Abstract
Type 1 diabetes mellitus (T1DM) can lead to severe diabetic osteopathy, largely driven by alterations in the bone marrow hypoxic microenvironment and damage to type H vessels. This study employed multimodal imaging—including DCE-MRI, Micro-CT, and USPIO-enhanced MRI—to enable early in vivo assessment of hypoxic changes and type H vessel impairment in a T1DM rabbit model. Experiments involved 20 rabbits with alloxan-induced T1DM and controls, evaluated four months post-modeling. Imaging revealed significant differences in bone marrow microcirculatory perfusion and vascular permeability in T1DM rabbits, along with elevated USPIO uptake and regional heterogeneity that correlated with type H vessel distribution. Accompanying pathological changes were confirmed via immunofluorescence, qPCR, and transmission electron microscopy, suggesting an association of the AGEs/ROS-HIF-1α-VEGF pathway with these microvascular lesions. Our findings offer visual and quantitative imaging evidence to inform future clinical strategies targeting type H vessel hypoxia in diabetic osteopathy.
Similar content being viewed by others
Introduction
Diabetic bone disease (DBD) is a skeletal disorder in patients with diabetes mellitus caused by metabolic dysregulation, microvascular complications, and chronic inflammation. It manifests as abnormal bone mineral density, deterioration of bone microstructure, and significantly increased fracture risk, particularly in type 1 diabetes mellitus (T1DM), where progression is more rapid and severe 1,2. Recent studies highlight hypoxia in the bone marrow microenvironment as a critical mechanism underlying DBD pathogenesis. Ameliorating bone marrow hypoxia has emerged as an effective strategy to mitigate diabetic bone marrow complications, including marrow adiposity, dysregulated bone metabolism, and fibrosis 3.
The advanced glycation endproducts (AGEs)/reactive oxygen species (ROS)-hypoxia-inducible factor-1α (HIF-1α) pathway plays a pivotal role in diabetic bone disease. A bidirectional vicious cycle exists between AGEs and ROS, characterized by mutual promotion of their generation and synergistic amplification of inflammation and oxidative stress 4,5,6. HIF-1α serves as a critical downstream effector in the AGEs/ROS signaling axis, collectively forming a pathogenic triad of metabolic disturbance, hypoxia, and oxidative stress. Furthermore, HIF-1α overexpression modulates bone marrow neovascularization via vascular endothelial growth factor (VEGF). AGEs/ROS and the HIF-1α-VEGF axis exhibit synergistic and overlapping regulatory effects on inflammation, oxidative stress, and microvascular perfusion, ultimately contributing to the pathological cascade of disruption of bone microstructure, increased bone fragility, and elevated fracture risk 7,8.
Previous studies identified a novel capillary subtype—type H vessels—located near the metaphyseal growth plate, periosteum, and endosteum of long bones. These vessels are characterized by endothelial cells with high expression of CD31 and endomucin (CD31hiEmcnhi) and are distinct from type L vessels predominantly distributed in the diaphysis 9. Under physiological conditions, type H vessels exhibit a higher partial pressure of oxygen (pO2) compared to type L vessels and play a critical role in regulating skeletal development, repair, and remodeling.In T1DM, NOX2-mediated ROS accumulate intra- and extracellularly, inducing endothelial oxidative stress that disrupts bone marrow hypoxia homeostasis. The HIF-1α-VEGF pathway exacerbates bone formation impairment through angiogenic-osteogenic decoupling mechanisms in type H vessels, which may underlie early-stage T1DM-related osteogenic dysfunction and frequent fragility fractures10,11.
Our group has previously investigated diabetic microvascular complications and microstructural changes in bone marrow and skeletal muscle using single-modality quantitative imaging and radiomic analysis 12,13,14,15,16,17,18. Nonetheless, the mecshanisms driving hypoxic microenvironments and type H vessel injury in T1DM remain unclear. This study employs multimodal imaging to quantitatively visualize type H vascular architecture in T1DM rabbit bone marrow and to examine potential spatiotemporal heterogeneity in HIF-1α-mediated microenvironmental alterations and type H vessel dysfunction. The outcomes may support the development of hypoxia-targeted therapies aimed at preserving vascular integrity and mitigating diabetic bone disease.
Materials and methods
Animal grouping and model preparation
This study was conducted at the Animal Experiment Center and Department of Radiology, Renmin Hospital of Wuhan University, with ethical approval from the Institutional Animal Care and Use Committee (IACUC Approval No. WP20230346). All procedures followed the ARRIVE guidelines and the National Research Council’s Guide for animal care. Forty male Japanese Big-ear white rabbits (2–3 months old, 1.2–1.6 kg) were obtained from Wanqian Jiaxing Biotechnology Co., Ltd. [SCXK (E) 2018-0022]. After one week of acclimatization, the rabbits were randomly assigned to a T1DM group (n=20) or a non-diabetic control group (n=20).
T1DM was induced via two intravenous injections of alloxan (5% solution, Sigma, USA) administered one week apart: the first at 60 mg/kg and the second at 100 mg/kg. Control animals received equivalent volumes of saline. All injections were performed under anesthesia with 3% pentobarbital sodium after 12 hours of fasting. Diabetes was confirmed 48 hours post-injection by a blood glucose level exceeding 14 mmol/L once or 11 mmol/L twice consecutively 19. Blood glucose was monitored monthly for four months. All rabbits were euthanized humanely via sodium pentobarbital injection four months after model validation, followed by standardized tissue collection.
Dynamic contrast-enhanced MRI (DCE-MRI) scanning of bone marrow and quantitative parameter evaluation
Four months post-model induction, rabbits from both T1DM and control groups were anesthetized with pentobarbital sodium (1 mL/kg) via the marginal ear vein and scanned in a supine, feet-first position using a 3.0-T MRI system (Signa Architect, GE Healthcare) with an 8-channel knee coil. Oblique coronal DCE-MRI was acquired using a DISCO sequence with the following parameters: TR = 5.4 ms, TE = 1.5 ms, slice thickness = 1.5 mm, FOV = 160 × 160 mm, matrix = 192 × 192, flip angle = 15°. A 12-second mask scan preceded intravenous injection of gadodiamide (0.2 mmol/kg at 1.0 mL/s), followed by a 5 mL saline flush. A 48-phase dynamic scan was performed, each phase lasting 5 s, totaling 4 minutes and 12 seconds.
