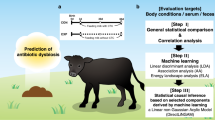
Abstract
This study evaluated the effects of traditional Chinese herbal medicine compounds (TCHMCs) on gut microbiota, maternal health, and calf performance in perinatal yaks. Thirty-two yaks with expected calving dates (two months pre-calving to one month post-calving) were randomly assigned to four groups (n = 8). Groups WA, WB, and WC received normal diets supplemented with different TCHMC formulations, while group WD served as the control. High-throughput sequencing revealed that TCHMC supplementation significantly increased gut microbial diversity and altered community composition. Compared with controls, supplemented yaks exhibited higher abundances of Bacteroidetes and enrichment of short-chain fatty acid (SCFA)-producing genera, including Phascolarctobacterium, Butyricicoccus, Bulleidia, and Blautia. Potentially beneficial taxa with anti-inflammatory properties, such as Adlercreutzia, Akkermansia, Odoribacter, Dehalobacterium, and Sporosarcina, were also enriched. Calves born to treated yaks demonstrated significantly greater birth weights, reduced serum levels of pro-inflammatory cytokines (TNF-α, IL-6), and improved antioxidant capacity, as indicated by increased T-AOC and SOD activity. Among the three formulations, TCHMCⅡ (WB) exerted the most pronounced effects, enhancing microbial shifts, maternal physiological resilience, and neonatal health outcomes. In conclusion, dietary supplementation with TCHMCs during the perinatal period modulated gut microbiota, reduced maternal inflammation, strengthened antioxidant defenses, and improved calf growth performance. These findings provide new evidence supporting the use of TCHMCs as practical nutritional interventions to improve reproductive performance and offspring vitality in yak husbandry under high-altitude conditions.
Similar content being viewed by others
Introduction
Yaks (Bos grunniens) inhabit the extreme conditions of the Qinghai-Tibet Plateau at altitudes ranging from 2,500 to 6,000 m. They play an indispensable role in the daily lives of Tibetan herders, providing milk, meat, fur, and fuel (manure used as a fuel source), all of which are vital for both sustenance and economic income1. Therefore, improving the breeding efficiency of yaks is essential for advancing the yak farming industry and supporting the economic growth of Tibetan regions. Despite their significance, yaks remain semi-domesticated, with most still grazing freely on grasslands and breeding without human intervention. This dependence on natural resources poses challenges, particularly during the calving season when forage on the plateau becomes scarce2. The perinatal period is a critical phase for female yaks, as it affects not only their health but also the growth, development, and future reproductive performance of their calves3.
During late pregnancy, fetal development accelerates, greatly increasing the mother’s nutritional demands. However, the scarcity of high-quality forage during the plateau’s cold season creates a mismatch between these demands and the nutrients available to the mother. This imbalance often results in inadequate fetal nourishment, leading to lower birth weight and weakened immunity in calves. Addressing these challenges by enhancing the physical fitness of female yaks during the perinatal period, supporting their reproductive performance, and improving calf health is essential.
Chinese herbal medicine, a natural and residue-free feed additive, offers a promising solution. In the livestock industry, it has been used to improve the animal’s performance and enhance the quality of livestock products4,5,6. The active ingredients in Chinese herbal medicine may strengthen the physical fitness and immunity of female yaks, reduce disease incidence, and provide a healthier environment for fetal development. Healthier yaks can better absorb and utilize nutrients, thereby supporting fetal growth and development4. In addition, certain herbal medicines contain vitamins, minerals, amino acids, and other bioactive substances that directly promote fetal development7. Some traditional Chinese medicines also stimulate the appetite in female yaks, improve the feed conversion efficiency, help to maintain optimal nutritional status during the perinatal period, and provide sufficient energy for reproduction and lactation8.
The gastrointestinal tract is a vital organ responsible for digestion, nutrient absorption, endocrine regulation, and immunity. Consequently, the digestive tract microbiome is closely linked to overall health9. In ruminants, the composition of the intestinal microbiota is influenced by diet, feed form, and stressors such as pregnancy and dietary changes. Fluctuations in micro-biome composition and structure often reflect an animal’s immune function, metabolic state, and disease status. Serum biochemical parameters are key indicators of physiological status, insight into tissue health, nutritional metabolism, inflammation, and oxidative stress. These parameters serve as reliable indicators of animal health.
Fluctuations in micro-biome composition and structure often reflect an animal’s immune function, metabolic state, and disease status. Serum biochemical parameters are also key indicators of physiological status, providing insight into tissue health, nutritional metabolism, inflammation, and oxidative stress. These parameters serve as reliable indicators of animal health.
In this study, the physiological status and nutritional requirements of perinatal female yaks were dialectically studied using traditional Chinese medicine. The formulation of the herbal compounds was guided by Tibetan and Chinese medical theory, which emphasizes nourishing blood, replenishing qi, enhancing digestion, and stabilizing pregnancy during the perinatal period. Specific herbs were selected based on their documented pharmacological properties, including antioxidant, anti-inflammatory, and immunomodulatory effects, as well as their historical use in Tibetan pastoral medicine. For example, Astragalus propinquus and Codonopsis pilosula are known to enhance energy metabolism and immunity, while Angelica sinensis and White Peony Root support blood nourishment and fetal protection. Preliminary screenings from livestock studies and literature reports further supported their inclusion.
Three herbal mixtures were prepared by combining traditional Chinese herbal medicine with locally sourced Tibetan herbs from Xizang, designed to achieve synergistic effects among active ingredients rather than relying on a single-compound intervention. These compounds were incorporated into the diets of perinatal female yaks at specific proportions. The study then assessed their impact on the health status and reproductive performance of pregnant yaks, as well as the growth and immunity of their calves. These findings were based on changes observed in the gut microbiota of female yaks and the serum biochemical parameters of their calves.
Results
Statistical analysis of initial weight, age, and body condition variations
The statistical analysis confirms that there were no significant differences in age, initial body weight, or body condition score among the four treatment groups at the study’s outset (P > 0.05) (Table 1).
