Abstract
Atrial fibrillation (AF) risk is influenced by sex hormones and steatotic liver disease (SLD), but their interplay in women remains unclear. We aimed to evaluate the impact of various SLD statuses on AF risk among premenopausal and postmenopausal women. Total 2,181,691 women from the 2009 Korean National Health Insurance Service screenings were analyzed, excluding those with prior AF, hysterectomy, liver cancer, or transplantation. Participants were classified by menopausal status and SLD type: No SLD, metabolic dysfunction-associated steatotic liver disease (MASLD), MASLD with combined etiologies, MASLD and increased alcohol intake (MetALD), and alcohol-associated liver disease (ALD). Over 8.3 years of follow-up, postmenopausal women (n = 903,079) were older with higher body mass index and comorbidities, while premenopausal women (n = 1,278,613) more often smoked and consumed alcohol. In premenopausal women, AF incidence ranged from 0.65 per 1,000 person-years without SLD to 1.67 in ALD, with the highest hazard ratio in ALD (HR 2.16, 95% CI 1.27–3.67). Postmenopausal women showed higher absolute AF incidence (3.43–5.44) but attenuated hazard ratios, peaking at 1.66 in ALD. SLD is significantly associated with increased AF risk, peaking in premenopausal women with MASLD or ALD; these findings necessitate early cardiovascular risk assessment and AF screening, warranting further research into preventive strategies. These novel findings establish a significant association between SLD and increased AF risk across all menopausal stages, demonstrating that the highest relative risk is disproportionately concentrated in premenopausal women with MASLD or ALD, thus underscoring the urgent need for enhanced cardiovascular surveillance and status-specific primary prevention strategies in this overlooked younger population.
Similar content being viewed by others
Introduction
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia, affecting millions of individuals globally, and is associated with significant morbidity and mortality1. While traditional risk factors for AF, such as hypertension, obesity, and diabetes, are well-established, emerging evidence suggests that sex hormones and metabolic dysfunction may also play a crucial role in AF development2,3. Notably, the hormonal shifts associated with menopause have been connected to a heightened risk of cardiovascular diseases, including AF.4
Metabolic dysfunction-associated Steatotic liver disease (MASLD), previously known as non-alcoholic fatty liver disease (NAFLD), is increasingly recognized as a healthcare burden. 5,6,7 MASLD is risk factor of various diseases including gastrointestinal disease, cardiovascular disease, type 2 diabetes, and chronic kidney disease8,9,10. Given its close relationship to metabolic syndrome, MASLD shares common risk factors with AF, such as obesity and dyslipidemia. MASLD may independently elevate AF risk through mechanisms like systemic inflammation, oxidative stress, and alterations in cardiac electrophysiology11. Notably, women have distinct metabolic and hormonal profiles that may affect their cardiovascular risk profile differently than men12. Observed differences in the prevalence and clinical presentation of MASLD indicate that gender plays a modulating role in disease trajectory and severity, warranting further investigation into sex specific risk factors13,14. However, research on how MASLD and menopausal status impact AF risk remains sparse, highlighting a significant gap in understanding these interacting factors.
The aim of this study was to evaluate the differential impact of specific steatotic liver disease (SLD) subtypes on AF risk in women, specifically examining the association in premenopausal compared to postmenopausal women using a nationwide population-based cohort.
Methods
Data were sourced from the comprehensive nationwide claims database of the Korean National Health Insurance Service (NHIS), which covers nearly the entire population of South Korea. The NHIS database includes a broad spectrum of information, such as demographic details, mortality records, medical costs, diagnoses coded according to the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM), as well as data on inpatient and outpatient services and prescriptions15. The National Health Screening Program for chronic diseases, available to individuals aged 19 and older, provides additional data from physical examinations, laboratory tests, and self-reported questionnaires16.
This study complied with the Declaration of Helsinki’s ethical principles. The study protocol was reviewed and exempted from full review by the Institutional Review Board (IRB no. E-2411-036-1585) due to data anonymization. The requirement for informed consent was waived by the IRB as the NHIS data were de-identified. Authorization to use the NHIS database from 2009 to 2018 was granted in 2024.
Study populations
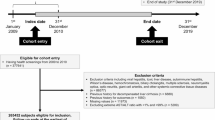
Figure 1 illustrates the patient selection process. The initial cohort comprised 3,106,111 women aged over 40 who underwent a National Health Insurance Corporation (NHIC) health examination and cancer survey in 2009. After excluding individuals with missing data (n = 509,184), a history of hysterectomy (n = 206,896), outliers for age at menarche or menopause (n = 186,899), liver cancer or transplantation (n = 1,508), prevalent AF (n = 15,500), and AF occurrence within one year of follow-up (n = 4,432), a total of 2,181,692 women remained. After further exclusion of those with specific etiology or cryptogenic SLD (n = 526), the final study population consisted of 2,181,166 participants, categorized into premenopausal (n = 902,825) and postmenopausal (n = 1,278,341) groups.
Study flow. Among women over age 40 years old who received NHIC health examination and cancer survey in 2009, a total of 2,181,166 subjects remained in the final study population after excluding the specified groups.
Definition of steatotic liver disease, fatty liver index, and cardiometabolic criteria
SLD is defined as hepatic steatosis identified by imaging or biopsy. It is further divided by the presence of cardiometabolic criteria and the cause of steatosis into five categories: MASLD, MASLD with increased alcohol intake (MetALD), alcohol-associated liver disease (ALD), specific etiology SLD, and cryptogenic SLD. 17 Due to the lack of imaging or biopsy data in the NHIS database, hepatic steatosis was instead assessed using the Fatty Liver Index (FLI).