Motion correction was applied using the non-rigid registration tool within the PHI software (GE Omni-Kinetics). All subsequent image analyses were performed by two experienced researchers who were blinded to the group assignment to minimize bias. The arterial input function (AIF) was determined by manually drawing an ROI within the lumen of the major tibial nutrient artery, which was clearly identified on pre-contrast anatomical scans. For pharmacokinetic modeling, a standard hematocrit value of 0.45 was assumed for the conversion of blood signal to plasma contrast concentration. For tissue quantification, ROIs were manually drawn within the bone marrow of metaphyseal and diaphyseal regions, carefully avoiding cortical bone and visible vessels. Inter- and intra-observer reliability for ROI placement was assessed and showed excellent agreement (intra-class correlation coefficient > 0.85). The Extended Tofts model was used to calculate pharmacokinetic parameters: Ktrans (volume transfer constant), Kep (rate constant), and Ve (extravascular extracellular volume fraction). Each ROI was measured three times, and the mean value was recorded.
In vivo USPIO-enhanced MRI of bone marrow vascular endothelial cells and quantitative parameter evaluation
Four months post-model induction, anesthetized rabbits (pentobarbital sodium, 1 mL/kg) from both T1DM and control groups were scanned supine in a 3.0-T MRI system (Signa Architect, GE Healthcare) using an 8-channel knee coil. An oblique coronal synthetic MRI (MAGiC sequence) was acquired, which provides quantitative T2 relaxometry maps directly. The acquisition parameters were as follows: TR = 9942 ms, TE = 21.2 ms, NEX = 1, slice thickness = 0.8 mm, spacing = 0.2 mm, 40 slices, FOV = 120 × 120 mm, matrix = 240 × 240; scan time = 7 min 32 s.
Paired baseline scans were acquired prior to contrast administration. USPIO (1 mg Fe/kg) was then manually injected via the marginal ear vein. Post-contrast scans were performed 1 hour after injection using the identical sequence and geometry. Image reconstruction and T2 map generation were performed on a GE AW 4.7 workstation using the vendor-provided synthetic MRI processing package with optimized settings. For analysis, the change in transverse relaxation rate (ΔR2) was calculated voxel-wise by subtracting the post-contrast R2 map from the baseline R2 map. This metric reflects USPIO uptake.
ROIs were manually drawn at metaphyseal and diaphyseal levels bilaterally on the coregistered baseline images, avoiding vessels and bone islands. These same ROIs were then applied to the corresponding ΔR2 maps. All analyses were performed by two researchers blinded to the experimental groups. To assess reproducibility, both intra- and inter-observer reliability were evaluated by having each observer repeat the ROI placements and measurements on a randomly selected subset of subjects with a two-week interval. The intra-class correlation coefficients for ΔR2 measurements exceeded 0.90, confirming high consistency. For each subject, T2/ΔR2 values within each ROI were measured three times, and the mean was recorded for statistical analysis.
Ex vivo bone marrow microvascular imaging using micro-CT and quantitative parameter evaluation
Four months post-model induction, anesthetized T1DM and control rabbits underwent cardiac catheterization followed by perfusion with saline (1.5 L), 4% PFA (AnyIn Biotechnology) for fixation, and 300 mL of Microfil MV-117 (Flow Tech) contrast agent. The contrast was polymerized overnight at 0–4 °C. All perfused specimens were then visually inspected; those showing incomplete or fragmented contrast filling in the tibial microvasculature (assessed under a dissection microscope) were excluded from further analysis to mitigate perfusion artifact. Tibiae were then harvested, decalcified, and scanned using a Skyscan 1276 Micro-CT system (Bruker) at 70 kV and 200 μA with a resolution of 20.2 μm. Vessel volume (VV), tissue volume (TV), vessel number (VN), and vessel volume fraction (VV/TV) were quantified metaphyseally and diaphyseally using CTAn 2.6 software on samples that passed the perfusion quality check.
Isolation and characterization of type H vascular endothelial cells in bone marrow
Four months after successful model induction, flow cytometry was performed to compare the proportion of type H vascular endothelial cells (CD31hiEmcnhi) between the T1DM and control groups. Bone Marrow Collection: Tibial bone marrow was harvested from T1DM and control rabbits and divided into metaphyseal and diaphyseal regions. Single-Cell Suspension Preparation: Red blood cells were lysed using ACK buffer. Cells were resuspended in complete medium at a density of 1 × 107cells/ml. Antibody Staining: Primary Antibodies: CD31 (ab212712, abcam, USA), EMCN (sc65495, Santa Cruz Biotechnology, USA); Secondary Antibodies: Donkey anti-Mouse IgG conjugated with Alexa Fluor® 647 (ANT034, antgene, China), Donkey anti-Rat IgG conjugated with Alexa Fluor® 488 (ANT035, antgene, China). Staining Procedure: Sequential incubation with primary and secondary antibodies (30 min each, room temperature, protected from light). Cells were washed and resuspended in PBS after each staining step. Flow Cytometric Sorting: Stained cells were analyzed using a Beckman Coulter flow cytometer. Gating Strategy: Live cells were selected based on forward/side scatter. CD31hiEmcnhi populations were identified using fluorescence thresholds. Quantification: The percentage of type H vascular endothelial cells was calculated for each group and anatomical region (metaphysis vs. diaphysis).
Fluorescence-based quantification of type H vessels, HIF-1α, VEGF expression, and neovascular sprouting in bone marrow
Four months after successful model induction, tibial bones from T1DM and control group rabbits were fixed in 4% PFA, followed by decalcification, trimming, dehydration, and paraffin embedding. Tissues were sectioned along the oblique coronal plane and subjected to immunofluorescence staining using CD31 antibody (ab212712, abcam, USA), EMCN antibody (sc65495, Santa Cruz Biotechnology, USA), VEGF antibody (MA5-13182, thermofisher, USA) and HIF-1α antibody (NB100-105, Novus, USA). Nuclei were counterstained with DAPI to visualize anatomical structures including the metaphysis, diaphysis, and growth plate, with images captured under a fluorescence microscope. Image-Pro Plus 6.0 software was utilized to measure the integrated fluorescence intensity (IntDen) and positive pixel area of CD31, EMCN, VEGF and HIF-1α in the metaphyseal and diaphyseal regions. The mean fluorescence intensity (MFI) was calculated as MFI = IntDen / Area. Additionally, vascular sprouts along the metaphyseal growth plate were quantified by counting CD31-positive structures per unit length of the growth plate.