High-throughput sequencing data analysis
High-throughput sequencing yielded a total of 3,552,385 raw sequences (WZ1: 1,614,707; WZ2: 1,937,678), averaging 74,008 ± 1,716 raw sequences (mean ± SEM) per sample. Following denoising, 3,175,911 effective sequences (WZ1: 1,439,898; WZ2: 1,736,013) were obtained, with an average of 66,164 ± 1,518 effective sequences per sample. The percentage of valid sequences exceeded 86%, with an average of 89 ± 0.12% per sample. Of these, 1,856,636 sequences were retained for species classification annotation (Table 2).
Rarefaction curves were plotted to examine the trend of alpha diversity with sequencing depth. The observed features curve plateaued as sequencing depth increased, indicating that the sequencing data were sufficient to capture the diversity within the samples and approached saturation, thus meeting the requirements for further analysis (Fig. 1a). Rank abundance curves revealed differences in ASV abundance distribution patterns between groups. The experimental groups exhibited a flatter curve, suggesting a higher level of community composition uniformity in the experimental groups (Fig. 1b).
High-throughput sequencing data analysis of rectal microbiota in periparturient yaks. (a) Rarefaction curves show that sequencing depth was sufficient to capture the majority of microbial diversity across all groups. (b) Rank abundance curves indicate differences in species richness and evenness among treatments. (c) Venn diagram illustrates both shared and unique bacterial OTUs, with overlapping regions reflecting core microbiota and distinct regions highlighting treatment-specific taxa.
A Venn diagram analysis of the intestinal microbiota community revealed distinct sequence variant (ASV) distributions among the four groups. In the midterm samples, 1,155 core ASVs (7.64%) were shared across all groups, while specific ASVs numbered 2,939 (19.45%) in WAZ1, 2,793 (18.48%) in WBZ1, 2,950 (19.52%) in WCZ1, and 3,075 (20.35%) in WDZ1 (Fig. 1c). For samples collected at the end of the experiment, 1,207 core ASVs (7.1%) were identified, with specific ASVs totaling 3,535 (20.79%) in WAZ2, 3,178 (18.69%) in WBZ2, 3,378 (19.87%) in WCZ2, and 2,960 (17.41%) in WDZ2.
The addition of traditional Chinese herbal medicine compounds affected the diversity, composition and function of gut microbiota in perinatal female yaks
Alpha diversity analysis revealed no significant differences in microbial diversity among the groups in the initial sample (Fig. 2a). However, as the experiment progressed, the richness and diversity of microorganisms in the experimental groups demonstrated significant improvement, as indicated by various diversity metrics (Fig. 2b). Principal coordinate analysis (PCoA) and non-metric multidimensional scaling (NMDS) of fecal samples collected on the 30th day of the experiment showed clear separation between the experimental groups and the control group (Fig. 2d). In contrast, no significant separation was observed for samples collected on the 15th day (Fig. 2c).
Diversity analysis of intestinal bacteria in yaks supplemented with different TCHMCs. (a, b) Alpha diversity indices (Shannon, Simpson, Chao1) at days 15 and 30 demonstrate that herbal supplementation groups (WA, WB, WC) generally exhibited greater richness and diversity compared with the control (WD). (c, d) Beta diversity analysis (PCoA) at days 15 and 30 shows distinct clustering of microbial communities by treatment, suggesting that TCHMCs significantly altered overall gut microbial structure over time.
Figure 3 illustrates the relative abundance of bacteria at various taxonomic levels. Firmicutes, Bacteroidetes, and Actinobacteria were the most abundant phyla across all samples, although their relative proportions varied among the groups. Firmicutes were more abundant in the control group (15d: 70.88%, 30d: 71.54%) compared to the three experimental groups receiving Chinese herbal medicine (15d: 57.97%, 30d: 64.43%). Conversely, the abundance of Bacteroidetes was lower in group FD, with this trend becoming more pronounced over time (15d: 18.24%, 30d: 16.95%). In the experimental groups, Bacteroidetes abundance was 18.16% on day 15 and increased to 22.56% by day 30, with a notable increase in group WA (Fig. 3a, f). Among all identifiable families, Ruminococcaceae was the most prevalent across all groups. Ruminococcaceae abundance was lower in group WD at the halfway point of the experiment, and this trend became more evident by the conclusion of the study (Fig. 3d, i).
Relative abundance and distribution of intestinal bacteria at different taxonomic levels. (a–e) Taxonomic composition at phylum, class, order, family, and genus levels on day 15. (f–j) Corresponding taxonomic distributions on day 30. Notably, TCHMC supplementation shifted the dominance of Firmicutes and Bacteroidetes, and enriched beneficial genera such as Blautia and Butyricicoccus, indicating modulation of the gut microbial ecosystem compared with the control group.
At the genus level, the most abundant genera in the Z1 groups were Arthrobacter, Akkermansia, and Solibacillus. Arthrobacter was more prevalent in the groups that received traditional Chinese medicine, particularly in group WC, while Akkermansia was notably richer in group WA. The abundance of Solibacillus was higher in the group with a normal diet (Fig. 3e). In the Z2 groups, Akkermansia, Clostridium, and Oscillospira were the most abundant genera, with Akkermansia remaining dominant in group WA, whereas Clostridium was more abundant in group WD. Additionally, the relative abundance of Solibacillus and Arthrobacter decreased during the later stages of the experiment, though Arthrobacter remained more abundant in the experimental groups (Fig. 3j).
PICRUSt2 (Phylogenetic Investigation of Communities by Reconstruction of Unobserved States) was employed to annotate the 16 S rRNA gene sequence in the KEGG database. The results indicated that the microbial functions in the experimental groups (those receiving traditional Chinese medicine) diverged from those in the control group by the end of the experiment (Fig. 4a, b). KEGG pathway analysis identified six primary pathways (Cellular Processes, Environmental Information Processing, Genetic Information Processing, Human Diseases, Metabolism, and Organismal Systems) and 33 secondary pathways. The predicted abundance map of KEGG secondary functional pathways is presented in Fig. 4c and d. No significant differences were observed in the differential analysis.