FLI incorporates body mass index (BMI), waist circumference, triglycerides, and gamma-glutamyl transpeptidase (GGT)18. Cardiometabolic factors used to define MASLD were identified by meeting at least one of the following five criteria17: (1) BMI ≥ 25 kg/m² (or ≥ 23 kg/m² for Asian populations), waist circumference > 94 cm for men or > 80 cm for women, or ethnicity-adjusted values (> 90 cm for men and > 80 cm for women was used for Asian populations); (2) Fasting serum glucose ≥ 5.6 mmol/L (100 mg/dL), 2-hour post-load glucose levels ≥ 7.8 mmol/L (140 mg/dL), HbA1c ≥ 5.7% (39 mmol/mol), a diagnosis of type 2 diabetes, or treatment for type 2 diabetes (2-hour post-load glucose levels and HbA1c were not assessed in the health examination and thus excluded); (3) Blood pressure ≥ 130/85 mmHg or specific antihypertensive drug treatment; (4) Plasma triglycerides ≥ 1.70 mmol/L (150 mg/dL) or lipid-lowering treatment; and (5) Plasma HDL-cholesterol ≤ 1.0 mmol/L (40 mg/dL) for men or ≤ 1.3 mmol/L (50 mg/dL) for women, or lipid-lowering treatment.
The study population was classified into seven groups based on the FLI, cardiometabolic criteria, concomitant liver disease (CLD), and alcohol consumption: no SLD, MASLD, MASLD with other combined etiology, MetALD, ALD, specific etiology SLD, and cryptogenic SLD (Supplementary Table S1). Subjects with an FLI below 30 were categorized as having no SLD. Those with an FLI ≥ 30, cardiometabolic risk factors, alcohol consumption below 30 g/day for men or 20 g/day for women, and no evidence of CLD or ALD were classified as MASLD. The MASLD with other combined etiology group, included individuals meeting the same criteria as MASLD but with concurrent CLD or ALD. The MetALD group consisted of subjects with an FLI ≥ 30, cardiometabolic risk factors, and moderate alcohol consumption (30–60 g/day for men or 20–50 g/day for women). ALD was defined by an FLI ≥ 30 and high alcohol consumption (> 60 g/day for men or > 50 g/day for women). Specific etiology SLD included those with an FLI ≥ 30, no cardiometabolic risk factors but significant alcohol consumption (≥ 30 g/day for men or ≥ 20 g/day for women) or evidence of either CLD or ALD. Cryptogenic SLD comprised individuals with an FLI ≥ 30, no cardiometabolic risk factors, low alcohol consumption (< 30 g/day for men or < 20 g/day for women), and no evidence of CLD or ALD. Due to the small number of subjects in the specific etiology SLD and cryptogenic SLD groups, these individuals were excluded from the final analysis.
Covariates
The detailed definitions for exclusion criteria regarding AF, along with key comorbidities (diabetes mellitus, hypertension, dyslipidemia, chronic kidney disease [CKD], heart failure, prior myocardial infarction, prior ischemic stroke, and concomitant liver disease), and health exam questionnaire (smoking, alcohol consumption, and exercise, and household income) are provided in Supplementary Table S1. Reproductive history data included age of menarche and menopause, parity, breastfeeding practices, use of oral contraceptives, hormone replacement therapy, and history of benign breast tumors. Smoking habits (never, former, or current), alcohol consumption (none, mild to moderate, or heavy), and incomes were self-reported through a questionnaire19,20. General health assessments measured height, weight, BMI, waist circumference, and both systolic and diastolic blood pressure. Laboratory tests included fasting glucose levels, estimated glomerular filtration rate (eGFR), lipid profiles (total cholesterol, triglycerides, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol), and liver function tests (aspartate aminotransferase [AST], alanine aminotransferase [ALT], and gamma-glutamyl transferase [GGT])20,21,22,23. Each analysis utilized all available data without excluding entire cases.
Study outcomes and follow-up
The primary outcome during the follow-up period was the development of AF. AF was identified based on the initial recording of relevant ICD-10-CM codes (I48; AF and atrial flutter) on at least two outpatient visits or a hospital admission20. Patients were followed from the index date until the first occurrence of AF, loss of eligibility from the NHIS due to emigration, death, or the end of the study period (December 31, 2018), whichever came first.
Statistical analysis
Continuous variables are expressed as mean ± standard deviation for normally distributed variables and as geometric mean with interquartile range for non-normally distributed variables. Categorical variables are presented as frequencies and percentages. Baseline characteristics were compared using one-way analysis of variance for continuous variables and chi-square tests for categorical variables. The incidence rate (IR) of AF was calculated as the number of new AF cases per 1,000 person-years. Kaplan–Meier survival curves were used to assess cumulative AF incidence, with comparisons made via the log-rank test. Cox proportional hazards models were employed to estimate hazard ratios (HRs) and 95% confidence intervals (CIs), using five models with increasing covariate adjustments: (1) unadjusted (model 1); (2) adjusted for age (model 2); (3) adjusted for age and comorbidities (CKD, heart failure, prior MI, prior ischemic stroke) (model 3); (4) adjusted for age, socioeconomic factors (income, smoking, regular exercise), and reproductive history (parity, breastfeeding, oral contraceptive use, age at menarche, age at menopause, hormone replacement therapy) (model 4); and (5) further adjusted for all the mentioned covariates in the previous models (model 5). For sensitivity analyses, subjects with significant cardiovascular comorbidities were excluded. Subgroup analyses were conducted by menopausal status and comorbidities to assess potential interactions. Further analysis stratified by age evaluated the impact of age on AF risk stratified menopausal status.
Statistical significance was set at p < 0.05. All statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, North Carolina, USA).
