₂ nanoparticles in basic environments.
Experimental design
The Box-Behnken Design (BBD), as a Response Surface Methodology-based model, effectively optimizes the adsorption process by statistically modeling factors to maximize removal efficiency. It reduces the number of experiments, minimizing waste while maintaining data quality33. Herein, BBD was employed to examine the impact of factors that include the amount of sorbent, duration of agitation, and pH of the buffer on the adsorption efficiency. The utilization of BBD provides advantages for exploring complex response functions with relatively few experimental runs. The accurate modeling of quadratic response surfaces is achieved by systematically varying the influential parameters and focusing on the middle levels. In this study, a total of 17 experiments were performed and absorbance value measured in FAAS system was taken as the response to be minimized. The quadratic equation representing the relationship of the response as a function of independent variables is given below (Eq. 4):
In this equation, three experimental parameters, namely pH of the buffer (A), SnO2 nanoparticle amount (B), and agitation period (C) were found to have a significant impact on the response value (y, %Removal). The ANOVA table for Cd removal is given in Table 3. The ANOVA test showed that the model was highly significant, with a very low probability (p-value) of less than 0.0001. The significance of the variables was confirmed with a p-value < 0.0533. Among the individual factors, pH (A) and adsorbent amount (B) exhibited the most significant impact, both with p-values below 0.0001. Additionally, the quadratic terms (A2 and B2 were also significant, indicating non-linearity in their effects. The interaction term (AB) suggested a strong combined influence of pH and adsorbent amount on the response. Conversely, the mixing period (C) was not statistically significant (p > 0.05), implying a minimal contribution to adsorption efficiency. As seen in parity plot given in Fig. 3a, the observed data points and anticipated values of the response showed a strong agreement, as evidenced by the model’s correlation coefficient (R2 of 0.9826, adjusted-R2 value of 0.9722, and predicted R2 value of 0.9090. The higher values of R2 indicated the accuracy of the model for experimental purposes. The closeness of adjusted-R2 value to R2 value confirmed that the model was not overfitted and the predicted R2 value remained within an acceptable range. All these findings proved the reliability of the model in predicting removal percentage. The lack of fit refers to the model’s failure to accurately explain the relationship between factors and the response34. The lack of fit, which was not statistically significant (p > 0.05), suggested that the model was appropriate for the data. Additionally, the model itself was statistically significant and suitable for further tests.
(a) Comparison of experimental and predicted values using a parity plot and (b) 3D-RSP showing the effects of adsorbent amount and pH of aqueous medium on the removal of cadmium.
Figure 3b displays three-dimensional response surface plots (3D-RSP) that illustrate the primary and interaction effects of independent variables on the dependent variable, which is the response value. The response value exhibited a positive correlation with both the sorbent amount and the increasing pH of the buffer. The percent removal was only increased from %91.78 to 99.34% with the more than two-times increase of the adsorbent amount. With an increase in pH value from 5.0 to 9.0, the percent removal was improved from 69.31 to 99.14%. Consequently, the effectiveness of pollutant removal was improved by increasing the amount of sorbent and pH level of aqueous medium. At low pH values below the pHpzc point < 5.8, electrostatic repulsion occurs between the metal ions and the positively charged adsorbent surface due to the hurdle created by H3O+ ions. In contrast, electrostatic attraction strengthens as the negative charge on the adsorbent surface strengthens when the solution’s pH exceeds the pHpzc value of 5.8, hence corroborating the findings from the point of zero charge characterization35. In this study, pH values above 9.0 was not tested due to the formation of Cd(OH)2 precipitates under alkaline conditions. At pH 9.0, there was only about 10% precipitation observed and thus, this value was chosen for further experiments.
The agitation period did not show noticeable effects on the adsorption process. The quadratic model determined according to the desirability function on the model which was aimed to get maximum percent removal result (100%). Several combinations were obtained with a desirability score of 1.0 and the following parameters were chosen for achieving maximum removal percentage with minimum sorbent amount: 15 mg of SnO2 sorbent, 20 min of agitation period, and pH value of 9.0.