Functional predictions of intestinal microbiota in yaks receiving TCHMCs. (a, b) PCA plots of predicted microbial functions on days 15 and 30 reveal functional clustering that differentiates treatment groups from the control. (c, d) KEGG pathway analysis shows that TCHMCs enhanced microbial functions related to carbohydrate metabolism, energy production, and antioxidant activity, suggesting potential links to improved host physiological resilience (46).
The addition of traditional Chinese herbal medicine compounds significantly impacted gut microbiota in perinatal female yaks
T-test analysis revealed that Firmicutes and Spirochaetes consistently maintained a significantly higher abundance in the control group throughout the experiment. Actinobacteria and Tenericutes were notably more abundant in groups WB and WC at the midpoint of the experiment (Fig. 5a). Bacteroidetes were enriched in groups WA and WC, while Verrucomicrobia exhibited higher abundance in group WA. Additionally, Proteobacteria showed increased abundance in group WD compared to groups WA and WB (Fig. 5b). It is noteworthy that the Firmicutes/Bacteroidetes ratio increased in the group of yaks fed with a normal diet by the end of the experiment.
T-test analysis of bacterial composition at the phylum level. (a, b) Significant differences in dominant bacterial phyla between treatment groups (WA, WB, WC) and the control (WD) at days 15 and 30. TCHMC supplementation increased Bacteroidetes abundance while reducing Firmicutes, a shift often associated with improved metabolic health.
In the midterm samples, Turicibacter, Blautia, and Treponema were significantly enriched in the group of yaks fed a normal diet. In contrast, L7A_E11, Arthrobacter, and Butyrivibrio showed higher abundance in the groups receiving added Chinese herbal medicine. Furthermore, the abundance of Phascolarctobacterium, Blautia, Sharpea, and Bifidobacterium was higher in group WB than in group WD. Group WC exhibited a higher abundance of Paenibacillus, YRC22, and BF311, while the abundance of [Clostridium] was lower in group WC compared to group WD (Fig. 6a). In the samples collected at the end of the experiment, 5-7N15, CF231, RC4-4, Odoribacter, and BF311 were significantly enriched in the experimental groups. Conversely, [Clostridium], Treponema, Coprococcus, Blautia, and Succinivibrio were more abundant in the control group. Additionally, Akkermansia and Serratia were more abundant in group WA than in group WD, while SMB53 and Anaerostipes exhibited the opposite trend. Adlercreutzia was notably more abundant in group WB, while Parabacteroides and Neisseria were more prevalent in group WD. Group WC also displayed a visible increase in the abundance of Dehalobacterium and Sporosarcina (Fig. 6b).
T-test analysis of bacterial composition at the genus level. (a, b) Comparison at days 15 and 30 shows enrichment of short-chain fatty acid (SCFA)-producing genera such as Blautia and Phascolarctobacterium in TCHMC-treated groups, supporting their potential role in modulating gut health and anti-inflammatory responses.
LEfSe (Linear Discriminant Analysis Effect Size) analysis highlighted the significantly enriched species and their importance within each group, yielding results consistent with the t-test analysis. The order Pasteurellales, family Pasteurellaceae, family Streptococcaceae, and genus Streptococcus were enriched in the WAZ1 group. The phylum Actinobacteria, class Actinobacteria, family Lactobacillaceae, and genus Lactobacillus were enriched in the WCZ1 group. Additionally, the phylum Firmicutes, family Clostridiaceae, and genus Clostridium were enriched in the WDZ1 group (Fig. 7a). In group WAZ2, the genus [Eubacterium] from order Erysipelotrichi, class Erysipelotrichales, and family Erysipelotrichaceae was more abundant. The order Bacillales, family Peptostreptococcaceae, and genus [Clostridium] were enriched in group WBZ2. Notably, the phylum Firmicutes remained more abundant in the control group, with the family Clostridiaceae and genus Clostridium also being more abundant in the WDZ2 group (Fig. 7b).
LEfSe analysis identifying bacterial taxa differentially enriched among groups. (a, b) On both days 15 and 30, TCHMC supplementation promoted enrichment of beneficial taxa associated with SCFA production and immune modulation, while the control group showed higher levels of opportunistic or less beneficial bacteria.
The addition of traditional Chinese herbal medicine compounds in the diet of perinatal female yaks affected the weight, inflammation level, and antioxidant capacity of newborn yaks
This experiment indicated that newborn yaks in the experimental groups demonstrated obviously higher birth weight (p < 0.01) (Fig. 8a). The average birth weight was 10.6 ± 0.83 kg (mean ± SEM) for the control group, 13.25 ± 0.29 kg for the WA group, 15.36 ± 0.47 kg for the WB group, and 14.93 ± 1.07 kg for the WC group. This represents a significant increase of approximately 25% in the WA group, 44.91% in the WB group, and 40.85% in the WC group. T-test analysis showed that T-AOC (p < 0.05) and SOD (p < 0.01) were significantly lower in group WD, while MDA were significantly higher (p < 0.05) (Fig. 8b). The level of TNF-α (p < 0.01) and IL-6 (p < 0.05) in yaks in WD were significantly higher in the experimental groups (Fig. 8c).
Effects of TCHMC supplementation on calf birth weight and maternal serum biomarkers. (a) Calf birth weights were significantly higher in treatment groups, particularly in WB, compared with control (WD). (b) Serum antioxidant indices (T-AOC, SOD, GSH-Px) were elevated and MDA levels reduced in treated yaks, indicating enhanced antioxidant capacity. (c) Inflammatory markers showed lower TNF-α and IL-6 but higher IL-10 in TCHMC groups, reflecting improved anti-inflammatory status. Together, these results suggest that TCHMC supplementation improved maternal health and neonatal outcomes.