Results
Baseline characteristics
The study included 2,181,166 women (mean age 54.8 ± 10.7 years, mean follow-up duration 8.3 ± 0.7 years). Of these, 41.4% were premenopausal, and 58.6% were postmenopausal. The baseline characteristics of the study population are described in Table 1. Postmenopausal women had later menarche (16.4 ± 1.8 vs. 15.1 ± 1.6 years). They had more children, longer breastfeeding, and more oral contraceptive use. Comorbidities were more frequent in postmenopausal women. While most women in the study never smoked, premenopausal women showed higher rates of current smoking (3.3% vs. 2.6%) and heavy drinking (2.1% vs. 0.8%). Regular exercise was similar in both groups (18.3% vs. 17.3%). In general, postmenopausal women had higher BMI, waist circumference, blood pressure, and worse lipid profiles overall. Within the same type of SLD, premenopausal subjects were generally heavier, with a higher proportion having a BMI over 30 kg/m² and larger waist circumferences. Detailed baseline characteristics by SLD group are described in Supplemental Table S2.
Steatotic liver disease and the risk of incident AF with different menopausal status
Over a mean follow-up duration of 8.3 ± 0.71 years, 47,061 (2.1%) developed new-onset AF. The Kaplan-Meier curves show that the cumulative incidence of AF is higher in women with SLD compared to those without SLD across both menopausal groups (Fig. 2), with a more marked increase in postmenopausal women. Log-rank test indicates a statistically significant difference between the groups (P < 0.001), suggesting a higher AF risk associated with liver disease in both menopausal statuses.
Cumulative Incidence of Atrial Fibrillation Based on Different Liver Disease Etiologies (a) Premenopausal Women (b) Postmenopausal Women. The Kaplan-Meier curves show that the cumulative incidence of AF is higher in women with SLD compared to those without SLD across both menopausal groups.
Table 2 presents the number of events, crude IRs, and HRs for AF, based on both unadjusted (model 1) and adjusted analyses (models 2–5). The postmenopausal group exhibited higher crude IRs than the premenopausal group (3.43 to 5.44 vs. 0.65 to 1.67 per 1,000 person-years). AF risk was significantly elevated across all SLD groups, irrespective of menopausal status, compared to individuals without SLD (all P-values < 0.001). Among liver disease categories, individuals with MetALD and ALD consistently demonstrated the highest adjusted HRs in both menopausal groups (MetALD: adjusted HR 2.07 [95% CI 1.60–2.66], ALD: adjusted HR 2.16 [95% CI 1.27–3.67] for premenopausal; MetALD: adjusted HR 1.57 [95% CI 1.34–1.83], ALD: adjusted HR 1.66 [95% CI 1.18–2.34] for postmenopausal).
Sensitivity analyses
In the sensitivity analyses, which excluded subjects with significant cardiovascular comorbidities (chronic kidney disease, heart failure, prior myocardial infarction, and prior ischemic stroke), AF risk remained elevated across all SLD groups compared to those without SLD (Supplemental Table S3).
In the premenopausal group, the adjusted HRs for AF were highest among individuals with MetALD (HR 2.03 [95% CI 1.56–2.63]) and ALD (HR 2.35 [95% CI 1.38–3.98]). Similarly, in the postmenopausal group, individuals with MetALD (HR 1.63 [95% CI 1.38–1.94]) and ALD (HR 1.72 [95% CI 1.19–2.48]) demonstrated the highest adjusted HRs for AF.
Subgroup analysis
The subgroup analysis on menopausal status and comorbidities revealed that AF risk is significantly influenced by menopausal status (Table 3). Other comorbidities, including chronic kidney disease, heart failure, prior myocardial infarction, and prior ischemic stroke, exhibited no significant interaction. Further subgroup analysis demonstrates the influence of age on AF risk, stratified by menopausal status (Supplementary Table S4). In both premenopausal and postmenopausal women, AF IRs increased progressively with age. The SLD group with the highest HRs varied across different age groups.
Discussion
Our study provides novel insights into the relationship between SLD, menopausal status, and AF risk in a large, nationwide cohort of Korean women. The key findings can be summarized as follows: (1) SLD is associated with an increased risk of AF in both premenopausal and postmenopausal women; (2) the impact of SLD on AF risk is more pronounced in premenopausal women, with higher hazard ratios observed in this group; (3) among the different categories of SLD, MetALD and ALD demonstrate the highest risk for AF in both menopausal groups; and (4) the association between SLD and AF risk persists even after adjusting for various confounding factors and in sensitivity analyses excluding individuals with significant cardiovascular comorbidities.
As far as we are aware this is the first analysis of the impact of various types of SLD on incident AF, in relation to menopausal status in women. This is important given that MASLD is one of the most common chronic liver diseases globally, with an estimated prevalence of 30–40% varying across populations24,25 Subjects with MASLD and related SLD have shown a higher risk of cardiovascular diseases—such as myocardial infarction, ischemic stroke, and heart failure—as well as associated mortality24,26. In the context of arrhythmias, biopsy-confirmed MASLD is linked to a significantly higher rate of incident AF compared to controls (adjusted HR 1.26, 95% CI 1.18–1.35). This effect is consistent regardless of the histological group27.
Generally, the prevalence of MASLD and its severity increases after menopause28,29,30,31. However, one recent study projected an increasing trend in MASLD prevalence among premenopausal women through 204028, highlighting that the importance of MASLD in this group should not be underestimated. Our results support this notion, as premenopausal women, despite a lower absolute incidence rate of AF, exhibit higher adjusted hazard ratios than postmenopausal women. These findings clearly emphasize the need for heightened awareness of SLD in premenopausal women.