As a result, the high adsorption efficiency observed at optimal conditions can be attributed to the physicochemical properties of tin oxide nanoflowers, including their functionalized surface, high crystallinity, moderate surface area, and flower-like morphology. The hydroxyl-rich surface, along with a porous and heterogeneous structure, plays an important role in enhancing the adsorption capacity by providing numerous active sites and promoting favorable adsorbate-surface interactions. These properties collectively confirmed the effectiveness of the material for removal of Cd(II) ions and supported the results obtained from the experimental design and optimization study.
Adsorption isotherms
Researchers from various disciplines are motivated to gain a deeper understanding of the adsorption process due to its significant impact on industrial applications. Several empirical and theoretical investigations, including molecular dynamics and density functional theory (DFT) approaches, have been carried out to achieve this objective36. Within this assortment of studies, the adsorption isotherms and their accompanying equations provide a comprehensive and meticulous depiction of the adsorption processes. The adsorption isotherm is a mathematical model that predicts the amount of material that is adsorbed as a result of pressure (or concentration) changes, while keeping the temperature constant37. The adsorption procedure was employed to acquire equilibrium data by altering initial concentrations of pollutants. The results were then examined using nonlinear forms of mathematical isotherm models: Langmuir, Freundlich, Temkin, D-R, Sips and Toth.
Langmuir-type adsorption is classified as a monolayer phenomenon. The maximum adsorption capacity per unit mass of the adsorbent is determined in tandem with the Langmuir constant which indicates the substance’s affinity for the adsorbent. Conversely, Freundlich-type adsorption is recognized as a multi-layer phenomenon where an amount of the substance adsorbed per unit mass of adsorbent gradually increases38. The Sips model integrates the adsorption characteristics of both the Langmuir and Freundlich models. The adsorption process in Sips model is described as a combination of both monolayer and multi-layer adsorption39. On the other hand, the Toth isotherm is a modified Langmuir equation, designed to minimize the difference between experimental and predicted values for adsorption equilibrium. It is an effective model to describe the adsorption on heterogeneous surfaces across a wide range of adsorbate concentrations, from low to high levels40,41. According to the Temkin isotherm, the adsorption process is characterized by a decrease in adsorption heat as the surface coverage increases, assuming a logarithmic variation in adsorption energy. D-R isotherm considers a Gaussian distribution of adsorption sites and energies, and accounts for the energy variation across the surface of adsorbent42.
Table 4 presents the projected parameters of Langmuir, Freundlich, D-R, Temkin, Sips and Toth isotherms that have been estimated from the nonlinear curve fitting results. The experimental data and predicted \(\:{q}_{e}\) values by the model equations are illustrated in Fig. 4. The Toth model demonstrated the finest fitting accuracy, as indicated by the highest R2 value and the lowest X2, ARE, RMSE and HYBRID errors. The Langmuir model simulation is valid when the value of \(\:{t}_{o}\) is equal to 1. The value of \(\:{t}_{o}\)<1 characterizes the heterogeneity of the adsorption surface, where the interactions between adsorbent and adsorbate ions are stronger than the interactions among the adsorbed ions themselves40.
Two and three parameter isotherms obtained using the nonlinear method for the adsorption of Cd(II) onto tin oxide nanoflowers (constructed by Matplot Library of Python).
The value of \(\:{t}_{o}\) obtained from the nonlinear regression of the Toth model was 0.1945, which confirms the development of multilayer adsorption of Cd(II) onto tin oxide nanoparticles. However, considering the plateaus of the isotherms at the equilibrium, the predicted \(\:{q}_{mL}\) values in the Toth and D-R isotherm models were abnormally high. Hence, in spite of the high R2 and low error values, Toth and D-R models were unable to fit the experimental data.
In addition to Toth isotherm model, the adsorption of Cd(II) was reasonably well presented by Freundlich and Sips models with R2 values above 0.98. The Sips isotherm constant \(\:{\beta\:}_{S}\) was estimated as 0.3786, indicating that the isotherm model is approaching to Freundlich isotherm rather than Langmuir isotherm model. Sips model correctly simulated the experimental data with the comparable prediction of maximum adsorption capacity (57.1264 mg/g). The \(\:{X}^{2}\), \(\:ARE\), \(\:RMSE\) and \(\:HYBRID\) error values were obtained as 0.2517, 3.51, 0.6921 and 4.2814 respectively. Consequently, a good representation of experimental data was achieved by Sips model, which signifies the heterogeneous nature of the flower-shaped tin oxide nanoparticles.