Assessment of ROS levels in bone marrow
Four months after successful model induction, fresh bone marrow tissues from the tibiae of T1DM and control group rabbits were prepared into single-cell suspensions. According to the instructions of the SOD activity assay kit(A001-3-2; Nanjing Jiancheng Bioengineering Institute, Nanjing, China), reagents were added proportionally based on tissue weight. The samples were homogenized in an ice bath and then centrifuged at 8000 × g for 10 minutes at 4°C. The supernatant was collected, and absorbance was measured at 560 nm using a spectrophotometer. SOD enzyme activity was calculated according to the formula provided in the kit protocol.
Detection and quantification of secretory vesicles and USPIO uptake in bone marrow endothelial cells
Four months after successful model induction, fresh bone marrow tissue from the tibiae of T1DM and control group rabbits was processed into single-cell suspensions. The suspensions were layered onto isosmotic Percoll solution (AnyIn Biotechnology, China) and centrifuged to separate three distinct layers. The intermediate layer cells were aspirated, washed to remove residual Percoll, resuspended, and counted. Cells were seeded at a density of 1 × 106/mL in endothelial cell-specific medium and cultured in a 5% CO2 incubator at 37°C. When cells reached approximately 80% confluency, they were detached using trypsin digestion. For USPIO labeling, cells were incubated with 25 μg/mL USPIO for 24 hours. Subsequently, labeled endothelial cells were fixed with PFA and subjected to transmission electron microscopy (TEM; HITACHI HT7800, Japan) and Prussian blue staining. Quantitative analysis: TEM images of metaphyseal and diaphyseal bone marrow endothelial cells from T1DM and control groups were analyzed. Intracellular vesicle count per field and percentage area occupied by USPIO were measured using image analysis software.
Detection of angiogenesis-related gene expression levels in hypoxic injury
Four months after successful modeling, tibial bone marrow tissues from T1DM and control group rabbits were cryopreserved at -80°C. Total RNA was extracted using Trizol reagent, reverse-transcribed into cDNA, and subsequently subjected to qPCR amplification analysis. β-actin served as the endogenous reference gene, with expression levels calculated according to the 2−ΔΔCt method. The primer sequences are listed in Table 1.
Statistics and reproducibility
All statistical analyses were performed using SPSS 26.0 and R (version 4.3.0). The normality of data distribution for each variable was assessed using the Shapiro-Wilk test, and the homogeneity of variance was evaluated using Levene’s test.
To account for the non-independence of multiple measurements (ROIs) obtained from the same animal, linear mixed-effects models were employed for all intergroup comparisons. In these models, the experimental group (T1DM vs. Control) and/or anatomical region (Metaphysis vs. Diaphysis) were included as fixed effects, while the animal identity was included as a random intercept to control for within-subject correlations. For post-hoc pairwise comparisons, p-values were adjusted using the Tukey-Kramer method. Data derived from these models are presented as estimated marginal means ± standard error.
For bivariate correlations between quantitative parameters (e.g., imaging parameters vs. metabolic indices), Pearson’s correlation analysis was conducted. To control the family-wise error rate arising from multiple comparisons within the correlation matrix, the False Discovery Rate (FDR) correction (Benjamini-Hochberg procedure) was applied. Only FDR-adjusted p-values (q-values) < 0.05 were considered statistically significant.
Results
Outcomes of animal model establishment
In the T1DM group, longitudinal blood glucose monitoring demonstrated sustained hyperglycemia with one exclusion due to spontaneous normoglycemia. The remaining subjects met the diagnostic criteria of either a single measurement ≥14 mmol/L or two consecutive readings >11 mmol/L, confirming successful model establishment. During the experimental period, two T1DM rabbits died from diabetes-related complications, ultimately yielding 17 T1DM and 20 control rabbits for final statistical analysis.
Comparative evaluation of quantitative parameters between DCE-MRI and USPIO contrast-enhanced MRI
Analysis using linear mixed-effects models revealed significant alterations in bone marrow perfusion, permeability, and USPIO uptake in T1DM rabbits. For DCE-MRI parameters, there was a significant main effect of Group (T1DM vs. Control) on Ktrans (F(1, 76) = 1865.2, p < 0.001), Kep (F(1, 76) = 2110.6, p < 0.001), and Ve (F(1, 76) = 3947.7, p < 0.001), with T1DM rabbits showing markedly higher estimated marginal means (Table 2, Fig. 1a-b). A significant Group × Region interaction was also found for all three parameters (all p < 0.001). Post-hoc tests (Tukey-adjusted) confirmed that within the T1DM group, metaphyseal values were significantly greater than diaphyseal values (all p < 0.001), whereas no such regional difference existed in the control group (all p > 0.05). Similarly, USPIO-enhanced MRI results showed a highly significant main effect of Group on both the maximum T2 change value (F(1, 76) = 22178, p < 0.001) and rate (F(1, 76) = 22076, p < 0.001), with T1DM rabbits exhibiting substantially higher values than controls (Fig. 1c-e). Significant Group × Region interactions were also present (both p < 0.001). Post-hoc comparisons revealed that in the T1DM group, metaphyseal values significantly exceeded diaphyseal values for both metrics (both p < 0.001), a pattern absent in controls.