Discussion
Yaks (Bos grunniens) are an essential resource in the plateau region, serving as the backbone of the Tibetan economy. Enhancing yak breeding practices could significantly benefit both the yak industry and the broader economic development of Xizang. The climate on the Qinghai-Tibet Plateau is harsh, with average temperatures ranging from − 15 to -5 °C and substantial snowfall from October to May. During the cold season, grass becomes scarce and withers under snow cover. Currently, yak breeding primarily relies on traditional grazing methods and natural pastures, with limited supplementary feeding. This results in a shortage of forage resources, particularly during the cold season10. In addition, the natural reproductive cycle of yaks includes mating from June to October and calving from May to September of the following year11. Consequently, the entirety of the gestation period occurs during the cold season, especially in the later stages when fetal development accelerates. This period necessitates a considerable increase in maternal nutrient intake to accommodate the growing needs of the developing fetus. However, the scarcity of nutritional resources during this time leads to inadequate nutrient supply, resulting in decreased birth weights and diminished resistance in newborn yaks10. Therefore, enhancing the management of mother yaks during the perinatal period is crucial for the health of both mothers and their offspring. The application of traditional Chinese medicine (TCM) offers a novel approach to improving the health management of female yaks.
Tibetan herbal medicine provides a variety of beneficial effects, including anti-pathogen, regulatory, anti-stress, antioxidant, immune-enhancing, and nutritional properties, which are particularly relevant to the high-altitude stressors faced by yaks, such as chronic hypoxia, cold temperatures, and seasonal forage scarcity12,13,14. These advantages address the diverse needs of the livestock industry for feed additives. As a natural plant-based solution, TCM ingredients are relatively mild, with minimal side effects, and do not induce drug resistance or leave harmful residues in animal products, thereby ensuring the safety of livestock products. Additionally, TCM reduces reliance on chemical drugs, which in turn lowers environmental pollution in breeding systems. Due to these benefits, TCM has been increasingly integrated into the livestock industry4,5,6.
Perinatal female yaks undergo significant changes in nutrition, metabolism, and endocrine function, which can lead to immune dysfunction and a heightened incidence of metabolic and infectious diseases15. During this period, yaks often experience decreased feed intake due to limited food sources or the demands of pregnancy, while their nutritional requirements for fetal growth and delivery increase. This situation leads to a state of negative energy balance16,17. Furthermore, late-stage pregnancy prompts fat mobilization in preparation for lactation, resulting in lipid peroxidation, oxidative stress, and immune dysfunction18. In response to the physical condition of periparturient female yaks, we have selected Chinese Tibetan medicine with anti-inflammatory, anti-stress, blood-nourishing, and fetal-protection effects to suit the remedy to the case.
The TCM compounds used in this study include Lycium chinense Miller, Astragalus propinquus, Tibetan Codonopsis pilosula, Atractylodes macrocephala, and Angelica sinensis, among others, which are known to enhance the body’s non-specific immunity and improve disease resistance. Codonopsis pilosula and Astragalus propinquus play a role in alleviating the body’s stress response by regulating adrenal, thyroid, and gonadal functions. Scutellaria baicalensis Georg exhibits anti-heat stress effects, while Glycyrrhiza uralensis Fish provides essential nutrients for fetal development, including vitamins, minerals, and amino acids. Other herbs, such as Citrus reticulata Blanco and Perilla Stem, promote digestion and improve feed conversion rates, thereby assisting female yaks in maintaining optimal nutritional status during the perinatal period19,20,21,22,23,24. Furthermore, White Peony Root and Prepared Rehmannia glutinosa enhance antioxidant resistance, nourish the blood, and stabilize pregnancy25,26. These herbs contribute to the restoration of normal physiological functions following parturition27. Antimicrobial resistance (AMR) issues have sparked a growing interest in traditional Chinese medicine among scientists. An important herb in traditional Chinese medicine (TCM), Atractylodes macrocephala has long been valued for its many health advantages. The inclusion of TCHMFs in the diet of weaned yaks considerably (P < 0.05) enhanced microbial diversity and richness as well as microbial stability in the gut microbiota. In the experiment groups, the ratio of Firmicutes/Bacteroidetes increased (P < 0.05 or P < 0.01), whereas the relative abundance of Firmicutes and Actinobacteria grew and Bacteroidetes dropped. TCHMFs have been shown to improve livestock productivity in a number of trials28. For example, TCHMFs as feed additives have been demonstrated to affect intestinal morphology and growth parameters in weaned piglets29. Furthermore, TCHMFs’ antioxidant mode of action has improved perinatal cows’ reproductive performance30. Liang et al. stated that, compared to the Hay diet, the TCM diet resulted in lower nitrogen excretion in feces and higher nitrogen (N) uptake and digestibility. Essential oils, a significant bioactive component of the rhizome of Atractylodes and the root of Angelica, are known to have antibacterial qualities against a variety of pathogens, including viruses, fungi, and both Gram-positive and Gram-negative bacteria31. Essential oils are thought to be a practical way to increase the effectiveness of ruminant nutrient consumption and have been thoroughly investigated for their potential as modulators of rumen microbial fermentation32. In particular, Soltan et al. found that supplementing dairy cows with essential oils marginally increased the perceived digestibility of dietary protein33. According to a prior study conducted on cattle by Yang et al. giving essential oils could improve ruminal digestion of food protein by 11% when compared to the control34. In their discussion of the beneficial effects of essential oils on ruminal fermentation, Hart et al. claimed that the primary function of essential oils in the rumen was to control the breakdown of proteins and amino acids by acting selectively on specific rumen microbes35. Consequently, it was hypothesized that TCM elements, such as essential oils, would have a major impact on ruminal fermentation and improve sheep’s nitrogen digestibility. Astragalus root and Angelica root together increased protein turnover in nephrotic patients, according to a prior human investigation by Li et al. 36. The results of their study are consistent with what is currently being observed in sheep. Additionally, almost two-thirds of the amino acids that ruminants receive come from the microbial protein source in the lower gut31.
Analysis of gut microbiota diversity indicated that the administration of Tibetan Chinese medicine significantly increased both the richness and evenness of gut microbiota in periparturient female yaks. This suggests that the herbal additives played a vital role in maintaining gut microbiota stability and health. Beta diversity analysis also revealed significant differences in gut microbiota composition between the experimental and control groups, indicating that the herbal additives had a substantial impact on gut microbial composition. However, the effects on microbial function were less pronounced. The dominant bacterial phyla in yaks, Firmicutes and Bacteroidetes37, remained the most abundant in the gut following TCM supplementation. The increased abundance of Bacteroidetes in the experimental groups suggests a favorable effect of TCM on intestinal health.