The mean free energy (E, kJ/mol) can be estimated using the D-R constant (β) with the following equation:
When E value falls between 8 and 16 kJ/mol, the adsorption process occurs through chemisorption. Conversely, when E is less than 8 kJ/mol, the process is of a physical nature43. In this study, E value was found to be 17.8 kJ/mol, indication that the process proceeded via chemisorption.
Adsorption kinetics
The accurate estimation of the rate at which adsorption occurs, specifically the movement of the adsorbate from the solution to the binding sites of the adsorbent material, is an essential step for the efficient development of an adsorption process. The kinetic data sets were analyzed using three different kinetic models, namely the PFO model, the PSO model and WM intra particle diffusion (IPD) model. The PFO kinetic model postulates that the adsorption process adopts first-order kinetics, with the rate being exactly proportional to the difference in adsorbate uptake between equilibrium and any point in time. Conversely, the PSO model relies on a rate law of second order pertaining to the active sites of the adsorbent. Addition to these kinetic models, WM-IPD model was also evaluated in order to gain insight into the diffusion mechanism of adsorbate ions44.
Nonlinear regression was applied to experimental data using three kinetic models and calculated regression parameters are given in Table 5. Nonlinear regression did not converge when PFO kinetic model was implemented. By contrast, PSO model yielded to adequate fit of the data set with a satisfactory \(\:{R}^{2}\) value of 0.86 and low error values. As can be seen from Fig. 5a, the estimated \(\:{q}_{t}\) values were close to the experimental values, confirming the applicability of PSO model where the chemisorption is the dominant mechanism. Additionally, the linear plot of \(\:{q}_{t}\) versus \(\:{t}^{0.5}\) is presented in Fig. 5b. Herein, the line does not pass through the origin, confirming that intra-particle diffusion is not the sole rate-controlling step. The high \(\:{R}^{2}\) value indicates the involvement of an intra-particle diffusion, while positive C value suggests the contribution of the boundary layer diffusion or some other processes to the overall adsorption kinetics45,46.
PSO and WM-IPD plots for the adsorption Cd(II) onto tin oxide nanoflowers (constructed by Matplot Library of Python).
Regeneration and reusability of the adsorbent
The regeneration and recovery of consumed adsorbent represent significant challenges in the adsorption process, impacting both economic viability and environmental sustainability. The regeneration process of SnO2 nanoparticles entailed the separation of the used adsorbent within optimum conditions following each adsorption cycle, washing it with distilled water, and subsequently regenerating it through treatment with 0.1 mol L− 1 HNO3 solution. The regenerated adsorbent was reused in following cycles, and its efficacy in adsorbing Cd(II) ions was assessed. The experiments were conducted under optimal conditions with an initial Cd(II) concentration of 30 mg/L. The removal efficiencies after four cycles are illustrated in Fig. 6. The figure indicates that, throughout the four cycles, the removal efficiency for Cd(II) remained above 99%, demonstrating the effectiveness and stability of the adsorbent utilized. These results indicate that the adsorbent possesses favorable characteristics for recycling and reusability.
The reusability graph for Cd(II) ion removal with tin oxide nanoflowers. Reaction conditions: temp 298 K, concentration of Cd(II) ion (30 mg/L), pH 9.0 buffer, mixing time (20 min), shaking speed (250 rpm).
Comparison with previous studies
The adsorption capacity of SnO2 nanoparticles, as estimated by the Sips isotherm model, was assessed in relation to the maximum adsorption capacity of other adsorbents (Table 6). Evaluating the SnO2 nanoparticles employed in the present study with those reported in the literature was challenging since diverse conditions for experiments were used in these studies. While all the adsorbents listed in this table have been found to be efficient in removing heavy metals, the fundamental objective of adsorption methods in practical applications is to achieve economic viability by minimizing the amount of adsorbent used via high adsorption capacity. Table 6 demonstrates that the SnO2 nanoparticle had a high maximum adsorption capacity value of 57.12 mg/g compared to the various adsorbents utilized for removing Cd(II) ions. SnO2 nanoparticles demonstrated high efficiency in removing Cd(II) ions. High efficiency results were achieved with regard to the use of a minimal amount of adsorbent at a room temperature of 298.15 K for a duration of 20 min.