a Quantitative DCE-MRI Parameters Analysis of Tibial Bone in T1DM Rabbits. Red circles: Metaphyseal region of interest (ROI); Yellow circles: Diaphyseal ROI. Ktrans: Volume transfer constant; Kep: Rate constant; Ve: Extravascular extracellular volume fraction. b Estimated marginal means of DCE-MRI parameters (Ktrans, Kep, Ve) in bone marrow of T1DM and control rabbits. Error bars represent 95% confidence intervals. T1DM rabbits showed significantly higher values than controls for all parameters (all p < 0.001). Within the T1DM group, metaphyseal regions exhibited significantly elevated values compared to diaphyseal regions (all p < 0.001), a pattern not observed in controls. Statistical analyses were performed using linear mixed-effects models with Tukey-adjusted post-hoc tests. c USPIO-Enhanced MRI of Metaphysis and Diaphysis in T1DM and Control Rabbits. Red circles: Metaphysis; Black circles: Diaphysis. T1DM bone marrow demonstrated significantly increased T2 changes compared to controls (P < 0.05). d Temporal T2 Change Curves from USPIO-Enhanced MRI in T1DM and Control Rabbits. T1DM metaphysis showed significantly higher maximum T2 value and maximum T2 change rate than diaphysis (P < 0.05), whereas no significant differences were observed between metaphyseal and diaphyseal regions in controls (P > 0.05). e. Estimated marginal means of USPIO-enhanced MRI parameters (maximum T2 change and T2 change rate) in bone marrow of T1DM and control rabbits. Error bars represent 95% confidence intervals. T1DM rabbits showed significantly higher values than controls for both parameters (both p < 0.001). Within the T1DM group, metaphyseal regions exhibited significantly elevated values compared to diaphyseal regions (both p < 0.001), a pattern not observed in controls.
Quantitative evaluation of ex vivo bone marrow microvasculature parameters via micro-CT imaging
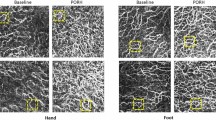
Micro-CT microvascular imaging revealed that type H vasculature is predominantly distributed in the metaphysis, presenting a dense dendritic branching network. In contrast, type L vasculature is mainly located in the diaphysis, displaying simpler, longitudinal tubular structures. Quantitative analysis using linear mixed-effects models demonstrated a significant main effect of Group (T1DM vs. Control) on both the vessel volume fraction (VV/TV) (F(1, 38) = 6708.2, p < 0.001) and vessel number (VN) (F(1, 38) = 4708.1, p < 0.001). T1DM rabbits exhibited markedly reduced estimated marginal means for both parameters compared to controls (Fig. 2). A significant main effect of Region (Metaphysis vs. Diaphysis) was also observed for both VV/TV (F(1, 38) = 141.5, p < 0.001) and VN (F(1, 38) = 153.5, p < 0.001). Crucially, a significant Group × Region interaction was detected for both parameters (VV/TV: F(1, 38) = 26.1, p < 0.001; VN: F(1, 38) = 22.4, p < 0.001). Post-hoc tests (Tukey-adjusted) elucidated this interaction: within the control group, metaphyseal VV/TV and VN were significantly higher than diaphyseal values (both p < 0.001). In stark contrast, this physiological regional gradient was abolished in the T1DM group, where metaphyseal and diaphyseal values did not differ significantly (VV/TV: p=0.124; VN: p=0.097). Direct between-group comparisons confirmed severe reductions in both the metaphysis and diaphysis of T1DM rabbits compared to their respective regional controls (all p < 0.001).
a, b Schematic Diagrams of Bone Marrow Vascular Spatial Distribution and Corresponding Spatial Cartesian Coordinate System Orientations in T1DM and Control Rabbits. Panel A: Tibial bone marrow microvasculature in T1DM rabbits; Panel B: Tibial bone marrow microvasculature in control rabbits. Yellow circles: Metaphysis; Green circles: Diaphysis; Red arrow: Horizontal X-axis direction; Green arrow: Vertical Y-axis direction; Blue arrow: Depth Z-axis direction. The metaphyseal and diaphyseal bone marrow microvessel counts in T1DM rabbits showed significant reductions compared to the control group (P < 0.05). c Comparison of Vascular Volume Fraction (VV/TV) and Vessel Number (VN) between T1DM and control rabbits in metaphysis and diaphysis. T1DM rabbits showed significant reductions in both VV/TV and VN compared to controls (both p < 0.001). In control rabbits, metaphyseal values were significantly higher than diaphyseal values (both p < 0.001), whereas no such regional difference was observed in T1DM rabbits.
Sorting and identification of endothelial cells in bone marrow type H vasculature
Comparative dual-positive sorting of CD31 and EMCN enabled the quantification of type H endothelial cells. Analysis using a linear mixed-effects model revealed a highly significant main effect of Group (T1DM vs. Control) on the percentage of type H endothelial cells (F(1, 38) = 1325.7, p < 0.001), with T1DM rabbits exhibiting a profound reduction compared to controls (Fig. 3). A significant main effect of Region (Metaphysis vs. Diaphysis) was also observed (F(1, 38) = 2046.3, p < 0.001), confirming the expected anatomical predominance of type H vasculature in the metaphysis. The analysis further identified a significant Group × Region interaction (F(1, 38) = 11.3, p = 0.0018). Post-hoc tests (Tukey-adjusted) elucidated these effects in detail. The proportion of type H endothelial cells was significantly higher in the metaphysis than in the diaphysis within both the T1DM group (p < 0.001) and the control group (p < 0.001). Most importantly, the model confirmed that the T1DM group had significantly lower type H endothelial cell percentages than the control group in both anatomical regions (metaphysis: p < 0.001; diaphysis: p < 0.001).
Flow cytometric analysis of type H vasculature endothelial cell proportion in bone marrow of T1DM and control rabbits. a Representative flow cytometry plots showing CD31hiEmcnhi population (type H endothelial cells) in the upper right quadrant. b Estimated marginal means of type H endothelial cell percentage in metaphysis and diaphysis. Error bars represent 95% confidence intervals. T1DM rabbits showed significantly reduced percentage of type H endothelial cells compared to controls (p < 0.001). In both groups, metaphyseal values were significantly higher than diaphyseal values (both p < 0.001).