The relative abundance of Firmicutes (which facilitate fiber digestion) and Bacteroidetes (which degrade non-fibrous carbohydrates) did not exhibit significant changes, indicating that TCM supplementation did not disrupt core microbial populations. However, the increase in the relative abundance of Bacteroidetes may suggest a positive effect on nutrient utilization and intestinal immunity. Additionally, the abundance of the RC4-4 genus, which is associated with obesity, increased in the experimental groups, indicating that TCM may enhance feed absorption and promote fetal weight gain38. This is supported by the relative increase in the birth weight of newborn calves in the experimental groups.
Short-chain fatty acids (SCFAs), produced by gut microbes, are essential for host health, including the regulation of immune homeostasis, improvement of gastrointestinal function, and alleviation of inflammation39. In this study, Tibetan herbal medicine supplementation was associated with an increased abundance of SCFA-producing bacteria such as Phascolarctobacterium, Butyricicoccus, Bulleidia, and Blautia. Although we did not directly measure the concentration of SCFAs, the enrichment of these specific bacteria suggests a potential mechanism for the observed improvement, as these bacteria are mature producers of beneficial SCFAs such as butyrate40,41,42. Under normal circumstances, the gut microbes may form a symbiotic connection with the host and encourage the creation of SCFAs and the fermentation of complex carbohydrates to strengthen the intestinal barrier. Butyrate and other SCFAs can cause the synthesis of IL-18, which is necessary for the synthesis of AMPs such as lipocalin, calprotectin, and defensins. When it comes to inhibiting the growth of certain harmful bacteria, like Staphylococcus, AMPs are crucial. The development of colonic inflammation is influenced by both the microbiota’s makeup and the amounts of AMPs43. These bacteria contribute to reducing inflammation and regulating metabolism, which may explain the improved health of yaks in the experimental groups. Despite some reductions in Clostridium and Anaerostipes, which are butyrate producers, other probiotics such as Akkermansia, Odoribacter, Dehalobacterium, and Sporosarcina exhibited increased abundance in the WA and WC groups, supporting the anti-inflammatory effects of TCM. These findings confirm that TCM supplementation improves both the health of female yaks and the development of their offspring. Serum biochemical indicators, including inflammatory cytokines (IL-6, IL-10, TNF-α) and antioxidant capacity (T-AOC), were also measured. The experimental groups showed decreased levels of inflammatory cytokines, indicating reduced inflammation. Additionally, the T-AOC was higher in the experimental groups, particularly in group WC, suggesting stronger antioxidant capacity and better resistance to oxidative stress. These findings confirm that TCM supplementation improves both the health of female yaks and the development of their offspring.
It has been demonstrated that TCHM formulations enhance the perinatal reproductive performance of cows, their antioxidant capacity30, the growth performance of weaned piglets, and the reduction of heat stress in beef cattle, which in turn increases the apparent nutrient digestibility44. Through the regulation of cell growth, the enhancement of anti-inflammatory and antioxidant responses, and the improvement of immunological function, TCM may help weaned yaks become more physically fit and lower their risk of diarrhea. For the purpose of further assessing the effectiveness of these formulae, this study offers initial insights into the pharmacological mechanism of TCM in avoiding diarrhea and enhancing physical fitness in weaned yaks.
Our findings reveal the impact of TCM on the gut microbiota of yaks during the late gestation period (60 to 30 days pre-calving). Further research is needed to investigate whether these prenatal changes persist into the postpartum period and how they contribute to the long-term health of both the dam and the calf. Although this study did not focus on gut eukaryotes or viruses, their interactions with bacteria may significantly influence the health and reproductive capacity of yaks. Future research should investigate these interactions more comprehensively. The main limitation of this study is the inability to directly measure the concentration of microbial metabolites such as short-chain fatty acids, thus making it impossible to establish a direct causal relationship. Future research will use metabolomics techniques such as GC-MS to quantitatively analyze the SCFA content in serum and feces and combine in vitro culture experiments to further validate the causal mechanism between traditional Chinese medicine regulation of microbial metabolic function and host anti-inflammatory/antioxidant phenotype. Additionally, the unique characteristics of perinatal yaks pose challenges in collecting large sample sizes. Therefore, future studies should aim for more extensive sample collection, including serum and rumen fluid, to further assess the effects of TCM on the health and recovery of both perinatal female yaks and their calves.
Conclusion
This study demonstrated the positive effects of Chinese Tibetan medicine (TCHMC Ⅱ) on the health and reproductive performance of perinatal female yaks. The use of herbal compounds significantly enhanced gut microbiota diversity, improved nutrient absorption, and strengthened immune function. These improvements contributed to higher calf birth weights and better maternal health during late pregnancy. Furthermore, the experimental groups showed reduced inflammatory markers, enhanced antioxidant capacity, and improved physical condition, suggesting that Chinese herbal additives may provide a safe and effective strategy to promote yaks` health under challenging environmental conditions. Future studies should focus on a larger sample size and explore the long-term benefits of these herbal treatments for both female yaks and calves.