While the mechanisms linking MASLD and AF remain incompletely characterized, emerging evidence implicates shared inflammatory and fibrotic pathways. Preliminary studies indicate that MASLD correlates with cardiac remodeling, such as increased left atrial stiffness and autonomic and conduction abnormalities, which may predispose individuals to AF.32–37 These observations may be partially explained by mitochondrial dysfunction, systemic inflammation, and oxidative stress observed in both metabolic conditions and AF.38,39 Specifically, MASLD pathogenesis involves elevated levels of proinflammatory cytokines (e.g., IL-6, CRP, and TNF), which are also implicated in AF.37,40 In parallel, heightened collagen turnover, as evidenced by increased collagen type I carboxy-terminal telopeptide, is linked to structural changes that further predispose to AF, suggesting a role for maladaptive cardiac remodeling41,42. These processes mirror findings in metabolic syndrome, where elevated galectin-3 and procollagen peptides independently predict AF risk43.
An important consideration in interpreting our findings is that SLD is defined by FLI which comprises BMI, waist circumference, triglycerides, and GGT. Since these factors are independently associated with AF risk,44–46 there is an inherent overlap between our definition of SLD and traditional cardiovascular risk factors. Therefore, SLD in our study should be interpreted as an integrative metabolic risk marker rather than a single isolated risk factor. SLD captures the cumulative burden of cardiometabolic dysfunction. Therefore, SLD in our study should be interpreted as an integrative metabolic risk marker that captures the cumulative burden of cardiometabolic dysfunction. While the observed associations may reflect shared upstream metabolic pathways rather than a direct effect of hepatic steatosis, the clinical relevance remains significant. Specifically, the clustering of these metabolic components manifesting as steatosis, may create a potent arrhythmogenic substrate, making SLD a valuable phenotype for identifying individuals at high AF risk.
Notably, our study observed the highest risks of AF in the MetALD and ALD groups, suggesting that alcohol consumption exerts an independent and potentially synergistic deleterious contribution when superimposed on metabolic dysfunction47. While MASLD drives AF primarily through structural remodeling linked to systemic inflammation, alcohol contributes via distinct electrophysiological mechanisms, including the shortening of the atrial effective refractory period, autonomic modulation, and direct toxic effects on cardiomyocytes48,49,50,51. Individuals with MetALD thus experience a ‘double hit’ phenomenon: the metabolic substrate provides a permissive milieu for inflammatory and fibrotic processes, while alcohol accelerates structural remodeling and triggers arrhythmogenesis. This synergistic toxicity may explain why MetALD group demonstrate substantially higher AF risk than MASLD alone, emphasizing alcohol’s independent contribution to atrial dysfunction.
The menopausal transition also marks a critical period in women, characterized by an increased prevalence of cardiovascular disease52. Early menopause, in particular, has been associated with elevated cardiovascular risk, including AF.53,54 The heightened cardiovascular risk among menopausal women can be partly attributed to menopause-related changes in cardiovascular risk profiles such as alterations in lipid metabolism, emergence of metabolic syndrome, and adverse vascular and body composition shifts55,56,57,58. These changes stem, in part, from declining sex hormones,59 which have also been linked to AF risk54. Indeed, one proposed mechanism involves the influence of sex hormones on immune function and gut microbiome composition, leading to exacerbated metabolic dysfunction60,61. Consistent with these observations, our study found that postmenopausal women exhibited a higher absolute IR of AF compared to premenopausal women. However, the impact of SLD is notably greater among premenopausal women, as reflected by a higher adjusted HR and a significant p-for-interaction value (< 0.0001). This pattern persisted even in individuals without major cardiovascular comorbidities, suggesting that SLD confers a more pronounced effect on AF risk in premenopausal women.
The greater impact of SLD in premenopausal women may be attributable to an inherent susceptibility to metabolic factors among these individuals. Although postmenopausal women generally exhibit worse metabolic profiles, premenopausal women who developed SLD tend to be heavier, with higher rates of obesity (BMI > 30 kg/m²) and larger waist circumferences. Mechanistically, the discrepancy between high relative risk and low absolute risk in premenopausal women offers a critical insight. Endogenous estrogen typically shields premenopausal women from visceral adiposity and liver fibrosis62,63. Consequently, the onset of SLD during this protective window implies the presence of particularly aggressive metabolic driver capable of overriding physiological defenses This may result in heightened systemic inflammation and epicardial fat accumulation, which are established triggers for atrial remodeling. Beyond metabolic profiles, genetic interactions may further define this susceptibility. Specific variants affecting lipid metabolism and estrogen signaling pathways could potentially amplify AF risk, particularly in premenopausal women39,63,64,65. In contrast, postmenopausal women face a high baseline risk of AF driven by age-related remodeling and estrogen withdrawal, which likely dilutes the relative statistical impact of SLD66. Future research should specifically investigate these gene-hormone interactions to refine risk stratification.
While premenopausal women exhibited higher relative hazard ratios, it is crucial to recognize that their absolute AF incidence remains substantially lower compared to postmenopausal women. Clinically, this distinction may warrant a stratified healthcare strategy. For premenopausal women, although the absolute risk is lower, the diagnosis of SLD should serve as a ‘red flag’ for metabolic susceptibility. Management in this group might optimally focus on aggressive risk factor modification. Future interventions could target weight management and strict alcohol cessation, particularly for those with MetALD, to reverse the metabolic substrate before atrial remodeling becomes before permanent structural changes occur. Regarding screening, given the lower absolute incidence, our data suggest that opportunistic screening may be more appropriate than universal electrocardiogram (ECG) screening for this younger demographic. In contrast, for postmenopausal women, the high absolute incidence of AF warrants systematic surveillance. For postmenopausal women, the high absolute incidence of AF may justify systematic surveillance as a hypothesis-generating strategy. Future research should evaluate the potential benefits of incorporating routine annual ECG screenings into standard care for postmenopausal women with SLD, particularly those with coexisting hypertension or diabetes, to determine if such approaches improve clinical outcomes before they are considered for routine implementation.