Fluorescence detection of type H vasculature, HIF-1α, VEGF and Neovascular sprout quantity in bone marrow
DAPI staining visualized the structures of the tibial metaphysis, diaphysis, and growth plate. Quantitative analysis of immunofluorescence intensity using linear mixed-effects models revealed distinct alterations in the bone marrow expression of key vascular markers in T1DM rabbits (Fig. 4). For the type H vasculature markers, the model identified significant main effects of Group (T1DM lower than Control) and Region (Metaphysis higher than Diaphysis) for both CD31 intensity (Group: F(1, 38) = 2546.7, p < 0.001; Region: F(1, 38) = 539.8, p < 0.001) and EMCN intensity (Group: F(1, 38) = 3193.5, p < 0.001; Region: F(1, 38) = 1351.1, p < 0.001). Significant Group × Region interactions were also present (CD31: F(1, 38) = 43.1, p < 0.001; EMCN: F(1, 38) = 26.5, p < 0.001). Post-hoc tests (Tukey-adjusted) confirmed that within both the T1DM and control groups, metaphyseal intensities were significantly higher than diaphyseal intensities (all p < 0.001). Crucially, the T1DM group exhibited significantly reduced expression in both regions compared to their respective controls (all p < 0.001). In contrast, HIF-1α intensity demonstrated a divergent pattern. A significant main effect of Group (F(1, 38) = 5069.7, p < 0.001) indicated markedly higher expression in T1DM rabbits. A main effect of Region (F(1, 38) = 1311.5, p < 0.001) and a significant Group × Region interaction (F(1, 38) = 15.6, p < 0.001) were also observed. Post-hoc analysis revealed that within the T1DM group, metaphyseal HIF-1α intensity was significantly higher than diaphyseal intensity (p < 0.001), whereas no significant regional difference was found in the control group (p = 0.883). VEGF intensity paralleled the trends of CD31 and EMCN, showing significant main effects of Group (F(1, 38) = 5920.8, p < 0.001) and Region (F(1, 38) = 1355.3, p < 0.001), and a significant interaction (F(1, 38) = 49.5, p < 0.001). Post-hoc tests confirmed the characteristic metaphyseal predominance in controls (p < 0.001) and a significant reduction in the T1DM group across both regions (both p < 0.001). Consistent with the impairment in vascular markers and pro-angiogenic VEGF, the number of neovascular sprouts in the metaphyseal growth plate was significantly lower in T1DM rabbits (5.0 ± 0.2) compared to controls (12.1 ± 0.6), as determined by an unpaired t-test (t(78) = 63.9, p < 0.001).
a CD31 Immunofluorescence and DAPI Staining in T1DM and Control Rabbits. Scale bars in top-left corners indicate magnification. Dashed lines: Growth plate at metaphysis; White boxes: Neovascular sprouts on growth plate; Blue: DAPI; Red: CD31. a, e: DAPI staining in control and T1DM rabbits; b, f: CD31 staining in control and T1DM rabbits; c, g: Magnified CD31 staining in metaphysis of control and T1DM rabbits; d, h: Magnified CD31 staining in diaphysis of control and T1DM rabbits. b EMCN Immunofluorescence and DAPI Staining in T1DM and Control Rabbits. Scale bars in top-left corners indicate magnification. Dashed lines: Growth plate at metaphysis; Blue: DAPI; Cyan: EMCN. a, e: DAPI staining in control and T1DM rabbits; b, f: EMCN staining in control and T1DM rabbits; c, g: Magnified EMCN staining in metaphysis of control and T1DM rabbits; d, h: Magnified EMCN staining in diaphysis of control and T1DM rabbits. c CD31 + EMCN Dual Immunofluorescence in Metaphysis of T1DM and Control Rabbits. Scale bars in top-left corners indicate magnification. Dashed lines: Growth plate at metaphysis; Red: DAPI; Cyan: EMCN; Merge: CD31 + EMCN co-staining. d Estimated marginal means of CD31 and EMCN immunofluorescence intensity in T1DM and control rabbit bone marrow. Error bars represent 95% confidence intervals. T1DM rabbits showed significantly reduced intensity compared to controls for both markers (both p < 0.001). In both groups, metaphyseal values were significantly higher than diaphyseal values (both p < 0.001). e HIF-1α Immunofluorescence and DAPI Staining in Tibial Bone Marrow. Dashed lines: Growth plate at metaphysis; Red circles: Metaphysis; Yellow circles: Diaphysis; Blue: DAPI; Green: HIF-1α. a, e: Tibial DAPI staining in control and T1DM rabbits; b, f: Tibial HIF-1α staining in control and T1DM rabbits; c, g: Magnified HIF-1α staining in metaphysis of control and T1DM rabbits; d, h: Magnified HIF-1α staining in diaphysis of control and T1DM rabbits. f VEGF Immunofluorescence and DAPI Staining in Tibial Bone Marrow. Dashed lines: Growth plate at metaphysis; Red circles: Metaphysis; Yellow circles: Diaphysis; Blue: DAPI; Purple: VEGF. a, e: Tibial DAPI staining in control and T1DM rabbits; b, f: Tibial VEGF staining in control and T1DM rabbits; c, g: Magnified VEGF staining in metaphysis of control and T1DM rabbits; d, h: Magnified VEGF staining in diaphysis of control and T1DM rabbits. g Estimated marginal means of HIF-1α and VEGF immunofluorescence intensity and neovascular sprout quantity in T1DM and control rabbits. (A-B) Error bars represent 95% confidence intervals. T1DM rabbits showed significantly higher HIF-1α intensity (p < 0.001) and lower VEGF intensity (p < 0.001) compared to controls. (C) Neovascular sprout quantity (mean ± SEM) was significantly reduced in T1DM rabbits compared to controls (p < 0.001). Within the T1DM group, metaphyseal HIF-1α intensity was significantly higher than diaphyseal intensity (p < 0.001). In the control group, metaphyseal VEGF intensity was significantly higher than diaphyseal intensity (p < 0.001).