Conclusion
This study introduces an intuitive, inexpensive, and practical approach for removing cadmium from wastewater. The process involves utilizing flower-shaped SnO2 nanoparticles for the batch adsorption process. The synthesized nanoparticles exhibited the capability to remove Cd(II) ions from aqueous sample solutions, with the sorption capacity being significantly influenced by the specific buffer solution and the mass of the adsorbent. The optimal parameters for the removal of Cd(II) ions were a pH value of 9.0, a mixing time of 20 min, and an adsorbent amount of 15 mg, which were determined to be applicable for varying Cd(II) concentrations in domestic wastewater. This method offers important benefits including low-cost production of adsorbent material, operation convenience regarding rapidity and high removal efficiency. Adsorption studies were performed to achieve equilibrium using synthetic domestic wastewater. The removal efficacy of SnO2-based batch removal for different initial concentrations of cadmium ranged from 98.8 to 78.7%. The analysis of adsorption isotherms and kinetics indicated the simultaneous presence of both monolayer and multilayer adsorption. It was found that chemical sorption, which involves the sharing or exchange of electrons between metal ions and the adsorbent material, was the primary rate-limiting process facilitated by valence forces. The adsorption capacity of SnO2 nanoparticles for Cd(II) ions was determined to be 57.12 mg g− 1, aligning with the Sips isotherm model and the experimental results. Additionally, the batch removal time was significantly reduced compared to prior studies utilizing alternative nanomaterials, thus enhancing the practicality of the method. The observations conclusively demonstrate that SnO2 nanoparticles are very suitable for efficiently removing cadmium from wastewater, offering an environmentally acceptable and cost-effective alternative combined with its reusable properties. The adsorption capacity of SnO₂ nanoparticles can potentially be enhanced through the application of surface functionalization that is appropriate for the specific application. In addition, it is of the utmost importance that future research attempts to investigate the adsorption mechanism of other heavy metals onto tin oxide nanomaterial in a persistent manner. This will enable the development of viable systems for real-world applications.
Data availability
Data and materials will be available on reasonable request from the corresponding author.
References
Sagheer, U. et al. Environmental pollution and cardiovascular disease: part 2 of 2: soil, water, and other forms of pollution. JACC: Adv. 3, 100815 (2024).
Kavisri, M. et al. Adsorption isotherm, kinetics and response surface methodology optimization of cadmium (Cd) removal from aqueous solution by Chitosan biopolymers from cephalopod waste. J. Environ. Manage. 335, 117484 (2023).
Fanfani, A. et al. Cadmium in biological samples and site-specific cancer risk and mortality: A systematic review of original articles and meta-analyses. Cancer Epidemiol. 102550 https://doi.org/10.1016/J.CANEP.2024.102550 (2024).
Bracher, C. et al. Tracing the fate of phosphorus fertilizer derived cadmium in soil-fertilizer-wheat systems using enriched stable isotope labeling. Environ. Pollut. 287, 117314 (2021).
Arabkhani, P. & Asfaram, A. A novel biowaste-derived magnetic adsorbent for efficient removal of cadmium, Cobalt and strontium ions from industrial wastewater. Inorg. Chem. Commun. 174, 113956 (2025).
Al-Meer, S., Al-Saad, K., Aledamat, R., El-Shafie, A. S. & El-Azazy, M. Unveiling the Power of Surfactant-Based Carbon Dots: Ultrasensitive Detection of Cadmium in Tap and Drinking Water Samples. Processes 12(10), 2239 (2024).
Kabata-Pendias, A. Soil–plant transfer of trace elements—an environmental issue. Geoderma 122, 143–149 (2004).
McLaughlin, M. J., Smolders, E., Zhao, F. J., Grant, C. & Montalvo, D. Managing cadmium in agricultural systems. Adv. Agron. 166, 1–129 (2021).
Bhattacharyya, K., Sen, D., Banik, A. K. & Ganguly, S. Adsorptive removal of cadmium from aqueous medium-a critical review. Phys. Chem. Earth Parts A/B/C. 134, 103538 (2024).