Study limitations
This study had several limitations. First, as an observational study using nationwide health insurance data, it can only establish associations between SLD and AF risk, not causal relationships. Second, the diagnosis of SLD relied on the FLI rather than imaging or biopsy, which may have introduced some misclassification. However, the FLI has demonstrated robust predictive capability across diverse populations, with area under receiver operating characteristic curves of 0.785, specifically in Korean cohorts67,68. Third, in certain groups, particularly the ALD category, the small number of events limited the reliability of some hazard ratio estimates. Fourth, while we adjusted for numerous confounding factors, the possibility of residual confounding from unmeasured variables remains. Fifth, the study population was limited to Korean women, which limits the generalizability of our findings to other ethnic groups. which may affect the generalizability of our findings to other ethnic groups or populations. Given that White ethnicity is associated with a higher risk of AF compared to Asians, and the impact of metabolic risk factors may vary across populations69,70,71,72,73, further studies in diverse cohorts are needed. Sixth, the disease severity of SLD could not be assessed. Sixth, menopausal status was assessed only at baseline and not updated during follow-up. Women who were premenopausal at baseline may have transitioned to postmenopausal status during the follow-up period, resulting in time-varying misclassification that may have attenuated the observed associations. However, such misclassification likely leads to a conservative bias, suggesting that the true associations between SLD and AF risk may be even more robust than those reported in this study. Lastly, we lacked detailed information on the severity and duration of liver disease, which could impact its association with AF risk. Despite these limitations, the large sample size and comprehensive nature of the national health insurance database provide valuable insights into the relationship between SLD, menopausal status, and AF risk.
Conclusion
This comprehensive study reveals statistically significant association between SLD and increased AF risk across all menopausal stages. The highest relative risk of AF was observed in premenopausal women with MASLD or ALD. These results emphasize the necessity of early cardiovascular risk assessments and targeted interventions including AF screening for women with SLD for this previously underrecognized subgroup. Further research is needed to uncover the underlying mechanisms and develop preventive strategies for this population.
Data availability
The data that support the findings of this study are available from the NHIS but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. However, data are available from the NHIS for researchers who meet the criteria for access to confidential data. To request access, researchers should visit the National Health Insurance Sharing Service website (https://nhiss.nhis.or.kr/) to submit a research proposal and proof of IRB approval. Subject to review and approval by the NHIS committee, and payment of a data usage fee, access to the requested datasets will be provided.
Abbreviations
- AF:
-
Atrial fibrillation
- ALD:
-
Alcoholic liver disease
- CI:
-
Confidence interval
- HR:
-
Hazard ratio
- MASLD:
-
Metabolic dysfunction-associated steatotic liver disease
- MetALD:
-
Metabolic-associated alcoholic liver disease
- SLD:
-
Steatotic liver disease
References
Linz, D. et al. Atrial fibrillation: epidemiology, screening and digital health. Lancet Reg. Health Eur. 37, 100786. https://doi.org/10.1016/j.lanepe.2023.100786 (2024).
Shantsila, E., Choi, E. K., Lane, D. A., Joung, B. & Lip, G. Y. H. Atrial fibrillation: comorbidities, lifestyle, and patient factors. Lancet Reg. Health Eur. 37, 100784. https://doi.org/10.1016/j.lanepe.2023.100784 (2024).
Elliott, A. D., Middeldorp, M. E., Van Gelder, I. C., Albert, C. M. & Sanders, P. Epidemiology and modifiable risk factors for atrial fibrillation. Nat. Rev. Cardiol. 20, 404–417. https://doi.org/10.1038/s41569-022-00820-8 (2023).
Lu, Z. et al. Association between sex-specific risk factors and risk of new-onset atrial fibrillation among women. JAMA Netw Open 5, e2229716 (2022). https://doi.org/10.1001/jamanetworkopen.2022.29716
Zhou, X. D. et al. Global burden of disease attributable to metabolic risk factors in adolescents and young adults aged 15–39, 1990–2021. Clin. Nutr. 43, 391–404. https://doi.org/10.1016/j.clnu.2024.11.016 (2024).
Zhou, X. D. et al. An international multidisciplinary consensus statement on MAFLD and the risk of CVD. Hepatol. Int. 17, 773–791. https://doi.org/10.1007/s12072-023-10543-8 (2023).
Nakano, M., Kawaguchi, M., Kawaguchi, T. & Yoshiji, H. Profiles associated with significant hepatic fibrosis consisting of Alanine aminotransferase > 30 U/L, exercise habits, and metabolic dysfunction-associated steatotic liver disease. Hepatol. Res. 54, 655–666. https://doi.org/10.1111/hepr.14020 (2024).
Cho, T. et al. Metabolic dysfunction-associated steatotic liver disease is a risk factor for gallstones: A multicenter cohort study. Hepatol. Res. 55, 663–674. https://doi.org/10.1111/hepr.14170 (2025). https://doi.org:
Tanaka, H. et al. MetALD was a more notable independent risk factor for reflux esophagitis than MASLD: A multicenter cohort study using directed acyclic graphs. Hepatol. Res. https://doi.org/10.1111/hepr.70028
Targher, G., Valenti, L. & Byrne, C. D. Metabolic Dysfunction-Associated steatotic liver disease. N Engl. J. Med. 393, 683–698. https://doi.org/10.1056/NEJMra2412865 (2025).
Stahl, E. P. et al. Nonalcoholic fatty liver disease and the heart: JACC State-of-the-Art review. J. Am. Coll. Cardiol. 73, 948–963. https://doi.org/10.1016/j.jacc.2018.11.050 (2019).
Vogel, B. et al. The lancet women and cardiovascular disease commission: reducing the global burden by 2030. Lancet 397, 2385–2438. https://doi.org/10.1016/s0140-6736(21)00684-x (2021).
Tsutsumi, T. et al. Low HDL cholesterol levels in women and hypertriglyceridemia in men: predictors of MASLD onset in individuals without steatosis. J. Gastroenterol. 60, 891–904. https://doi.org/10.1007/s00535-025-02242-y (2025).