Detection of ROS levels and secretory vesicle quantity in bone marrow vascular endothelial cells and USPIO uptake measurement
Quantitative analysis using linear mixed-effects models revealed significant alterations in oxidative stress and USPIO handling in the bone marrow of T1DM rabbits. For SOD activity, a marker indicative of compensatory response to oxidative stress, the model showed a significant main effect of Group (F(1, 38) = 272.0, p < 0.001), with T1DM rabbits exhibiting higher estimated marginal means than controls. A significant main effect of Region (F(1, 38) = 550.6, p < 0.001) and a significant Group × Region interaction (F(1, 38) = 15.8, p < 0.001) were also detected. Post-hoc tests (Tukey-adjusted) confirmed that SOD activity was significantly elevated in the T1DM group compared to controls in both the metaphysis (p < 0.001) and diaphysis (p < 0.001). Within the T1DM group, metaphyseal activity was significantly higher than diaphyseal activity (p < 0.001), a regional difference that was less pronounced but still present in controls (p = 0.004) (Fig. 5a). Prussian blue staining confirmed the uptake of USPIO by bone marrow vascular endothelial cells (Fig. 5b), and transmission electron microscopy (TEM) visualized that this uptake occurred via vesicle-mediated mechanisms (Fig. 5c-d). Quantitative TEM analysis further demonstrated that both the percentage area occupied by USPIO and the number of vesicles per field were significantly increased in T1DM rabbits. For USPIO-occupied area, there were significant main effects of Group (F(1, 38) = 1664.7, p < 0.001) and Region (F(1, 38) = 1259.5, p < 0.001), and a significant interaction (F(1, 38) = 39.7, p < 0.001). Similarly, for vesicle number, significant main effects of Group (F(1, 38) = 1308.7, p < 0.001) and Region (F(1, 38) = 1483.8, p < 0.001), and a significant interaction (F(1, 38) = 25.3, p < 0.001) were observed. Post-hoc analysis for both parameters revealed an identical pattern: values in the T1DM group were significantly higher than in controls in both anatomical regions (all p < 0.001). Furthermore, within the T1DM group, metaphyseal endothelial cells exhibited significantly higher USPIO-occupied area and vesicle counts than diaphyseal cells (both p < 0.001). This pronounced regional gradient was absent in the control group for both parameters (both p > 0.05) (Fig. 5e).
a Estimated marginal means of superoxide dismutase (SOD) activity in bone marrow of T1DM and control rabbits. Error bars represent 95% confidence intervals. T1DM rabbits showed significantly higher SOD activity compared to controls (p < 0.001). Within both groups, metaphyseal activity was significantly higher than diaphyseal activity (both p < 0.001), with this regional difference being more pronounced in T1DM rabbits. b Prussian Blue Staining of USPIO-Labeled Bone Marrow Vascular Endothelial Cells in T1DM and Control Groups. Scale bars in top-left corners indicate magnification. Red boxes: USPIO-labeled Prussian blue-stained bone marrow vascular endothelial cells. The number of stained endothelial cells in the T1DM group was significantly higher than in the control group (P < 0.05). c, d TEM Images of USPIO Uptake via Vesicles in Metaphyseal and Diaphyseal Bone Marrow Endothelial Cells. a, c: Metaphyseal bone marrow endothelial cells in T1DM and control rabbits; b, d: Magnified vesicles within metaphyseal endothelial cells; e, g: Diaphyseal bone marrow endothelial cells in T1DM and control rabbits; f, h: Magnified vesicles within diaphyseal endothelial cells; Red boxes: Intracellular vesicles; Yellow arrows: USPIO within vesicles. The T1DM group showed significant increases in vesicle counts and USPIO-occupied area percentage compared to controls (P < 0.05). Within the T1DM group, metaphyseal endothelial cells exhibited significantly higher vesicle counts and USPIO-occupied area percentage than diaphyseal regions (P < 0.05). e Estimated marginal means of vesicle counts and USPIO-occupied area percentage per field in bone marrow of T1DM and control rabbits. (A) Vesicle counts and (B) USPIO-occupied area percentage are presented as estimated marginal means with 95% confidence intervals. T1DM rabbits showed significantly higher values compared to controls for both parameters in both anatomical regions (all p < 0.001). Within the T1DM group, metaphyseal values were significantly higher than diaphyseal values (both p < 0.001), whereas no such regional differences were observed in the control group.
Hypoxic injury and angiogenesis-related gene expression levels and correlation analysis
Quantitative analysis of mRNA expression using linear mixed-effects models revealed significant dysregulation of key hypoxic and angiogenic genes in the bone marrow of T1DM rabbits. For HIF-1α mRNA, the model identified significant main effects of Group (F(1, 38) = 654.3, p < 0.001) and Region (F(1, 38) = 1502.1, p < 0.001), and a significant Group × Region interaction (F(1, 38) = 154.9, p < 0.001). Post-hoc tests (Tukey-adjusted) confirmed that HIF-1α mRNA expression was significantly elevated in the T1DM group compared to controls in both the metaphysis (p < 0.001) and diaphysis (p < 0.001). Within the T1DM group, metaphyseal expression was significantly higher than diaphyseal expression (p < 0.001), while no significant regional difference was observed in the control group (p = 0.999) (Fig. 6a). Similarly, AGER mRNA expression showed significant main effects of Group (F(1, 38) = 1365.6, p < 0.001) and Region (F(1, 38) = 2545.7, p < 0.001), and a significant interaction (F(1, 38) = 235.0, p < 0.001). Post-hoc analysis revealed an identical pattern: markedly higher expression in T1DM rabbits across both regions (both p < 0.001), with a significant metaphyseal predominance within the T1DM group (p < 0.001) that was absent in controls (p = 0.997). In contrast, VEGF mRNA expression exhibited a divergent trend. While a significant main effect of Region was present (F(1, 38) = 1523.5, p < 0.001), the main effect of Group was also significant (F(1, 38) = 13246, p < 0.001), indicating dramatically lower expression in T1DM rabbits. A significant Group × Region interaction was observed (F(1, 38) = 318.7, p < 0.001). Post-hoc tests confirmed that VEGF mRNA was significantly downregulated in the T1DM group in both regions compared to controls (both p < 0.001). Within the T1DM group, metaphyseal expression remained higher than diaphyseal expression (p < 0.001), recapitulating the physiological pattern seen in controls (p < 0.001), albeit at a profoundly reduced overall level (Fig. 6a).