Materials and methods
Yaks and experimental design
This experiment was conducted at the Gesangtang Yak Breeding Base (Linzhou County, Lhasa, Xizang) (29.89 N 91.26E). A total of 32 late-pregnancy female yaks (defined as the period from two months pre-calving to one month post-calving) were randomly selected into four groups (8 yaks per group). We selected 32 late-pregnancy female yaks (defined as the period from two months pre-calving to one month post-calving) in four to six years from 1000 female yaks; those selected animals were healthy and in the late-pregnancy phase, confirmed by a local veterinarian. Those female yaks were four to six years old with an initial body weight of around 400–430 kg, with a good body condition score (Table 1). The experimental intervention starts 60 days before the expected delivery date and lasts for one month. Samples were collected 15 days after the start of the intervention (approximately 45 days before the expected delivery date, denoted as WZ1) and at the end of the intervention (on the 30th day of the intervention, approximately 30 days before the expected delivery date, denoted as WZ2). Based on the nutritional requirements of the perinatal yaks and the efficacy of Chinese herbs, three traditional Chinese herbal medicine compounds (TCHMCⅠ, TCHMCⅡ, and TCHMCⅢ) were formulated. These compounds were derived primarily from medicinal plants native to Xizang and were designed to nourish blood, replenish qi, stabilize pregnancy, and enhance immune function. All herbal ingredients were sourced from licensed Chinese herbal medicine wholesalers, then dried, crushed, and mixed in specified proportions for dietary supplementation. The experimental groups were treated as follows: (1) Group WA: 5% dietary addition of TCHMCІ (21% Scutellaria baicalensis Georgi, 21% Tibetan Codonopsis pilosula, 21% Astragalus propinquus, 21% White Peony Root, 16% Glycyrrhiza uralensis Fisch.); (2) Group WB: 5% dietary addition of TCHMC Ⅱ (18% Lycium chinense Miller; 18% Angelica sinensis; 18% Tibetan Codonopsis pilosula; 18% Atractylodes macrocephala; 18% Scutellaria baicalensis Georgi; 10% Glycyrrhiza uralensis Fisch.); (3) Group WC: 5% dietary addition of TCHMC Ⅲ (10% Boehmeria nivea Root; 5% Ligusticum sinense ‘Chuanxiong’; 11% prepared Rehmannia glutinosa; 7% White Peony Root; 7% Astragalus propinquus; 9% Tibetan Codonopsis pilosula; 13% Stir fried Atractylodes macrocephala; 7% Poncirus trifoliata; 7% Citrus reticulata Blanco; 7% Scutellaria baicalensis Georgi; 7% Perilla Stem; 5% Argy Wormwood Leaf; 5% Glycyrrhiza uralensis Fisch.) (4) Group WD: the control group, fed ad libitum. The experimental period consisted of 7 days of adaptation and 30 days of measurements.
The experiment included a 7-day acclimation period followed by 30 days of feeding. Samples were collected on Days 15 (WZ1) and 30 (WZ2). However, no Day 0 samples were collected, which limits the ability to determine true pre-treatment baselines for gut microbiota and serum biochemical markers. Yaks that delivered prematurely were excluded from the analysis.
Sample collection and processing
All sample collections were conducted at 8:00 a.m. before the yaks were fed to avoid postprandial variation. To minimize the risk of miscarriage when handling, rectal sampling was not performed. Instead, approximately 10 g of freshly excreted central fecal matter was collected immediately after defecation using a sterile, disposable fecal collection tube. This method ensured minimal contamination from soil, hair, and other environmental sources. The fecal samples were immediately placed into insulated containers with ice packs and transported to the laboratory, where they were stored at -80 °C until further analysis.
At the end of the 30-day experimental period, the newborn calves from the study yaks were weighed. Fresh blood samples were collected from the jugular vein of each calf using sterile blood collection needles and vacuum-sealed tubes. After resting at room temperature (~ 20 °C) for 2 h, samples were centrifuged at 4000 rpm for 10 min to separate the serum, which was then stored at -80 °C for subsequent biomarker analyses. Serum samples were promptly analyzed for inflammation and oxidative stress markers. Inflammatory cytokines, including interleukin-6 (IL-6), interleukin-10 (IL-10), and tumor necrosis factor-alpha (TNF-α), were quantified using commercial enzyme-linked immunosorbent assay (ELISA) kits (Enzyme-linked Biotechnology, Shanghai, China). Oxidative stress parameters were assessed using diagnostic kits (Nanjing Jiancheng Bioengineering Research Institute, China) to measure levels of malondialdehyde (MDA), total antioxidant capacity (T-AOC), superoxide dismutase (SOD) activity, and glutathione peroxidase (GSH-Px) activity.
DNA extraction, PCR amplification, and sequencing library preparation
Rectal content samples were collected on days 15 and 30 of the trial, with six samples randomly selected from each group (WA, WB, WC, WD) for 16 S rRNA sequencing. Genomic DNA (gDNA) was extracted using the Stool Genomic DNA Extraction Kit (Solarbio®, Beijing, China), according to the manufacturer`s instructions. DNA concentration was measured with a Nanodrop 2000 spectrophotometer, and integrity was verified by 1.2% agarose gel electrophoresis.
The V3/V4 hypervariable regions of the bacterial 16 S rRNA gene were amplified using specific primers and Pfu high-fidelity DNA polymerase (Fullmetal Corporation)46. PCR products were purified with Vazyme VAHTS™ DNA Clean Beads and quantified with the Quant-iT PicoGreen dsDNA Assay Kit. Fluorescence measurements were performed on a BioTek FLx800 Microplate Reader. Based on the quantification results, samples were pooled in appropriate proportions to meet sequencing requirements.
Sequencing libraries were prepared using the TruSeq Nano DNA LT Library Prep Kit (Illumina, San Diego, USA). Library fragments were selected and purified via 2% agarose gel electrophoresis, and quality was assessed with the Agilent High Sensitivity DNA Kit. Libraries passing quality control were sequenced on the Illumina NovaSeq 6000 platform with the NovaSeq 6000 SP Reagent Kit (500 cycles), generating high-quality paired-end sequencing data.
Bioinformatics and statistical analysis
High-quality raw sequencing data were processed by demultiplexing libraries and samples according to index and barcode information, followed by barcode removal. Sequence denoising was performed using the QIIME2 dada2 pipeline, and the taxonomic composition was assigned at various taxonomic levels.
Alpha diversity was assessed based on the distribution of amplicon sequence variants (ASVs). For beta diversity, distance matrices were generated at the ASV level, and group differences were evaluated using ordination and clustering methods, combined with statistical testing.
To assess species composition, LEfSe analysis and t-tests were applied to identify differentially abundant taxa and potential microbial biomarkers. Functional prediction was performed using the 16 S rRNA gene data to infer microbial metabolic pathways, with differential pathway enrichment and taxonomic contributors analyzed. The experimental data were analyzed using GraphPad Prism (version 9.0) and SPSS (version 29.0). The t-test, ANOVA analysis, Kruskal-Wallis test, and Dunn’s test were employed for statistical analysis, with a P-value of less than 0.05 considered statistically significant. A one-way Analysis of Variance (ANOVA) was conducted for initial body weight, body condition scoring, and age to test for significant differences between the four groups (WA, WB, WC, WD) (Table 1).