Kalligeros, M. et al. Prevalence of steatotic liver disease (MASLD, MetALD, and ALD) in the united states: NHANES 2017–2020. Clin. Gastroenterol. Hepatol. 22, 1330–1332e1334. https://doi.org/10.1016/j.cgh.2023.11.003 (2024).
Cheol Seong, S. et al. Data resource profile: the National health information database of the National health insurance service in South Korea. Int. J. Epidemiol. 46, 799–800. https://doi.org/10.1093/ije/dyw253 (2017).
Lee, W. C. & Lee, S. Y. National health screening program of Korea. Jkma 53, 363–370. https://doi.org/10.5124/Jkma.2010.53.5.363 (2010).
Rinella, M. E. et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology 78, 1966–1986. https://doi.org/10.1097/hep.0000000000000520 (2023).
Bedogni, G. et al. The fatty liver index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 6, 33. https://doi.org/10.1186/1471-230x-6-33 (2006).
Lee, S. R. et al. Association between clustering of unhealthy lifestyle factors and risk of new-onset atrial fibrillation: a nationwide population-based study. Sci. Rep. 10, 19224. https://doi.org/10.1038/s41598-020-75822-y (2020).
Choi, E. K. Cardiovascular research using the Korean National health information database. Korean Circ. J. 50, 754–772. https://doi.org/10.4070/kcj.2020.0171 (2020).
Bansal, N. et al. eGFR and albuminuria in relation to risk of incident atrial fibrillation: A Meta-Analysis of the Jackson heart study, the Multi-Ethnic study of Atherosclerosis, and the cardiovascular health study. Clin. J. Am. Soc. Nephrol. 12, 1386–1398. https://doi.org/10.2215/cjn.01860217 (2017).
Lee, S. R., Choi, E. K., Han, K. D., Lee, S. H. & Oh, S. Effect of the variability of blood pressure, glucose level, total cholesterol level, and body mass index on the risk of atrial fibrillation in a healthy population. Heart Rhythm. 17, 12–19. https://doi.org/10.1016/j.hrthm.2019.07.006 (2020).
Alonso, A. et al. Circulating levels of liver enzymes and incidence of atrial fibrillation: the atherosclerosis risk in communities cohort. Heart 100, 1511–1516. https://doi.org/10.1136/heartjnl-2014-305756 (2014).
Lee, H. H. et al. Metabolic dysfunction-associated steatotic liver disease and risk of cardiovascular disease. Gut 73, 533–540. https://doi.org/10.1136/gutjnl-2023-331003 (2024).
Wong, V. W., Ekstedt, M., Wong, G. L. & Hagström, H. Changing epidemiology, global trends and implications for outcomes of NAFLD. J. Hepatol. 79, 842–852. https://doi.org/10.1016/j.jhep.2023.04.036 (2023).
Adams, L. A., Anstee, Q. M., Tilg, H. & Targher, G. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 66, 1138–1153. https://doi.org/10.1136/gutjnl-2017-313884 (2017).
Simon, T. G. et al. Incident cardiac arrhythmias associated with metabolic dysfunction-associated steatotic liver disease: a nationwide histology cohort study. Cardiovasc. Diabetol. 22, 343. https://doi.org/10.1186/s12933-023-02070-5 (2023).
Danpanichkul, P. et al. Metabolic syndrome and metabolic Dysfunction-Associated steatotic liver disease in premenopausal women: global trends and projections to 2040. Mayo Clin. Proc. 99, 1615–1628. https://doi.org/10.1016/j.mayocp.2023.12.025 (2024).
Jaroenlapnopparat, A. et al. Menopause is associated with increased prevalence of nonalcoholic fatty liver disease: a systematic review and meta-analysis. Menopause 30, 348–354. https://doi.org/10.1097/gme.0000000000002133 (2023).
Park, S. H. et al. Prevalence and risk factors of non-alcoholic fatty liver disease among Korean adults. J. Gastroenterol. Hepatol. 21, 138–143. https://doi.org/10.1111/j.1440-1746.2005.04086.x (2006).
Yang, J. D. et al. Gender and menopause impact severity of fibrosis among patients with nonalcoholic steatohepatitis. Hepatology 59, 1406–1414. https://doi.org/10.1002/hep.26761 (2014).
Simon, T. G., Bamira, D. G., Chung, R. T., Weiner, R. B. & Corey, K. E. Nonalcoholic steatohepatitis is associated with cardiac remodeling and dysfunction. Obes. (Silver Spring). 25, 1313–1316. https://doi.org/10.1002/oby.21879 (2017).
Houghton, D. et al. The degree of hepatic steatosis associates with impaired cardiac and autonomic function. J. Hepatol. 70, 1203–1213. https://doi.org/10.1016/j.jhep.2019.01.035 (2019).
VanWagner, L. B. et al. Association of nonalcoholic fatty liver disease with subclinical myocardial remodeling and dysfunction: A population-based study. Hepatology 62, 773–783. https://doi.org/10.1002/hep.27869 (2015).
Kim, N. H. et al. Non-alcoholic fatty liver disease, metabolic syndrome and subclinical cardiovascular changes in the general population. Heart 100, 938–943. https://doi.org/10.1136/heartjnl-2013-305099 (2014).
Ozveren, O. et al. Doppler tissue evaluation of atrial conduction properties in patients with Non-alcoholic Fatty-liver disease. Ultrason. Imaging. 38, 225–235. https://doi.org/10.1177/0161734615595015 (2016).
Mahfouz, R. A., Gouda, M., Galal, I. & Ghareb, M. S. Interatrial septal fat thickness and left atrial stiffness are mechanistic links between nonalcoholic fatty liver disease and incident atrial fibrillation. Echocardiography 36, 249–256. https://doi.org/10.1111/echo.14229 (2019).