a Estimated marginal means of HIF-1α, AGER, and VEGF mRNA expression levels in the tibial metaphysis and diaphysis of T1DM and control rabbits. Error bars represent 95% confidence intervals. HIF-1α & AGER: Expression was significantly higher in T1DM rabbits compared to controls in both metaphysis and diaphysis (all p < 0.001). Within the T1DM group, metaphyseal expression exceeded diaphyseal expression (p < 0.001). VEGF: Expression was significantly lower in T1DM rabbits compared to controls in both regions (all p < 0.001). The physiological pattern of higher metaphyseal than diaphyseal expression was preserved in both groups (both p < 0.001). Statistical analyses were performed using linear mixed-effects models with Tukey-adjusted post-hoc tests. b Correlation heatmap of multimodal parameters in diabetic bone marrow. Pearson correlation matrix showing relationships among metabolic exchange (ΔT2_max, T2 change rate, vesicle number, USPIO_Area%), angiogenic (VEGF_IF, VEGF_mRNA), microvascular (VV/TV, vessel number), oxidative stress (SOD_Activity, AGER_mRNA), perfusion/permeability (Ktrans, Kep, Ve), HIF-1α (HIF1α_IF, HIF1α_mRNA), and type H vessel parameters. FDR-corrected significance: *q < 0.05, **q < 0.01, ***q < 0.001. Key correlations: positive between HIF-1α and metabolic/oxidative/perfusion parameters; negative between HIF-1α and angiogenic/microvascular parameters, supporting the AGEs/ROS-HIF-1α-VEGF axis in diabetic microvascular dysfunction.
Pairwise correlation analysis of all quantitative parameters from metaphyseal and diaphyseal regions in T1DM and control rabbits was performed based on experimental objectives, generating a correlation heatmap (Fig. 6b). To control the family-wise error rate arising from multiple comparisons, the False Discovery Rate (FDR) correction (Benjamini-Hochberg procedure) was applied to all correlation p-values. Significance thresholds in the heatmap are defined as: FDR-adjusted p (q) < 0.05: significant correlation; q < 0.01: highly significant correlation; q < 0.001: extremely significant correlation.
Discussion
This multimodal imaging study demonstrates, for the first time, a spatially heterogeneous microangiopathy in the bone marrow of T1DM rabbits, characterized by region-specific impairments in type H vessels that correlate with dysregulation of the AGEs/ROS–HIF‑1α–VEGF axis.
The observed microvascular dysfunction is mechanistically anchored in the dysregulated AGEs/ROS–HIF-1α–VEGF axis 20. In the diabetic bone marrow, accumulation of AGEs and concurrent ROS overproduction create a state of oxidative stress and pseudohypoxia 21,22. This is evidenced by increased SOD activity and, crucially, a marked upregulation of HIF-1α 23. Notably, this pathogenic signaling exhibits a distinct spatial gradient, with the metaphysis—a site enriched in type H vessels critical for osteogenic coupling 9,10 and showing a significant reduction in CD31hiEmcnhi endothelial cells in T1DM—demonstrating significantly higher levels of AGER, SOD, and HIF-1α than the diaphysis. Paradoxically, despite HIF-1α induction, downstream pro-angiogenic output is blunted: VEGF expression and neovascular sprouting are substantially reduced. This dissociation between hypoxic signaling and functional angiogenesis indicates a severe impairment of vascular-osteogenic coupling, likely contributing to the site-specific fragility of the metaphysis in T1DM 24,25,26.
Our multimodal imaging data provide in vivo and ex vivo visualisation of this dysregulated axis and its spatial heterogeneity. The functional consequences are captured by DCE MRI, which revealed elevated permeability (Ktrans, Kep) and extracellular volume (Ve), indicating a leaky, dysfunctional microvasculature 27. Structurally, micro-CT confirmed a loss of vascular density (VV/TV, vessel number), with the physiological metaphyseal predominance being abolished in T1DM. Furthermore, USPIO enhanced MRI and correlative TEM revealed altered endothelial vesicular activity—increased vesicle count and USPIO uptake 28,29,30—that was significantly more pronounced in the metaphysis. Importantly, quantitative correlation analysis integrated these disparate measures: HIF-1α levels positively correlated with metabolic/oxidative parameters (USPIO uptake, SOD, AGER) and perfusion dysfunction, but negatively correlated with angiogenic markers (VEGF, sprout density) and microvascular density. This coordinated pattern solidifies the imaging findings as direct correlates of the activated AGEs/ROS–HIF-1α–VEGF pathway.
Collectively, these results map a spatially heterogeneous microangiopathy in diabetic bone marrow, driven by the aforementioned axis and preferentially targeting the metaphyseal type H vasculature. The functional imaging signatures of permeability, altered metabolism, and rarefaction, concentrated in this osteogenic niche, offer a potential explanatory model for region-specific bone fragility 31,32.
Several limitations warrant consideration. First, the alloxan-induced rabbit model may not fully recapitulate human T1DM pathophysiology. Second, potential under-perfusion artifacts could affect vascular quantification accuracy. Third, the functional impact of type H vessel impairment on osteogenic coupling remains to be directly investigated. Fourth, the observational nature of this study establishes association, not causation; future interventional studies targeting the implicated pathway are needed.
Conclusion
This study establishes the feasibility of employing multimodal imaging for assessing HIF-1α-driven microenvironmental changes and structural and functional impairments of type H vessels in the bone marrow of type 1 diabetic rabbits. Our results uncover a spatially heterogeneous microangiopathy, associated with dysregulation of the AGEs/ROS–HIF‑1α–VEGF axis, which leads to region-specific dysfunction and maladaptive remodeling of type H endothelium. These findings offer quantitative and visualizable in vivo evidence that supports the development of hypoxia-targeted therapies to rejuvenate vascular-osteogenic coupling and combat diabetic bone disease.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Safarova, S. S. Alterations of bone metabolism in patients with diabetes mellitus. Int. J. Endocrinol. 59, 84681 (2019).
Faienza, M. F., Pontrelli, P. & Brunetti, G. Type 2 diabetes and bone fragility in children and adults. World J. Diabetes. 13(11), 900–911 (2022).
Lao, A. et al. Functionalized metal-organic framework-modified hydrogel that breaks the vicious cycle of inflammation and ros for repairing of diabetic bone defects. Small. 19(36), e2206919 (2023).