Data availability
All sequence data in this study were deposited in the NCBI Sequence Read Archive database under accession number: PRJNA1204184.
References
Qiu, Q. et al. The Yak genome and adaptation to life at high altitude. Nat. Genet. 44, 946–949 (2012).
Wang, Y. et al. Effects of milk Replacer-Based Lactobacillus on growth and gut development of yaks’ calves: a gut Microbiome and metabolic study. Microbiol. Spectr. 10, e0115522 (2022).
Overton, T. R., McArt, J. A. A. & Nydam, D. V. A 100-Year review: metabolic health indicators and management of dairy cattle. J. Dairy. Sci. 100, 10398–10417 (2017).
Ran, M. et al. Traditional Chinese herbal medicine complex supplementation improves reproductive performance, serum biochemical parameters, and anti-oxidative capacity in periparturient dairy cows. Animal Biotechnol. 33, 647–656 (2022).
Wang, M. et al. Effects of dietary supplementation with herbal extract mixture on growth performance, organ weight and intestinal morphology in weaning piglets. J. Anim. Physiol. Anim. Nutr. (Berl). 104, 1462–1470 (2020).
Bozkurt, M. et al. Effects of dietary supplementation with a herbal extract on the performance of broilers infected with a mixture of Eimeria species. Br. Poult. Sci. 53, 325–332 (2012).
Huang, X. et al. Administration of an herbal powder based on traditional Chinese veterinary medicine enhanced the fertility of Holstein dairy cows affected with retained placenta. Theriogenology 121, 67–71 (2018).
Zhu, Z. et al. Effects of traditional Chinese medicine formula on ruminal fermentation, enzyme activities and nutrient digestibility of beef cattle. Anim. Sci. J. 89, 661–671 (2018).
Zhang, Z. et al. Convergent evolution of rumen microbiomes in High-Altitude mammals. Curr. Biol. 26, 1873–1879 (2016).
Long, R., Dong, S., Wei, X. H. & Pu, X. J. L. P. S. The effect of supplementary feeds on the bodyweight of Yaks in cold season. 93:197–204. (2005).
Gu, Y. et al. Dietary supplementation with tributyrin prevented weaned pigs from growth retardation and lethal infection via modulation of inflammatory cytokines production, ileal expression, and intestinal acetate fermentation. J. Anim. Sci. 95, 226–238 (2017).
Cui, H. R. et al. Immunometabolism at the service of traditional Chinese medicine. Pharmacol. Res. 176, 106081 (2022).
Li, Y. et al. Mechanisms of traditional Chinese medicine in modulating gut microbiota metabolites-mediated lipid metabolism. J. Ethnopharmacol. 278, 114207 (2021).
Zhang, S. Q., Jiang, X. X. & Li, J. C. Traditional Chinese medicine in human diseases treatment: new insights of their potential mechanisms. Anat. Rec (Hoboken). 306, 2920–2926 (2023).
Ribeiro, E. S. et al. Prevalence of periparturient diseases and effects on fertility of seasonally calving grazing dairy cows supplemented with concentrates. J. Dairy. Sci. 96, 5682–5697 (2013).
McArt, J. A. A., Nydam, D. V. & Oetzel, G. R. Epidemiology of subclinical ketosis in early lactation dairy cattle. J. Dairy. Sci. 95, 5056–5066 (2012).
Esposito, G., Irons, P. C., Webb, E. C. & Chapwanya, A. Interactions between negative energy balance, metabolic diseases, uterine health and immune response in transition dairy cows. Anim. Reprod. Sci. 144, 60–71 (2014).
Abuelo, A., Hernández, J., Benedito, J. L. & Castillo, C. Association of oxidative status and insulin sensitivity in periparturient dairy cattle: an observational study. J. Anim. Physiol. Anim. Nutr. (Berl). 100, 279–286 (2016).
Kim, H. J. et al. Immunostimulating activity of lycium Chinense miller root extract through enhancing cytokine and chemokine production and phagocytic capacity of macrophages. J. Food Biochem. 44, e13215 (2020).
Zhu, X. M. et al. Effects of dietary astragalus propinquus schischkin polysaccharides on growth performance, immunological parameters, antioxidants responses and inflammation-related gene expression in Channa Argus. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 249, 109121 (2021).
Zhang, Y. et al. Astragalus polysaccharides alleviate DSS-induced ulcerative colitis in mice by restoring SCFA production and regulating Th17/Treg cell homeostasis in a microbiota-dependent manner. Carbohydr. Polym. 349, 122829 (2025).
Zou, Y. F. et al. A Polysaccharide Isolated from Codonopsis pilosula with Immunomodulation Effects Both In Vitro and In Vivo. Molecules 24. (2019).
Song, Q., Peng, S. & Zhu, X. Baicalein protects against MPP(+)/MPTP-induced neurotoxicity by ameliorating oxidative stress in SH-SY5Y cells and mouse model of parkinson’s disease. Neurotoxicology 87, 188–194 (2021).
Du, K. et al. Angelica sinensis polysaccharide antagonizes 5-Fluorouracil-induced spleen injury and dysfunction by suppressing oxidative stress and apoptosis. Biomed. Pharmacother. 162, 114602 (2023).
Yan, B. F. et al. Aqueous extract of paeoniae radix Alba (Paeonia lactiflora Pall.) ameliorates DSS-induced colitis in mice by tunning the intestinal physical barrier, immune responses, and microbiota. J. Ethnopharmacol. 294, 115365 (2022).
Fangfang, Z., Hongyan, W. & Hui, Z. Analysis of Baishao and Shudihuang in regulating menstruation chapter of Fuqingzhu Nvke. China’s Naturopathy. 29, 11–12 (2021).