Nishida, K. & Otsu, K. Inflammation and metabolic cardiomyopathy. Cardiovasc. Res. 113, 389–398. https://doi.org/10.1093/cvr/cvx012 (2017).
Zhong, B. et al. Identification of key genes increasing susceptibility to atrial fibrillation in nonalcoholic fatty liver disease and the potential mechanisms: mitochondrial dysfunction and systemic inflammation. Front. Pharmacol. 15, 1360974. https://doi.org/10.3389/fphar.2024.1360974 (2024).
Shitole, S. G. et al. Assessment of inflammatory biomarkers and incident atrial fibrillation in older adults. J. Am. Heart Assoc. 13, e035710. https://doi.org/10.1161/jaha.124.035710 (2024).
Odeh, A. et al. Interrelationship between inflammatory biomarkers and collagen remodeling proteins in atrial fibrillation. Clin. Appl. Thromb. Hemost. 29, 10760296231165055. https://doi.org/10.1177/10760296231165055 (2023).
Duprez, D. A. et al. Collagen biomarkers and incidence of new onset of atrial fibrillation in subjects with no overt cardiovascular disease at baseline: the Multi-Ethnic study of atherosclerosis. Circ. Arrhythm. Electrophysiol. 11, e006557. https://doi.org/10.1161/circep.118.006557 (2018).
Ionin, V. A. et al. Galectin-3, N-terminal propeptides of type I and III Procollagen in patients with atrial fibrillation and metabolic syndrome. Int. J. Mol. Sci. 21 https://doi.org/10.3390/ijms21165689 (2020).
Jeon, W. K. et al. Association between the accumulation of elevated serum γ-glutamyltransferase level and risk of atrial fibrillation: a nationwide cohort study. Sci. Rep. 13, 13771. https://doi.org/10.1038/s41598-023-40689-2 (2023).
Aune, D. et al. Body mass index, abdominal fatness, fat mass and the risk of atrial fibrillation: a systematic review and dose-response meta-analysis of prospective studies. Eur. J. Epidemiol. 32, 181–192. https://doi.org/10.1007/s10654-017-0232-4 (2017).
Alonso, A. et al. Blood lipids and the incidence of atrial fibrillation: the Multi-Ethnic study of atherosclerosis and the Framingham heart study. J. Am. Heart Assoc. 3, e001211. https://doi.org/10.1161/jaha.114.001211 (2014).
Moon, J. H., Jeong, S., Jang, H., Koo, B. K. & Kim, W. Metabolic dysfunction-associated steatotic liver disease increases the risk of incident cardiovascular disease: a nationwide cohort study. EClinicalMedicine 65, 102292. https://doi.org/10.1016/j.eclinm.2023.102292 (2023).
Voskoboinik, A., Prabhu, S., Ling, L. H., Kalman, J. M. & Kistler, P. M. Alcohol and atrial fibrillation: A sobering review. J. Am. Coll. Cardiol. 68, 2567–2576. https://doi.org/10.1016/j.jacc.2016.08.074 (2016).
Gould, L., Reddy, C. V., Becker, W., Oh, K. C. & Kim, S. G. Electrophysiologic properties of alcohol in man. J. Electrocardiol. 11, 219–226. https://doi.org/10.1016/s0022-0736(78)80120-4 (1978).
Mäki, T. et al. Effect of ethanol drinking, hangover, and exercise on adrenergic activity and heart rate variability in patients with a history of alcohol-induced atrial fibrillation. Am. J. Cardiol. 82, 317–322. https://doi.org/10.1016/s0002-9149(98)00299-9 (1998).
Aberle, N. S. 2, Burd, L., Zhao, B. H., Ren, J. & nd, & Acetaldehyde-induced cardiac contractile dysfunction May be alleviated by vitamin B1 but not by vitamins B6 or B12. Alcohol Alcohol. 39, 450–454. https://doi.org/10.1093/alcalc/agh085 (2004).
El Khoudary, S. R. et al. Menopause transition and cardiovascular disease risk: implications for timing of early prevention: a scientific statement from the American heart association. Circulation 142, e506-e532 (2020). https://doi.org/10.1161/cir.0000000000000912
Zhu, D. et al. Age at natural menopause and risk of incident cardiovascular disease: a pooled analysis of individual patient data. Lancet Public. Health. 4, e553–e564. https://doi.org/10.1016/s2468-2667(19)30155-0 (2019).
Yang, S. et al. Association of total reproductive years with incident atrial Fibrillation, and subsequent ischemic stroke in women with natural menopause. Circ. Arrhythm. Electrophysiol. 12, e007428. https://doi.org/10.1161/circep.119.007428 (2019).
Kamińska, M. S. et al. Menopause predisposes women to increased risk of cardiovascular disease. J. Clin. Med. 12 https://doi.org/10.3390/jcm12227058 (2023).
de Kat, A. C. et al. Unraveling the associations of age and menopause with cardiovascular risk factors in a large population-based study. BMC Med. 15, 2. https://doi.org/10.1186/s12916-016-0762-8 (2017).
Gurka, M. J., Vishnu, A., Santen, R. J. & DeBoer, M. D. Progression of metabolic syndrome severity during the menopausal transition. J. Am. Heart Assoc. 5 https://doi.org/10.1161/jaha.116.003609 (2016).
Franklin, R. M., Ploutz-Snyder, L. & Kanaley, J. A. Longitudinal changes in abdominal fat distribution with menopause. Metabolism 58, 311–315. https://doi.org/10.1016/j.metabol.2008.09.030 (2009).
El Khoudary, S. R. & Thurston, R. C. Cardiovascular implications of the menopause transition: endogenous sex hormones and vasomotor symptoms. Obstet. Gynecol. Clin. North. Am. 45, 641–661. https://doi.org/10.1016/j.ogc.2018.07.006 (2018).