Zhang, Q. et al. A factor-free hydrogel with ROS scavenging and responsive degradation for enhanced diabetic bone healing. Small. 20(24), e2306389 (2024).
Qian, Y. et al. Immunoregulation in diabetic wound repair with a photoenhanced glycyrrhizic acid hydrogel scaffold. Adv Mater. 34(29), e2200521 (2022).
Shen, C. Y. et al. The development of maillard reaction, and advanced glycation end product (AGE)-receptor for AGE(RAGE) signaling inhibitors as novel therapeutic strategies for patients with AGE-related diseases. Molecules 25(23), 5591 (2020).
Bixel, M. G. et al. Angiogenesis is uncoupled from osteogenesis during calvarial bone regeneration. Nat Commun. 15(1), 4575 (2024).
Wang, J. et al. A triple-integrated 3D-printed composite scaffold of high-activity peptide-metal ion-bone cement facilitates Osteo-vascular regenerative repair of diabetic bone defects. Adv. Funct. Mater. 13, 2422950 (2025).
Kusumbe, A. P., Ramasamy, S. K. & Adams, R. H. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature 507(7492), 323–328 (2014).
Hu, X. F. et al. Impairment of type H vessels by NOX2-mediated endothelial oxidative stress: critical mechanisms and therapeutic targets for bone fragility in streptozotocin-induced type 1 diabetic mice. Theranostics. 11(8), 3796–3812 (2021).
Wu, Z. et al. ROS-reactive PMS/PC drug delivery system improves new bone formation under diabetic conditions by promoting angiogenesis-osteogenesis coupling via down-regulating NOX2-ROS signalling axis. Biomaterials 291, 121900 (2022).
Wang, Y. et al. Integration of texture analysis based on DCE-MRI Ktrans map and metabolomics of early bone marrow microvascular changes in alloxan-induced diabetic rabbits. BMC Med Imaging. 24(1), 247 (2024).
Hu, L. et al. Quantitative evaluation of vertebral microvascular permeability and fat fraction in alloxan-induced diabetic rabbits. Radiology 287(1), 128–136 (2018).
Yang, L. et al. Microvascular permeability and texture analysis of bone marrow in diabetic rabbits with critical limb ischemia based on dynamic contrast-enhanced magnetic resonance imaging. J. Diabetes. Investig. 15(5), 584–593 (2024).
Liu, B. et al. Evaluation of microvascular permeability of skeletal muscle and texture analysis based on DCE-MRI in alloxan-induced diabetic rabbits. Eur. Radiol. 31(8), 5669–5679 (2021).
Chen, P. et al. Evaluation of bone marrow texture and trabecular changes with quantitative DCE-MRI and QCT in alloxan-induced diabetic rabbit models. Front. Endocrinol. (Lausanne). 12, 785604 (2021).
Yang, Q. et al. Microvascular permeability and texture analysis of the skeletal muscle of diabetic rabbits with critical limb ischaemia based on DCE-MRI. Front. Endocrinol. (Lausanne) 13, 783163 (2022).
Zeitoun, D. et al. Microcomputed tomography of the femur of diabetic rats: alterations of trabecular and cortical bone microarchitecture and vasculature- a feasibility study. Europ. Radiol. Experim. 3(1), 17 (2019).
Liu, T. et al. Rosiglitazone attenuates atrial structural remodeling and atrial fibrillation promotion in alloxan-induced diabetic rabbits. Cardiovasc Ther. 32(4), 178–183 (2014).
Gao, H. et al. Role of hypoxia in cellular senescence. Pharmacol Res. 194, 106841 (2023).
Gao, Q. et al. Advanced glycation end products mediate biomineralization disorder in diabetic bone disease. Cell Rep Med. 5(9), 101694 (2024).
Wang, Y. et al. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol. 217(6), 1915–1928 (2018).
Qiu, M. et al. Activation of the osteoblastic HIF-1α pathway partially alleviates the symptoms of STZ-induced type 1 diabetes mellitus via RegIIIγ. Exp Mol Med. 56(7), 1574–1590 (2024).
Koh, B. I. et al. Adult skull bone marrow is an expanding and resilient haematopoietic reservoir. Nature 636(8041), 172–181 (2024).
Bi, J., Zhou, W. & Tang, Z. Pathogenesis of diabetic complications: Exploring hypoxic niche formation and HIF-1α activation. Biomed Pharmacother. 172, 116202 (2024).
Peng, Y. et al. Type H blood vessels in bone modeling and remodeling. Theranostics. 10(1), 426–436 (2020).
Liu, Z. et al. Immunosenescence: molecular mechanisms and diseases. Signal Transduct Target Ther. 8(1), 200 (2023).
Tiwari, A. et al. Cross-modal imaging reveals nanoparticle uptake dynamics in hematopoietic bone marrow during inflammation. ACS Nano 18(9), 7098–7113 (2024).
Sindhwani, S. et al. The entry of nanoparticles into solid tumours. Nat Mater. 19(5), 566–575 (2020).
Wang, X. et al. Hypoxia promotes EV secretion by impairing lysosomal homeostasis in HNSCC through negative regulation of ATP6V1A by HIF-1α. J Extracell Vesicles. 12(2), e12310 (2023).
Iwamoto, J. et al. Effect of vitamin K2 on cortical and cancellous bone mass and hepatic lipids in rats with combined methionine-choline deficiency. Bone 48(5), 1015–1021 (2011).
Aydingoz, U. Imaging osteomyelitis: An update. Rofo. 195(4), 297–308 (2023).
Acknowledgements
We acknowledge the financial support of the National Natural Science Foundation of China (No. 82171895).
Funding
This study was supported by the National Natural Science Foundation of China (No. 82171895).
Author information
Authors and Affiliations
Contributions
H. L. designed the study and wrote the paper. K. W. analyzed the images. L. L. and C. L. analyzed the data. Y. Z. was responsible for the study design, and the final approval of the version to be published. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lei, H., Wang, K., Li, L. et al. Multimodal imaging evaluation of hypoxic bone marrow microenvironment and type H vascular injury in diabetes. Sci Rep 16, 7206 (2026). https://doi.org/10.1038/s41598-026-37164-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37164-z