Cang, L., Xing, J., Liu, C., Wang, Y. & Zhou, D. Effects of different water management strategies on the stability of cadmium and copper immobilization by Biochar in rice-wheat rotation system. Ecotoxicol. Environ. Saf. 202, 110887 (2020).
Pushkar, B., Sevak, P., Parab, S. & Nilkanth, N. Chromium pollution and its bioremediation mechanisms in bacteria: A review. J. Environ. Manage. 287, 112279 (2021).
Sulaiman, M. S., Ajayi, O. A. & Olakunle, M. S. Optimization of cadmium adsorption on metal organic frameworks MIL-53(Fe) using response surface methodology. Inorg. Chem. Commun. 159, 111765 (2024).
Mahvi, A. H., balarak & Bazrafshan, E. Remarkable reusability of magnetic Fe3O4-graphene oxide composite: a highly effective adsorbent for Cr(VI) ions. Int. J. Environ. Anal. Chem. 103, 3501–3521 (2023).
Pakade, V. E., Tavengwa, N. T. & Madikizela, L. M. Recent advances in hexavalent chromium removal from aqueous solutions by adsorptive methods. RSC Adv. 9, 26142–26164 (2019).
Baig, N., Kammakakam, I., Falath, W. & Kammakakam, I. Nanomaterials: a review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2, 1821–1871 (2021).
Rani, M. & Shanker, U. Highly efficient removal of endocrine disrupting pesticides by metal ferrites loaded Guar gum based green nanomaterials. J. Mol. Liq. 387, 122611 (2023).
Li, F., Yin, H., Zhu, T. & Zhuang, W. Understanding the role of manganese oxides in retaining harmful metals: insights into oxidation and adsorption mechanisms at microstructure level. Eco-Environment Health. 3(1), 89-106 (2024).
Liu, D. et al. Recent advances in MOF-derived carbon-based nanomaterials for environmental applications in adsorption and catalytic degradation. Chem. Eng. J. 427, 131503 (2022).
Abd-Elhamid, A. I., Abu Elgoud, E. M. & Aly, H. F. Adsorption of palladium from chloride aqueous solution using silica alginate nanomaterial. Int. J. Biol. Macromol. 253, 126754 (2023).
Ciğeroğlu, Z. et al. Clay-based nanomaterials and their adsorptive removal efficiency for dyes and antibiotics: A review. Mater. Today Sustain. 26, 100735 (2024).
Kader, M. A., Azmi, N. S., Kafi, A. K. M. & Alim, S. Recent advances in functionalization of copper and Tin oxide nanomaterials for application in sensing: A comparative review. Mater. Today Chem. 37, 102003 (2024).
Ebrahimian, J., Mohsennia, M. & Khayatkashani, M. Photocatalytic-degradation of organic dye and removal of heavy metal ions using synthesized SnO2 nanoparticles by Vitex agnus-castus fruit via a green route. Mater. Lett. 263, 127255 (2020).
Wang, W., Song, F., Du, C. & Su, Y. Durable and eco-friendly peroxymonosulfate activation over Cobalt/tin oxides-based heterostructures for antibiotics removal: insight to mechanism, degradation pathway. J. Colloid Interface Sci. 625, 479–492 (2022).
Mokarizadeh, H. et al. Solar-activated Tin oxide photocatalysis for efficient naphthenic acids removal and toxicity reduction in oil sands process water. J. Environ. Chem. Eng. 12(6), 114168 (2024).
Turan, N. B. et al. Feasibility studies on the effect of natural plant compounds on sludge characteristics in a batch-type aerobic reactor and N-butyryl-L Homoserine lactone. Anal. Lett. 53, 2431–2444 (2020).
Jin, W. X. et al. Synthesis of hierarchical SnO2 nanoflowers with enhanced acetic acid gas sensing properties. Appl. Surf. Sci. 353, 71–78 (2015).
Bezerra, M. A., Santelli, R. E., Oliveira, E. P., Villar, L. S. & Escaleira, L. A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76, 965–977 (2008).
Zeng, Q. et al. High adsorption capacity and super selectivity for Pb (II) by a novel adsorbent: nano Humboldtine/almandine composite prepared from natural Almandine. Chemosphere 253, 126650 (2020).