Hoffmann, J. P., Liu, J. A., Seddu, K. & Klein, S. L. Sex hormone signaling and regulation of immune function. Immunity 56, 2472–2491. https://doi.org/10.1016/j.immuni.2023.10.008 (2023).
Cross, T. L. et al. Gut Microbiome responds to alteration in female sex hormone status and exacerbates metabolic dysfunction. Gut Microbes. 16, 2295429. https://doi.org/10.1080/19490976.2023.2295429 (2024).
Villa, A. et al. Tetradian Oscillation of Estrogen receptor α is necessary to prevent liver lipid deposition. Proc. Natl. Acad. Sci. U S A. 109, 11806–11811. https://doi.org/10.1073/pnas.1205797109 (2012).
Xiang, D., Liu, Y., Zhou, S., Zhou, E. & Wang, Y. Protective effects of estrogen on cardiovascular disease mediated by oxidative stress. Oxid. Med. Cell Longev. 5523516 (2021).https://doi.org/10.1155/2021/5523516
Cherubini, A. et al. Interaction between Estrogen receptor-α and PNPLA3 p.I148M variant drives fatty liver disease susceptibility in women. Nat. Med. 29, 2643–2655. https://doi.org/10.1038/s41591-023-02553-8 (2023).
Schwarz, K. G., Vicencio, S. C., Inestrosa, N. C. & Villaseca, P. Del Rio, R. Autonomic nervous system dysfunction throughout menopausal transition: A potential mechanism underpinning cardiovascular and cognitive alterations during female ageing. J. Physiol. 602, 263–280. https://doi.org/10.1113/jp285126 (2024).
Ying, W. et al. Associations between menopause, cardiac remodeling, and diastolic function: the CARDIA study. Menopause 28, 1166–1175. https://doi.org/10.1097/gme.0000000000001815 (2021).
Kim, J. H., Kwon, S. Y., Lee, S. W. & Lee, C. H. Validation of fatty liver index and lipid accumulation product for predicting fatty liver in Korean population. Liver Int. 31, 1600–1601. https://doi.org/10.1111/j.1478-3231.2011.02580.x (2011).
Koehler, E. M. et al. External validation of the fatty liver index for identifying nonalcoholic fatty liver disease in a population-based study. Clin. Gastroenterol. Hepatol. 11, 1201–1204. https://doi.org/10.1016/j.cgh.2012.12.031 (2013).
Kim, M. H. et al. Racial differences and similarities in atrial fibrillation epidemiology and risk factors in UK biobank and Korean NHIS-HEALS cohort studies. Heart Rhythm. https://doi.org/10.1016/j.hrthm.2025.02.013 (2025).
Kang, D. S. et al. Racial differences in ischemic and hemorrhagic stroke: an ecological epidemiological study. Thromb. Haemost. 124, 883–892. https://doi.org/10.1055/a-2278-8769 (2024).
Dewland, T. A., Olgin, J. E., Vittinghoff, E. & Marcus, G. M. Incident atrial fibrillation among Asians, Hispanics, blacks, and Whites. Circulation 128, 2470–2477. https://doi.org/10.1161/circulationaha.113.002449 (2013).
Ugowe, F. E., Jackson, L. R. 2, Thomas, K. L. & nd & Racial and ethnic differences in the prevalence, management, and outcomes in patients with atrial fibrillation: A systematic review. Heart Rhythm. 15, 1337–1345. https://doi.org/10.1016/j.hrthm.2018.05.019 (2018).
Rodriguez, F. et al. Racial and ethnic differences in atrial fibrillation risk factors and predictors in women: findings from the women’s health initiative. Am. Heart J. 176, 70–77. https://doi.org/10.1016/j.ahj.2016.03.004 (2016).
Acknowledgements
We thank the National Health Insurance Service for their approval of data use.
Funding
This research was supported by a grant from the Patient-Centered Clinical Research Coordinating Center funded by the Ministry of Health & Welfare, Republic of Korea (RS-2021-KH119931), and supported by the National Research Foundation of Korea grant funded by the Korea government (MSIT) (IRIS RS-2024-00340590). The funders had no role in study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
J.M. Choi attributed visualization, writing-original draft, and writing - review & editing. K-D. Han attributed conceptualization, data curation, formal analysis. Lee, S. Oh, G.Y. H. Lip attributed supervision and writing - review & editing. K.Y. Lee and H.J. Ahn attributed visualization and writing - review & editing. B. Kim attributed data curation, formal analysis, methodology. E-K Choi attributed conceptualization, funding acquisition and supervision. All authors contributed to the overall data interpretation, reviewed, and approved the final draft for submission.
Corresponding author
Ethics declarations
Competing interests
SRL: Speaking fees from Bayer, BMS/Pfizer, Biosense Webster, Daiichi-Sankyo, Sanofi-Aventis, Daewoong Pharmaceutical Co., Samjinpharm, Seers Technology, Biotronik, Boston Scientific, and Medtronic. Consultant for Biosense Webster. EKC: Research grants or speaking fees from Abbott, Bayer, BMS/Pfizer, Biosense Webster, Chong Kun Dang, Daewoong Pharmaceutical Co., Daiichi-Sankyo, DeepQure, Dreamtech Co., Ltd., Jeil. GYHL: Consultant and speaker for BMS/Pfizer, Boehringer Ingelheim, Anthos, and Daiichi-Sankyo. GYHL is a National Institute for Health and Care Research (NIHR) Senior Investigator and co-principal investigator of the AFFIRMO project on multimorbidity in AF, which has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No. 899871. No personal fees were received. The remaining authors have nothing to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Choi, J., Han, K., Lee, S. et al. The influence of menopausal phase on atrial fibrillation risk in steatotic liver disease patients: a nationwide cohort study. Sci Rep 16, 8205 (2026). https://doi.org/10.1038/s41598-026-38049-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-38049-x