Jang, Y., Kim, T. K. & Shim, W. S. Naringin exhibits in vivo prokinetic activity via activation of Ghrelin receptor in Gastrointestinal motility dysfunction rats. Pharmacology 92, 191–197 (2013).
Zhu, Y. et al. Protective effects of traditional Chinese herbal medicine formulas (TCHMFs) via influencing Anti-Oxidative Capacity, inflammatory Mediators, and gut microbiota in weaned Yaks. Pakistan Veterinary Journal, 45(1). (2025).
Wang, M., Huang, H., Hu, Y., Liu, Y., Zeng, X., Zhuang, Y., He, S. (2020). Effects of dietary supplementation with herbal extract mixture on growth performance, organ weight and intestinal morphology in weaning piglets. Journal of animal physiology and animal nutrition, 104(5), 1462–1470.
Ran, M. et al. Traditional Chinese herbal medicine complex supplementation improves reproductive performance, serum biochemical parameters, and anti-oxidative capacity in periparturient dairy cows. Animal Biotechnol. 33 (4), 647–656 (2022).
Liang, X., Bi, X., Sano, H. & Jin, J. Supplementing forage with traditional Chinese medicine can increase microbial protein synthesis in sheep. Ruminants 4 (4), 613–622. https://doi.org/10.3390/ruminants4040042 (2024).
Benchaar, C. et al. A review of plant-derived essential oils in ruminant nutrition and production. Anim. Feed Sci. Technol. 145 (1–4), 209–228 (2008).
Soltan, M. A. E., Shewita, R. S. & Al-Sultan, S. I. Influence of essential oils supplementation on digestion, rumen fermentation, rumen microbial populations and productive performance of dairy cows. (2009).
Yang, W. Z. et al. Effects of Garlic and Juniper berry essential oils on ruminal fermentation and on the site and extent of digestion in lactating cows. J. Dairy Sci. 90 (12), 5671–5681 (2007).
Hart, K. J., Yáñez-Ruiz, D. R., Duval, S. M., McEwan, N. R. & Newbold, C. J. Plant extracts to manipulate rumen fermentation. Anim. Feed Sci. Technol. 147 (1–3), 8–35 (2008).
Li, L., Yu, H. & Pan, J. A study on protein metabolism in nephrotic patients treated with Chinese herbs. Zhonghua Nei Ke Za Zhi. 34 (10), 670–672 (1995).
Liu, J. et al. Comparative analysis of gut microbiota in healthy and diarrheic Yaks. Microb. Cell. Fact. 21, 111 (2022).
Ziętak, M. et al. Altered microbiota contributes to reduced Diet-Induced obesity upon cold exposure. Cell. Metab. 23, 1216–1223 (2016).
Koh, A., De Vadder, F., Kovatcheva-Datchary, P. & Bäckhed, F. From dietary fiber to host physiology: Short-Chain fatty acids as key bacterial metabolites. Cell 165, 1332–1345 (2016).
Zhou, X. et al. Coffee leaf tea extracts improve hyperuricemia nephropathy and its associated negative effect in gut microbiota and amino acid metabolism in rats. J. Agric. Food Chem. 71, 17775–17787 (2023).
Zhu, W. et al. Garlic skin induces shifts in the rumen Microbiome and metabolome of fattening lambs. Animal 15, 100216 (2021).
Zhang, D. et al. Sex-dependent changes in the microbiota profile, serum metabolism, and hormone levels of growing pigs after dietary supplementation with Lactobacillus. Appl. Microbiol. Biotechnol. 105, 4775–4789 (2021).
Yao, Y., Liu, Y., Xu, Q. & Mao, L. Short chain fatty acids: essential weapons of traditional medicine in treating inflammatory bowel disease. Molecules 29 (2), 379. https://doi.org/10.3390/molecules29020379 (2024).
Lin, Z. N., Ye, L., Li, Z. W., Huang, X. S., Lu, Z., Yang, Y. Q., Ying, Z. Y. Chinese herb feed additives improved the growth performance, meat quality, and nutrient digestibility parameters of pigs. AMEM 3 (1), 47–54 (2020).
Kanehisa, M., Furumichi, M., Sato, Y., Matsuura, Y. & Ishiguro-Watanabe, M. KEGG: biological systems database as a model of the real world. Nucleic Acids Res. 53 (D1), D672–D677 (2025).
Xu, C., He, Q., Zhu, Z.Y., Li, K. Propolis improves intestinal barrier function againstCryptosporidium parvum via NLRP6 inflammasome. mBio 16 (11), e0231725 (2025).
Funding
This research was funded by the Nagqu City Major Science and Technology Project (NQKJ-2023-10), the Department of Science and Technology of the Tibet Autonomous Region (XZ202101 ZD0002N), the breeding and efficient propagation of yaks in Gesangtang in Linzhou County (QYXTZX-LS2020-01), and the Ministry of Agriculture and Rural Affairs of China (CARS-37).
Author information
Authors and Affiliations
Contributions
YZ, WB, and KL developed the study concept and design. YZ, ZC, SL, MA, YC, and XZ conducted the experiment and collected the data. YC, ZC, SL, PP, MA, PS, ZB, and DZ analyzed the data and prepared the article. WB and KL provided critical revisions to the manuscript. All authors have reviewed and approved the final version of the manuscript for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study involving animals was approved by the ethics committee of Gesangtang Yak Breeding Base, Institute of Animal Husbandry and Veterinary Medicine, Tibet Academy of Agriculture and Animal Husbandry Science, and the ethics committee of Nanjing Agricultural University (NJAU.No20220305025). This study is performed in accordance with relevant guidelines and regulations. All methods are reported by the ARRIVE guidelines. For this experiment, yaks were obtained from Gesangtang Yak Breeding Base, a government-owned company, and cooperated with the Tibet Academy of Agriculture and Animal Husbandry Sciences. And before the experiment, we obtained informed consent orally from the manager of this company.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cidan, Y., Cisang, Z., Lu, S. et al. Tibetan herbal medicine improved the health status of calves by regulating the antioxidant ability, inflammatory reaction and microbiota of female Yaks. Sci Rep 16, 6685 (2026). https://doi.org/10.1038/s41598-026-37500-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-37500-3