Mani, R., Vivekanandan, K. & Subiramaniyam, N. P. Photocatalytic activity of different organic dyes by using pure and Fe doped SnO 2 nanopowders catalyst under UV light irradiation. J. Mater. Sci.: Mater. Electron. 28, 13846–13852 (2017).
Preethi, T. et al. Enhanced Humidity Sensing Properties of Surfactant-Free Hydrothermally Synthesized Tin Oxide Nanoparticles. 13 (6) 06003-1-06003-5 (2021).
Wang, J., Duan, X. & Ren, Y. Efficient electrochemical degradation of Ceftazidime by Ti3 + self-doping TiO2 nanotube-based Sb–SnO2 nanoflowers as an intermediate layer on a modified PbO2 electrode. Chemosphere 356, 141853 (2024).
Singh, K., Bharose, R., Verma, S. K. & Singh, V. K. Potential of powdered activatedmustard cake for decolorising Raw sugar. J. Sci. Food Agric. 93, 157–165 (2013).
El-Shafie, A. S. & El-Azazy, M. Crosslinked chitosan–montmorillonite composite and its magnetized counterpart for the removal of basic Fuchsin from wastewater: parametric optimization using Box-Behnken design. Int. J. Biol. Macromol. 263, 130224 (2024).
Faravelli, L. Response-surface approach for reliability analysis. J. Eng. Mech. 115, 2763–2781 (1989).
Isaac, R. & Siddiqui, S. Adsorption of divalent copper from aqueous solution by magnesium chloride co-doped Cicer arietinum husk Biochar: isotherm, kinetics, thermodynamic studies and response surface methodology. Bioresour Technol. Rep. 18, 101004 (2022).
Ahmad, R., Ali, Z., Khan, A. A. & Rehman, N. U. Terbium extraction by functionalized surface: experimental and DFT approach. Adsorption 26, 117–125 (2020).
Majd, M. M., Kordzadeh-Kermani, V., Ghalandari, V., Askari, A. & Sillanpää, M. Adsorption isotherm models: A comprehensive and systematic review (2010 – 2020). Sci. Total Environ. 812, 151334 (2022).
Chung, H. K. et al. Application of Langmuir and Freundlich isotherms to predict adsorbate removal efficiency or required amount of adsorbent. J. Ind. Eng. Chem. 28, 241–246 (2015).
An, F. Q. et al. Adsorption of heavy metal ions by iminodiacetic acid functionalized D301 resin: kinetics, isotherms and thermodynamics. React. Funct. Polym. 118, 42–50 (2017).
Chen, X. et al. Isotherm models for adsorption of heavy metals from water - A review. Chemosphere 307, 135545 (2022).
Tóth, J. State equation of the solid-gas interface layers. Acta Chim. Hung. 69, 311–328 (1971).
Varnaseri, M. et al. Linear and nonlinear isotherm, kinetic, and thermodynamic behavior of Safranine T adsorption using porous geopolymer prepared from coal fly Ash wastes. Water Conserv. Sci. Eng. 9(2), 45 (2024).
Kord Mostafapour, F. et al. Removal of acid blue 113 from aqueous medium using a novel magnetic adsorbent derived from activated carbon fiber. Int. J. Environ. Anal. Chem. 104(17), https://doi.org/10.1080/03067319.2022.2130061 5732 - 5747 (2022).
Tan, K. L. & Hameed, B. H. Insight into the adsorption kinetics models for the removal of contaminants from aqueous solutions. J. Taiwan. Inst. Chem. Eng. 74, 25–48 (2017).
Rahman, N. & Haseen, U. Equilibrium modeling, kinetic, and thermodynamic studies on adsorption of Pb(II) by a hybrid inorganic-organic material: polyacrylamide zirconium(IV) iodate. Ind. Eng. Chem. Res. 53, 8198–8207 (2014).
Al-Musawi, T. J. et al. Levofloxacin Adsorption onto MWCNTs/CoFe2O4 Nanocomposites: Mechanism, and Modeling Using Non-Linear Kinetics and Isotherm Equations. Magnetochemistry 9(1), 9 (2023).
Brdar, M., Šćiban, M., Takači, A. & Došenović, T. Comparison of two and three parameters adsorption isotherm for Cr(VI) onto kraft lignin. Chem. Eng. J. 183, 108–111 (2012).
Wang, J., Guo, X., Tsang, D. & Ma, L. Q. Adsorption kinetics and isotherm models of heavy metals by various adsorbents: an overview. Crit. Rev. Environ. Sci. Technol. 53, 1837–1865 (2023).
Mahanty, B., Behera, S. K. & Sahoo, N. K. Misinterpretation of Dubinin–Radushkevich isotherm and its implications on adsorption parameter estimates. Sep. Sci. Technol. (Philadelphia). 58, 1275–1282 (2023).
Zhou, X., Maimaitiniyazi, R. & Wang, Y. Some consideration triggered by misquotation of Temkin model and the derivation of its correct form. Arabian Journal of Chemistry 15(11), Preprint at https://doi.org/10.1016/j.arabjc.2022.104267 104267 (2022).
RezaeiKalantary, R. et al. Adsorption and magnetic separation of lead from synthetic wastewater using carbon/iron oxide nanoparticles composite. J. Mazandaran Univ. Med. Sci. 24, 172–183 (2014).
Bagheri, S., Esrafili, A., Kermani, M., Mehralipour, J. & Gholami, M. Performance evaluation of a novel rGO-Fe0/Fe3O4-PEI nanocomposite for lead and cadmium removal from aqueous solutions. J. Mol. Liq. 320, 114422 (2020).
Dong, Z. et al. Bio-inspired surface-functionalization of graphene oxide for the adsorption of organic dyes and heavy metal ions with a superhigh capacity. J. Mater. Chem. Mater. 2, 5034–5040 (2014).
Hur, J., Shin, J., Yoo, J. & Seo, Y. S. Competitive Adsorption of Metals onto Magnetic Graphene Oxide: Comparison with Other Carbonaceous Adsorbents. The Scientific World Journal 2015, 836287 (2015).
Vuković, G. D. et al. Removal of lead from water by amino modified multi-walled carbon nanotubes. Chem. Eng. J. 173, 855–865 (2011).
Mahmoud, M. E., Abdelwahab, S. & Ibrahim, G. A. A. M. The design of SnO2-crosslinked-chitosan nanocomposite for microwave-assisted adsorption of aqueous cadmium and mercury ions. Sustain. Chem. Pharm. 28, 100731 (2022).
Haq, S. et al. Adsorption of Cd2 + ions on plant mediated SnO2 nanoparticles. Mater. Res. Express 3, 105019 (2016).
Acknowledgements
The authors would like to thank İstanbul Technical University Scientific Research Projects Coordination Unit for the project supported in Project Number: MAB-2023-44836.
Author information
Authors and Affiliations
Contributions
Selim Gürsoy: Formal analysis; Investigation; Methodology; Validation; Visualization; Roles/Writing - original draft.Miray Bombom: Formal analysis; Methodology; Validation; Roles/Writing - original draft.Buse Tuğba Zaman: Formal analysis; Methodology; Validation; Roles/Writing - original draft.Fatma Turak: Formal analysis; Methodology; Validation; Roles/Writing - original draft.Sezgin Bakırdere: Conceptualization; Investigation; Methodology; Supervision; Validation; Writing - review & editing.Elif Öztürk Er: Conceptualization; Investigation; Methodology; Supervision; Validation; Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gürsoy, S., Bombom, M., Zaman, B.T. et al. Investigation of cadmium removal using tin oxide nanoflowers through process optimization, isotherms and kinetics. Sci Rep 15, 14597 (2025). https://doi.org/10.1038/s41598-025-99636-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-99636-y
Keywords
This article is cited by
-
Equilibrium modelling and kinetic studies on adsorption of cadmium from lake water by a magnetic covalent organic framework
Scientific Reports (2026)
-
A comprehensive review of cadmium removal by adsorptive mechanism from wastewater using carbon-based nanotubes
Applied Water Science (2026)
-
Mechanistic and performance evaluation of SnO2-AcCS composite in the adsorptive remediation of hg (II) ions from aqueous systems
Discover Chemistry (2025)